Abstract
The disruption of the spatial order of electromechanical junctions at myocyte-intercalated disks (ICDs) is a poorly understood characteristic of many cardiac disease states. Here, in vitro and in vivo evidence is provided that zonula occludens-1 (ZO-1) regulates the organization of gap junctions (GJs) and adherens junctions (AJs) at ICDs. We investigated the contribution of ZO-1 to cell-cell junction localization by expressing a dominant-negative ZO-1 construct (DN-ZO-1) in rat ventricular myocytes (VMs). The expression of DN-ZO-1 in cultured neonatal VMs for 72 h reduced the interaction of ZO-1 and N-cadherin, as assayed by colocalization and coimmunoprecipitation, prompting cytoplasmic internalization of AJ and GJ proteins. DN-ZO-1 expression in adult VMs in vivo also reduced N-cadherin colocalization with ZO-1, a phenomenon not observed when the connexin-43 (Cx43)-ZO-1 interaction was disrupted using a mimetic of the ZO-1-binding ligand from Cx43. DN-ZO-1-infected VMs demonstrated large GJs at the ICD periphery and showed a loss of focal ZO-1 concentrations along plaque edges facing the disk interior. Additionally, there was breakdown of the characteristic ICD pattern of small interior and large peripheral GJs. Continuous DN-ZO-1 expression in VMs over postnatal development reduced ICD-associated Cx43 GJs and increased lateralized and cytoplasmic Cx43. We conclude that ZO-1 regulation of GJ localization is via an association with the N-cadherin multiprotein complex and that this is a key determinant of stable localization of both AJs and GJs at the ICD.
Keywords: N-cadherin, connexin-43, postsynaptic density 95/Drosophila disk large/zonula occludens-1, heart
the intercalated disk (ICD) is a specialized domain of electromechanical coupling between myocytes (6, 31). The disk incorporates three types of intercellular junctions: adherens junctions (AJs), desmosomes, and gap junctions (GJs). AJs and desmosomes provide for cadherin-mediated adhesion between myocytes and serve as a platform for cytoskeletal attachment. GJs are aggregates of cell-to-cell channels that mediate the exchange of small molecules (<1,000 Da) between cells and in the myocardium conduct the ion transients responsible for the intercellular propagation of the cardiac action potential. Based on early electron microscopic studies, the ICD was perceived more as a locus at which multiple junctional types were found rather than as an integrated unitary structure in its own right. However, the disk is now recognized as a functional unit specialized for electromechanical coupling in which AJs, GJs, and desmosomes, together with cytoskeletal proteins, are interlinked via a web of protein-protein interactions (31, 32).
Over postnatal ventricular development, the remodeling of electromechanical junctions from lateral, side-by-side contacts between myocytes results in a preferential accumulation of AJs, desmosomes, and GJs at ICDs in the adult (2, 13, 16). The average size of GJs also increases during the postnatal growth of the ventricle (16, 36). A distinct population of large GJs composed of connexin-43 (Cx43) differentiates at the periphery of ICDs, with smaller GJs localizing to the disk interior (12). Organizational patterns seen homogenously throughout the ventricle early during the postnatal period, including the lateralization of junctional proteins such as Cx43 and desmoplakin, are recapitulated in adult ventricles of some arrhythmic cardiomyopathies (7, 15, 34, 35, 38, 42). The fact that the normal ordered pattern of intercellular junctions at the mature ICD is disrupted by mutations in mechanical junction proteins (30, 34) suggests a hierarchical dependence for mechanical junction formation to precede normal GJ localization. Understanding the mechanism of how intercellular junctions are patterned and integrated into the mature ICD will provide insight into the origins of arrhythmic pathologies.
Zonula occludens-1 (ZO-1) is an actin-binding scaffolding protein that localizes at the ICD (4, 19, 20, 40). Originally discovered in association with the tight junction (8, 10, 39), ZO-1 has since been shown to interact with components of various other junction types (9, 25) and is required for the formation of polarized AJs in epithelial cells (22). The amino-terminal (NT) half of ZO-1 binds a number of proteins thought to contribute to the structure and function of the AJs, including α-catenin, ALL1-fused gene from chromosome 6 protein/afadin, ZO-2, ZO-3, and others (1, 11, 17). ZO-1 also interacts with Cx43, the main GJ protein of the ventricular myocardium, via a consensus class II postsynaptic density 95/Drosophila disk large/ZO-1 (PDZ)-binding motif (40). This interaction involves the Cx43 carboxyl terminus (CT) and the second PDZ domain (PDZ2) in the NT of ZO-1 (9).
We have previously shown that a short peptide designed to disrupt ZO-1 interaction with Cx43 by mimicking the PDZ2-binding ligand of the Cx43 CT increased GJ size between cultured myocytes (20). Here we report that a dominant-negative ZO-1 construct (DN-ZO-1) perturbed the AJ and GJ organization between rat ventricular myocytes in vitro and in vivo. The expression of DN-ZO-1 in ventricular myocardium over postnatal development reduced the accumulation of GJs at ICDs, with complementary increases in lateralized GJs similar to those seen in the immature and diseased ventricle (7). Moreover, an acute and long-term expression of DN-ZO-1 disrupted the physiological patterns of N-cadherin and ZO-1, associated with a breakdown of the characteristic segregation of small interior and large peripheral GJ populations occurring at ICDs. Furthermore, a cytoplasmic internalization and a reduction in localization at sites of cell-cell interaction of ZO-1, N-cadherin, and Cx43 were observed. The alterations in AJ and GJ localization were not due to DN-ZO-1 weakening the interaction between Cx43 and ZO-1, as no such disruption to AJ protein localization was observed in the presence of the PDZ2-binding peptide derived from Cx43. We conclude that the level of ZO-1 association with the N-cadherin multiprotein complex determines the localization of GJs at cell-cell interactions between myocytes, primarily via the disruption of AJ organization.
MATERIALS AND METHODS
Animal care was approved by and in accordance with institutional guidelines at the Medical University of South Carolina, Charleston, SC (Animal welfare assurance number: A4328-01). To confirm reproducibility, the experiments were repeated a minimum of three times. The quantification of confocal images was performed on optical sections from at least three animals per time point.
Neonatal ventricular myocyte isolation and culture.
The isolation and culture of neonatal rat myocytes were performed according to the method of Simpson et al. (37) with modifications. Hearts were removed from neonatal rats at 1–4 postnatal days and minced in Hanks' balanced salt solution (HBSS; Gibco). Following extensive washes with HBSS, the tissue was incubated on a Nutator in digestion solution (HBSS with 0.5 mg/ml of collagenase and 10 mg/ml cytosine arabinoside) for periods of 20 min. Following each incubation, the solution containing isolated myocytes was removed and fresh digestion solution was added. The isolated myocytes were spun at 200 g for 10 min, and the supernatant was discarded. FBS (5 ml; Hyclone) was added to each pellet following centrifugation. After six digestion incubation periods, the pellets were pooled, placed in maintenance media (medium 199 with 20% FBS and 10 mg/ml cytosine arabinoside), and plated on a 150-ml culture dished for 1 h to selectively remove fibroblasts. Myocytes were then centrifuged, diluted in maintenance media, and plated on 0.02% gelatin (Sigma)-coated coverslips or culture dishes. The culture media was changed every 48 h. The myocytes were plated for 72 h before adenoviral infection to allow the formation of stable cell-cell contacts.
Expression of DN-ZO-1 postnatally in vitro and in vivo.
Full-length human ZO-1 cDNA in pSK-Bluescript+ was a gift from Dr. J. Anderson (Yale University, New Haven, CT). The open reading frame of ZO-1 from bases 150 to 1,227 was cloned using the polymerase chain reaction (forward primer: 5′-GAG ACA AGA TGT CCG CCA GA-3′; reverse primer: 5′-TTG AGA CAG CCC CAG GTT TAG-3′) into pGem-T Promega easy vector system. After the sequence and orientation were confirmed, the sequence was cloned into an AdEasy adenovirus expression vector upstream of a myc sequence (Q-Biogene, Carlsbad, CA). The construct incorporated a bicistronic cytomegalovirus promoter enabling the coexpression of the 369 amino acid truncated ZO-1-myc sequence and green fluorescent protein (GFP). Primary neonatal myocyte cultures were infected with Ad-DN-ZO-1, an empty GFP control adenovirus (Ad-GFP), or no adenovirus for a period of 24 h. The cultures were infected with Ad-DN-ZO-1 or Ad-GFP at an multiplicity of infection between 3 and 5, which resulted in an infection efficiency of >70%. Following 24 h of exposure to the adenovirus, the cultures were washed in PBS and new media was added. The cultures were harvested for Western blot analysis or immunohistochemistry after 72 h of protein expression.
Adenovirus injection in vivo was carried out either long term over the course of postnatal development or acutely over a 48–72-h time period. In the instance of the long-term expression of DN-ZO-1, microinjections of adenovirus into neonatal hearts were performed according to the method of Christensen et al. (5) with modifications. Neonatal rats (postnatal day 1) were placed on ice for 2 min. Approximately 15 μl of adenovirus were loaded into a flame stretched capillary tube. The capillary tube/needle was then mounted on a micro-manipulator system, and the chest cavity was penetrated. The entrance of pulsatile blood into the capillary tube indicated the penetration of the ventricle. A Picospritzer II (General Valve, Fairfield, NJ) was used to provide pressure for the injection of adenovirus into the ventricle. Following the injection, the rats were euthanized at 1-, 10-, 40-, and 90-day time points.
The acute expression of DN-ZO-1 was carried out in the adult rat heart. Female 8-wk-old Sprague-Dawley rats (Charles River) were anesthetized and intubated, and the thorax was exposed through an incision in the fourth intercostal space. The pericardial sac was removed, and ∼30 μl of Ad-DN-ZO-1 virus was injected subepicardially in the left ventricular free wall. The incisions were closed, and the rats were revived. The animals were euthanized 48 h postinjection. The hearts were fixed in 4% paraformaldehyde for 10 min and frozen in liquid nitrogen-cooled optimal temperature cutting media.
Coimmunoprecipitation analyses.
Coimmunoprecipitations were based on previously reported protocols from Barker et al. (3). Lysates were collected 72 h postinfection in 500 μl/well complete lysis buffer containing 50 mM Tris·HCl (pH 7.4), 150 mM NaCl, 2 mM EGTA, 1% Nonidet P-40, 0.1% SDS, 0.25% sodium deoxycholate, 0.01 mg/ml DNase, and complete protease-phosphatase inhibitors for 10 min at 4°C on orbital rotater (maximum rotation). The cells were scraped and syringed with a 22-gauge needle 5×, followed by a 27-gauge 6×, and lysis continued for 1 h at 4°C with rotation. The samples were centrifuged at 14,000 rpm for 20 min at 4°C. The pellets were extracted in one-tenth volume lysis buffer supplemented with 1% SDS and brought to the same volume as the supernatant in lysis buffer to give 0.1% SDS. Protein concentrations were determined with Pierce Micro BCA. Rabbit polyclonal antibodies were precoupled to n Protein A Sepharose Fast Flow (Amersham 17-5280-01) and mouse monoclonal antibodies to Protein G Sepharose Fast Flow (Amersham 17-0618-01). Coupling buffer (750 μl) containing (in mM) 150 NaCl, 50 Tris (pH 8), and 1 EDTA was added to the 65-μl packed washed beads/reaction, followed by 5 μg of either rabbit anti-ZO-1 (H300; Santa Cruz) or mouse anti-N-cadherin (33-3900; Zymed).
The coupling reactions were incubated for 1 h at 4°C with end-over-end rotation. The coupled beads were collected by centrifugation for 30 s at 600 g and washed 3× in cold PBS. Equal volumes of lysate were added to the coupled beads and incubated overnight at 4°C with end-over-end rotation. The complexes were centrifuged at 12,000 rpm for 20 s and washed 4× in complete lysis buffer. The complexes were released from the beads in 50 μl 2×T sample buffer (161-0791; Bio-Rad) and subsequently heated at 95°C for 5 min. The samples were resolved on 3–8% Tris-acetate gel (Criteron XT 345-0129; Bio-Rad) in 1×T Tricine running buffer (161-0790; Bio-Rad). Resolved proteins were transferred to Immoblion PVDF at 100 V for 1 h (15 mM Tris, 192 mM glycine, 10% vol/vol methanol, and 0.01% SDS) with cooling. The membranes were blocked in 2% nonfat dry milk/TBS-Tween-20 and probed with the following primary antibodies: rabbit anti-ZO-1 (1:2,000; 61-7300; Zymed) and rabbit anti-N-cadherin (1:100; H63; Santa Cruz). Subsequently, all the blots were probed with a goat anti-rabbit alkaline phosphatase secondary (1:10,000; 4010-04; Southern Biotechnology). Immunoreactive proteins were visualized by chemiluminescence using CDP STAR (Applied Biosystems T2306) as a substrate.
Confocal microscopy.
Frozen sections of rat heart were prepared for immunolabeling as previously described (2, 14), as were myocyte cultures (21). For intact tissue, hearts from various postnatal ages (days 1, 10, 40, and 90) were surgically removed, fixed in 4% paraformaldehyde, and washed thrice in PBS. Before being placed in TBS tissue freezing compound (Triangle Biomedical Sciences), the hearts were examined by fluorescence microscopy to assess and photograph the regions of GFP expression and to orient the hearts for sectioning. The specimens were then immediately immersed in liquid nitrogen until frozen solid. Fresh frozen sections (10 μm) were prepared at −32°C and placed on glass slides. The sections were then washed with PBS-A with 0.01% Triton X-100 added and blocked with 1% bovine serum albumin in PBS-A for 1 h. The primary antibodies used for immunolabeling were rabbit anti-ZO-1 (1:100; Zymed), mouse anti-Cx43 (1:100, Chemicon, Temecula, CA), mouse anti-Cx43 immunofluorescence-1 (1:1,000), and rabbit anti-Cx43 (1:8,000; Sigma). As in our earlier studies (3, 43), Alexa488 (1:3,000; Molecular Probes) and Cy5 (1:125; Jackson )-conjugated secondary antibodies were used for double labeling to ensure that the signal overlap between detection channels did not occur. Experiments requiring double labeling in the presence of the viral GFP signal were labeled with Alexa 568 and 633 (1:3,000; Molecular Probes) or Cy5-conjugated secondary antibodies, and the images were obtained sequentially to prevent signal overlap. High-magnification optical sections were captured with either a Leica TCS SP2, Leica TCS SP5, or a Perkin-Elmer spinning disk laser confocal microscope (LCM) equipped with a 63× or 100× oil-immersion objective. The microscope model, objective, pinhole, gain, and black level settings were held constant for each experiment. For quantitative analyses of Cx43 GJ size, cellular distribution, and colocalization with ZO-1, confocal optical sections were taken at random locations from left ventricular myocardium (at postnatal days 1, 10, 20, and 90), isolated neonatal myocytes cells immunolabeled for Cx43 and/or ZO-1. Immunolabeled en face ICDs were optically sectioned and reconstructed as previously described (12).
Isolated neonatal rat ventricular myocytes (NRVMs) grown on gelatin-coated coverslips were fixed with 4% paraformaldehyde for 10 min, washed 3× in PBS-A, and then blocked with 1% bovine serum albumin in PBS-A with 0.01% Triton X-100 added for 1 h. The slides or coverslips were incubated overnight at 4°C in a combination of primary antibodies directed against ZO-1 (1:100; Zymed), Cx43 (1:100; Chemicon; Sigma), c-myc ( 1:100; Santa Cruz Biotechnology, Santa Cruz, CA), or N-cadherin (1:100; Sigma). Secondary antibodies conjugated to the fluorescent probes Alexa 546 and Alexa 633 were used to label the specific primary antibodies. Antifade agent (Molecular Probes) was added, and the sections were coverslipped and sealed.
Adult rat hearts were surgically removed 48 h after Ad-DN-ZO-1 injection, 10-μm sections were obtained, and the hearts were double immunolabeled using the following antibodies: ZO-1 (1:100, rabbit, Zymed), Cx43 (1:1,000, mouse immunofluorescence-1, and 1:8,000, rabbit; Sigma), and N-cadherin, (1:500, mouse, BD transductions) using anti-rabbit Alexa 568 and anti-mouse 633 secondary antibodies respectively (1:3,000; Invitrogen). A Leica LCM equipped with a 63× oil-immersion objective and a digital zoom of 4 was used to obtain a 32-slice Z-series (0.17 μm step size) of ICDs inside and outside the virally infected (GFP positive) regions. Proprietary Leica software was used to generate maximum projections of these slices and a colocalization mask.
Image analysis.
For analyses of ventricular myocardium, the quantification was replicated on three to six animals per time point. For analyses of isolated neonatal myocytes, the measurements were replicated on four independent cultures at each time point (24 and 48 h). The areas of individual Cx43 plaques/particles (>2,000 per condition and/or time point) were measured from 9–16 images using National Institutes of Health Image as previously described (2, 3, 14). The quantification of postnatal ZO-1 colocalization with Cx43 plaques was carried out according to Zhu et al. (43). Measurements of ZO-1 colocalization with the inner/outer edge of peripheral disk GJs was performed in ImageJ by bisecting the outer Cx43 plaques and dividing the area of the colocalized ZO-1 by the total Cx43 immunolabeled area. Cytofluorograms were generated using Colocalizer Pro 2.6 (Colocalization Research Software). Statistical analyses were done using Microsoft Excel. Unless otherwise noted, means were compared using Student's t-test; P values > 0.05 were rejected as not significant. Data shown with P values as means ± SE.
RESULTS
GJ distribution and Cx43-ZO-1 interaction in the adult ventricle.
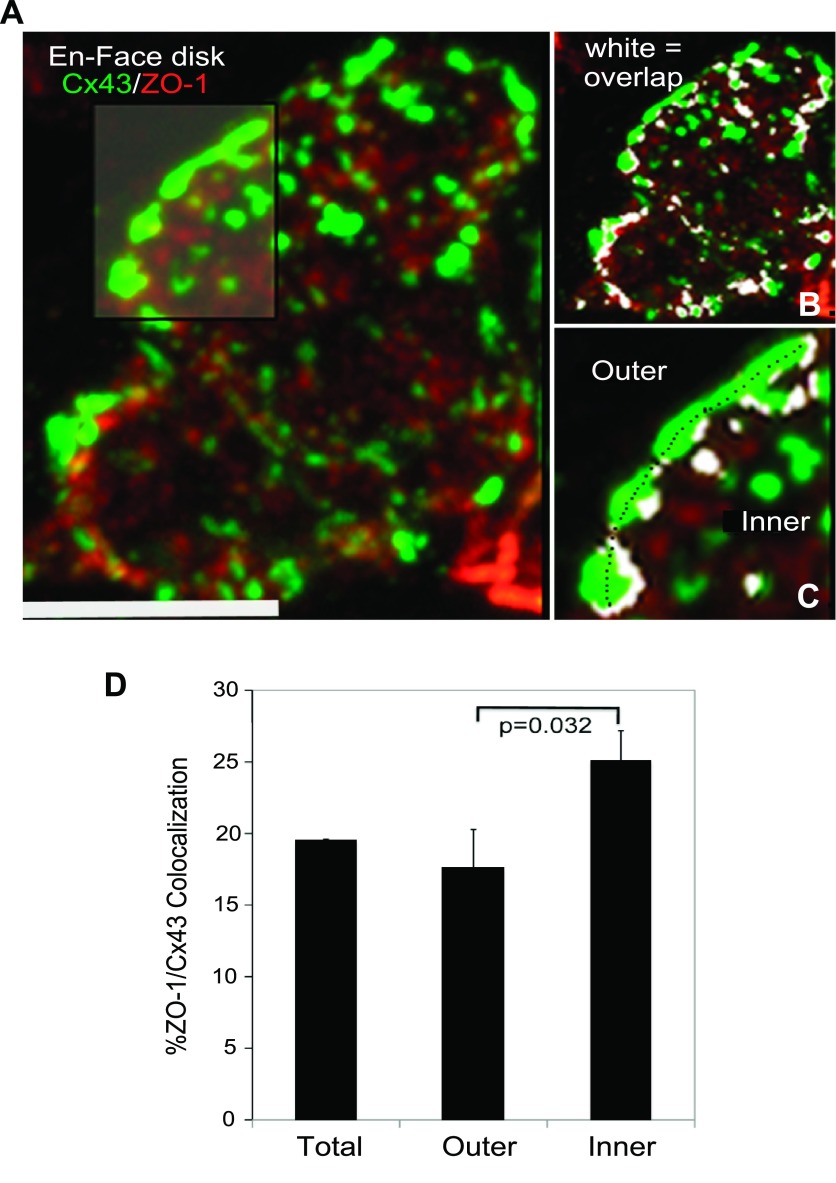
In the adult mammalian ventricle, the largest GJs ring the periphery of ICDs (Fig. 1A and Ref. 12). To gain further insight into the differentiation of this distinct class of large GJs, we examined ZO-1 colocalization with Cx43 in ICDs reconstructed from LCM optical sections. Consistent with previous studies (3, 43), GJs in the disk showed modest levels of colocalized ZO-1 mainly occurring at plaque edges (Fig. 1, A–C). However, we also identified a novel phenomenon not previously described. A radial asymmetry in ZO-1 localization at the large peripheral GJs was noted (Fig. 1C). Specifically, the outer edge of the large GJs demonstrated reduced levels of colocalized ZO-1 compared with the inner edge facing into the disk interior. An example of this organization is illustrated in Fig. 1C, inset, where a Cx43 aggregate at the periphery of the ICD can be seen to have its inner edge bordered by colocalized ZO-1 (white), whereas its outer edge is free of ZO-1. The quantification confirmed that ZO-1 colocalization at the outer edge of the large peripheral plaques was significantly lower (P < 0.05, n = 5 animals) than the edges facing into the disk interior (Fig. 1D).
Fig. 1.
A: connexin-43 (Cx43; green) and zonula occludens-1 (ZO-1; red) colocalized in an adult rat intercalated disk oriented en face as reconstructed from a maximum projection of a confocal optical section series recorded on a Perkin-Elmer Spinning disk laser confocal microscope with a ×100 objective. Scale bar = 10 μm. B: same disk as in A showing areas of overlap between Cx43 and ZO-1 signal highlighted in a white colocalization mask. C: subregion of the colocalization mask image shown in B as indicated by highlight showing asymmetric polarization of colocalized ZO-1 (white) on the inside edges (as defined by dashed line) of large gap junctions (GJs) ringing the intercalated disk. D: quantification of the average percentage of colocalized area in the total disk ring, the inner ring edge, and outer ring edge of 11 intercalated disks obtained from 5 adult rats. Colocalized area was significantly greater in inner ring edge vs. outer ring edge (Mann-Whitney, P = 0.032).
Expression of a DN-ZO-1 in cultured NRVMs.
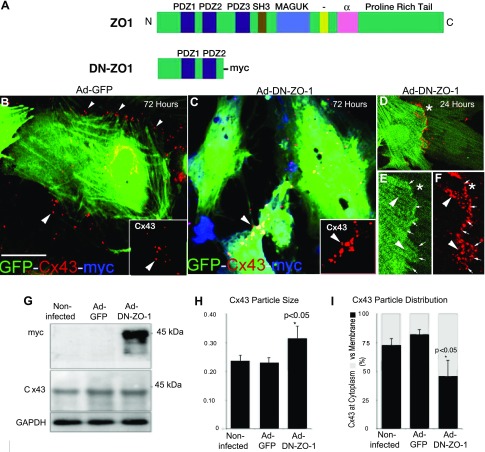
We next examined the effects of targeting ZO-1 on intercellular junctions in cultures of NRVMs. Previous groups have reported a DN-ZO-1 construct comprising a truncated NT sequence of ZO-1 incorporating its first and second PDZ domains. This sequence lacks the CT actin-binding domain of full-length ZO-1 and has been shown to disrupt AJs and GJs in cell culture models (27, 33, 41). We generated a bicistronic adenoviral vector that coexpressed this DN-ZO-1 sequence together with GFP. The DN construct incorporated amino acids 1 through 359 of ZO-1 tagged at its CT with the 10 amino acid myc sequence (Fig. 2A).
Fig. 2.
A: domain organization of full-length ZO-1 and a ZO-1 truncation [dominant negative (DN)-ZO-1] construct incorporating the first 2 postsynaptic density 95/Drosophila disk large/ZO-1 (PDZ) domains of ZO-1 and a 10 amino acid myc sequence. N, NH2 terminus; C, COOH terminus. B–F: confocal optical sections of neonatal myocytes cultured for 72 h or 24 h (D–F) following infection with adenovirus green fluorescent protein (Ad-GFP; B) or Ad-DN-ZO-1 (D–F) and immunolabeled for Cx43 (red) and myc (blue). *Position of high magnification. GFP localization occupies the green channel. Cx43 distribution in Ad-GFP-infected myocytes was localized primarily to the borders of myocytes (small arrowheads). Extensive Cx43 labeling in large particles was observed in the cytoplasm of DN-ZO-1-expressing cells (large arrowheads) and high magnification in B and C. G. Western blots for myc (top), Cx43 (middle), and GAPDH (bottom) in neonatal myocytes infected with Ad-GFP, Ad-DN-ZO-1, or no virus. H–I: quantification of Cx43 immunolabeling indicates significant increases in the size (H) and cytoplasmic distribution (I) of Cx43 aggregates following DN-ZO-1 expression compared with Ad-GFP-infected (GFP) and noninfected controls. SH3, SRC homology 3 domain; MAGUK, membrane-associated guanylate kinase. Scale bar = 20 μm. *P < 0.05.
Primary cultures of NRVMs were infected with Ad-DN-ZO-1, a control adenovirus expressing GFP (Ad-GFP), or no virus. Following 72 h of expression, the cultures were double immunolabeled for Cx43 and myc (Fig. 2, B–F). As expected, only NRVMs overexpressing DN-ZO-1 showed immunolabeling for both myc and GFP (blue and green channel in Figs. 2B and 3D, respectively). Western blot analysis with anti-myc antibodies indicated that a 42-kDa band, consistent with the molecular mass of DN-ZO-1, was expressed in cultures infected with Ad-DN-ZO-1 (Fig. 2G). NRVMs not exposed to virus or infected with Ad-GFP did not show this band (Fig. 2G, top). Western blot analysis indicated that the overall Cx43 levels did not vary between Ad-DN-ZO-1, Ad-GFP, and noninfected NRVMs (Fig. 2G, middle).
Fig. 3.
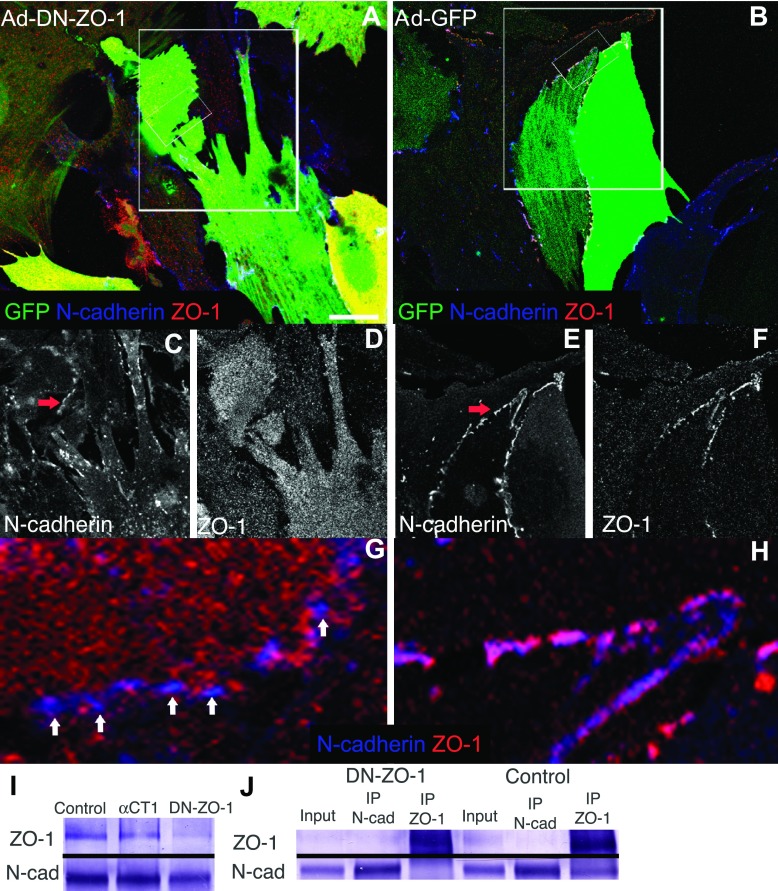
Neonatal rat ventricular myocytes (NRVMs) infected with either Ad-DN-ZO-1 (A, C, D, and G) or Ad-GFP (B, E, F, and H) and immunolabeled for N-cadherin (blue) and ZO-1 (red). Boxed regions in A and B are shown at higher magnification in C–H. GFP localization occupies the green channel. C–F demonstrate single channel images of Cx43 and ZO-1 expression of boxed regions in A and B. Arrows indicate regions of cell borders that are magnified and shown in G and H without the green channel. Note that N-cadherin and ZO-1 colocalize frequently at myocyte-myocyte contacts in control (GFP) cultures, and the same 2 proteins are largely internalized in the cytoplasm of DN-ZO-1-expressing cultures. Scale Bar = 25 μm. I. Western Blot of noninfected (left), DN-ZO-1-infected, or α-carboxy terminal-1 (α-CT1)-treated NRVM lysates probes with anti ZO-1 or N-cadherin antibodies. J: NRVM lysates immunoprecipitated (IP) with anti-ZO-1, -N-cadherin (N-Cad), or nonimmunoprecipitated (input). Note: no N-cadherin detected in DN-ZO-1-infected cardiomyocyte lysates immunoprecipitated with ZO-1 antibodies compared with uninfected cells (control).
The Cx43 distribution in DN-ZO-1-expressing cells was then examined. Conveniently, differential GFP expression enabled a ready discrimination of individual myocytes, providing a straightforward means of visualizing cell borders and sites of cell-cell contact. When this ability to visualize cell borders was used in conjunction with single confocal optical sectioning, Cx43 immunolabeling localized at cell borders could be distinguished from that in the cytoplasm. With the use of this approach, alterations in the distribution of Cx43 GJs were observed in DN-ZO-1 expressing NRVMs. NRVMs infected with Ad-DN-ZO-1 had larger Cx43 GJ particles (Fig. 2B, inset) relative to noninfected or Ad-GFP-infected cells (Fig. 2B, inset). Also, Cx43 immunolabeling in the cytoplasm was increased in DN-ZO-1-expressing NRVMs (Fig. 2, D–F). This phenomenon is unequivocally illustrated in Fig. 2 where cytoplasmic Cx43 particles are observed proximal to, but disassociated from, a cell-cell border delineated by GFP expression (Fig. 2E). The quantification confirmed that significant increases in Cx43 particle size (P < 0.05, Fig. 2H) and cytoplasmic localization of Cx43 (P < 0.05, Fig. 3I) had occurred in Ad-DN-ZO-1-infected NRVMs relative to Ad-GFP-infected and noninfected controls.
We next determined the acute effect of DN-ZO-1 on AJs as delineated by N-cadherin immunolabeling (Fig. 3, A–H). In control myocytes, ZO-1 colocalized with frequent N-cadherin punctae at intercellular contacts [Fig. 3, B, E (arrow), and H (high magnification)]. In contrast, at 72 h postinfection, DN-ZO-1-expressing NRVMs exhibited a prominent accumulation of N-cadherin and ZO-1 within the cytoplasm and reduced N-cadherin at cell-cell borders (compare Fig. 3C with 3E and 3D with 3F). Junction-like N-cadherin punctae could be seen at points of cell-to-cell contact between myocytes (Fig. 3, C and G, high magnification). However, this cell border-localized N-cadherin showed a reduction of colocalized ZO-1. Most notably, the distribution of ZO-1 shifted from a cell-cell border to cytoplasmic localization with DN-ZO-1 expression (compare Fig. 3D with 3F). To investigate this further, we undertook Western blot analysis and coimmunoprecipitation studies of ZO-1 and N-cadherin in NRVMs. Western blot analysis of NRVM lysates revealed a reduction in ZO-1 in DN-ZO-1-infected cells compared with noninfected cells or cells treated with a PDZ-binding peptide derived from the Cx43 carboxy terminus and designed to inhibit ZO-1 PDZ interactions (α-CT1) (Fig. 3I). Consistent with these results, the treatment of NRVMs with the α-CT1 PDZ-binding peptide resulted in no discernable change to N-cadherin localization (supplementary Fig. 1; note: supplemental figure may be found posted with the online version of this article). We were unable to detect changes in ZO-1 coimmunoprecipitated with N-cadherin in our assay; however, consistent with immunocytochemical results, we found a loss of the N-cadherin band coimmunoprecipitated with ZO-1 antibodies from lysates of NRVMs infected with Ad-DN-ZO-1 compared with uninfected NRVMs (Fig. 3J).
Effects on ICD organization in vivo of acute Ad-DN-ZO-1 infection.
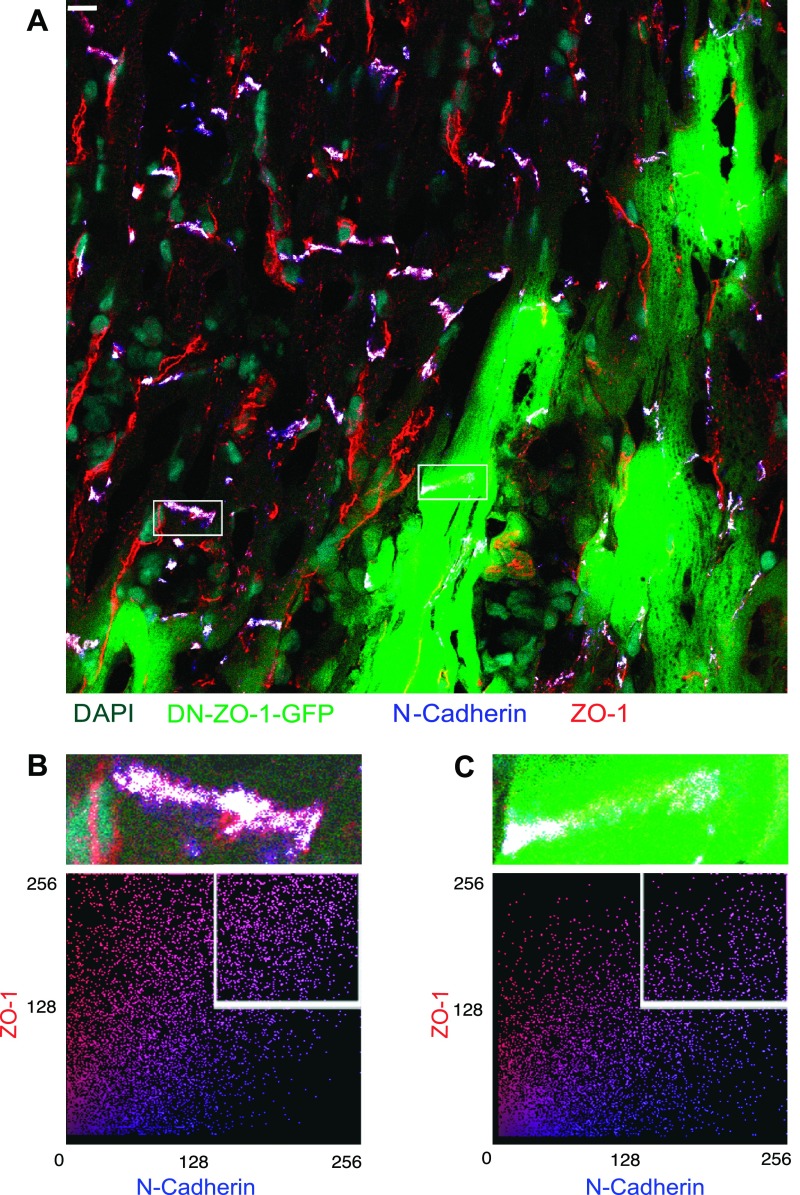
The effect of the acute expression of DN-ZO-1 over 48 h in adult ventricular myocardium was subsequently investigated. Infected adult rat hearts demonstrated frequent GFP-positive myocytes in the ventricle corresponding to sites of viral injection (Fig. 4A). For virally infected cells in vivo, GFP expression provided a further morphological marker in addition to demarcating individual myocytes as described in vitro. In vivo GFP enabled a discrimination of end to end (ICDs) from lateral domains of contact between longitudinally arrayed myocytes (Figs. 4 and 5). Figure 4A demonstrates an LCM field exhibiting areas of both virally infected and noninfected myocytes. Figure 4, B and C, shows a superimposition of N-cadherin signal with the N-cadherin/ZO-1 colocalization mask to highlight the colocalized pixels of disks indicated by boxed regions in Fig. 4A, bottom. A high-magnification colocalization analysis of these two ICDs in this field reveals a decrease in colocalization of N-cadherin and ZO-1 in the disk associated with the DN-ZO-1-expressing cell (compare Fig. 4B, top, with 4C, top). A cytofluorogram plot of the pixels in two selected disk regions exhibits less colocalization in the DN-ZO-1-positive region as indicated by the number of pixels in the upper right quartile (Fig. 4B, bottom, and 4C, bottom).
Fig. 4.
A: adult rat left ventricular myocardium harvested 48 h postinjection with DN-ZO-1. GFP-positive regions indicate areas of DN-ZO-1 expression. Scale bar = 10 μm. DAPI, 4,6-diamidino-2-phenylindole. B (top) and C (top): high magnification regions of A showing an intercalated disk from a GFP-negative (Ad-DN-ZO-1 noninfected; B) or GFP-positive (Ad-DN-ZO-1 infected; C) region with the ZO-1 N-cadherin colocalized pixels highlighted in white. B (bottom) and C (bottom): cytofluorogram plotting red ZO-1 signal vs. blue N-cadherin signal for the GFP-negative (B) and GFP-positive (C) region indicated above.
Fig. 5.
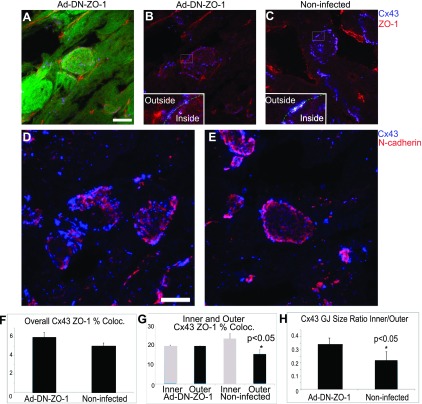
A–E: en face views of intercalated disks from 48 h Ad-DN-ZO-1-infected adult rat hearts from GFP-positive areas (A, B, and D) and GFP-negative areas (C and E). A: GFP labeling demonstrating Ad-DN-ZO-1-infected intercalated disk. Scale bar = 10 μm. B: same as A without green channel. Inset: Cx43-ZO-1 colocalization mask (white). C: noninfected disk from same heart shows normal disk morphology and Cx43-ZO-1 colocalization mask (white inset). D: Ad-DN-ZO-1-infected disks labeled for Cx43 (blue) and N-cadherin (red). E: control disk from DN-ZO-1-nonexpressing cell from same heart as D. F: total Cx43 ZO-1 colocalization (Coloc) did not significantly differ in fields containing GFP compared with areas absent GFP expression (n = 3; P > 0.05). G: quantification of inner vs. outer colocalization of Cx43 and ZO-1; n = 3 animals. H: quantification of Cx43 GJ size in the center vs. peripheral regions of adult disks; *P < 0.05.
Three-dimensional projections of LCM optical sections were used to generate en face views of ICDs of DN-ZO-1-expressing regions (Fig. 5, A–E). A colocalization analysis revealed no significant change in the overall Cx43/ZO-1 colocalization compared with noninfected regions (Fig. 5F). However, changes were noted in the geometry of the Cx43/ZO-1 colocalization pattern, as well as the characteristic distribution of GJ sizes at ICDs in Ad-DN-ZO-1-infected myocytes (Fig. 5, B and C). Specifically, GJs located at the disk periphery demonstrated uniform ZO-1 localization around plaque edges (Fig. 5B), i.e., a loss of the novel asymmetric colocalization pattern we report at normal disks found in noninfected hearts (Figs. 5C and 1C). The quantification of ZO-1 localized with GJs at the ICD periphery in DN-ZO-1-expressing myocytes indicated a loss of the normal pattern in which ZO-1 was focally concentrated at the inner facing plaque edge (Fig. 5G). Instead, ZO-1 was uniformly localized around the plaque edge of ICD peripheral GJs in DN-ZO-1-expressing myocytes. Concomitant with this altered ZO-1 colocalization pattern, there was a loss of the characteristic distinction between the large GJs at the disk periphery and smaller GJs in the disk interior. The size ratio of the inner region Cx43 GJs to those at the disk periphery was significantly (P < 0.05) increased in the Ad-DN-ZO-1-infected disks (Fig. 5H). The en face views of ICDs in Ad-DN-ZO-1-infected cells also revealed a disruption of normal N-cadherin organization (compare Fig. 5D with 5E). In addition to the decreased colocalization between N-cadherin and ZO-1 (Fig. 4), disks of cells expressing DN-ZO-1 showed distinct heterogeneity, with nonuniform clumps of N-cadherin occurring in the en face views (Fig. 5D) that were not evident in the noninfected myocytes (Fig. 5E).
Long-term DN-ZO-1 expression by ventricular myocytes in vivo increases lateralization of Cx43 GJs.
The experiments in the previous sections demonstrated that an acute expression of DN-ZO-1 disrupted the junctional complexes at the ICD in both neonatal and adult myocytes. We next determined how constitutive long-term DN-ZO-1 expression influences ICD differentiation over postnatal development. Adenovirus was injected into the ventricles of neonatal rats and expressed DN-ZO-1 over postnatal development using a method previously described by Chien and colleagues (5).
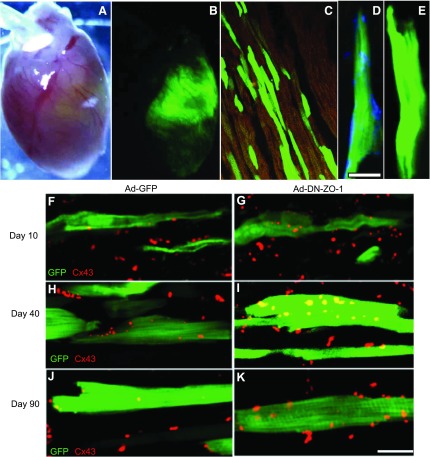
Following the injection of Ad-DN-ZO-1 and Ad-GFP into the neonatal ventricle, we found the foci of infected myocytes that numbered from a few to hundreds of cells that showed persistent GFP expression at 10, 40, and 90 postnatal days (Fig. 6, A–E). Myc-labeling, indicating the presence of the DN-ZO-1 protein, was also maintained in Ad-DN-ZO-1-infected myocytes in the ventricle (Fig. 6D). In contrast, Ad-GFP-infected and noninfected control cells exhibited no evidence of myc immunolabeling (Fig. 6E). Confocal imaging of single optical sections of DN-ZO-1 ventricles at 10, 40, and 90 days revealed a decrease in end-to-end localization of Cx43 and an increase in Cx43 GJ size and cytoplasmic localization over 90 postnatal days. (Fig. 6, F–K).
Fig. 6.
A: a postnatal day 10 heart infected on postnatal day 1 with Ad-DN-ZO-1. B: green GFP fluorescence indicates that the ventricle of this heart has been extensively infected with virus. Adult 90-day myocytes infected in vivo with Ad-GFP (C) or Ad-DN-ZO-1 (D) were labeled for myc (shown in blue). Myocytes infected with Ad-DN-ZO-1 showed labeling for myc (D), indicating the expression of DN-ZO-1, whereas Ad-GFP-infected myocytes (E) did not. GFP localization occupies the green channel. Scale bar = 25 μm. F–K: single confocal optical sections of Cx43-immunolabeled rat cardiac myocytes infected with Ad-GFP (F, H, and J) or Ad-DN-ZO-1 (G, I, and K) and euthanized at 10 (A–B), 40 (C–D), or 90 (E–F) postnatal days. Scale bar =10 μm. Over the course of postnatal development, the Cx43 GJs exhibit a cytoplasmic distribution in DN-ZO-1-infected myocytes.
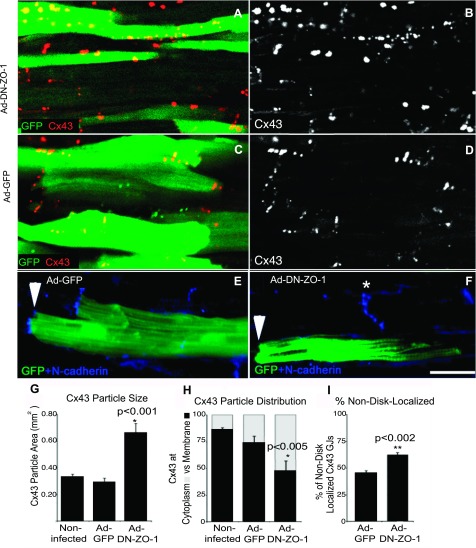
Immunolabeled Cx43 GJ size and distribution were measured in single confocal optical sections from the ventricle of 40-day-old animals, i.e., the approximate midpoint in the postnatal accumulation of GJs into ICDs (2) (Fig. 7, A–I). GFP labeling and confocal optical sectioning were again used to discriminate ICD, lateral, and cytoplasmic localizations of junctional components. Ventricular myocytes expressing DN-ZO-1 displayed alterations in GJ size and distribution compared with the Ad-GFP and noninfected controls (Fig. 7, A–D). Cx43 aggregate size was significantly increased (P < 0.001) in Ad-DN-ZO-1-infected myocytes, relative to controls (compare Fig. 7B with 7D; quantified in Fig. 7G). In addition, significantly increased levels (P < 0.005) of cytoplasmic Cx43 particles were found in DN-ZO-1-infected myocytes compared with those of Ad-GFP and noninfected control cells (Fig. 7H). A decrease in immunolabeled Cx43 GJs at ICDs was evident at 40 postnatal days (Fig. 7, A–F) relative to GFP-expressing myocytes. Concomitant with this decrease, Ad-DN-ZO-1-infected myocytes demonstrated notable increases in punctate Cx43 immunolabeling localized at lateral myocyte-myocyte interfaces. This was confirmed quantitatively (Fig. 7I). Significant increases (P < 0.005) in the proportion of Cx43 GJs localizing at lateral domains of cell-cell contacts were observed in Ad-DN-ZO-1-infected relative to Ad-GFP expressing myocytes. Additionally, consistent with our results in acute expression models (Figs. 3 and 4), N-cadherin immunolabeling was reduced at cell-cell contacts in ventricular myocytes expressing DN-ZO-1 relative to uninfected myocytes (Fig. 7, E and F).
Fig. 7.
A–D: myocytes from hearts at 40 days of postnatal development were immunolabeled for Cx43 following infection in vivo with either Ad-DN-ZO-1 (A, B, and F) or Ad-GFP viruses at postnatal day 1 (C–E). Cx43 signal is shown in red and GFP fluorescence is in green. The Cx43 signal is displayed in black and white single channel images in B and D. Ad-GFP-infected cells maintained typical distributions of Cx43 at cell borders and at developing intercalated disks (D–F). Striking increases in lateralization and cytoplasmic labeling of large Cx43 particles were detected in Ad-DN-ZO-1-infected myocytes (A–D). N-cadherin staining of Ad-GFP (E) in Ad-DN-ZO-1 (F)-infected myocytes. *Adjacent uninfected cell with N-cadherin labeling at the intercalated disk. Quantification of immunolabeling in single optical sections from 40-day-old ventricles indicate significant increases in Cx43 GJ aggregate size (G), cytoplasmic internalization (H), and lateralization (I) following Ad-DN-ZO-1 infection compared with Ad-GFP-infected and noninfected controls. Scale bar =12 μm.
DISCUSSION
Previous work from our laboratory has focused on the role of direct ZO-1 interaction with Cx43 on GJs. Specifically, we have shown that Cx43 GJ size is related to the level of ZO-1 interaction with the CT of Cx43 and that inhibiting this interaction increases GJ size in cell culture models (20). Here we report two novel findings. First, the population of large GJs that ring the periphery of ICDs exhibit a decreased association with ZO-1 along the outer GJ edge compared with the inner GJ edge, i.e., the edge facing the disk interior (Fig. 1). This new observation in the adult rodent heart is consistent with our previous in vitro studies that showed ZO-1 at the plaque edge limited GJ size. The larger size of junctions at the ICD periphery appears to result from the asymmetric loss of ZO-1 at the outer edge of GJs, leading to the disinhibition of size growth regulation at the outer edge of the plaque. This asymmetry in colocalized ZO-1 at the inner versus the outer edge of GJs was not observed at ICDs from hearts expressing the DN-ZO-1, demonstrating that the mutant sequence disrupted normal ZO-1 Cx43 association at the ICD.
The second novel finding reported here is that the regulation of GJ localization by ZO-1 is not exclusive to the direct ZO-1-Cx43 interaction. The data presented indicate that ZO-1 regulation of GJ localization at sites of cell-cell interaction is not related to a direct interaction with the GJ and likely occurs via an effect on the AJ. We further demonstrated that the loss of Cx43 GJ localization at cell borders occurred downstream of AJ disruption by DN-ZO-1. It is long established that ZO-1 interaction with the AJ complex is necessary for the “strong state” of adhesion that occurs in sheet-like junctions such as those at the ICD or in polarized epithelia (23). The loss of ZO-1 association with the cadherin multiprotein complex and cytoplasmic internalization as demonstrated here in response to DN-ZO-1 (Fig. 2G) is characteristic with a “weak state” of AJ adhesion. This state is exemplified by small punctate cell-cell contacts such as those found between fibroblasts (22). Our immunolabeling and coimmunoprecipitation results show a dissociation of N-cadherin and ZO-1 at AJs and smaller N-cadherin-positive punctae at myocyte-myocyte contacts in the presence of DN-ZO-1. These changes are consistent with a reduction in the “strong state” of adhesion. Those AJs that persist in DN-ZO-1-expressing myocytes lack colocalized ZO-1 and appear to resemble “weak state” AJs (22).
A downstream dependence of GJ stability on the AJ “strong state” may have implications for differentiation of the ICD during maturational growth of the heart. In 1997, our group showed that the assembly of AJs into ICDs during postnatal ventricular development preceded that of GJs (2). Based on this observation, we hypothesized that the maintenance of disk-localized GJs was related to their physical proximity to AJs (2). ICD differentiation over postnatal development was proposed to result from a process that involved the retention of GJs at AJ-rich regions of sarcolemma (i.e., at ICDs) over the loss of GJs from AJ-sparse regions at lateral membranes. Consistent with this hypothesis, we show in the present report that a constitutive expression of the AJ-destabilizing molecule DN-ZO-1 from birth in the ventricle leads to large increases in lateralized GJs (Fig. 6).
The disruption of cadherin-mediated contacts and GJs similar to that seen in postnatal development is recapitulated in certain myocardial pathologies, particularly those diseases associated with arrhythmia (34, 35). The lateralization of Cx43 GJs to AJ-sparse regions of membrane has been reported in myocardial infarct border tissue and zones of myofiber disarray in patients with hypertrophic cardiomyopathy (35). Similar to our observations with DN-ZO-1, Hertig and coworkers (18) found that the expression of a DN-N-cadherin in rat myocytes resulted in the disruption of GJ contacts between myocytes (18). Additionally, in studies by Luo and Radice (28), the cardiac-specific loss of the N-cadherin gene in mice showed reduced Cx43 GJs (28). Subsequent work by the same group indicated that these N-cadherin-deficient mice also died of ventricular arrhythmias (26).
In humans, it is known that diseases associated with AJ mutations lead to electrical disturbances. Rare hereditable syndromes such as Carvajal syndrome and Naxos disease that are marked by mutation of desmosomal proteins (34) are characterized by arrhythmias and a profound disruption of myocyte GJs. These disruptions include reduced Cx43 expression and cytoplasmic internalization of both GJs and desmosomal components. Additionally, the lateralization of Cx43 GJs to AJ-sparse regions of membrane has been reported in myocardial infarct border tissue and zones of myofiber disarray in patients with hypertrophic cardiomyopathy (35).
Our data in myocytes and in the heart using DN-ZO-1 are consistent with previous results, demonstrating that ZO-1 truncations, including a sequence nearly identical to DN-ZO-1, cause a disruption of AJs between corneal epithelial cells (33) and induced delayed AJ formation in another epithelial cell line (22). An older report using a ZO-1 mutant protein similar to the one used in our study (i.e., DN-ZO-1) yielded disparate results (40). Consistent with our data, Toyofuku and coworkers (40) showed a loss of Cx43 from cell-cell contacts following the expression of a DN-ZO-1-like sequence. However, this early investigation also reported that the ZO-1-truncation mutant caused no detectable change in AJ organization in HEK293 cells. These data stand in contrast to our results in myocytes and those of Ryoem et al. in an epithelial cell line derived from adult cornea (33). The reason for the discrepancy is not clear; however, the most obvious difference between these studies is the cell types involved. HEK293 cells are a weakly polarized cell line derived from human embryonic kidney, which do not express tight junctions (24). The level of differentiation of intercellular junctions in this embryonal cell line is low relative to polarized epithelial lines and neonatal myocytes.
Here we show that the targeting of PDZ2 alone is insufficient to destabilize AJs. Myocytes were treated with a PDZ2-binding peptide designed to inhibit the interaction between the Cx43 CT and ZO-1 (supplementary Fig. 1). This inhibitory peptide caused no change in AJ distribution or ZO-1 colocalization at AJs between cultured NRVMs (supplementary Fig. 1). In previous reports, we have demonstrated that this PDZ2-binding peptide increases GJ size and showed no evidence of inducing a redistribution of Cx43 GJs from the membrane to the cytoplasm in either Cx43-expressing HeLa cells or NRVMs (20). Indeed, consistent with the study of Maass and coworkers (29), rather than causing GJ internalization, we observed that the targeting of the ZO-1 PDZ2 domain by the peptide resulted in an increase in membrane-localized pools of Cx43 in GJs (20), i.e., the promotion of GJ stability at cell membranes rather than the destablization and cytoplasmic internalization observed here in response to DN-ZO-1. Therefore, it appears unlikely that DN-ZO-1 mediates its effects on GJ localization at myocyte-myocyte contacts via the disruption of PDZ2 function.
In conclusion, ZO-1 association with the N-cadherin multiprotein complex between myocytes is a key determinant of GJ localization at the ICD. Dynamic variance of membrane-associated guanylate kinase protein-protein interactions at the AJ appears to strongly influence the organization of GJs between myocardial cells. Further understanding of the regulation of ZO-1 dynamics at the AJ may provide insights into the disease mechanism of fatal cardiac arrhythmias.
GRANTS
This work was supported in part by National Institutes of Health Grants F30-HL-095320, HL-56728, HL-36059, and HD-39946 and American Heart Association Grant 09GRNT2261357.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
Supplementary Material
ACKNOWLEDGMENTS
We thank Cheng Gang for assistance in generating and characterizing the DN-ZO-1 adenovirus. The excellent technical assistance of Ursula Kelly and Yuhua Zhang is acknowledged with gratitude.
REFERENCES
- 1. Anderson JM, Fanning AS, Lapierre L, Van Itallie CM. Zonula occludens (ZO)-1 and ZO-2: membrane-associated guanylate kinase homologues (MAGuKs) of the tight junction. Biochem Soc Trans 23: 470–475, 1995. [DOI] [PubMed] [Google Scholar]
- 2. Angst BD, Khan LU, Severs NJ, Whitely K, Rothery S, Thompson RP, Magee AI, Gourdie RG. Dissociated spatial patterning of gap junctions and cell adhesion junctions during postnatal differentiation of ventricular myocardium. Circ Res 80: 88–94, 1997. [DOI] [PubMed] [Google Scholar]
- 3. Barker RJ, Price RL, Gourdie RG. Increased association of ZO-1 with connexin43 during remodeling of cardiac gap junctions. Circ Res 90: 317–324, 2002. [DOI] [PubMed] [Google Scholar]
- 4. Bruce AF, Rothery S, Dupont E, Severs NJ. Gap junction remodeling in human heart failure is associated with increased interaction of connexin43 with ZO-1. Cardiovasc Res 77: 757–765, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Christensen G, Minamisawa S, Gruber PJ, Wang Y, Chien KR. High-efficiency, long-term cardiac expression of foreign genes in living mouse embryos and neonates. Circulation 101: 178–184, 2000. [DOI] [PubMed] [Google Scholar]
- 6. Delmar M. The intercalated disk as a single functional unit. Heart Rhythm 1: 12–13, 2004. [DOI] [PubMed] [Google Scholar]
- 7. Dupont E, Matsushita T, Kaba RA, Vozzi C, Coppen SR, Khan N, Kaprielian R, Yacoub MH, Severs NJ. Altered connexin expression in human congestive heart failure. J Mol Cell Cardiol 33: 359–371, 2001. [DOI] [PubMed] [Google Scholar]
- 8. Fanning AS, Jameson BJ, Jesaitis LA, Anderson JM. The tight junction protein ZO-1 establishes a link between the transmembrane protein occludin and the actin cytoskeleton. J Biol Chem 273: 29745–29753, 1998. [DOI] [PubMed] [Google Scholar]
- 9. Giepmans BN, Moolenaar WH. The gap junction protein connexin43 interacts with the second PDZ domain of the zona occludens-1 protein. Curr Biol 8: 931–934, 1998. [DOI] [PubMed] [Google Scholar]
- 10. Gonzalez-Mariscal L, Betanzos A, Avila-Flores A. MAGUK proteins: structure and role in the tight junction. Semin Cell Dev Biol 11: 315–324, 2000. [DOI] [PubMed] [Google Scholar]
- 11. Gonzalez-Mariscal L, Betanzos A, Nava P, Jaramillo BE. Tight junction proteins. Prog Biophys Mol Biol 81: 1–44, 2003. [DOI] [PubMed] [Google Scholar]
- 12. Gourdie RG, Green CR, Severs NJ. Gap junction distribution in adult mammalian myocardium revealed by an anti-peptide antibody and laser scanning confocal microscopy. J Cell Sci 99: 41–55, 1991. [DOI] [PubMed] [Google Scholar]
- 13. Gourdie RG, Green CR, Severs NJ, Thompson RP. Immunolabelling patterns of gap junction connexins in the developing and mature rat heart. Anat Embryol (Berl) 185: 363–378, 1992. [DOI] [PubMed] [Google Scholar]
- 14. Green CR, Peters NS, Gourdie RG, Rothery S, Severs NJ. Validation of immunohistochemical quantification in confocal scanning laser microscopy: a comparative assessment of gap junction size with confocal and ultrastructural techniques. J Histochem Cytochem 41: 1339–1349, 1993. [DOI] [PubMed] [Google Scholar]
- 15. Grikscheit K, Thomas N, Bruce AF, Rothery S, Chan J, Severs NJ, Dupont E. Coexpression of connexin 45 with connexin 43 decreases gap junction size. Cell Commun Adhes 15: 185–193, 2008. [DOI] [PubMed] [Google Scholar]
- 16. Gros D, Mocquard JP, Challice CE, Schrevel J. Formation and growth of gap junctions in mouse myocardium during ontogenesis: a freeze-cleave study. J Cell Sci 30: 45–61, 1978. [DOI] [PubMed] [Google Scholar]
- 17. Hartsock A, Nelson WJ. Adherens and tight junctions: structure, function and connections to the actin cytoskeleton. Biochim Biophys Acta 1778: 660–669, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hertig CM, Eppenberger-Eberhardt M, Koch S, Eppenberger HM. N-cadherin in adult rat cardiomyocytes in culture. I. Functional role of N-cadherin and impairment of cell-cell contact by a truncated N-cadherin mutant. J Cell Sci 109: 1–10, 1996. [DOI] [PubMed] [Google Scholar]
- 19. Herve JC, Bourmeyster N, Sarrouilhe D, Duffy HS. Gap junctional complexes: from partners to functions. Prog Biophys Mol Biol 94: 29–65, 2007. [DOI] [PubMed] [Google Scholar]
- 20. Hunter AW, Barker RJ, Zhu C, Gourdie RG. Zonula occludens-1 alters connexin43 gap junction size and organization by influencing channel accretion. Mol Biol Cell 16: 5686–5698, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hunter AW, Jourdan J, Gourdie RG. Fusion of GFP to the carboxyl terminus of connexin43 increases gap junction size in HeLa cells. Cell Commun Adhes 10: 211–214, 2003. [DOI] [PubMed] [Google Scholar]
- 22. Ikenouchi J, Umeda K, Tsukita S, Furuse M. Requirement of ZO-1 for the formation of belt-like adherens junctions during epithelial cell polarization. J Cell Biol 176: 779–786, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Imamura Y, Itoh M, Maeno Y, Tsukita S, Nagafuchi A. Functional domains of alpha-catenin required for the strong state of cadherin-based cell adhesion. J Cell Biol 144: 1311–1322, 1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Inai T, Sengoku A, Hirose E, Iida H, Shibata Y. Freeze-fracture electron microscopic study of tight junction strands in HEK293 cells and MDCK II cells expressing claudin-1 mutants in the second extracellular loop. Histochem Cell Biol 131: 681–690, 2009. [DOI] [PubMed] [Google Scholar]
- 25. Itoh M, Nagafuchi A, Moroi S, Tsukita S. Involvement of ZO-1 in cadherin-based cell adhesion through its direct binding to alpha catenin and actin filaments. J Cell Biol 138: 181–192, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kostetskii I, Li J, Xiong Y, Zhou R, Ferrari VA, Patel VV, Molkentin JD, Radice GL. Induced deletion of the N-cadherin gene in the heart leads to dissolution of the intercalated disc structure. Circ Res 96: 346–354, 2005. [DOI] [PubMed] [Google Scholar]
- 27. Laing JG, Manley-Markowski RN, Koval M, Civitelli R, Steinberg TH. Connexin45 interacts with zonula occludens-1 and connexin43 in osteoblastic cells. J Biol Chem 276: 23051–23055, 2001. [DOI] [PubMed] [Google Scholar]
- 28. Luo Y, Radice GL. Cadherin-mediated adhesion is essential for myofibril continuity across the plasma membrane but not for assembly of the contractile apparatus. J Cell Sci 116: 1471–1479, 2003. [DOI] [PubMed] [Google Scholar]
- 29. Maass K, Shibayama J, Chase SE, Willecke K, Delmar M. C-terminal truncation of connexin43 changes number, size, and localization of cardiac gap junction plaques. Circ Res 101: 1283–1291, 2007. [DOI] [PubMed] [Google Scholar]
- 30. Oxford EM, Musa H, Maass K, Coombs W, Taffet SM, Delmar M. Connexin43 remodeling caused by inhibition of plakophilin-2 expression in cardiac cells. Circ Res 101: 703–711, 2007. [DOI] [PubMed] [Google Scholar]
- 31. Palatinus JA, Gourdie RG. Xin and the art of intercalated disk maintenance. Am J Physiol Heart Circ Physiol 293: H2626–H2628, 2007. [DOI] [PubMed] [Google Scholar]
- 32. Pieperhoff S, Borrmann C, Grund C, Barth M, Rizzo S, Franke WW. The area composita of adhering junctions connecting heart muscle cells of vertebrates. VII. The different types of lateral junctions between the special cardiomyocytes of the conduction system of ovine and bovine hearts. Eur J Cell Biol 89: 365–378, 2010. [DOI] [PubMed] [Google Scholar]
- 33. Ryeom SW, Paul D, Goodenough DA. Truncation mutants of the tight junction protein ZO-1 disrupt corneal epithelial cell morphology. Mol Biol Cell 11: 1687–1696, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Saffitz JE. Dependence of electrical coupling on mechanical coupling in cardiac myocytes: insights gained from cardiomyopathies caused by defects in cell-cell connections. Ann NY Acad Sci 1047: 336–344, 2005. [DOI] [PubMed] [Google Scholar]
- 35. Sepp R, Severs NJ, Gourdie RG. Altered patterns of cardiac intercellular junction distribution in hypertrophic cardiomyopathy. Heart 76: 412–417, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Shibata Y, Nakata K, Page E. Ultrastructual changes during development of gap junctions in rabbit left ventricular myocardial cells. J Ultrastruct Res 71: 258–271, 1980. [DOI] [PubMed] [Google Scholar]
- 37. Simpson DG, Terracio L, Terracio M, Price RL, Turner DC, Borg TK. Modulation of cardiac myocyte phenotype in vitro by the composition and orientation of the extracellular matrix. J Cell Physiol 169: 89–105, 1994. [DOI] [PubMed] [Google Scholar]
- 38. Smith JH, Green CR, Peters NS, Rothery S, Severs NJ. Altered patterns of gap junction distribution in ischemic heart disease. An immunohistochemical study of human myocardium using laser scanning confocal microscopy. Am J Pathol 139: 801–821, 1991. [PMC free article] [PubMed] [Google Scholar]
- 39. Stevenson BR, Siliciano JD, Mooseker MS, Goodenough DA. Identification of ZO-1: a high molecular weight polypeptide associated with the tight junction (zonula occludens) in a variety of epithelia. J Cell Biol 103: 755–766, 1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Toyofuku T, Yabuki M, Otsu K, Kuzuya T, Hori M, Tada M. Direct association of the gap junction protein connexin-43 with Zo-1 in cardiac myocytes. J Biol Chem 273: 12725–12731, 1998. [DOI] [PubMed] [Google Scholar]
- 41. Toyofuku T, Akamatsu Y, Zhang H, Kuzuya T, Tada M, Hori M. c-Src regulates the interaction between connexin-43 and ZO-1 in cardiac myocytes. J Biol Chem 276: 1780–1788, 2001. [DOI] [PubMed] [Google Scholar]
- 42. Yao JA, Hussain W, Patel P, Peters NS, Boyden PA, Wit AL. Remodeling of gap junctional channel function in epicardial border zone of healing canine infarcts. Circ Res 92: 437–443, 2003. [DOI] [PubMed] [Google Scholar]
- 43. Zhu C, Barker RJ, Hunter AW, Zhang Y, Jourdan J, Gourdie RG. Quantitative analysis of ZO-1 colocalization with Cx43 gap junction plaques in cultures of rat neonatal cardiomyocytes. Microsc Microanal 11: 244–248, 2005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.