Abstract
According to hygiene hypothesis, a lower exposure to infection is associated with increased prevalence of allergic diseases. This study aimed to investigate the association between atopy and Toxoplasma gondii (Tg) infection by analyzing the antibody and cytokine responses to house dust mite allergens and T. gondii antigens in Brazilian subjects. A total of 275 individuals were assessed and divided into atopics (n=129) and non-atopics (n=146) based on markers of allergy (positive skin prick test and ELISA-IgE to mite allergens) or Tg-seropositive (n=116) and Tg-seronegative (n=159) groups according to infection markers (positive ELISA-IgG to T. gondii). Tg-seropositive individuals presented lower allergenic sensitization (37%) to mite allergens than Tg-seronegative subjects (54%). A significant association was found between atopy and negative serology to T. gondii (OR: 2.0; 95% CI: 1.23–3.26; P<0.05). Proliferative responses and cytokine production after antigenic stimulation showed predominant synthesis of Th1-cytokines as IFN-γ in Tg-seropositive patients, whether atopics or non-atopics. Conversely, Th2-cytokines as IL-5 prevailed in atopics compared to non-atopics, regardless the seropositivity to T. gondii. Levels of IL-10, IL-13, IL-17, and TGF-β were not able to discriminate the groups. Hence, a negative association between atopy and infection by T. gondii was demonstrated for the first time in Brazilian subjects, focusing on the antibody and cytokine responses and indicating that the immunomodulation induced by the parasite may play a protective role in the development of allergic diseases.
Keywords: Toxoplasma gondii, Allergic diseases, House dust mites, Hygiene hypothesis, IgE antibodies, Cytokines
Introduction
The increasing prevalence of allergic diseases such as asthma, allergic rhinitis and atopic dermatitis, observed in Western countries over the last four decades has been associated with a complex interaction of genetic and environmental factors related to Westernized lifestyles and urbanization [1]. Among the lifestyle factors, the improved hygiene is implicated, resulting in the hygiene hypothesis [2]. Populations of urban areas have higher rates of sensitization to house dust mites and fungi than those from rural areas, reinforcing the hypothesis of the influence of urban pollution in the increase of respiratory allergy [3].
Another factor that has given special attention focuses on the indiscriminate use of antibiotics in early life. The colonization of the gastrointestinal tract by commensal bacteria is prerequisite for the normal development of local and systemic immune responses. The decrease of intestinal bacterial flora is associated with altered immune response and Th2-type anti-inflammatory responses [4,5]. Thus, there is increasing evidence, although non-conclusive, in favor of the hygiene hypothesis, linking high socioeconomic status and improved hygiene with a lesser exposure to infections and microbial components, and consequently with an increased risk for the development of allergic diseases [6].
Infections of gastrointestinal tract seem to play a protective role in the development of allergies and Toxoplasma gondii (Tg) has been considered as a marker of “poor” hygiene and associated with a lower prevalence of allergic diseases due to its peculiar features: (i) it is an obligate intracellular protozoan distributed worldwide; (ii) the parasite infection induces a strong T cell-mediated immune response characterized by highly Th1-type polarized responses in early stages that remain during chronic infection, and (iii) the immune response is predominant and usually directs the kind of response in co-infections with other parasites [7].
Based on the fact that Dermatophagoides pteronyssinus and Dermatophagoides farinae are the most prevalent house dust mites worldwide and considered as major sources of inhaled allergens for inducing airway allergen sensitization in genetically predisposed individuals and that T. gondii infection has been considered a marker for “poor” hygiene, we aimed to investigate the association between atopy and T. gondii infection by analyzing the antibody and cytokine responses to house dust mite allergens and T. gondii antigens in Brazilian subjects.
Methods
Subjects
A total of 275 individuals were assessed and divided into atopics (n = 129) and non-atopics (n = 146) based on markers of allergy. Atopic patients, male and female, age from 18 to 60 years, were selected for the study at the Laboratory of Allergy and Clinical Immunology, Federal University of Uberlandia, Uberlandia, MG, Brazil. As inclusion criteria patients should have (i) perennial allergic rhinitis with or without intermittent or persistent, mild-to-moderate asthma, (ii) history of respiratory symptoms related to house dust exposure, (iii) positive skin prick test (SPT) to at least one of mite allergens (D. pteronyssinus and D. farinae), and (iv) presence of specific serum IgE to mite allergens determined by ELISA. Exclusion criteria were the presence of upper airway infections in the last 30 days prior to the study, the use of antihistaminic drugs in the week before SPT, and the use of oral or topic corticosteroids in the previous 4 weeks. Non-atopic healthy subjects, male and female, between 18 and 60 years old, were selected based on the absence of allergy history or clinical symptoms and negative SPT to a panel of standardized aeroallergens. The study was approved by the Ethics Committee in Human Research of the Federal University of Uberlandia and written informed consent was obtained from all participants.
Skin prick test and serum samples
All subjects underwent SPT as described elsewhere [8], with the following allergen extracts: mite (D. pteronyssinus, D. farinae and B. tropicalis) extracts prepared as previously described [9], and standardized commercial extracts of cockroach (Blattella germanica and Periplaneta americana), mold (Alternaria alternata) and pet danders (Felis domesticus and Canis familiaris) (IPI/ASAC, São Paulo, Brazil). A wheal mean 3-mm diameter larger than the negative control (diluent) was considered to be positive. In parallel, blood samples (5 ml) were collected from all individuals and the serum was stored at −20 °C until serological assays.
Mite extracts
D. pteronyssinus (Dp), D. farinae (Df) and B. tropicalis (Bt) crude extracts were obtained from mite bodies and feces as described elsewhere [9]. Briefly, mite powder was triturated in liquid nitrogen and allergens were extracted in 5 mM borate-buffered saline containing protease inhibitors (10 μg/ml aprotinin; 50 μg/ml leupeptin; 1.6 mM phenyl-methylsulfonyl fluoride–PMSF; Sigma Chemical Co., St Louis, USA). After centrifugation (20,000g for 45 min at 4 °C), the supernatant was dialyzed, filtered through a 0.22-μm membrane and protein concentration was determined [10]. Allergen extract aliquots were stored at −20 °C until being used in serological and proliferation assays.
Parasites and antigens
T. gondii RH strain tachyzoites were maintained in Swiss mice by intraperitoneal serial passage at 48-h intervals [11]. Mouse peritoneal exudates were harvested in sterile RPMI 1640 medium and washed twice (720g, 10 min, 4 °C) in medium. Parasite suspensions were adjusted to 1 × 108 tachyzoites/ml, treated with protease inhibitors as above mentioned, and then lysed by six freeze–thaw cycles followed by ultrasound (six 60 Hz cycles for 1 min each) on ice [12]. After centrifugation (10,000g, 30 min, 4 °C), supernatants were collected, filtered through a 0.22-μm membrane and the protein concentration was determined [10]. Soluble tachyzoite antigen (STAg) aliquots were stored at −20 °C until being used in serological and proliferation assays.
Measurement of specific IgE to mite allergens
Levels of specific IgE to mite allergens were measured by ELISA as described [9]. Briefly, high-affinity microtiter plates were coated with Dp or Df extracts (1 μg/well), blocked with phosphate-buffered saline (PBS, pH 7.2) containing 0.05% Tween 20 and 1% bovine serum albumin (PBS-T-BSA), and incubated with serum samples diluted 1:2 in PBS-T-BSA. After washing, plates were incubated with biotinylated anti-human IgE (Kierkegaard and Perry Lab., Gaithersburg, USA) and subsequently with streptavidin–peroxidase (Sigma). The assay was developed with 10 mM 2,2′-azinobis-(3-ethylbenzthiazoline) sulfonic acid (ABTS; Sigma) and 0.03% H2O2. Absorbance was determined in a plate reader at 405 nm up to a maximum absorbance value of 1.5 for which the assay was linear in positive control sera. Antibody titers were reported as ELISA index (EI) and determined as follows: EI = Absorbance of test sample/cutoff, where cutoff was calculated as the mean absorbance of negative control sera plus three standard deviations, as described elsewhere [9]. EI values >1.2 were considered to be positive in order to exclude borderline reactivity values close to EI = 1.0.
Measurement of total serum IgE
Total serum IgE was measured with a monoclonal antibody-based ELISA [13]. Briefly, plates were coated with monoclonal anti-human IgE (1:5000; Sigma), blocked with PBS-T-BSA and subsequently incubated with serum samples (1:5, 1:50 and 1:500) and biotinylated goat anti-human IgE (1:4000; Kierkegaard and Perry Lab). Subsequent steps were similar to specific IgE-ELISA. Results were reported as international units per milliliter (IU/ml) and calculated on the basis of a standard curve constructed with a reference serum that contained 500 IU/ml of total IgE.
Measurement of IgG anti-T. gondii
An indirect ELISA was carried out to detect IgG anti-T. gondii as previously described [11], with modifications. Briefly, low-affinity microtiter plates were coated with STAg (10 μg/ml), washed with PBS plus 0.05% Tween 20 (PBS-T) and incubated with serum samples diluted 1:64 in PBS-T plus 5% skim milk (PBS-T-M) for 1 h at 37 °C. After washing, goat anti-human IgG labeled with horseradish peroxidase (1:2000; Sigma) was added and incubated for 1 h at 37 °C. The reaction was revealed with 0.03% H2O2 in 1 mg/ml of ortho-phenylenediamine (OPD; Merck, Germany) in citrate-phosphate buffer (pH 5.0). The reaction was stopped with 2 N H2SO4 and absorbance was determined in a plate reader at 492 nm. Antibody titers were reported as ELISA index (EI) as described for specific IgE-ELISA and samples were considered positive when EI>1.2.
Proliferation assays
To evaluate the proliferative response to mite allergens and T. gondii antigen, 40 patients were selected based on the allergy and infection markers, and equally divided into four groups: atopic and Tg-seropositive, atopic and Tg-seronegative, non-atopic and Tg-seropositive, and non-atopic and Tg-seronegative. Peripheral blood mononuclear cells (PBMC) were freshly isolated from 20 ml heparinized blood samples by density gradient centrifugation over Ficoll–Hypaque solution (d = 1.077; Amersham Pharmacia Biotech AB, Uppsala, Sweden). PBMC were cultured in quadruplicate (2×105 cells/200 μl per well) in 96-well plates in RPMI 1640 medium supplemented with 5% heat-inactivated calf fetal serum (Cultilab, Campinas, Brazil) and stimulated with mitogen (phytohemagglutinin—PHA; 5 μg/ml), mite allergen extracts (Dp or Df; 10 μg/ml) or T. gondii antigen (STAg; 10 μg/ml). Control cultures were incubated with medium alone. Cells were incubated at 37 °C and 5% CO2 for 5 days and pulsed with 0.5 μCi/well of tritiated thymidine (New England Nuclear, Buckinghamshire, UK) for the final 8 h of culture. Cells were then harvested on glass fiber filters using cell harvester (Cambridge Technology Inc., Cambridge, USA) and counted in a liquid scintillation β-counter (Packard, Downers Grove, USA) and results were reported as counts per minute (cpm).
Cytokine production assays
PBMC (1×106 cells/1000 μl per well) were cultured in 24-well plates and stimulated with mitogen (PHA), mite allergen extracts (Dp or Df), T. gondii antigen (STAg), or medium alone, at the same conditions used for proliferation assays. Supernatants were collected after 3 days of culture, and stored in aliquots at −70 °C until determination of cytokine levels. IFN-γ, IL-5, IL-10, IL-13, IL-17 and TGF-β levels were measured in supernatants from cell cultures with an ELISA sandwich according to manufacturer’s instructions (R&D Systems, Minneapolis, USA) in comparison with a standard curve built with the respective recombinant human cytokines. The sensitivity of the assays was 15 pg/ml (IL-5) or 31 pg/ml (IFN-γ, IL-10, IL-17, and TGF-β) or 94 pg/ml (IL-13). Data below the sensitivity of each assay were calculated by extrapolation of the corresponding control curve on a cubic regression plot. The intra- and inter-assay coefficients of variations for these cytokines were below 10% and 15%, respectively.
Statistical analysis
Statistical analysis was performed using the GraphPad Prism 4.0 software (GraphPad Software, Inc., San Diego, CA). Differences between groups were analyzed using the Kruskal–Wallis or Mann–Whitney tests, when appropriate, and the Dunn multiple comparison test was applied to examine all possible pairwise comparisons. Differences in the percentages of positive samples for IgE antibodies to mite allergens and IgG to T. gondii between groups were analyzed by the χ2 test. Association between atopy and serology for T. gondii was analyzed by odds ratio (OR) with 95% confidence interval (CI). Differences were considered to be statistically significant when P<0.05.
Results
Subject data
The 275 analyzed subjects (129 atopics and 146 non-atopics) were distributed according to infection serological markers into two groups: TG+ (116 Tg-seropositive) and TG− (159 Tg-seronegative) (Table 1). The percentage of Tg-seropositive subjects (42%) was significantly lower as compared to Tg-seronegative ones (58%) (P=0.0002), and Tg-seropositive patients had age (median: 29 years) significantly higher than Tg-seronegative ones (median: 22 years) (P<0.0001). There was no significant difference regarding the sex between two groups of patients. SPT results to Dp and Df allergens revealed positivity rates significantly higher in TG− (54%) as compared to TG+ (37%) groups (P=0.0052). Although levels of total serum IgE in TG+ (median: 39 IU/ml) were lower than TG− (median: 78 IU/ml) groups, these differences were not significant.
Table 1.
Demographic and immunological characteristics of study individuals distributed according to serology to Toxoplasma gondii
| Characteristics | Groups |
||
|---|---|---|---|
| TG+ | TG− | P value | |
| Number of individuals (%) | 116 (42%) | 159 (58%) | 0.0002* |
| Age (years) Median (25–75%) |
29 (23-42) | 22 (20–28) | <0.0001* |
| Sex (male/female) | 42/74 | 62/97 | 0.6379 |
| SPT positive to Dp and Df (n, %)a |
43 (37%) | 86 (54%) | 0.0052* |
| Total serum IgE (IU/ml) Median (25-75%) |
39 (12-136) | 78 (14–234) | 0.2204 |
TG+: seropositive patients to Toxoplasma gondii;
TG?: seronegative patients to Toxoplasma gondii;
Values statistically significant (P<0.05).
Positivity of skin prick test (SPT) to D. pteronyssinus (Dp) and D. farinae (Df) allergens was determined for a wheal mean 3 mm-diameter larger than the negative control.
IgE to mite allergens in Tg-seropositive and Tg-seronegative subjects
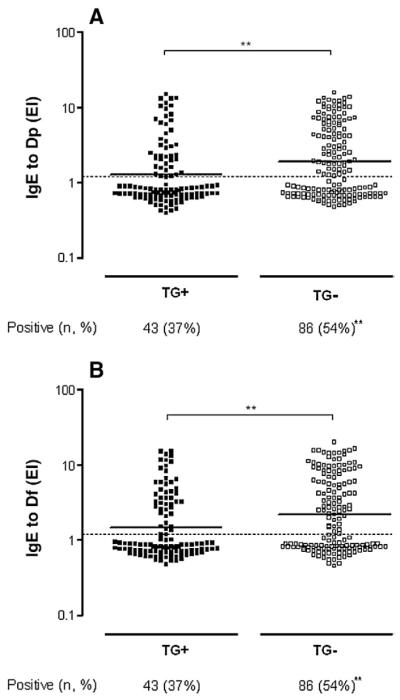
Levels of IgE to Dp and Df in Tg-seropositive and Tg-seronegative subjects are shown in Fig. 1. Levels of IgE to Dp (Fig. 1A) were significantly higher in TG− (geometric mean [gm]: 1.9; 95% CI: 1.6–2.2) than TG+ (gm: 1.3; 95% CI: 1.0–1.5; P = 0.0056) groups. Similar results were observed for IgE to Df (gm: 2.2; 95% CI: 1.8–2.6 vs. gm: 1.4; 95% CI: 1.2–1.7; P=0.001) (Fig. 1B). Also, the percentages of positive samples for IgE to Dp and Df were significantly higher in TG− (54%) than TG+ (37%) groups (P=0.0052).
Figure 1.
Levels of IgE to allergens of Dermatophagoides pteronyssinus (Dp, A) and D. farinae (Df, B), expressed in ELISA index (EI), in serum samples of Toxoplasma gondii seropositive (TG+, n=116) and seronegative (TG−, n=159) subjects. Horizontal bars indicate the geometric mean obtained for each group and dotted lines represent the cut off value (EI>1.2). Absolute (n) and relative (%) frequencies of positive samples for IgE to each allergen are also demonstrated. **P<0.01.
IgG to T. gondii in atopic and non-atopic patients
Levels of IgG to T. gondii were significantly higher in non-atopic individuals (gm: 2.4; 95% CI: 1.9–3.0) than atopic patients (gm: 1.5; 95% CI: 1.2–1.9; P=0.0038) (Fig. 2). Similarly, seropositivity to T. gondii was significantly higher in non-atopics (50%) than atopics (33%) (P=0.0052).
Figure 2.
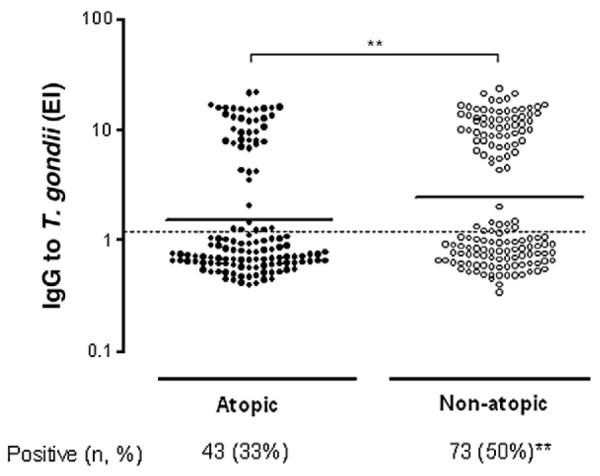
Levels of IgG to T. gondii, expressed in ELISA index (EI), in serum samples of atopic (n=129) and non-atopic (n=146) patients. Horizontal bars indicate the geometric mean obtained for each group and dotted lines represent the cut off value (EI>1.2). Absolute (n) and relative (%) frequencies of positive samples for IgG to T. gondii are also demonstrated. **P<0.01.
Total serum IgE in Tg-seropositive and Tg-seronegative patients
As shown in Fig. 3, total serum IgE levels were significantly higher in atopics (median: 198.5 IU/ml) than non-atopics (median: 26.0 IU/ml) within the TG+ group (P<0.0001). The same was observed in the TG− group, where atopic patients had higher levels of total serum IgE (median: 187.3 IU/ml) than non-atopics (median: 14.9 IU/ml) (P<0.0001). There was no significant difference in total serum IgE levels between Tg-seropositive and Tg seronegative individuals, whether atopics or non-atopics.
Figure 3.
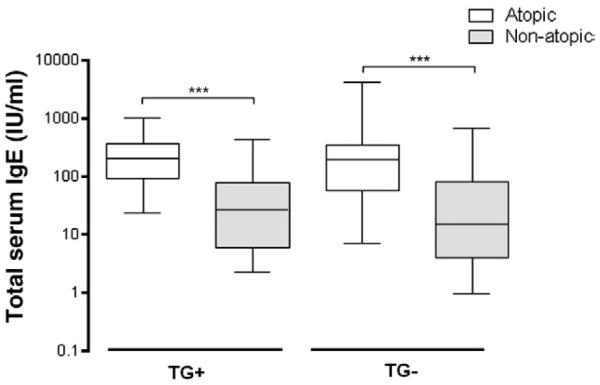
Total serum IgE levels, expressed in international units per milliliter (IU/ml), in serum samples of T. gondii seropositive (TG+, n=116) and seronegative (TG−, n=159) subjects. Data are represented in box and whisker plot, with box indicating median and 25–75% percentiles, and whiskers indicating the range of the data. ***P<0.0001.
Association between atopy and T. gondii infection
The overall proportion of TG+ atopics (16%) was significantly lower as compared to TG+ non-atopics (26%) (P= 0.0017) and TG− atopics (31%) (P<0.0001) (data not shown). The odds ratio (OR) calculated for association between atopy and positive serology to T. gondii was 0.50 (95% CI: 0.31–0.81; P<0.05) while the association between atopy and negative serology to T. gondii resulted in OR of 2.0 (95% CI: 1.2–3.3; P<0.05).
Proliferative response
Proliferative responses were analyzed in 40 individuals equally distributed in four groups: TG+ atopic, TG+ non-atopic, TG− atopic, and TG− non-atopic (Fig. 4). Proliferative response induced by STAg was higher in TG+ groups, atopic and non-atopic, than in both TG− groups (P<0.0001). Moreover, only TG+ groups showed higher proliferative response in relation to medium (P<0.0001). After stimulation with Dp allergen, no significant difference was found among the groups, although proliferative responses in all groups were higher in relation to medium (P<0.05). Proliferative response to Df allergen was higher in TG− atopic than non-atopic patients (P<0.0001), but only the TG− atopic group showed higher proliferative response in relation to medium (P<0.0001). As expected, after mitogen stimulation, proliferative responses were higher in all groups in relation to medium (P<0.0001), predominantly in the TG− atopic and non-atopic groups (P<0.05).
Figure 4.
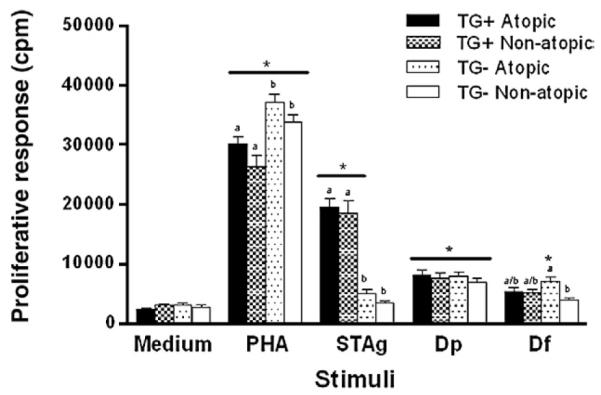
Proliferative response of peripheral blood mononuclear cells of 40 individuals equally distributed in four groups according to allergy and infection markers: TG+ atopic, TG+ non-atopic, TG− atopic and TG− non-atopic. Cells were stimulated with phytohemagglutinin (PHA; 5 μg/ml) or soluble antigen of T. gondii (STAg; 10 μg/ml) or D. pteronyssinus (Dp; 10 μg/ml) and D. farinae (Df; 10 μg/ml) allergen extracts. Data are expressed as mean and standard error of the mean (SEM). a,bDifferent letters indicate significant differences between the groups for each stimulus; *Statistically significant in relation to medium (control) for each group (P<0.05 determined by the Kruskal–Wallis and Dunn multiple comparison tests).
Cytokine profile
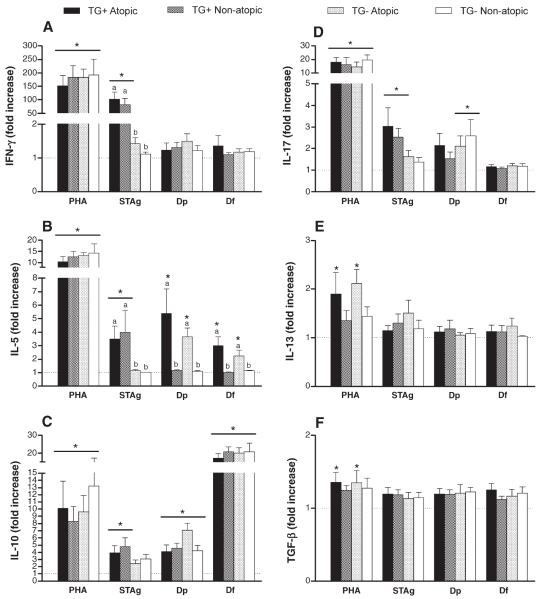
Cytokine levels were measured in PBMC culture supernatants after mitogen (PHA), antigen (STAg) or allergen (Dp and Df) stimulation (Fig. 5). After STAg stimulation, elevated IFN-γ levels were detected in both TG+ groups and were higher than in TG− atopic and non-atopic groups as well as in relation to baseline (P<0.001) (Fig. 5A). After stimulation with Dp or Df, IFN-γ levels were low with no significant difference among the groups and in relation to baseline. On the other hand, after mitogen stimulation, all groups showed increased IFN-γ levels as compared to baseline (P<0.0001).
Figure 5.
Cytokine levels in culture supernatants of peripheral blood mononuclear cells of 40 individuals equally distributed in four groups according to allergy and infection markers: T. gondii seropositive atopic (TG+ atopic), T. gondii seropositive non-atopic (TG+ non-atopic), T. gondii seronegative atopic (TG− atopic) and T. gondii seronegative non-atopic (TG− non-atopic). Cells were stimulated with phytohemagglutinin (PHA; 5 μg/ml) or soluble antigen of T. gondii (STAg; 10 μg/ml) or D. pteronyssinus (Dp; 10 μg/ml) and D. farinae (Df; 10 μg/ml) allergen extracts. Supernatants were collected after 3 days of culture and analyzed for detection of IFN-γ (A), IL-5 (B), IL-10 (C), IL-17 (D), IL-13 (E) and TGF-β (F). Data are expressed as fold increase in relation to medium (baseline) obtained from each patient and represented as mean and standard error of the mean (SEM). The dashed lines indicate the baseline. a,bDifferent letters indicate significant differences between the groups for each stimulus; *Statistically significant in relation to medium (baseline) for each patient group (P<0.05 determined by the Kruskal–Wallis and Dunn multiple comparison tests).
Low levels of IL-5 were detected after STAg stimulation, although higher in both TG+ groups than in TG− groups and in relation to baseline (P<0.05) (Fig. 5B). In contrast, after stimulation with Dp, IL-5 levels were higher in atopic than non-atopic patients, whether in TG+ or TG− groups, as well as in relation to baseline (P<0.01). Similar results were observed after stimulation with Df, although with lower IL-5 levels than Dp stimulation (P<0.05). After mitogen stimulation, all groups showed elevated levels of IL-5 as compared to baseline (P<0.0001).
Increased levels of IL-10 were detected after Df stimulation in all groups of patients in relation to baseline (P<0.0001), but with no significant differences among them (Fig. 5C). After STAg stimulation, IL-10 levels were higher in relation to baseline (P<0.001), and slightly greater in both TG+ groups than in the TG− counterparts. No significant difference was found among the groups after Dp or PHA stimulation, although all groups showed increased IL-10 levels as compared to baseline (P<0.05).
High levels of IL-17 were detected after PHA stimulation in all groups of patients in relation to baseline (P<0.0001), but with no significant differences among them (Fig. 5D). Increased IL-17 levels were also detected after STAg stimulation in TG+ groups and after Dp stimulation in TG− groups in comparison to baseline (P<0.05), while no change was observed after stimulation with Df in all the groups.
IL-13 (Fig. 5E) and TGF-β (Fig. 5F) levels were increased only after PHA stimulation in atopic groups (P<0.05), whether TG+ or TG− groups, and all other stimulations resulted in baseline levels.
Discussion
In the present study, we evaluated the association between atopy and T. gondii infection, analyzing the humoral and cellular immune responses in four subject groups according to allergy and infection markers. Analyzing the allergy markers among TG+ and TG− subjects, the higher percentage of positive SPT to mite allergens (Dp and Df) seen in the TG− group (54%) indicates a negative association between atopy and T. gondii infection in the population under study. Similarly, results of specific serum IgE to Dp and Df allergens showing a higher mite sensitization in the TG− group reinforce the inverse relationship between parasite infection and allergenic sensitization. However, total serum IgE response showed no significant differences between TG+ and TG− subjects, indicating that the parasite infection does not appear to influence the total serum IgE levels, in opposition to what was seen with specific serum IgE levels to mite allergens.
Analyzing the infection markers among atopic and non-atopic patients, the humoral immune response to T. gondii showing higher levels of parasite-specific IgG and higher seropositivity among non-atopic subjects emphasizes that the presence of a serological marker of chronic infection in toxoplasmosis was inversely related to the allergenic sensitization. These findings support that parasite infection and type of immune response induced by a pathogen as T. gondii can exert inhibitory effects on the induction of mite allergen sensitization.
Conflicting results have been reported in several epidemiological studies on the association between T. gondii infection and atopic diseases [14-20]. Our findings agree with those reports of lower prevalence of T. gondii infection in atopic patients compared to non-atopics [15-17,20,21], reinforcing the hypothesis that a higher exposure to T. gondii reduces sensitization on population level. On the other hand, other studies did not find any association between seropositivity to T. gondii and atopy [14,18,19]. One likely explanation for these contradictory findings may be the selection criteria used for definition of atopy [21], since the prevalence of respiratory allergic diseases increases linearly with the increase in specific serum IgE levels [22]. In the present study, atopy was defined by the concomitant presence of positive SPT and ELISA-IgE to Dp or Df allergens, with values of EI>1.2, that is, 20% above the threshold to exclude cut off borderline values.
Analyzing the cellular immune response, a strong proliferative response associated with elevated IFN-γ synthesis was observed after STAg stimulation only in TG+ groups, whether atopics or non-atopics. These findings demonstrate a predominance of T cell-mediated immunity specific to the parasite, characterized by a Th1-type polarized immune response with production of high IFN-γ levels even in chronic phase of infection [23]. Recently, experimental evidence of the hypothesis that T. gondii contributes to protect against atopy was demonstrated in a murine model of allergic inflammation, verifying that both acute and chronic parasite infections substantially block the development of airway inflammation and are related to high concentrations of Th1-type cytokines [7]. On the other hand, we have demonstrated the interference of Th2 immune responses induced by the acarian Myocoptes musculinus in naturally infected mice in the outcome of experimental T. gondii infection [24].
Another pro-inflammatory cytokine investigated in the present study was IL-17, which is produced by Th17 cells and is also involved in inflammatory allergic responses [25,26]. However, we were unable to demonstrate significant differences in IL-17 levels among the groups after any stimuli, probably because patients defined as atopics had no allergy clinical symptoms at the time of analysis. On the other hand, IL-17 levels were markedly increased after PHA stimulation in all patient groups, indicating a strong response to mitogen stimulation. Interestingly, IL-17 levels were also increased in TG+ groups after STAg stimulus and in TG− groups after Dp stimulus, suggesting that PBMCs from TG+ patients are capable to produce IL-17 after antigen specific restimulation, while the same was not observed for PBMCs from atopic patients after allergen specific restimulation. These findings may be explained by the fact that IL-17 is often associated with the severity of allergic diseases and considered a marker of severe allergy [27].
In the present study, Th2 typical cytokines as IL-5 prevailed in atopic than non-atopic groups, regardless of seroprevalence to T. gondii. Stimulation with Dp or Df allergens induced weak proliferative response in all subject groups, although the Df allergen was able to induce significant proliferative responses only in TG− atopic patients. Therefore, our results indicate that cells from atopic patients are prone to produce Th2 profile cytokines inductively by allergen restimulation [28], even in the presence of concurrent infections. Furthermore, Tg-sero-positive patients also produced detectable IL-5 levels in response to STAg, although it is noticeable the high ratio IFN-γ/IL-5 after specific antigen stimulation. Another Th2 type cytokine analyzed was IL-13, but none of the stimuli was able to induce significant differences among the groups, except for slightly increased IL-13 levels after PHA stimulation in atopic groups only, suggesting that PBMCs of atopic patients seem to be more responsive to mitogen stimulation than non-atopic patients.
Response of regulatory cytokines, such as IL-10, was verified in all groups, markedly after stimulation with Df allergen. Studies have shown that PBMCs from atopic patients produce higher levels of IL-10 than non-atopics after allergen restimulation [28]. Additionally, in the present study, higher IL-10 production after STAg stimulation was detected in PBMC from TG+ than TG− groups, confirming previous reports that T. gondii infection also induces the production of IL-10 with regulatory activities [29]. In this context, infections may protect against the development of allergies by inducing the production of regulatory cytokines such as IL-10 and TGF-β, which control both Th1 and Th2 responses, reducing the manifestations of disease and damage to the host [30,31]. Moreover, IL-10 levels have been inversely correlated with the incidence or severity of allergic diseases [32]. Therefore, the findings of IL-10 and IL-17 in our study may be related to the absence the allergic clinical symptoms in atopic patients at the time of analysis. Another regulatory cytokine analyzed was TGF-β, but similarly to IL-13, no significant differences were found among the groups, except for PHA stimulation in atopic groups only, suggesting that PBMCs of atopic patients are prone to produce TGF-β after mitogen stimulation.
Altogether, the inverse association between infection and atopy may be representative for the prevention of sensitization rather than clinical disease. The immunological mechanisms involved in this protective effect of exposure to infections are still controversial. They could be mediated, at least in part, by Th1-type cytokines, such as IFN-γ, induced by pathogens, leading to changes in Th2/Th1 balance, but the role of regulatory cytokines such as IL-10 should not be neglected. In conclusion, a negative association between atopy and infection by T. gondii was demonstrated for the first time in Brazilian subjects, focusing on the antibody and cytokine responses and indicating that the presence of T. gondii infection can influence negatively in the induction of allergen sensitization rather than the manifestation of allergic symptoms. Therefore, the immunomodulation induced by the parasite may play a protective role in the development of allergic diseases.
Acknowledgments
The project was supported by grants of the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG; F-0590/06 and F-5394/07) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; No. 480010/2007/02) from the Brazilian Foundation Agencies. J.F.C.F. was funded by a Master Science scholarship of the CNPq.
References
- [1].Garn H, Renz H. Epidemiological and immunological evidence for the hygiene hypothesis. Immunobiology. 2007;212:441–452. doi: 10.1016/j.imbio.2007.03.006. [DOI] [PubMed] [Google Scholar]
- [2].Strachan DP. Hay fever, hygiene, and household size. BMJ. 1989;299:1259–1260. doi: 10.1136/bmj.299.6710.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Galvão CES, Kalil J, Castro FFM. Sensibilização a aeroalérgenos em dois grupos escolares nas zonas rural e urbana de São Paulo, Brasil. Rev. Bras. Alergia Imunopatol. 2002;25:2–9. [Google Scholar]
- [4].Gore C, Custovic A. Protective parasites and medicinal microbes? The case for hygiene hypothesis, Prim. Care Respir. J. 2004;13:68–75. doi: 10.1016/j.pcrj.2004.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Vercelli D. Mechanisms of the hygiene hypothesis—molecular and otherwise. Curr. Opin. Immunol. 2006;18:733–737. doi: 10.1016/j.coi.2006.09.002. [DOI] [PubMed] [Google Scholar]
- [6].Sheikh A, Strachan DP. The hygiene theory: fact of fiction? Curr. Opin. Otolaryngol. Head Neck Surg. 2004;12:232–236. doi: 10.1097/01.moo.0000122311.13359.30. [DOI] [PubMed] [Google Scholar]
- [7].Fenoy I, Giovannoni M, Batalla E, Martin V, Frank FM, Piazzon I, Goldman A. Toxoplasma gondii infection blocks the development of allergic airway inflammation in BALB/c mice. Clin. Exp. Immunol. 2009;155:275–284. doi: 10.1111/j.1365-2249.2008.03813.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Oppenheimer J, Nelson HS. Skin Testing. Ann. Allergy Asthma Immunol. 2006;96:6–12. doi: 10.1016/s1081-1206(10)60895-2. [DOI] [PubMed] [Google Scholar]
- [9].Pereira EAL, Silva DAO, Cunha-Júnior JP, Almeida KC, Alves R, Sung SJ, Taketomi EA. IgE, IgG1, and IgG4 antibody responses to Blomia tropicalis in atopic patients. Allergy. 2005;60:401–406. doi: 10.1111/j.1398-9995.2005.00738.x. [DOI] [PubMed] [Google Scholar]
- [10].Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- [11].Mineo JR, Camargo ME, Ferreira AW. Enzyme-linked immunosorbent assay for antibodies to Toxoplasma gondii polysaccharides in human toxoplasmosis. Infect. Immun. 1980;27:283–287. doi: 10.1128/iai.27.2.283-287.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Silva DAO, Lobato J, Mineo TW, Mineo JR. Evaluation of serological tests for the diagnosis of Neospora caninum infection in dogs: optimization of cut off titers and inhibition studies of cross-reactivity with Toxoplasma gondii. Vet. Parasitol. 2007;143:234–244. doi: 10.1016/j.vetpar.2006.08.028. [DOI] [PubMed] [Google Scholar]
- [13].Silva DAO, Gervásio AM, Sopelete MC, Arruda-Chaves E, Arruda KL, Chapman MD, Sung-Sang JS, Taketomi EA. A sensitive reverse ELISA for the measurement of specific IgE to Der p 2, a major Dermatophagoides pteronyssinus allergen. Ann. Allergy Asthma Immunol. 2001;86:545–550. doi: 10.1016/S1081-1206(10)62903-1. [DOI] [PubMed] [Google Scholar]
- [14].Bodner C, Anderson WJ, Reid TS, Godden DJ. Childhood exposure to infection and risk of adult onset wheeze and atopy. Thorax. 2000;55:383–387. doi: 10.1136/thorax.55.5.383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Matricardi PM, Rosmini F, Rapicetta M, Gasbarrini G, Stroffolini T, San Marino Study Group Atopy, hygiene, and anthroposophic lifestyle. Lancet. 2000;354:430. doi: 10.1016/s0140-6736(05)75853-0. [DOI] [PubMed] [Google Scholar]
- [16].Matricardi PM, Rosmini F, Panetta V, Ferrigno L, Bonini S. Hay fever and asthma in relation to markers of infection in the United States. J. Allergy Clin. Immunol. 2002;110:381–387. doi: 10.1067/mai.2002.126658. [DOI] [PubMed] [Google Scholar]
- [17].Linneberg A, Ostergaard C, Tvede M, Andersen LP, Nielsen NH, Frolund L, Dirksen A, Jorgensen T. IgG antibodies against microorganisms and atopic disease in Danish adults: the Copenhagen Allergy Study. J. Allergy Clin. Immunol. 2003;111:847–853. doi: 10.1067/mai.2003.1335. [DOI] [PubMed] [Google Scholar]
- [18].Radon K, Windstetter D, Eckart J, Dressel H, Leitritz L, Reichert J, Schmid M, Praml G, Schosser M, Von Mutius E, Nowak D. Farming exposure in childhood, exposure to markers of infections and the development of atopy in rural subjects. Clin. Exp. Allergy. 2004;34:1178–1183. doi: 10.1111/j.1365-2222.2004.02005.x. [DOI] [PubMed] [Google Scholar]
- [19].Birgisdóttir A, Asbjörnsdottir H, Cook E, Gislason D, Jansson C, Olafsson I, Gislason T, Jogi R, Thjodleifsson B. Seroprevalence of Toxoplasma gondii in Sweden, Estonia and Iceland. Scand. J. Infect. Dis. 2006;38:625–631. doi: 10.1080/00365540600606556. [DOI] [PubMed] [Google Scholar]
- [20].Janson C, Asbjornsdottir H, Birgisdottir RB, Gunnbjörnsdottir M, Gislason D, Olafsson I, Cook E, Jögi R, Gislason T, Thjodleifsson B. The effect of infectious burden on the prevalence of atopy and respiratory allergies in Iceland, Estonia, and Sweden. J. Allergy Clin. Immunol. 2007;120:673–679. doi: 10.1016/j.jaci.2007.05.003. [DOI] [PubMed] [Google Scholar]
- [21].Ellersten LK, Hetland G, Lovik M. Specific IgE to respiratory allergens and IgG antibodies to Toxoplasma gondii and Streptococcus pneumoniae in Norwegian military recruits. Clin. Immunol. 2008;67:496–500. doi: 10.1111/j.1365-3083.2008.02090.x. [DOI] [PubMed] [Google Scholar]
- [22].Simpson A, Soderstrom L, Ahristedt S, Murray CS, Woodcock A, Custovic A. IgE antibody quantification and the probability of wheeze in preschool children. J. Allergy Clin. Immunol. 2005;116:744–749. doi: 10.1016/j.jaci.2005.06.032. [DOI] [PubMed] [Google Scholar]
- [23].Denkers EY, Gazzinelli RT, Martin D. Emergence of NK1.1+ cells as effectors of IFN-γ dependent immunity to Toxoplasma gondii in MHC class 1-deficient mice. J. Exp. Med. 1993;178:1465–1472. doi: 10.1084/jem.178.5.1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Welter A, Mineo JR, Silva DAO, Lourenço EV, Ferro EAV, Roque-Barreira MC, Silva NM. An opposite role is exerted by the acarian Myocoptes musculinus in the outcome of Toxoplasma gondii infection according to the route of the protozoa inoculation. Microbes Infect. 2006;8:2618–2628. doi: 10.1016/j.micinf.2006.07.006. [DOI] [PubMed] [Google Scholar]
- [25].Oboki K, Ohno T, Saito H, Nakae S. Th17 and allergy. Allergol. Intern. 2008;57:121–134. doi: 10.2332/allergolint.R-07-160. [DOI] [PubMed] [Google Scholar]
- [26].Wang Y, Liu Y. The IL-17 cytokine family and their role in allergic inflammation. Curr. Opin. Immunol. 2008;20:697–702. doi: 10.1016/j.coi.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Ciprandi G, Fenoglio D, De Amici M, Quaglini S, Negrini S, Filaci G. Serum IL-17 levels in patients with allergic rhinitis. J. Allergy Clin. Immunol. 2008;122:650–651. doi: 10.1016/j.jaci.2008.06.005. [DOI] [PubMed] [Google Scholar]
- [28].Hales BJ, Pearce LJ, Kusel MM, Holt PG, Sly PD, Thomas WR. Differences in the antibody response to a mucosal bacterial antigen between allergic and non-allergic subjects. Thorax. 2008;63:221–227. doi: 10.1136/thx.2006.069492. [DOI] [PubMed] [Google Scholar]
- [29].Jankovic D, Kullberg MC, Feng CG, Goldszmid RS, Collazo CM, Wilson M, Wynn TA, Kamanaka M, Flavell RA, Sher A. Conventional T-bet(+)Foxp3(−) Th1 cells are the major source of host-protective regulatory IL-10 during intracellular protozoan infection. J. Exp. Med. 2007;204:273–283. doi: 10.1084/jem.20062175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Robinson DS, Larché M, Durham SR. Tregs and allergic disease. J. Clin. Invest. 2004;114:1389–1397. doi: 10.1172/JCI23595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Romagnani S. Regulatory T cells: which role in the pathogenesis and treatment of allergic disorders? Allergy. 2006;61:3–14. doi: 10.1111/j.1398-9995.2006.01005.x. [DOI] [PubMed] [Google Scholar]
- [32].Akdis M, Verhagen J, Taylor A, Karamloo F, Karagiannidis C, Crameri R, Thunberg S, Deniz G, Valenta R, Fiebig H, Kegel C, Disch R, Schmidt-Weber CB, Blaser K, Akdis CA. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J. Exp. Med. 2004;199:1567–1575. doi: 10.1084/jem.20032058. [DOI] [PMC free article] [PubMed] [Google Scholar]