Abstract
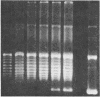
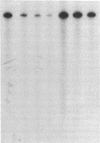
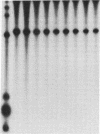
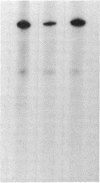
The fraction DE-B obtained by fractionating an extract from rat mammary adenocarcinoma cells on a DEAE-Sephadex column was used for transcribing linear and supercoiled rRNA gene (rDNA). This fraction, which is known to contain RNA polymerase I and essential transcription factors, also contains DNA topoisomerase I activity. Inhibition of this topoisomerase activity by the selective inhibitor camptothecin markedly diminished transcription of supercoiled rDNA, and at a concentration of 150 microM, camptothecin almost completely inhibited DNA topoisomerase I activity and supercoiled rDNA transcription. Addition of exogenous calf thymus DNA topoisomerase I to the sample containing the drug restored the ability of the extract to transcribe supercoiled rDNA. Camptothecin, even at a concentration of 500 microM, had no significant effect on the transcription of linear rDNA. These studies show that relaxation of supercoiled rDNA by DNA topoisomerase I is essential for its transcription. The preferential inhibition of rRNA synthesis in vivo following treatment with camptothecin is probably due to selective camptothecin inhibition of DNA topoisomerase I activity.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abelson H. T., Penman S. Selective interruption of high molecular weight RNA synthesis in HeLa cells by camptothecin. Nat New Biol. 1972 May 31;237(74):144–146. doi: 10.1038/newbio237144a0. [DOI] [PubMed] [Google Scholar]
- Abraham J., Feldman J., Nasmyth K. A., Strathern J. N., Klar A. J., Broach J. R., Hicks J. B. Sites required for position-effect regulation of mating-type information in yeast. Cold Spring Harb Symp Quant Biol. 1983;47(Pt 2):989–998. doi: 10.1101/sqb.1983.047.01.113. [DOI] [PubMed] [Google Scholar]
- Cozzarelli N. R. DNA gyrase and the supercoiling of DNA. Science. 1980 Feb 29;207(4434):953–960. doi: 10.1126/science.6243420. [DOI] [PubMed] [Google Scholar]
- Fleischmann G., Pflugfelder G., Steiner E. K., Javaherian K., Howard G. C., Wang J. C., Elgin S. C. Drosophila DNA topoisomerase I is associated with transcriptionally active regions of the genome. Proc Natl Acad Sci U S A. 1984 Nov;81(22):6958–6962. doi: 10.1073/pnas.81.22.6958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gocke E., Bonven B. J., Westergaard O. A site and strand specific nuclease activity with analogies to topoisomerase I frames the rRNA gene of Tetrahymena. Nucleic Acids Res. 1983 Nov 25;11(22):7661–7678. doi: 10.1093/nar/11.22.7661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harland R. M., Weintraub H., McKnight S. L. Transcription of DNA injected into Xenopus oocytes is influenced by template topology. Nature. 1983 Mar 3;302(5903):38–43. doi: 10.1038/302038a0. [DOI] [PubMed] [Google Scholar]
- Horwitz S. B., Chang C. K., Grollman A. P. Studies on camptothecin. I. Effects of nucleic acid and protein synthesis. Mol Pharmacol. 1971 Nov;7(6):632–644. [PubMed] [Google Scholar]
- Hsiang Y. H., Hertzberg R., Hecht S., Liu L. F. Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J Biol Chem. 1985 Nov 25;260(27):14873–14878. [PubMed] [Google Scholar]
- Kurl R. N., Jacob S. T. Accurate initiation of rat ribosomal RNA gene transcription using a fractionated nuclear extract from normal liver and a hepatoma. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1059–1063. doi: 10.1073/pnas.82.4.1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurl R. N., Jacob S. T. Characterization of a factor that can prevent random transcription of cloned rDNA and its probable relationship to poly(ADP-ribose) polymerase. Nucleic Acids Res. 1985 Jan 11;13(1):89–101. doi: 10.1093/nar/13.1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurl R. N., Rothblum L. I., Jacob S. T. A purified fraction containing RNA polymerase I that can accurately transcribe rat ribosomal RNA gene. Proc Natl Acad Sci U S A. 1984 Nov;81(21):6672–6675. doi: 10.1073/pnas.81.21.6672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luchnik A. N., Bakayev V. V., Zbarsky I. B., Georgiev G. P. Elastic torsional strain in DNA within a fraction of SV40 minichromosomes: relation to transcriptionally active chromatin. EMBO J. 1982;1(11):1353–1358. doi: 10.1002/j.1460-2075.1982.tb01322.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller M. T., Pfund W. P., Mehta V. B., Trask D. K. Eukaryotic type I topoisomerase is enriched in the nucleolus and catalytically active on ribosomal DNA. EMBO J. 1985 May;4(5):1237–1243. doi: 10.1002/j.1460-2075.1985.tb03766.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruitt S. C., Reeder R. H. Effect of topological constraint on transcription of ribosomal DNA in Xenopus oocytes. Comparison of plasmid and endogenous genes. J Mol Biol. 1984 Mar 25;174(1):121–139. doi: 10.1016/0022-2836(84)90368-1. [DOI] [PubMed] [Google Scholar]
- Rothblum L. I., Reddy R., Cassidy B. Transcription initiation site of rat ribosomal DNA. Nucleic Acids Res. 1982 Nov 25;10(22):7345–7362. doi: 10.1093/nar/10.22.7345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G. R. DNA supercoiling: another level for regulating gene expression. Cell. 1981 Jun;24(3):599–600. doi: 10.1016/0092-8674(81)90085-4. [DOI] [PubMed] [Google Scholar]
- Wang J. C. DNA topoisomerases. Annu Rev Biochem. 1985;54:665–697. doi: 10.1146/annurev.bi.54.070185.003313. [DOI] [PubMed] [Google Scholar]
- Weintraub H., Cheng P. F., Conrad K. Expression of transfected DNA depends on DNA topology. Cell. 1986 Jul 4;46(1):115–122. doi: 10.1016/0092-8674(86)90865-2. [DOI] [PubMed] [Google Scholar]
- Weisbrod S. Active chromatin. Nature. 1982 May 27;297(5864):289–295. doi: 10.1038/297289a0. [DOI] [PubMed] [Google Scholar]
- Wu R. S., Kumar A., Warner J. R. Ribosome formation is blocked by camptothecin, a reversible inhibitor of RNA synthesis. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3009–3014. doi: 10.1073/pnas.68.12.3009. [DOI] [PMC free article] [PubMed] [Google Scholar]