Abstract
Background
Standard care for infants on extracorporeal life support (ECLS) relies on intermittent measurement of blood glucose (BG); however, this can lead to significant changes in BG that go unrecognized for several hours. The present study was designed to assess performance and clinical applicability of a subcutaneous glucose sensor technology modified for use as a blood-contacting sensor within the ECLS circuit.
Methods
Twelve children, aged 3 years or less, requiring ECLS support were studied. Three continuous glucose sensors (Medtronic MiniMed) were inserted into hubs placed in line with the ECLS circuit. Blood glucose was assessed with a laboratory analyzer (BGLAB; Bayer Rapidlab 860) approximately every 5 h (mean 4.9 ± 3.3 h) with more frequent samples obtained with a bedside monitor (HemoCue) as needed. Sensor current (ISIG) was transmitted to a laptop computer and retrospectively calibrated using BGLAB. Sensor performance was assessed by mean absolute relative difference (MARD), linear regression slope and intercept, and correlation, all with BGLAB as reference.
Results
The BGLAB averaged 107.6 ± 36.4 mg/dl (mean ± standard deviation) ranging from 58 to 366 mg/dl. The MARD was 11.4%, with linear regression slope (0.86 ± 0.030) and intercept (9.0 ± 3.2 mg/dl) different from 1 and 0, respectively (p < .05), and correlation (r2 = 0.76; p < .001). The system was not associated with any adverse events, and placement and removal into the hubs was easily accomplished. Instances in which more frequent BG values were obtained using a bedside HemoCue (BGHEMO) monitor showed the sensor to respond rapidly to changes.
Conclusions
We conclude that continuous sensors can be adapted for use in an ECLS circuit with accuracy similar to or better than that achieved with the subcutaneous site. Continuous glucose monitoring in this population can rapidly detect changes in BG that would not otherwise be observed. Further studies will be needed to assess the benefit of continuous glucose monitoring in this population.
Keywords: cardiopulmonary bypass, continuous monitor, extracorporeal, extracorporeal life support, glucose
Background
Infants on extracorporeal life support (ECLS) are among the most tenuous of pediatric critically ill patients, with a mortality rate of 33–62% and significant variations in blood glucose (BG) concentration1,2 secondary to stress, corticosteroid administration, and exogenous glucose. Hyperglycemia is the most common abnormality of glucose metabolism seen in ECLS patients, but hypoglycemia resulting from insufficient endogenous glucose production or insulin therapy can also occur and may exacerbate adverse neurologic outcomes.3,4 Current standard care utilizes intermittent laboratory measurement of glucose concentration; however, this practice is inherently flawed, as significant changes may go unrecognized for several hours. We previously investigated the performance of a first-generation extracorporeal glucose monitoring system designed to be blood contacting5 and adapted for the ECLS circuit in babies.6 In the present study, a second-generation system based on subcutaneous (SC)-glucose sensor technology (Medtronic MiniMed, Northridge, CA) was evaluated. The study was designed to assess performance and clinical applicability of the system in pediatric surgical patients on ECLS.
Methods
Patient Enrollment
A convenience sample of children 3 years old and under who were expected to require support for more than 24 hours were recruited between March 22, 2005, and February 16, 2006. Patients with evidence of thrombus in the circuit, determined by visual inspection by the respiratory therapist monitoring the ECLS circuit, were excluded from the study. The protocol was initiated after the patient was stabilized on ECLS and informed consent obtained. The study was approved by the Children’s Hospital Boston Committee on Clinical Investigation. The device was deemed nonsignificant risk in consultation with the Food and Drug Administration, with the opinion confirmed by the local committee on clinical investigation.
Continuous Glucose Monitor System
Three glucose sensors were introduced into the circuit using sterile technique. Each sensor was inserted into a hub (Figure 1A) placed in the venous (afferent) limb of thecircuit. The sensors were given 5 minutes to hydrate before being connected to transmitters (Figure 1B) that wirelessly relayed the sensor signal (nA current) each minute to a laptop computer running proprietary Medtronic MiniMed software (sensor placement shown; Figure 1C). Each sensor signal was saved to a file for later retro-spective calibration as described later, with the variability between sensors placed in the same circuit assessed by correlation. Sensors were replaced every 48 hours or removed if the ECLS circuit was changed or discontinued. During the course of the study, a modified transmitter was introduced to reduce run-in time, with the two transmitters denoted throughout the article as TX1 (unmodified) and TX2 (modified). Run-in time is defined as the time between first inserting and powering the sensor and the time the sensor is deemed sufficiently stable to report glucose values (typically 2–10 hours depending on sensor manufacturer).
Figure 1.
(A) Pre-inserted hub with glucose sensor; (B) transmitter; (C) afferent circuit showing placement of redundant sensors. Sensor signals were transmitted to a bedside laptop computer and retrospectively calibrated.
Reference Glucose Measurements
Blood glucose was determined by a central laboratory (BGLAB) using a Bayer Rapidlab 860 (Tarrytown, NY) analyzer approximately every 5 hours, with more frequent 5–10 minute values (BGHEMO) obtained as needed using a bedside monitor (HemoCue; Ängelholm, Sweden).
Calibration Algorithm
One-point calibration was performed using the first BGLAB value following 2 h of equilibration time (run in)and the first available value following 6 h intervals thereafter. At each calibration time, a calibration factor (CF) was obtained by dividing BGLAB by sensor current (ISIG); sensor glucose (SG) was subsequently calculated as SG = CF × ISIG. On occasions when frequent BGHEMO values were obtained by bedside clinicians in order to assess rapid changes in BG, a second retrospective calibration was performed using linear regression to determine sensor sensitivity (CFLR = 1/slope of regression; mg/dl pernA) and offset current (OS; nA; intercept of regression). Offset current is defined as the expected current as the glucose concentration approaches 0. For cases in which frequent BGHEMO values were available, identified in the text as examples, SG was calculated by first subtracting the offset (SGLR = CFLR × [ISIG - OS]).
Statistical Analysis
Mean absolute relative difference (MARD) between blood and sensor glucose, linear regression of the blood value versus sensor value, and correlation were all performed using BGLAB. At each calibration time point, the sensor reading prior to calibration was paired with the BG values (BGLAB) when assessing accuracy to allow all possible BGLAB values to be used in assessing performance metrics (MARD, SG versus BGLAB slope and intercept, and correlation). Analysis was performed separately for the two transmitters used in the study (TX1 and TX2) with the data combined when no statistically significant difference was observed. Example profiles in which the more frequent BGHEMO determinations were used to assess rapid changes in glucose are identified within the manuscript, with the calibration method (linear regression versus one point) and source of the BG measurement (laboratory versus HemoCue) noted. Retrospective one-point calibration was performed using MATLAB version 7.6(MathWorks, Inc., Natick, MA). Statistical analysis (linear regression for slope and offset; Mann–Whitney for comparisons of MARD) was performed using GraphPad Prizm (version 5.00 for Windows, GraphPad Software, San Diego, CA).
Results
Twelve patients [5 male, 7 female, median age 10 days (range 1–998), and body weight 3.5 kg (range 2.8–14.7 kg)] were enrolled in the study. Six patients were diagnosed with congenital cardiac disease, 5 with congenital diaphragmatic hernias, and 1 with respiratory failure. Fifty-four sensor profiles were obtained, 36 with TX1 and 18 with TX2, totaling 2095 h of data collection and 365 paired BGLAB–SG measurements. Median sensor duration was 45.5 h (range 18.3–48 h). Median time from start of ECLS to sensor insertion was 70.5 h (range 14.0–263.0). Sensors were inserted into and removed from the ECLS circuit without incident. There were no adverse outcomes from the sensor use and no discernable increase in the incidence of clot formation, air embolus, or need for circuit changes.
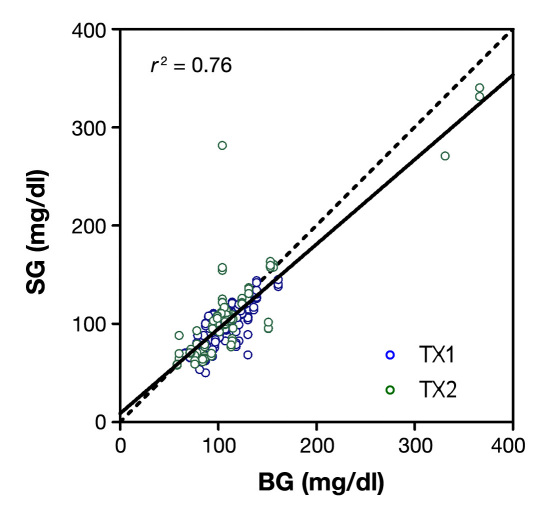
Average BG concentration during the study was 106 ± 36 mg/dl (mean ± standard deviation), ranging from 58 to 366 mg/dl. Regression analysis performed separately on profiles obtained with TX1 and TX2 did not indicate a difference (p = 0.14; Figure 2, regression lines not shown) and data were combined to form a single regression with slope and intercept (0.86 and 9 mg/dl, respectively) different from the unbiased BG = SG line (p < .05; Figure 2, dashed line). Blood glucose and SG were significantly correlated (r2 = 0.76; p < .001). The MARDwas 10.1% and 14.1% for TX1 and TX2, respectively (p = not significant), with a combined value of 11.4%.
Figure 2.
Regression showing paired sensor (SG) and reference glucose (BG) values. Regression slopes were not different using TX1 and TX2 (not shown), and the data were combined to form a single regression line (solid line), which was determined to have slope and offset different from the unbiased BG = SG line (dashed line).
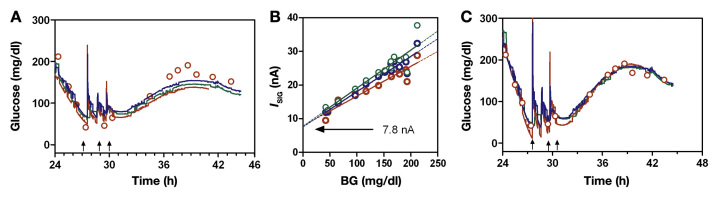
The regression slope less than 1 (Figure 2) suggests that SG was overestimated at values below where the regression line intersected the SG = BG line and under-estimated above this point. Closer examination in a subject treated for hypoglycemia (Figure 3A, treatment with exogenous glucose shown with arrows; frequent blood samples obtained with HemoCue) confirmed the one-point calibration overestimated BGHEMO in the low region (time ~28 h) and underestimated it in the high region (time ~38 h). To determine if the bias was due to a difference in reference meters (laboratory values versus bedside HemoCue) the sensors were recalibrated based on the linear regression slope and offset of BGHEMO versus ISIG. All three sensors in the circuit were observed to be highly correlated with the HemoCue determinations (Figure 3B; r2 = 0.9942, 0.9905, and 0.9911) but showed significant OS current (~7.8 nA). Recalibration with the OS removed showed all three sensors to track both the low and high glucose values (Figure 3C, with BGHEMO versus SG regression slope and intercept not different from 1 and 0, respectively, regressions not shown) and to respond to exogenous glucose boluses (arrows) with no discernable delay.
Figure 3.
(A) Triplicate SG profiles obtained in one subject requiring repeat glucose boluses (marked as upward-pointing arrows) to maintain euglycemia. Sensors were calibrated using laboratory glucose values and the one-point calibration algorithm but are shown here with the more frequent bedside BGHEMO measurements. (B) Recalibration using linear regression of BGHEMO and sensor current, with sensor OS current identified as ~7.8 nA for all 3 sensors. (C) Recalibrated SG signals with OS removed.
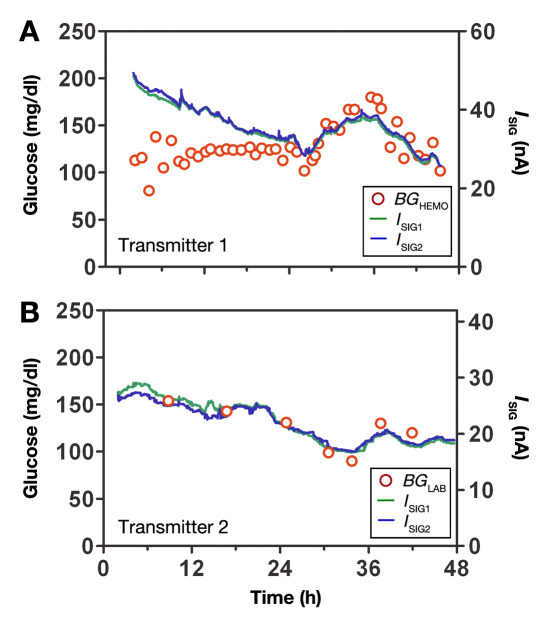
Some profiles obtained with TX1 (example shown in Figure 4A) were observed to decrease for almost 24 h despite a relatively stable BG profile. Thereafter, the current was observed to be well correlated with the subsequent glucose excursion (r2 = 0.78 and 0.81 for t > 24 h). During the excursion, both sensors had substantial OS current (13.5 and 13.1 nA; BGHEMO versus ISIG regressions not shown). In contrast, profiles obtained with TX2 (Figure 4B) began tracking BGLAB almost immediately (r2 = 0.92 and 0.85 for t > 2 h), albeit many still had significant OS current (3.8 and 2.4 nA; BGHEMO versus ISIG regressions not shown).
Figure 4.
(A) Current in two sensors recorded with transmitter 1 (unmodified) compared to BG assessed with a HemoCue. (B) Sensor current obtained in two sensors connected to transmitter 2 (modified for rapid run in) compared with BG assessed with a laboratory analyzer.
Discussion
The present study demonstrated that continuous glucose sensor technology similar to that used in earlier ambulatory studies7–10 can be safely modified for placement into a heart–lung bypass circuit. Accuracy, expressed as MARD (11.4%), was similar to or better than that observed when similar sensors are placed subcutaneously (16–18%10,11). We observed a slope less than 1 in the regression of BG and SG (Figure 2), which would limit the ability to detect hypoglycemia12 (Figure 3A); however, this was easily corrected (Figure 3C) by identifying a nonspecific background current (Figure 3B). A more surprising observation within this study was that some sensor signals showed long run-in times (Figure 4A) that were corrected (Figure 4B) with a transmitter designed to reduce the run in observed when sensors are placed subcutaneously. The run-in phenomenon is widely believed to be due to a local tissue response13,14 to wound healing and would not be expected to occur in the extracorporeal circuit. Generally, sensor performance in an extracorporeal blood contacting loop should be expected to be better than that observed when sensors are placed subcutaneously, as putative delays in SC interstitial fluid glucose15–17 do not exist. Also noteworthy in the present study is the high correlation between sensor signals placed in the same circuit (Figures 3 and 4). Although a high correlation is desirable, it can generate instances in which an artifact observed in one sensor is also observed in multiple sensors of the same type.
The present study was not designed to look at sensor-related differences in infection or mortality or the ability of continuous glucose monitoring to aid in managing BG levels. Nonetheless, it is apparent that if a real-time calibration algorithm were used, the sensors would have allowed recognition of critically high and low BG concentrations faster than standard intermittent monitoring. Of the two calibration algorithms used here, one-point and linear regression, only the one‑point can be applied in real time. The linear regression calibration used all the data points to estimated sensor sensitivity and offset, a process that can only be applied retrospectively. A real-time linear regression calibration using all available glucose points up to the time of calibration can be performed but often results in poor estimates of the OS current when the available reference BG values do not span a wide range of glucose values.12 It is also possible to perform a one-point calibration with an arbitrary OS,18 but this requires the OS to be similar in different sensors. In the present study, OS was similar in sensors placed in the same circuit but varied between circuits (e.g., ~7.6 nA in all the sensors shown in Figure 3 and ~14 nA in both sensors shown in Figure 4). Underestimation of the true OS results in an inability of the monitor to detect hypoglycemia (Figure 3A).12
Once the OS issue is resolved, the ability to see changes in glucose in real time should allow clinicians to identify when the glucose concentration becomes abnormal and identify trends toward hyperglycemia or hypoglycemia that could generate an appropriate clinical response before severe abnormalities develop (analogous to the use of portable oxygen saturation monitoring). This ability to watch and preemptively treat evolving metabolic derangements is critical if these patients are to be maintained in a euglycemic state while minimizing risk of hypoglycemia. Continuous glucose monitoring may also allow insulin to be more effectively utilized in managing glucose levels.19,20
Although ECLS patients represent an important group for targeting advances in treatment, they account for only a small percentage of critically ill children. Broader benefits may be achieved in adapting the technology for patients on short-term cardiopulmonary bypass or those receiving hemodialysis or continuous hemofiltration. Tight glycemic control has been widely adopted in the treatment of adult critically ill cardiac surgical patients, and studies suggest that further benefit may be gained through maintenance of euglycemia during cardiopulmonary bypass for open heart surgery.21–24 Success in achieving intraoperative tight glycemic control is variable, in part, due to difficulties in monitoring and responding to rapid changes in BG during cardiopulmonary bypass.25–27 The risk of hypoglycemia has led some centers to abandon attempts at intraoperative control.26,28 Dialysis patients, many of whom also suffer from diabetes mellitus, may also be a particularly important group of patients to target therapies that improve glycemic control.
Conclusions
We show here that continuous sensors can be adapted for use in an ECLS circuit. This technology may be useful in improving the safety of tight glycemic control in patient populations that have similar vascular access, including short-term cardiopulmonary bypass and dialysis. Changes in the sensor design, or calibration algorithm, may still be required to address problems associated with offset current. Further research will also be needed to ensure the monitor can be used in systems in which clots may be present (excluded from the present study). Nonetheless, while the blood contacting sensor evaluated here remains a work in progress, it is clear that continuous glucose monitoring can be introduced into the ECLS circuit and that doing so will allow critically high or low BG concentration to be determined more rapidly than standard intermittent monitoring.
Abbreviations
- BG
blood glucose
- CF
calibration factor
- ECLS
extracorporeal life support
- MARD
mean absolute relative difference
- OS
offset current
- SC
subcutaneous
- SG
sensor glucose
References:
- 1.Conrad SA, Rycus PT, Dalton H. Extracorporeal Life Support Registry Report 2004. ASAIO J. 2005;51(1):4–10. doi: 10.1097/01.mat.0000151922.67540.e9. [DOI] [PubMed] [Google Scholar]
- 2.Kyle UG, Jolliet P, Genton L, Meier CA, Mensi N, Graf JD, Chevrolet JC, Pichard C. Clinical evaluation of hormonal stress state in medical ICU patients: a prospective blinded observational study. Intensive Care Med. 2005;31(12):1669–1675. doi: 10.1007/s00134-005-2832-9. [DOI] [PubMed] [Google Scholar]
- 3.Glass P, Wagner AE, Papero PH, Rajasingham SR, Civitello LA, Kjaer MS, Coffman CE, Getson PR, Short BL. Neurodevelopmental status at age five years of neonates treated with extracorporeal membrane oxygenation. J Pediatr. 1995;127(3):447–457. doi: 10.1016/s0022-3476(95)70082-x. [DOI] [PubMed] [Google Scholar]
- 4.Short BL. The effect of extracorporeal life support on the braa focus on ECMO. Semin Perinatol. 2005;29(1):45–50. doi: 10.1053/j.semperi.2005.02.007. [DOI] [PubMed] [Google Scholar]
- 5.Renard E, Costalat G, Chevassus H, Bringer J. Artificial beta-cell: clinical experience toward an implantable closed-loop insulin delivery system. Diabetes Metab. 2006;32(5 Pt 2):497–502. doi: 10.1016/s1262-3636(06)72802-6. [DOI] [PubMed] [Google Scholar]
- 6.Javid PJ, Halwick DR, Betit P, Thompson JE, Long K, Zhang Y, Jaksic T, Agus MS. The first use of live continuous glucose monitoring in patients on extracorporeal life support. Diabetes Technol Ther. 2005;7(3):431–439. doi: 10.1089/dia.2005.7.431. [DOI] [PubMed] [Google Scholar]
- 7.Steil GM, Rebrin K, Mastrototaro J, Bernaba B, Saad MF. Determination of plasma glucose during rapid glucose excursions with a subcutaneous glucose sensor. Diabetes Technol Ther. 2003;5(1):27–31. doi: 10.1089/152091503763816436. [DOI] [PubMed] [Google Scholar]
- 8.Steil GM, Rebrin K, Hariri F, Jinagonda S, Tadros S, Darwin C, Saad MF. Interstitial fluid glucose dynamics during insulin-induced hypoglycaemia. Diabetologia. 2005;48(9):1833–1840. doi: 10.1007/s00125-005-1852-x. [DOI] [PubMed] [Google Scholar]
- 9.Steil GM, Rebrin K, Darwin C, Hariri F, Saad MF. Feasibility of automating insulin delivery for the treatment of type 1 diabetes. Diabetes. 2006;55(12):3344–3350. doi: 10.2337/db06-0419. [DOI] [PubMed] [Google Scholar]
- 10.Panteleon AE, Rebrin K, Steil GM. The role of the independent variable to glucose sensor calibration. Diabetes Technol Ther. 2003;5(3):401–410. doi: 10.1089/152091503765691901. [DOI] [PubMed] [Google Scholar]
- 11.Keenan DB, Cartaya R, Mastrototaro JJ. Accuracy of a new real-time continuous glucose monitoring algorithm. J Diabetes Sci Technol. 2010;4(1):111–118. doi: 10.1177/193229681000400114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rebrin K, Sheppard NF Jr, Steil GM. Use of subcutaneous interstitial fluid glucose to estimate blood glucose: revisiting delay and sensor offset. J Diabetes Sci Technol. 2010;4(5):1087–1098. doi: 10.1177/193229681000400507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Henninger N, Woderer S, Kloetzer HM, Staib A, Gillen R, Li L, Yu X, Gretz N, Kraenzlin B, Pill J. Tissue response to subcutaneous implantation of glucose-oxidase-based glucose sensors in rats. Biosens Bioelectron. 2007;23(1):26–34. doi: 10.1016/j.bios.2007.03.016. [DOI] [PubMed] [Google Scholar]
- 14.Chen T, Schmidtke DW, Heller A. Defining the period of recovery of the glucose concentration after its local perturbation by the implantation of a miniature sensor. Clin Chem Lab Med. 2002;40(8):786–789. doi: 10.1515/CCLM.2002.135. [DOI] [PubMed] [Google Scholar]
- 15.Monsod TP, Flanagan DE, Rife F, Saenz R, Caprio S, Sherwin RS, Tamborlane WV. Do sensor glucose levels accurately predict plasma glucose concentrations during hypoglycemia and hyper-insulinemia? Diabetes Care. 2002;25(5):889–893. doi: 10.2337/diacare.25.5.889. [DOI] [PubMed] [Google Scholar]
- 16.Voskanyan G, Barry Keenan D, Mastrototaro JJ, Steil GM. Putative delays in interstitial fluid (ISF) glucose kinetics can be attributed to the glucose sensing systems used to measure them rather than the delay in ISF glucose itself. J Diabetes Sci Technol. 2007;1(5):639–644. doi: 10.1177/193229680700100507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stout PJ, Racchini JR, Hilgers ME. A novel approach to mitigating the physiological lag between blood and interstitial fluid glucose measurements. Diabetes Technol Ther. 2004;6(5):635–644. doi: 10.1089/dia.2004.6.635. [DOI] [PubMed] [Google Scholar]
- 18.Youssef JE, Castle JR, Engle JM, Massoud RG, Ward WK. Continuous glucose monitoring in subjects with type 1 diabetes: improvement in accuracy by correcting for background current. Diabetes Technol Ther. 2010;12(11):921–928. doi: 10.1089/dia.2010.0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Agus MS, Javid PJ, Ryan DP, Jaksic T. Intravenous insulin decreases protein breakdown in infants on extracorporeal membrane oxygenation. J Pediatr Surg. 2004;39(6):839–844. doi: 10.1016/j.jpedsurg.2004.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Agus MS, Javid PJ, Piper HG, Wypij D, Duggan CP, Ryan DP, Jaksic T. The effect of insulin infusion upon protein metabolism in neonates on extracorporeal life support. Ann Surg. 2006;244(4):536–544. doi: 10.1097/01.sla.0000237758.93186.c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Doenst T, Wijeysundera D, Karkouti K, Zechner C, Maganti M, Rao V, Borger MA. Hyperglycemia during cardiopulmonary bypass is an independent risk factor for mortality in patients undergoing cardiac surgery. J Thorac Cardiovasc Surg. 2005;130(4):1144. doi: 10.1016/j.jtcvs.2005.05.049. [DOI] [PubMed] [Google Scholar]
- 22.Furnary AP, Wu Y, Bookin SO. Effect of hyperglycemia and continuous intravenous insulin infusions on outcomes of cardiac surgical procedures: the Portland Diabetic Project. Endocr Pract. 2004;10(Suppl 2):21–33. doi: 10.4158/EP.10.S2.21. [DOI] [PubMed] [Google Scholar]
- 23.Najmaii S, Redford D, Larson DF. Hyperglycemia as an effect of cardiopulmonary bypass: intra-operative glucose management. J Extra Corpor Technol. 2006;38(2):168–173. [PMC free article] [PubMed] [Google Scholar]
- 24.Preissig CM, Rigby MR. Pediatric critical illness hyperglycemia: risk factors associated with development and severity of hyperglycemia in critically ill children. J Pediatr. 2009;155(5):734–739. doi: 10.1016/j.jpeds.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 25.Carvalho G, Moore A, Qizilbash B, Lachapelle K, Schricker T. Maintenance of normoglycemia during cardiac surgery. Anesth Analg. 2004;99(2):319–324. doi: 10.1213/01.ANE.0000121769.62638.EB. [DOI] [PubMed] [Google Scholar]
- 26.Chaney MA, Nikolov MP, Blakeman BP, Bakhos M. Attempting to maintain normoglycemia during cardiopulmonary bypass with insulin may initiate postoperative hypoglycemia. Anesth Analg. 1999;89(5):1091–1095. doi: 10.1213/00000539-199911000-00004. [DOI] [PubMed] [Google Scholar]
- 27.Smith CE, Styn NR, Kalhan S, Pinchak AC, Gill IS, Kramer RP, Sidhu T. Intraoperative glucose control in diabetic and nondiabetic patients during cardiac surgery. J Cardiothorac Vasc Anesth. 2005;19(2):201–208. doi: 10.1053/j.jvca.2005.01.032. [DOI] [PubMed] [Google Scholar]
- 28.Brunkhorst FM, Engel C, Bloos F, Meier-Hellmann A, Ragaller M, Weiler N, Moerer O, Gruendling M, Oppert M, Grond S, Olthoff D, Jaschinski U, John S, Rossaint R, Welte T, Schaefer M, Kern P, Kuhnt E, Kiehntopf M, Hartog C, Natanson C, Loeffler M, Reinhart K, German Competence Network Sepsis (SepNet) Intensive insulin therapy and pentastarch resuscitation in severe sepsis. N Engl J Med. 2008;358(2):125–139. doi: 10.1056/NEJMoa070716. [DOI] [PubMed] [Google Scholar]