Abstract
The promise of pluripotent stem cells as a research and therapeutic tool is partly undermined by the technical challenges of generating and maintaining these cells in culture. Human embryonic stem cells (hESCs) are exquisitely sensitive to culture conditions, and require constant signaling by growth factors and cell–cell and cell–matrix interactions to prevent apoptosis, senescence, and differentiation. Previous work from our laboratory demonstrated that overexpression of the prosurvival gene BCL2 in mouse embryonic stem cells overrode the requirement of serum factors and feeder cells to maintain mESCs in culture. To determine whether this prosurvival gene could similarly protect hESCs, we generated hESC lines that constitutively or inducibly express BCL2. We find that BCL2 overexpression significantly decreases dissociation-induced apoptosis, resulting in enhanced colony formation from sorted single cells, and enhanced embryoid body formation. In addition, BCL2-hESCs exhibit normal growth in the absence of serum, but require basic fibroblast growth factor to remain undifferentiated. Furthermore, they maintain their pluripotency markers, form teratomas in vivo, and differentiate into all three germ layers. Our data suggest that the BCL2 signaling pathway plays an important role in inhibiting hESC apoptosis, such that its overexpression in hESCs offers both a survival benefit in conditions of stress by resisting apoptosis and obviates the requirement for serum or a feeder layer for maintenance.
Keywords: embryonic stem cell survival, viability
Embryonic development proceeds through a series of tightly regulated and precisely timed fate decisions to generate the complex tissues and organs that comprise the developing organism. The cells that form the early embryo are sensitive to improper signals or conditions that could derail the developmental program. Embryonic stem cells, derived from the inner cell mass of blastocysts, mirror this sensitivity and are prone to apoptosis, senescence, or differentiation when the in vitro culture conditions are less than optimal. As a result, human embryonic stem cells (hESCs) and their progeny, including tissue stem cells, are technically difficult to maintain in culture, impeding their use in studying intermediate developmental states. In particular, hESCs undergo significant cell death upon dissociation into single cells, which has been a major hurdle for their differentiation, rapid expansion, and genetic manipulation (1, 2). Additionally, hESCs and their early progeny are vulnerable to apoptosis upon sorting through a flow cytometer, hindering efforts to isolate rare populations. It is thought that a combination of soluble growth factors and cell–cell or cell–matrix interactions acts in concert to maintain hESCs in a viable and pluripotent state, and these favorable interactions may be disrupted during single-cell dissociation or flow cytometry (3).
Human ESC media has been carefully formulated to provide a myriad of prosurvival signals. The standard media used for maintenance of hESCs on mouse embryonic fibroblasts (MEFs) is supplemented with basic fibroblast growth factor (bFGF) and knockout replacement serum (KOSR), a proprietary formulation containing a variety of growth factors for hESC growth (4). Efforts have been made to develop defined culture systems to eliminate the need for animal-derived products in the media and the supporting feeder-cell layer for the maintenance and expansion of hESCs (5–7). Although several defined media have optimized the growth and maintenance condition for hESCs without the requirement of a feeder layer, these media still rely on bFGF supplementation. FGF signaling plays a critical role in the survival, self-renewal, proliferation, and maintenance of the undifferentiated hESC state in vitro (8–14).
Despite the optimization of culture media for growth and survival, apoptosis upon manipulation of hESCs remains a major problem. Hence, several recent studies have identified specific prosurvival factors to enhance hESC survival after dissociation and to facilitate cloning efficiency. A selective Rho-associated kinase (ROCK) inhibitor (Y-27632) has been shown to markedly diminish dissociation-induced apoptosis of hESCs (15). Other compounds have been reported to enhance hESC survival by stabilizing E-cadherin signaling, one of the key signals disrupted during single cell dissociation (3).
Many of the prosurvival signals converge at the BCL2 pathway. The BCL2 family of proteins contains three major subfamilies: a proapoptotic family (e.g., BAX, BAK, BOK, and so forth), a prosurvival family (e.g., BCL2, BCL-XL, MCL1), and the BH3-only subfamily (e.g., BAD, BID, BIP, PUMA, NOXA) (16). Growth factors continuously stimulate tyrosine kinase activity to suppress the proapoptotic and BH3-only family members, and thus prevent initiation of apoptosis (17, 18). Enforced expression of the 26-kDa BCL2 antiapoptotic protein has been reported to result in enhanced survival and proliferation in several different systems (16, 19, 20). We demonstrated that mouse embryonic stem-cell (mESC) lines expressing the human BCL2 transgene could self-renew continuously under serum-free and feeder cell-free conditions (21), and improved efforts to identify and isolate mouse ES-derived HSC (22).
Because signaling pathways that interact with the BCL2 pathway play a major role in the survival of hESCs, we evaluated the effect of overexpression of BCL2 on growth and survival of hESCs. We established hESC clones that constitutively or transiently express human BCL2. After serial passages for more than 30 generations, these BCL2-hESC lines continue to maintain their pluripotency, ability to differentiate in vitro, and potential to form teratomas in vivo. We show that BCL2-hESCs display increased viability following single-cell dissociation, cell sorting, and in cultures lacking feeders and serum factors. Furthermore, the enhanced survival of the BCL2-expressing cells in the absence of serum factors is partially a result of their resistance to apoptosis. Our results collectively demonstrate that the overexpression of BCL2 substantially promotes hESC survival without compromising their self-renewal and developmental potency.
Results
Expression of BCL2 in hESCs.
To determine whether overexpression of human BCL2 could improve the viability of hESCs in conditions of stress, we generated hESC lines that either constitutively or inducibly express BCL2 (Figs. S1 and S2 ). Constitutive BCL2-expressing hESC lines were generated using lentiviral constructs expressing BCL2 linked to GFP (C306) or CFP (C342), driven by the constitutively active EF1α promoter (Fig. S1A). Alternatively, Tet-inducible BCL2 hESC lines were generated, in which the expression of BCL2 is induced by exogenous addition of doxycycline (DOX) (Fig. S2). Expression of GFP/CFP was confirmed by microscopy, and BCL2 expression was confirmed by intracellular staining (Fig. S1 B and C). The primary hESC line induced to constitutively express BCL2, and used for all subsequent experiments, was generated via the introduction of construct C306; it is hereafter denoted as “BCL2-hESC.” We estimated the copy number of the primary BCL2-hESC line by quantitative real-time PCR of genomic DNA using primers that amplify the endogenous locus and lentiviral construct equally (Fig. S1D). Based on this assay, we estimated that 28 copies of the BCL2 construct were present in the BCL2-hESC line. Using quantitative real-time RT-PCR, we examined mRNA expression of BCL2 in this line, and found an over 4,600-fold increase in BCL2 transcripts compared with GFP-expressing hESC line, although this high fold-increase is partially because of the fact that hESCs normally express very low, if any, levels of BCL2 (Fig. S1E). For BCL2-inducible lines, we found that the maximal BCL2 expression was exhibited in the first 24 h after DOX addition (Fig. S2), and decreased gradually over the course of 5 d following DOX withdrawal.
BCL2-hESC lines have been serially passed for more than 30 generations yet still maintain the characteristic colony morphology and normal karyotype (Fig. S3A). Furthermore, BCL2-hESCs express the pluripotency markers Oct3/4, Nanog, and SSEA4 (Fig. S3B). When BCL2-hESCs were injected subcutaneously or under the kidney capsule of immunocompromised mice, they generated complex teratomas consisting of all three germ layers (Fig. S3 C–E). Additionally, we observed no defects in the ability of BCL2-hESCs to differentiate to either the cardiomyocyte (Fig. S4 and Movie S1) or hematopoietic/endothelial lineages (Fig. S5). Thus, to the best of our knowledge, the constitutive overexpression of BCL2 in hESC lines affects neither their pluripotency nor differentiation potential.
BCL2-hESC Cells Exhibit Improved Colony Formation Efficiency After Sorting and Enhanced Survival During Embryoid Body Formation.
The efficiency of colony formation from a single hESC is notoriously poor, particularly after sorting via flow cytometer. The poor survival is attributed partially to disruption of favorable cell–cell and cell–matrix interactions that prevent apoptosis. When single wild-type hESCs were FACS-sorted directly into the wells of a 96-well plate coated with Matrigel, we noted one colony out of ∼100 plated cells (1 in 100), which improved to one colony per 60 cells (1 in 60) in the presence of ROCK-inhibitor (Fig. 1 A and B). However, when BCL2-hESCs were sorted onto matrigel-coated wells in the absence or presence of ROCK inhibitor, we observed colony-formation efficiencies of 1 in 26 cells and 1 in 21 cells, respectively, a three- to fourfold improvement over wild-type hESCs. Similarly, when sorted onto MEFs, wild-type hESCs yielded colony-forming efficiencies of 1 in 10, which improved to 1 in 5 with ROCK inhibitor (Fig. 1 C and D). The efficiency of colony formation of BCL2-hESC on MEFs increased to 1 in 4.5 and 1 in 2 cells plated in the absence or presence of ROCK-inhibitor, respectively. Taken together, BCL2 expression improved colony-formation from sorted single cells between two- and fivefold, depending on the precise plating conditions.
Fig. 1.
Improvement in colony and EB formation with BCL2-hESCs. (A–D) Colony formation from FACS-sorted wild-type (red line), GFP- (green line), and BCL2- (blue line) hESCs when plated onto matrigel (A and B) or MEFs (C and D) in the absence (A and C) or presence (B and D) of ROCK inhibitor. Colony formation efficiency was plotted versus cells plated per well. Cell counts were capped at 50 colonies per well. (E and F) EBs were generated from RFP-hESCs (E) and BCL2-hESCs (F) after single-cell dissociation. Shown are brightfield (Left) and RFP/GFP fluorescence (Right) of hESC clusters in microwells. (G) Chimeric EBs generated from BCL2-hESCs and RFP-hESCs, mixed at multiple ratios. Plotted is the percentage of BCL2-hESCs within the EBs at 0, 1, and 4 d after generation, determined by FACS. (H) Absolute number of live hESC-derived cells per EB of GFP-hESCs (orange), constitutively expressed BCL2-hESCs (red), and inducible BCL2-hESCs in the absence (green) or presence (blue) of DOX. Two-thousand hESCs were seeded per well (40 wells per line) at day 0, then pooled at day 1. Half the EBs were harvested and counted at day 1, and the other half at day 4. Error bars are SD (n = 3) of three independent experiments.
A large number of cells also undergo cell death in the initial stages of embryoid body (EB) formation, particularly when EBs are formed from single-cell aggregation. To determine whether BCL2-hESCs exhibit improved survival during EB formation, BCL2-hESCs were mixed with wild-type hESCs that constitutively express red fluorescent protein (RFP). BCL2-hESCs and RFP-hESCs were dissociated into single cells and were mixed at different ratios to form chimeric EBs. EBs formed from 100% BCL2-hESC cells exhibited greater uniformity, were symmetrically spherical with distinct borders, and relatively few detached single cells were noted in the wells (Fig. 1 E and F). Conversely, RFP-hESC EBs were less uniform and more vulnerable to disruption upon transfer. Furthermore, when chimeric EBs generated from different mixtures of BCL2- and RFP-hESC were evaluated 24 h after EB formation, we found a significant increase in the ratio of BCL2- to RFP-hESCs within the EBs (Fig. 1G). This result was found to be caused by a combination of factors, including continued growth of BCL2-hESC cells and poor survival of RFP-hESCs, especially in the first 24 h of EB formation (Fig. 1H). In EBs formed from control hESCs alone, the total number of cells decreased by 45% after 24 h and returned to baseline after 4 d. However, the total number of cells in EBs formed from constitutive BCL2-hESCs increased by 13% and nearly doubled after 4 d. A similar trend was noted when EBs were formed from inducible BCL2-hESCs, but only when BCL2 expression was induced by DOX addition 24 h before EB formation (Fig. 1H). These results indicate that during the first 24 h after EB formation from single cells, massive cell death occurs that can be largely rescued by expression of BCL2.
BCL2-hESC Cells Exhibit Normal Growth Under Serum-Free Conditions.
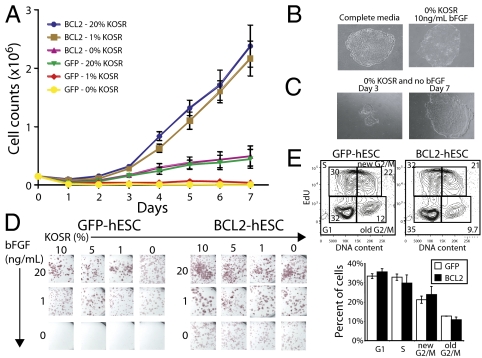
Conventional hESC media contains factors that provide prosurvival signals to hESCs. We have previously shown that mESCs that express BCL2 are able to survive in media lacking serum or feeders, the primary sources of these prosurvival signals (21). To determine whether BCL2 overexpression can similarly compensate for the absence of prosurvival signals in hESCs, parental and BCL2-hESC cells were cultured in feeder-free conditions (Matrigel) and with decreasing concentrations of KOSR. The growth kinetics were determined over the course of two passages. Unmanipulated hESCs required 20% KOSR for normal growth and failed to survive when KOSR was decreased to a concentration of 1% or removed entirely (Fig. 2A). However, BCL2-expressing hESCs grew normally in the absence of KOSR, and displayed increased growth kinetics in 20 and 1% KOSR. Thus, overexpression of BCL2 appears to override the requirement for KOSR in hESC culture.
Fig. 2.
Enhanced survival of BCL2-hESCs in serum-free media. (A) Growth kinetics of hESCs at different KOSR concentrations. Wild-type and BCL2-hESCs were plated on Matrigel and cultured in media with KOSR concentrations of 20, 1, and 0%, and cells were counted daily. Error bars are SD (n = 3). (B) Representative BCL2-hESC colonies grown in media with 20% KOSR (Left) and in 0% KOSR (Right). (C) Representative colony of BCL2-hESCs grown in media lacking both KOSR and bFGF. The same colony is shown at day 3 (Left) and day 7 (Right) after plating. (D) Alkaline phosphatase activity of BCL2-hESCs (Left) and wild-type hESCs (Right) cultured for two passages under different concentrations of KOSR and bFGF. (E) Cell-cycle analysis of GFP- (Upper Left plot and Lower graph, white bars) or BCL2-hESCs (Upper Right plot and Lower graph, black bars) cultured in complete hESC media. EdU was added 2 h before analysis. Shown are the percentages of cells at each cell cycle stage.
BCL2-hESC colonies appeared morphologically normal, whether grown in “complete” media—containing the standard concentrations of KOSR (20%) and bFGF (10 ng/mL)—or in media lacking KOSR (0%) but containing normal bFGF (Fig. 2B). BCL2-hESCs expanded independently of KOSR and feeders but required bFGF to form tightly packed colonies with indistinct cell boundaries, a hallmark of undifferentiated hESCs. When bFGF was withdrawn from the serum-free media, the hESC colonies survived for a couple of passages before extensive differentiation was noted (Fig. 2C). Additionally, BCL2-hESCs cultured in decreasing KOSR and bFGF concentrations after one passage exhibited alkaline phosphatase activity (a marker of pluripotent cells of embryonic origin), although parental hESC cells lost alkaline phosphatase activity upon KOSR withdrawal (Fig. 2D).
To determine whether the increased growth kinetics of BCL2-hESCs is because of increased cycling, we cultured GFP- and BCL2-hESCs in the presence of a BrdU analog, EdU, and measured the rate of EdU incorporation and DNA content after 2 h (Fig. 2E). We found that both hESC lines incorporated EdU at an equivalent rate, indicating that overexpression of BCL2 does not cause hESCs to cycle faster. We also examined 11 different BCL2-hESC clones, bearing a range of copy numbers from 1 to 10, to determine the relationship between BCL2 copy number and expression level to viability in minimal media, and found a general trend of enhanced survival in clones with higher copy numbers (Fig. S6). Taken together, these results suggest that BCL2-hESCs can grow and expand in the absence of KOSR supplementation in the medium, but require bFGF to prevent differentiation. Furthermore, the increase in the growth kinetics noted in BCL2-hESCs is not due to an augmented cell cycle activity.
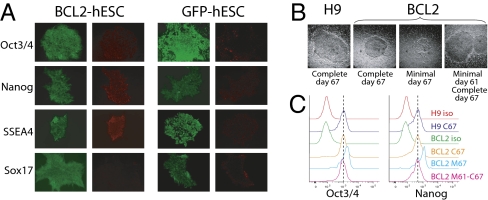
To determine whether culture conditions devoid of serum affected hESC pluripotency, we examined pluripotency marker expression in BCL2-hESCs cultured for three passages in the absence of knockout serum. We found that BCL2-hESCs maintained high expression of Oct3/4, Nanog, and SSEA4 (Fig. 3A), similar to BCL2-hESCs cultured in complete media (Fig. S3B). Conversely, control hESCs lost pluripotency marker expression after three passages in minimal media. To determine whether BCL2-hESCs remain pluripotent after long-term culture without serum, we cultured control and BCL2-hESCs in complete media, and in media lacking serum for 12 passages (67 d). Control hESCs did not survive beyond a few passages in the absence of serum, whereas BCL2-hESCs grew normally for several passages, although at later passages their growth kinetics began to slow and their colonies remained small (Fig. 3B). Despite their reduced growth rate, BCL2-hESCs continued to express high levels of Oct3/4 and Nanog proteins (Fig. 3C). In addition, they rapidly returned to normal colony size and growth kinetics when the same colonies were cultured back onto complete media. These data indicate that BCL2-hESCs retain their pluripotency expression profiles even when deprived of serum factors for over 2 mo.
Fig. 3.
Human ESCs overexpressing BCL2 maintain their pluripotency markers when cultured for short-term (3 passages) or long-term (12 passages) in serum-free media. (A) Immunofluorescence staining of BCL2-hESCs after three passages in minimal media containing no KOSR with anti-Oct3/4, anti-Nanog, anti-SSEA4, and anti-Sox 17. Compared with control GFP-hESCs, BCL2 expressing hESCs had normal expression of Oct3/4, Nanog, and SSEA4. Absence of Sox 17 indicates lack of differentiation in the BCL2-hESC colonies. (B) Representative colonies of parental H9 and BCL2 expressing hESCs cultured on MEFs for 67 d (12 passages) in complete media, in minimal media lacking serum, or in minimal media for 61 d, then passaged into complete media for additional 6 d. Unmanipulated H9 cells in minimal media did not survive beyond three passages, and therefore were not analyzed. (C) Intracellular FACS profile of Oct3/4 (Left) and Nanog (Right) expression of hESC cultures at day 67. H9 and BCL2 isotype controls are shown in red and green lines, respectively. For ease of comparison, the crest of Oct3/4 and Nanog expression curves for the H9 culture in complete media are indicated by the dotted line.
BCL2 Partially Blocks Apoptosis in hESCs Cultured in Serum-Free Media.
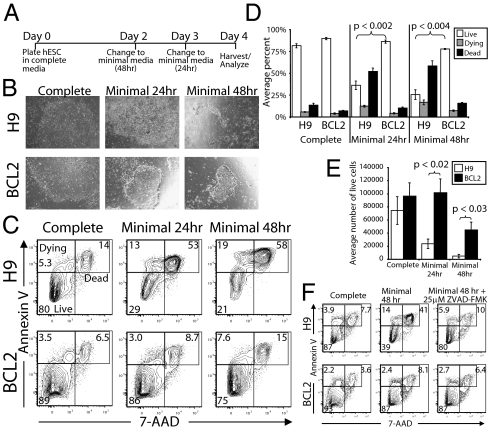
We hypothesized that the enhanced survival of BCL2-hESCs in serum-free media was caused by increased resistance to apoptosis. To determine whether BCL2 overexpression protects hESCs from apoptosis, we passaged BCL2- and control hESCs onto Matrigel-coated dishes in complete media (20% KOSR, 10 ng/mL bFGF), then after 2 to 3 d switched them into a minimal media with 0% KSR and 1 ng/mL of bFGF, and measured the frequency of apoptotic cells by Annexin V staining after 24 and 48 h in minimal media (Fig. 4). We found that control hESCs cultured in minimal media for 24 or 48 h rapidly underwent apoptosis and anoikis compared with cells cultured in complete media for the duration of the experiment (Fig. 4 B–D). However, BCL2-hESCs exhibited minimal apoptosis despite the lack of survival factors in minimal media, such that both the frequency and absolute number of live nonapoptotic cells remained near that of BCL2-hESCs cultured in complete media (Fig. 4 B–E).
Fig. 4.
Apoptosis analysis of BCL2-hESCs in minimal media. (A) Experimental design. Control and BCL2 hESCs were passaged from feeder plates onto Matrigel-coated wells in complete media. At 48 (day 2) or 24 (day 3) h before analysis, wells were changed into minimal (0% KOSR, 1 ng/mL bFGF) media for the 48 and 24 h time points, respectively. At day 4, all floating and attached cells were harvested and analyzed. (B) Photographs of hESC colonies taken at day 4 from control (H9, Upper) and BCL2 (Lower) wells in complete media (Left), or in minimal media 24 h (Center) or 48 h (Right) before analysis. (C) Apoptosis analysis of control and BCL2 hESCs, using Annexin V (y axis) and 7-AAD (x axis). Percentages of live cells (Annexin V−, 7-AAD−), dying cells (Annexin V+, 7AAD−), and dead cells (Annexin V+, 7AAD+) are shown. (D) Average percent of live (white), dying (gray), and dead (cells) from control and BCL2 hESCs in complete and minimal media. Error bars are SD (n = 3) and P values are calculated by the t test. (E) Average absolute number of live cells from control (white) and BCL2 (black) hESCs. Error bars are SD (n = 3) and P values are calculated by the t test. (F) Analysis of apoptosis of H9- and BCL2-hESCs treated with the caspase inhibitor ZVAD-FMK in minimal media. H9- and BCL2-hESCs were plated as described in A, but some wells were pretreated for 1 h with 25 μM ZVAD-FMK before changing to minimal media, then retreated with an additional 25 μM ZVAD-FMK after each media change. Human ESCs were harvested 48 h after changing to minimal media, and analyzed for Annexin V binding (y axis) and 7-AAD incorporation (x axis). Values shown are the percent of live, dying, and dead cells.
To determine if the apoptosis that results from the removal of KOSR and reduction of bFGF is caspase-dependent, we pretreated wild-type hESCs and BCL2-hESCs with the caspase-inhibitor ZVAD-FMK at 25 μM 1 h before changing to minimal media, and measured apoptosis after 48 h (Fig. 4F). We found that wild-type hESCs cultured in minimal media containing ZVAD-FMK were more resistant to apoptosis than untreated cells, indicating that the growth factor starvation-induced apoptosis is caspase-dependent. However, treatment with ZVAD-FMK only reduced apoptosis in wild-type hESCs, as BCL2-hESCs appeared unaffected by ZVAD-FMK. BCL2 hESCs showed higher viability than the ZVAD-FMK treated H9 cells.
To eliminate the possibility that the apoptosis resistance is an integration artifact of this particular BCL2-hESC line, we repeated these experiments using an inducible BCL2 hESC line (Fig. S7). BCL2 expression was induced by addition of DOX 24 h before changing to minimal media, and thus we could compare the effects of BCL2 expression within the same cell line (Fig. S7A). Like their constitutively-expressed counterparts, BCL2-induced cells were more resistant to apoptosis in minimal media compared with uninduced cells (Fig. S7 B–E). Taken together, these results indicate that the increased growth kinetics of BCL2-expressing hESCs is likely in large part the result of an increased resistance to apoptosis when critical survival factors contained in KOSR are reduced or removed.
Discussion
We have previously shown that transgenic mESCs expressing human BCL2 continue to self-renew in serum- and feeder-free conditions when supplemented with leukemia inhibitory factor (21). These studies suggest that leukemia inhibitory factor and BCL2 overexpression are sufficient to maintain and expand mESCs in culture. The data summarized here is a logical extension of the previous study, where we show that BCL2 overexpression, in the presence of bFGF, is sufficient to maintain hESCs in an undifferentiated state for prolonged periods and offers a survival benefit in conditions of stress by resisting apoptosis. Although differentiation to only the mesoderm-derived cardiomyocyte, endothelial, and hematopoietic lineages were specifically tested, BCL2-hESCs can form all three germ layers upon teratoma formation.
It is known that growth factors and cytokines present in KOSR can stimulate multiple receptor tyrosine kinase signaling, leading to activation of a variety of tyrosine kinases, such as AKT and ERK, which can directly inhibit proapoptotic BCL2 family members (23). In the absence of serum factors, those proapoptotic proteins may accumulate and override the activity of BCL2 and the other prosurvival family members. The overexpression of BCL2 proteins may increase the threshold level of proapoptotic signaling required for initiating apoptosis, and thereby prevent apoptosis when other survival signals are removed. This theory may explain our finding that the protective effect of BCL2 correlates with its expression level. That BCL2 overexpression alone may obviate the requirement for KOSR suggests that the primary function of the myriad growth factors contained in KOSR is to provide survival signals, and that the BCL2 pathway lies at the hub of those signals.
Although we found KOSR dispensable in maintaining BCL2-hESC lines, bFGF was not. When bFGF was removed from the culture media, BCL2-hESCs continued to proliferate but gradually differentiated. Accumulating evidence suggests that FGF signaling plays a central role in sustaining hESCs in an undifferentiated state by providing both survival and antidifferentiation signals (8, 10, 11, 24). Our results suggest that although BCL2 can replace the prosurvival function of bFGF, it cannot supplant the antidifferentiation signal that bFGF provides. This finding may partially explain why BCL2-hESCs can grow in low concentrations of bFGF (1 ng/mL) but not in its complete absence. Analogous to the requirement for leukemia inhibitory factor in the serum-free and feeder-free culture of BCL2-expressing mESCs, bFGF appears to be required for maintaining the pluripotency of hESCs. Additionally, signaling from TGF-β family members has been demonstrated to play an integral role in hESC self-renewal, maintenance of pluripotency, and regulation of differentiation (25). Although activities of FGF and TGF-β superfamily may partially overlap, each is necessary to sustain hESCs in an undifferentiated state. Our data suggest that short-term culture conditions in which KOSR is lacking, overexpression of BCL2 in conjunction with the presence of bFGF in the medium may partially override TGF-β signaling for maintenance of pluripotency.
Human ESCs are especially vulnerable to apoptosis when dissociated into single cells. The molecular mechanisms that underlie this sensitivity are not completely understood. Recently, Xu et al. reported the essential role of E-cadherin signaling for hESC survival: E-cadherin-mediated cell–cell interactions constitute an essential regulatory mechanism that controls hESC survival and self-renewal (3). Dissociation of hESCs into single cells disrupts this signal, perhaps leading to cell death. Here, we show that BCL2 expression offers a significant survival benefit for hESCs upon single-cell dissociation and cell sorting. Additionally, we have shown that EB formation from single-cell suspensions and their subsequent growth is considerably more efficient when BCL2 is overexpressed. As the survival benefit conferred by BCL2 is most obvious during the first day of EB formation, when individual cells must aggregate and join, it is possible that BCL2’s benefit in EB formation is caused by its protection of hESCs when they exist as single cells.
Human ESC lines that overexpress BCL2, either constitutively or transiently, have several practical uses: first, BCL2 improves the viability of hESCs in protocols that require single cell dissociation, such as (i) clonal isolation following genetic modification, (ii) EB formation, (iii) single-cell differentiation protocols, and (iv) culture of populations of hESCs after sorting by magnetic-activated cell sorting or flow cytometry. Second, BCL2 overexpression allows for the efficient differentiation of hESCs in protocols that require the removal of survival factors. In support of this notion, BCL2 overexpression was shown to enhance mESC-derived hematopoietic differentiation (22). Finally, BCL2-hESC lines can be cultured in serum- and feeder-free conditions to assess the effect of individual growth factors, cytokines, and small molecules on the proliferation and differentiation of hESCs, without concern for survival.
Although increased expression of BCL2, whether by transgene or translocation, appears to predispose some cell types to neoplasia, overexpression alone is not oncogenic (26). In fact, we previously showed that chimeric mice generated from mouse ES cells expressing human BCL2 appear normal and remain tumor-free despite ubiquitous expression of BCL2 (21). Nonetheless, the constitutive expression of an antiapoptotic gene in an hESC line may gradually alter its epigenetic state over time. Although the constitutive BCL2-expressing hESC lines used in this study have remained healthy and pluripotent throughout the many passages they have undergone, there remains the possibility that gradual genomic alterations that usually signal apoptosis may accumulate over time and compromise the potential of these lines. To circumvent this limitation, we generated an inducible system that allows for temporal expression of BCL2 only during the brief periods where viability is most challenged. Specifically, the addition of DOX the day before challenge ensures maximal BCL2 protection when needed most. BCL2 expression returns to baseline ∼5 d after removal of DOX (Fig. S2D), thereby preventing any long-term effects that may occur. Although BCL2 overexpression in pluripotent stem cells has a variety of applications, the genetic modification required for expression and the uncertainty of genomic instability from permanent inhibition of apoptosis may preclude their use in clinical applications. To that end, we have found that the caspase inhibitor ZVAD-FMK can partially protect hESCs from apoptosis when survival factors are removed. Although this protection does not appear to be as robust as BCL2 overexpression, this inhibitor can be safely added to cultures without genetic modification or use of animal products.
In summary, overexpression of BCL2 enables hESCs to expand and proliferate under serum- and feeder-free conditions, and this effect was mediated primarily by increased resistance to apoptosis. Additionally, we have demonstrated a significant survival benefit when BCL2-hESCs are dissociated into single cells or formed into EBs. Furthermore, BCL2-hESCs maintain their pluripotency, are able to differentiate to all three germ layers, and display a normal karyotype. We find that BCL2 overexpression can be used to enhance hESC viability in any assay performed under harsh conditions. Our data suggest that numerous survival signals are required for hESC culture, but these signals intersect at the BCL2 pathway and thus overexpression of BCL2 can obviate the need for many of these signals, such as those provided by KOSR. Further studies are warranted to delineate the molecular mechanism by which BCL2 exerts its effect.
Materials and Methods
Flow Cytometry.
All flow cytometry was performed using a FACSAria 2 (BD Bioscience).
Intracellular staining.
Human ESCs were dissociated into single cells, then fixed in 2% paraformaldehyde, and permeabilized with 3% saponin. Human ESCs were stained with PE-conjugated mouse antihuman BCL2 antibody (Cat. no. MHBCL04, clone 100; Invitrogen) at 1:100 dilution. For intracellular Oct3/4 and Nanog stains, cells were first treated with Fixable Viability Dye eFluor450 according to the manufacturer's protocol (eBioscience; Cat. no. 65–0863), then stained with αOct3/4-PE, αNanog-PE, or isotype control PE at 1:10 dilutions.
Surface stains.
Single-cell suspensions of hESCs or derived products were stained in staining media (PBS with 2% FCS), on ice, for 15 min.
Cell cycle analysis.
The BrdU analog EdU was added to healthy hESC cultures at 10 μM for 2 h. Human ESCs were then dissociated into single cells and the cell cycle status analyzed using the Click-iT EdU flow cytometry assay kit (Cat. no. A10202; Invitrogen) according to the manufacturer's protocol.
Antibodies.
Clone numbers, catalog numbers, and conjugates of all antibodies used in this study are listed in Table S1.
Immunostaining and Immunohistochemistry.
Cells were fixed in 4% paraformaldehyde (Electron Microscopy Sciences) for 15 min, permeabilized with 0.1% Triton X-100 (Fisher Scientific) in PBS for 5 min, and then blocked with 1% goat serum for 15 min. Cells were incubated overnight with primary antibodies against Oct3/4, Nanog, SSEA4, Sox 17, troponin I, and β-myosin heavy-chain isoform, washed, and then incubated with goat anti-mouse Alexa 594, goat anti-mouse Alexa 488, or donkey anti-goat Alexa 594 secondary antibody for 1 h. Teratomas formed under the kidney capsule were fixed as described in SI Materials and Methods, and 6-μm sections were prepared for immunohistochemical analysis.
Cell Proliferation and Viability Assays.
To assess the effect of KOSR and bFGF on the growth of BCL2-expressing cells, the concentration of bFGF and KOSR were varied, respectively, to 5 ng/mL, 1 ng/mL, 0 ng/mL, and 1%, 0%. Wild-type and BCL2 hESCs were passaged onto Matrigel-coated wells in the different media formulations, and maintained in those media for two passages before their growth kinetics were evaluated. Cumulative cell counts at designated days were determined by dissociating the colonies into single cells using Trypsin 0.05% and manually counting. Cells were passaged at day 4.
Apoptosis Assays.
Human ESCs were passaged onto Matrigel-coated dishes in complete hESC media. At day 2 to 3 after plating, wells were washed in PBS, and then either complete (20% KSR, 10 ng/mL bFGF) or minimal (0% KSR, 1 ng/mL bFGF) hESC media was added. After 24 to 48 h, supernatants were aspirated, then hESCs were harvested with accutase and pooled with the supernatants. Human ESCs were then stained with Annexin-APC (Cat. no. 550474; BD Biosciences) and 7-AAD (Cat. no. 00–6993-50; eBioscience) according to the manufacturer's protocol (BD Bioscience). For all cultures, media was changed daily, whether to complete or minimal media. For the inducible BCL2 hESCs, DOX (1 μg/mL) was added to the media of selected wells the day before changing to minimal media, and was re-added at each media change until analysis. To count the absolute number of cells in each well, a set number of accudrop beads (Cat. no. 345249; BD Biosciences) was added to samples before FACS analysis. After FACS analysis, the percent of sample analyzed could be estimated based on the amount of accudrop beads counted in relation to the amount added. The caspase inhibitor ZVAD-FMK was used at 25 μM according to the manufacturer's instructions (R&D; Cat. no. FMK001).
Supplementary Material
Acknowledgments
The authors thank Stephen Willingham for helpful discussion, Libuse Jerabek for excellent laboratory management, and members of the I.L.W. laboratory for helpful advice and assistance. This work was supported in part by Grant RCI 00354 from the California Institute for Regenerative Medicine; Fellowship TG2-01159 from the California Institute for Regenerative Medicine Training Program (to R.A.); fellowships from the National Cancer Institute (Public Health Service Grant CA09151), the California Institute for Regenerative Medicine (T1-00001), and the National Institutes of Health (5 T32 AI07290, Molecular and Cellular Immunobiology) (to M.A.I.); fellowships from the Stanford Medical Scholars Research Program and the Howard Hughes Medical Institute (to S.R.A. and C.T.); and the Soros Fellowships for New Americans (to S.R.A.).
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1019047108/-/DCSupplemental.
References
- 1.Hasegawa K, Fujioka T, Nakamura Y, Nakatsuji N, Suemori H. A method for the selection of human embryonic stem cell sublines with high replating efficiency after single-cell dissociation. Stem Cells. 2006;24:2649–2660. doi: 10.1634/stemcells.2005-0657. [DOI] [PubMed] [Google Scholar]
- 2.Pyle AD, Lock LF, Donovan PJ. Neurotrophins mediate human embryonic stem cell survival. Nat Biotechnol. 2006;24:344–350. doi: 10.1038/nbt1189. [DOI] [PubMed] [Google Scholar]
- 3.Xu Y, et al. Revealing a core signaling regulatory mechanism for pluripotent stem cell survival and self-renewal by small molecules. Proc Natl Acad Sci USA. 2010;107:8129–8134. doi: 10.1073/pnas.1002024107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Skottman H, Narkilahti S, Hovatta O. Challenges and approaches to the culture of pluripotent human embryonic stem cells. Regen Med. 2007;2:265–273. doi: 10.2217/17460751.2.3.265. [DOI] [PubMed] [Google Scholar]
- 5.Chin AC, Padmanabhan J, Oh SK, Choo AB. Defined and serum-free media support undifferentiated human embryonic stem cell growth. Stem Cells Dev. 2010;19:753–761. doi: 10.1089/scd.2009.0210. [DOI] [PubMed] [Google Scholar]
- 6.Ludwig TE, et al. Feeder-independent culture of human embryonic stem cells. Nat Methods. 2006;3:637–646. doi: 10.1038/nmeth902. [DOI] [PubMed] [Google Scholar]
- 7.Ludwig TE, et al. Derivation of human embryonic stem cells in defined conditions. Nat Biotechnol. 2006;24:185–187. doi: 10.1038/nbt1177. [DOI] [PubMed] [Google Scholar]
- 8.Dvorak P, et al. Expression and potential role of fibroblast growth factor 2 and its receptors in human embryonic stem cells. Stem Cells. 2005;23:1200–1211. doi: 10.1634/stemcells.2004-0303. [DOI] [PubMed] [Google Scholar]
- 9.Eiselleova L, et al. A complex role for FGF-2 in self-renewal, survival, and adhesion of human embryonic stem cells. Stem Cells. 2009;27:1847–1857. doi: 10.1002/stem.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.James D, Levine AJ, Besser D, Hemmati-Brivanlou A. TGFbeta/activin/nodal signaling is necessary for the maintenance of pluripotency in human embryonic stem cells. Development. 2005;132:1273–1282. doi: 10.1242/dev.01706. [DOI] [PubMed] [Google Scholar]
- 11.Levenstein ME, et al. Basic fibroblast growth factor support of human embryonic stem cell self-renewal. Stem Cells. 2006;24:568–574. doi: 10.1634/stemcells.2005-0247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li J, et al. MEK/ERK signaling contributes to the maintenance of human embryonic stem cell self-renewal. Differentiation. 2007;75:299–307. doi: 10.1111/j.1432-0436.2006.00143.x. [DOI] [PubMed] [Google Scholar]
- 13.Wang X, Lin G, Martins-Taylor K, Zeng H, Xu RH. Inhibition of caspase-mediated anoikis is critical for basic fibroblast growth factor-sustained culture of human pluripotent stem cells. J Biol Chem. 2009;284:34054–34064. doi: 10.1074/jbc.M109.052290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu RH, et al. NANOG is a direct target of TGFbeta/activin-mediated SMAD signaling in human ESCs. Cell Stem Cell. 2008;3:196–206. doi: 10.1016/j.stem.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Watanabe K, et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol. 2007;25:681–686. doi: 10.1038/nbt1310. [DOI] [PubMed] [Google Scholar]
- 16.Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 1999;13:1899–1911. doi: 10.1101/gad.13.15.1899. [DOI] [PubMed] [Google Scholar]
- 17.Cory S, Adams JM. The Bcl2 family: Regulators of the cellular life-or-death switch. Nat Rev Cancer. 2002;2:647–656. doi: 10.1038/nrc883. [DOI] [PubMed] [Google Scholar]
- 18.Opferman JT, Korsmeyer SJ. Apoptosis in the development and maintenance of the immune system. Nat Immunol. 2003;4:410–415. doi: 10.1038/ni0503-410. [DOI] [PubMed] [Google Scholar]
- 19.Kutschka I, et al. Adenoviral human BCL-2 transgene expression attenuates early donor cell death after cardiomyoblast transplantation into ischemic rat hearts. Circulation. 2006;114(1)(Suppl):I174–I180. doi: 10.1161/CIRCULATIONAHA.105.001370. [DOI] [PubMed] [Google Scholar]
- 20.Sasaki T, et al. Bcl2 enhances survival of newborn neurons in the normal and ischemic hippocampus. J Neurosci Res. 2006;84:1187–1196. doi: 10.1002/jnr.21036. [DOI] [PubMed] [Google Scholar]
- 21.Yamane T, Dylla SJ, Muijtjens M, Weissman IL. Enforced Bcl-2 expression overrides serum and feeder cell requirements for mouse embryonic stem cell self-renewal. Proc Natl Acad Sci USA. 2005;102:3312–3317. doi: 10.1073/pnas.0500167102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yamane T, Hosen N, Yamazaki H, Weissman IL. Expression of AA4.1 marks lymphohematopoietic progenitors in early mouse development. Proc Natl Acad Sci USA. 2009;106:8953–8958. doi: 10.1073/pnas.0904090106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang L, et al. Self-renewal of human embryonic stem cells requires insulin-like growth factor-1 receptor and ERBB2 receptor signaling. Blood. 2007;110:4111–4119. doi: 10.1182/blood-2007-03-082586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vallier L, et al. Activin/Nodal signalling maintains pluripotency by controlling Nanog expression. Development. 2009;136:1339–1349. doi: 10.1242/dev.033951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vallier L, Alexander M, Pedersen RA. Activin/Nodal and FGF pathways cooperate to maintain pluripotency of human embryonic stem cells. J Cell Sci. 2005;118:4495–4509. doi: 10.1242/jcs.02553. [DOI] [PubMed] [Google Scholar]
- 26.Jamieson CHM, Passegue E, Weissman IL. In: Stem Cells in the Nervous System: Functional and Clinical Implications. Gage R, editor. Berlin: Springer; 2004. pp. 157–182. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.