Abstract
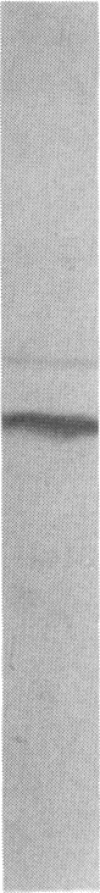
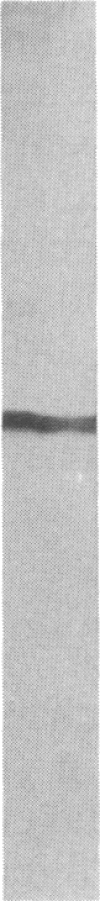
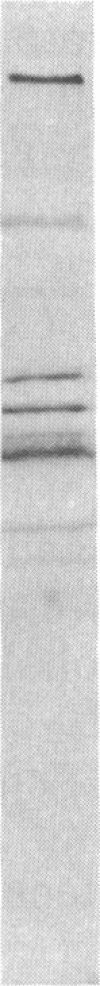
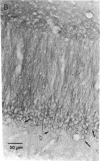
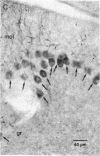
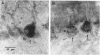
High-affinity antibodies against calmodulin (CaM)-dependent cyclic nucleotide phosphodiesterase and protein phosphatase (calcineurin) were purified and characterized. Rabbit anti-phosphodiesterase antibody did not react with other phosphodiesterases or with the regulatory subunits of cAMP-dependent protein kinase. Affinity-purified goat anti-calcineurin antibody recognized both the 61-kDa catalytic subunit and the 18-kDa Ca2+-binding subunit of the phosphatase. Neither antibody reacted with CaM, several CaM-binding proteins (calmodulin-dependent protein kinase, myosin light chain kinase, fodrin), or other cytosolic proteins from brain. The antibodies were used to compare the cellular localization of these two CaM-dependent enzymes in rat brain. Both calcineurin and phosphodiesterase were found predominantly in nerve cells; however, phosphodiesterase was restricted to very specific neuronal populations. Phosphodiesterase was prominent in the somatic cytoplasm and dendrites of regional output neurons--e.g., cerebellar Purkinje cells and hippocampal and cortical pyramidal cells. The extensive and uniform staining in the dendrites was consistent with postsynaptic localization and suggested an important function for this enzyme in neurons that integrate multiple convergent inputs. Calcineurin was present in virtually all classes of neurons, with immunoreactivity confined primarily to cell bodies. Both diffuse cytoplasmic staining and characteristic punctate staining of cell bodies were observed; the latter suggested compartmentalization of calcineurin at or near the plasma membrane. The results of this study demonstrate that calcineurin and phosphodiesterase are differentially localized in the central nervous system. Thus, the expression and compartmentalization of CaM-binding proteins may be highly regulated and specific for particular differentiated nerve cell types.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baehr W., Devlin M. J., Applebury M. L. Isolation and characterization of cGMP phosphodiesterase from bovine rod outer segments. J Biol Chem. 1979 Nov 25;254(22):11669–11677. [PubMed] [Google Scholar]
- Berridge M. J. The interaction of cyclic nucleotides and calcium in the control of cellular activity. Adv Cyclic Nucleotide Res. 1975;6:1–98. [PubMed] [Google Scholar]
- Billingsley M. L., Pennypacker K. R., Hoover C. G., Brigati D. J., Kincaid R. L. A rapid and sensitive method for detection and quantification of calcineurin and calmodulin-binding proteins using biotinylated calmodulin. Proc Natl Acad Sci U S A. 1985 Nov;82(22):7585–7589. doi: 10.1073/pnas.82.22.7585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brostrom C. O., Huang Y. C., Breckenridge B. M., Wolff D. J. Identification of a calcium-binding protein as a calcium-dependent regulator of brain adenylate cyclase. Proc Natl Acad Sci U S A. 1975 Jan;72(1):64–68. doi: 10.1073/pnas.72.1.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caceres A., Bender P., Snavely L., Rebhun L. I., Steward O. Distribution and subcellular localization of calmodulin in adult and developing brain tissue. Neuroscience. 1983 Oct;10(2):449–461. doi: 10.1016/0306-4522(83)90145-8. [DOI] [PubMed] [Google Scholar]
- Carlin R. K., Grab D. J., Siekevitz P. Function of a calmodulin in postsynaptic densities. III. Calmodulin-binding proteins of the postsynaptic density. J Cell Biol. 1981 Jun;89(3):449–455. doi: 10.1083/jcb.89.3.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen P. The role of protein phosphorylation in neural and hormonal control of cellular activity. Nature. 1982 Apr 15;296(5858):613–620. doi: 10.1038/296613a0. [DOI] [PubMed] [Google Scholar]
- Cumming R., Krigman M. R., Steiner A. L. Post-natal ontogenesis of calmodulin and cyclic AMP-dependent protein kinase subunits in the Purkinje cell using immunofluorescence. Neurosci Lett. 1982 Mar 5;28(3):247–252. doi: 10.1016/0304-3940(82)90065-9. [DOI] [PubMed] [Google Scholar]
- De Camilli P., Cameron R., Greengard P. Synapsin I (protein I), a nerve terminal-specific phosphoprotein. I. Its general distribution in synapses of the central and peripheral nervous system demonstrated by immunofluorescence in frozen and plastic sections. J Cell Biol. 1983 May;96(5):1337–1354. doi: 10.1083/jcb.96.5.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Camilli P., Miller P. E., Levitt P., Walter U., Greengard P. Anatomy of cerebellar Purkinje cells in the rat determined by a specific immunohistochemical marker. Neuroscience. 1984 Apr;11(4):761–817. doi: 10.1016/0306-4522(84)90193-3. [DOI] [PubMed] [Google Scholar]
- DeLorenzo R. J. Role of calmodulin in neurotransmitter release and synaptic function. Ann N Y Acad Sci. 1980;356:92–109. doi: 10.1111/j.1749-6632.1980.tb29603.x. [DOI] [PubMed] [Google Scholar]
- Detre J. A., Nairn A. C., Aswad D. W., Greengard P. Localization in mammalian brain of G-substrate, a specific substrate for guanosine 3',5'-cyclic monophosphate-dependent protein kinase. J Neurosci. 1984 Nov;4(11):2843–2849. doi: 10.1523/JNEUROSCI.04-11-02843.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eccles J. C. An instruction-selection theory of learning in the cerebellar cortex. Brain Res. 1977 May 27;127(2):327–352. doi: 10.1016/0006-8993(77)90550-9. [DOI] [PubMed] [Google Scholar]
- Ferrendelli J. A., Kinscherf D. A., Chang M. M., Morgan J. F. Comparison of the effects of biogenic amines on cyclic GMP and cycle AMP levels in mouse cerebellum in vitro. Brain Res. 1975 Jan 24;84(1):63–73. doi: 10.1016/0006-8993(75)90800-8. [DOI] [PubMed] [Google Scholar]
- Fung B. K., Hurley J. B., Stryer L. Flow of information in the light-triggered cyclic nucleotide cascade of vision. Proc Natl Acad Sci U S A. 1981 Jan;78(1):152–156. doi: 10.1073/pnas.78.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greengard P. Possible role for cyclic nucleotides and phosphorylated membrane proteins in postsynaptic actions of neurotransmitters. Nature. 1976 Mar 11;260(5547):101–108. doi: 10.1038/260101a0. [DOI] [PubMed] [Google Scholar]
- Jones E. G., Wise S. P. Size, laminar and columnar distribution of efferent cells in the sensory-motor cortex of monkeys. J Comp Neurol. 1977 Oct 15;175(4):391–438. doi: 10.1002/cne.901750403. [DOI] [PubMed] [Google Scholar]
- Kelly P. T., McGuinness T. L., Greengard P. Evidence that the major postsynaptic density protein is a component of a Ca2+/calmodulin-dependent protein kinase. Proc Natl Acad Sci U S A. 1984 Feb;81(3):945–949. doi: 10.1073/pnas.81.3.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M. B., Bennett M. K., Erondu N. E. Biochemical and immunochemical evidence that the "major postsynaptic density protein" is a subunit of a calmodulin-dependent protein kinase. Proc Natl Acad Sci U S A. 1983 Dec;80(23):7357–7361. doi: 10.1073/pnas.80.23.7357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kincaid R. L., Manganiello V. C., Odya C. E., Osborne J. C., Jr, Stith-Coleman I. E., Danello M. A., Vaughan M. Purification and properties of calmodulin-stimulated phosphodiesterase from mammalian brain. J Biol Chem. 1984 Apr 25;259(8):5158–5166. [PubMed] [Google Scholar]
- Kincaid R. L., Vaughan M. Affinity chromatography of brain cyclic nucleotide phosphodiesterase using 3-(2-pyridyldithio)propionyl-substituted calmodulin linked to thiol-sepharose. Biochemistry. 1983 Feb 15;22(4):826–830. doi: 10.1021/bi00273a018. [DOI] [PubMed] [Google Scholar]
- King M. M., Huang C. Y., Chock P. B., Nairn A. C., Hemmings H. C., Jr, Chan K. F., Greengard P. Mammalian brain phosphoproteins as substrates for calcineurin. J Biol Chem. 1984 Jul 10;259(13):8080–8083. [PubMed] [Google Scholar]
- Klee C. B., Crouch T. H., Krinks M. H. Calcineurin: a calcium- and calmodulin-binding protein of the nervous system. Proc Natl Acad Sci U S A. 1979 Dec;76(12):6270–6273. doi: 10.1073/pnas.76.12.6270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostopoulos G. K., Limacher J. J., Phillis J. W. Action of various adenine derivatives on cerebellar Purkinje cells. Brain Res. 1975 Apr 25;88(1):162–165. doi: 10.1016/0006-8993(75)90966-x. [DOI] [PubMed] [Google Scholar]
- Lin C. T., Dedman J. R., Brinkley B. R., Means A. R. Localization of calmodulin in rat cerebellum by immunoelectron microscopy. J Cell Biol. 1980 May;85(2):473–480. doi: 10.1083/jcb.85.2.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Llinás R., Sugimori M. Electrophysiological properties of in vitro Purkinje cell dendrites in mammalian cerebellar slices. J Physiol. 1980 Aug;305:197–213. doi: 10.1113/jphysiol.1980.sp013358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Llinás R., Sugimori M. Electrophysiological properties of in vitro Purkinje cell somata in mammalian cerebellar slices. J Physiol. 1980 Aug;305:171–195. doi: 10.1113/jphysiol.1980.sp013357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohmann S. M., Walter U., Miller P. E., Greengard P., De Camilli P. Immunohistochemical localization of cyclic GMP-dependent protein kinase in mammalian brain. Proc Natl Acad Sci U S A. 1981 Jan;78(1):653–657. doi: 10.1073/pnas.78.1.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meibach R. C., Siegel A. Efferent connections of the hippocampal formation in the rat. Brain Res. 1977 Mar 25;124(2):197–224. doi: 10.1016/0006-8993(77)90880-0. [DOI] [PubMed] [Google Scholar]
- Proctor W. R., Dunwiddie T. V. Adenosine inhibits calcium spikes in hippocampal pyramidal neurons in vitro. Neurosci Lett. 1983 Feb 21;35(2):197–201. doi: 10.1016/0304-3940(83)90550-5. [DOI] [PubMed] [Google Scholar]
- Robinson P. J., Dunkley P. R. Depolarisation-dependent protein phosphorylation and dephosphorylation in rat cortical synaptosomes is modulated by calcium. J Neurochem. 1985 Feb;44(2):338–348. doi: 10.1111/j.1471-4159.1985.tb05422.x. [DOI] [PubMed] [Google Scholar]
- Schwartzkroin P. A., Slawsky M. Probable calcium spikes in hippocampal neurons. Brain Res. 1977 Oct 21;135(1):157–161. doi: 10.1016/0006-8993(77)91060-5. [DOI] [PubMed] [Google Scholar]
- Sharma R. K., Adachi A. M., Adachi K., Wang J. H. Demonstration of bovine brain calmodulin-dependent cyclic nucleotide phosphodiesterase isozymes by monoclonal antibodies. J Biol Chem. 1984 Jul 25;259(14):9248–9254. [PubMed] [Google Scholar]
- Shenolikar S., Thompson W. J., Strada S. J. Characterization of a Ca2+-calmodulin-stimulated cyclic GMP phosphodiesterase from bovine brain. Biochemistry. 1985 Jan 29;24(3):672–678. doi: 10.1021/bi00324a020. [DOI] [PubMed] [Google Scholar]
- Shields S. M., Ingebritsen T. S., Kelly P. T. Identification of protein phosphatase 1 in synaptic junctions: dephosphorylation of endogenous calmodulin-dependent kinase II and synapse-enriched phosphoproteins. J Neurosci. 1985 Dec;5(12):3414–3422. doi: 10.1523/JNEUROSCI.05-12-03414.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siggins G. R., Hoffer B. J., Bloom F. E. Cyclic adenosine monophosphate: possible mediator for norepinephrine effects on cerebellar Purkinje cells. Science. 1969 Sep 5;165(3897):1018–1020. doi: 10.1126/science.165.3897.1018. [DOI] [PubMed] [Google Scholar]
- Stewart A. A., Ingebritsen T. S., Manalan A., Klee C. B., Cohen P. Discovery of a Ca2+- and calmodulin-dependent protein phosphatase: probable identity with calcineurin (CaM-BP80). FEBS Lett. 1982 Jan 11;137(1):80–84. doi: 10.1016/0014-5793(82)80319-0. [DOI] [PubMed] [Google Scholar]
- Teo T. S., Wang J. H. Mechanism of activation of a cyclic adenosine 3':5'-monophosphate phosphodiesterase from bovine heart by calcium ions. Identification of the protein activator as a Ca2+ binding protein. J Biol Chem. 1973 Sep 10;248(17):5950–5955. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Eldik L. J., Wolchok S. R. Conditions for reproducible detection of calmodulin and S100 beta in immunoblots. Biochem Biophys Res Commun. 1984 Nov 14;124(3):752–759. doi: 10.1016/0006-291x(84)91022-2. [DOI] [PubMed] [Google Scholar]
- Walaas S. I., Aswad D. W., Greengard P. A dopamine- and cyclic AMP-regulated phosphoprotein enriched in dopamine-innervated brain regions. Nature. 1983 Jan 6;301(5895):69–71. doi: 10.1038/301069a0. [DOI] [PubMed] [Google Scholar]
- Wood J. G., Wallace R. W., Whitaker J. N., Cheung W. Y. Immunocytochemical localization of calmodulin and a heat-labile calmodulin-binding protein (CaM-BP80) in basal ganglia of mouse brain. J Cell Biol. 1980 Jan;84(1):66–76. doi: 10.1083/jcb.84.1.66. [DOI] [PMC free article] [PubMed] [Google Scholar]