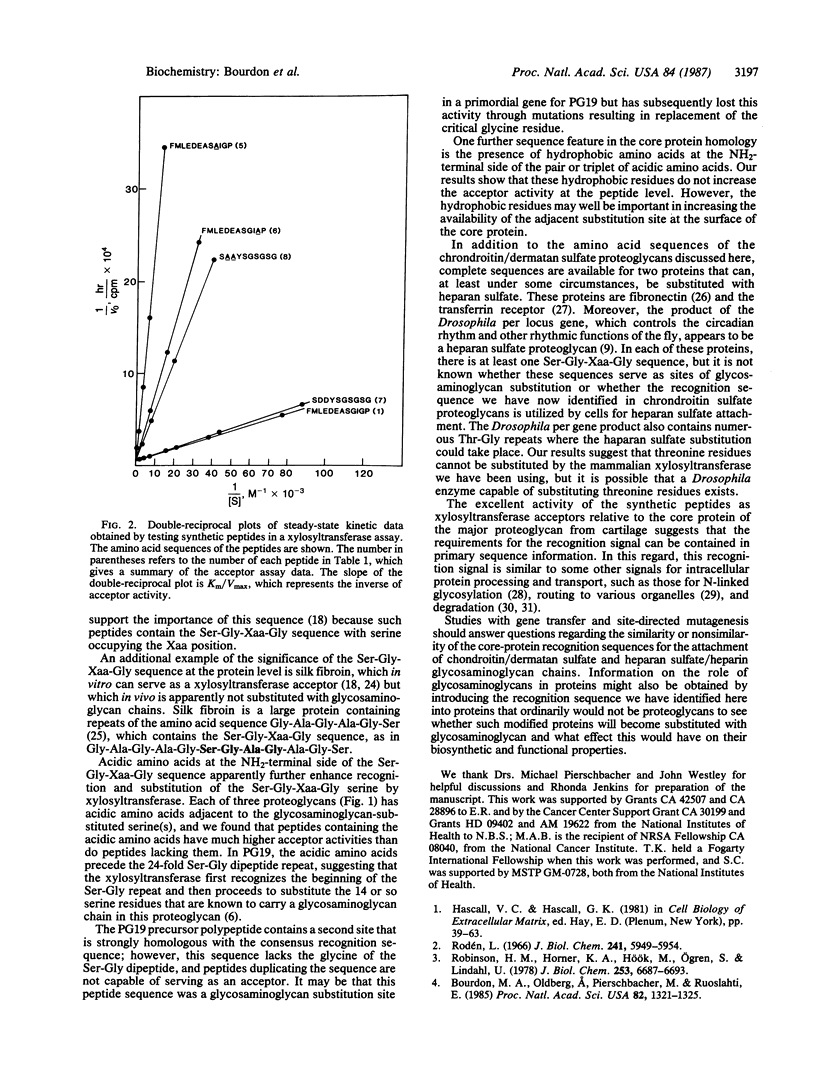
Abstract
Comparison of the amino acid sequences of three different proteoglycan core proteins reveals a 12-amino acid sequence that is about 50% homologous among these proteoglycans. In each of the proteoglycans, this sequence surrounds the serine-glycine dipeptide in which the serine is known or presumed to be substituted with a chondroitin/dermatan sulfate glycosaminoglycan chain. Peptides containing this sequence from two proteoglycans were examined for their ability to serve as acceptors for xylosyltransferase, the enzyme that begins the assembly of glycosaminoglycan chains. Those peptides corresponding to amino acid sequences known to contain glycosaminoglycan-substituted serine residues in the protein were efficient xylosyltransferase acceptors, whereas peptides from sequences with no glycosaminoglycan-substituted serine residues were not. Amino acid substitutions at four critical sites in the acceptor peptides showed that single substitutions could completely abolish acceptor activity or greatly reduce it. The results suggest that the proteoglycan recognition consensus sequence for the attachment of glycosaminoglycans to core proteins consists of acidic amino acids closely followed by the tetrapeptide Ser-Gly-Xaa-Gly, where Xaa is any amino acid. The signal appears to be contained in the primary sequence information. In this regard it resembles a number of other signals for protein processing and intracellular routing.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bachmair A., Finley D., Varshavsky A. In vivo half-life of a protein is a function of its amino-terminal residue. Science. 1986 Oct 10;234(4773):179–186. doi: 10.1126/science.3018930. [DOI] [PubMed] [Google Scholar]
- Backer J. M., Dice J. F. Covalent linkage of ribonuclease S-peptide to microinjected proteins causes their intracellular degradation to be enhanced during serum withdrawal. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5830–5834. doi: 10.1073/pnas.83.16.5830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baenziger J., Kornfeld S. Structure of the carbohydrate units of IgA1 immunoglobulin. II. Structure of the O-glycosidically linked oligosaccharide units. J Biol Chem. 1974 Nov 25;249(22):7270–7281. [PubMed] [Google Scholar]
- Bourdon M. A., Oldberg A., Pierschbacher M., Ruoslahti E. Molecular cloning and sequence analysis of a chondroitin sulfate proteoglycan cDNA. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1321–1325. doi: 10.1073/pnas.82.5.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourdon M. A., Shiga M., Ruoslahti E. Identification from cDNA of the precursor form of a chondroitin sulfate proteoglycan core protein. J Biol Chem. 1986 Sep 25;261(27):12534–12537. [PubMed] [Google Scholar]
- Brennan M. J., Oldberg A., Pierschbacher M. D., Ruoslahti E. Chondroitin/dermatan sulfate proteoglycan in human fetal membranes. Demonstration of an antigenically similar proteoglycan in fibroblasts. J Biol Chem. 1984 Nov 25;259(22):13742–13750. [PubMed] [Google Scholar]
- Campbell P., Jacobsson I., Benzing-Purdie L., Rodén L., Fessler J. H. Silk--a new substrate for UDP-d-xylose:proteoglycan core protein beta-D-xylosyltransferase. Anal Biochem. 1984 Mar;137(2):505–516. doi: 10.1016/0003-2697(84)90119-2. [DOI] [PubMed] [Google Scholar]
- Chopra R. K., Pearson C. H., Pringle G. A., Fackre D. S., Scott P. G. Dermatan sulphate is located on serine-4 of bovine skin proteodermatan sulphate. Demonstration that most molecules possess only one glycosaminoglycan chain and comparison of amino acid sequences around glycosylation sites in different proteoglycans. Biochem J. 1985 Nov 15;232(1):277–279. doi: 10.1042/bj2320277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Claesson L., Larhammar D., Rask L., Peterson P. A. cDNA clone for the human invariant gamma chain of class II histocompatibility antigens and its implications for the protein structure. Proc Natl Acad Sci U S A. 1983 Dec;80(24):7395–7399. doi: 10.1073/pnas.80.24.7395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cossu G., Warren L. Lactosaminoglycans and heparan sulfate are covalently bound to fibronectins synthesized by mouse stem teratocarcinoma cells. J Biol Chem. 1983 May 10;258(9):5603–5607. [PubMed] [Google Scholar]
- Fransson L. A., Carlstedt I., Cöster L., Malmström A. Binding of transferrin to the core protein of fibroblast proteoheparan sulfate. Proc Natl Acad Sci U S A. 1984 Sep;81(18):5657–5661. doi: 10.1073/pnas.81.18.5657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giacoletto K. S., Sant A. J., Bono C., Gorka J., O'Sullivan D. M., Quaranta V., Schwartz B. D. The human invariant chain is the core protein of the human class II-associated proteoglycan. J Exp Med. 1986 Nov 1;164(5):1422–1439. doi: 10.1084/jem.164.5.1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes R. C. Glycoproteins as components of cellular membranes. Prog Biophys Mol Biol. 1973;26:189–268. doi: 10.1016/0079-6107(73)90020-5. [DOI] [PubMed] [Google Scholar]
- Isemura M., Hanyu T., Kosaka H., Ono T., Ikenaka T. Comparative study of carbohydrate-protein complexes. III. Peptide structures of the linkage region in proteoglycans of human, porcine and shark cartilages. J Biochem. 1981 Apr;89(4):1113–1119. [PubMed] [Google Scholar]
- Jackson F. R., Bargiello T. A., Yun S. H., Young M. W. Product of per locus of Drosophila shares homology with proteoglycans. Nature. 1986 Mar 13;320(6058):185–188. doi: 10.1038/320185a0. [DOI] [PubMed] [Google Scholar]
- Krusius T., Ruoslahti E. Primary structure of an extracellular matrix proteoglycan core protein deduced from cloned cDNA. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7683–7687. doi: 10.1073/pnas.83.20.7683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldberg A., Hayman E. G., Ruoslahti E. Isolation of a chondroitin sulfate proteoglycan from a rat yolk sac tumor and immunochemical demonstration of its cell surface localization. J Biol Chem. 1981 Nov 10;256(21):10847–10852. [PubMed] [Google Scholar]
- Olson C. A., Krueger R., Schwartz N. B. Deglycosylation of chondroitin sulfate proteoglycan by hydrogen fluoride in pyridine. Anal Biochem. 1985 Apr;146(1):232–237. doi: 10.1016/0003-2697(85)90420-8. [DOI] [PubMed] [Google Scholar]
- Pearson C. H., Winterbottom N., Fackre D. S., Scott P. G., Carpenter M. R. The NH2-terminal amino acid sequence of bovine skin proteodermatan sulfate. J Biol Chem. 1983 Dec 25;258(24):15101–15104. [PubMed] [Google Scholar]
- Reddy P., Jacquier A. C., Abovich N., Petersen G., Rosbash M. The period clock locus of D. melanogaster codes for a proteoglycan. Cell. 1986 Jul 4;46(1):53–61. doi: 10.1016/0092-8674(86)90859-7. [DOI] [PubMed] [Google Scholar]
- Robinson H. C., Horner A. A., Hök M., Ogren S., Lindahl U. A proteoglycan form of heparin and its degradation to single-chain molecules. J Biol Chem. 1978 Oct 10;253(19):6687–6693. [PubMed] [Google Scholar]
- Rodén L., Koerner T., Olson C., Schwartz N. B. Mechanisms of chain initiation in the biosynthesis of connective tissue polysaccharides. Fed Proc. 1985 Feb;44(2):373–380. [PubMed] [Google Scholar]
- Rodén L., Smith R. Structure of the neutral trisaccharide of the chondroitin 4-sulfate-protein linkage region. J Biol Chem. 1966 Dec 25;241(24):5949–5954. [PubMed] [Google Scholar]
- Sant A. J., Cullen S. E., Schwartz B. D. Identification of a sulfate-bearing molecule associated with HLA class II antigens. Proc Natl Acad Sci U S A. 1984 Mar;81(5):1534–1538. doi: 10.1073/pnas.81.5.1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin H. S., Bargiello T. A., Clark B. T., Jackson F. R., Young M. W. An unusual coding sequence from a Drosophila clock gene is conserved in vertebrates. Nature. 1985 Oct 3;317(6036):445–448. doi: 10.1038/317445a0. [DOI] [PubMed] [Google Scholar]
- Singer P. A., Lauer W., Dembić Z., Mayer W. E., Lipp J., Koch N., Hämmerling G., Klein J., Dobberstein B. Structure of the murine Ia-associated invariant (Ii) chain as deduced from a cDNA clone. EMBO J. 1984 Apr;3(4):873–877. doi: 10.1002/j.1460-2075.1984.tb01899.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiess M., Lodish H. F. An internal signal sequence: the asialoglycoprotein receptor membrane anchor. Cell. 1986 Jan 17;44(1):177–185. doi: 10.1016/0092-8674(86)90496-4. [DOI] [PubMed] [Google Scholar]
- Strubin M., Mach B., Long E. O. The complete sequence of the mRNA for the HLA-DR-associated invariant chain reveals a polypeptide with an unusual transmembrane polarity. EMBO J. 1984 Apr;3(4):869–872. doi: 10.1002/j.1460-2075.1984.tb01898.x. [DOI] [PMC free article] [PubMed] [Google Scholar]



