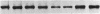Abstract
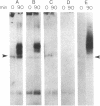
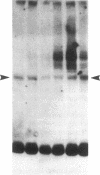
Phytochrome is the photoreceptor that controls red light-mediated morphogenesis in higher plants. It exists in two photointerconvertible forms, a red light-absorbing form, Pr, and a far-red light-absorbing form, Pfr. Because photoconversion of Pr to Pfr by a brief light pulse decreases the in vivo half-life of this chromoprotein by a factor of ≈100, this system offers a unique way to modulate the turnover rate of a specific protein and hence study the mechanisms responsible for selective protein degradation. In etiolated oat [Avena sativa (L.)] seedlings, degradation of phytochrome as Pfr follows zero-order kinetics as measured both spectrally and by ELISA, with 50% of Pfr lost in ≈130 min at 27°C. Immunoblot analysis of the destruction process with anti-oat phytochrome immunoglobulins reveals that degradation involves the loss of the 124-kDa phytochrome monomer and that proteolytic intermediates of apparent molecular mass lower than 124 kDa do not accumulate to detectable levels in vivo (<0.015% of total phytochrome). The latter observation suggests that proteolytic breakdown of the protein is extremely rapid. However, a series of polypeptides with higher apparent molecular mass and recognized by anti-phytochrome immunoglobulins (principally 129 and 134 kDa) appears after photoconversion to Pfr. These polypeptides represent no more than a few percent of the total immunologically detectable phytochrome pool and have incremental differences in apparent molecular mass of 5 kDa. They appear within 5 min after Pfr formation, reach maximal levels between 90 and 180 min, and decline thereafter. These polypeptides and others of apparent molecular mass up to 160 kDa are also detectable with immunoglobulins directed against either oat or human ubiquitin, indicating that they are ubiquitin-phytochrome conjugates. Since ubiquitin conjugation is involved in intracellular protein turnover and since formation and degradation of Pfr-ubiquitin conjugates coincide with the turnover of Pfr, these data suggest that the Pfr form of phytochrome is degraded via a ubiquitin-dependent proteolytic pathway.
Keywords: protein degradation, regulatory photoreceptor, posttranslational modification
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Butler W. L., Lane H. C., Siegelman H. W. Nonphotochemical Transformations of Phytochrome in Vivo. Plant Physiol. 1963 Sep;38(5):514–519. doi: 10.1104/pp.38.5.514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciechanover A., Finley D., Varshavsky A. Ubiquitin dependence of selective protein degradation demonstrated in the mammalian cell cycle mutant ts85. Cell. 1984 May;37(1):57–66. doi: 10.1016/0092-8674(84)90300-3. [DOI] [PubMed] [Google Scholar]
- Colbert J. T., Hershey H. P., Quail P. H. Autoregulatory control of translatable phytochrome mRNA levels. Proc Natl Acad Sci U S A. 1983 Apr;80(8):2248–2252. doi: 10.1073/pnas.80.8.2248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniels S. M., Quail P. H. Monoclonal antibodies to three separate domains on 124 kilodalton phytochrome from Avena. Plant Physiol. 1984 Nov;76(3):622–626. doi: 10.1104/pp.76.3.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg A. L., Dice J. F. Intracellular protein degradation in mammalian and bacterial cells. Annu Rev Biochem. 1974;43(0):835–869. doi: 10.1146/annurev.bi.43.070174.004155. [DOI] [PubMed] [Google Scholar]
- Haas A. L., Bright P. M. The immunochemical detection and quantitation of intracellular ubiquitin-protein conjugates. J Biol Chem. 1985 Oct 15;260(23):12464–12473. [PubMed] [Google Scholar]
- Hershey H. P., Barker R. F., Idler K. B., Lissemore J. L., Quail P. H. Analysis of cloned cDNA and genomic sequences for phytochrome: complete amino acid sequences for two gene products expressed in etiolated Avena. Nucleic Acids Res. 1985 Dec 9;13(23):8543–8559. doi: 10.1093/nar/13.23.8543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Ciechanover A., Heller H., Haas A. L., Rose I. A. Proposed role of ATP in protein breakdown: conjugation of protein with multiple chains of the polypeptide of ATP-dependent proteolysis. Proc Natl Acad Sci U S A. 1980 Apr;77(4):1783–1786. doi: 10.1073/pnas.77.4.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Ciechanover A. Mechanisms of intracellular protein breakdown. Annu Rev Biochem. 1982;51:335–364. doi: 10.1146/annurev.bi.51.070182.002003. [DOI] [PubMed] [Google Scholar]
- Hershko A., Eytan E., Ciechanover A., Haas A. L. Immunochemical analysis of the turnover of ubiquitin-protein conjugates in intact cells. Relationship to the breakdown of abnormal proteins. J Biol Chem. 1982 Dec 10;257(23):13964–13970. [PubMed] [Google Scholar]
- Hershko A., Heller H., Eytan E., Kaklij G., Rose I. A. Role of the alpha-amino group of protein in ubiquitin-mediated protein breakdown. Proc Natl Acad Sci U S A. 1984 Nov;81(22):7021–7025. doi: 10.1073/pnas.81.22.7021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Heller H. Occurrence of a polyubiquitin structure in ubiquitin-protein conjugates. Biochem Biophys Res Commun. 1985 May 16;128(3):1079–1086. doi: 10.1016/0006-291x(85)91050-2. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Levinger L., Varshavsky A. Selective arrangement of ubiquitinated and D1 protein-containing nucleosomes within the Drosophila genome. Cell. 1982 Feb;28(2):375–385. doi: 10.1016/0092-8674(82)90355-5. [DOI] [PubMed] [Google Scholar]
- Litts J. C., Kelly J. M., Lagarias J. C. Structure-function studies on phytochrome. Preliminary characterization of highly purified phytochrome from Avena sativa enriched in the 124-kilodalton species. J Biol Chem. 1983 Sep 25;258(18):11025–11031. [PubMed] [Google Scholar]
- Polson A., von Wechmar M. B., van Regenmortel M. H. Isolation of viral IgY antibodies from yolks of immunized hens. Immunol Commun. 1980;9(5):475–493. doi: 10.3109/08820138009066010. [DOI] [PubMed] [Google Scholar]
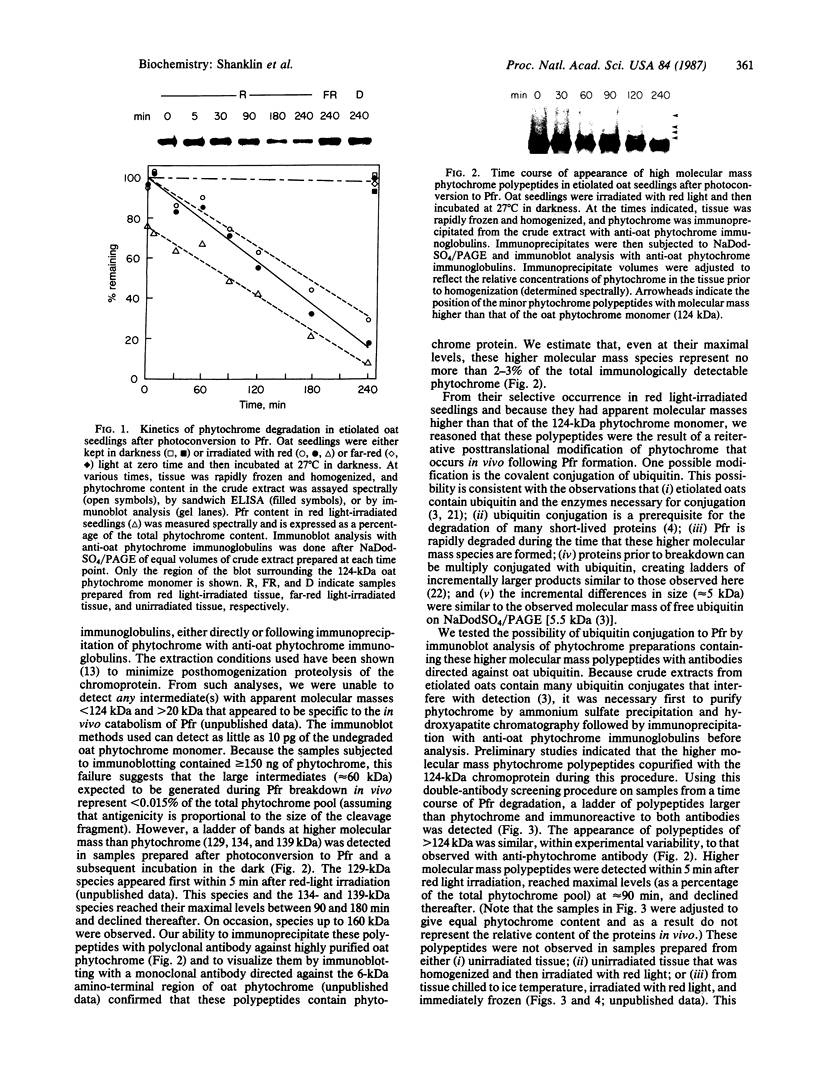
- Quail P. H., Schäfer E., Marmé D. Turnover of phytochrome in pumpkin cotyledons. Plant Physiol. 1973 Aug;52(2):128–131. doi: 10.1104/pp.52.2.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schäfer E., Lassig T. U., Schopfer P. Photocontrol of phytochrome destruction in grass seedlings. The influence of wavelength and irradiance. Photochem Photobiol. 1975 Nov;22(5):193–202. doi: 10.1111/j.1751-1097.1975.tb06736.x. [DOI] [PubMed] [Google Scholar]
- Siegelman M., Bond M. W., Gallatin W. M., St John T., Smith H. T., Fried V. A., Weissman I. L. Cell surface molecule associated with lymphocyte homing is a ubiquitinated branched-chain glycoprotein. Science. 1986 Feb 21;231(4740):823–829. doi: 10.1126/science.3003913. [DOI] [PubMed] [Google Scholar]
- Vierstra R. D., Langan S. M., Haas A. L. Purification and initial characterization of ubiquitin from the higher plant, Avena sativa. J Biol Chem. 1985 Oct 5;260(22):12015–12021. [PubMed] [Google Scholar]
- Vierstra R. D., Quail P. H. Photochemistry of 124 kilodalton Avena phytochrome in vitro. Plant Physiol. 1983 May;72(1):264–267. doi: 10.1104/pp.72.1.264. [DOI] [PMC free article] [PubMed] [Google Scholar]