Abstract
Background:
Bone morphogenetic protein-7 (BMP-7) is a signalling molecule belonging to the transforming growth factor--superfamily. Recent studies have demonstrated the clinical impact of BMP-7 expression in various human cancers. However, there have been few reports detailing this in gastric cancer.
Methods:
We immunohistochemically investigated the expression of BMP-7 in 233 gastric cancer patients to disclose the clinicopathological features of BMP-7-positive gastric cancer.
Results:
Immunohistochemically, in human gastric cancer, BMP-7 expression was identified in cellular membranes but also in the cytoplasm of cancer cells. Bone morphogenetic protein-7-positive expression was found in 129 of 233 patients (55%). Bone morphogenetic protein-7 expression was correlated with tumour size, nodal involvement, lymphatic invasion, venous invasion and histology (P<0.05). Bone morphogenetic protein-7 expression was significantly correlated with patient postoperative outcome, especially in the undifferentiated group. Multivariate analysis revealed BMP-7 expression as one of the independent prognostic factors next to the depth of invasion and nodal involvement (P<0.01).
Conclusions:
From the data collected, it would be appropriate to conclude on the possible regulation of gastric cancer progression by autocrine or paracrine BMP-7 loops. We can use BMP-7 expression as one of the strong predictors of risk of tumour recurrence in gastric cancer.
Keywords: gastric cancer, BMP-7, nodal involvement, histology, prognosis
Gastric cancer is the fourth most common cancer. East Asian countries in particular, including Japan, are the most high-risk areas for gastric cancer. In Japan, the establishment of mass screening by photo fluoroscopy or gastrointestinal fibroscopy has led to gastric cancer being diagnosed asymptomatically and curatively treated with gastrectomy; the survival rate of gastric cancer has drastically improved. However, even for patients who have received curative surgery, the recurrence of gastric cancer occurs (Parkin et al, 2005; Brenner et al, 2009). In this context, it is necessary to find novel cancer-related factors to use as markers for diagnosis and treatment of gastric cancer.
Bone morphogenetic proteins (BMPs) are signalling molecules belonging to the transforming growth factor (TGF)--superfamily, with >30 subtypes in mammals, Drosophila, Xenopus and sea urchin (Wozney et al, 1988; Ducy and Karsenty, 2000). Bone morphogenetic proteins were originally identified as cytokines inducing ectopic chondro-osteogenesis and have a role in skeletal and joint morphogenesis, bone remodelling and fracture repair (Urist, 1965; Chen et al, 2004). However, the function of BMPs is linked not only to bone tissue, but also to cellular homeostasis and embryonic development. Several studies demonstrated that mice deficient in different BMPs exhibited impairments and abnormalities of various organs (Dudley et al, 1995; Luo et al, 1995; McPherron et al, 1997; Solloway and Robertson, 1999).
Expression of BMP-7 is highest in the kidney and it is thought to be related to kidney and eye development and skeletal patterning (Dudley et al, 1995; Luo et al, 1995). Moreover, BMP-7 reverses chronic renal injury by counteracting TGF-1-induced epithelial-to-mesenchymal transition (Zeisberg et al, 2003). Recent studies demonstrated that BMP-7 expression is found in various human cancers, and regulates cell differentiation, proliferation, migration, invasion and apoptosis (Andrews et al, 1994; Ro et al, 2004; Yang et al, 2005, 2006; Grijelmo et al, 2007; Alarmo et al, 2009). However, to our knowledge, there have been no reports demonstrating the association between BMP-7 expression and clinicopathological factors including prognosis in gastric cancer. Thus, the purpose of this study was to investigate BMP-7 expression by immunohistochemistry in gastric cancer and evaluate the clinical impact of BMP-7-positive gastric cancer.
Materials and methods
Detection of mRNA expression of BMP-7 in gastric cancer cell lines
We used five gastric cancer cell lines (KATOIII, NUGC-4, MKN45, MKN74 and MKN7 (Riken Cell Bank, Tsukuba, Japan)) to detect mRNA expression of BMP-7 by RT–PCR. First, five gastric cancer cell lines were cultured in RPMI-1640 (Sigma-Aldrich Co., St Louis, MO, USA) supplemented with antibiotics (100 units ml–1 penicillin and 100 μg ml–1 streptomycin) and 10% fetal bovine serum (MBL, Nagoya, Japan) at 37 °C in a humidified atmosphere of 5% CO2 in air. Proliferated cells were used to detect mRNA expression of BMP-7. Total RNA was extracted from the cell lines using RN easy Mini-Kit (Qiagen, Valencia, CA, USA) according to the manufacturer's instructions. Complementary DNA (0.05 μg μl–1) was synthesised as described previously (Yokomakura et al, 2007). Amplification of BMP-7 and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) cDNA was performed in a total volume of 30 μl, which included 1 μl of the cDNA product sample, 0.5 μM each of BMP-7 primer (Roche, Mannheim, Germany) and GAPDH primer (forward primer: 5′-TTGGTATCGTGGAAGGACTCA-3′ and reverse primer: 5′-TGTCATCATATTTGGCAGGTTT-3′), GeneAmp 10 × PCR Buffer (Applied Biosystems, Foster City, CA, USA), 0.5 U AmpliTaq DNA polymerase (Applied Biosystems) and 25 mM dNTP mixture (Takara Bio Inc., Otsu, Japan). The GAPDH gene served as an internal control. The BMP-7 and GAPDH amplification reactions were 30 cycles (BMP-7) or 26 cycles (GAPDH) of denaturation at 94 °C for 60 s (BMP-7) or 30 s (GAPDH), annealing at 64 °C for 60 s (BMP-7) or 54 °C for 30 s (GAPDH), and elongation at 72 °C for 60 s (BMP-7) or 30 s (GAPDH) in the GeneAmp PCR System 9700 (Applied Biosystems). Amplified DNA fragments were electrophoresed on 1.5% agarose gels containing ethidium bromide with a DNA molecular weight marker for comparison.
Patients and specimens
This study included 233 patients with gastric adenocarcinoma invading deeper than the submucosal layer. All patients underwent curative gastrectomy with lymph node dissection at Kagoshima University Hospital from January 1995 to December 2004 (Table 1). In all, 75 patients underwent distal, 21 proximal, 124 total and 13 partial gastrectomy. A total of 233 gastric cancer patients were classified, including 161 male and 72 female patients (range, 31–85 years; average, 66 years). The final pathological examination disclosed that cases of stage I, II, III and IV gastric cancers numbered 70, 45, 63 and 55, respectively. The patients were histopathologically classified as 118 differentiated (papillary, well-differentiated and moderately differentiated tubular adenocarcinoma) or 115 undifferentiated (poorly differentiated adenocarcinoma, mucinous adenocarcinoma and signet-ring cell carcinoma) according to the Japanese classification of gastric cancer (Japanese Gastric Cancer Association, 1998). The study was approved by the Institutional Review Board of Kagoshima University and performed according to the Helsinki Declaration.
Table 1. Patient characteristics.
| No. | |
|---|---|
| Gender | |
| Male | 161 |
| Female | 72 |
| Age | |
| Mean | 66 |
| Range | 31–85 |
| Operation | |
| Total gastrectomy | 124 |
| Distal | 75 |
| Proximal | 21 |
| Partial | 13 |
| Stage | |
| I | 70 |
| II | 45 |
| III | 63 |
| IV | 55 |
| Histology | |
| Differentiated | 118 |
| Undifferentiated | 115 |
Immunohistochemistry of BMP-7 in gastric cancer and its evaluation
The specimens of the gastric cancer were formalin-fixed and paraffin-embedded tissues; they were cut into 3-μm-thick sections and mounted on glass slides for immunohistochemistry. They were deparaffinised in xylene and dehydrated with a series of graded ethanol. The endogenous peroxidase activity of specimens was blocked by immersing the slides in a 0.3% H2O2 solution in methanol for 30 min at room temperature. After washing three times with phosphate-buffered saline (PBS) for 5 min each, the sections were treated with 1% bovine serum albumin for 30 min to block nonspecific reactions at room temperature. The blocked sections were incubated with the mouse monoclonal antibody against human BMP-7 (1 : 500; R&D Systems, Inc., Minneapolis, MN, USA) and left 24 h at 4 °C, followed by staining with a streptavidin–biotin peroxidase kit (Vector Laboratories, Inc., Burlingame, CA, USA). The sections were washed in PBS for 5 min three times and the immune complex was visualised by incubating the sections with diaminobenzidine tetrahydrochloride. The sections were rinsed briefly in water, counterstained with haematoxylin and mounted. Noncancerous kidney samples were used as positive controls for BMP-7. Bone morphogenetic protein-7 expression was determined by counting the number of cancer cells in which the cytoplasm was stained with the anti-BMP-7 antibody. Evaluation of immunohistochemistry was independently carried out by two investigators (MA and SI). To evaluate this, 10 fields within the tumour were selected, and expression in 1000 cancer cells (100 cells per field) was evaluated using high-power ( × 200) microscopy. The average labelling index of BMP-7 was assessed according to the proportion of positive cells in each field. Bone morphogenetic protein-7 expression was graded as the BMP-7-positive group if >10% of cancer cells were stained or as the BMP-7-negative group if <10% of cancer cells were stained.
Statistical analysis
A statistical analysis of group differences was performed using χ2 test. The Kaplan–Meier method was used for survival analysis and evaluated by the log-rank test. The Cox proportional hazard model was used in multivariate analysis. P<0.05 was considered statistically significant.
Results
BMP-7 expression in gastric cancer and its association with clinical results
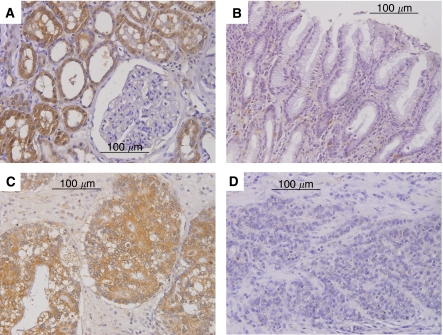
Immunohistochemically, in human gastric cancer, BMP-7 expression was identified in cellular membranes but also in the cytoplasm of cancer cells (Figure 1). According to the immunohistochemical evaluation, 129 of 233 patients (55%) were placed in the BMP-7-positive group. Bone morphogenetic protein-7 expression correlated with clinicopathological variables. Namely, tumour diameter, nodal involvement, lymphatic invasion and venous invasion were significantly greater in the BMP-7-positive group than in the BMP-7-negative group (P=0.01, <0.01, <0.01 and <0.01, respectively). Moreover, BMP-7 expression was significantly higher in the differentiated histology group than in the undifferentiated group (P<0.05). No significant difference was observed regarding age, gender or tumour depth (Table 2). The overall survival rate was significantly lower in the BMP-7-positive group than in the BMP-7-negative group (P<0.01; Figure 2A). Furthermore, we evaluated the correlation between expression of BMP-7 and prognosis in the differentiated and the undifferentiated histology groups. In the differentiated histology group, BMP-7 expression was not associated with postoperative outcome (Figure 2B). On the other hand, in the undifferentiated group, the BMP-7-positive group had significantly poorer survival than the BMP-7-negative group (P=0.01) (Figure 2C).
Figure 1.
Expression of BMP-7 in clinical samples. Immunostaining of BMP-7 (original magnification, × 400): Examples of (A) noncancerous kidney tissue, (B) noncancerous gastric epithelium, (C) BMP-7-positive gastric cancer and (D) BMP-7-negative gastric cancer are shown. Staining is detected in cell membranes and the cytoplasm (A, C; they obviously stain brown).
Table 2. Correlation between expression of BMP-7 and clinical factors.
|
Expression of BMP-7
|
|||
|---|---|---|---|
| Clinical factors | Positive (%) n=129 (55) | Negative n=104 | P-value |
| Age | |||
| <65 | 40 (48) | 44 | 0.08 |
| ⩾65 | 89 (60) | 60 | |
| Gender | |||
| Male | 94 (58) | 67 | 0.2 |
| Female | 35 (49) | 37 | |
| Tumour size | |||
| <50 mm | 40 (44) | 50 | 0.01 |
| ⩾50 mm | 89 (62) | 54 | |
| Depth | |||
| T1 (sm)/T2 (mp) | 85 (57) | 65 | 0.68 |
| T3 (ss)/T4 (se) | 44 (53) | 39 | |
| Nodal involvement | |||
| Yes | 87 (63) | 52 | <0.01 |
| No | 42 (45) | 52 | |
| Lymphatic invasion | |||
| Yes | 107 (61) | 69 | <0.01 |
| No | 22 (39) | 35 | |
| Venous invasion | |||
| Yes | 88 (67) | 44 | <0.01 |
| No | 41 (41) | 60 | |
| Histology | |||
| Differentiated | 73 (62) | 45 | <0.05 |
| Undifferentiated | 56 (49) | 59 | |
Abbreviation: BMP-7=bone morphogenetic protein-7.
Figure 2.
Postoperative survival curves of patients according to their expression of BMP-7 in gastric cancer. The BMP-7-positive group had significantly lower overall survival than the BMP-7-negative group (P<0.01, log-rank test) (A). In the differentiated group, no significant difference was observed in overall survival (P=0.11, log-rank test) (B). In the undifferentiated group, the BMP-7-positive group had significantly lower overall survival than the BMP-7-negative group (P=0.01, log-rank test) (C).
Univariate and multivariate analyses of survival
Table 3 shows the results of univariate and multivariate analyses of factors related to patient prognosis. Univariate analysis showed that tumour histology, the depth of invasion, tumour size, lymph node metastasis, lymphatic invasion, venous invasion and BMP-7 expression were significantly related to postoperative survival (P<0.01). Multivariate analysis indicated that BMP-7 expression was one of the independent prognostic factors of overall survival for the patients with gastric cancer next to the depth of invasion and nodal involvement (P<0.01).
Table 3. Univariate and multivariate analysis of prognostic factor in gastric cancer.
| Clinical factors | Univariate P | Multivariate P | Hazard ratio | 95% Confidence interval |
|---|---|---|---|---|
| Age | 0.4 | — | — | — |
| Gender | 0.88 | — | — | — |
| Histology | <0.01 | <0.05 | 1.893 | 1.021–3.510 |
| Depth | <0.01 | <0.01 | 3.239 | 1.650–6.361 |
| Tumour size | <0.01 | 0.34 | 1.518 | 0.641–3.595 |
| Nodal involvement | <0.01 | <0.01 | 3.868 | 1.450–10.321 |
| Lymphatic invasion | <0.01 | — | — | — |
| Venous invasion | <0.01 | 0.18 | 1.171 | 0.555–2.474 |
| BMP-7 | <0.01 | <0.01 | 2.934 | 1.524–5.648 |
Abbreviation: BMP-7=bone morphogenetic protein-7.
Discussion
Bone morphogenetic protein-7 induces differentiation of mesenchymal cells to osteoblastic cells in bone and cartilage tissue (Ducy and Karsenty, 2000). Moreover, in kidney, it is related to MET and reverses chronic renal injury (Zeisberg et al, 2003). On the other hand, BMP-7 expression has been identified and clinical features of BMP-7 expression in several human cancers such as osteosarcoma, malignant melanoma, breast cancer, prostate cancer, colorectal cancer and renal cell cancer have been discussed (Sulzbacher et al, 2002; Masuda et al, 2004; Alarmo et al, 2006; Kwak et al, 2007; Rothhammer et al, 2007; Motoyama et al, 2008 ). In this study, robust expression of the BMP-7 mRNA was identified by RT–PCR in MKN45 and MKN74 gastric cancer cell lines, which were derived from liver metastasis (data not shown).
We showed that BMP-7 expression was significantly associated with clinical factors such as lymphatic invasion, venous invasion and nodal involvement. Their previous studies demonstrated that BMP-7 promoted breast cancer cell migration and invasion, prostate cancer cell mobility and related metastasis in colorectal cancer (Yang et al, 2005; Grijelmo et al, 2007; Alarmo et al, 2009). In this context, BMP-7 expression may be connected with tumour aggressiveness beyond the specific organ. In this study, we showed that BMP-7 expression strongly correlated with nodal involvement. Thus, BMP-7 expression of biopsy may be informative to predict lymph node metastasis, when we should choose between endoscopic resection or gastrectomy for early gastric cancer.
As a prognostic parameter, Motoyama et al (2008) reported that overexpression of BMP-7 mRNA was significantly associated with lower overall survival in colorectal cancer. Moreover, BMP-7 expression by immunohistochemistry was also significantly associated with lower recurrence-free survival in malignant melanoma and breast cancer (Rothhammer et al, 2007; Alarmo et al, 2008). We showed that BMP-7 expression in gastric cancer was an independent prognostic marker in accordance with results for other cancers. Only in renal cell carcinoma was BMP-7 expression significantly associated with better surgical outcome (Kwak et al, 2007). This may be explained by the fact that normal kidney cell usually highly expresses BMP-7 unlike normal gastric mucosa. Such differences might have caused the inverse result in prognostic implication.
In this study, BMP-7-positive gastric cancer was shown to be correlated with well-differentiated tumour histology. Andrews et al (1994) demonstrated that BMP-7-induced differentiation of pluripotent human embryonal carcinoma cells. Moreover, Lombardo et al (2011) reported that BMP-4-induced differentiation of colorectal cancer stem cells and increased their response to chemotherapy in mice. We demonstrated that BMP-7 expression was a more significant prognostic factor only in the undifferentiated group (Figures 2B and C). In short, although BMP-7 was highly expressed in the BMP-7-positive and undifferentiated group, the differentiation of this group was restricted, and the prognosis was poor. In the undifferentiated group, other factors that are associated with regulation of BMP signalling pathway might be associated with this result. The BMP signalling pathway is regulated in a complex manner by extracellular and intracellular factors and crosstalk with other signalling pathways, including TGF--signalling pathway (Zwijsen et al, 2003; Cao and Chen, 2005; Gazzerro and Canalis, 2006; Lombardo et al, 2011; Luo et al, 2010). It is necessary to further study the relationship between BMP signalling pathway and its related factors to open the view of chemotherapy in gastric cancer.
In conclusion, from the data collected, it would be appropriate to conclude on the possible regulation of gastric cancer progression by autocrine or paracrine BMP-7 loops. We can use BMP-7 expression as a predictor of lymph node metastasis and postoperative outcome in gastric cancer. As previously mentioned, the signals activated by BMP-7 are involve intracellular and extracellular factors, so further analysis seems to be necessary to determine the mechanism involved.
References
- Alarmo EL, Korhonen T, Kuukasjävi T, Huhtala H, Holli K, Kallioniemi A (2008) Bone morphogenetic protein 7 expression associates with bone metastasis in breast carcinomas. Ann Oncol 19(2): 308–314 [DOI] [PubMed] [Google Scholar]
- Alarmo EL, Pärssinen J, Ketolain JM, Savinainen K, Karhu R, Kallioniemi A (2009) BMP7 influences proliferation, migration, and invasion of breast cancer cells. Cancer Lett 275(1): 35–43 [DOI] [PubMed] [Google Scholar]
- Alarmo EL, Rauta J, Kauraniemi P, Karhu R, Kuukasjävi T, Kallioniemi A (2006) Bone morphogenetic protein 7 is widely overexpressed in primary breast cancer. Genes Chromosomes Cancer 45(4): 411–419 [DOI] [PubMed] [Google Scholar]
- Andrews PW, Damjanov I, Berends J, Kumpf S, Zappavigna V, Mavilio F, Sampath K (1994) Inhibition of proliferation and induction of differentiation of pluripotent human embryonal carcinoma cells by osteogenic protein-1 (or bone morphogenetic protein-7). Lab Invest 71(2): 243–251 [PubMed] [Google Scholar]
- Brenner H, Rothenbacher D, Arndt V (2009) Epidemiology of stomach cancer. Methods Mol Biol 472: 467–477 [DOI] [PubMed] [Google Scholar]
- Cao X, Chen D (2005) The BMP signaling and in vivo bone formation. Gene 357: 1–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D, Zhao M, Mundy GR (2004) Bone morphogenetic proteins. Growth Factors 22: 233–241 [DOI] [PubMed] [Google Scholar]
- Ducy P, Karsenty G (2000) The family of bone morphogenetic proteins. Kidney Int 57: 2207–2214 [DOI] [PubMed] [Google Scholar]
- Dudley AT, Lyons KM, Robertson EJ (1995) A requirement for bone morphogenetic protein-7 during development of the mammalian kidney and eye. Genes Dev 9(22): 2795–2807 [DOI] [PubMed] [Google Scholar]
- Gazzerro E, Canalis E (2006) Bone morphogenetic proteins and their antagonists. Rev Endcr Metab Disord 7(1–2): 51–65 [DOI] [PubMed] [Google Scholar]
- Grijelmo C, Rodrigue C, Svrcek M, Bruyneel E, Hendrix A, de Wever O, Gespach C (2007) Proinvasive activity of BMP-7 through SMAD4/src-independent and ERK/Rac/JNK-dependent signaling pathways in colon cancer cells. Cell Signal 19(8): 1722–1732 [DOI] [PubMed] [Google Scholar]
- Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma –2nd English edition. Gastric Cancer 1: 10–24 [DOI] [PubMed] [Google Scholar]
- Kwak C, Park YH, Kim IY, Moon KC, Ku JH (2007) Expression of bone morphogenetic proteins, the subfamily of the transforming growth factor-beta superfamily, in renal cell carcinoma. J Urol 178(3 Part 1): 1062–1067 [DOI] [PubMed] [Google Scholar]
- Lombardo Y, Scopelliti A, Cammareri P, Tadaro M, Iovino F, Ricci-Vitiani L, Gulotta G, Dieli F, De Maria R, Stassi G (2011) BMP4 induces differentiation of colorectal cancer stem cells and increases their response to chemotherapy in mice. Gastroenterology 140(1): 279–309 [DOI] [PubMed] [Google Scholar]
- Luo DD, Phillips A, Fraser D (2010) Bone morphogenetic protein-7 inhibits proximal tubular epithelial cell Smad3 signaling via SnoN expression. Am J Pathol 176(3): 1139–1147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G, Hoffmann C, Bronckers AL, Sohochi M, Bradley A, Karsent G (1995) BMP-7 is an inducer of nephrogenesis, and is also required for eye development and skeletal patterning. Genes Dev 9: 2808–2820 [DOI] [PubMed] [Google Scholar]
- Masuda H, Hukabori Y, Nakano K, Shimizu N, Yamanaka H (2004) Expression of bone morphogenetic protein-7 (BMP-7) in human prostate. Prostate 59(1): 101–106 [DOI] [PubMed] [Google Scholar]
- McPherron AC, Lawler AM, Lee SJ (1997) Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387(6628): 83–90 [DOI] [PubMed] [Google Scholar]
- Motoyama K, Tanaka F, Kosaka Y, Mimori K, Uetake H, Inoue H, Sugihara K, Mori M (2008) Clinical significance of BMP7 in human colorectal cancer. Ann Surg Oncol 15(5): 1530–1537 [DOI] [PubMed] [Google Scholar]
- Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55(2): 74–108 [DOI] [PubMed] [Google Scholar]
- Ro TB, Holt RU, Brenne AT, Hjorth-Hansen H, Waage A, Hjertner O, Sundan A, Borset M (2004) Bone morphogenetic protein-5, -6 and -7 inhibit growth and induce apoptosis in human myeloma cells. Oncogene 23(17): 3024–3032 [DOI] [PubMed] [Google Scholar]
- Rothhammer T, Wild PJ, Meyer S, Bataille F, Pauer A, Klinkhammer-Schalke M, Hein R, Hofstaedter F, Bosserhoff AK (2007) Bone morphogenetic protein 7 (BMP7) expression is a potential novel prognostic marker for recurrence in patients with primary melanoma. Cancer Biomark 3(2): 111–117 [DOI] [PubMed] [Google Scholar]
- Solloway MJ, Robertson EJ (1999) Early embryonic lethality in Bmp5: Bmp7 double mutant mice suggests functional redundancy within the 60A subgroup. Development 126(8): 1753–1768 [DOI] [PubMed] [Google Scholar]
- Sulzbacher I, Birner P, Trieb K, Pichlbauer E, Lang S (2002) The expression of bone morphogenetic proteins in osteosarcoma and its relevance as a prognostic parameter. J Clin Pathol 55(5): 381–385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urist MR (1965) Bone: formation by autoinduction. Science 150: 893–899 [DOI] [PubMed] [Google Scholar]
- Wozney JM, Rosen V, Celeste AJ, Mitsock LM, Whitters MJ, Kriz RW, Hewick RW, Wang EA (1988) Novel regulators of bone formation: molecular clones and activities. Science 242(4885): 1528–1534 [DOI] [PubMed] [Google Scholar]
- Yang S, Lim M, Pham LK, Kendall SE, Reddi AH, Altieri DC, Roy-Burman P (2006) Bone morphogenetic protein 7 protects prostate cancer cells from stress-induced apoptosis via both Smad and c-Jun NH2-terminal kinase pathways. Cancer Res 66(8): 4285–4290 [DOI] [PubMed] [Google Scholar]
- Yang S, Zhong C, Frenkel B, Reddi AH, Roy-Burman P (2005) Diverse biological effect and Smad signaling of bone morphogenetic protein 7 in prostate tumor cells. Cancer Res 65(13): 5769–5777 [DOI] [PubMed] [Google Scholar]
- Yokomakura N, Natsugoe S, Okumura H, Ikeda R, Uchikado Y, Mataki Y, Takatori H, Matsumoto M, Owaki T, Ishigami S, Aikou T (2007) Improvement in radiosenseitivity using small interfering RNA targeting p53R2 in esophageal squamous cell carcinoma. Oncol Rep 18(3): 561–567 [PubMed] [Google Scholar]
- Zeisberg M, Hanai J, Sugimoto H, Mammoto T, Charytan D, Strutz F, Kalluri R (2003) BMP-7 counteracts TGF-β1-induced epithelial-to-mesenchymal transition and reverses chronic renal injury. Nat Med 9(7): 964–968 [DOI] [PubMed] [Google Scholar]
- Zwijsen A, Verchueren K, Huylebroeck D (2003) New intracellular components of bone morphogenetic protein/Smad signaling cascades. FEBS Lett 546: 133–139 [DOI] [PubMed] [Google Scholar]