Abstract
The potential of human embryonic stem (ES) cells as experimental therapies for neuronal replacement has recently received considerable attention. In view of the organization of the mature nervous system into distinct neural circuits, key challenges of such therapies are the directed differentiation of human ES cell-derived neural precursors (NPs) into specific neuronal types and the directional growth of axons along specified trajectories. In the present study, we cultured human NPs derived from the NIH-approved ES line BGO1 on polycaprolactone fiber matrices of different diameter (i.e., nanofibers and microfibers) and orientation (i.e., aligned and random); fibers were coated with poly-L-ornithine/laminin to mimic the extracellular matrix and support the adhesion, viability, and differentiation of NPs. On aligned fibrous meshes, human NPs adopt polarized cell morphology with processes extending along the axis of the fiber, whereas NPs on plain tissue culture surfaces or random fiber substrates form nonpolarized neurite networks. Under differentiation conditions, human NPs cultured on aligned fibrous substrates show a higher rate of neuronal differentiation than other matrices; 62% and 86% of NPs become TUJ1 (+) early neurons on aligned micro- and nanofibers, respectively, whereas only 32% and 27% of NPs acquire the same fate on random micro- and nanofibers. Metabolic cell activity/viability studies reveal that fiber alignment and diameter also have an effect on NP viability, but only in the presence of mitogens. Our findings demonstrate that fibrous substrates serve as an artificial extracellular matrix and provide a microenviroment that influences key aspects of the neuronal differentiation of ES-derived NPs.
Introduction
Neural stem cells presently used for transplantation are derived from human embryonic stem (ES) cells that are differentiated in vitro into the neural lineage or from neural precursors (NPs) isolated from the fetal or the adult nervous system.1–3 As recently demonstrated, neural stem cells can be also derived from somatic cells reprogrammed to pluripotency with combinations of transcription factors.4–6 At this juncture, human ES cells may represent the best solution for transplantation therapies as they can provide a potentially unlimited source of human neural stem cells for single or repeated administrations in the nervous system.7 The substantial plasticity and broad differentiation potential of human ES cells have been demonstrated in several in vitro studies where these cells were induced to specific neuronal and glial lineages, including motor8 and dopaminergic9,10 neurons as well as oligodendrocytes.11
One way to control the growth and differentiation of grafted neural stem cells is the use of suitable scaffolding materials serving as three-dimensional microenvironments for the cells to attach and grow and also functioning as temporary extracellular matrix (ECM). Fibrous matrices are particularly useful in neural tissue engineering as they can be engineered to have high porosity, high surface-to-volume ratio, and high spatial interconnectivity,12 all physical properties that can promote cellular adhesion and proliferation.13,14 Fibrous substrates of desired sizes and dimensions can be easily prepared using electrospinning,15–17 that is, a convenient fabrication process to produce random or aligned fibrous matrices with fiber diameters ranging from tens of nanometers to several microns.18–20 The topography of electrospun nanofibers resembles that of the fibrous ECM proteins.21 Various studies have shown that the alignment of micro-to-nano topography can significantly influence the morphology, proliferation rate, phenotype, and function of various cell types,22–24 as well as adult and ES cell types.25,26 For example, when cultured on aligned microgrooves, human keratinocytes tend to elongate and their longitudinal axis is co-aligned with the grooves.27 Surface-aminated electrospun nanofibers have been shown to promote the adhesion and proliferation of umbilical cord blood hematopoietic stem/progenitor cells.28 These adaptations are believed to involve rearrangement of cytoskeletal constituents, a process that in turn can influence cell phenotype and function via established links with intracellular signaling pathways.
Some of the above principles have been recently found to be useful in controlling neural stem cell differentiation for the purpose of neural tissue engineering. For example, aligned poly-L-lactic acid (PLLA) nanofibrous substrates support the adhesion of immortalized neural stem cell-like C17.2 cells and cause extensive neurite outgrowth along the longitudinal axis of nanofibers.12,29 Mouse neural stem cells show increased rate of differentiation on nanofibers than on microfibers; and aligned electrospun fibers further promote neurite outgrowth.12,30 Fiber diameter of electrospun polymer nanofiber matrix can also influence the proliferation and differentiation of neural stem cells derived from the rat hippocampus.31
In this study, fiber matrices of different diameter (nanofibers and microfibers) and orientation (aligned and random) were prepared by electrospinning and functionalized with ECM molecules to create an environment biocompatible with human ES cell-derived NPs. The effects of topography on the morphology, viability, and differentiation of human ES cell-derived NPs were examined. Our findings suggest that fiber diameter and orientation can impact the adhesion and viability of human NPs and that cells seeded on aligned fibrous substrates appear to have the best viability. Aligned fiber orientation also has a profound effect on the differentiation of cells in the neuronal lineage irrespective of diameter.
Materials and Methods
Nanofiber preparation and characterization
Polycaprolactone (PCL) fiber matrices were prepared by electrospinning. Solutions of PCL (Sigma) were prepared in a mixture of dichloromethane:methanol (80:20, v/v). The PCL solution was dispensed through a syringe pump at 2.5 mL/h through a 12-kV potential field, resulting in the formation of a thin, charged polymer jet that was deposited onto glass coverslips affixed to a grounded collecting plate. Random fiber meshes were collected by depositing fibers onto a stationary plate, and aligned fiber meshes were collected by depositing the polymer fibers onto the edge of a grounded rotating aluminum disc. Microfiber substrates were fabricated by electrospinning 12 wt% PCL solution. Nanofiber matrices were fabricated by doping a 12 wt% solution with 0.01% octadecyl rhodamine B chloride (R-18; Invitrogen), a cationic amphiphile that serves to stabilize the polymer jet and yield smaller diameter fibers. For characterization of the fiber diameter, meshes that had been sputter-coated with a 4-nm layer of platinum were imaged using a JEOL 6700F scanning electron microscope. High-magnification images (10,000 ×) of the matrices were acquired, and fiber diameters were measured using NIH ImageJ software. A minimum of 50 fibers were measured for each sample group.
Fibrous matrices were prepared for cell culture studies as follows: small droplets of medical-grade silicone adhesive (Factor I) were used to secure the mesh onto the coverslips. Matrices were sterilized by washing in ethanol and phosphate-buffered saline (PBS), followed by exposure to UV in the biosafety cabinet for 30 min. Fibers were then coated with poly-L-ornithine (PLO)/laminin. Briefly, matrices were immersed in 10 μg/mL PLO (Sigma) for 2 h at 37°C, washed three times with PBS, and then soaked in 20 μg/mL laminin (Sigma) overnight at 37°C, and washed three times with PBS at room temperature before cell seeding.
Human NP cultures and cell seeding on fiber substrates
Human NPs were differentiated from the human ES cell line BG01 included in the NIH Human Embryonic Stem Cell Registry (http://stemcells.nih.gov/research/registry). NP cultures were established and expanded as previously described.32 For analysis of their disposition and differentiation on PCL substrates, NPs were detached from the culture plate using TrypLE™ solution (Gibco BRL), harvested and collected by centrifugation, and resuspended in the culture medium with or without 20 ng/mL human recombinant epidermal growth factor (EGF; Sigma) and 20 ng/mL human recombinant basic fibroblast growth factor (bFGF; Sigma). Cells were seeded at a density of 5 × 105 cells/well onto PLO/laminin-coated electrospun PCL matrices that were placed into sterile 24-well culture dishes in triplicate. Fiber substrates were of varying diameters (∼250 nm to ∼1 μm) and different orientation, including aligned versus randomly oriented fibers. Tissue culture polystyrene wells coated with PLO/laminin served as positive controls. Cells were cultured on fiber matrices in complete neurobasal medium (NBM) supplemented with 20 ng/mL each of EGF and bFGF. The complete NBM contains 94% NBM (v/v), 1% 100 × Pen-Strep, 1% L-glutamine, 1% MEM nonessential amino acid solution (all above from Invitrogen), 0.1% bovine serum albumin (w/v, Fraction V; Sigma), 0.1 mM β-mercaptoethanol (Sigma), and supplements B27 (without retinoic acid) and N2 (both from Invitrogen). For the PLO/laminin-coated wells, cells were seeded at a density of 5 × 104 cells/well. A separate plate including control wells was used to study the effect of fiber matrices in the differentiation of human NPs. The same cell densities were used but mitogens were removed from the media. NPs were then cultured at 37°C under 5% of CO2 atmosphere.
Immunocytochemistry and imaging of NPs on fiber matrices
NP cultures were fixed with 4% neutral-buffered, freshly depolymerized paraformaldehyde for 15 min. Fixed cells were washed twice with PBS before staining. After washing, cells were incubated for 1 h in blocking solution (5% donkey serum, 1 × PBS, with or without Triton X-100), and primary antibodies were applied in blocking buffer overnight at room temperature. Antibodies used for immunocytochemistry include primary antibodies against nestin (1:200; neural progenitor marker; mouse anti-human nestin IgG1; Millipore/Chemicon), beta III tubulin (1:600; neuronal marker; rabbit polyclonal anti-TUJ1; Covance), glial fibrillary acidic protein (1:700; astrocyte marker; rabbit polyclonal anti-GFAP; Dako Cytomation), and RIP (1:100; oligodendrocyte marker; mouse anti-RIP; Chemicon). Cells were then washed in PBS and treated with secondary antibodies coupled to Cy2 or Cy3 (1:400 dilution; Jackson ImmunoResearch) for 30–60 min. After two washes in PBS, cells were stained with 4′,6′-diamidino-2-phenylindole (DAPI) and then coverslipped using ProLong Gold anti-fade reagent (Invitrogen Corporation). In control sections, primary antibodies were omitted from the staining procedure. Cells were studied with epifluorescence and confocal microscopy. Microscopy was performed using a Nikon TE2000 fluorescence microscope and confocal microscopic images were captured with a Zeiss LSM 510 inverted confocal microscope (Carl Zeiss, Inc.). Double staining for nestin or TUJ1 and DAPI was performed to determine the percentage rates of NPs and early neurons. Three samples from each type of matrix were examined under the microscope. Ten visual fields were randomly selected and cells from each field were manually counted. Analysis was conducted with one-way analysis of variance followed by Tukey's multiple-comparison post hoc test.
Metabolic activity/viability assay
To measure the viability of NPs seeded on matrices of different type of orientation (aligned and random) and diameter (nano- and microfibers) and on tissue culture polystyrene surface, a colorimetric methyl thiazoletetrazolium (MTT) assay was used (Promega). Mitochondrial dehydrogenases of living cells reduce the tetrazolium ring of MTT and yield a blue formazan product, which can be measured spectrophotometrically. The amount of formazan produced is proportional to the number of viable cells in cell culture. Metabolic activity was analyzed on day 3 of cell culture. Briefly, cells seeded on fiber substrates were washed with PBS and then incubated with 20 μL of MTT (5 mg/mL in PBS) for 3 h at 37°C. After incubation, the dye solution was discarded and formazan was extracted with 500 μL of dimethyl sulfoxide (Sigma). A total of 100 μL of the extract was transferred into the wells of a 96-well polysterene plate and optical absorbance at 490 nm was recorded with a spectrophotometric plate reader. Variation in metabolic activity was studied with one-way analysis of variance followed by Tukey's multiple comparison post hoc test. Cell proliferation experiments were performed in triplicate and the results were expressed as mean ± standard error.
Scanning electron microscopy of cells cultured on fiber matrices
Samples prepared for scanning electron microscopy (SEM) analysis were fixed with a mixture of 2% glutaraldehyde/1% paraformaldehyde in PBS for 45 min and then treated with 1% osmium tetroxide in 100 mM sodium cacodylate buffer for 15 min at room temperature. Samples were processed and imaged on a field-emission SEM (JEOL 6700F) according to the method reported previously.31
Results
Preparation of fibrous substrates and responses of proliferating human NPs to PCL matrices
We adopted a previously reported method to prepare PCL fiber matrices for this study.31 Aligned and randomly oriented PCL fibers prepared by electrospinning were collected onto coverslips, forming a dense mat that covered the whole surface of glass coverslips. Aligned fibrous substrates containing fibers of an average diameter of ∼250 nm were named aligned nanofibers (ANF), and those with fibers of average diameter of ∼1 μm were named aligned microfibers (AMF). To explore the effects of variable fiber diameter and orientation on the biology of NPs, random PCL fibrous matrices with similar fiber diameters were also fabricated and named random nanofibers (RNF) and random microfibers (RMF). Fiber substrates were coated with PLO/laminin, based on previous findings that such coatings support the adhesion and growth of NPs.33
The morphology of electrospun PCL fiber substrates was observed with SEM (Fig. 1). Most of the fibers in the oriented fibrous meshes were well aligned along one direction. To understand how cells would respond to electrospun PCL substrates, dissociated NPs were seeded on aligned or random fibrous matrices and incubated in normal growth media in the presence of 20 ng/mL of FGF-2 and EGF, that is, requisite mitogens for human NP proliferation.
FIG. 1.
Scanning electron microscopy characterization of electrospun fiber matrices. Aligned (A) and random (A′) microfiber scaffolds prepared with 12 wt% polycaprolactone solution. Aligned (B) and random (B′) nanofiber scaffolds fabricated with a 12 wt% solution/0.01% octadecyl rhodamine B chloride. Images were acquired at high magnification (10,000 ×). Scale bars are 1 μm.
Cell morphologies and distributions of NP cells on the PCL fiber meshes were studied with both SEM and immunocytochemistry. Representative SEM micrographs of human NPs cultured for 3 days on aligned or randomly oriented fibers revealed the close interaction between human NPs and fiber matrices (Fig. 2). A representative SEM image of human NPs cultured on ANF is shown in Figure 2A and indicates that cell bodies had an elongated appearance with their longitudinal axis parallel to the direction of PCL fibers. In contrast, NPs spread across the nanofiber network forming a continuous flat cell layer (Fig. 2B). In both cases, NPs maintained substantial cell–fiber adhesion, indicating the ability of matrices to facilitate cell attachment/interaction (Fig. 2B, C).
FIG. 2.
Scanning electron microscopy images of human NPs seeded on poly-L-ornithine/laminin-coated fiber meshes for 3 days. (A) Cells cultured on an aligned nanofiber substrate. Human NPs are attached on the scaffold and acquire an elongated shape when they grow on aligned fibrous substrates. (B, C) Cells on a random nanofiber substrate. NPs spread across the fiber mat surfaces, forming a continuous cell layer (B) where multiple cell–cell interactions are observed (C). Panels (B) and (C) are low- and high-power photographs, respectively, of cells on randomly oriented nanofibers. Scale bars are 10 μm. NPs, neural precursors.
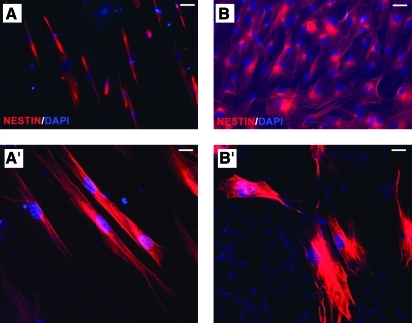
The effect of fibrous substrates on NP disposition was also studied using immunocytochemistry for nestin, a neural stem/progenitor marker, observed with epifluorescence or confocal microscopy (Fig. 3). As in the case of SEM, the general topographical disposition of nestin+ NPs followed the underlying structure of the fibrous meshes. Cells were well attached on both nano- and microfiber meshes. The ones seeded on the aligned fibrous matrices assumed a longitudinal shape and grew parallel to the PCL fibers (Fig. 3A, A'). In contrast, NPs on random fiber matrices assumed a polygonal shape and appeared to grow without particular directionality (Fig. 3B, B'). These patterns indicate that PCL fibrous substrates can determine the shape and guide the growth of human NPs in culture according to fiber alignment.
FIG. 3.
Immunofluorescent images of nestin (+) human NPs cultured onto different substrates. The cytology of human NPs cultured on aligned (A, A′) and random (B, B′) nanofiber matrices was examined following nestin immunocytochemistry (NPs are in red) and epifluorescence (A, B) or confocal microscopy (A′, B′). DAPI counterstaining (blue) was used to observe cell nuclei. Scale bars are 50 μm. DAPI, 4′,6′-diamidino-2-phenylindole. Color images available online at www.liebertonline.com/ten.
Viability of cultured human NPs on different substrates in the presence or absence of mitogens
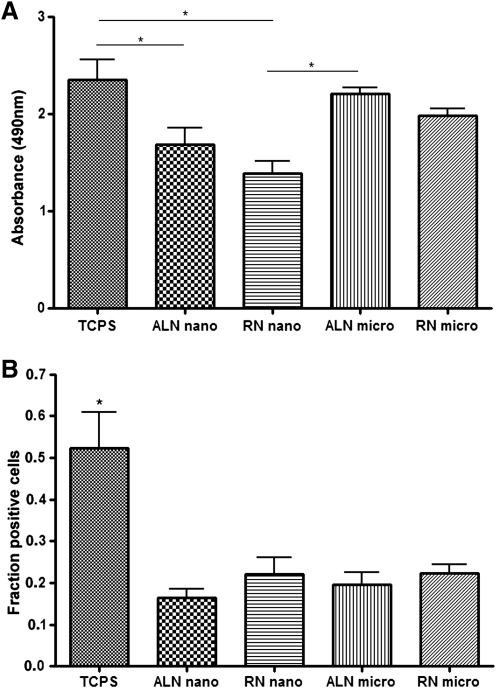
To test the effect of fiber alignment and diameter on human NP viability, we expanded NPs in culture media first in the presence of FGF-2 and EGF. Cell viability was evaluated using the MTT metabolic activity assay (Fig. 4A). Optical absorbance data indicate that cells growing in tissue culture plate present significantly higher metabolic activity than the ones on fibers of the nanometer scale. Among the fibrous matrices tested, cells seeded on AMF appeared to have the greatest metabolic activity, an effect suggesting that cell alignment and diameter might have a positive effect on cell metabolic activity and survival.
FIG. 4.
Effect of fiber alignment and diameter on human NPs viability. (A) The MTT assay was used to assess the viability of cultured human NPs on different fibrous scaffolds in the presence of mitogens. Overall variance was studied with ANOVA and was significant (p < 0.05). Significant differences between groups based on Tukey's post hoc multiple comparison test are indicated by an asterisk. (B) Quantitative analysis of nestin-positive cells cultured on different substrates in the absence of growth factors. As detailed in the Materials and Methods section, experiments were performed three times; 10 visual fields were randomly selected per culture and over 1500 cells were manually counted. Results were grouped per substrate type and studied with ANOVA. Variance was significant (p < 0.05). Significant differences between groups based on post hoc testing are indicated by an asterisk. Bars represent mean ± standard error in both panels. ANOVA, analysis of variance; TCPS, tissue culture polystyrene surface; ALN nano, aligned nanofibrous substrate; RN nano, random nanofibrous substrate; ALN micro, aligned microfibrous substrate; RN micro, random microfibrous substrate.
The effect of fiber alignment and diameter on human NP viability was also tested in the absence of exogenously added growth factors. Cells were cultured for 3 days after growth factor withdrawal and the viability of human NPs on different substrates was assessed by nestin immunocytochemistry (Fig. 4B). The rate of nestin+ cells was 52% on tissue culture surface and decreased substantially compared with all fiber substrates including ANF (16%), RNF (22%), AMF(19%), and RMF (22%) substrates.
Taken together, data from the MTT metabolic cell activity assay and nestin immunocytochemistry indicate that fiber alignment and diameter had an effect on NP viability only in the presence of FGF-2/EGF. In the absence of mitogens, there was no significant difference between nano- and microfibers or aligned versus random fibers, as all fibrous substrates appeared to have a negative effect on survival.
Effect of fiber diameter/alignment on the neuronal differentiation of human NPs
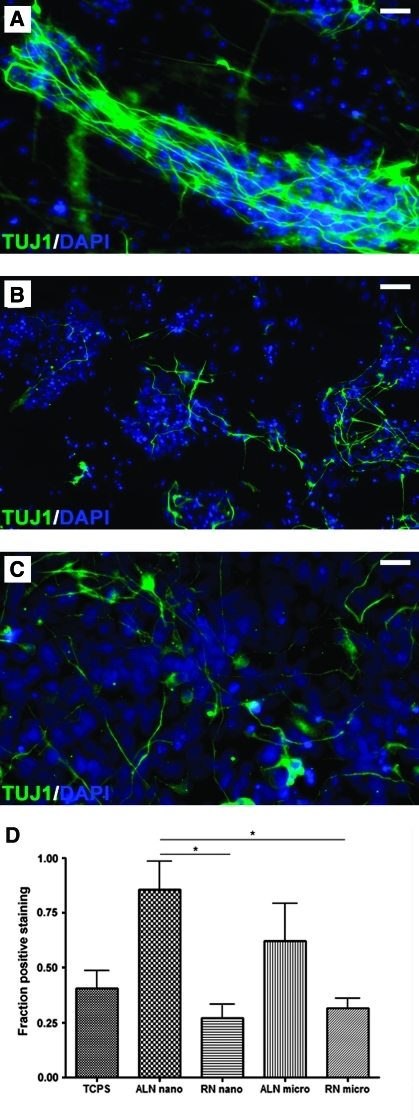
To explore the effect of fiber diameter and alignment on the neuronal differentiation of human NPs on fibrous substrates, we withdrew bFGF and EGF and allowed NPs to differentiate for 15 days. Phenotypic maturation of differentiating NPs was studied with immunofluorescence for the class III β-tubulin epitope TUJ1. The examination of such stained preparations showed that differentiated NPs grown on AMF and ANF extended axons in a highly oriented manner, that is, along the axis of the aligned fibers (Fig. 5A). This aligned morphology was significantly different from that of cells grown on random fibers (Fig. 5B) or on tissue culture plate surface (Fig. 5C) where neurites exhibited random orientation (Fig. 5B).
FIG. 5.
Effect of various types of substrates on the neuronal differentiation of human NPs. Fluorescence microscopy images of cells cultured on aligned (A) and random (B) fibrous substrates and on TCPS (C) for 15 days in the absence of growth factors. The phenotypic differentiation of human NPs was assessed by immunocytochemistry for TUJ1, a class III β-tubulin protein that marks early neurons (green). DAPI (blue) was used for counterstaining of nuclei. The fractions of TUJ1+ cells are shown in (D). Data represent three independent experiments, and 10 randomly selected fields were counted per culture. Significant differences between groups based on post hoc testing (p < 0.05, ANOVA test) are indicated by an asterisk. Bars represent mean ± standard error of the mean. Color images available online at www.liebertonline.com/ten.
The effects of PCL fiber diameter and alignment on differentiation of NPs were assessed by counting TUJ1+ cells from ten randomly selected fields of NP cultures on various types of fibrous matrices (Fig. 5D). Cells cultured on aligned fiber substrates showed higher levels of TUJ1 immunoreactivity. There were significantly higher rates of TUJ1+ NPs on AMF (62%) and ANF (86%) than on tissue culture plate surface cultures (40%) or to cultures involving RNF and RMF (27% and 32%, respectively). Immunoreactivity of differentiating NPs for the astrocyte marker GFAP and the oligondendrocyte markers O4 or RIP was negligible (data not shown). These findings show that aligned fiber matrices significantly promote the neuronal differentiation of human NPs as compared with random fiber substrates. The effect of fiber diameter was less significant for fibers with diameters in the range of 250 nm to 1 μm that were examined in the present study.
Discussion
In this present study, we explored the effects of electrospun fibrous substrates of various diameters and orientations on the viability and differentiation of human ES cell-derived NPs. Although the effects of electrospun fibers on a variety of neuronal and non-neuronal cell types have been addressed in previous studies, to the best of our knowledge, the present findings represent the first description of the responses of human ES cell-derived NPs to different fibrous matrices. With the electrospinning technique used here we were able to produce fibers with average diameters of 250 nm and 1 μm, as representative of the nanometer and micron scale ranges that have been previously shown to have an impact on the adhesion, spreading, migration, survival, and lineage specification of neural stem cells.12,31
The diameter of electrospun fibers was varied by changing a number of parameters related to the spinning process of the PCL solution used in the present study—a biodegradable polymer commonly used in tissue engineering applications.26,34 Some of these parameters include polymer concentration, flow rate, and distance between needle and collector. The incorporation of ECM molecules, that is, laminin on to electrospun fiber surface, renders matrices adhesive to cells and promotes cell survival.35
Tissue engineering scaffolds provide a three-dimensional template for cells to adhere, proliferate, and differentiate.36,37 The ECM-like properties of electrospun nanofibers have been shown to have an effect on the proliferation and differentiation/fate choice of a variety of cell types, including adult somatic and stem cells. For example, the proliferation and differentiation of osteoblasts have been promoted on carbon nanofiber scaffolds.38 Also, surface-aminated electrospun nanofibers have been shown to promote the adhesion and proliferation of human umbilical cord blood-derived hematopoietic progenitor cells.28 Human mesenchymal stem cells seeded on nano- and microfibrous scaffolds maintained the cell alignment on both types of scaffolds, but nanofibrous scaffolds significantly enhanced the chondrogenic differentiation of cells.39
In the present in vitro study, human NPs were found to interact favorably with fibrous substrates based on evidence that they attached well and responded with morphological changes. Human NPs seeded on aligned fibers acquired a spindle-like shape and extended processes parallel to the fiber axis. This morphology is probably due to contact guidance and cytoskeletal reorganization in response to substrate topography.39,40 Our findings also demonstrate that the type of substrate where cells grow significantly influences their survival. The two-dimensional tissue culture plate substrate was associated with higher numbers of surviving human NPs than nanofibrous matrices; among the fibrous substrates, cells seeded on AMF showed the highest viability. These results are consistent with the previous report that osteoprogenitor cells cultured on larger-diameter poly(D,L-lactide) fiber substrates (diameter 2.1 μm) yielded higher cell densities than when cultured on smaller-diameter fibers (0.14 μm), a pattern consistent with higher cell proliferation rates in the former scenario.41
Our immunocytochemical findings using phenotypic markers for mature cells of the nervous system showed that the propensity of neuronal differentiation was higher on ANF and AMF than on random fibers and on the two-dimensional tissue culture plate substrate. We also found that axonal outgrowth from differentiated neurons follows the direction of aligned fibers. Previous studies have shown that by altering the size of electrospun fiber diameter, one may significantly influence stem cell spreading, migration, proliferation, and differentiation. In one study,12 PLLA was electrospun into matrices with varied diameter and orientation by changing PLLA solution concentration and collection method; immortalized neural stem cells from neonatal mouse cerebellum seeded onto thus prepared nanofiber meshes showed increased neurofilament immunostaining (a measure of neuronal differentiation) compared with cells seeded on microfiber substrates. Further, neurite outgrowth was parallel to the direction of aligned PLLA fibers. On the basis of their ability to sense the topography of their tissue-specific microenvironment, stem cells can alter responses related to migration, adhesion, proliferation, and differentiation. By modifying different structural parameters of the electrospun fibrous matrix, we can recapitulate the hierarchical structure of the ECM in a synthetic substrate and, ideally, induce the desired cellular responses. As shown in our study, human NPs assumed a longitudinal shape when seeded on aligned fibers and their tendency for neuronal differentiation was higher, a pattern consistent with a shape-dependent control of lineage specification. Although the exact signaling pathways responsible for the effects of matrix architecture on stem cell function have yet to be elucidated, such a lineage specification mechanism may involve cytoskeletal and nuclear rearrangements induced by the acquisition of a particular cell shape.42,43 On the other hand, the promotional effects of the aligned fibrous substrates on differentiation may explain their adverse effects of survival; in the developing nervous system, differentiation is typically associated with reduced proliferation and enhanced apoptosis.44–46
Biomaterial technologies and specifically fibrous scaffolds offer great opportunities to promote repair of the nervous system after injury. In experiments on the mammalian visual system, it has been shown that a self-assembled peptide nanofiber scaffold can support axon regeneration through the site of an acute injury.47 The present study also demonstrates that aligned fibers can promote the neuronal differentiation and direct neurite growth of human NPs. Such contact guidance effect by fibrous scaffolds may be enhanced through the incorporation of growth factors and ECM proteins that serve to optimize the cell–fiber interactions and to also influence the differentiation of NPs into specific neural lineages, including neurons, astrocytes, and oligodendrocytes.48 The scaffolds may function as a carrier system for the local delivery of stem cells and provide the necessary trophic support and biochemical signals that may not be available to neural stem cells in the environment of injury.49 The utility of ANF in promoting neuronal differentiation and guiding regenerating axons has been demonstrated in vivo in peripheral nerve, where these preparations have been reported to promote axonal regrowth and Schwann cell migration.50 However, the application of these nanofibers to NP delivery to central nervous system remains to be investigated.
In conclusion, in this report, we evaluated a set of electrospun fibers as a substrate for culturing and differentiating human ES cell-derived NPs. Our results indicate that fiber diameter and orientation are important factors that influence the adhesion properties, as well as viability and differentiation of NPs. Effects on viability and differentiation are important factors to consider when grafting human ES cell-derived NPs for therapeutic purposes in vivo32,51 because such effects influence, in turn, the engraftment, survival, and neuronal differentiation of grafts and may prevent the formation of teratomas.
Acknowledgments
This work was supported by grants from the National Institutes of Health (NS45140, V.E.K.) and the Muscular Dystrophy Association (V.E.K.). V.M. is a recipient of the Maryland Stem Cell Research Fund Postdoctoral Fellowship.
Disclosure Statement
No competing financial interests exist.
References
- 1.Cummings B.J. Uchida N. Tamaki S.J. Salazar D.L. Hooshmand M. Summers R. Gage F.H. Anderson A.J. Human neural stem cells differentiate and promote locomotor recovery in spinal cord-injured mice. Proc Natl Acad Sci U S A. 2005;102:14069. doi: 10.1073/pnas.0507063102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yan J. Xu L. Welsh A.M. Hatfield G. Hazel T. Johe K. Koliatsos V.E. Extensive neuronal differentiation of human neural stem cell grafts in adult rat spinal cord. PLoS Med. 2007;4:e39. doi: 10.1371/journal.pmed.0040039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tabar V. Panagiotakos G. Greenberg E.D. Chan B.K. Sadelain M. Gutin P.H. Studer L. Migration and differentiation of neural precursors derived from human embryonic stem cells in the rat brain. Nat Biotechnol. 2005;23:601. doi: 10.1038/nbt1088. [DOI] [PubMed] [Google Scholar]
- 4.Takahashi K. Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 5.Karumbayaram S. Novitch B.G. Patterson M. Umbach J.A. Richter L. Lindgren A. Conway A.E. Clark A.T. Goldman S.A. Plath K. Wiedau-Pazos M. Kornblum H.I. Lowry W.E. Directed differentiation of human-induced pluripotent stem cells generates active motor neurons. Stem Cells. 2009;27:806. doi: 10.1002/stem.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hu B.Y. Zhang S.C. Differentiation of spinal motor neurons from pluripotent human stem cells. Nat Protoc. 2009;4:1295. doi: 10.1038/nprot.2009.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mitalipova M. Calhoun J. Shin S. Wininger D. Schulz T. Noggle S. Venable A. Lyons I. Robins A. Stice S. Human embryonic stem cell lines derived from discarded embryos. Stem Cells. 2003;21:521. doi: 10.1634/stemcells.21-5-521. [DOI] [PubMed] [Google Scholar]
- 8.Li X.J. Du Z.W. Zarnowska E.D. Pankratz M. Hansen L.O. Pearce R.A. Zhang S.C. Specification of motoneurons from human embryonic stem cells. Nat Biotechnol. 2005;23:215. doi: 10.1038/nbt1063. [DOI] [PubMed] [Google Scholar]
- 9.Perrier A.L. Tabar V. Barberi T. Rubio M.E. Bruses J. Topf N. Harrison N.L. Studer L. Derivation of midbrain dopamine neurons from human embryonic stem cells. Proc Natl Acad Sci U S A. 2004;101:12543. doi: 10.1073/pnas.0404700101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zeng X. Cai J. Chen J. Luo Y. You Z.B. Fotter E. Wang Y. Harvey B. Miura T. Backman C. Chen G.J. Rao M.S. Freed W.J. Dopaminergic differentiation of human embryonic stem cells. Stem Cells. 2004;22:925. doi: 10.1634/stemcells.22-6-925. [DOI] [PubMed] [Google Scholar]
- 11.Keirstead H.S. Nistor G. Bernal G. Totoiu M. Cloutier F. Sharp K. Steward O. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. J Neurosci. 2005;25:4694. doi: 10.1523/JNEUROSCI.0311-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang F. Murugan R. Wang S. Ramakrishna S. Electrospinning of nano/micro scale poly(L-lactic acid) aligned fibers and their potential in neural tissue engineering. Biomaterials. 2005;26:2603. doi: 10.1016/j.biomaterials.2004.06.051. [DOI] [PubMed] [Google Scholar]
- 13.Chiu J. Luu Y.K. Hsiao B.S. Chu B. Hadjiargyrou M. Electrospun nanifibrous scaffolds for miomedical applications. J Biomed Nanotechnol. 2005;1:115. [Google Scholar]
- 14.Ma Z. Kotaki M. Inai R. Ramakrishna S. Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng. 2005;11:101. doi: 10.1089/ten.2005.11.101. [DOI] [PubMed] [Google Scholar]
- 15.Yoshimoto H. Shin Y.M. Terai H. Vacanti J.P. A biodegradable nanofiber scaffold by electrospinning and its potential for bone tissue engineering. Biomaterials. 2003;24:2077. doi: 10.1016/s0142-9612(02)00635-x. [DOI] [PubMed] [Google Scholar]
- 16.Bhattarai S.R. Bhattarai N. Yi H.K. Hwang P.H. Cha D.I. Kim H.Y. Novel biodegradable electrospun membrane: scaffold for tissue engineering. Biomaterials. 2004;25:2595. doi: 10.1016/j.biomaterials.2003.09.043. [DOI] [PubMed] [Google Scholar]
- 17.Pham Q.P. Sharma U. Mikos A.G. Electrospinning of polymeric nanofibers for tissue engineering applications: a review. Tissue Eng. 2006;12:1197. doi: 10.1089/ten.2006.12.1197. [DOI] [PubMed] [Google Scholar]
- 18.Huang Z.M. Zhang Y.Z. Kotaki M. Ramakrishna S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Comp Sci Technol. 2003;63:2223–2253. [Google Scholar]
- 19.Li D. Xia Y. Electrospinning of Nanofibers: reinventing the wheel? Adv Mater. 2004;16:1151. [Google Scholar]
- 20.Jayaraman K. Kotaki M. Zhang Y. Mo X. Ramakrishna S. Recent advances in polymer nanofibers. J Nanosci Nanotechnol. 2004;4:52. [PubMed] [Google Scholar]
- 21.Smith L.A. Ma P.X. Nano-fibrous scaffolds for tissue engineering. Colloids Surf B Biointerfaces. 2004;39:125. doi: 10.1016/j.colsurfb.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 22.Bozkurt A. Deumens R. Beckmann C. Olde D.L. Schugner F. Heschel I. Sellhaus B. Weis J. Jahnen-Dechent W. Brook G.A. Pallua N. In vitro cell alignment obtained with a Schwann cell enriched microstructured nerve guide with longitudinal guidance channels. Biomaterials. 2009;30:169. doi: 10.1016/j.biomaterials.2008.09.017. [DOI] [PubMed] [Google Scholar]
- 23.Deumens R. Koopmans G.C. Den Bakker C.G. Maquet V. Blacher S. Honig W.M. Jerome R. Pirard J.P. Steinbusch H.W. Joosten E.A. Alignment of glial cells stimulates directional neurite growth of CNS neurons in vitro. Neuroscience. 2004;125:591. doi: 10.1016/j.neuroscience.2004.02.010. [DOI] [PubMed] [Google Scholar]
- 24.Gerardo-Nava J. Fuhrmann T. Klinkhammer K. Seiler N. Mey J. Klee D. Moller M. Dalton P.D. Brook G.A. Human neural cell interactions with orientated electrospun nanofibers in vitro. Nanomedicine (Lond) 2009;4:11. doi: 10.2217/17435889.4.1.11. [DOI] [PubMed] [Google Scholar]
- 25.Li W.J. Laurencin C.T. Caterson E.J. Tuan R.S. Ko F.K. Electrospun nanofibrous structure: a novel scaffold for tissue engineering. J Biomed Mater Res. 2002;60:613. doi: 10.1002/jbm.10167. [DOI] [PubMed] [Google Scholar]
- 26.Ma Z.W. Kotaki M. Inai R. Ramakrishna S. Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng. 2005;11:101. doi: 10.1089/ten.2005.11.101. [DOI] [PubMed] [Google Scholar]
- 27.Teixeira A.I. Nealey P.F. Murphy C.J. Responses of human keratocytes to micro- and nanostructured substrates. J Biomed Mater Res A. 2004;71:369. doi: 10.1002/jbm.a.30089. [DOI] [PubMed] [Google Scholar]
- 28.Chua K.N. Chai C. Lee P.C. Tang Y.N. Ramakrishna S. Leong K.W. Mao H.Q. Surface-aminated electrospun nanofibers enhance adhesion and expansion of human umbilical cord blood hematopoietic stem/progenitor cells. Biomaterials. 2006;27:6043. doi: 10.1016/j.biomaterials.2006.06.017. [DOI] [PubMed] [Google Scholar]
- 29.Yang F. Xu C.Y. Kotaki M. Wang S. Ramakrishna S. Characterization of neural stem cells on electrospun poly(L-lactic acid) nanofibrous scaffold. J Biomater Sci Polym Ed. 2004;15:1483. doi: 10.1163/1568562042459733. [DOI] [PubMed] [Google Scholar]
- 30.Xie J. Willerth S.M. Li X. Macewan M.R. Rader A. Sakiyama-Elbert S.E. Xia Y. The differentiation of embryonic stem cells seeded on electrospun nanofibers into neural lineages. Biomaterials. 2009;30:354. doi: 10.1016/j.biomaterials.2008.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Christopherson G.T. Song H. Mao H.Q. The influence of fiber diameter of electrospun substrates on neural stem cell differentiation and proliferation. Biomaterials. 2009;30:556. doi: 10.1016/j.biomaterials.2008.10.004. [DOI] [PubMed] [Google Scholar]
- 32.Nasonkin I. Mahairaki V. Xu L.Y. Hatfield G. Cummings B.J. Eberhart C. Ryugo D.K. Maric D. Bar E. Koliatsos V.E. Long-term, stable differentiation of human embryonic stem cell-derived neural precursors grafted into the adult mammalian neostriatum. Stem Cells. 2009;27:2414. doi: 10.1002/stem.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Song H. Stevens C.F. Gage F.H. Astroglia induce neurogenesis from adult neural stem cells. Nature. 2002;417:39. doi: 10.1038/417039a. [DOI] [PubMed] [Google Scholar]
- 34.Schnell E. Klinkhammer K. Balzer S. Brook G. Klee D. Dalton P. Mey J. Guidance of glial cell migration and axonal growth on electrospun nanofibers of poly-epsilon-caprolactone and a collagen/poly-epsilon-caprolactone blend. Biomaterials. 2007;28:3012. doi: 10.1016/j.biomaterials.2007.03.009. [DOI] [PubMed] [Google Scholar]
- 35.Koh H.S. Yong T. Chan C.K. Ramakrishna S. Enhancement of neurite outgrowth using nano-structured scaffolds coupled with laminin. Biomaterials. 2008;29:3574. doi: 10.1016/j.biomaterials.2008.05.014. [DOI] [PubMed] [Google Scholar]
- 36.Ingber D.E. Mow V.C. Butler D. Niklason L. Huard J. Mao J. Yannas I. Kaplan D. Vunjak-Novakovic G. Tissue engineering and developmental biology: going biomimetic. Tissue Eng. 2006;12:3265. doi: 10.1089/ten.2006.12.3265. [DOI] [PubMed] [Google Scholar]
- 37.Goldberg M. Langer R. Jia X. Nanostructured materials for applications in drug delivery and tissue engineering. J Biomater Sci Polym Ed. 2007;18:241. doi: 10.1163/156856207779996931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Elias K.L. Price R.L. Webster T.J. Enhanced functions of osteoblasts on nanometer diameter carbon fibers. Biomaterials. 2002;23:3279. doi: 10.1016/s0142-9612(02)00087-x. [DOI] [PubMed] [Google Scholar]
- 39.Wise J.K. Yarin A.L. Megaridis C.M. Cho M. Chondrogenic differentiation of human mesenchymal stem cells on oriented nanofibrous scaffolds: engineering the superficial zone of articular cartilage. Tissue Eng Part A. 2009;15:913. doi: 10.1089/ten.tea.2008.0109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dunn G.A. Heath J.P. A new hypothesis of contact guidance in tissue cells. Exp Cell Res. 1976;101:1. doi: 10.1016/0014-4827(76)90405-5. [DOI] [PubMed] [Google Scholar]
- 41.Badami A.S. Kreke M.R. Thompson M.S. Riffle J.S. Goldstein A.S. Effect of fiber diameter on spreading, proliferation, and differentiation of osteoblastic cells on electrospun poly(lactic acid) substrates. Biomaterials. 2006;27:596. doi: 10.1016/j.biomaterials.2005.05.084. [DOI] [PubMed] [Google Scholar]
- 42.Guilak F. Cohen D.M. Estes B.T. Gimble J.M. Liedtke W. Chen C.S. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. 2009;5:17. doi: 10.1016/j.stem.2009.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lim S.H. Mao H.Q. Electrospun scaffolds for stem cell engineering. Adv Drug Deliv Rev. 2009;61:1084. doi: 10.1016/j.addr.2009.07.011. [DOI] [PubMed] [Google Scholar]
- 44.Oppenheim R.W. Cell death during development of the nervous system. Annu Rev Neurosci. 1991;14:453. doi: 10.1146/annurev.ne.14.030191.002321. [DOI] [PubMed] [Google Scholar]
- 45.Hamburger V. Cell death in the development of the lateral motor column of the chick embryo. J Comp Neurol. 1975;160:535. doi: 10.1002/cne.901600408. [DOI] [PubMed] [Google Scholar]
- 46.Hamburger V. Levi-Montalcini R. Proliferation, differentiation and degeneration in the spinal ganglia of the chick embryo under normal and experimental conditions. J Exp Zool. 1949;111:457. doi: 10.1002/jez.1401110308. [DOI] [PubMed] [Google Scholar]
- 47.Ellis-Behnke R.G. Liang Y.-X. You S.-W. Fay D.K.C. Zhang S. So K.-F. Schneider G.E. Nano neuro knitting: peptide nanofiber scaffold for brain repair and axon regeneration with functional return of vision. Eur J Neurosci. 2004;20:1984. doi: 10.1073/pnas.0600559103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Silva G.A. Czeisler C. Niece K.L. Beniash E. Harrington D.A. Kessler J.A. Stupp S.I. Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science. 2004;303:1352. doi: 10.1126/science.1093783. [DOI] [PubMed] [Google Scholar]
- 49.Willerth S.M. Sakiyama-Elbert S.E. Cell therapy for spinal cord regeneration. Adv Drug Deliv Rev. 2008;60:263. doi: 10.1016/j.addr.2007.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jiang X. Lim S.H. Mao H.Q. Chew S.Y. Current applications and future perspectives of artificial nerve conduits. Exp Neurol. 2010;223:86. doi: 10.1016/j.expneurol.2009.09.009. [DOI] [PubMed] [Google Scholar]
- 51.Xu L. Ryugo D.K. Pongstaporn T. Johe K. Koliatsos V.E. Human neural stem cell grafts in the spinal cord of SOD1 transgenic rats: differentiation and structural integration into the segmental motor circuitry. J Comp Neurol. 2009;514:297. doi: 10.1002/cne.22022. [DOI] [PMC free article] [PubMed] [Google Scholar]