Abstract
Fructose consumption in the USA has increased over the past three decades. During this time, obesity, insulin resistance and the metabolic syndrome have also increased in prevalence. While diets high in fructose have been shown to promote insulin resistance and increase TAG concentrations in animals, there are insufficient data available regarding the long-term metabolic effects of fructose consumption in humans. The objective of the present study was to investigate the metabolic effects of 10-week consumption of fructose-sweetened beverages in human subjects under energy-balanced conditions in a controlled research setting. Following a 4-week weight-maintaining complex carbohydrate diet, seven overweight or obese (BMI 26.8–33.3 kg/m2) postmenopausal women were fed an isoenergetic intervention diet, which included a fructose-sweetened beverage with each meal, for 10 weeks. The intervention diet provided 15% of energy from protein, 30% from fat and 55% from carbohydrate (30% complex carbohydrate, 25% fructose). Fasting and postprandial glucose, insulin, TAG and apoB concentrations were measured. Fructose consumption increased fasting glucose concentrations and decreased meal-associated glucose and insulin responses (P=0.0002, P=0.007 and P=0.013, respectively). Moreover, after 10 weeks of fructose consumption, 14 h postprandial TAG profiles were significantly increased, with the area under the curve at 10 weeks being 141% higher than at baseline (P=0.04). Fructose also increased fasting apoB concentrations by 19% (P=0.043 v. baseline). In summary, consumption of fructose-sweetened beverages increased postprandial TAG and fasting apoB concentrations, and the present results suggest that long-term consumption of diets high in fructose could lead to an increased risk of CVD.
Keywords: Fructose, Glucose, Insulin, Hypertriacylglycerolaemia, Apolipoprotein-B
Dietary fructose consumption in the USA has increased by about 30% during the past three decades(1) and has coincided with a dramatic rise in the prevalence of obesity and its metabolic consequences(2,3). There is substantial evidence from studies conducted in animals and some in human subjects to indicate that high-fructose diets can promote the development of the metabolic syndrome(1,3,4). Fructose is primarily metabolised by the liver, where it is a highly lipogenic substrate(5). In animals, fructose consumption enhances hepatic de novo lipogenesis(6) and increases the secretion of apoB-containing VLDL particles(7). Similarly, in humans, consumption of a high-fructose diet can increase both fasting(8) and postprandial(9) plasma TAG concentrations relative to diets containing starch or glucose. High-fructose diets can promote insulin resistance in animals by directly interfering with hepatic insulin signalling(10–12); and the elevated postprandial TAG in response to fructose may further impair insulin sensitivity(13). Consistent with these effects, overfeeding human subjects with a high-fructose diet for 6 d not only led to increased de novo lipogenesis and plasma TAG, but also reduced hepatic insulin sensitivity(14).
Unlike glucose, fructose does not stimulate insulin secretion, due to its hepatic metabolism and the low level of expression of the fructose transporter GLUT5 in pancreatic β-cells(15). The metabolic effects of long-term consumption of high-fructose diets have not been fully investigated in human subjects. In a short-term study in normal-weight women, we reported that consumption of fructose-sweetened beverages (providing 30% of total energy) with meals resulted in lowered postprandial glucose and insulin excursions, reduced 24 h circulating leptin concentrations relative to when the subjects consumed glucose-sweetened beverages, and produced sustained increases of postprandial plasma TAG(16). The present study was designed to investigate the effects of an isoenergetic 10-week intervention, in which 25% of daily energy requirements was provided as fructose-sweetened beverages, on fasting and postprandial glucose, insulin, TAG and plasma apoB concentrations.
Subjects and methods
Subjects
Seven overweight or obese (BMI 26.8–33.3 kg/m2) postmenopausal women (age 50–72 years) were recruited. All subjects provided informed consent to participate in the study, and reported being non-smokers, relatively sedentary and weight stable. Subjects were given a complete medical examination.
Experimental protocol
The experimental protocol was approved by the Human Subjects Review Committee at the University of California (Davis, CA, USA). Subjects resided in a residential facility during the entire 14-week period, where body weight, food intake and blood pressure were monitored. Body composition measurements and 14 h postprandial blood collections were performed at the Ragle Human Nutrition Research Facility (University of California, Davis, CA, USA). Exercise was limited to three supervised 20 min walks per week.
Subjects were studied at three different times; at the end of a 4-week baseline period (before dietary intervention) and at 2 and 10 weeks during the intervention. During the initial baseline period, subjects consumed a weight-maintaining diet. Energy requirements for weight maintenance were estimated using the Harris–Benedict equation(17) and with a physical activity factor of 1.5(18). Subjects were weighed daily, and energy intake was adjusted every fourth day to maintain body weight. Following this stabilisation period, subjects were fed 100% of their established energy requirements. Body composition was determined by dual-energy X-ray absorptiometry (Lunar DPX; Lunar Corp., Madison, WI, USA).
Diets
Diets provided 15% of energy from protein, 30% from fat and 55% from carbohydrate. During the baseline period, the carbohydrate was composed almost entirely of complex carbohydrate (i.e. starches from bread, rice and pasta), and the fructose content was < 3%. During intervention, complex carbohydrate was replaced with beverages sweetened with free fructose (at 25% of daily energy requirements), while the protein and fat content of the diet were unchanged. Beverages were prepared as 12% (w/w) solutions, flavoured with a popular unsweetened drink mix. The diets provided about 20 g dietary fibre per d.
The meals consisted of a 5 d rotating menu, served as three meals (breakfast at 09.00 hours, lunch at 13.00 hours, dinner at 18.00 hours) and a snack (at 21.00 hours). The subjects consumed all food provided. On each test day, a specific (sixth) menu was served to standardise test diet conditions. Prepared lunch and dinner items were obtained from Life Spring (Richmond, CA, USA).
Blood sampling
Blood samples were collected between 08.00 and 22.00 hours, at 30 min intervals around meal ingestion and at hourly intervals at other times. At each collection time, 4ml blood was obtained and placed into tubes containing EDTA. Samples were stored on ice, centrifuged, and stored at −80°C before assay. Full postprandial profiles were not obtained in all subjects due to inadequate patency of intravenous catheters in two subjects. Thus, the area-under-the-curve data for the 14 h profiles represent five subjects consuming fructose.
Assays
Fasting serum lipids (total cholesterol, LDL- and HDL-cholesterol and TAG) were determined with a Beckman Coulter Synchron LX (Fullerton, CA, USA). Fasting apoB was measured in the 08.00 hours sample, as described(19). Plasma glucose was measured with a YSI 2300 Glucose Analyser (Yellow Springs Instruments, Yellow Springs, OH, USA). Plasma insulin was measured by RIA using 125I human insulin (Amersham, Piscataway, NJ, USA) and human insulin standards from Linco Research (St Charles, MO, USA). Homeostatic model assessment of insulin resistance was calculated as described(20).
Data analysis
Fasting concentrations of glucose, insulin and TAG were determined as the average of the first three measurements (08.00 hours, 08.30 hours and 09.00 hours). Areas under the curve for glucose, insulin and TAG were calculated by the trapezoidal method using Microsoft Excel. Statistical analysis was performed with Prism 2.01 (GraphPad Software, San Diego, CA, USA). The significance of changes of parameters between baseline and 2 and 10 weeks of dietary intervention was determined by one-way repeated-measures ANOVA. The difference in apoB concentrations at 10 weeks was assessed by the paired t test. The two-sided level of significance was set at P<0.05.
Results
Body weight, body composition and blood pressure
There was a small (about 1.5%) decrease of body weight over the 10-week intervention period (Table 1). Body composition was not significantly changed, and blood pressure remained stable over the 10-week intervention.
Table 1.
Anthropometric, biochemical and postprandial measurements of subjects consuming fructose-sweetened beverages at 25% of daily energy requirements for 10 weeks† (Mean values with their standard errors)
| Baseline |
2 weeks |
10 weeks |
|||||
|---|---|---|---|---|---|---|---|
| Variables | Mean | SE | Mean | SE | Mean | SE | P |
| Anthropometric measurements | |||||||
| Body weight (kg) | 75.7 | 9.2 | 75.3 | 9.2 | 74.6** | 9.6 | 0.016 |
| BMI (kg/m2) | 29.1 | 2.2 | 29.0 | 2.1 | 28.7** | 2.4 | 0.011 |
| Body fat (%) | 49.2 | 2.7 | 49.4 | 3.0 | 49.1 | 2.9 | 0.64 |
| Fat mass (kg) | 37.2 | 5.0 | 37.2 | 5.3 | 36.7 | 5.3 | 0.097 |
| Lean mass (kg) | 38.5 | 5.0 | 38.1 | 4.8 | 37.9 | 5.2 | 0.30‡ |
| SBP (mmHg) | 130 | 14 | 128 | 7 | 133 | 14 | 0.52 |
| DBP (mmHg) | 70 | 12 | 69 | 8 | 69 | 12 | 0.67 |
| Biochemical measurements | |||||||
| Fasting glucose (mmol/l) | 4.6 | 0.4 | 4.9*** | 0.4 | 4.9*** | 0.4 | 0.0002 |
| Fasting insulin (pmol/l) | 58 | 18 | 68 | 26 | 62 | 27 | 0.32 |
| HOMA-IR | 1.71 | 0.61 | 2.13 | 0.80 | 1.96 | 0.91 | 0.15 |
| Total cholesterol (mmol/l) | 4.99 | 1.26 | 5.15 | 0.92 | 4.76 | 0.99 | 0.11 |
| HDL-cholesterol (mmol/l) | 1.07 | 0.29 | 1.13 | 0.23 | 1.04 | 0.27 | 0.51 |
| LDL-cholesterol (mmol/l) | 2.90 | 0.89 | 2.95 | 0.95 | 2.72 | 1.04 | 0.26 |
| TAG (mmol/l) | 1.69 | 1.30 | 1.93 | 1.98 | 1.93 | 2.49 | 0.49‡ |
| Postprandial measurements | |||||||
| Glucose AUC (mmol/l × 14 h) | 15.4 | 3.4 | 5.2** | 2.3 | 4.9** | 2.1 | 0.007 |
| Insulin AUC (pmol/l × 14 h) | 3229 | 611 | 2014* | 410 | 2132* | 306 | 0.013 |
| TAG AUC (mmol/l × 14 h) | 5.1 | 2.5 | 7.3 | 1.8 | 12.3* | 2.5 | 0.040 |
SBP, systolic blood pressure; DBP, diastolic blood pressure; HOMA-IR, homeostatic model assessment of insulin resistance; AUC, area under the curve.
Mean value was significantly different from that at baseline:
P<0.05,
P<0.01,
P<0.001.
For details of subjects and procedures, see Subjects and methods.
The Friedman test was used.
Fasting plasma concentrations of glucose, insulin and lipids
Consumption of the fructose-sweetened beverages significantly increased fasting glucose concentrations at 2 weeks and 10 weeks (P=0.0002 v. baseline) (Table 1). Fasting plasma insulin concentrations and homeostatic model assessment of insulin resistance were not significantly increased. There were no significant effects of fructose consumption on fasting total cholesterol, LDL, HDL or plasma TAG concentrations (Table 1).
Postprandial glucose, insulin and triacylglycerol responses and fasting apolipoprotein-B concentrations
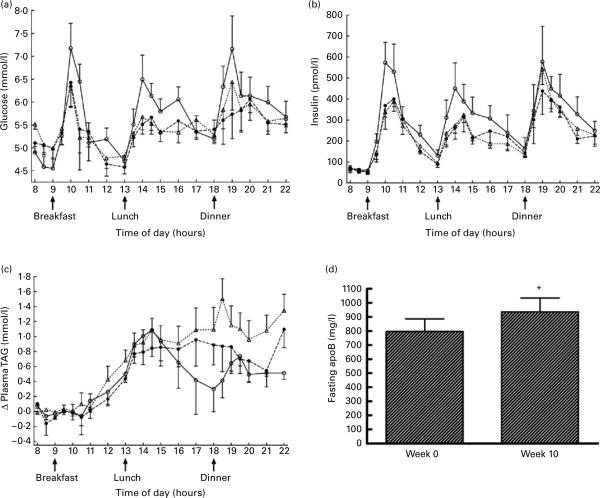
Consumption of fructose reduced meal-related plasma glucose excursions by 67% at 2 weeks and by 73% at 10 weeks (Fig. 1 (a)). The insulin area under the curve was reduced in subjects consuming fructose-sweetened beverages, by 42% at 2 weeks and 39% at 10 weeks, relative to baseline (Fig. 1 (b)).
Fig. 1.
Changes of plasma glucose (a), insulin (b) and TAG (c) over the three 14 h sampling periods during which subjects consumed either a high-complex carbohydrate diet (0 weeks; –◯–) or an isoenergetic diet with fructose-sweetened beverages at 25% of daily energy requirements (2 weeks (- -●- -) and 10 weeks (- -△- -)). Fasting apoB concentrations at 0 and 10 weeks of fructose beverage consumption are shown in (d). Values are means, with standard errors represented by vertical bars. * Mean value was significantly different from that at week 0 (P<0.05).
Relative to the baseline diet, consumption of fructose-sweetened beverages resulted in a progressive increase of postprandial TAG concentrations. The TAG area under the curve was increased by 44% after 2 weeks and by 141% at 10 weeks in the subjects consuming fructose (Fig. 1 (c)). After 10 weeks of fructose consumption, fasting apoB concentrations increased by 19.3 (SE 7.8) % (P=0.043; Fig. 1 (d)). In four subjects who underwent the same protocol, but consumed glucose-sweetened beverages for 10 weeks, no increases of postprandial TAG profiles or fasting apoB concentrations were observed (data not shown).
Discussion
In the present study, we investigated the effects of consuming fructose-sweetened beverages (25% of energy requirements) for 10 weeks on circulating glucose, insulin, TAG and apoB concentrations in overweight or obese postmenopausal women. Compared with a diet high in complex carbohydrates, consumption of fructose-sweetened beverages as part of an isoenergetic diet increased fasting plasma glucose concentrations, reduced meal-induced glucose and insulin excursions, and increased postprandial TAG concentrations and fasting plasma apoB concentrations.
A major strength of the present study is the high degree of control over the subjects' diets, with the subjects residing in a supervised setting and consuming standardised meals for the entire 14-week period. Another strength of the present study is that in addition to fasting concentrations, postprandial glucose, insulin and TAG responses were measured. Examination of only the fasting concentrations would not have revealed the marked effects of fructose consumption on postprandial TAG profiles. Subjects consuming the fructose-sweetened beverages for 10 weeks exhibited increased circulating TAG concentrations of > 1 mmol/l over fasting concentrations for the entire period from after lunch until 22.00 hours in the evening (Fig. 1 (c)). A number of studies support the existence of an independent relationship between non-fasting (postprandial) TAG concentrations and CVD(21,22). Moreover, because circulating apoB concentrations reflect the total number of potentially atherogenic lipoprotein particles, the increase of apoB concentrations observed here suggests that long-term consumption of high amounts of fructose could be atherogenic(23).
The main limitations of the present study are that the observations were restricted to overweight, postmenopausal women. Due to the high cost of the study, enrolment was limited to postmenopausal women, in order to avoid the possible confounding effects of age, sex and menstrual cycle on postprandial TAG metabolism(24,25).
The finding that consuming fructose-sweetened beverages promotes postprandial hypertriacylglycerolaemia is well supported by studies in animals fed high-fructose diets(1), and by several studies in human subjects, although there are some conflicting results in the literature(2). While the precise biochemical mechanisms underlying fructose-induced hepatic VLDL overproduction are unknown, studies in mice have shown that long-term fructose feeding induces the hepatic expression of a number of lipogenic genes, including stearoyl-CoA desaturase(26). Other rodent studies indicate that protein tyrosine phosphatase-1B, a negative regulator of insulin signalling, is required for fructose-associated increases in VLDL secretion(27). Impaired TAG uptake by adipose tissue, possibly due to reduced adipose tissue lipoprotein lipase activation from the lower insulin excursions, may also contribute to fructose-induced hypertriacylglycerolaemia in humans(28). In the present study, the relative contributions of increased TAG production v. reduced clearance were not examined; however, this issue is currently being addressed in our ongoing studies.
In human subjects, several short-term studies indicate that fructose consumption induces postprandial hypertriacylglycerolaemia(29–31). In our previous study, consumption of fructose-sweetened beverages (30% of energy) with mixed-nutrient meals in normal-weight women led to a rapid and prolonged increase of plasma TAG concentrations compared with when the same subjects consumed an equal amount of glucose(16). We have obtained similar results in overweight and obese men and women(32).
Bantle et al. fed men and women diets containing 17% of energy as either fructose or glucose for 6 weeks(9). Men on the fructose diet had significantly higher fasting plasma TAG concentrations and 24 h circulating TAG responses than the men consuming glucose, but neither of these effects were observed in women. The lack of an observable effect of fructose on postprandial TAG concentrations in women may be due to differences in age between the women in present study (mean age 64 (SE 3) years) and those studied by Bantle et al.(9) (overall mean about 40 years). Postprandial TAG responses are known to be greater in older subjects(24). Other factors, such as the presence of obesity and/or insulin resistance, the amount of fructose included in the diet, and the use of in-patient v. out-patient studies may also explain the inconsistencies that have been observed between studies. The effects of fructose consumption on postprandial TAG may also be exacerbated by pre-existing hypertriacylglycerolaemia and hyperinsulinaemia(33,34). Study duration may also be an important factor for the development of postprandial hypertriacylglycerolaemia, as it was much more pronounced after 10 weeks of fructose consumption than at 2 weeks.
Drinking fructose-sweetened beverages as part of a weight-maintaining diet also increased fasting plasma glucose concentrations, while meal-related excursions of plasma glucose and insulin were reduced. These findings are consistent with our short-term results(16) and some(35), but not all, studies of human subjects(2). Increases of fasting plasma glucose concentrations, within the normoglycaemic range, have been reported to independently predict the development of type 2 diabetes in men(36).
The amount of added fructose (25% of energy) investigated here is higher than the average US daily consumption, as the most recent estimate for the mean intake of added sugars by Americans (the 1994–6 Continuing Survey of Food Intakes by Individuals(37)) was 15.8 %. It is likely, however, that intake of sugar-sweetened beverages as well as total energy intake has increased in the 12 years since this survey, and is likely to be higher in certain groups, such as adolescents(38). Moreover, as sugar, fat and energy consumption are often under-reported in surveys(39), we propose that consuming 25% of total energy as sugars is not uncommon. However, since high-fructose corn syrup and sucrose are the predominant sweetners in the US food supply and are composed of approximately equal parts fructose and glucose, it is unlikely that fructose itself is often consumed at 25% of total energy. Interestingly, we have preliminary data which demonstrate that high-fructose corn syrup and sucrose both increase postprandial TAG comparably to pure fructose alone, and to support the relationship between fructose consumption and elevated postprandial TAG and apoB concentrations in a larger group of overweight and obese male and female subjects (n 18)(40).
In summary, consumption of fructose-sweetened beverages as part of a weight-maintaining diet for 10 weeks increased fasting glucose concentrations, postprandial TAG concentrations, as well as fasting plasma apoB concentrations in overweight and obese postmenopausal women. Together, these results suggest that obtaining a significant proportion of daily energy intake from fructose may promote or exacerbate altered lipid and carbohydrate metabolism associated with the metabolic syndrome, the prevalence of which has increased dramatically in parallel with fructose consumption in the USA. Furthermore, the increased fasting apoB and postprandial TAG concentrations observed in response to fructose consumption suggest that prolonged consumption of diets high in fructose could contribute to an increased risk for development of CVD.
Acknowledgements
The present study was supported by a National Research Initiative Competitive Grant from the United States Department of Agriculture (NRI#199900698) and a Co-operative Agreement with the Western Human Nutrition Research Center (58-5330-9-069). The University of California, Davis, Clinical and Translational Science Center's Clinical Research Center is supported by National Institutes of Health (NIH) grant RR-019975. Dr Havel's laboratory receives research support from NIH grants HL-075675, AT-002599, AT-002993, AT-003645, and the American Diabetes Association.
The authors also wish to acknowledge the efforts of Kim Oostema, Valerie Francisco, Sharon Martin and Kelli Kolstad, who coordinated the study.
Footnotes
There are no conflicts of interest to declare.
References
- 1.Elliott SS, Keim NL, Stern JS, Teff K, Havel PJ. Fructose, weight gain, and the insulin resistance syndrome. Am J Clin Nutr. 2002;76:911–922. doi: 10.1093/ajcn/76.5.911. [DOI] [PubMed] [Google Scholar]
- 2.Havel PJ. Dietary fructose: implications for dysregulation of energy homeostasis and lipid/carbohydrate metabolism. Nutr Rev. 2005;63:133–157. doi: 10.1301/nr.2005.may.133-157. [DOI] [PubMed] [Google Scholar]
- 3.Bray GA, Nielsen SJ, Popkin BM. Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am J Clin Nutr. 2004;79:537–543. doi: 10.1093/ajcn/79.4.537. [DOI] [PubMed] [Google Scholar]
- 4.Basciano H, Federico L, Adeli K. Fructose, insulin resistance, and metabolic dyslipidemia. Nutr Metab (Lond) 2005;2:5. doi: 10.1186/1743-7075-2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mayes PA. Intermediary metabolism of fructose. Am J Clin Nutr. 1993;58:754S–765S. doi: 10.1093/ajcn/58.5.754S. [DOI] [PubMed] [Google Scholar]
- 6.Park OJ, Cesar D, Faix D, Wu K, Shackleton CH, Hellerstein MK. Mechanisms of fructose-induced hypertriglyceridaemia in the rat. Activation of hepatic pyruvate dehydrogenase through inhibition of pyruvate dehydrogenase kinase. Biochem J. 1992;282:753–757. doi: 10.1042/bj2820753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Taghibiglou C, Carpentier A, Van Iderstine SC, Chen B, Rudy D, Aiton A, Lewis GF, Adeli K. Mechanisms of hepatic very low density lipoprotein overproduction in insulin resistance. Evidence for enhanced lipoprotein assembly, reduced intracellular apoB degradation, and increased microsomal triglyceride transfer protein in a fructose-fed hamster model. J Biol Chem. 2000;275:8416–8425. doi: 10.1074/jbc.275.12.8416. [DOI] [PubMed] [Google Scholar]
- 8.Reiser S, Powell AS, Scholfield DJ, Panda P, Ellwood KC, Canary JJ. Blood lipids, lipoproteins, apoproteins, and uric acid in men fed diets containing fructose or high-amylose cornstarch. Am J Clin Nutr. 1989;49:832–839. doi: 10.1093/ajcn/49.5.832. [DOI] [PubMed] [Google Scholar]
- 9.Bantle JP, Raatz SK, Thomas W, Georgopoulos A. Effects of dietary fructose on plasma lipids in healthy subjects. Am J Clin Nutr. 2000;72:1128–1134. doi: 10.1093/ajcn/72.5.1128. [DOI] [PubMed] [Google Scholar]
- 10.Taghibiglou C, Rashid-Kolvear F, Van Iderstine SC, Le-Tien H, Fantus IG, Lewis GF, Adeli K. Hepatic very low density lipoprotein-apoB overproduction is associated with attenuated hepatic insulin signaling and overexpression of protein-tyrosine phosphatase 1B in a fructose-fed hamster model of insulin resistance. J Biol Chem. 2002;277:793–803. doi: 10.1074/jbc.M106737200. [DOI] [PubMed] [Google Scholar]
- 11.Wei Y, Pagliassotti MJ. Hepatospecific effects of fructose on c-jun NH2-terminal kinase: implications for hepatic insulin resistance. Am J Physiol Endocrinol Metab. 2004;287:E926–E933. doi: 10.1152/ajpendo.00185.2004. [DOI] [PubMed] [Google Scholar]
- 12.Kelley GL, Allan G, Azhar S. High dietary fructose induces a hepatic stress response resulting in cholesterol and lipid dysregulation. Endocrinology. 2004;145:548–555. doi: 10.1210/en.2003-1167. [DOI] [PubMed] [Google Scholar]
- 13.Pedrini MT, Niederwanger A, Kranebitter M, Tautermann C, Ciardi C, Tatarczyk T, Patsch JR. Postprandial lipaemia induces an acute decrease of insulin sensitivity in healthy men independently of plasma NEFA levels. Diabetologia. 2006;49:1612–1618. doi: 10.1007/s00125-006-0262-z. [DOI] [PubMed] [Google Scholar]
- 14.Faeh D, Minehira K, Schwarz JM, Periasamy R, Park S, Tappy L. Effect of fructose overfeeding and fish oil administration on hepatic de novo lipogenesis and insulin sensitivity in healthy men. Diabetes. 2005;54:1907–1913. doi: 10.2337/diabetes.54.7.1907. [DOI] [PubMed] [Google Scholar]
- 15.Sato Y, Ito T, Udaka N, Kanisawa M, Noguchi Y, Cushman SW, Satoh S. Immunohistochemical localization of facilitated-diffusion glucose transporters in rat pancreatic islets. Tissue Cell. 1996;28:637–643. doi: 10.1016/s0040-8166(96)80067-x. [DOI] [PubMed] [Google Scholar]
- 16.Teff KL, Elliott SS, Tschöp M, Kieffer TJ, Rader D, Heiman M, Townsend RR, Keim NL, D'Alessio D, Havel PJ. Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J Clin Endocrinol Metab. 2004;89:2963–2972. doi: 10.1210/jc.2003-031855. [DOI] [PubMed] [Google Scholar]
- 17.Harris J, Benedict F. A Biometric Study of Basal Metabolism in Man. Carnegie Institute of Washington; Washington, DC: 1919. [Google Scholar]
- 18.Keim NL, Stern JS, Havel PJ. Relation between circulating leptin concentrations and appetite during a prolonged, moderate energy deficit in women. Am J Clin Nutr. 1998;68:794–801. doi: 10.1093/ajcn/68.4.794. [DOI] [PubMed] [Google Scholar]
- 19.Olin-Lewis K, Krauss RM, La Belle M, Blanche PJ, Barrett PH, Wight TN, Chait A. ApoC-III content of apoB-containing lipoproteins is associated with binding to the vascular proteoglycan biglycan. J Lipid Res. 2002;43:1969–1977. doi: 10.1194/jlr.m200322-jlr200. [DOI] [PubMed] [Google Scholar]
- 20.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and b-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 21.Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA. 2007;298:299–308. doi: 10.1001/jama.298.3.299. [DOI] [PubMed] [Google Scholar]
- 22.Bansal S, Buring JE, Rifai N, Mora S, Sacks FM, Ridker PM. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA. 2007;298:309–316. doi: 10.1001/jama.298.3.309. [DOI] [PubMed] [Google Scholar]
- 23.Marcovina S, Packard CJ. Measurement and meaning of apolipoprotein AI and apolipoprotein B plasma levels. J Intern Med. 2006;259:437–446. doi: 10.1111/j.1365-2796.2006.01648.x. [DOI] [PubMed] [Google Scholar]
- 24.Cohn JS, McNamara JR, Cohn SD, Ordovas JM, Schaefer EJ. Postprandial plasma lipoprotein changes in human subjects of different ages. J Lipid Res. 1988;29:469–479. [PubMed] [Google Scholar]
- 25.van Beek AP, de Ruijter-Heijstek FC, Erkelens DW, de Bruin TW. Menopause is associated with reduced protection from postprandial lipemia. Arterioscler Thromb Vasc Biol. 1999;19:2737–2741. doi: 10.1161/01.atv.19.11.2737. [DOI] [PubMed] [Google Scholar]
- 26.Miyazaki M, Dobrzyn A, Man WC, Chu K, Sampath H, Kim HJ, Ntambi JM. Stearoyl-CoA desaturase 1 gene expression is necessary for fructose-mediated induction of lipogenic gene expression by sterol regulatory element-binding protein-1c-dependent and -independent mechanisms. J Biol Chem. 2004;279:25164–25171. doi: 10.1074/jbc.M402781200. [DOI] [PubMed] [Google Scholar]
- 27.Qiu W, Avramoglu RK, Dubé N, Chong TM, Naples M, Au C, Sidiropoulos KG, Lewis GF, Cohn JS, Tremblay ML, Adeli K. Hepatic PTP-1B expression regulates the assembly and secretion of apolipoprotein B-containing lipoproteins: evidence from protein tyrosine phosphatase-1B overexpression, knockout, and RNAi studies. Diabetes. 2004;53:3057–3066. doi: 10.2337/diabetes.53.12.3057. [DOI] [PubMed] [Google Scholar]
- 28.Chong MF, Fielding BA, Frayn KN. Mechanisms for the acute effect of fructose on postprandial lipemia. Am J Clin Nutr. 2007;85:1511–1520. doi: 10.1093/ajcn/85.6.1511. [DOI] [PubMed] [Google Scholar]
- 29.Cohen JC, Schall R. Reassessing the effects of simple carbohydrates on the serum triglyceride responses to fat meals. Am J Clin Nutr. 1988;48:1031–1034. doi: 10.1093/ajcn/48.4.1031. [DOI] [PubMed] [Google Scholar]
- 30.Abraha A, Humphreys SM, Clark ML, Matthews DR, Frayn KN. Acute effect of fructose on postprandial lipaemia in diabetic and non-diabetic subjects. Br J Nutr. 1998;80:169–175. [PubMed] [Google Scholar]
- 31.Jeppesen J, Chen YI, Zhou MY, Schaaf P, Coulston A, Reaven GM. Postprandial triglyceride and retinyl ester responses to oral fat: effects of fructose. Am J Clin Nutr. 1995;61:787–791. doi: 10.1093/ajcn/61.4.787. [DOI] [PubMed] [Google Scholar]
- 32.Teff KL, Kleim NL, Townsend RR, Havel PJ. Fructose-sweetened beverages decrease circulating leptin levels and increase postprandial triglycerides in obese men and women. Diabetes. 2005;54:A385. [Google Scholar]
- 33.Crapo PA, Kolterman OG. The metabolic effects of 2-week fructose feeding in normal subjects. Am J Clin Nutr. 1984;39:525–534. doi: 10.1093/ajcn/39.4.525. [DOI] [PubMed] [Google Scholar]
- 34.Hallfrisch J, Reiser S, Prather ES. Blood lipid distribution of hyperinsulinemic men consuming three levels of fructose. Am J Clin Nutr. 1983;37:740–748. doi: 10.1093/ajcn/37.5.740. [DOI] [PubMed] [Google Scholar]
- 35.Le KA, Faeh D, Stettler R, Ith M, Kreis R, Vermathen P, Boesch C, Ravussin E, Tappy L. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am J Clin Nutr. 2006;84:1374–1379. doi: 10.1093/ajcn/84.6.1374. [DOI] [PubMed] [Google Scholar]
- 36.Tirosh A, Shai I, Tekes-Manova D, Israeli E, Pereg D, Shochat T, Kochba I, Rudich A. Normal fasting plasma glucose levels and type 2 diabetes in young men. N Engl J Med. 2005;353:1454–1462. doi: 10.1056/NEJMoa050080. [DOI] [PubMed] [Google Scholar]
- 37.Guthrie JF, Morton JF. Food sources of added sweeteners in the diets of Americans. J Am Diet Assoc. 2000;100:43–50. doi: 10.1016/S0002-8223(00)00018-3. [DOI] [PubMed] [Google Scholar]
- 38.West DS, Bursac Z, Quimby D, Prewitt TE, Spatz T, Nash C, Mays G, Eddings K. Self-reported sugar-sweetened beverage intake among college students. Obes (Silver Spring) 2006;14:1825–1831. doi: 10.1038/oby.2006.210. [DOI] [PubMed] [Google Scholar]
- 39.Lissner L, Heitmann BL, Bengtsson C. Population studies of diet and obesity. Br J Nutr. 2000;83(Suppl. 1):S21–S24. doi: 10.1017/s000711450000091x. [DOI] [PubMed] [Google Scholar]
- 40.Stanhope KL, Griffen SC, Bair B, Swarbrick MM, Keim NL, Havel PJ. Endocrine and metabolic effects of consuming high fructose corn syrup-, sucrose-, fructose-, and glucose-sweetened beverages with meals in normal weight and overweight humans. Am J Clin Nutr. 2008 doi: 10.1093/ajcn/87.5.1194. (In the Press) [DOI] [PMC free article] [PubMed] [Google Scholar]