Abstract
Serotonin (5HT) is a platelet-stored vasoconstrictor. Altered concentrations of circulating 5HT are implicated in several pathologic conditions, including hypertension. The actions of 5HT are mediated by different types of receptors and terminated by a single 5HT transporter (SERT). Therefore, SERT is a major mechanism that regulates plasma 5HT levels to prevent vasoconstriction and thereby secure a stable blood flow. In this study, the response of platelet SERT to the plasma 5HT levels was examined within two models: (i) in subjects with chronic hypertension or normotension; (ii) on platelets isolated from normotensive subjects and pretreated with 5HT at various concentrations. The platelet 5HT uptake rates were lower during hypertension due to a decrease in Vmax with a similar Km; also, the decrease in Vmax was primarily due to a decrease in the density of SERT on the platelet membrane, with no change in whole cell expression. Additionally, while the platelet 5HT content decreased 33%, the plasma 5HT content increased 33%. Furthermore, exogenous 5HT altered the 5HT uptake rates by changing the density of SERT molecules on the plasma membrane in a biphasic manner. Therefore, we hypothesize that in a hypertensive state, the elevated plasma 5HT levels induces a loss in 5HT uptake function in platelets via a decrease in the density of SERT molecules on the plasma membrane. Through the feedback effect of this proposed mechanism, plasma 5HT controls its own concentration levels by modulating the uptake properties of platelet SERT.
Keywords: hypertension, plasma serotonin level, platelet serotonin uptake, serotonin, serotonin transporter
Serotonin (5HT) is an intermediate product of tryptophan metabolism located primarily in the enterochromaffin cells of the intestines, serotonergic neurons of the brain, and blood platelets, and is a well established neurotransmitter in the central nervous system. Although altered concentrations of circulating 5HT are implicated in several pathologic conditions, including hypertension, the mechanism underlying involvement of 5HT remains elusive. 5HT has potent vasoconstrictor activity (Watts 2005) and also enhances the hypertensive effects of known vasoconstrictors (Golino et al. 1991;Gujrati et al. 1994; Azzadin et al. 1995; Yildiz et al. 1996; Watts 2005). In hypertension and atherosclerosis (Golino et al. 1991; Yildiz et al. 1996), the blood vessels become more sensitive to the vasoconstrictor effects of 5HT, and this effect is further amplified by subcontractile levels of other vasoconstrictors such as angiotensin and endothelin (Yildiz et al. 1998; Watts 2005). Furthermore, 5HT levels and turnover are increased in primary hypertension and certain types of secondary hypertension, such as pregnancy, erythropoietin, and cyclosporine-induced hypertension (Gujrati et al. 1994; Azzadin et al. 1995; Watts 2005). The actions of 5HT are mediated by different types of receptors, but are terminated by a single 5HT transporter (SERT) (Blakely et al. 1991; Hoffman et al. 1991; Lesch et al. 1993; Ramamoorthy et al. 1993b). The uptake mechanism of platelet SERT regulates plasma 5HT levels and secures stable blood flow by decreasing the possibility of platelet-activation, which produces clots in blood vessels, resulting in platelet aggregation, and subsequently, hypertension (Pettinger et al. 1973; Shulman et al. 1989; Lichtman et al. 2002; Blann et al. 2003; Stoddard et al. 2003; Pidgeon et al. 2004). The relation between the plasma 5HT level and the uptake ability of SERT has been demonstrated using SERT knock-out mice (Bengel et al. 1998), polymorphisms in the SERT-linked promoter region, and SERT inhibitors (Heils et al. 1996; Lesch et al. 1996; Serebruany et al. 2001; Johnson et al. 2003). Overall, these studies emphasize that the 5HT uptake efficiency of SERT depends on the plasma 5HT level and the number of transporters on the plasma membrane (Ramamoorthy and Blakely 1999). However, neither the contributions of these phenomena nor the order of events in the hypertensive state are known.
In the present study, we first measured 5HT levels in platelets and platelet-poor plasma (PPP). We then characterized the platelet 5HT uptake rates, the catalytic properties (Vmax), and the substrate-dependence (Km) of SERT in platelets from subjects with chronic hypertension or normotension. The platelet 5HT uptake rate during hypertension was lower due to a decrease in Vmax with no change in Km for the platelet SERT. To understand the mechanism for the reduction in 5HT uptake, the expressions of SERT on the plasma membrane and in the intracellular pools were measured. The findings indicated that the decrease in Vmax was due to a decrease in the amount of SERT molecules expressed on the platelet membrane but not in whole cell expression. Furthermore, we studied the interaction between the plasma 5HT levels and the density of SERT molecules on the platelet membrane. The effects of exogenous 5HT on the uptake rates and plasma membrane densities of SERT were measured in isolated platelets after pre-treating them with various concentrations of 5HT.
Together, our findings demonstrated that exogenous 5HT affects the uptake capacity of SERT by controlling the density of transporter molecules on the plasma membrane in a biphasic manner. Therefore, we hypothesize that in a hypertensive state, the elevated plasma 5HT levels induce a loss in the uptake function of platelets via a decrease in the density of SERT molecules on the plasma membrane. Through the feedback effect, plasma 5HT controls its own concentration levels by modulating the kinetic properties of platelet SERT.
Materials and methods
Subjects
Seventeen adult men between the ages of 45 and 50 years and with high blood pressure and 25 subjects with normal blood pressure (Table 1) were recruited for this study. Our study was carried out as part of a larger University of Arkansas for Medical Sciences (UAMS) IRB-approved investigation, which included these procedures, and for which subjects had previously provided written informed consent. The subjects had no other medical conditions. Women were not included to avoid the potential confounding influence of variations in average platelet age across the menstrual cycle on platelet 5HT uptake and storage, which could occur because younger platelets, proportionately more prevalent during menstruation, are larger, with a greater number of storage vesicles (Corash et al. 1984), and are very likely have relatively more SERT molecules (Corash et al. 1984; Arora and Meltzer 1989; Ozaki et al. 1994). Subjects who had medical problems, smoked, or took medications in the preceding 2 weeks were excluded from the study.
Table 1.
Clinical evaluation of the subject
| Hypertensive | Normotensive | |||
|---|---|---|---|---|
| Age (years) | Systolic (mmHg) | Diastolic (mmHg) | Systolic (mmHg) | Diastolic (mmHg) |
| 47 ± 3 | 212.67 ± 23.27a* | 110.5 ± 8.52a** | 113.6 ± 15.20a* | 81.00 ± 16.8a** |
Mean ± SD.
p = 0.034;
p = 0.017.
Blood sampling and platelet preparation
A blood sample (10 mL) was drawn into a heparin-containing tube by venipuncture from an antecubital vein. To one 5-mL aliquot of blood, Tyrode Hepes buffer (134 mmol/L NaCl, 0.34 mmol/L Na2HPO4, 2.9 mmol/L KCl, 12 mmol/L NaHCO3, 20 mmol/L HEPES, 5 mmol/L glucose, 1 mmol/L MgCl2 pH 7.3) was added and centrifuged at 200 g for 7.5 min at 20–25°C to separate platelet rich plasma (PRP). The supernatant, PRP, was transferred to another tube and Tyrode Hepes was added in a 1:0.5 volume. Samples were centrifuged at 200 g for 7.5 min at 20–25°C. The supernatant was centrifuged again at 1000 g for 5 min and platelet poor plasma (PPP) was separated from the pellet. The platelet pellet was then re-suspended in the appropriate buffer.
Quantitative measurement of 5HT levels by enzyme-linked immunosorbent assay
The 5HT levels of platelets and PPP prepared from 16 hypertensive and 16 normotensive subjects were measured by competitive enzyme-linked immunosorbent assay (ELISA) technique by following the manufacturer’s instructions (IBL Immuno-Biological Laboratories, Hamburg, Germany). Briefly, 5HT in samples and controls were acylated with acetic anhydride in acetone and samples, controls, and standards were applied to 96-well microtitre plates coated with goat anti-rabbit IgG. Biotinylated 5HT and rabbit antiserum to 5HT were added to each well and incubated overnight at 4°C. Para-nitrophenylphosphate in a diethanolamine solution was used as a substrate following the application of alkaline phosphatase conjugated goat anti-biotin antibody. Samples were read at 405 nm on an ELISA plate reader (Molecular Devices Union City, CA, USA). The amounts of 5HT were quantified using standards supplied by the manufacturer and analyzed using Origin software (Microcal Software, Northampton, MA, USA).
Platelet 5HT uptake assay
Platelet pellet was quickly washed with phosphate-buffered saline containing 0.1 mmol/L CaCl2 and 1 mmol/L MgCl2 (PBSCM) then resuspended in PBSCM with 14.6 nmol/L 3H-5HT at RT for 10 min, to include only the initial linear phase of transport in human platelets. Platelets were collected by rapid filtration through Whatman GF/B filters and were washed twice with 5 mL of ice-cold phosphate-buffered saline (PBS). Filters were placed in scintillation vials containing 5 mL scintillation cocktail and immediately counted.
Background accumulation of 3H-5HT that occurred independent of SERT was measured in the same experiment treating platelets with the high-affinity cocaine analog, 0.1 µmol/L 2β-carbomethoxy-3-tropane (β-CIT) (Chemical Synthesis Service, NIMH, Bethesda, MD, USA) and subtracted from each experimental value. As much as 0.1 mmol/L cocaine totally inactivates the 5HT uptake function of SERT (Heils et al. 1996; Ramamoorthy and Blakely 1999; Johnson et al. 2003; Ozaslan et al. 2003). In parallel, the protein concentration for 0.2 × 106 (0.015 mg cellular protein) platelets was determined using the Micro BCA protein Assay Kit (Pierce, Rockford, IL, USA). The 5HT uptake rates of transporters were calculated as means of standard deviation values from three independent experiments.
Western blot analysis
Platelets were washed, lysed and solubilized in PBS containing 0.44% sodium dodecyl sulfate (SDS), 1 mmol/L phenylmethylsulfonyl fluoride (PMSF), and protease inhibitor mixture (PIM). The PIM contained 5 mg/mL pepstatin and 50 mg/mL leupeptin, and 5 mg/mL aprotinin was included with each lysis buffer (Kilic et al. 2003), which also contained the alkylating agent N-ethylmaleimide (NEM) to a final concentration of 5 mmol/L to prevent oxidation and formation of non-specific disulfide bonds during lysis and to retain the native monomeric structures in the gel (Kocabas et al. 2003). Samples were analyzed by 8% SDS–polyacrylamide gel electrophoresis (PAGE) and transferred to the nitrocellulose membrane. Western blot analysis was performed, first with monoclonal human SERT antibody (hSERT-Ab) (MAb Technologies, Inc., Stone Mountain, GA, USA) (diluted 1 : 1000), and then with HRP-conjugated anti-mouse secondary antibodies at a dilution of 1 : 10 000. The signals were visualized using the ECL western blot detection system. Blots were visualized under a VersaDoc 1000 gel visualization and analysis system (BioRad Lab, Hercules, CA, USA).
Cell surface biotinylation
Platelet plasma membrane expression levels of hSERT were compared using the membrane-impermeant biotinylation reagent, NHS-SS-biotin (Pierce, Inc., Rockford, IL, USA). Platelets from 3 mL blood samples were prepared and washed twice with of ice-cold PBS/CM. Next, the platelets were incubated with NHS-SS-biotin (1.5 mg/mL) on ice with very gentle shaking. After biotinylation, platelets were first briefly rinsed and then incubated with PBS/CM containing 100 mmol/L glycine on ice. Platelets were lysed with 1% SDS–1% TX100. Then the biotinylated proteins were recovered from the platelet lysate using streptavidin–agarose beads (Pierce, Inc.) (Kilic and Rudnick 2000). The beads were washed with high salt, low salt and 50 mmol/L Tris-HCl (pH 7.5). The biotinylated proteins were eluted from the beads in SDS–PAGE sample buffer. The mercaptoethanol cleaves the disulfide bond of NHS-SS-biotin, releasing the recovered proteins from the biotin moiety and consequently from the streptavidin-agarose beads. The cell surface proteins were separated using SDS–PAGE and SERT was detected by western blot (Towbin et al. 1979) using the monoclonal hSERT-antibody. In human platelet one band of molecular weights 90 kDa is recognized for SERT.
Data analysis
Non-linear regression fits of experimental and calculated data were performed with Origin, which uses the Marquardt–Levenberg nonlinear least squares curve fitting algorithm. Each figure shows a representative experiment that was performed at least twice. The statistical analysis given in the Results section is from multiple experiments. Data with error bars represent the mean ± SD for triplicate samples. Comparison of paired continuous variables, chronic hypertensive and normotensive, was done by paired t-test. The level of significance was set at <0.05.
Results
Platelet and PPP 5HT levels
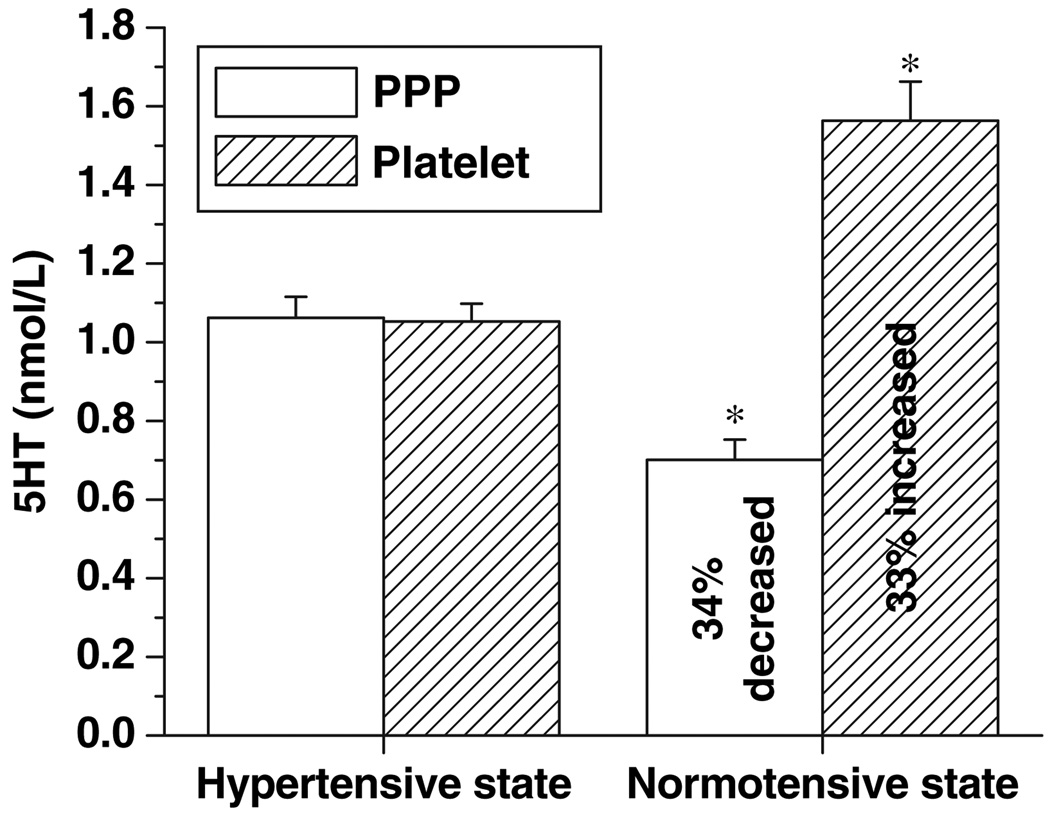
There is a growing body of evidence that plasma 5HT concentrations are directly related to systolic and diastolic blood pressure (Azzadin et al. 1995; Yildiz et al. 1996;Watts 2005). Before studying the relationship between plasma 5HT and platelet SERT, we measured the 5HT levels in the plasma and PPP of 16 subjects with chronic hypertension and normotension. The mean platelet 5HT concentration was lower during hypertension than normotension (1.06 ± 0.04 vs. 1.56 ± 0.09 nmol/L, respectively; p < 0.001). On the other hand, the mean PPP 5HT concentration was higher during hypertension than normotension (1.06 ± 0.050 vs. 0.70 ± 0.05 nmol/L, respectively; p < 0.001; Fig. 1).
Fig. 1.
Serotonin (5HT) contents of platelet and platelet-poor plasma (PPP) in hypertensive and normotensive states. Using a competitive ELISA technique, platelet and PPP 5HT concentrations from 16 subjects with hypertension were measured and compared with the values from normotension following the manufacturer’s instructions. Samples were read at 405 nm on an ELISA plate reader. 5HT amounts were quantified using standards supplied by the manufacturer, and analyzed using Origin software. The (*) represents the results of a two-tailed Student’s t-test with p < 0.001 (compared with normotensive subjects).
Kinetic characterization of platelet hSERT
For this and the following sections all the measurements were performed from the same samples in parallel. As much as 10 mL blood samples from seven hypertensive and seven normotensive subjects were drawn, and platelets from the blood samples were prepared (10 mL blood sample = 1.5 × 108 ± 350 platelets) and re-suspended in 10 mL PBCM; 1 mL from the platelet suspension was utilized to measure the 5HT uptake rates; 7 mL was used to study the Vmax and Km values; 2 mL was used to measure the whole platelet and plasma membrane density of transporters.
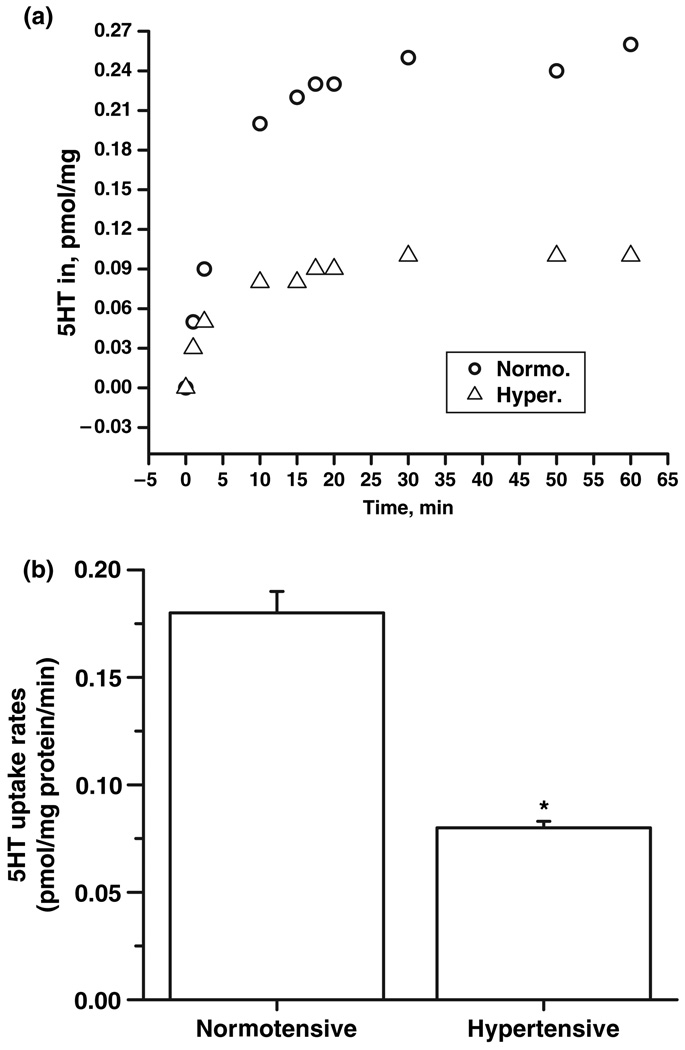
In order to understand the involvement of platelet SERT in the accumulation of 5HT in plasma during hypertensive states, 5HT rates of platelets from hypertensive and normotensive subjects were determined in platelet (c. 1.5 × 107 platelet/1 mL suspension) as described in the Materials and methods section and calculated as pmol of 5HT transported in a minute. Platelet pellet was resuspended in PBSCM with 14.6 nmol/L 3H-5HT for various times (Fig. 2a). As shown in Table 2, the 5HT uptake rate of platelet SERT during chronic hypertension was lower than normotensive state (Fig. 2b). The 45% reduction in 5HT uptake rates of platelets from hypertensive subjects was most pronounced at the earliest time points.
Fig. 2.
Serotonin (5HT) uptake rates of platelet single 5HT transporter (SERT) from hypertensive and normotensive subjects. (a) Platelets from the blood samples of seven hypertensive and seven normotensive subjects were prepared and assayed for [3H]-5HT uptake at 1, 2.5, 10, 15, 17.5, 20, 30, 50 and 60 min as described in Materials and methods. Background accumulation of [3H]-5HT was measured in the same experiment by treating platelets with 0.1 µmol/L high-affinity cocaine analog, 2β-carbomethoxy-3-tropane (β-CIT) and subtracted from each experimental value. The difference between the 5HT uptake rates of hypertensive and normotensive samples was most pronounced at the earliest time points, 10 min. (b) 5HT uptake rates of platelets prepared from 1 mL blood samples of seven hypertensive and seven normotensive subjects were measured by incubating with [3H]-5HT for 10 min as described in Materials and methods. The (*) represents the results of a two-tailed Student’s t-test with p < 0.001 (compared with normotensive subjects).
Table 2.
Kinetic characteristics of single 5HT transporter (SERT) in platelets from hypertensive subjects and after hypertension has resolved (normotensive)
| Rate (pmol/min/mg protein) | Km (µmol/L) * | Vmax** (pmol/min/mg protein) | |
|---|---|---|---|
| Hypertensive | 0.28 ± 8.5 ×10−5 a | 0.96 ± 0.06a* | 0.61 ± 0.06a** |
| Normotensive | 0.51 ± 0.0004a | 0.98 ± 0.04a* | 1.58 ± 0.06a** |
Mean ± SD.
p = 0.26;
p = 0.03.
Transport measurements were performed as described under Materials and Methods. Km and Vmax values and their associated uncertainties were obtained by nonlinear regression analysis.
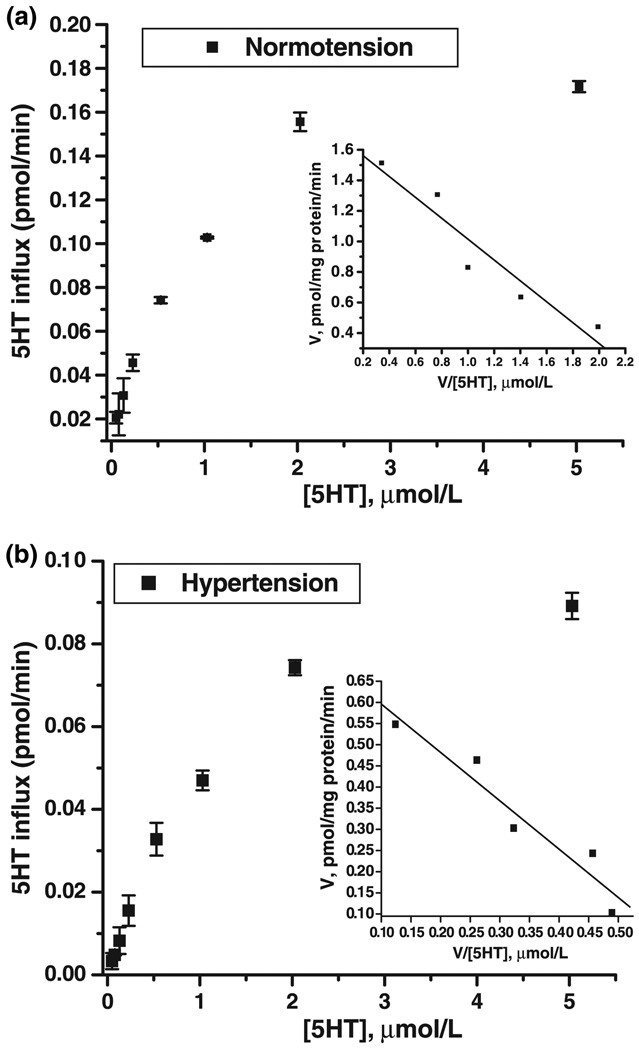
To analyze the mechanism involved in the decrease of 5HT uptake we measured the transport rate over a range of 5HT concentrations (0.02–5 µmol/L).The 5HT uptake measurements of platelets were performed in a triplicate experiment. Therefore, platelets in 7 mL PBSCM were distributed equally into the 27 tubes (4 × 106 platelets/tube). The cold 5HT in PBSCM at various concentrations (0.02–5 µmol/L) were mixed with 14.6 nmol/L 3H-5HT. Platelets were incubated at 20–25°C for 10 min an intervals as it is presented in Fig. 3. The 5HT uptake assays were followed as described in the Materials and methods section. Vmax values were determined by fitting the rate versus concentration data. The inset shows an Eadie–Hofstee plot of the data with lines drawn from the derived kinetic constants.
Fig. 3.
Substrate dependence of platelet hSERT from hypertensive and normotensive subjects. Platelets prepared from 7 mL blood samples of seven hypertensive and seven normotensive subjects were prepared as described under Materials and methods. Initial rates of 5HT uptake in platelets from hypertensive subjects (a) and normotensive subjects (b) were measured over the indicated range of 5HT concentrations using 20 nmol/L 3H-5HT with unlabeled 5HT added to the final concentration. Km and Vmax values were determined by fitting the rate versus concentration data. The inset shows an Eadie–Hofstee plot of the data with lines drawn from the derived kinetic constants. See Table 2 for a summary of the calculated data. Background SERT-independent accumulation of 3H-5HT was measured in the same experiment by treating platelets with 0.1 µmol/L high-affinity cocaine analog, 2β-carbomethoxy-3-tropane (β-CIT), and subtracted from each experimental value. Cocaine (0.1 mmol/L) totally inactivates the 5HT uptake function of SERT (Kilic and Rudnick 2000; Kilic et al. 2003; Kocabas et al. 2003; Ozaslan et al. 2003). The 5HT transporter uptake rates were calculated as means of standard deviation values from three independent experiments performed in triplicate.
The results in Fig. 3a demonstrate that the decreased uptake rate of platelets in hypertensive subjects was maintained over a wide range of substrate concentrations and those high substrate concentrations did not eliminate the difference between hypertensive and normotensive states (Figs 3a and b). These data were analyzed by fitting them to hyperbolic saturation kinetics, yielding a 1.17-fold difference in Vmax value and a similar Km value. SERT obtained in the hypertensive state had a lower maximal rate and saturated at a similar 5HT concentration than that obtained during the normotensive state (see the Eadie–Hofstee plot shown as an inset to Fig. 3a). The line representing normotensive state intersected the y-axis at a higher value (Vmax) than that of hypertensive state and with a similar slope (−KM).
SERT expression in platelets
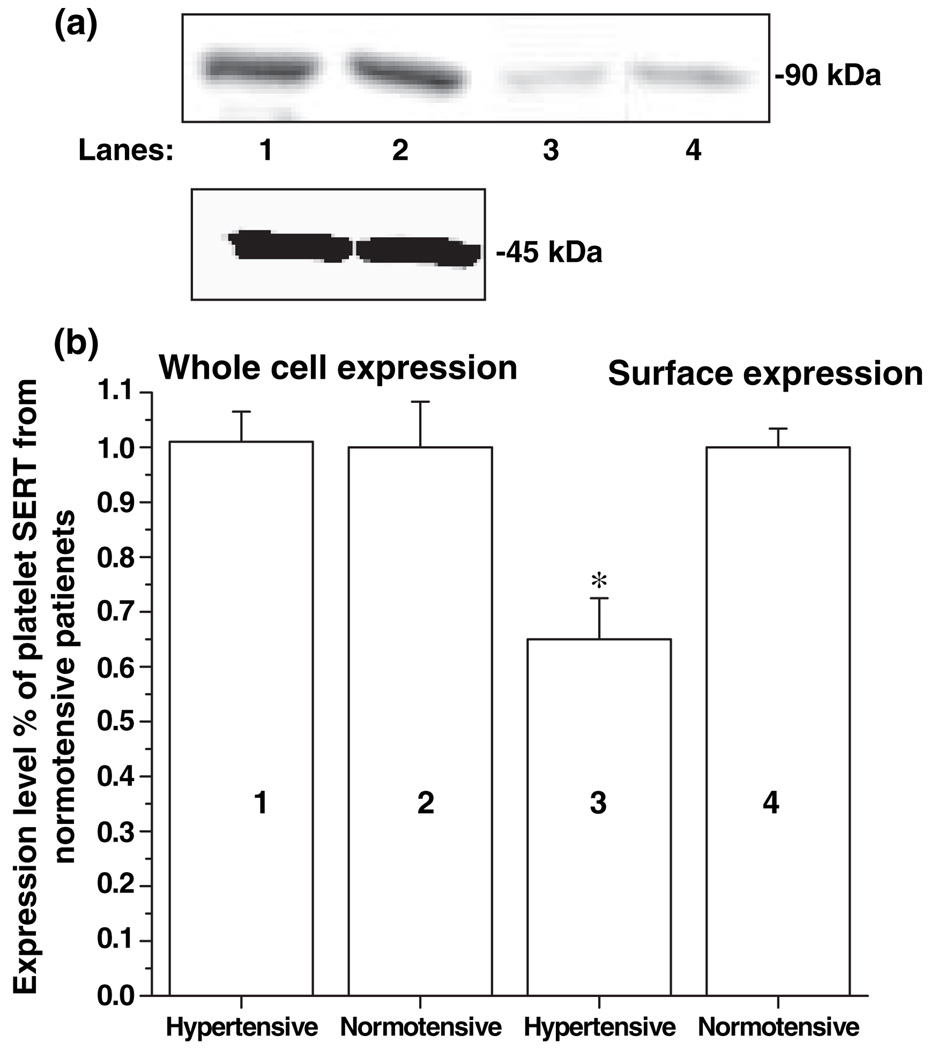
An additional possible explanation for the decreased Vmax is that the distribution of transporters between the cell surface and intracellular locations. To test this possibility, the intact platelets (c. 3 × 107 platelets) prepared from 2 mL blood samples of seven hypertensive and seven normotensive subjects were treated with membrane-impermeant biotinylating reagent sulfo-NHS-SS-biotin to label proteins on the cell surface. The platelets were then solubilized and biotinylated proteins were extracted using streptavidin-agarose. The plasma membrane SERT was then separated from the intracellular SERT. Both intracellular and plasma membrane SERT were analyzed by quantitative western blot analysis. The intracellular protein actin was used as a loading control for both plasma membrane and intracellular SERT.
There was a 35% decrease in the surface density of SERT molecules on platelets from hypertensive subjects when compared with the normotensive ones (p < 0. 01) (Figs 4a and b). The decreased surface expression during the hypertensive state explains the reason for decrease in Vmax of SERT.
Fig. 4.
Analysis of single 5HT transporter (SERT) expression in platelets from hypertensive and normotensive subjects. For whole-cell expression, platelets from 2 mL blood samples of seven hypertensive and seven normotensive subjects with (lanes 1 and 2, respectively) were lysed, and proteins were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and visualized by western blot analysis with anti-SERT-Ab (a). The integrated density values were determined by densitometry (b). For cell surface expression, platelets from the subjects with hypertension and normotension (lanes 3 and 4, respectively) were biotinylated, and the labeled cell surface proteins were precipitated with streptavidin beads and then separated and visualized as above. The results of western blot analysis are shown above a summary of combined data from four densitometric scans. The (*) represents the results of a two-tailed Student’s t-test with p < 0.001 (compared with normotensive subjects).
In order to determine the relationship between the plasma 5HT levels and the platelet SERT in the hypertensive state, we characterized SERT in isolated platelets from normotensive subjects after a pretreatment with 5HT at various concentrations levels.
5HT uptake rates of SERT in isolated platelet pretreated with 5HT at different concentrations
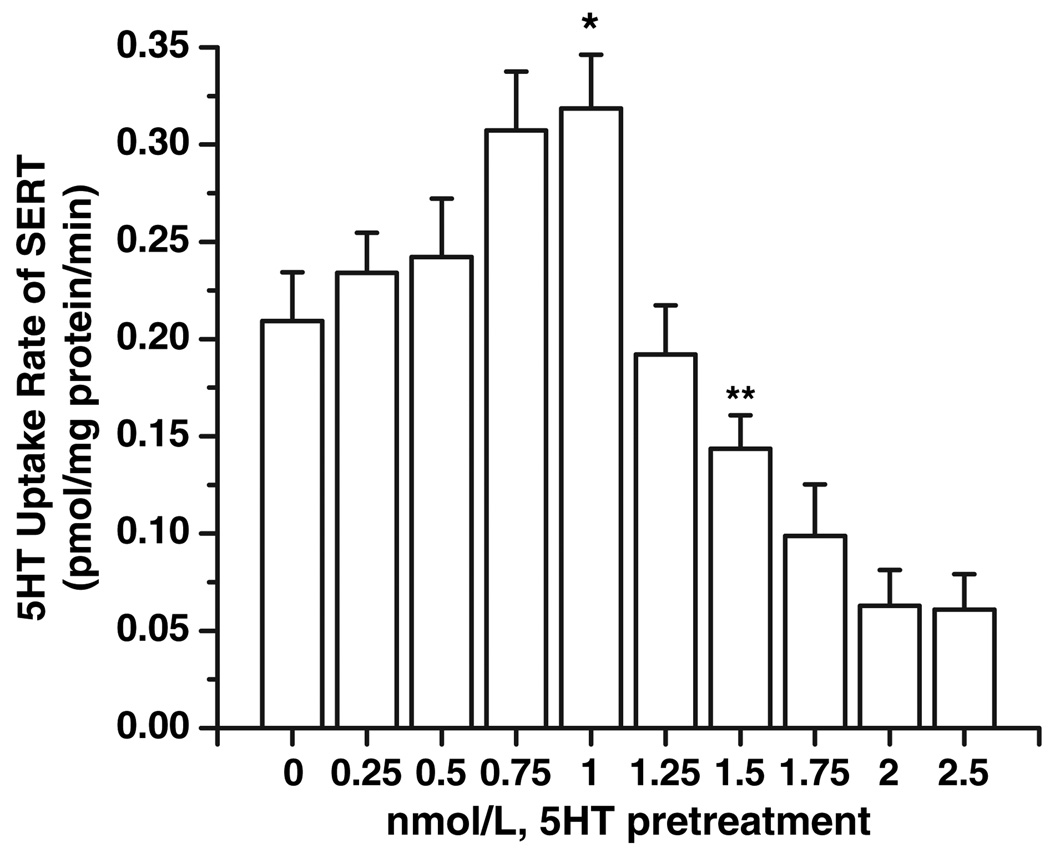
The uptake rates of SERT were measured in isolated platelets (1.5 × 108 ± 350) after 30 min pre-treatment with 5HT in a wide range of concentrations (0–2.5 nmol/L) as described under the Materials and methods section. A pretreatment with (0.75 nm ≤ 5HT ≤ 1 nmol/L) increased the 5HT uptake rate of platelet SERT system by 32%; however, pre-treatment with 1.5 nmol/L 5HT decreased the 5HT uptake rates of platelet SERT by 31% (Fig. 5). Background accumulation of 3H-5HT that occurred independent of SERT was measured in the same experiment by treating platelets with 0.1 µmol/L β-CIT. These findings are very similar to our observation on 5HT uptake rates of platelets from hypertensive and normotensive subjects. Therefore, we next investigated the density of transporters on the plasma membrane and in whole platelet.
Fig. 5.
Serotonin (5HT) uptake rates of single 5HT transporter (SERT) on isolated platelets pretreated with 5HT at different concentrations. Platelets prepared from 5 mL blood samples of three normotensive subjects were first pre-treated with 5HT over a wide concentration range (0–2.5 nmol/L) for 30 min. Then the [3H]-5HT uptake rates were measured in intact platelet. Rate of uptake is expressed as the means and SD values of triplicate determinations from three independent experiments. The (*) and (**) represent the results of a two-tailed Student’s t-test with both p < 0.001 (compared with untreated platelet uptake rates).
Impact of 5HT-pretreatment on SERT expression on the plasma membrane and in whole platelets
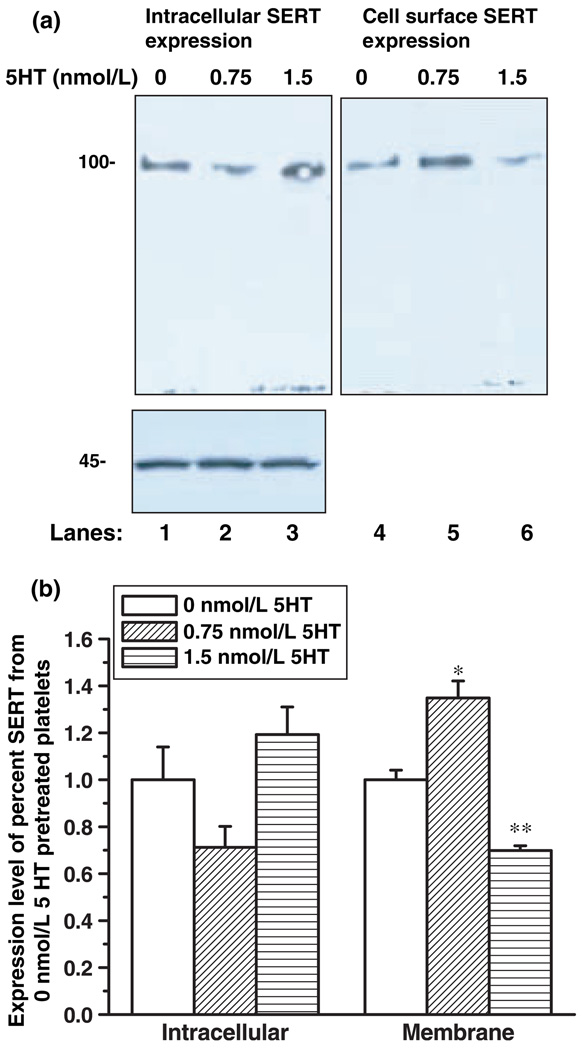
Platelets (1.5 × 108 ± 350) were pelleted, washed with PBSCM and treated with 0, 0.75 or 1.5 nmol/L 5HT for 30 min. Then, the biotinylation procedure was followed as described under the Method section. The intact platelets were incubated with membrane-impermeant NHS-SS-biotin first then lysed, and biotinylated membrane proteins were collected on streptavidin beads. The expressed transporters on the plasma membrane and in intracellular locations were analyzed by quantitative Western blot with anti-SERT antibody (Fig. 6a). The intracellular protein actin was used as a loading control. The expression of actin (inset of Fig. 6a) was not altered for all concentrations of 5HT pre-treatment, indicating that equal amounts of protein were loaded in each well.
Fig. 6.
Expression of single 5HT transporter (SERT) on isolated platelets with and without 5HT pre-treatment. Platelets prepared from 5 mL blood samples of 3 normotensive subjects were first pretreated with 0, 7.5, 1.5 nmol/L 5HT for 30 min. Then, they were biotinylated, and the labeled cell surface proteins were precipitated with streptavidin beads and then separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and visualized by western blot analysis using anti-SERT antibody followed by HRP-conjugated rabbit IgG (a). The integrated density values were determined by densitometry (b). The results of western blot analysis are shown in figure is the summary of combined data from three densitometric scans (Table 3). Actin was used as the loading control (inset in Figure 45 kD). The (* and **) represents the two-tailed Student’s t-test with p < 0.001 (compared with 0 nmol/L 5HT-matched samples).
5HT pretreatment (0.75 nmol/L; lane 5) increased SERT expression on the plasma membrane compared with the untreated (lane 4). In three independent experiments under steady-state experimental conditions, the streptavidin beads removed 59% of the biotinylated hSERT from the total transporters in platelets cells pretreated with 0.75 nmol/L 5HT. In cells pretreated with 1.5 nmol/L 5HT, however, the streptavidin beads removed only 35% of the biotinylated hSERT. In the untreated condition, 44% of the biotinylated hSERT was removed from the pool of biotinylated proteins. After removing the biotinylated membrane proteins from the lysed platelets (depleted lysate) with an excess of streptavidin beads, we determined the remaining amount of intracellular hSERT. Quantitative western blot analysis with SERT-antibody revealed that 1.5 nmol/L 5HT treatment maintained 65% of the total synthesized hSERT (Fig. 6a, lane 3) in the intracellular pools, and 35% of it appeared on the platelet membrane.
Following a pre-treatment with 1.5 nmol/L 5HT, the density of SERT on the platelet membrane decreased 30% when compared with non-treated controls. These findings explain why platelets pre-treated with 1.5 nmol/L 5HT exhibited a 32% decrease in 5HT uptake rates (Fig. 5).
Discussion
5HT was first isolated 55 years ago and identified as a vasoconstrictor compound in serum (Freyburger et al. 1952; Willerson 1995; Reis et al. 2000). Today the impact of 5HT on the cardiovascular system is well-established. Specifically, in hypertension and atherosclerosis (Golino et al. 1991; Yildiz et al. 1996), there is a very high sensitivity of blood vessels to the vasoconstrictive effects of 5HT, and these effects are amplified further by even subcontractile concentrations of endogenous hypertensive mediators (Watts 2005). Thus, 5HT, which markedly induces vasoconstriction, also enhances the hypertensive effects of known vasoconstrictors.
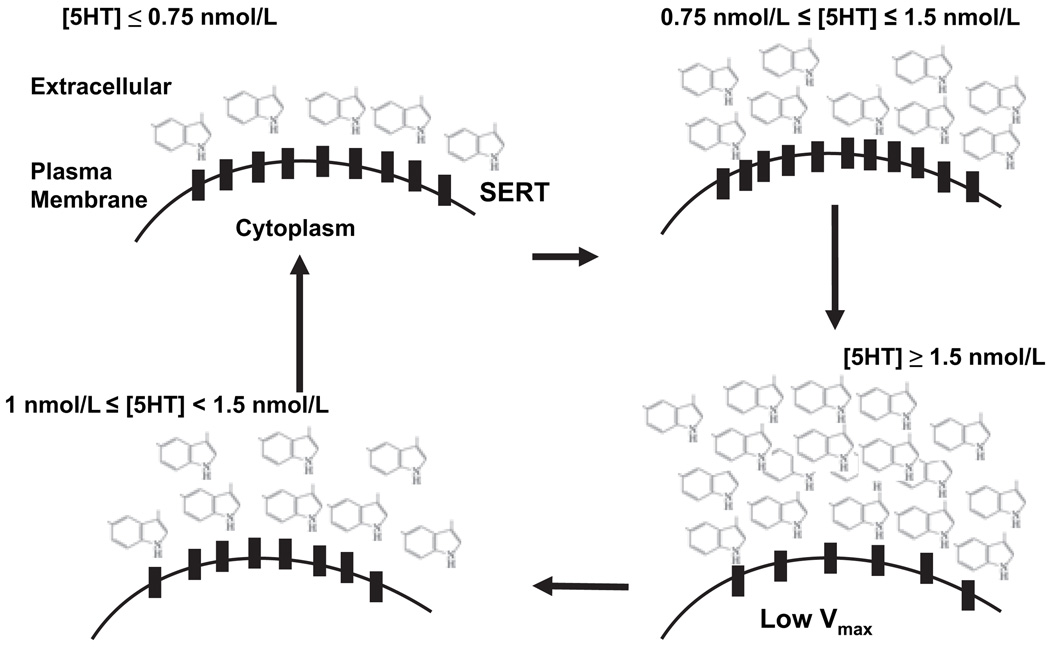
Altered 5HT plasma level is one of the multiple mechanisms associated with certain types of hypertension (Woittiez et al. 1986; Dabire et al. 1987; Egan et al. 1988; Wocial et al. 1990; Gujrati et al. 1994; Azzadin et al. 1995; Yildiz et al. 1998; Brass 1999; Reis et al. 2000; Watts 2005). Plasma 5HT plasma levels are regulated by SERT in the platelet membranes (Pettinger et al. 1973; Shulman et al. 1989; Lichtman et al. 2002; Blann et al. 2003; Doggrell 2003); Stoddard et al. 2003; Pidgeon et al. 2004; Ni et al., 2006). Therefore, the uptake efficiency of platelet SERT is one of the major factors in controlling blood pressure levels by regulating plasma 5HT concentration. In the present study, we first demonstrated that plasma 5HT levels of hypertensive subjects are increased while the amounts of 5HT in their platelets are decreased. These findings are consistent with previous reports (Jafri et al., 1992; Watts 2005). Then, we compared the biochemical characteristics of platelet SERT from hypertensive with the normotensive subjects to determine whether platelet SERT undergoes hypertension-associated alterations. We performed our studies in two model systems (i) the platelets prepared from hypertensive and normotensive subjects; and (ii) the platelets isolated from normotensive subjects and pretreated with various concentrations of 5HT. The main conclusions from our findings are that the 5HT uptake rate of SERT is lower in platelets from hypertensive subjects than those from normotensive subjects due to the decrease in Vmax and the decreased number of SERT molecules on the platelet membrane. Several independent lines of evidence support these conclusions. First, we differentiated the expression levels of transporter protein on the platelet membrane and in whole cells by biotinylation of membrane proteins. The density of SERT on the plasma membrane of platelets from hypertensive subjects was less than that of the normotensive subjects. Second, the decreased Vmax of SERT during the hypertensive state could be due to the decreased surface expression of SERT. Third, we confirmed these findings in an in vitro model system, isolated platelets pretreated with 5HT at various concentrations (Fig. 7). Our data demonstrated that exogenous 5HT at lower concentrations (0.75 nmol/L ≤ [5HT] ≤ 1.5 nmol/L) increases the uptake rates and surface density of platelet SERT by 32% and 35%, respectively. However, at higher concentrations ([5HT] ≥ 1.5 nmol/L) 5HT decreases the uptake rates and surface density of platelet SERT by 31% and 30%, respectively (Table 3). These findings nicely agreed with the data obtained from the platelets of hypertensive subjects when the plasma 5HT levels were around 1 nmol/L, the 5HT uptake rates and the surface density of platelet SERT decreases 45% and 35%, respectively. The overall data show that in serum during the formation of hypertension plasma 5HT levels are important and have more impact than the one we obtained with the in vitro system. This is a reasonably expected outcome.
Fig. 7.
Model for Serotonin (5HT)-dependent plasma membrane density of platelet single 5HT transporter (SERT). In the presence of high (>1 nmol/L) plasma 5HT levels, the density and the 5HT uptake rates of platelet SERT decrease in platelets samples of hypertensive subjects and 5HT-pre-treated platelets obtained from normotensive subjects. Therefore, we hypothesize that the low 5HT uptake capacity of platelet SERT results from a feedback effect of increased plasma 5HT levels. Individuals with hypertension might have an impaired 5HT uptake mechanism, which abates as the hypertensive state improves.
Table 3.
Effect of 5HT pretreatment on platelet single 5HT transporter (SERT)
| % Change of SERT in 5HT pretreated platelets compare to the untreated ones |
5HT pre-treatment | |
|---|---|---|
| 0.75 nmol/L | 1.5 nmol/L | |
| 5HT uptake rate | 32% increase | 31% decrease |
| Surface expression | 35%* increase | 30%** decrease |
| Intracellular expression | 29% decrease | 20% increase |
p = 0.001;
p = 0.003.
The regulatory roles of substrates and inhibitors on their transporters are well established (Wuller et al. 2004). Glutamate increases the surface expression of EAAT1 (Duan et al. 1999) and GABA increases GAT1 activity, through an increase in cell surface expression, which occurs within minutes (Bernstein and Quick 1999; Quick 2003). Dopamine transporter function and plasma membrane expression are regulated by dopamine, amphetamine, and cocaine (Kahlig and Galli 2003). Protein kinase C activators decrease the numbers of SERT molecules; 5HT pretreatment altered the density of SERT on the plasma membrane by interfering with SERT phosphorylation and internalization (Ramamoorthy and Blakely 1999). Cyclic adenosine monophosphate activators, however, increase SERT density in cell membranes (Ramamoorthy et al. 1993a). These studies confirm that the functional efficiency of SERT depends on the density of functionally active transporters on the plasma membrane (Ramamoorthy et al. 1993a; Ramamoorthy and Blakely 1999; Koldzic-Zivanovic et al. 2004). Therefore, we then examined whether plasma 5HT levels play a role in regulating the density of SERT molecules on the plasma membrane. Our findings indicate that at lower concentrations (≤1 nmol/L) 5HT-pre-treatment promotes the plasma membrane expression of SERT which subsequently increases the rate of 5HT uptake (Table 3). Pre-treatment with 1.5 nmol/L 5HT decreased the 5HT uptake rate and the density of SERT on the plasma membrane. On the basis of these findings, we hypothesize that during hypertension, the 5HT uptake rate of the platelet SERT system decreases due to the number of available SERT molecules on the platelet membrane; therefore, the average rate of transport per cell surface SERT molecule is decreased (Fig. 7). Our findings, together with those of others, suggest that plasma 5HT regulates its own concentration via altering the density of SERT molecules on the platelet membrane. Elucidating the molecular basis of communication between 5HT and the SERT protein will lead to the identification of the intracellular activities of platelets in hypertension.
During hypertension, plasma 5HT levels are higher than during normotension. Our current findings indicate that the 5HT uptake rates of SERT undergo hypertension-associated alterations. This is an important finding establishing that SERT on the platelet membrane can be a major regulator of plasma 5HT levels. More specifically, we demonstrated that the 5HT uptake rate of SERT decreases due to a decrease in Vmax and the amount of SERT molecules on the platelet membrane. These findings support the idea that SERT is an attractive candidate for studies of the control of hypertension via regulating the plasma 5HT levels. On the basis of these findings, without further investigation, it would be speculative to relate the plasma 5HT levels directly with the uptake capacity of platelet SERT.
Acknowledgement
We would like to thank Drs L. DeFelice (Vanderbilt University, Nashville, TN, USA) and J. Ware (UAMS, Little Rock, AR, USA) for their critical review of this manuscript. Our sincere thanks to Dr Robin Bohra for his participation, helpful advice and discussion related to this work. This work was supported by grants from the National Alliance for Research on Schizophrenia and Depression and the American Heart Association (F.K.).
Abbreviations used
- 5HT
Serotonin
- hSERT-Ab
human SERT antibody
- NEM
N-ethylmaleimide
- PIM
protease inhibitor mixture
- PPP
platelet-poor plasma
- SERT
single 5HT transporter
- β-CIT
2β-carbomethoxy-3-tropane
References
- Arora RC, Meltzer HY. 3H-imipramine binding in the blood platelets of normal twins. Psychiatry Res. 1990;32:265–273. doi: 10.1016/0165-1781(90)90031-y. [DOI] [PubMed] [Google Scholar]
- Azzadin A, Mysliwiec J, Wollny T, Mysliwiec M, Buczko W. Serotonin is involved in the pathogenesis of hypertension developing during erythropoietin treatment in uremic rats. Thromb. Res. 1995;77:217–224. doi: 10.1016/0049-3848(95)91609-o. [DOI] [PubMed] [Google Scholar]
- Bengel D, Murphy DL, Andrews AM, Wichems CH, Feltner D, Heils A, Mossner R, Westphal H, Lesch K. Altered brain serotonin homeostasis and locomotor insensitivity to 3,4-methylenedioxymethamphetamine (“Ecstasy”) in serotonin transporter-deficient mice. Mol. Pharmacol. 1998;53:649–655. doi: 10.1124/mol.53.4.649. [DOI] [PubMed] [Google Scholar]
- Bernstein EM, Quick MW. Regulation of gamma-aminobutyric acid (GABA) transporters by exogenous GABA. J. Biol. Chem. 1999;274:889–895. doi: 10.1074/jbc.274.2.889. [DOI] [PubMed] [Google Scholar]
- Blakely RD, Berson HE, Fremeau RT, Jr, Caron MC, Peek MM, Priace HK, Bradley CC. Cloning and expression of a functional the 5HT transporter from rat brain. Nature. 1991;354:66–70. doi: 10.1038/354066a0. [DOI] [PubMed] [Google Scholar]
- Blann AD, Nadar S, Lip GY. Pharmacological modulation of platelet function in hypertension. Hypertension. 2003;42:1–7. doi: 10.1161/01.HYP.0000077901.84467.E1. [DOI] [PubMed] [Google Scholar]
- Brass LF. More pieces of the platelet activation puzzle slide into place. J. Clin. Invest. 1999;104:1663–1665. doi: 10.1172/JCI8944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corash L, Costa JL, Shafer B, Donlon JA, Murphy DL. Heterogeneity of human whole blood platelet subpopulations. III. Density-dependent differences in subcellular constituents. Blood. 1984;64:185–193. [PubMed] [Google Scholar]
- Dabire H, Cherqui C, Fournier B, Schmitt H. Comparison of effects of some 5-HT1 agonists on blood pressure and heart rate of normotensive anaesthetized rats. Eur. J. Pharmacol. 1987;140:259–266. doi: 10.1016/0014-2999(87)90282-2. [DOI] [PubMed] [Google Scholar]
- Doggrell SA. Pharmacotherapy of intermittent claudication. Expert Opin. Pharmacother. 2001;2:1725–1736. doi: 10.1517/14656566.2.11.1725. [DOI] [PubMed] [Google Scholar]
- Duan S, Anderson CM, Stein BA, Swanson RA. Glutamate induces rapid upregulation of astrocyte glutamate transport and cell-surface expression of GLAST. J. Neurosci. 1999;19:10193–10200. doi: 10.1523/JNEUROSCI.19-23-10193.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egan B, Conlon ME, Campbell R, Schork N, Zwiefler A, Julius S. Effects of ketanserin on blood pressure and platelet aggregation in elderly men with mild hypertension. Am. J. Hypertens. 1988;1:324S–330S. doi: 10.1093/ajh/1.3.324s. [DOI] [PubMed] [Google Scholar]
- Freyburger WA, Graham BE, Rapport MM, Seay PH, Govier WM, Swoap OF, Vander Brook MJ. The pharmacology of 5-hydroxytryptamine (serotonin) J. Pharmacol. Exp. Ther. 1952;105:80–86. [PubMed] [Google Scholar]
- Golino P, Piscione F, Willerson JT, et al. Divergent effects of serotonin on coronary-artery dimensions and blood flow in patients with coronary atherosclerosis and control patients. New Engl. J. Med. 1991;324:641–648. doi: 10.1056/NEJM199103073241001. [DOI] [PubMed] [Google Scholar]
- Gujrati VR, Goyal A, Gaur SP, Singh N, Shanker K, Chandravati Relevance of platelet serotonergic mechanisms in pregnancy induced hypertension. Life Sci. 1994;55:327–335. doi: 10.1016/0024-3205(94)00735-7. [DOI] [PubMed] [Google Scholar]
- Heils A, Teufel A, Petri S, Stober G, Riederer P, Bengel D, Lesch KP. Allelic variation of human serotonin transporter gene expression. J. Neurochem. 1996;66:2621–2624. doi: 10.1046/j.1471-4159.1996.66062621.x. [DOI] [PubMed] [Google Scholar]
- Hoffman BJ, Mezey E, Browstein MJ. Cloning of the 5HT transporter affected by antidepressants. Science. 1991;254:579–580. doi: 10.1126/science.1948036. [DOI] [PubMed] [Google Scholar]
- Jafri SM, Chandra N, Dhawan S, Soni D, Chandra M, Shanker K. Factors influencing platelet serotonin uptake in essential hypertension. Int. J. Cardiol. 1992;34:327–333. doi: 10.1016/0167-5273(92)90031-w. [DOI] [PubMed] [Google Scholar]
- Johnson GJ, Leis LA, Dunlop PC, Weir EK. The effect of the anorectic agent, d-fenfluramine, and its primary metabolite, d-norfenfluramine, on intact human platelet serotonin uptake and efflux. J. Thromb. Haemost. 2003;1:2663–2668. doi: 10.1046/j.1538-7836.2003.00474.x. [DOI] [PubMed] [Google Scholar]
- Kahlig KM, Galli A. Regulation of dopamine transporter function and plasma membrane expression by dopamine, amphetamine, and cocaine. Eur. J. Pharmacol. 2003;479:153–158. doi: 10.1016/j.ejphar.2003.08.065. [DOI] [PubMed] [Google Scholar]
- Kilic F, Murphy D, Rudnick G. A Human 5HT Transporter Mutation Causes Constitutive Activation of Transport Activity. (A Human 5HT Transporter Mutation Causes Constitutive Activation of Transport Activity. Mol. Pharmacol. 2003;64:4–12. doi: 10.1124/mol.64.2.440. [DOI] [PubMed] [Google Scholar]
- Kilic F, Rudnick G. Oligomerization of 5HT transporter and its functional consequences. Proc. Natl. Acad. Sci. USA. 2000;97:3106–3111. doi: 10.1073/pnas.060408997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kocabas AM, Rudnick G, Kilic F. Functional consequences of Homo- But Not Hetero-Oligomerization between transporters for the biogenic amine neurotransmitters. J. Neurochem. 2003;85:1513–1520. doi: 10.1046/j.1471-4159.2003.01793.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koldzic-Zivanovic N, Seitz PK, Watson CS, Cunningham KA, Thomas ML. Intracellular signaling involved in estrogen regulation of serotonin reuptake. Mol. Cell Endocrinol. 2004;226:33–42. doi: 10.1016/j.mce.2004.07.017. [DOI] [PubMed] [Google Scholar]
- Lesch KP, Bengel D, Heils A, Sabol SZ, Greenberg BD, Petri S, Benjamin J, Muller CR, Hamer DH, Murphy DL. Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science. 1996;29:1527–1531. doi: 10.1126/science.274.5292.1527. [DOI] [PubMed] [Google Scholar]
- Lesch KP, Wolozin BL, Murphy DL, Riderer P. Primary structure of the human platelet 5HT uptake site: identity with the brain 5HT transporter. J. Neurochem. 1993;60:2319–2322. doi: 10.1111/j.1471-4159.1993.tb03522.x. [DOI] [PubMed] [Google Scholar]
- Lichtman JH, Krumholz HM, Wang Y, Radford MJ, Brass LM. Risk and predictors of stroke after myocardial infarction among the elderly: results from the Cooperative Cardiovascular Project. Circulation. 2002;5:1082–1087. doi: 10.1161/hc0902.104708. [DOI] [PubMed] [Google Scholar]
- Ni W, Watts SW. 5-hydroxytryptamine in the cardiovascular system: focus on the serotonin transporter (SERT) Clin. Exp. Pharmacol. Physiol. 2006;3:575–583. doi: 10.1111/j.1440-1681.2006.04410.x. [DOI] [PubMed] [Google Scholar]
- Ozaki N, Rosenthal NE, Mazzola P, Chiueh CC, Hardin T, Garcia-Borreguero D, Schwartz PJ, Turner E, Oren DA, Murphy DL. Platelet [3H]paroxetine binding, 5-HT-stimulated Ca2+ response, and 5-HT content in winter seasonal affective disorder. Biol. Psychiatry. 1994;36:458–466. doi: 10.1016/0006-3223(94)90641-6. [DOI] [PubMed] [Google Scholar]
- Ozaslan D, Wang S, Ahmed B, Bene A, Kocabas AM, Kilic F. Glycosyl Modification Facilitates Homo- and Hetero-Oligomerization of Serotonin Transporter. A Specific Role for the Sialic Acid Residues. J. Biol. Chem. 2003;278:43991–44000. doi: 10.1074/jbc.M306360200. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Pettinger W, Sheppard H, Palkoski Z, Renyi E. Angiotensin antagonism and antihypertensive activity of phosphodiesterase inhibiting agents. Life Sci. I. 1973;15:49–62. doi: 10.1016/0024-3205(73)90061-1. [DOI] [PubMed] [Google Scholar]
- Pidgeon GP, Tamosiuniene R, Chen G, Leonard I, Belton O, Bradford A, Fitzgerald DJ. Intravascular thrombosis after hypoxia-induced pulmonary hypertension: regulation by cyclooxygenase-2. Circulation. 2004;26:2701–2707. doi: 10.1161/01.CIR.0000145613.01188.0B. [DOI] [PubMed] [Google Scholar]
- Quick MW. Regulating the conducting states of a mammalian serotonin transporter. Neuron. 2003;40:537–549. doi: 10.1016/s0896-6273(03)00605-6. [DOI] [PubMed] [Google Scholar]
- Ramamoorthy S, Cool DR, Mahesh VB, Leibach FH, Melikian HE, Blakely RD, Ganapathy V. Regulation of the human serotonin transporter. Cholera toxin-induced stimulation of serotonin uptake in human placental choriocarcinoma cells is accompanied by increased serotonin transporter mRNA levels and serotonin transporter-specific ligand binding. J. Biol. Chem. 1993a;268:21626–21631. [PubMed] [Google Scholar]
- Ramamoorthy S, Baumen AL, Moore KR, Han H, Yang-Fen T, Chang AS, Ganapathy V, Blakely RD. Antidepressant- and cocaine-sensitive human the 5HT transporter: molecular cloning, expression, and chromosomal localization. Proc. Natl. Acad. Sci. USA. 1993b;90:2542–2546. doi: 10.1073/pnas.90.6.2542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramamoorthy S, Blakely RD. Phosphorylation and sequestration of serotonin transporters differentially modulated by psychostimulants. Science. 1999;285:763–766. doi: 10.1126/science.285.5428.763. [DOI] [PubMed] [Google Scholar]
- Reis F, Tavares P, Rito LC, Teixeira HM, Santos Dias JD, Ferrer-Antunes C, Mesquita JF, Teixeira F. Platelet activation is increased in cyclosporin A-induced hypertensive rats. J. Cardiovasc. Pharmacol. 2000;36:56–64. doi: 10.1097/00005344-200007000-00008. [DOI] [PubMed] [Google Scholar]
- Serebruany VL, Gurbel PA, O’Connor CM. Platelet inhibition by sertraline and N-desmethylsertraline: a possible missing link between depression, coronary events, and mortality benefits of selective serotonin reuptake inhibitors. Pharmacol. Res. 2001;43:453–461. doi: 10.1006/phrs.2001.0817. [DOI] [PubMed] [Google Scholar]
- Shulman NB, Ford CE, Hall WD, Blaufox MD, Simon D, Langford HG, Schneider KA. Prognostic value of serum creatinine and effect of treatment of hypertension on renal function. Results from the hypertension detection and follow-up program. The Hypertension Detection and Follow-up Program Cooperative Group. Hypertension. 1989:180–193. doi: 10.1161/01.hyp.13.5_suppl.i80. [DOI] [PubMed] [Google Scholar]
- Stoddard MF, Singh P, Dawn B, Longaker RA. Left atrial thrombus predicts transient ischemic attack in patients with atrial fibrillation. Am. Heart J. 2003;145:676–682. doi: 10.1067/mhj.2003.91. [DOI] [PubMed] [Google Scholar]
- Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA. 1979;76:4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yildiz O, Cicek S, Ay I, Demirkilic U, Tuncer M. Hypertension increases the contractions to sumatriptan in the human internal mammary artery. Ann. Thorac. Surg. 1996;62:1392–1396. doi: 10.1016/0003-4975(96)00674-1. [DOI] [PubMed] [Google Scholar]
- Yildiz O, Smith JR, Purdy RE. Serotonin and vasoconstrictor synergism. Life Sci. 1998;62:1723–1732. doi: 10.1016/s0024-3205(97)01166-1. [DOI] [PubMed] [Google Scholar]
- Watts S. 5-HT in systemic hypertension. Clin. Sci. 2005;108:399–412. doi: 10.1042/CS20040364. [DOI] [PubMed] [Google Scholar]
- Willerson JT. Conversion from chronic to acute coronary heart disease syndromes. Role of platelets and platelet products. Tex Heart Inst. J. 1995;22:13–19. [PMC free article] [PubMed] [Google Scholar]
- Woittiez AJ, Wenting GJ, van den Meiracker AH, Ritsema van Eck HJ, Man in’t Veld AJ, Zantvoort FA, Schalekamp MA. Chronic effect of ketanserin in mild to moderate essential hypertension. Hypertension. 1986;8:167–173. doi: 10.1161/01.hyp.8.2.167. [DOI] [PubMed] [Google Scholar]
- Wocial B, Wasowska-Ciszek T, Lapinski M, Januszewicz A, Grzesiuk W, Stepniakowski K, Januszewicz W. Free serotonin level in the blood of patients with borderline and essential hypertension. Kardiol. Pol. 1990;33:4–7. [PubMed] [Google Scholar]
- Wuller S, Wiesner B, Loffler A, Furkert J, Krause G, Hermosilla R, Schaefer M, Schulein R, Rosenthal W, Oksche A. Pharmacochaperones post-translationally enhance cell surface expression by increasing conformational stability of wild-type and mutant vasopressin V2 receptors. J. Biol. Chem. 2004;279:47254–47263. doi: 10.1074/jbc.M408154200. [DOI] [PubMed] [Google Scholar]