Abstract
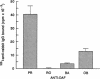
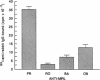
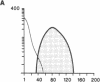
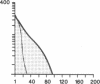
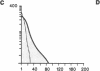
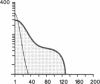
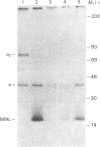
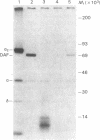
Susceptibility to hemolysis initiated by activated cobra venom factor (CoF) complexes is a characteristic that distinguishes the most complement-sensitive type III erythrocytes of paroxysmal nocturnal hemoglobinuria (PNH) from the intermediately sensitive type II and the normally sensitive type I cells. Recently we isolated a membrane constituent from normal erythrocytes that inhibits CoFBb-initiated hemolysis, and this protein was designated membrane inhibitor of reactive lysis (MIRL). To investigate the molecular basis of the variability in complement sensitivity among PNH erythrocytes, the surface expression of MIRL and decay accelerating factor (DAF) on the three phenotypes of PNH was quantified immunochemically. Both complement regulatory proteins were markedly deficient on the erythrocytes from a patient with predominately type III cells. The erythrocytes from patients with a majority of either type II or I cells were also significantly deficient in both MIRL and DAF. While cytofluorometric analysis confirmed the quantitative deficiencies, segregation of erythrocytes into discrete subpopulations that expressed either no MIRL or normal amounts of MIRL was not observed. The results of immunoprecipitation studies were consistent with quantitative, but not qualitative abnormalities of MIRL and DAF. Selective removal of the sensitive erythrocytes indicated that approximately 20% of the normal amount of MIRL is sufficient to protect cells from CoF-initiated lysis. These studies suggest that relatively subtle quantitative differences in membrane complement regulatory proteins underlie the variability in complement sensitivity of PNH erythrocytes.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burroughs S. F., Devine D. V., Browne G., Kaplan M. E. The population of paroxysmal nocturnal hemoglobinuria neutrophils deficient in decay-accelerating factor is also deficient in alkaline phosphatase. Blood. 1988 Apr;71(4):1086–1089. [PubMed] [Google Scholar]
- Davitz M. A., Low M. G., Nussenzweig V. Release of decay-accelerating factor (DAF) from the cell membrane by phosphatidylinositol-specific phospholipase C (PIPLC). Selective modification of a complement regulatory protein. J Exp Med. 1986 May 1;163(5):1150–1161. doi: 10.1084/jem.163.5.1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson M. A., Williams A. F. Cell-surface anchoring of proteins via glycosyl-phosphatidylinositol structures. Annu Rev Biochem. 1988;57:285–320. doi: 10.1146/annurev.bi.57.070188.001441. [DOI] [PubMed] [Google Scholar]
- Haziot A., Chen S., Ferrero E., Low M. G., Silber R., Goyert S. M. The monocyte differentiation antigen, CD14, is anchored to the cell membrane by a phosphatidylinositol linkage. J Immunol. 1988 Jul 15;141(2):547–552. [PubMed] [Google Scholar]
- Holguin M. H., Fredrick L. R., Bernshaw N. J., Wilcox L. A., Parker C. J. Isolation and characterization of a membrane protein from normal human erythrocytes that inhibits reactive lysis of the erythrocytes of paroxysmal nocturnal hemoglobinuria. J Clin Invest. 1989 Jul;84(1):7–17. doi: 10.1172/JCI114172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hänsch G. M., Schönermark S., Roelcke D. Paroxysmal nocturnal hemoglobinuria type III. Lack of an erythrocyte membrane protein restricting the lysis by C5b-9. J Clin Invest. 1987 Jul;80(1):7–12. doi: 10.1172/JCI113065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hänsch G. M., Weller P. F., Nicholson-Weller A. Release of C8 binding protein (C8bp) from the cell membrane by phosphatidylinositol-specific phospholipase C. Blood. 1988 Sep;72(3):1089–1092. [PubMed] [Google Scholar]
- Kinoshita T., Rosenfeld S. I., Nussenzweig V. A high m.w. form of decay-accelerating factor (DAF-2) exhibits size abnormalities in paroxysmal nocturnal hemoglobinuria erythrocytes. J Immunol. 1987 May 1;138(9):2994–2998. [PubMed] [Google Scholar]
- Low M. G., Saltiel A. R. Structural and functional roles of glycosyl-phosphatidylinositol in membranes. Science. 1988 Jan 15;239(4837):268–275. doi: 10.1126/science.3276003. [DOI] [PubMed] [Google Scholar]
- Medof M. E., Gottlieb A., Kinoshita T., Hall S., Silber R., Nussenzweig V., Rosse W. F. Relationship between decay accelerating factor deficiency, diminished acetylcholinesterase activity, and defective terminal complement pathway restriction in paroxysmal nocturnal hemoglobinuria erythrocytes. J Clin Invest. 1987 Jul;80(1):165–174. doi: 10.1172/JCI113043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medof M. E., Kinoshita T., Nussenzweig V. Inhibition of complement activation on the surface of cells after incorporation of decay-accelerating factor (DAF) into their membranes. J Exp Med. 1984 Nov 1;160(5):1558–1578. doi: 10.1084/jem.160.5.1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medof M. E., Walter E. I., Roberts W. L., Haas R., Rosenberry T. L. Decay accelerating factor of complement is anchored to cells by a C-terminal glycolipid. Biochemistry. 1986 Nov 4;25(22):6740–6747. doi: 10.1021/bi00370a003. [DOI] [PubMed] [Google Scholar]
- Nicholson-Weller A., Burge J., Fearon D. T., Weller P. F., Austen K. F. Isolation of a human erythrocyte membrane glycoprotein with decay-accelerating activity for C3 convertases of the complement system. J Immunol. 1982 Jul;129(1):184–189. [PubMed] [Google Scholar]
- Nicholson-Weller A., March J. P., Rosenfeld S. I., Austen K. F. Affected erythrocytes of patients with paroxysmal nocturnal hemoglobinuria are deficient in the complement regulatory protein, decay accelerating factor. Proc Natl Acad Sci U S A. 1983 Aug;80(16):5066–5070. doi: 10.1073/pnas.80.16.5066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pangburn M. K., Schreiber R. D., Müller-Eberhard H. J. Deficiency of an erythrocyte membrane protein with complement regulatory activity in paroxysmal nocturnal hemoglobinuria. Proc Natl Acad Sci U S A. 1983 Sep;80(17):5430–5434. doi: 10.1073/pnas.80.17.5430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker C. J., Baker P. J., Rosse W. F. Increased enzymatic activity of the alternative pathway convertase when bound to the erythrocytes of paroxysmal nocturnal hemoglobinuria. J Clin Invest. 1982 Feb;69(2):337–346. doi: 10.1172/JCI110457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker C. J., Soldato C. M., Rosse W. F. Abnormality of glycophorin-alpha on paroxysmal nocturnal hemoglobinuria erythrocytes. J Clin Invest. 1984 Apr;73(4):1130–1143. doi: 10.1172/JCI111299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker C. J., White V. F., Falk R. J. Site-specific activation of the alternative pathway of complement. Synthesis of a hybrid molecule consisting of antibody and cobra venom factor. Complement. 1986;3(4):223–235. [PubMed] [Google Scholar]
- Parker C. J., Wiedmer T., Sims P. J., Rosse W. F. Characterization of the complement sensitivity of paroxysmal nocturnal hemoglobinuria erythrocytes. J Clin Invest. 1985 Jun;75(6):2074–2084. doi: 10.1172/JCI111927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosse W. F., Dacie J. V. Immune lysis of normal human and paroxysmal nocturnal hemoglobinuria (PNH) red blood cells. I. The sensitivity of PNH red cells to lysis by complement and specific antibody. J Clin Invest. 1966 May;45(5):736–748. doi: 10.1172/JCI105388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosse W. F., Parker C. J. Paroxysmal nocturnal haemoglobinuria. Clin Haematol. 1985 Feb;14(1):105–125. [PubMed] [Google Scholar]
- Rosse W. F. Variations in the red cells in paroxysmal nocturnal haemoglobinuria. Br J Haematol. 1973 Mar;24(3):327–342. doi: 10.1111/j.1365-2141.1973.tb01657.x. [DOI] [PubMed] [Google Scholar]
- Schönermark S., Rauterberg E. W., Shin M. L., Löke S., Roelcke D., Hänsch G. M. Homologous species restriction in lysis of human erythrocytes: a membrane-derived protein with C8-binding capacity functions as an inhibitor. J Immunol. 1986 Mar 1;136(5):1772–1776. [PubMed] [Google Scholar]
- Selvaraj P., Dustin M. L., Silber R., Low M. G., Springer T. A. Deficiency of lymphocyte function-associated antigen 3 (LFA-3) in paroxysmal nocturnal hemoglobinuria. Functional correlates and evidence for a phosphatidylinositol membrane anchor. J Exp Med. 1987 Oct 1;166(4):1011–1025. doi: 10.1084/jem.166.4.1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selvaraj P., Rosse W. F., Silber R., Springer T. A. The major Fc receptor in blood has a phosphatidylinositol anchor and is deficient in paroxysmal nocturnal haemoglobinuria. Nature. 1988 Jun 9;333(6173):565–567. doi: 10.1038/333565a0. [DOI] [PubMed] [Google Scholar]
- Seya T., Farries T., Nickells M., Atkinson J. P. Additional forms of human decay-accelerating factor (DAF). J Immunol. 1987 Aug 15;139(4):1260–1267. [PubMed] [Google Scholar]
- Sirchia G., Dacie J. V. Immune lysis of AET-treated normal red cells (PNH-like cells). Nature. 1967 Aug 12;215(5102):747–748. doi: 10.1038/215747a0. [DOI] [PubMed] [Google Scholar]
- Sugita Y., Nakano Y., Tomita M. Isolation from human erythrocytes of a new membrane protein which inhibits the formation of complement transmembrane channels. J Biochem. 1988 Oct;104(4):633–637. doi: 10.1093/oxfordjournals.jbchem.a122524. [DOI] [PubMed] [Google Scholar]
- Vogel C. W., Smith C. A., Müller-Eberhard H. J. Cobra venom factor: structural homology with the third component of human complement. J Immunol. 1984 Dec;133(6):3235–3241. [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Zalman L. S., Wood L. M., Frank M. M., Müller-Eberhard H. J. Deficiency of the homologous restriction factor in paroxysmal nocturnal hemoglobinuria. J Exp Med. 1987 Feb 1;165(2):572–577. doi: 10.1084/jem.165.2.572. [DOI] [PMC free article] [PubMed] [Google Scholar]