Abstract
Th-2-biased immune responses are known to play a key role in the pathogenesis of atopic dermatitis. In particular, the macrophage-derived chemokine CCL22 is directly implicated in Th-2-associated skin inflammatory reactions, and its levels are significantly elevated in serum and are correlated with disease severity in atopic dermatitis. In this study, we tested the development of genetic therapeutic options to treat atopic dermatitis using bacteria expressing miRNA. We constructed a recombinant strain of Salmonella typhimurium expressing CCL22 miRNA (ST-miRCCL22) for the in vivo knockdown of CCL22. The CCL22 gene was downregulated with CCL22 miRNA in activated lymphocytes. In mice with a cutaneous disease similar to atopic dermatitis, interleukin-4 was inhibited and interferon-γ was induced after treatments with ST-miRCCL22. Furthermore, CCL22 levels were suppressed in the atopic mice treated with ST-miRCCL22. These results suggest that ST-miRCCL22 may be an effective genetic agent for treating atopic dermatitis.
Keywords: biological therapy; chemokine CCL22; dermatitis, atopic; immunotherapy; RNA interference; Salmonella typhimurium
Introduction
Atopic dermatitis (AD) is a chronic and persistent inflammatory skin disease with recurrent inflammatory episodes that results from a combination of genetic predisposition, imbalanced immune responses, epidermal barrier abnormalities and severe pruritus (Chen et al., 2004). In particular, imbalances between the Th-1 and Th-2 immune responses (Th-2-biased immune response) are known to be key factors in the pathogenesis of AD (Akkoc et al., 2008). These two types of helper T-cell responses differ in their functional effects and cytokine production. Cytokines regulate the function of T cells in allergic immune responses. IL-2 and IFN-γ are typically produced by T helper 1 (Th 1) cells, whereas T helper 2 (Th 2) cells predominantly produce IL-4, IL-5 and IL-10 (Schwarz et al., 1993). Furthermore, the humoral immune response is dominated by enhanced IgE secretion. Thus, in allergic diseases such as AD, Th-2 responses are considered to play primary roles in inducing disease (Yanai et al., 2007).
CCL22 levels were significantly elevated and were correlated with the severity of disease in atopic patients (Nakazato et al., 2008). The imbalance in serum CCL22 concentrations contributes to AD development (Narbutt et al., 2009). To knockdown the CCL22 gene, we used an RNA interference (RNAi) mechanism. RNAi induces the specific inhibition of eukaryotic gene expression through the degradation of complementary mRNA (Zeng and Cullen, 2002). In spite of the great potential of RNAi, clinical trials with RNAi are difficult because of their transient nature, their instability and the lack of appropriate delivery methods (Yang et al., 2008).
Furthermore, the use of vector-based, short hairpin RNA (shRNA) for RNAi analysis requires the screening of a large number of sequences to identify active sequences. Moreover, the use of Pol III promoters limits their applications to tissue-specific expression (Giering et al., 2008).
To overcome these limitations of siRNA and shRNA, we used an engineered vector expressing RNAi that enables the expression of engineered microRNA (miRNA) sequences from Pol II promoters (Xia et al., 2006). miRNAs are expressed as small single-stranded RNA (ssRNA) sequences that are 22 nucleotides in length and naturally direct gene silencing through components shared with the RNAi pathway (Bartel, 2004). Furthermore, S. typhimurium was transformed with the CCL22 miRNA expression vector (miRCCL22) to solve the problems associated with RNAi delivery.
In this study, we assessed the efficacy of live, attenuated S. typhimurium as a vector for oral gene therapy and tested the efficacy of ST-miRCCL22 in mice with AD.
Results
Creation of S. typhimurium-expressing CCL22 miRNA bacteria
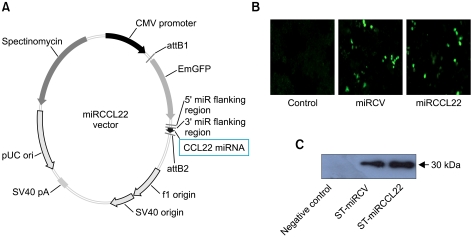
To construct the CCL22 miRNA plasmid for RNAi against CCL22, we designed two single-stranded DNA oligonucleotides encoding the CCL22 target pre-miRNA using an RNAi design program. A 22-nucleotide miRNA double-stranded oligonucleotide was inserted into the pcDNA™ 6.2-GW/EmGFP-miR expression vector (Figure 1A). We observed that successful transfer of CCL22 miRNA expression into mammalian cells occurred. Raw 264.7 cells were transfected with miRCCL22. miRNA expression in the cell was detected with fluorescence microscopy (× 200) after transfection for 48 hours (Figure 1B). Green fluorescence was detected in the Raw 264.7 cells transfected with both miRCCL22 and the EmGFP-positive control vector but not with the negative control vector. These data show that the miRNA expression vector in S. typhimurium can be expressed in mammalian cells.
Figure 1.
Construction of the CCL22 miRNA expression vector (miRNACCL22) and miRNA expression of recombinant S. typhimurium expressing CCL22 miRNA. The dsDNA oligo of CCL22 cloned into the pcDNATM6.2-GW/EmGFP-miR expression vector using T4 DNA ligase (A). To examine the expression of the miRNA vector and S. typhimurium-expressing CCL22 miRNA, Raw 264.7 cells were infected with ST-miRCCL22, ST-miRCV or EmGFP negative controltransfected counterparts. Expression of the miRNA vector was observed in cells with ST-miRCCL22 and ST-miRCV (B). GFP from recombinant Salmonella was also detected in ST-miRCCL22 and ST-miRCV by Western blots, but not in control samples (C).
E. coli DH5α cells were transformed with miRCCL22, and the plasmid was isolated and used to transform S. typhimurium SF586. The plasmid from the transformed SF586 cells was used to further transform S. typhimurium BRD509, and this was used for further experiments.
To evaluate the expression of miRCCL22 in bacteria, we also observed green fluorescent proteins in ST-miRCCL22 and ST-miRCV by Western blot analysis. It should be noted that miRCCL22 contains the EmGFP coding sequence under the control of the CMV promoter (Figure 1C).
Gene silencing of CCL22
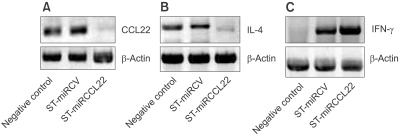
To examine whether ST-miRCCL22 successfully silenced the CCL22 gene, whole mouse splenocytes were extracted. Splenocytes were treated with lectin and IL-4 to induce the overexpression of CCL22; whole mouse splenocytes were then transfected with ST-miRCCL22 (6 × 108 c.f.u.). The expression of CCL22 was only silenced in mouse splenocytes treated with ST-miRCCL22 (Figure 2A). These data showed that ST-miRCCL22 induced specific silencing of the CCL22 gene.
Figure 2.
Gene silencing against CCL22 and the alteration of inflammatory cytokine levels. Expression of CCL22 was silenced in splenocytes after treatment with ST-miRCCL22; ST-miRCV did not affect CCL22 expression (A). Specific gene silencing against CCL22 suppressed IL-4 (B) and increased IFN-γ levels (C). One experiment, representative of three independent analyses with similar results, is shown.
Cytokines are known to be important factors in AD; hence, we also tested the expression levels of the inflammatory cytokines IFN-γ and IL-4. Mouse splenocytes were extracted to analyze changes in the in vitro levels of these inflammatory cytokine. Splenocyte cells successfully overexpressed CCL22 after treatment with lectin and IL-4. Mouse splenocytes were then infected with ST-miRCCL22 (6 × 108 c.f.u.). Total RNA was isolated, and cDNA was synthesized from mouse splenocytes after treatment with ST-miRCCL22. RT-PCR analysis showed changes in the cytokine levels of the treated cells. IL-4 levels were suppressed in cells treated with ST-miRCCL22 but were unchanged in the ST-miRCV treatment group (Figure 2B). The ST-miRCCL22 treatment groups also showed greater induction of IFN-γ production than ST-miRCV-treated cells (Figure 2C). These results suggested that ST-miRCCL22 altered the levels of inflammatory cytokines.
Modulation of IL-4, IFN-γ, IL-10, TNF-α and IgE in mice with cutaneous disease after treatment with ST-miRCCL22
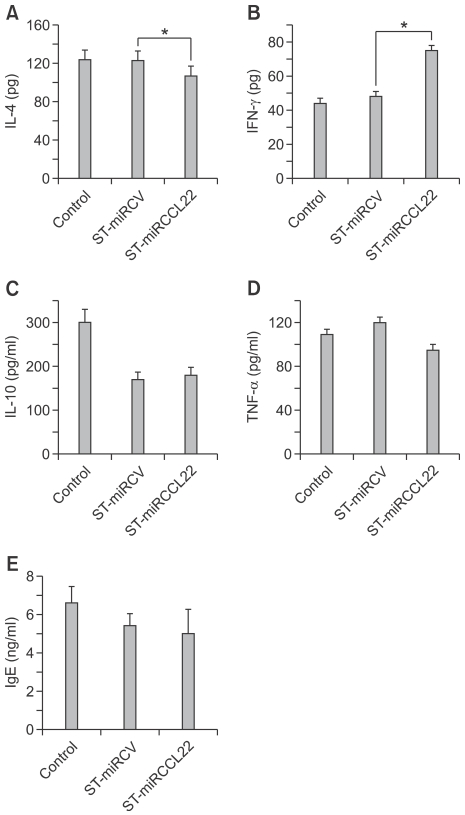
IL-4 levels were elevated in an AD mouse model. Thus, the changes in IL-4 levels in mice with AD were also examined after ST-miRCCL22 treatment. For this test, mice with AD were orally inoculated with 1.6 × 108 c.f.u. ST-miRCCL22, ST-miRCV and PBS. One week after inoculation, total serum was collected from each mouse for the detection of IL-4 by ELISA. As shown in Figure 3A, the total IL-4 levels in ST-miRCCL22-treated mice were lower than those in the PBS- and ST-miRCV-treated mice. This result showed that specific gene silencing against CCL22 suppressed IL-4 levels (Figure 3A). The Th-1 cytokine IFN-γ is also an important factor in the primary immune response, and the levels of IFN-γ were reduced in both AD patients and the AD mouse model. Therefore, to check the IFN-γ production level in mice treated with ST-miRCCL22, mice with AD were orally inoculated with ST-miRCCL22, ST-miRCV and PBS. One week after inoculation, total serum from each mouse was collected for the detection of IFN-γ by ELISA. Total IFN-γ levels in ST-miRCCL22-treated mice were increased compared to levels in mice without ST-miRCCL22 treatment (Figure 3B), but were not increased compared to levels in mice treated with PBS or ST-miRCV. These data showed that specific gene silencing against CCL22 affected IFN-γ levels.
Figure 3.
Changes in IL-4, IFN-γ, IL-10, TNF-α and IgE levels in serum from mice suffering from atopic dermatitis that were treated with ST-miRCCL22. Mice were orally infected with 1.6 × 108 c.f.u. ST-miRCCL22 and ST-miRCV. Total IL-4 (A), IFN-γ (B), IL-10 (C) and TNF-α (D) levels of ST-miRCCL22 treated mice were examined by ELISA. Total IgE levels in ST-miRCCL22-treated mice were lower when compared with the PBS or ST-miRCV groups (E). *P < 0.05 when comparing the ST-miRCCL22 group with the ST-miRCV-control group (n = 5). The results are expressed as the mean ± SEM (n = 5).
We also examined the inhibition of IL-10, TNF-α and IgE in mice with cutaneous disease after treatment with ST-miRCCL22. Although IL-10 and TNF-α were reduced after treatment with ST-miRCCL22 compared to their levels in control mice, all orally Salmonella-treated mice displayed reduced IL-10 levels in blood. Atopic dermatitis is frequently accompanied by high levels of IgE, which is known to be a key pathogenic factor in AD. Thus, we tested the IgE production levels in mice treated with ST-miRCCL22. For this analysis, mice with AD were orally inoculated with 1.6 × 108 c.f.u. ST-miRCCL22, ST-miRCV and PBS. One week after inoculation, total serum from each mouse was collected for the detection of IgE by ELISA. The total IgE level of ST-miRCCL22-treated mice was suppressed compared to mice treated with PBS or ST-miRCV (Figure 3E). These data show that specific gene silencing against CCL22 decreased the hypersensitivity caused by IgE secretion.
Suppression of CCL22 in blood and skin from mice after treatment with ST-miRCCL22
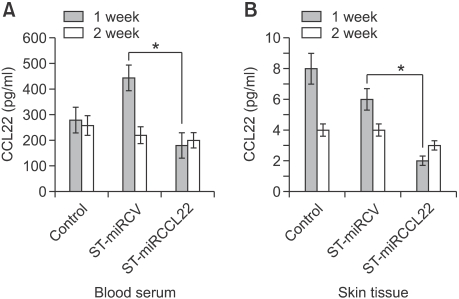
We investigated whether CCL22 could be detected in blood and skin tissues after treatment with ST-miRCCL22. At 1 week after oral treatment with ST-miRCCL22, mice demonstrated suppression of CCL22 protein in blood (Figure 4A). CCL22 (MDC) levels in serum were examined by ELISA.
Figure 4.
Suppression of CCL22 in serum and skin tissue from mice suffering from atopic dermatitis treated with ST-miRCCL22. Serum and skin tissue were obtained from mice at one week after treatment with ST-miRCCL22. Skin samples were isolated with NucleoSpin RNA/Protein Columns (MACHEREY-NAGEL, Germany). The concentrations of CCL22 were measured by ELISA (R&D Systems). *P < 0.05 when comparing the ST-miRCCL22 group with the ST-miRCV-control group (n = 5).
Interestingly, we were able to detect CCL22 in skin tissue. After oral treatment, skin tissues were homogenized and CCL22 was isolated using a protein column. CCL22 expression was suppressed in tissue after oral treatment with ST-miRCCL22; these suppressions were reduced after two weeks. Altogether, these data suggest that oral treatment with ST-miRCCL22 induced the suppression of CCL22 in mice (Figure 4B).
Improvement of skin inflammation in mice treated with ST-miRCCL22
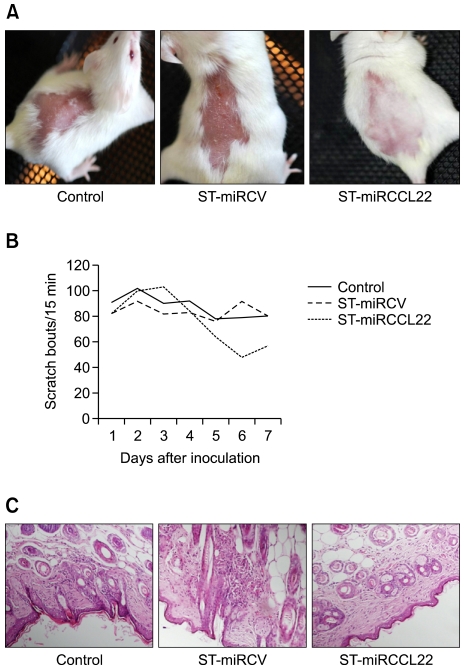
To examine the efficacy of ST-miRCCL22 in mice, we studied mice treated with DNCB to induce an inflammatory reaction and elevated levels of IgE and IL-4 in blood (data not shown). In mice treated with ST-miRCCL22, skin regeneration and hair re-growth were exhibited, but control mice did not show these effects (Figure 5A). Because pruritus is one of the major symptoms of AD, we examined the scratching behavior of mice. The total scratching counts in the ST-miRCCL22-treated group were significantly lower than those for the mice treated with PBS or ST-miRCV (Figure 5B). To verify the effect of recombinant bacteria on tissue, we examined the histological changes of skin tissues after oral administration of recombinant Salmonella. In ST-miRCCL22-treated mice, skin inflammations were reduced and other adverse effects were not observed. These data showed that specific gene silencing against CCL22 relieved pruritus (Figure 5C).
Figure 5.
Improvement of the symptoms of atopic dermatitis by oral administration of ST-miRCCL22 in mice. (A) The skin of ST-miRCCL22-treated mice showed greater improvement when compared to the PBS or ST-miRCV groups. (B) The total scratch counts of ST-miRCCL22-treated mice were dramatically lower compared with the PBS or ST-miRCV groups. *P < 0.05, when comparing the ST-miRCCL22 group with the ST-miRCV-control group (n = 10). These experiments were repeated at least three times with similar results. (C) Histological analyses were performed to examine the reduced inflammation due to ST-miRCCL22. Histological samples were collected seven days after oral inoculation with 108 bacterial cells. Mice were treated with PBS, ST-miRCV and ST-miRCCL22. As shown with H/E staining, skin tissue demonstrated reduced inflammation when compared to the skin of control mice treated with ST-miRCCL22.
Discussion
This study showed that the suppression of CCL22 induced therapeutic effects and that S. typhimurium could be used as a vector for miRNA delivery in mice with AD. In particular, S. typhimurium-harboring plasmids that expressed CCL22 miRNA reduced in vitro and in vivo CCL22 gene expression in mouse models of AD.
In this study, we used an RNAi mechanism to knockdown CCL22. Although siRNA or shRNA treatments are limited by problems associated with stability and delivery (Brummelkamp et al., 2002; Paddison et al., 2002), we engineered bacteria with vector-based miRNA for easy, stable delivery. Previous research showed that S. typhimurium could transfer eukaryotic vector-based RNAi-expressing plasmids in vitro or in vivo by oral administration. Oral administration of ST-miRCCL22 reduced CCL22 gene expression in the mouse spleen and induced therapeutic effects.
Our plasmid DNA, under the control of the CMV promoter, can be expressed in both eukaryotic and prokaryotic systems. This plasmid was partially generated with bacterial promoters placed in the CMV promoter sequence (Goussard et al., 2003; Gahan et al., 2009).
We evaluated GFP expression in ST-miRCCL22 and ST-miRCV bacteria using Western blot analysis. GFP was observed in ST-miRCCL22 and ST-miRCV but was not observed in the control group (Figure 1B). These data showed that the miRNA vector system used in this study, which included miRCCL22, could be expressed in both eukaryotic and prokaryotic systems (Figure 1C).
CCL22 principally attracts CD4+ T cells, which express a skin-homing receptor known as cutaneous lymphocyte-associated antigen (CLA) (Campbell and Kemp, 1999; Ghia et al., 2002). These cells also bind CCR4, which is preferentially expressed on Th-2 cells. Accordingly, CCL22 is directly implicated in Th-2-associated skin inflammatory responses and is significantly elevated and correlated with disease severity in AD (Gilet et al., 2009).
In this study, S. typhimurium expressing CCL22 miRNA reduced IL-4 levels. ST-miRCCL22 also reduced IL-4 and induced IFN-γ. IL-4 is a key factor in the induction of IgE isotype switching in B cells (Fiset et al., 2006; Howell et al., 2006), whereas IFN-γ inhibits IgE secretion through the antagonistic effect of the Th-2 cytokines (Herberth et al., 2010). These data demonstrated that CCL22 plays an important role in the shift and balance of Th-2 in AD. Furthermore, the inhibition of the CCL22 gene induced a Th-1 immune response and downregulated the Th-2 immune response. These responses contributed to the Th-1/Th-2 immune response in vitro.
The IgE response was elevated in the AD model, as evidenced by increased Th-2 cytokine responses and the simultaneous decrease of IFN-γ production in patients with AD (Yoshizawa et al., 2002).
Atopic dermatitis is characterized by increased IL-4 secretion and decreased IFN-γ secretion in the peripheral circulation (Clarke et al., 2008). IL-4 is known to assist in the switching of B-cells to allergen-specific IgE (Gauchat et al., 1990). Activation of the IL-4 receptor-signaling pathway increased serum IgE levels in AD (Chan et al., 2001). IFN-γ plays the opposite role to that of IL-4 (Liu et al., 1999). Therefore, imbalanced Th-1/Th-2 immune responses and Th-2-biased immune responses are related to AD. In addition, itching is an important symptom in AD (Takaoka et al., 2006), which causes histamines to be secreted through the activation of Th-2 and IgE/mast-cell driven responses (Thurmond et al., 2008). In our research, the recombinant microbe was treated to modulate imbalances in the disease. The treatment of ST-miRCCL22 induced therapeutic effects on the scratching behavior of mice with AD.
In conclusion, recombinant S. typhimurium that expresses the CCL22 miRNA reduced CCL22 gene expression in cells and induced therapeutic effects with Th-1 immune responses.
These results suggest that S. typhimurium expressing CCL22 miRNA could potentially be used as an effective therapeutic agent for treating AD.
Methods
Design of the CCL22 pre-miRNA and construction of the CCL22 miRNA expression vector
We designed two single-stranded DNA oligonucleotides encoding the CCL22 target pre-miRNA (GenBank accession number NM_009137) using an RNAi design program. The top and bottom single-strand oligos (Top strand: 5'-TGCTGTTATGGAGTAGCTTCTTCACCGTTTTGGCCACTGACTGACGGTGAAGACTACTCCATAA-3', Bottom strand: 5'-CCTGTTATGGAGTAGTCTTCACCGTCAGTCAGTGGCCAAAACGGTGAAGAAGCTACTCCATAAC-3') were annealed to generate a double-stranded oligonucleotide for cloning into the miRNA expression vector. Once the double-stranded oligos were generated, they were cloned into the miRNA expression vector using T4 DNA Ligase. This new plasmid was named miRCCL22, and the scrambled miRNA expressing plasmid was named miRCV.
Creation of CCL22 miRNA-expressing Salmonella strains and growth conditions
Competent DH5α cells were transformed with the resultant plasmid vector, and plasmid DNA was isolated from the DH5α cells using a plasmid mini-prep kit (GeneAll, Korea). Then the S. typhimurium SF586 strain (SF586) was transformed with the plasmid DNA by electroporation. Plasmids from the transformed SF586 were used to transform S. typhimurium BRD 509 (BRD 509) cells, which are mutant aroA/aroD variants of SL1344. The engineered S. typhimurium expressing CCL22 miRNA was designated as ST-miRCCL22. S. typhimurium containing the scrambled miRNA-expressing plasmid and S. typhimurium that were used as controls were termed ST-miRCV. Each bacterial strain was grown in Luria-Bertani (LB) broth, consisting of 1% Tryptone, 0.5% NaCl and 0.5% Yeast with spectinomycin at 50 µg/ml for E. coli, 250 µg/ml for the SF586 strain and 500 µg/ml for the BRD 509 strain.
CCL22 miRNA transfection
Raw 264.7 cells were purchased from KCLB (the Korean Cell Line Bank, Seoul) and cultured in DMEM supplemented with 100 units/ml penicillin and gentamycin (Welgene, Korea), 100 mg/ml streptomycin and 10% FBS (Welgene, Korea). One day before transfection, cells were plated on culture slides at a density of 2 × 104 and incubated overnight at 37℃ in a 5% CO2 incubator so that the monolayer would be 90% to 95% confluent at the transfection time. After incubation, 0.5 µg of plasmid DNA was transfected with lipofectamine 2000 (Invitrogen) or transformed S. typhimurium, and the cells were incubated at 37℃ in a 5% CO2 incubator for 48 h.
Whole splenocyte culture and infection of Salmonella strains
Mice were sacrificed by dislocation of the cervical vertebrae, and the spleen was dissociated under aseptic conditions. The extracted spleen was transferred to 10 ml of RPMI1640. Connective tissues and fat were removed. The spleen was then homogenized using a homogenizer, and red blood cells were lysed in red blood cell lysing buffer (Sigma, St Louis, MO). The cells were cultured in RPMI1640 media supplemented with 100 units/ml penicillin, 100 mg/ml streptomycin and 10% FBS (Welgene, Korea). Cells were plated at a density of 5.0 × 107 in 6-well culture plates and treated with Lectin (10 µg/ml, Sigma) and IL-4 (20 ng/ml, Invitrogen, Carlsbad, CA) to overexpress CCL22 for 24 h. Mouse splenocytes were transfected with ST-miRCCL22 for one h, and gentamycin was added (50 µg/ml, Welgene; Korea) for 90 min to kill any remaining extracellular bacteria. The mixtures were incubated for 48 h to express the eukaryotic plasmid vector in the mammalian cell.
RNA isolation & RT-PCR
Cultured cells were collected by centrifugation at 1500 × g, and total RNA was isolated by TRIzol (Invitrogen) according to the manufacturer's instructions. RNA was reverse transcribed using random primers and the M-MLV-RT enzyme for cDNA synthesis (Promega). The resulting cDNA was used in semi-quantitative PCR reactions (final volume of 20 µl) with Taq Polymerase enzyme and its reaction mix (Han-taq, Korea), 0.2 mM dNTP (Takara, Japan) and 0.3 µM forward and reverse primers. The primers used for semi-quantitative PCR were as follows: CCL22: 5'ATGAGGTCACTTCAGATGCT and 5'AGGTCACGGCCTTGGGTTTT (284 bp) IL-4: 5'GAGCCATATCCACGGATGCGACAA and 5'CATGGTGGCTCAGTACTACGAGTA3'; IFN-γ: pre-made primers (Bioneer, Korea); and β-actin: pre-made primers (Bioneer, Korea). CCL22 was amplified from 1 µl (~20 ng) cDNA in a 35-cycle PCR program with cycling conditions as follows: 95℃ for 30 s, 50℃ for 30 s, 72℃ for 30 s. IL-4 and IFN-γ were amplified from 1 µl (~20 ng) of cDNA in a 35-cycle PCR program with cycling conditions as follows: 95℃ for 1 min, 50℃ for 1 min, and 72℃ for 1 min. Normalized template volumes were subjected to 31 to 35 PCR cycles using mouse β-actin primers.
Allergic mouse model
Four-week-old CD-1 (ICR) female mice were purchased from Orient Co. Ltd. (Korea) and fed a standard diet for mice (Purina). The mice were housed in an animal room maintained at 24 ± 2℃ with a 12-h light/dark cycle. Mice were acclimatized to the animal facility for one week before experimentation. The hair covering specific body regions of each mouse was removed by hair clippers and a hair remover. After 48 h, 2,4-Dinitro-1-chlorobenzene (DNCB; Sigma) was used as a contact allergen. The test group of mice were subjected to 100 µl of 1% DNCB (Olive oil: Acetone, 1:3) for three days. After 48 h, the mice were treated with 120 µl of 2% DNCB daily for three days (Shah et al., 2010; Yamashita et al., 2010).
SDS-PAGE and western blot assay
The supernatant from the bacterial culture was run on a 12% SDS-PAGE and electrophoretically transferred to nitrocellulose membranes. The membranes were pre-equilibrated overnight at 4℃ with a TBS-T solution containing 5% skim milk and were incubated with mouse anti-GFP antibodies for one hour at room temperature. After three washes with TBS-T, the membranes were incubated with a goat anti-mouse Ig-G HRP conjugate for one hour at room temperature. Immunoreactive protein bands were visualized using the BM Chemiluminescence Blotting Substrate (Roche).
ELISA
Serum samples of the control and test groups were collected from mice by eye bleeding at one week post-bacterial delivery. Additionally, 96-well plates were coated with 100 µl/well of each of the antibodies (IL-4, IFN-γ, IL-10, TNF-α, IgE capture antibody) and incubated overnight at 4℃ according to manufacturer's instruction (BD). The wells were aspirated and washed three times with PBS-T (PBS with 0.05% Tween 20) and were blocked with dilution buffer (PBS with 10% FBS) for two hours at room temperature. After aspiration, 100 µl of each sample was pipetted into the wells and incubated for two hours at room temperature. The wells were aspirated and washed five times. Then 100 µl of each detection antibody and SAv-HRP conjugate reagent were added and incubated for one hour at room temperature. The wells were aspirated by washing seven times followed by the addition of 100 µl of substrate solution to each well and incubation for 30 min at room temperature in the dark. Subsequently, 50 µl of stop solution (2N H2SO4) was added. The absorbance was read at 450 nm by a microplate ELISA reader within 30 min of stopping the reaction.
Scratching behavior test
The induced hypersensitivity of pruritic mice was observed for seven days. The total number of scratching bouts was scored over the entire 15-min period. A scratching bout was defined as one or more rapid hind paw movements directed toward the region that was in direct contact with the DNCB agent. The end point of a scratching bout was when the mouse bit or licked its toes or placed its hind paw on the floor. Grooming or other movements directed toward areas other than the DNCB hypersensitive site were not counted. Observers were always blinded to the treatment group and genotype.
Histological analysis
Each mouse skin tissue sample was collected and sequentially immersion-fixed in 10% paraformaldehyde/0.1 M phosphate buffer (PB) pH 7.4, 12.5% glucose/0.1 M PB pH 7.4 and 25% glucose/0.1 M PB pH 7.4 for two days. Afterwards, the tissue was decalcified using 5% ethylenediaminetetraacetic acid (EDTA)/25% glucose/0.1 M PB pH 7.4 for two days. Samples were routinely processed and embedded in 4% carboxy methyl cellulose. Standard sagittal sections measuring 5 µm in thickness were prepared from the frozen and fixed toe using a cryostat CM3050S (LIECA, Tokyo, Japan). The degree of inflammation was histologically assessed on each section, and the sections were stained with Mayer's hematoxylin and eosin (HE), toluidine blue (TB) pH 7.0 and tartrate-resistant acid phosphatase (TRAP) (Moon and Ryu, 2007). For analysis, an Olympus light microscope (× 100) was used.
Statistical analysis
Statistical significance was analyzed using Student's t-tests; *P < 0.05 was considered significant.
Acknowledgements
This work was supported by the Korea Science & Engineering Foundation (Grant No.E00156) and by a grant from the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea. (A090228). We are grateful to Hana Nim for expert technical assistance.
Abbreviations
- AD
atopic dermatitis
- ST-miRCCL22
Salmonella typhimurium expressing CCL22 miRNA
- miRNA
microRNA
References
- 1.Akkoc T, de Koning PJ, Ruckert B, Barlan I, Akdis M, Akdis CA. Increased activation-induced cell death of high IFN-gamma-producing T(H)1 cells as a mechanism of T(H)2 predominance in atopic diseases. J Allergy Clin Immunol. 2008;121:652–658.e1. doi: 10.1016/j.jaci.2007.12.1171. [DOI] [PubMed] [Google Scholar]
- 2.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 3.Brummelkamp TR, Bernards R, Agami R. Stable suppression of tumorigenicity by virus-mediated RNA interference. Cancer Cell. 2002;2:243–247. doi: 10.1016/s1535-6108(02)00122-8. [DOI] [PubMed] [Google Scholar]
- 4.Campbell DE, Kemp AS. Cutaneous lymphocyte-associated antigen expression in children with atopic dermatitis and non-atopic healthy children. Pediatr Allergy Immunol. 1999;10:253–257. doi: 10.1034/j.1399-3038.1999.00042.x. [DOI] [PubMed] [Google Scholar]
- 5.Chan LS, Robinson N, Xu L. Expression of interleukin-4 in the epidermis of transgenic mice results in a pruritic inflammatory skin disease: an experimental animal model to study atopic dermatitis. J Invest Dermatol. 2001;117:977–983. doi: 10.1046/j.0022-202x.2001.01484.x. [DOI] [PubMed] [Google Scholar]
- 6.Chen L, Martinez O, Overbergh L, Mathieu C, Prabhakar BS, Chan LS. Early up-regulation of Th2 cytokines and late surge of Th1 cytokines in an atopic dermatitis model. Clin Exp Immunol. 2004;138:375–387. doi: 10.1111/j.1365-2249.2004.02649.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Clarke E, Rahman N, Page N, Rolph MS, Stewart GJ, Jones GJ. Functional characterization of the atopy-associated gene PHF11. J Allergy Clin Immunol. 2008;121:1148–1154.e3. doi: 10.1016/j.jaci.2008.02.028. [DOI] [PubMed] [Google Scholar]
- 8.Fiset PO, Leung DY, Hamid Q. Immunopathology of atopic dermatitis. J Allergy Clin Immunol. 2006;118:287–290. doi: 10.1016/j.jaci.2006.03.046. [DOI] [PubMed] [Google Scholar]
- 9.Gahan ME, Webster DE, Wesselingh SL, Strugnell RA, Yang J. Bacterial antigen expression is an important component in inducing an immune response to orally administered Salmonella-delivered DNA vaccines. PLoS One. 2009;4:e6062. doi: 10.1371/journal.pone.0006062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gauchat JF, Lebman DA, Coffman RL, Gascan H, de Vries JE. Structure and expression of germline epsilon transcripts in human B cells induced by interleukin 4 to switch to IgE production. J Exp Med. 1990;172:463–473. doi: 10.1084/jem.172.2.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ghia P, Strola G, Granziero L, Geuna M, Guida G, Sallusto F, Ruffing N, Montagna L, Piccoli P, Chilosi M, Caligaris-Cappio F. Chronic lymphocytic leukemia B cells are endowed with the capacity to attract CD4+, CD40L+ T cells by producing CCL22. Eur J Immunol. 2002;32:1403–1413. doi: 10.1002/1521-4141(200205)32:5<1403::AID-IMMU1403>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 12.Giering JC, Grimm D, Storm TA, Kay MA. Expression of shRNA from a tissue-specific pol II promoter is an effective and safe RNAi therapeutic. Mol Ther. 2008;16:1630–1636. doi: 10.1038/mt.2008.144. [DOI] [PubMed] [Google Scholar]
- 13.Gilet J, Chang Y, Chenivesse C, Legendre B, Vorng H, Duez C, Wallaert B, Porte H, Senechal S, Tsicopoulos A. Role of CCL17 in the generation of cutaneous inflammatory reactions in Hu-PBMC-SCID mice grafted with human skin. J Invest Dermatol. 2009;129:879–890. doi: 10.1038/jid.2008.333. [DOI] [PubMed] [Google Scholar]
- 14.Goussard S, Grillot-Courvalin C, Courvalin P. Eukaryotic promoters can direct protein synthesis in Gram-negative bacteria. J Mol Microbiol Biotechnol. 2003;6:211–218. doi: 10.1159/000077252. [DOI] [PubMed] [Google Scholar]
- 15.Herberth G, Heinrich J, Roder S, Figl A, Weiss M, Diez U, Borte M, Herbarth O, Lehmann I. Reduced IFN-gamma- and enhanced IL-4-producing CD4+ cord blood T cells are associated with a higher risk for atopic dermatitis during the first 2 yr of life. Pediatr Allergy Immunol. 2010;21:5–13. doi: 10.1111/j.1399-3038.2009.00890.x. [DOI] [PubMed] [Google Scholar]
- 16.Howell MD, Gallo RL, Boguniewicz M, Jones JF, Wong C, Streib JE, Leung DY. Cytokine milieu of atopic dermatitis skin subverts the innate immune response to vaccinia virus. Immunity. 2006;24:341–348. doi: 10.1016/j.immuni.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 17.Liu M, Zheng S, Wang X, Wen Z. Regulatory roles of IL-12, IL-4 and IFN-gamma on IgE synthesis in atopic patients. Chin Med J (Engl) 1999;112:550–553. [PubMed] [Google Scholar]
- 18.Moon EY, Ryu SK. TACI:Fc scavenging B cell activating factor (BAFF) alleviates ovalbumin-induced bronchial asthma in mice. Exp Mol Med. 2007;39:343–352. doi: 10.1038/emm.2007.38. [DOI] [PubMed] [Google Scholar]
- 19.Nakazato J, Kishida M, Kuroiwa R, Fujiwara J, Shimoda M, Shinomiya N. Serum levels of Th2 chemokines, CCL17, CCL22, and CCL27, were the important markers of severity in infantile atopic dermatitis. Pediatr Allergy Immunol. 2008;19:605–613. doi: 10.1111/j.1399-3038.2007.00692.x. [DOI] [PubMed] [Google Scholar]
- 20.Narbutt J, Lesiak A, Sysa-Jedrzeiowska A, Zakrzewski M, Bogaczewicz J, Stelmach I, Kuna P. The imbalance in serum concentration of Th-1- and Th-2-derived chemokines as one of the factors involved in pathogenesis of atopic dermatitis. Mediators Inflamm. 2009;2009:269541. doi: 10.1155/2009/269541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Paddison PJ, Caudy AA, Hannon GJ. Stable suppression of gene expression by RNAi in mammalian cells. Proc Natl Acad Sci USA. 2002;99:1443–1448. doi: 10.1073/pnas.032652399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schwarz M, Sunder-Plassmann R, Cerwenka A, Pickl WF, Holter W. [Regulation of cytokine production by human T-lymphocytes in allergic immune response] Wien Klin Wochenschr. 1993;105:672–676. [PubMed] [Google Scholar]
- 23.Shah MM, Miyamoto Y, Yamada Y, Yamashita H, Tanaka H, Ezaki T, Nagai H, Inagaki N. Orally supplemented Lactobacillus acidophilus strain L-92 inhibits passive and active cutaneous anaphylaxis as well as 2,4-dinitroflurobenzene and mite fecal antigen induced atopic dermatitis-like skin lesions in mice. Microbiol Immunol. 2010;54:523–533. doi: 10.1111/j.1348-0421.2010.00251.x. [DOI] [PubMed] [Google Scholar]
- 24.Takaoka A, Arai I, Sugimoto M, Honma Y, Futaki N, Nakamura A, Nakaike S. Involvement of IL-31 on scratching behavior in NC/Nga mice with atopic-like dermatitis. Exp Dermatol. 2006;15:161–167. doi: 10.1111/j.1600-0625.2006.00405.x. [DOI] [PubMed] [Google Scholar]
- 25.Thurmond RL, Gelfand EW, Dunford PJ. The role of histamine H1 and H4 receptors in allergic inflammation: the search for new antihistamines. Nat Rev Drug Discov. 2008;7:41–53. doi: 10.1038/nrd2465. [DOI] [PubMed] [Google Scholar]
- 26.Xia XG, Zhou H, Samper E, Melov S, Xu Z. Pol II-expressed shRNA knocks down Sod2 gene expression and causes phenotypes of the gene knockout in mice. PLoS Genet. 2006;2:e10. doi: 10.1371/journal.pgen.0020010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yamashita H, Ito T, Kato H, Asai S, Tanaka H, Nagai H, Inagaki N. Comparison of the efficacy of tacrolimus and cyclosporine A in a murine model of dinitrofluorobenzene-induced atopic dermatitis. Eur J Pharmacol. 2010;645:171–176. doi: 10.1016/j.ejphar.2010.07.031. [DOI] [PubMed] [Google Scholar]
- 28.Yanai M, Sato K, Aoki N, Takiyama Y, Oikawa K, Kobayashi H, Kimura S, Harabuchi Y, Tateno M. The role of CCL22/macrophage-derived chemokine in allergic rhinitis. Clin Immunol. 2007;125:291–298. doi: 10.1016/j.clim.2007.08.002. [DOI] [PubMed] [Google Scholar]
- 29.Yang N, Zhu X, Chen L, Li S, Ren D. Oral administration of attenuated S. typhimurium carrying shRNA-expressing vectors as a cancer therapeutic. Cancer Biol Ther. 2008;7:145–151. doi: 10.4161/cbt.7.1.5195. [DOI] [PubMed] [Google Scholar]
- 30.Yoshizawa Y, Nomaguchi H, Izaki S, Kitamura K. Serum cytokine levels in atopic dermatitis. Clin Exp Dermatol. 2002;27:225–229. doi: 10.1046/j.1365-2230.2002.00987.x. [DOI] [PubMed] [Google Scholar]
- 31.Zeng Y, Cullen BR. RNA interference in human cells is restricted to the cytoplasm. RNA. 2002;8:855–860. doi: 10.1017/s1355838202020071. [DOI] [PMC free article] [PubMed] [Google Scholar]