Abstract
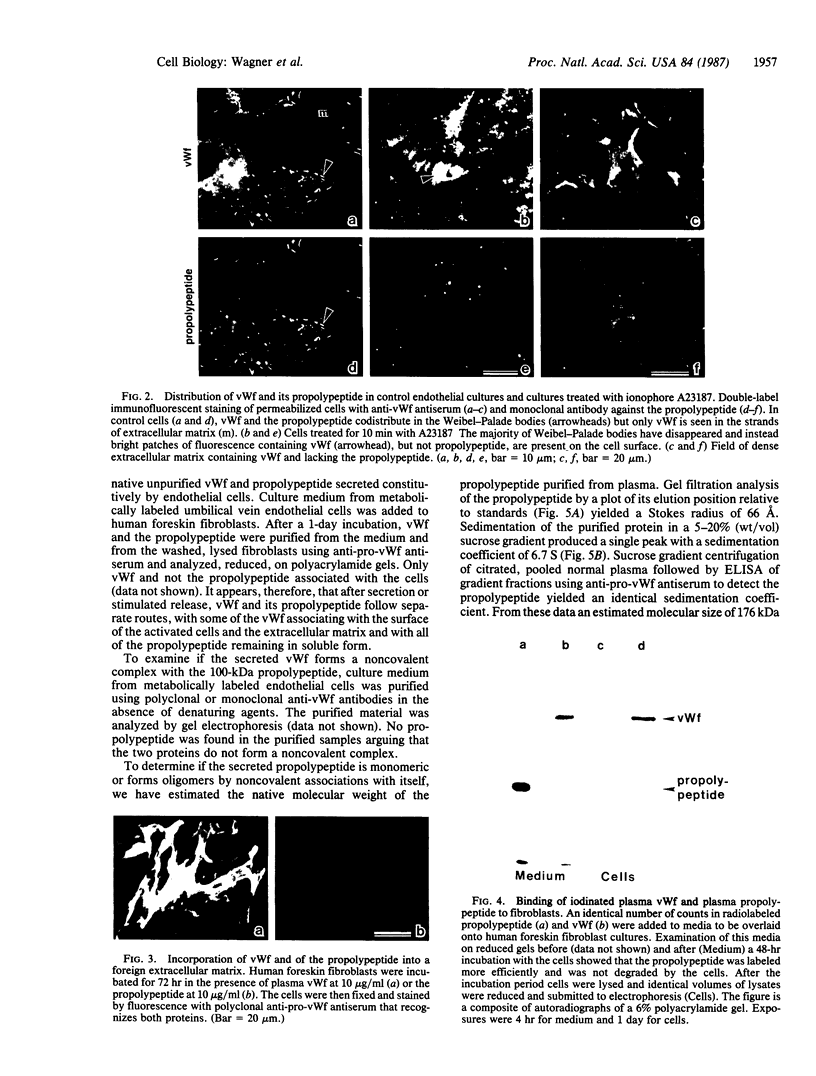
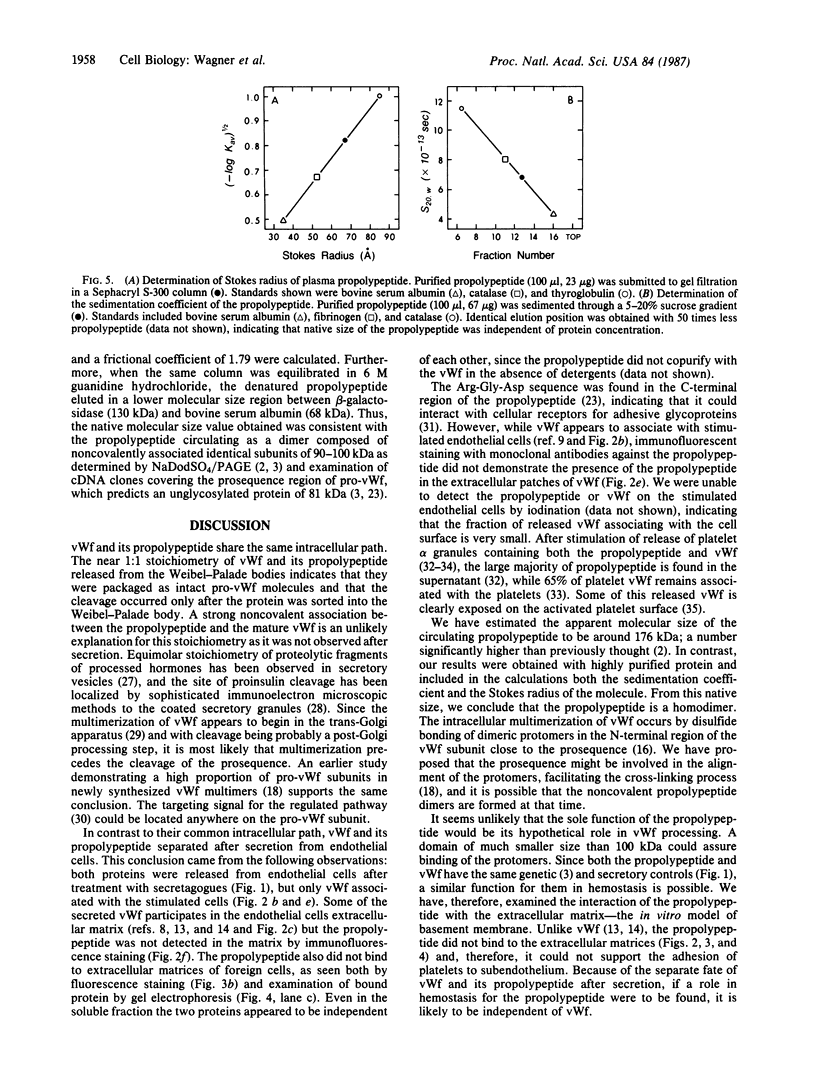
The intracellular site of cleavage of pro-von Willebrand factor subunit and the subsequent fate of the propolypeptide (von Willebrand antigen II) and of the mature von Willebrand factor (vWf) were investigated. Both the propolypeptide, which was found to be a homodimer of noncovalently linked subunits, and mature vWf were released from Weibel-Palade bodies of endothelial cells following stimulation with secretagogues. The stoichiometry of the two released proteins was essentially equimolar. This indicates that vWf and the propolypeptide were packaged into the Weibel-Palade bodies as one unit, pro-vWf, and that the proteolytic cleavage of pro-vWf is likely to be a post-Golgi event. The association of prosequences into dimers supports their hypothetical role in the multimerization process. After secretion, the two proteins were distributed differently, as based on the following observations. The propolypeptide did not associate with vWf in the culture medium, did not codistribute with vWf in the extracellular "patches of release" on stimulated endothelial cells, and was not detected in the endothelial cell extracellular matrix, which did contain vWf. Additionally, in contrast to vWf, the propolypeptide did not bind to the matrix of human foreskin fibroblasts. Since the propolypeptide does not associate with vWf and does not interact with extracellular matrices in vitro, it is highly unlikely that it would promote platelet adhesion to subendothelium in vivo.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Fay P. J., Anderson M. T., Chavin S. I., Marder V. J. The size of human factor VIII heterodimers and the effects produced by thrombin. Biochim Biophys Acta. 1986 Jun 23;871(3):268–278. doi: 10.1016/0167-4838(86)90208-6. [DOI] [PubMed] [Google Scholar]
- Fay P. J., Kawai Y., Wagner D. D., Ginsburg D., Bonthron D., Ohlsson-Wilhelm B. M., Chavin S. I., Abraham G. N., Handin R. I., Orkin S. H. Propolypeptide of von Willebrand factor circulates in blood and is identical to von Willebrand antigen II. Science. 1986 May 23;232(4753):995–998. doi: 10.1126/science.3486471. [DOI] [PubMed] [Google Scholar]
- Gimbrone M. A., Jr, Cotran R. S., Folkman J. Human vascular endothelial cells in culture. Growth and DNA synthesis. J Cell Biol. 1974 Mar;60(3):673–684. doi: 10.1083/jcb.60.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham J. M., Hynes R. O., Davidson E. A., Bainton D. F. The location of proteins labeled by the 125I-lactoperoxidase system in the NIL 8 hamster fibroblast. Cell. 1975 Apr;4(4):353–365. doi: 10.1016/0092-8674(75)90156-7. [DOI] [PubMed] [Google Scholar]
- Hormia M., Lehto V. P., Virtanen I. Factor VIII-related antigen. A pericellular matrix component of cultured human endothelial cells. Exp Cell Res. 1983 Dec;149(2):483–497. doi: 10.1016/0014-4827(83)90360-9. [DOI] [PubMed] [Google Scholar]
- Jaffe E. A., Hoyer L. W., Nachman R. L. Synthesis of antihemophilic factor antigen by cultured human endothelial cells. J Clin Invest. 1973 Nov;52(11):2757–2764. doi: 10.1172/JCI107471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly R. B. Pathways of protein secretion in eukaryotes. Science. 1985 Oct 4;230(4721):25–32. doi: 10.1126/science.2994224. [DOI] [PubMed] [Google Scholar]
- Koutts J., Walsh P. N., Plow E. F., Fenton J. W., 2nd, Bouma B. N., Zimmerman T. S. Active release of human platelet factor VIII-related antigen by adenosine diphosphate, collagen, and thrombin. J Clin Invest. 1978 Dec;62(6):1255–1263. doi: 10.1172/JCI109246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lahav J., Hynes R. O. Involvement of fibronectin, Von Willebrand factor, and fibrinogen in platelet interaction with solid substrata. J Supramol Struct Cell Biochem. 1981;17(4):299–311. doi: 10.1002/jsscb.380170402. [DOI] [PubMed] [Google Scholar]
- Levine J. D., Harlan J. M., Harker L. A., Joseph M. L., Counts R. B. Thrombin-mediated release of factor VIII antigen from human umbilical vein endothelial cells in culture. Blood. 1982 Aug;60(2):531–534. [PubMed] [Google Scholar]
- Loesberg C., Gonsalves M. D., Zandbergen J., Willems C., van Aken W. G., Stel H. V., Van Mourik J. A., De Groot P. G. The effect of calcium on the secretion of factor VIII-related antigen by cultured human endothelial cells. Biochim Biophys Acta. 1983 Sep 22;763(2):160–168. doi: 10.1016/0167-4889(83)90039-3. [DOI] [PubMed] [Google Scholar]
- Lynch D. C., Zimmerman T. S., Kirby E. P., Livingston D. M. Subunit composition of oligomeric human von Willebrand factor. J Biol Chem. 1983 Nov 10;258(21):12757–12760. [PubMed] [Google Scholar]
- Mains R. E., Eipper B. A. Coordinate, equimolar secretion of smaller peptide products derived from pro-ACTH/endorphin by mouse pituitary tumor cells. J Cell Biol. 1981 Apr;89(1):21–28. doi: 10.1083/jcb.89.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarroll D. R., Levin E. G., Montgomery R. R. Endothelial cell synthesis of von Willebrand antigen II, von Willebrand factor, and von Willebrand factor/von Willebrand antigen II complex. J Clin Invest. 1985 Apr;75(4):1089–1095. doi: 10.1172/JCI111802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montgomery R. R., Zimmerman T. S. von Willebrand's disease antigen II. A new plasma and platelet antigen deficient in severe von Willebrand's disease. J Clin Invest. 1978 Jun;61(6):1498–1507. doi: 10.1172/JCI109070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Orci L., Ravazzola M., Amherdt M., Madsen O., Vassalli J. D., Perrelet A. Direct identification of prohormone conversion site in insulin-secreting cells. Cell. 1985 Sep;42(2):671–681. doi: 10.1016/0092-8674(85)90124-2. [DOI] [PubMed] [Google Scholar]
- Rand J. H., Gordon R. E., Sussman I. I., Chu S. V., Solomon V. Electron microscopic localization of factor-VIII-related antigen in adult human blood vessels. Blood. 1982 Sep;60(3):627–634. [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. Arg-Gly-Asp: a versatile cell recognition signal. Cell. 1986 Feb 28;44(4):517–518. doi: 10.1016/0092-8674(86)90259-x. [DOI] [PubMed] [Google Scholar]
- Scott J. P., Montgomery R. R. Platelet von Willebrand's antigen II: active release by aggregating agents and a marker of platelet release reaction in vivo. Blood. 1981 Dec;58(6):1075–1080. [PubMed] [Google Scholar]
- Siegel L. M., Monty K. J. Determination of molecular weights and frictional ratios of proteins in impure systems by use of gel filtration and density gradient centrifugation. Application to crude preparations of sulfite and hydroxylamine reductases. Biochim Biophys Acta. 1966 Feb 7;112(2):346–362. doi: 10.1016/0926-6585(66)90333-5. [DOI] [PubMed] [Google Scholar]
- Sporn L. A., Marder V. J., Wagner D. D. Inducible secretion of large, biologically potent von Willebrand factor multimers. Cell. 1986 Jul 18;46(2):185–190. doi: 10.1016/0092-8674(86)90735-x. [DOI] [PubMed] [Google Scholar]
- Verweij C. L., Diergaarde P. J., Hart M., Pannekoek H. Full-length von Willebrand factor (vWF) cDNA encodes a highly repetitive protein considerably larger than the mature vWF subunit. EMBO J. 1986 Aug;5(8):1839–1847. doi: 10.1002/j.1460-2075.1986.tb04435.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner D. D., Lawrence S. O., Ohlsson-Wilhelm B. M., Fay P. J., Marder V. J. Topology and order of formation of interchain disulfide bonds in von Willebrand factor. Blood. 1987 Jan;69(1):27–32. [PubMed] [Google Scholar]
- Wagner D. D., Marder V. J. Biosynthesis of von Willebrand protein by human endothelial cells. Identification of a large precursor polypeptide chain. J Biol Chem. 1983 Feb 25;258(4):2065–2067. [PubMed] [Google Scholar]
- Wagner D. D., Marder V. J. Biosynthesis of von Willebrand protein by human endothelial cells: processing steps and their intracellular localization. J Cell Biol. 1984 Dec;99(6):2123–2130. doi: 10.1083/jcb.99.6.2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner D. D., Mayadas T., Marder V. J. Initial glycosylation and acidic pH in the Golgi apparatus are required for multimerization of von Willebrand factor. J Cell Biol. 1986 Apr;102(4):1320–1324. doi: 10.1083/jcb.102.4.1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner D. D., Olmsted J. B., Marder V. J. Immunolocalization of von Willebrand protein in Weibel-Palade bodies of human endothelial cells. J Cell Biol. 1982 Oct;95(1):355–360. doi: 10.1083/jcb.95.1.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner D. D., Urban-Pickering M., Marder V. J. Von Willebrand protein binds to extracellular matrices independently of collagen. Proc Natl Acad Sci U S A. 1984 Jan;81(2):471–475. doi: 10.1073/pnas.81.2.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zucker M. B., Broekman M. J., Kaplan K. L. Factor VIII-related antigen in human blood platelets: localization and release by thrombin and collagen. J Lab Clin Med. 1979 Nov;94(5):675–682. [PubMed] [Google Scholar]