Abstract
A paradoxical metabolic imbalance in inorganic phosphate occurs from the early onset of diabetes and may lead to a reduction of high energy phosphates and tissue hypoxia. These changes take place in the cells and tissues in which the entry of glucose is not controlled by insulin, and particularly in poorly regulated diabetes patients in whom long-term vascular complications are more likely to occur. Several therapeutic intervention trials have been carried out, including assessment of optimal glucose regulation, the effect of dietary inclusion of calcium diphosphate and pharmaceutical intake of etidronate disodium (EHDP), but none of these modalities wholly overcome the problem. The potential therapeutic application of fructose-1, 6-diphosphate, however, which also acts as human bioenergy, holds a great deal of promise as an efficacious and well-tolerated therapeutic regimen.
Keywords: diabetes, inorganic phosphate metabolism, early intervention trials
Diabetes mellitus is the most common human metabolic disorder, and a major health concern is the severe morbidity and mortality of the late diabetic complications. In our efforts to elucidate the mechanisms leading to the early functional changes in the retina and kidneys of diabetes patients, we noted repeated evidence of a disturbance in the metabolism of inorganic phosphate (Pi).1–6 Phylogenetic, experimental and clinical research data in nondiabetic conditions strongly suggests that the plasma and/or intracellular concentration of Pi may be a determining factor in regulation of energy metabolism and rate of oxygen consumption.7–13 Diabetes mellitus, however, demonstrates the opposite behavior, in that the highest oxygen consumption is associated with the lowest content of inorganic phosphate, and lowering oxygen consumption is associated with higher concentration of inorganic phosphate. Since a reduction of high energy phosphates and tissue hypoxia may be important factors in the development of long-term diabetic complications (DLC), the effects of Pi on the metabolism and function of the erythrocyte and renal tubular cell, as well as local and systemic consequences of severe hypophosphatemia in and during recovery from diabetic ketoacidosis, have recently been reviewed (J Ditzel and HH Lervang, unpublished data). The results indicate, that most conventionally treated diabetic patients respond as if their tissues are in a state of chronic hypoxia and suggesting that erythropoietin (EPO) is stimulated at an early stage in diabetes. A major disturbance in phosphate handling occurs in the kidney tubules, where the excessive sodium-dependent glucose reabsorption in diabetics depolarizes the electrochemical sodium gradient. Since Pi use the same driving force, but have less binding to sodium than glucose and amino acids such as alanine, the Pi reabsorption, particularly in poorly regulated patients, becomes impaired. This paradoxical phosphate imbalance may lead to affinity hypoxia and impaired formation of high energy phosphates. The lack of intracellular phosphate complementary to the increased intracellular glucose takes place in the insulin-insensitive cells and tissues, resulting in the possibility of DLC.
Many of the obvious dietary and pharmacological routes explored thus far to overcome this problem are evaluated herein.
Effect of optimal blood glucose control
Many investigators have found decreased concentrations of Pi in poorly regulated diabetic patients and slightly elevated levels when optimally controlled.
In newly diagnosed, nonacidotic insulin-dependent diabetic patient’s plasma Pi concentration was normal at admission, showed lower range on the day after initial insulin administration and slightly above normal level on the day of best metabolic control. Red cell 2, 3-diphosphoglycerate (2, 3-DPG) exhibited the same fluctuating pattern, and Pi correlated closely to 2, 3-DPG (r =0.61; P < 0.001). Red cell 2, 3-DPG concentration correlated equally well with P50 (oxygen tension at 50% oxygen saturation) of the oxyhemoglobin dissociation curve (ODC).14,15
Gertner et al16 studied mineral metabolism in 7 juvenile-onset diabetic patients before and after achieving near-normal glucose levels by 7 to 14 days treatment with a portable subcutaneous insulin infusion system. They found that as plasma glucose decreased from an average of 221 mg/dL to 95.9 mg/dL, serum Pi rose from 4.09 to 5.01 mg/dL (P < 0.001) due to a 25% rise in renal tubular threshold for phosphate. No change was noted in immunoreactive parathyroid hormone (PTH) and in 1, 25 hydroxy-vitamin D.
Ditzel et al4 studied renal handling of Pi in 26 conventionally treated diabetic children vs 28 healthy children and found fasting urinary phosphate excretion 3 times higher in the former group despite a significantly lower fasting Pi. The maximal capacity of renal tubular reabsorption of phosphate per liter of filtrate (TmPO4/GFR) was significantly suppressed in the diabetic patients. The increased urinary phosphate excretion correlated positively with both urinary glucose excretion and blood glucose concentration (P < 0.01). This finding was unrelated to serum PTH or to plasma growth hormone.
Raskin and Pak17 studied 21 diabetic patients in whom treatment results ranged from “suboptimal” to “optimal” control and found that, as the mean plasma glucose decreased from 17.1 mmol/L to 5.2 mmol/L over 4 to 10 days, serum phosphate level rose from 1.12 to 1.26 mmol/L (P < 0.001).
The same significant increase in Pi was found in 28 patients with type 2 diabetes who were examined both at admission when their disease was poorly controlled and following several days of hospitalization and treatment had markedly improved their metabolic status. In these patients, serum Pi levels increased significantly from 1.12 mmol/L to 1.21 mmol/L (P < 0.01), while serum calcium remained unchanged and urinary calcium and phosphorous excretion both decreased. On admission urinary calcium and phosphorous excretions showed a positive correlation with glucose excretion. Serum PTH decreased from a mid-normal to a low-normal value.18
Thus, in both type 1 and type 2 diabetes, there is a close correlation between the Pi concentration in plasma and an improvement in diabetes control and in intracellular phosphate with a stimulating influence on the rate of cell glycolysis.
Effect of dietary phosphate intake
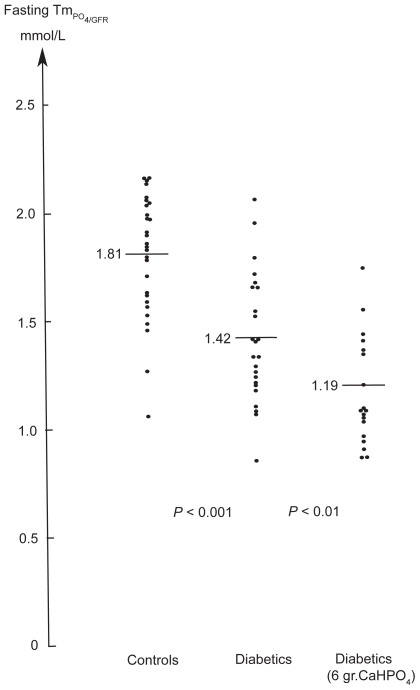
Pi concentration may also be increased by increasing dietary phosphate. Adding 2 g of calcium diphosphate to the three main daily meals over 4 weeks resulted in increased mean content of intraerythrocytic-2, 3-DPG and in the P50 of the ODC in both juvenile and adult diabetes patients.19 To determine whether the immediate effect of increased phosphate intake persisted a subsequent double-masked, placebo-controlled study was undertaken for 1 year to assess renal processing of phosphate and bone mineral content in 43 juvenile type 1 diabetic patients. No increase was seen in the concentration of Pi (active n =19: 1.33 mmol/L vs placebo; n =24: 1.42 mmol/L, P =ns).20 In the treatment group, the fasting urinary phosphate excretion increased as compared to the placebo group (median 1.28 vs 1.01 mmol/h; P < 0.001) and phosphate clearance/GFR was significantly enhanced (0.16 vs 0.10; P < 0.001). The threshold concentration of phosphate (TmPO4/GFR) was suppressed (1.19 vs 1.42 mmol/L; P < 0.01) in the treatment vs placebo group, and in contrast to the controls (1.81 vs 1.42 mmol/L; P < 0.001) (Figure 1). The threshold concentration TmPO4/GFR was not related to the level of parathyroid hormone or to growth hormone level in serum, but inversely correlated with the degree of hyperglycemia. No difference in bone mineral content. was found between the treatment and placebo groups. These observations demonstrate that the dysfunction of the tubular handling of phosphate in diabetic children associated with hyperglycemia cannot be compensated for by daily dietary supplements of calcium diphosphate.
Figure 1.
The phosphate threshold concentration, which is numerically equal to maximum tubular reabsorption rate for phosphate per unit volume of glomerular filtrate in 28 healthy children (1.81 mmol/L) vs 24 conventionally treated diabetic children without microvascular complications (1.42 mmol/L) and 19 diabetic children whom received 6 grams of calcium diphosphate daily for one year (1.19 mmol/L).
Effect of etidronate disodium (EHDP)
Intake of the diphosphonate, ethane-1, 1-diphosphonate (EHDP) is known to produce sustained hyperphosphatemia.21–23 This rise in Pi concentration occurs without a corresponding increase in urinary phosphate excretion, suggesting that EHDP partly elevates Pi by reducing its renal clearance. To study the effect of prolonged elevation of Pi on red cell metabolism and function, oxyhemoglobin dissociation curves (ODC) from zero to full saturation were performed on whole blood from 14 insulin-treated, nonacidotic diabetes patients and 5 healthy volunteers following oral intake of EHDP ( 20 mg kg−1day−1) or placebo for 28 days.24 The mean Pi increased from 1.18 to 1.57 mmol/L (P < 0.001) in the diabetes patients and from 1.14 to 1.69 mmol/L (P < 0.005) in healthy controls. A significant rise in red cell 2, 3-DPG was seen only in the diabetic group from 15.2 to 16.3 μmol/g of hemoglobin (P < 0.005) and probably indicates a suppression of the 2, 3-DPG formation pathway in diabetes patients. However, there was a significant relationship between the concentration of Pi and the P50 of the ODC in both diabetics (r =0.58, P < 0.01) and healthy controls (r =0.69, P < 0.005). Mean P50 in diabetic patients was significantly lower than controls despite normal 2, 3-DPG. The study emphasizes the importance of Pi on red cell function and indicates that elevated Pi tends to counteract the defect in oxygen-release capacity of the erythrocytes in diabetes patients. A subsequent randomized, 6-month, double-blind study of 26 type 1 diabetic patients with nonproliferative retinopathy confirmed the effect of EHDP on Pi, red cell 2, 3-DPG and P50 of ODC.25 Furthermore, the lack of changes in mean glucose and HbA1c indicated an interesting association between improvement in retinopathy as evaluated by retinal fluorescein angiography and increased Pi (P < 0.05).
Potential therapeutic application of fructose-1, 6-diphosphate
Fructose-1, 6-diphosphate (FDP) is a key intermediate in anaerobic glycolysis and is the product of the major regulatory enzyme in the pathway (phosphofructokinase). Natelson et al26 showed that orally administrated fructose diphosphate calcium salt was absorbed directly by the intestinal tract without splitting the phosphate linkage, and that 6 g led to an increase of serum inorganic phosphate averaging 15%, citric acid 10.7%, and nonprotein organic phosphate as much as 173%. Further preclinical and clinical data indicate that FDP can enter cells and serve as a metabolizable substrate of glycolysis.27–29 FDP acts as human bioenergy and can transport phosphorous intracellularly as well as deliver 4 moles of ATP per mole of FDP. Thus FDP has substantial cytoprotective effects on a variety of ischemia-induced tissue damage.30,31 Recent studies also show that FDP can modulate nitric oxide production in a variety of cellular injury events, and indicate that FDP can influence red cell rheology and can increase red cell ATP and 2, 3-DPG levels.32–34 FDP can be given orally and intravenously to humans and it is well tolerated at pharmacological doses. Although FDP appears to be highly efficacious, no previous controlled study has been reported in diabetic patients.
Discussion
This review demonstrates a close correlation between plasma concentration of Pi and the degree of metabolic control of diabetes. The relationship may involve Pi handling in the kidney tubules; the major regulator of Pi homeostasis. Glucose and Pi (eg, alanine, myoinositol) reabsorption are all examples of secondary active transport processes with sodium (Na+) as the driving force. The sodium entry results from the active extrusion of Na+ across the basolateral segment of the proximal tubular cells, energized by adenosine triphosphate (ATP) hydrolysis, and catalyzed by the Na/K-ATPase. As glucose is more potent than inorganic phosphate in stimulating the uptake of Na+ in renal microvillus vesicles,35 the elevated glucose concentrations depolarize the transmembrane electrochemical Na+ gradient of the brush border membrane for inorganic phosphate entry into the tubular cells and decrease intracellular phosphate leading to hyperphosphaturia. Therefore normalization of blood glucose levels leads to an improved capacity of the kidney tubules to reabsorb Pi and a subsequent increase in plasma Pi concentration. Over time this improved glucose regulation will positively influence or even prevent the long-term diabetic complications as has been proven in DCCT and UKPDS studies.36,37 Although effective, however, this method achieves only an approximation of normalcy given the chronic and fluctuating nature of diabetes and the nonphysiologic administration of daily injections vs immediate insulin secretion into the portal circulation in response to a rise in blood glucose in healthy organisms.
Increasing the dietary intake of Pi with 6 g of calcium diphosphate has been shown to be of no benefit since the maximal reabsorptive capacity for Pi in the kidney tubules does not increase, but actually decreases leading to a significant increase in Pi clearance (Figure 1).20 This effect was not found to be related to the serum level of parathyroid hormone or growth hormone, but inversely correlated with the degree of hyperglycemia.
The study of the effect of etidronate disodium (EHDP) superficially appears to be more promising since EHDP increased both plasma Pi and red cell 2, 3-diphophoglycerate levels and thereby the oxygen release capacity. Studies of nonproliferative retinopathy suggested that a 6-month course of EHDP administration may improve retinopathy.25 EHDP in large doses may also suppress bone turnover, inhibits bone mineralization and affect tooth development, deleterious effects that discourage general use.38,39
The therapeutic use of fructose-1, 6 diphosphate (FDP) in diabetes is much more attractive since it is a natural intermediate in glycolysis and also acts as human bioenergy, delivering intracellular phosphorus and 4 moles ATP per mole of FDP. The substance is well tolerated at pharmacological doses and can be given orally. Future controlled studies may lead to a prophylactic therapy to prevent vascular complications and may also counteract the metabolic disturbances leading to type 2 diabetes and the metabolic syndrome.40
Footnotes
Disclosures
The authors declare no conflicts of interest.
References
- 1.Ditzel J. Effect of plasma inorganic phosphate on tissue oxygenation during recovery from diabetic ketoacidosis. Adv Exp Med Biol. 1973;37A:163–172. doi: 10.1007/978-1-4684-3288-6_20. [DOI] [PubMed] [Google Scholar]
- 2.Ditzel J, Standl E. The problem of tissue oxygenation in diabetes mellitus. II Evidence of disordered oxygen release from the erythrocytes of diabetics in various conditions of metabolic control. Acta Med Scand. 1975;(Suppl 578):S59–68. doi: 10.1111/j.0954-6820.1975.tb06503.x. [DOI] [PubMed] [Google Scholar]
- 3.Ditzel J, Brøchner-Mortensen J, Rødbro P. Elevated glomerular filtration rate in early diabetes may be explained by increased sodium reabsorption secundary to impairment in renal tubular handling of phosphate. Hormone Metab Res. 1981;(Suppl 11):S87–89. [PubMed] [Google Scholar]
- 4.Ditzel J, Brøchner-Mortensen J, Kawahara R. Dysfunction of tubular phosphate reabsorption related to glomerular filtration and blood glucose control in diabetic children. Diabetologia. 1982;23(5):406–410. doi: 10.1007/BF00260952. [DOI] [PubMed] [Google Scholar]
- 5.Ditzel J, Brøchner-Mortensen J. Tubular reabsorption rates as related to elevated glomerular filtration in diabetic children. Diabetes. 1983;32 (Suppl 2):S28–S32. doi: 10.2337/diab.32.2.s28. [DOI] [PubMed] [Google Scholar]
- 6.Mathiassen B, Nielsen S, Johansen JS, et al. Long-term bone loss in insulin-dependent diabetic patients with microvascular complications. J Diabet Complications. 1990;4(4):145–149. doi: 10.1016/0891-6632(90)90012-t. [DOI] [PubMed] [Google Scholar]
- 7.Hardy HA, Wellman M. Oxidative phosphorylation. Role of inorganic phosphate and acceptor systems in control of metabolic rates. J Biol Chem. 1952;185:215–224. [PubMed] [Google Scholar]
- 8.Sestoft L. Regulation of fructose metabolism in the perfused rat liver. Interrelation with inorganic phosphate, glucose, ketone body and ethanol metabolism. Biochem Biophys Acta. 1974;343(1):1–16. doi: 10.1016/0304-4165(74)90235-9. [DOI] [PubMed] [Google Scholar]
- 9.Brazy PC, Mandel IJ. Does availability of inorganic phosphate regulate cellular oxidative metabolism? News Physiol Sci. 1986;1:100–103. [Google Scholar]
- 10.Bose S, French S, Evans FJ, Balaban RS. Metabolic network control of oxidative phosphorylation: Multiple roles of inorganic phosphate. J Biol Chem. 2003;278(40):39155–39165. doi: 10.1074/jbc.M306409200. [DOI] [PubMed] [Google Scholar]
- 11.Sestoft L. Is the relationship between plasma concentration of inorganic phosphate and the rate of oxygen consumption of significance in regulating energy metabolism in mammals? Scand J Clin Invest. 1979;39(3):191–197. doi: 10.3109/00365517909106093. [DOI] [PubMed] [Google Scholar]
- 12.Firmat G, Prunier J, Rawson RW, Roberts KE, Schwartz MK. Effect of phosphate enhancing the action of triiodothyronine. Endocrinology. 1956;59(5):565–570. doi: 10.1210/endo-59-5-565. [DOI] [PubMed] [Google Scholar]
- 13.Mosekilde L, Christensen MS. Decreased parathyroid function in hyperthyroidism: interrelationshipp between parathyroid hormone, calcium-phosporous metabolism and thyroid function. Acta Endocrinol (Copenh) 1977;84(3):566–575. doi: 10.1530/acta.0.0840566. [DOI] [PubMed] [Google Scholar]
- 14.Ditzel J, Standl E. Plasma Pi and erythrocyte 2, 3DPG concentrations of non-acidotic diabetics in various degree of metabolic control. Clin Chem. 1976;22(4):550–551. [PubMed] [Google Scholar]
- 15.Ditzel J, Jæger P, Standl E. An adverse effect of insulin on the oxygen-release capacity of red blood cells in nonacidotic diabetics. Metabolism. 1978;27(8):927–934. doi: 10.1016/0026-0495(78)90136-1. [DOI] [PubMed] [Google Scholar]
- 16.Gertner JM, Tamborlane WV, Horst RL, Sherwin RS, Felig P, Genel M. Mineral metabolism in diabetes mellitus. Changes accompanying treatment with portable subcutaneous insulin infusion. J Clin Endocrinol Metab. 1980;50(5):862–866. doi: 10.1210/jcem-50-5-862. [DOI] [PubMed] [Google Scholar]
- 17.Raskin P, Pak CYC. The effect of chronic insulin therapy on phosphate metabolism in diabetes mellitus. Diabetologia. 1981;21(1):50–53. doi: 10.1007/BF03216224. [DOI] [PubMed] [Google Scholar]
- 18.Nagasaka S, Murakami T, Uchikawa T, Ishikawa SE, Sato T. Effect of glycemic control on calcium and phosphorous handling and parathyroid level in patients with non-insulin-depemdemt diabetes mellitus. Endocr J. 1995;42(3):377–383. doi: 10.1507/endocrj.42.377. [DOI] [PubMed] [Google Scholar]
- 19.Ditzel J. The problem of tissue oxygenation in diabetes mellitus III. Acta Med Scand. 1975;(Suppl 578):S69–83. doi: 10.1111/j.0954-6820.1975.tb06503.x. [DOI] [PubMed] [Google Scholar]
- 20.Ditzel J, Lervang HH, Brøchner-Mortensen J, Rødbro P. The influence of a dietary supplement of calcium and phosphate on bone mineral content and mineral homeostasis of diabetic children. Diabetologia. 1993;(Suppl 1):A58. [Google Scholar]
- 21.Altman RD, Johnson CC, Khairi MRA, Wellman M, Serafini AN, Sankey RR. Influence of disodium etidronate (EHDPTM) on clinical and laboratory manifestastion of Paget’s disease of bone (osteitis deformans) N Engl J Med. 1973;289(26):1379–1384. doi: 10.1056/NEJM197312272892601. [DOI] [PubMed] [Google Scholar]
- 22.Recker RR, Hassing GS, Lau JR, Saville PD. The hyperphosphatemic effect of disodium-1-hydroxy-1, 1 diphophonate (EHDPTM): Renal handling of phosphorus and the renal response to parathyroid hormone. J Lab Clin Med. 1973;81(2):258–266. [PubMed] [Google Scholar]
- 23.Russell RGG, Smith R, Preston C, Walton RJ, Woods CG. Diphosphonates in Paget’s disease. Lancet. 1974;1(7863):894–898. doi: 10.1016/s0140-6736(74)90347-x. [DOI] [PubMed] [Google Scholar]
- 24.Ditzel J, Hau C, Daugaard N. Effect of diphosphonate ethane-1-hydroxy-1, 1-diphosphonate (EHDP) on hemoglobin oxygen affinity of diabetic and healthy subjects. Microvasc Res. 1977;13(4):355–361. doi: 10.1016/0026-2862(77)90102-9. [DOI] [PubMed] [Google Scholar]
- 25.Nielsen NV, Ditzel J, Jensen S, Kjærgaard JJ. The effect of etidronate disodium (EHDP) on retinopathy in insulin-dependent diabetic patients. Graefe’s Arch Clin Exp Ophthalmol. 1982;219(2):60–63. doi: 10.1007/BF02173441. [DOI] [PubMed] [Google Scholar]
- 26.Natelson S, Klein M, Kramer B. The effect of oral administration of calcium fructose diphosphate on the serum organic phosphate, inorganic phosphate, calcium protein, and citric acid levels. J Clin Invest. 1951;30(1):50–54. doi: 10.1172/JCI102415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hardin C, Lazzarino G, Tavazzi B, Di Pierro D, Roberts TM, Giardino B, et al. Myocardial metabolism of exogenous FDP is consistent with transport by a dicarboxylate transporter. Am J Physiol. 2001;281(6):H2654–H2660. doi: 10.1152/ajpheart.2001.281.6.H2654. [DOI] [PubMed] [Google Scholar]
- 28.Markow AK, Neely WA, Didioki RH, TerryIII J, Causey A, Lehan PH. Metabolic responses of Fructose-1, 6-diphosphate in healthy subjects. Metabolism. 2000;49(6):698–703. doi: 10.1053/meta.2000.6249. [DOI] [PubMed] [Google Scholar]
- 29.Ehringer WD, Niu W, Chiang B, Wang OL, Gordon L, Chien S. Membrane permeability of fructose.1, 6.diphosphate in lipid vesicles and endothelial cells. Mol Cell Biochem. 2000;210(1–2):35–45. doi: 10.1023/a:1007059214754. [DOI] [PubMed] [Google Scholar]
- 30.Takeuchi K, Cao-Danh H, Frichs I, Glynn P, DÀgostino D, Simplaceanu E, et al. Administration of fructose 1,6-diphosphate during early reperfusion significantly improves recovery of contractile function in the postischemic heart. J Thorac Cardiovasc Surg. 1998;116(2):335–339. doi: 10.1016/s0022-5223(98)70135-7. [DOI] [PubMed] [Google Scholar]
- 31.Antunes N, Martinusso CA, Takiya CM, de Silva AJR, de Omellas JFR, Elias PR, et al. Fructose-1, 6 diphosphate as a protective agent for experimental ischemic acute renal failure. Kidney Int. 2006;69(1):68–72. doi: 10.1038/sj.ki.5000013. [DOI] [PubMed] [Google Scholar]
- 32.Rao MR, Olinde KD, Markov AK. In vitro induction of nitric oxide by fructose-1, 6-diphosphate in the cardiovascular system of rats. Mol Cell Biochem. 1998;185(1–2):171–175. doi: 10.1023/a:1006840802595. [DOI] [PubMed] [Google Scholar]
- 33.Cacioli D, Clivate A, Pelosi P, Megevand J, Galeone M. Haemorheological effects of fructose-1, 6-diphosphate in patients with lower extremity ischemia. Curr Med Res Opin. 1988;10(10):668–674. doi: 10.1185/03007998809111117. [DOI] [PubMed] [Google Scholar]
- 34.Urso L, Brillante C, Orlandi M, Rotundo M, Ballati S. Evaluation of fructose-1, 6-diphosphate on erythrocyte 2, 3 diphosphoglycerate and ATP in surgical orthopedic patients. Aggressologie. 1982;23(3):115–117. [PubMed] [Google Scholar]
- 35.Barrett PQ, Aronson PS. Glucose and alanine inhibition of phosphate transport in renal microvillus membrane vesicles. Am J Physiol. 1982;242(2):F126–F131. doi: 10.1152/ajprenal.1982.242.2.F126. [DOI] [PubMed] [Google Scholar]
- 36.The Diabetes Control and Complication Trial Research Group. The effect of intensive treatment of diabetes in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977–986. doi: 10.1056/NEJM199309303291401. [DOI] [PubMed] [Google Scholar]
- 37.UK Prospective Diabetes Study (UKPDS) Group. Intensive blood glucose control with sulfonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes. (UKPDS 33) Lancet. 1998;352(9131):837–853. [PubMed] [Google Scholar]
- 38.Christoffersen J, Christoffersen NR, Ruben J, Arenda J. The effect of EHDP concentration on enamel demineralization in vitro. J Dent Res. 1991;70(2):123–126. doi: 10.1177/00220345910700020501. [DOI] [PubMed] [Google Scholar]
- 39.Hirano T, Turner CH, Forwood HR, Johnson CC, Burr DR. Does suppression bone turnover impair mechanical properties by allowing microdamage accumulation? Bone. 2000;27(1):13–20. doi: 10.1016/s8756-3282(00)00284-2. [DOI] [PubMed] [Google Scholar]
- 40.Håglin L, Lindblad A, Bygren LO. Hypophosphataemia in the metabolic syndrome. Gender differences in body weight and blood glucose. Eur J Clin Nutr. 2001;55(6):493–498. doi: 10.1038/sj.ejcn.1601209. [DOI] [PubMed] [Google Scholar]