To the Editors:
Quadriceps muscle dysfunction is well recognised in chronic obstructive pulmonary disease (COPD), and is associated with impaired exercise capacity and increased mortality [1]. Functionally, the quadriceps is characterised by reduced strength, increased fatigability and decreased endurance, associated with muscle fibre atrophy and a switch towards more glycolytic muscle fibres (types 1–2). The underlying molecular mechanisms of quadriceps dysfunction in COPD are not well clarified.
Myostatin is a member of the transforming growth factor-β family, and is a potent negative regulator of muscle mass, as demonstrated in naturally occurring animal and human genetic mutations, and genetic murine models [2]. Myostatin may therefore be a candidate regulator of muscle mass in disorders characterised by muscle atrophy, including COPD.
Little data exist regarding myostatin in COPD, but a recent cross-sectional study showed a three-fold increase in vastus lateralis myostatin mRNA transcripts in COPD patients with significant quadriceps weakness compared to healthy controls [3]. However, the relationship between quadriceps myostatin expression and functional characteristics of the muscle is not known. We hypothesised that myostatin expression would negatively correlate with quadriceps strength and exercise capacity in COPD.
18 patients with COPD were enrolled from clinics at the Royal Brompton Hospital (London, UK). Exclusion criteria included exacerbation in the previous 4 weeks, coexisting heart, renal or liver failure, or a systemic inflammatory or metabolic disorder. 16 healthy, age-matched controls were recruited by advertisement. All participants gave written informed consent and the protocol was approved by the local Research Ethics Committee.
The following were measured in study participants: lung function tests; fat-free mass index, calculated from bioelectrical impedance and normalised to height squared; quadriceps muscle strength, assessed by measuring supine isometric maximal voluntary contraction (MVC) of the leg ipsilateral to the dominant hand and corrected for body mass index (MVC/BMI); quadriceps muscle endurance, non-volitionally assessed by repetitive magnetic stimulation of the femoral nerve as recently described [4]; 6-min walk distance (6MWD), performed on a 30-metre level course according to American Thoracic Society (ATS) guidelines [5]; health status, assessed using the St. George's Respiratory Questionnaire; and daily physical activity, measured using a tri-axial accelerometer (Dynaport Activity Monitor) (McRoberts BV, The Hague, the Netherlands) for 12 h·day−1 for 2 week days during normal activities with calculation of average locomotion time (Loc). Quadriceps weakness was defined as a MVC/BMI <1.2 based on the prognostic significance of this cut-off [1], and quadriceps endurance was defined as the time taken for quadriceps force to fall to 80% of baseline (T80) [4].
Percutaneous needle biopsy of the vastus lateralis was performed under local anaesthesia using the Bergstrom technique. RNA was extracted, and first strand cDNA was generated. Real-time quantitative PCR analysis was carried out in duplicate on each cDNA sample for both the myostatin gene and for the reference housekeeping gene human RPLPO (large ribosomal protein), using a 10-μL reaction of SYBR® Green Quantitative Kit (Sigma Aldrich, Poole, UK) and the primer pair (2 pmol·μL−1). Myostatin mRNA levels were normalised to human RPLPO. Further details and primer sequences are in the on-line supplementary material.
Spearman's rank correlation was used to compare the relationship between myostatin mRNA levels and clinical variables. Comparison of myostatin mRNA levels between patients and controls, and between those with quadriceps weakness and preserved strength were performed using Mann–Whitney test. Statistical analyses were performed using GraphPad Prism 5 (GraphPad Software, San Diego, CA, USA). Significance was set at a two-tailed p-value of <0.05.
Baseline characteristics are summarised in table 1. Of the study participants, 10 of the 18 COPD patients, and three of the 16 controls had quadriceps weakness. Myostatin mRNA expression was significantly elevated in those with quadriceps weakness compared with those with preserved strength (median 0.0012 versus 0.0006 AU; p<0.005).
TABLE 1.
Clinical characteristics of subjects
| COPD | Controls | p-value | |
|---|---|---|---|
| Age yrs | 65±7 | 65±7 | ns |
| Males:females n | 12 : 6 | 9 : 7 | ns |
| BMI kg·m−2 | 24.5±4.7 | 25.7±4.8 | ns |
| Fat free mass index | 15.8±2.3 | 17.4±2.5 | ns |
| FEV1 L | 0.93±0.40 | 3.03±0.63 | <0.0001 |
| FEV1 % pred | 35±15 | 112±13 | <0.0001 |
| DL,CO % pred | 37.9±14.3 | 91.3±16.1 | <0.0001 |
| TLC % pred | 120±19 | 108±11 | 0.03 |
| RV % pred | 193±51 | 103±16 | <0.0001 |
| Quadriceps MVC kg | 29.8±8.6 | 37.7±9.8 | 0.02 |
| MVC/BMI | 1.2±0.3 | 1.5±0.5 | 0.04 |
| Quadriceps endurance# s | 91±47 | 127±72 | ns |
| 6MWD m | 362±145 | 630±86 | <0.0001 |
| SGRQ total score | 57±11 | 3±4 | <0.0001 |
| Locomotion time min | 48±45 | 106±53 | 0.004 |
Data are presented as mean± sd, unless otherwise stated. COPD: chronic obstructive pulmonary disease; BMI: body mass index; FEV1: forced expiratory volume in 1 s; %pred.: % predicted; DL,co: carbon monoxide diffusion capacity; TLC: total lung capacity; RV: residual volume; MVC: maximal voluntary contraction; 6MWD: 6-min walk distance; SGRQ: St George's Respiratory Questionnaire, ns: nonsignificant i.e. p>0.05.
defined as the time for force to fall to 80% of baseline.
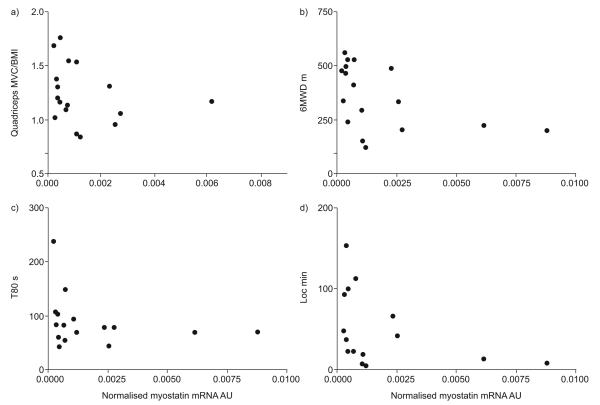
There was a significant inverse correlation between myostatin mRNA transcripts and quadriceps strength in COPD patients alone (r= −0.50, 95% CI −0.79— −0.02, p=0.04), and in COPD patients and healthy controls combined (r= −0.46, 95% CI −0.70– −0.13, p=0.006). Furthermore, in COPD patients, there was a significant inverse correlation between myostatin expression and T80 (r= −0.53, 95% CI −0.81– −0.05, p=0.03), 6MWD (r= −0.62, 95% CI −0.85– −0.21, p<0.01) and Loc (r= −0.54, 95% CI −0.82– −0.04, p=0.03) (fig. 1).
FIGURE 1.
Relationships between quadriceps myostatin mRNA expression and a) quadriceps maximum voluntary contraction normalised to body mass index (MVC/BMI) (r=0.50, p=0.04), b) 6-min walk distance (6MWD) (r= −0.62, p<0.01), c) quadriceps endurance (defined as the time for force to fall to 80% of the baseline (T80)) (r= −0.53, p=0.03) and d) daily physical activity (locomotion time (Loc)) (r= −0.54, p=0.03) in chronic obstructive pulmonary disease (COPD) patients.
This study quantified quadriceps muscle myostatin mRNA expression in well-characterised COPD patients and age-matched healthy controls. It presents the novel finding that quadriceps myostatin mRNA expression is inversely correlated with quadriceps strength and other clinically relevant physiological measures, including 6MWD, daily physical activity and quadriceps endurance in COPD patients. By directly seeking significant relationships between the expression of muscle myostatin and clinically relevant physiological outcome measures, our data complement existing quadriceps biopsy studies in COPD, where focus has either been solely on the molecular aspects, or where relationships between clinical physiological and molecular measurements have not been reported.
Several cattle breeds with an obvious increase in muscle mass have been shown to have natural mutations of the myostatin gene [6]. Knockout of the myostatin gene in mice results in a dramatic increase in muscle mass [2]. Putative factors that contribute to skeletal muscle dysfunction in COPD include immobility and corticosteroids. In normal mice, immobilisation leading to muscle atrophy induces a significant reversible rise in muscle myostatin expression [7], whilst myostatin null mice, compared with wild-type, appear to be protected from glucocorticoid-induced muscle atrophy [8].
Although myostatin appears to be a biologically plausible candidate regulator of muscle mass, there are few studies in COPD. Plant et al. [3] found significantly increased expression of myostatin transcripts in the quadriceps muscle of very weak COPD patients, compared with controls. Lewis et al. [9] found a “modest trend to reduction” in myostatin mRNA transcripts following pulmonary rehabilitation, with and without testosterone, or testosterone alone, in COPD patients compared with placebo. This did not reach statistical significance, but the relatively small sample size raises the possibility of a Type II error. More recently, Troosters et al. [10] showed that quadriceps myostatin mRNA was significantly lower in COPD patients who had completed 7 days of resistance training during a hospital admission, compared with usual care.
We speculate that myostatin is a driver of muscle wasting in selected patients. This concept is supported by examination of figure 1. In those with preserved quadriceps strength, 6MWD or daily locomotion time, upregulation of myostatin mRNA was not seen. However, increased myostatin expression was seen in some but not all patients with low quadriceps strength, exercise capacity or daily physical activity. Further work is required to elucidate the upstream and downstream signalling pathways for myostatin in COPD, including quantification of myostatin protein levels.
Although our cross-sectional study shows an association, rather than causation, previous studies have at least suggested that this is biologically plausible. This raises the intriguing possibility of myostatin as a therapeutic target. At least one myostatin antibody has been found safe in humans, though it proved only partially effective in increasing muscle size in muscular dystrophy patients [11].
In summary, our study has demonstrated a negative association between quadriceps muscle myostatin expression and strength, suggesting a putative biological role for myostatin in the skeletal muscle phenotype of COPD patients. Further work is required to elucidate the role of myostatin in COPD skeletal muscle dysfunction, including longitudinal studies and the closer interrogation of related signalling pathways.
Supplementary Material
Acknowledgements
W. D-C. Man is a National Institute for Health Research (NIHR) Clinician Scientist. S. A. Natanek is a Wellcome Trust Clinical Research Fellow. This project was undertaken, in part, at the NIHR Biomedical Research Unit in Advanced Lung Disease at the Royal Brompton and Harefield NHS Foundation Trust and Imperial College London; Professor Polkey's salary is part funded by the Biomedical Research Unit. The views expressed in this publication are those of the authors and not necessarily those of the NHS, The National Institute for Health Research nor the Department of Health.
Support Statement: W. D-C. Man is a National Institute for Health Research (NIHR) Clinician Scientist. S. A. Natanek is a Wellcome Trust Clinical Research Fellow. This project was undertaken, in part, at the NIHR Biomedical Research Unit in Advanced Lung Disease at the Royal Brompton and Harefield NHS Foundation Trust and Imperial College London; M.I. Polkey's salary is part funded by the Biomedical Research Unit. The views expressed in this publication are those of the authors and not necessarily those of the NHS, The National Institute for Health Research nor the Department of Health.
Footnotes
This article has supplementary material accessible from www.erj.ersjournals.com.
Statement of Interest: Statement of interest for P.R. Kemp and M.I. Polkey can be found at www.erj.esrjournals.com/misc/statements.dtl.
REFERENCES
- 1.Swallow EB, Reyes D, Hopkinson NS, et al. Quadriceps strength predicts mortality in patients with moderate to severe chronic obstructive pulmonary disease. Thorax. 2007;62:115–120. doi: 10.1136/thx.2006.062026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-β superfamily member. Nature. 1997;387:83–90. doi: 10.1038/387083a0. [DOI] [PubMed] [Google Scholar]
- 3.Plant PJ, Brooks D, Faughnan M, et al. Cellular markers of muscle atrophy in chronic obstructive pulmonary disease (COPD) Am J Respir Cell Mol Biol. 2009;42:461–471. doi: 10.1165/rcmb.2008-0382OC. [DOI] [PubMed] [Google Scholar]
- 4.Swallow EB, Gosker HR, Ward KA, et al. A novel technique for nonvolitional assessment of quadriceps muscle endurance in humans. J Appl Physiol. 2007;103:739–746. doi: 10.1152/japplphysiol.00025.2007. [DOI] [PubMed] [Google Scholar]
- 5.ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166:111–117. doi: 10.1164/ajrccm.166.1.at1102. [DOI] [PubMed] [Google Scholar]
- 6.McPherron AC, Lee SJ. Double muscling in cattle due to mutations in the myostatin gene. Proc Natl Acad Sci U S A. 1997;94:12457–12461. doi: 10.1073/pnas.94.23.12457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carlson CJ, Booth FW, Gordon SE. Skeletal muscle myostatin mRNA expression is fiber-type specific and increases during hindlimb unloading. Am J Physiol. 1999;277:R601–606. doi: 10.1152/ajpregu.1999.277.2.r601. [DOI] [PubMed] [Google Scholar]
- 8.Gilson H, Schakman O, Combaret L, et al. Myostatin gene deletion prevents glucocorticoid-induced muscle atrophy. Endocrinology. 2007;148:452–460. doi: 10.1210/en.2006-0539. [DOI] [PubMed] [Google Scholar]
- 9.Lewis MI, Fournier M, Storer TW, et al. Skeletal muscle adaptations to testosterone and resistance training in men with COPD. J Appl Physiol. 2007;103:1299–1310. doi: 10.1152/japplphysiol.00150.2007. [DOI] [PubMed] [Google Scholar]
- 10.Troosters T, Probst VS, Crul T, et al. Resistance Training Prevents Deterioration in Quadriceps Muscle Function During Acute Exacerbations of COPD. Am J Respir Crit Care Med. 2010;181:1016–1017. doi: 10.1164/rccm.200908-1203OC. [DOI] [PubMed] [Google Scholar]
- 11.Wagner KR, Fleckenstein JL, Amato AA, et al. A phase I/II trial of MYO-029 in adult subjects with muscular dystrophy. Ann Neurol. 2008;63:561–571. doi: 10.1002/ana.21338. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.