Abstract
Background & Aims
Aspirin and non-steroidal anti-inflammatory drugs (NSAIDs) lower risk of colorectal cancer (CRC). We investigated whether plasma inflammatory markers were associated with risk of CRC and if use of anti-inflammatory drugs was differentially associated with risk of CRC according to baseline levels of inflammatory markers.
Methods
We measured levels of high-sensitivity C-reactive protein (CRP), interleukin (IL)-6, and the soluble tumor necrosis factor receptor-2 (sTNFR-2) in blood samples from 32,826 women, collected from 1989 to 1990. Through 2004, we documented 280 cases of incident CRC; each case was matched for age to 2 randomly selected participants without cancer (controls). Information on anti-inflammatory drug (aspirin and NSAIDs) use was collected biennially.
Results
Compared to women in the lowest quartile of plasma levels of sTNFR-2, women in the highest quartile had an increased risk of CRC (multivariate relative risk [RR], 1.67; 95% confidence interval [CI], 1.05–2.68; P for the trend=0.03). Among women with high baseline levels of sTNFR-2, those that initiated aspirin/NSAID use after blood collection had significant reductions in subsequent risk of CRC (multivariate RR, 0.39; 95% CI, 0.18–0.86). In contrast, among women with low baseline levels of sTNFR-2, initiation of aspirin/NSAID use was not associated with significant risk reduction (multivariate RR, 0.86; 95% CI, 0.41–1.79). Plasma levels of CRP and IL-6 were not significantly associated with CRC risk.
Conclusions
Plasma levels of sTNFR-2, but not CRP or IL-6, are associated with an increased risk of CRC. Anti-inflammatory drugs appear to reduce risk of CRC among women with high, but not low, baseline levels of sTNFR-2. Certain subsets of the population, defined by inflammatory markers, might obtain different benefits from anti-inflammatory drugs.
Keywords: Colon cancer, prevention, inflammation, chemoprevention
INTRODUCTION
Aspirin and NSAID use reduces risk of colorectal adenoma and cancer.1–5 This effect may be mediated through abrogation of chronic inflammation. Considerable experimental, epidemiological, and clinical data provide strong evidence for a causative link between chronic inflammation and cancer, including the well-described association between inflammatory bowel disease and colonic dysplasia.6 Beyond inflammatory bowel disease, however, the importance of inflammation in sporadic colorectal cancer remains undefined. Inflammation may predispose to cancer through enhanced cellular proliferation and mutagenesis, inability to adapt to oxidative stresses, promotion of angiogenesis, inhibition of apoptosis, and secretion of mediators that may promote tumorigenesis.7
Chronic inflammation is characterized by abnormal production of circulating inflammatory factors that have been causally linked to obesity, diabetes, and vascular disease in animal models.8, 9 In humans, cytokines such as CRP, IL-6, and sTNFR-2 not only mediate the inflammatory response, but also serve as potential biomarkers of these chronic diseases.10–12 Although diabetes and vascular disease are associated with risk of colorectal cancer,13, 14 prospective studies specifically relating inflammatory markers to colorectal cancer have been equivocal. For CRP, some prospective studies have observed an increased risk of colorectal cancer,15–20 whereas other studies have shown an equivocal,21–23 non-existent,24, 25 or somewhat inverse association.26 The few studies that have also examined IL-6 have not observed strong relationships.18, 22 To our knowledge, no studies have specifically investigated sTNFR-2 in relation to risk.
Thus, to further elucidate the role of inflammation and anti-inflammatory drugs in relation to colorectal carcinogenesis, we examined whether CRP, IL-6, or sTNFR-2 levels are independently associated with risk of colorectal cancer in a nested case-control analysis among women enrolled in the Nurses’ Health Study (NHS) who provided a blood sample at baseline and were followed for 14 years. Because women provided aspirin/NSAID data both before and after collection of plasma, we also had the unique opportunity to prospectively evaluate the relationship between baseline levels of inflammatory markers and subsequent response to aspirin and NSAIDs on risk of colorectal cancer.
METHODS
Study Participants
Participants were drawn from the NHS, which began in 1976 when 121,701 U.S. female registered nurses, aged 30 to 55 years, completed a mailed questionnaire about their health history. We have mailed follow-up questionnaires to the participants every 2 years to update information on lifestyle factors, medication usage, and diagnoses of colorectal cancer and other diseases. In 1989 through 1990, we collected a blood specimen from 32,826 participants.27 As previously detailed,28 women who provided a blood specimen were generally similar to women who did not; however, the proportion of women who were current smokers was lower among women who gave a blood specimen (14.4%) than among women who did not (25.0%). Subsequent follow-up of this sub-cohort of women has been greater than 96%. The Human Research Committee at the Brigham and Women’s Hospital and the Harvard School of Public Health approved this study.
Selection of Colorectal Cancer Cases and Control Participants
Eligible women for this study provided a blood specimen and were free from inflammatory bowel disease or cancer (except non-melanoma skin). When a participant reported a diagnosis of colorectal cancer, we asked for permission to obtain hospital records and pathology reports. We identified deaths through the National Death Index and next-of-kin. For all deaths, we sought information to determine the cause, including death certificates, and, when appropriate, request permission from next-of-kin to review medical records. Mortality follow-up was more than 98 percent complete.29, 30 Study physicians, blinded to exposure data, reviewed all medical records to confirm cases of colorectal cancer. After blood collection through June 1, 2004, we confirmed 286 incident cases of colorectal cancer. Using risk-set sampling, we randomly selected controls in a 2:1 ratio who were matched on year of birth and month/year of blood sampling (n=572) from participants who were free of colorectal cancer at the same time the colorectal cancer was diagnosed in the cases.12 We excluded 6 cases and 15 controls due to insufficient plasma volume. Thus, we included 280 cases and 555 control participants in the final analysis.
Laboratory Procedures
We sent a phlebotomy kit to all women willing to provide a blood specimen in 1989–1990. After receipt by overnight courier, the chilled heparinized blood was immediately centrifuged, aliquoted into plasma, and stored in continuously monitored liquid nitrogen freezers. Over 97% of the blood samples arrived in our laboratory within 26 hours of phlebotomy. Quality-control samples were routinely frozen along with study samples to monitor for changes associated with storage and assay variability. To examine stability of plasma biomarkers according to our storage techniques, we previously compared the concentrations in samples taken from 17 female volunteers that were processed and plasma frozen immediately after venipuncture (the standard processing methods) to samples that were stored as heparinized whole blood for 24 to 36 hours before processing (mimicking our collection conditions). The mean IL-6, CRP, and sTNFR-2 values were almost identical and the intraclass correlations (ICC) between results of the two collection methods were 0.99 for CRP, 0.93 for IL-6, 0.91 for sTNFR-2, demonstrating that our collection methods did not adversely affect sample integrity.31
In a core laboratory facility, we used a highly sensitive immunoturbidimetric assay (Denka Seiken Co.) to measure CRP and enzyme-linked immunosorbent assays (R & D Systems) to measure IL-6 and sTNFR-2. Personnel blinded to quality control and case–control status conducted all assays. The intra-assay coefficients of variations from blinded quality control samples for each analyte were: CRP, 2.2%; IL-6, 10.6%; sTNFR-2, 6.7%.
Assessment of Aspirin/NSAID Use
Assessment of aspirin and NSAID use in the NHS has been described in detail previously. Briefly, since 1980, we assessed intake of aspirin biennially except in 1986 with specific questions on the number of standard aspirin tablets (325 mg) taken. Women were specifically asked to record their typical pattern of aspirin use in most weeks over the prior 2-year period. Early in the study, most women used standard-dose aspirin tablets of 325 mg; however, to reflect overall secular trends in consumption of low-dose, or baby aspirin, questionnaires after 1992 asked participants to convert intake of four baby aspirin to one adult tablet. Beginning in 1990, we also asked about the use of “other anti-inflammatory drugs (e.g. Ibuprofen, Naprosyn, Advil)” which we classified as NSAIDs.1, 3, 32 There were no important differences in baseline characteristics comparing regular aspirin users with NSAID users.
Statistical Analysis
We first calculated means (± standard deviation [SD]), medians (± interquartile ranges [IQR]), and proportions of baseline characteristics for the case subjects and control subjects at the time of blood draw. We used Wilcoxon signed-rank and χ2 tests for comparisons of the means and proportions of the baseline characteristics. We calculated Spearman coefficients to estimate the correlation between mean levels of plasma biomarkers and lifestyle factors. We divided the inflammatory markers into quartiles, from the lowest to highest levels, on the basis of the distribution among the controls. Because postmenopausal hormone use elevates CRP levels,33 consistent with prior studies we categorized women into quartiles of CRP based on the CRP distribution among never users or ever users of postmenopausal hormones at baseline.26 We estimated relative risks (RR) and corresponding 95% confidence intervals (CI) for associations between quartile-specific markers using logistic regression models. Tests for trend were conducted using the median values for each quartile of plasma biomarker as a continuous variable in the regression models. We obtained similar results using conditional logistic regression models or unconditional logistic regression models adjusting for matching factors. Because both analyses provided similar results, we present the results of unconditional logistic regression, which parallel the results in the subgroup analyses. We also examined the possibility of a non-linear association between each plasma analyte and colorectal cancer risk using restricted cubic splines.34 We selected 3 knots splines, consistent with prior analyes in this cohort and the frequency distribution of each analyte.35 We tested for overall significance of the spline curve using a likelihood ratio test that compared the −2 log likelihood of a model with the analytes expressed as spline terms with that of a model without these terms (only the covariates).
In our multivariate model, we further adjusted for known or suspected risk factors for colorectal cancer listed in Table 1. We have previously shown each of these covariates to be related to colorectal cancer within the overall cohort from which this study was nested.1 To reflect participant characteristics at the time of the measurement of the plasma biomarker, we used baseline data for all lifestyle covariate data at the time of blood collection. For our analyses evaluating the influence of aspirin/NSAID use after measurement of plasma biomarkers, we derived aspirin and NSAID use data from the biennial questionnaire after blood collection. Consistent with our prior analyses, regular aspirin use was defined as at least 2 standard (325-mg) aspirin tablets/week and NSAID use was defined as at least 2 tablets/week.1–3 Consistent with prior analyses in this cohort,36 we assessed additive interaction based on the work of Rothman which showed that independent risk factors adhere to an additive model and biological interaction results in the departure from additivity.37–39 We also assessed multiplicative interaction by using a cross-product term. We used SAS, version 9.1.3 (Cary NC), for all analyses. All P-values are two-sided.
Table 1.
Baseline characteristics of study participants
| Baseline characteristic | Case participants (N = 280) | Control participants (N = 555) |
|---|---|---|
| Mean age at blood draw, years (SD) | 59.3 ± 6.6 | 59.3 ± 6.6 |
| Nonwhite (%) | 9 (3) | 6 (1) |
| Current or past smoker No. (%) | 161 (56) | 317 (57) |
| Post-menopausal, No. (%) | 244 (87) | 490 (88) |
| Current use of hormones, No, (%) 1 | 86 (35) | 213 (44) |
| Body mass index, mean (SD) | 26.0 (6.0) | 25.5 (5.8) |
| Physical activity, mean (SD) 2 | 19.1 (19.7) | 17.4 (18.6) |
| Regular aspirin use (≥2 325-mg tablets/week), No. (%) | 114 (41) | 253 (46) |
| Regular NSAID use (≥2 tablets/week), No. (%) | 43 (15) | 107 (19) |
| Multivitamin use, No. (%) | 102 (36) | 208 (37) |
| Colorectal cancer in a parent or sibling | 40 (14) | 72 (13) |
| History of previous endoscopy, No. (%) | 34 (12) | 89 (16) |
| History of colon polyp, No. (%) | 21 (8) | 25 (5) |
| Calcium, mg/day | 995 (546) | 1069 (573) |
| Folate, mg/day | 436 (277) | 470 (238) |
| Beef, pork, or lamb as main dish, servings/day | 0.30 (0.18) | 0.30 (0.17) |
| Alcohol, g/day | 5.8 (9.9) | 5.4 (9.6) |
| C-reactive protein (mg/L), median (IQR) 3 | 1.02 (0.59–2.29) | 1.24 (0.55–3.14) |
| Interleukin-6 (pg/ml), median (IQR) | 1.28 (0.86–2.05) | 1.22 (0.84–1.98) |
| sTNF-R2 (pg/mL), median (IQR) | 2636 (2202–3083) | 2528 (2116–2997) |
Percentage is among post-menopausal women.
METs – metabolic equivalent task score hours per week.
Among 118 case participants and 224 control participants who never used postmenopausal hormones
RESULTS
The baseline characteristics of the 280 colorectal cancer case and 555 matched control participants at the time of blood collection are presented in Table 1. The mean age of the study cohort was 59.3 years at the time of blood draw. The median values of baseline plasma CRP were 1.24 mg/L among 224 control women who never used post-menopausal hormones and 1.99 mg/L among 331 control women who had ever used post-menopausal hormones (p<0.0001). For IL-6, the corresponding median values were 1.22 pg/ml for women who never used post-menopausal hormones and 1.23 for women who had ever used (p=0.88). For sTNFR-2, the corresponding median values were 2598 pg/ml for women who never used post-menopausal hormones and 2502 pg/ml for women who had ever used (p=0.13). Overall, women who were subsequently diagnosed with colorectal cancer were significantly less likely to have used postmenopausal hormones (p=0.03) and had non-significantly higher levels of baseline plasma sTNFR-2 (p=0.07). Plasma levels of CRP (among women who never used postmenopausal hormones) and IL-6 did not appear to significantly differ between cases and controls (p=0.24 for CRP and p=0.44 for IL-6).
The Spearman correlation coefficients between plasma inflammatory markers, age, body-mass index (BMI), and metabolic equivalent task score hours (MET-hrs) per week of physical activity are shown in Table 2. Plasma inflammatory markers directly correlated with age and BMI and negatively correlated with physical activity. Levels of plasma CRP appeared to correlate more strongly with levels of IL-6 than sTNFR-2.
Table 2.
Spearman correlation coefficients between CRP, IL-6, sTNF-R2, age, BMI, and MET-hrs of physical activity at blood draw among control subjects 1
| Variable | CRP | IL-6 | sTNF-R2 | Age | BMI | METs |
|---|---|---|---|---|---|---|
| CRP | 1.00 | 0.46 b | 0.29 b | 0.12 d | 0.37 b | −0.17 b |
| IL-6 | 0.46 b | 1.00 | 0.40 b | 0.09 f | 0.24 b | −0.12 e |
| sTNF-R2 | 0.29 b | 0.40 b | 1.00 | 0.24 b | 0.24 b | −0.03 |
| Age (years) | 0.12 d | 0.09 f | 0.24 b | 1.00 | −0.04 | 0.07 |
| BMI (kg/m2) | 0.37 b | 0.24 b | 0.24 b | −0.04 | 1.00 | −0.14 |
| METs | −0.17 b | −0.12 e | −0.03 | 0.07 | −0.14 | 1.00 |
BMI - body mass index; METs – metabolic equivalent task score hours per week of physical activity.
p=<.0001
p=.002
p=.004
p=.007
p=.04
We examined the relationship between baseline plasma inflammatory markers and risk of colorectal cancer based on the quartile distribution of each marker in the controls (Table 3). Consistent with prior studies and the known effect of post-menopausal hormones in raising CRP levels, we assigned quartile categories of CRP according to separate cutpoints derived for women who never used hormones and women who ever used hormones. The highest quartile of plasma sTNFR-2 compared to the lowest quartile was significantly associated with colorectal cancer, even after adjustment for other traditional lifestyle and dietary risk factors (multivariate RR, 1.67; 95% CI, 1.05–2.68; ptrend=0.03). In contrast, there was no association observed comparing extreme quartiles for CRP (multivariate RR, 0.65; 95% CI, 0.40–1.05; ptrend=0.17) or IL-6 (multivariate RR, 1.18; 95% CI, 0.75–1.85; ptrend=0.55).
Table 3.
Relative risk of colorectal cancer according to plasma inflammatory markers 1
| Analyte | Quartiles | Ptrend2 | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| CRP | |||||
| Median (mg/ml) | 0.39 | 0.95 | 2.13 | 5.37 | |
| No. of cases/controls | 77/133 | 68/138 | 75/142 | 60/142 | |
| Age-adjusted RR (95% CI) 3 | 1.00 (referent) | 0.85 (0.57–1.27) | 0.91 (0.61–1.35) | 0.73 (0.48–1.10) | 0.13 |
| Multivariate-adjusted RR (95% CI) 4 | 1.00 (referent) | 0.79 (0.51–1.21) | 0.89 (0.57–1.39) | 0.65 (0.40–1.05) | 0.17 |
| IL-6 | |||||
| Median (pg/ml) | 0.65 | 1.02 | 1.45 | 3.04 | |
| No. of cases/controls | 63/138 | 70/138 | 71/138 | 75/139 | |
| Age-adjusted RR (95% CI) 3 | 1.00 (referent) | 1.11 (0.73–1.68) | 1.13 (0.74–1.70) | 1.17 (0.78–1.78) | 0.52 |
| Multivariate-adjusted RR (95% CI) 4 | 1.00 (referent) | 1.13 (0.73–1.74) | 1.11 (0.72–1.72) | 1.18 (0.75–1.85) | 0.55 |
| sTNF-R2 | |||||
| Median (pg/ml) | 1900 | 2345 | 2743 | 3463 | |
| No. of cases/controls subjects | 54/138 | 63/139 | 80/139 | 80/139 | |
| Age-adjusted RR (95% CI) 3 | 1.00 (referent) | 1.18 (0.76–1.82) | 1.50 (0.98–2.30) | 1.51 (0.98–2.33) | 0.04 |
| Multivariate-adjusted RR (95% CI) 4 | 1.00 (referent) | 1.22 (0.78–1.92) | 1.53 (0.98–2.40) | 1.67 (1.05–2.68) | 0.03 |
CRP – C-reactive protein; IL-6 – interleukin-6; sTNF-R2 – soluble tumor necrosis factor-receptor 2. Quartiles of plasma inflammatory markers are based on the distribution in the controls. For CRP, women were categorized according to cutpoints of never users or ever users of postmenopausal hormones. For CRP, the median values in the table represent never users of postmenopausal hormones. The corresponding values for ever users of postmenopausal hormones were 0.68 mg/L for quartile 1, 1.62 mg/L for quartile 2, 3.15 mg/L for quartile 3, and 6.71 for quartile 4. One case and 2 controls had insufficient plasma for IL-6 measurements. 3 cases had insufficient plasma for sTNF-R2 measurements.
Tests for linear trend were conducted using the median values for each quartile of analyte.
Age-adjusted models including adjustment for matching factors (age at blood draw, date of blood draw).
Multivariate models were adjusted for age at blood draw, date of blood draw, body mass index (quintiles), physical activity (quintiles of MET task score per week), current or past smoking (yes or no), menopause status, current postmenopausal hormone use (yes or no), prior lower gastrointestinal endoscopy (yes or no), colorectal cancer in parent or sibling. (yes or no), regular use of multivitamins (yes or no), regular use of aspirin or NSAIDs (≥2 tablets/week), energy-adjusted intake (including supplements) of calcium and folate (quintiles), servings of beef, pork, or lamb as a main dish (0–3 times/month, 1 time/week, 2–4 times/week, 5+ times/week), alcohol consumption (0, 0.1–4.9, 5.0–14.9, or +15 g per day).
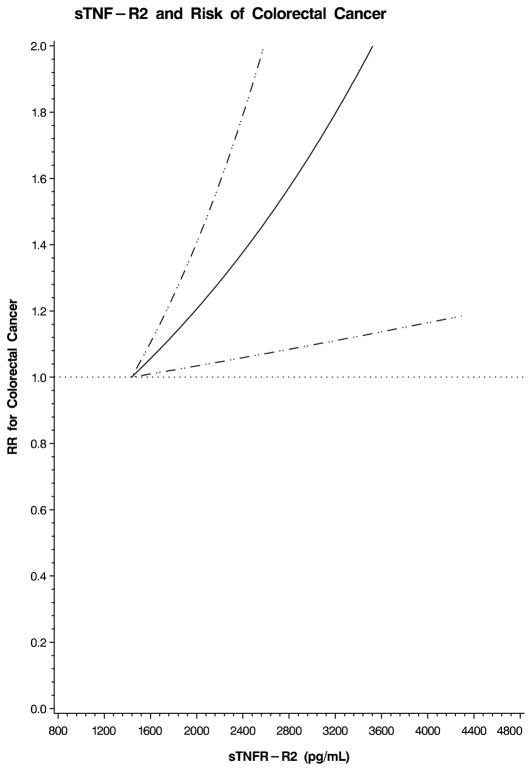
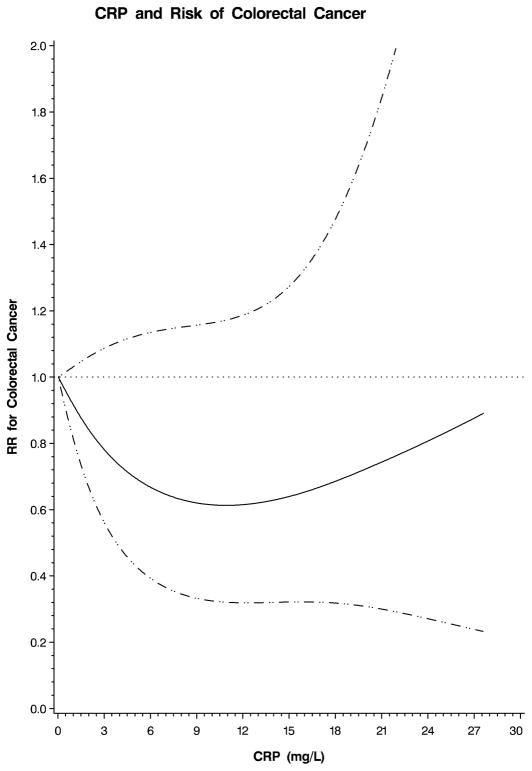
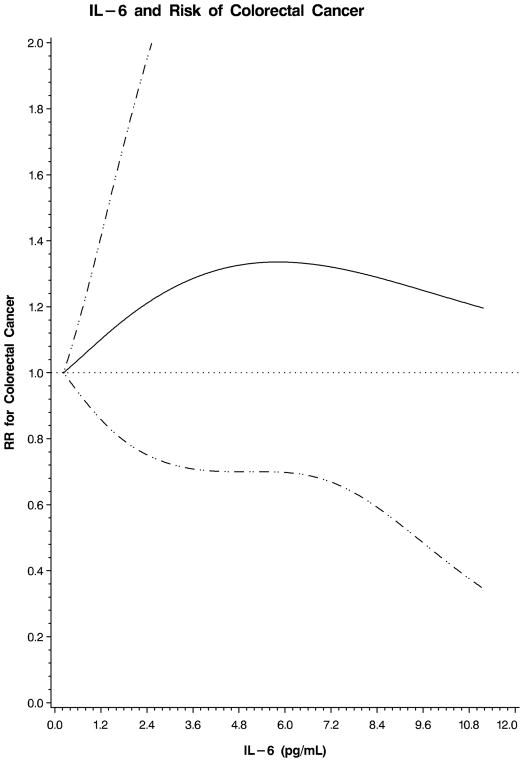
We also examined the possibility of a nonlinear relation between each plasma marker and colorectal cancer using restricted cubic splines. This flexible method allowed us to examine the relation to colorectal cancer risk without any categorization of each plasma marker or an assumption of linearity. A test for overall significance of the curve demonstrated a significant relationship between colorectal cancer and plasma sTNF-R2 (p=0.02; Figure 1). In contrast, there was no significant association between colorectal cancer and CRP (p=0.32; Figure 2) or IL-6 (p=0.49; Figure 3). Based on these analyses, we calculated multivariate RRs at specific values on the spline plots that have been previously shown to be clinically relevant. Because no prior studies have examined sTNF-R2 in relation to colorectal cancer, we selected a plasma level that has been previously associated with risk of coronary heart disease in this cohort.12 A plasma sTNF-R2 level of 3408 pg/mL was associated with a multivariate RR of colorectal cancer of 1.92 (95% CI, 1.12–3.30). In addition, for sTNF-R2, the relationship with colorectal cancer appeared linear; a 100 pg/mL increase in sTNF-R2 was associated with a multivariate RR of colorectal cancer of 1.04 (95% CI, 1.01–1.08). Prior studies have related IL-6 to risk of total cancer;18 a plasma IL-6 level of 3.0 pg/mL was associated with a multivariate RR of colorectal cancer of 1.25 (95% CI, 0.72–2.17) in our cohort. Finally, for CRP, we selected a value that has been specifically associated with risk of colorectal cancer;16 a plasma CRP level of 5.33 mg/L was associated with a multivariate RR of colorectal cancer of 0.58 (95% CI, 0.33–1.01) in our cohort.
Figure 1.
Restricted cubic spline plot for sTNF-R2 and risk of colorectal cancer. Relative risk (RR) of colorectal cancer is plotted according to plasma sTNF-R2 (pg/mL). Hatched lines represent 95% confidence intervals (CI). Spline was adjusted for the same factors as the multivariate model in Table 3. A test for overall significance of the curve was statistically significant (p=0.02).
Figure 2.
Restricted cubic spline plot for CRP and risk of colorectal cancer. Relative risk (RR) of colorectal cancer is plotted according to plasma CRP (mg/L). Hatched lines represent 95% confidence intervals (CI). Spline was adjusted for the same factors as the multivariate model in Table 3. A test for overall significance of the curve was non-significant (p=0.32).
Figure 3.
Restricted cubic spline plot for IL-6 and risk of colorectal cancer. Relative risk (RR) of colorectal cancer is plotted according to plasma IL-6 (pg/mL). Hatched lines represent 95% confidence intervals (CI). Spline was adjusted for the same factors as the multivariate model in Table 3. A test for overall significance of the curve was non-significant (p=0.49).
To address the potential bias that undiagnosed colorectal cancer itself could influence levels of plasma inflammatory markers at the time of blood collection, we excluded incident cases of colorectal cancer that were diagnosed within 2 years of follow-up. The multivariate RR of colorectal cancer for the highest quartile of plasma sTNFR-2 compared to the lowest quartile was not materially changed (multivariate RR, 1.61; 95% CI, 0.98–2.62; ptrend=0.04). There remained no significant association between plasma CRP (ptrend=0.08) or plasma IL-6 (ptrend=0.86) after excluding cases within 2 years of blood collection.
We conducted analyses (including spline-based models) of plasma inflammatory markers according to selected subgroups, including age, BMI, aspirin/NSAID use, multivitamin use, or folate intake at the time of blood collection. For each marker, there were no significant differences in the associations we observed within each subgroup, including those who were nonusers of aspirin/NSAIDs and those who were regular users of aspirin/NSAIDs at the time of blood draw. We also performed analyses adjusting for the presence of coronary heart disease or diabetes mellitus and our results were not materially altered. Because current use of post-menopausal hormones increases plasma CRP levels, we also conducted separate spline-based analyses of CRP using a model that did not include a term for current use of post-menopausal hormones. The spline plot was essentially unchanged; a test for overall significance of the curve relating CRP to colorectal cancer remained non-significant (p=0.15; supplementary Figure 1). In addition, we observed consistent results for spline analyses conducted among women who currently used post-menopausal hormones (p=0.75; supplementary Figure 2) and women who did not currently use post-menopausal hormones (p=0.44; supplementary Figure 3).
We have previously shown that aspirin and NSAID use in this cohort is associated with a lower risk of colorectal cancer.1, 3 Thus, in an exploratory analysis, we considered the possibility that initiating aspirin or NSAID use may be differentially associated with risk of colorectal cancer according to baseline levels of sTNFR-2 among the 286 women who were not using aspirin or NSAIDs at the time of blood draw (Table 4). Among women with high baseline levels of plasma sTNFR-2 (≥ median), women who began using aspirin (≥2 standard 325-mg tablets/week) or NSAIDs (≥2 tablets/week) after blood collection had a multivariate RR of colorectal cancer of 0.39 (95% CI, 0.18–0.86). In contrast, among women with low baseline plasma sTNFR-2 (< median), women who began using aspirin or NSAIDs had a multivariate RR of colorectal cancer of 0.86 (95% CI, 0.41–1.79). A formal test of additive interaction between sTNF-R2, use of aspirin/NSAIDs, and risk of colorectal cancer was statistically significant (p=0.03). A formal test of multiplicative interaction between sTNF-R2, use of aspirin/NSAIDs and risk of colorectal cancer approached statistical significance (P=0.10).
Table 4.
Relative risk of colorectal cancer according to initiation of regular aspirin or NSAID use after blood draw, stratified by sTNF-R2 level 1
| Characteristic | Non-user after blood draw | Regular user after blood draw |
|---|---|---|
| All sTNF-R2 levels | ||
| No. of cases/controls | 109/177 | 34/84 |
| Age-adjusted RR (95% CI) 2 | 1.00 | 0.65 (0.41–1.04) |
| Multivariable-adjusted RR (95% CI) 3 | 1.00 | 0.67 (0.41–1.09) |
| sTNF-R2 ≥ median (2636 pg/ml) | ||
| No. of cases/controls | 59/83 | 15/43 |
| Age-adjusted RR (95% CI) 2 | 1.00 | 0.49 (0.25–0.97) |
| Multivariable-adjusted RR (95% CI) 3 | 1.00 | 0.39 (0.18–0.86) |
| sTNF-R2 < median (2636 pg/ml) | ||
| No. of cases/controls | 48/94 | 19/41 |
| Age-adjusted RR (95% CI) 2 | 1.00 | 0.91 (0.48–0.74) |
| Multivariable-adjusted RR (95% CI) 3 | 1.00 | 0.86 (0.41–1.79) |
This analysis was restricted to the 404 participants who denied regular use of aspirin (≥2 standard 325-mg tablets/week) or NSAIDS (≥2 tablets/week) on the most recent questionnaire prior to blood draw. Non-users after blood draw denied regular use of aspirin on any questionnaire after blood draw but prior to diagnosis. Regular users after blood draw reported regular use after the most recent questionnaire after blood draw but prior to diagnosis.
Age-adjusted models including adjustment for matching factors (age at blood draw, date of blood draw).
Multivariate relative risks and 95% CI were adjusted for the same factors as the multivariate model in Table 3 with the exception of regular use of aspirin/NSAIDs.
DISCUSSION
Our results demonstrate an association between baseline plasma sTNFR-2, but not CRP or IL-6, and subsequent risk of colorectal cancer. Moreover, the inverse association between regular aspirin and NSAID use on risk of colorectal cancer appeared to be restricted to women with high baseline levels of sTNFR-2. To our knowledge, this study is the first to directly examine the role of sTNFR-2 in relation to risk of colorectal cancer.
Our data support a role for chronic inflammation in the pathogenesis of sporadic colorectal cancer, but suggest that circulating biomarkers of inflammation have varying ability to predict risk of incident cancer. Specifically, we did not find an association between colorectal cancer and CRP or IL-6, consistent with several other studies.18, 21–26 In contrast, prior studies have observed an increased risk of colorectal cancer associated with CRP.15–19 Each of these positive studies examined populations that included a significant proportion of men, with the strongest associations observed in a cohort comprised entirely of male smokers.15 In contrast, our null results for CRP and colorectal cancer were most consistent with those observed in the only other cohort comprised entirely of women.26 Thus, it is possible that some of the divergent results across studies of CRP may be related to sex. In support of this hypothesis, several studies have demonstrated significant differences in the inflammatory marker profile among women compared with men.12, 40 Animal data also demonstrate sex-based differences in cytokines associated with inflammation-related cancers.41 Nonetheless, the precise reasons for the different results for CRP and colorectal cancer across studies remain largely speculative.
Although our results for CRP and IL-6 were null, we did find an association between sTNFR-2 and risk of colorectal cancer. In support of our findings, previous studies in this cohort have shown that sTNFR-2 is associated with risk of coronary heart disease and diabetes mellitus,10, 12, 42 conditions related to systemic inflammation and also associated with risk of colorectal cancer.13, 14 sTNFR-2 is considered a reliable surrogate marker for tumor necrosis factor-α (TNF-α) since it is more stable in stored frozen samples and less influenced by diurnal variation.11, 43–47 Compared with CRP and IL-6, sTNFR-2 may be more strongly associated with colorectal cancer since TNF-α not only independently promotes cellular proliferation and inhibits apoptosis, but is also a key upstream regulator of CRP and IL-6.48 Thus, sTNFR-2 may be a superior marker of the specific pro-inflammatory milieu that predisposes to long-term development of cancer. Moreover, sTNFR-2 may also be a biomarker of deranged insulin resistance pathways through TNF-α-mediated inhibition of insulin signaling. Insulin resistance may represent an independent mechanistic pathway which promotes colorectal carcinogenesis.49
Our results for sTNFR-2 are also consistent with findings from the North Carolina Diet and Health Study (DHS). In this cross-sectional study, TNF-α levels were positively associated with prevalent colorectal adenomas.50 However, our findings contrast with a secondary analysis of a clinical trial of aspirin and/or folic acid in the prevention of recurrent colorectal adenomas. 51 In that study, changes over three years in plasma levels of sTNFR-2 were not associated with adenoma recurrence among high-risk patients with prior adenoma. The discordant findings in the aspirin trial compared with our study or the DHS may reflect the inability of inflammatory markers to determine risk of recurrent adenoma rather than initial adenoma or cancer.
We also observed that initiation of aspirin or NSAIDs was more strongly associated with lower risk of colorectal cancer among women with high levels of baseline sTNFR-2 than among women with low levels of sTNFR-2. This supports the hypothesis that aspirin and NSAIDs, at least in part, reduce risk of colorectal neoplasia through anti-inflammatory pathways. This is consistent with prior findings in this cohort in which we observed that aspirin use specifically reduced risk of colorectal cancers that over-expressed the pro-inflammatory COX-2 (PTGS2) isoenzyme.3 Other data examining the role of inflammatory markers in determining responsiveness to anti-inflammatory drugs are limited. Most prior studies relating anti-inflammatory drug use to inflammatory markers only examined drug exposure at the time of blood collection.15, 16, 18, 19, 21, 50 However, it is difficult to interpret these studies since the concurrent use of aspirin and NSAIDs directly influences the baseline level of inflammatory markers,50, 51 precluding an evaluation of the effect of these drugs according to pre-treatment levels. In the clinical trial of aspirin/folic acid neither baseline levels nor changes in post-treatment levels of sTNFR-2 modified the association of aspirin treatment with risk of recurrent adenoma.51 However, these results may reflect the potential limitation of using sTNFR-2 levels among high-risk patients with established neoplasia. Moreover, the doses of aspirin used by the women in our study (mean=7 standard aspirin tablets/week) were considerably higher than the aspirin dose (81 mg/day) that was effective in preventing recurrence in this randomized trial.1, 52 Finally, sTNFR-2 may influence the effect of aspirin/NSAID use on initial risk of invasive cancer but not recurrent adenoma.
The strengths of our study include its prospective design and high follow-up rate. Our measures of plasma inflammatory markers before diagnosis of cancer minimize potential bias related to elevation of these markers by the cancer itself. Moreover, because our risk estimates were essentially unchanged after excluding cases within 2 years of blood draw, our results are unlikely to be related to occult malignancy. We also had prospectively collected data on aspirin/NSAID use as well as potential lifestyle factors that could confound the relationship between colorectal cancer and inflammation. Notably, our associations between aspirin/NSAID use and risk of colorectal cancer have been validated by the results of randomized control trials.53 Finally, because we obtained data on aspirin/NSAID use both before and after blood collection, we were able to disentangle the effect of concurrent anti-inflammatory drug use on inflammatory marker levels with the specific influence of these drugs on colorectal cancer risk among treatment-naïve individuals.
We acknowledge several limitations to our study. First, we obtained only one baseline measure of inflammatory markers. However, others have shown that these markers are generally consistent over several years among the same individual.25, 54 Moreover, intraindividual variation in levels over time would tend to attenuate our observed associations.54, 55 Second, plasma inflammatory markers are likely to be a relatively non-specific assessment of the tissue-specific inflammatory pathways most relevant for colorectal carcinogenesis. Third, our cohort was comprised entirely of women. Thus, additional studies are needed to generalize our findings to men. Fourth, because our cohort was a case-control study of incident colorectal cancer nested within a prospective cohort, our study design did not permit an examination of adenoma, colorectal cancer-specific mortality, overall mortality, or the adverse consequences of aspirin use. However, we have previously shown significant relationships between aspirin use and risk of adenoma, colorectal-cancer specific mortality, overall mortality, and cardiovascular and gastrointestinal side effects in detailed, separate analyses of the larger cohort.32, 56–59 Finally, we cannot exclude the possibility that our associations with sTNFR-2 may reflect residual confounding resulting from imperfectly measured lifestyle risk factors. However, even if sTNFR-2 is not causally related to colorectal carcinogenesis, this would not diminish its potential as a surrogate biomarker for relevant pro-inflammatory, pro-carcinogenic lifestyle factors or exclude inflammation as a mechanistic basis for colorectal cancer.
In conclusion, plasma sTNFR-2, but not CRP or IL-6 is associated with the development of colorectal cancer. Anti-inflammatory drugs appear to reduce risk of colorectal cancer among women with high baseline levels of sTNFR-2 but not among women with low levels of sTNFR-2. Although these results should be viewed as exploratory and require confirmation, they support a role for chronic inflammation in colorectal cancer pathogenesis and the potential of using inflammatory markers to define subsets of the population that may obtain differential benefit from anti-inflammatory drugs.
Supplementary Material
Acknowledgments
The authors would like to acknowledge the continued dedication of the participants in the Nurses’ Health Study; Donna Spiegelman, ScD for statistical expertise; Ellen Hertzmark for assistance in programming; Gary Bradwin and Nader Rifai, PhD for measurements of the inflammatory analytes.
GRANT SUPPORT: R01 137178, U01 CA049449, P01CA87969, P01CA55075, P50 CA 127003, from the National Institutes of Health. Dr. Chan is a Damon Runyon Cancer Research Foundation Clinical Investigator.
ROLE OF THE SPONSOR: The sponsors had no role in the collection, analysis, or interpretation of the data.
Footnotes
CONFLICTS OF INTEREST: Dr. Chan has served as a consultant to Bayer HealthCare.
ROLES OF THE AUTHOR: Study concept and design (ATC, ELG, CSF); acquisition of data (ATC, ELG, CSF); analysis and interpretation of data (ATC, ELG, CSF); drafting of the manuscript (ATC, SO, ELG, CSF); critical revision of the manuscript for important intellectual content (ATC, SO, ELG, CSF); statistical analysis (ATC); obtained funding (ATC, ELG, CSF); technical, or material support (ATC, ELG, CSF); study supervision (ATC).
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Chan AT, Giovannucci EL, Meyerhardt JA, Schernhammer ES, Curhan GC, Fuchs CS. Long-term use of aspirin and nonsteroidal anti-inflammatory drugs and risk of colorectal cancer. Jama. 2005;294:914–23. doi: 10.1001/jama.294.8.914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chan AT, Giovannucci EL, Meyerhardt JA, Schernhammer ES, Wu K, Fuchs CS. Aspirin dose and duration of use and risk of colorectal cancer in men. Gastroenterology. 2008;134:21–8. doi: 10.1053/j.gastro.2007.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chan AT, Ogino S, Fuchs CS. Aspirin and the risk of colorectal cancer in relation to the expression of COX-2. N Engl J Med. 2007;356:2131–42. doi: 10.1056/NEJMoa067208. [DOI] [PubMed] [Google Scholar]
- 4.Cole BF, Logan RF, Halabi S, Benamouzig R, Sandler RS, Grainge MJ, Chaussade S, Baron JA. Aspirin for the chemoprevention of colorectal adenomas: meta-analysis of the randomized trials. J Natl Cancer Inst. 2009;101:256–66. doi: 10.1093/jnci/djn485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grau MV, Sandler RS, McKeown-Eyssen G, Bresalier RS, Haile RW, Barry EL, Ahnen DJ, Gui J, Summers RW, Baron JA. Nonsteroidal anti-inflammatory drug use after 3 years of aspirin use and colorectal adenoma risk: observational follow-up of a randomized study. J Natl Cancer Inst. 2009;101:267–76. doi: 10.1093/jnci/djn484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schottenfeld D, Beebe-Dimmer J. Chronic inflammation: a common and important factor in the pathogenesis of neoplasia. CA Cancer J Clin. 2006;56:69–83. doi: 10.3322/canjclin.56.2.69. [DOI] [PubMed] [Google Scholar]
- 7.Terzic J, Grivennikov S, Karin E, Karin M. Inflammation and colon cancer. Gastroenterology. 2010;138:2101–2114. e5. doi: 10.1053/j.gastro.2010.01.058. [DOI] [PubMed] [Google Scholar]
- 8.Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science. 1993;259:87–91. doi: 10.1126/science.7678183. [DOI] [PubMed] [Google Scholar]
- 9.Ventre J, Doebber T, Wu M, MacNaul K, Stevens K, Pasparakis M, Kollias G, Moller DE. Targeted disruption of the tumor necrosis factor-alpha gene: metabolic consequences in obese and nonobese mice. Diabetes. 1997;46:1526–31. doi: 10.2337/diab.46.9.1526. [DOI] [PubMed] [Google Scholar]
- 10.Hu FB, Meigs JB, Li TY, Rifai N, Manson JE. Inflammatory markers and risk of developing type 2 diabetes in women. Diabetes. 2004;53:693–700. doi: 10.2337/diabetes.53.3.693. [DOI] [PubMed] [Google Scholar]
- 11.Liu S, Tinker L, Song Y, Rifai N, Bonds DE, Cook NR, Heiss G, Howard BV, Hotamisligil GS, Hu FB, Kuller LH, Manson JE. A prospective study of inflammatory cytokines and diabetes mellitus in a multiethnic cohort of postmenopausal women. Arch Intern Med. 2007;167:1676–85. doi: 10.1001/archinte.167.15.1676. [DOI] [PubMed] [Google Scholar]
- 12.Pai JK, Pischon T, Ma J, Manson JE, Hankinson SE, Joshipura K, Curhan GC, Rifai N, Cannuscio CC, Stampfer MJ, Rimm EB. Inflammatory markers and the risk of coronary heart disease in men and women. N Engl J Med. 2004;351:2599–610. doi: 10.1056/NEJMoa040967. [DOI] [PubMed] [Google Scholar]
- 13.Hu FB, Manson JE, Liu S, Hunter D, Colditz GA, Michels KB, Speizer FE, Giovannucci E. Prospective study of adult onset diabetes mellitus (type 2) and risk of colorectal cancer in women. J Natl Cancer Inst. 1999;91:542–7. doi: 10.1093/jnci/91.6.542. [DOI] [PubMed] [Google Scholar]
- 14.Chan AO, Jim MH, Lam KF, Morris JS, Siu DC, Tong T, Ng FH, Wong SY, Hui WM, Chan CK, Lai KC, Cheung TK, Chan P, Wong G, Yuen MF, Lau YK, Lee S, Szeto ML, Wong BC, Lam SK. Prevalence of colorectal neoplasm among patients with newly diagnosed coronary artery disease. Jama. 2007;298:1412–9. doi: 10.1001/jama.298.12.1412. [DOI] [PubMed] [Google Scholar]
- 15.Gunter MJ, Stolzenberg-Solomon R, Cross AJ, Leitzmann MF, Weinstein S, Wood RJ, Virtamo J, Taylor PR, Albanes D, Sinha R. A prospective study of serum C-reactive protein and colorectal cancer risk in men. Cancer Res. 2006;66:2483–7. doi: 10.1158/0008-5472.CAN-05-3631. [DOI] [PubMed] [Google Scholar]
- 16.Erlinger TP, Platz EA, Rifai N, Helzlsouer KJ. C-reactive protein and the risk of incident colorectal cancer. Jama. 2004;291:585–90. doi: 10.1001/jama.291.5.585. [DOI] [PubMed] [Google Scholar]
- 17.Otani T, Iwasaki M, Sasazuki S, Inoue M, Tsugane S. Plasma C-reactive protein and risk of colorectal cancer in a nested case-control study: Japan Public Health Center-based prospective study. Cancer Epidemiol Biomarkers Prev. 2006;15:690–5. doi: 10.1158/1055-9965.EPI-05-0708. [DOI] [PubMed] [Google Scholar]
- 18.Il’yasova D, Colbert LH, Harris TB, Newman AB, Bauer DC, Satterfield S, Kritchevsky SB. Circulating Levels of Inflammatory Markers and Cancer Risk in the Health Aging and Body Composition Cohort. Cancer Epidemiol Biomarkers Prev %R. 2005;14:2413–2418. doi: 10.1158/1055-9965.EPI-05-0316. [DOI] [PubMed] [Google Scholar]
- 19.Trichopoulos D, Psaltopoulou T, Orfanos P, Trichopoulou A, Boffetta P. Plasma C-reactive protein and risk of cancer: a prospective study from Greece. Cancer Epidemiol Biomarkers Prev. 2006;15:381–4. doi: 10.1158/1055-9965.EPI-05-0626. [DOI] [PubMed] [Google Scholar]
- 20.Aleksandrova K, Jenab M, Boeing H, Jansen E, Bueno-de-Mesquita HB, Rinaldi S, Riboli E, Overvad K, Dahm CC, Olsen A, Tjonneland A, Boutron-Ruault MC, Clavel-Chapelon F, Morois S, Palli D, Krogh V, Tumino R, Vineis P, Panico S, Kaaks R, Rohrmann S, Trichopoulou A, Lagiou P, Trichopoulos D, van Duijnhoven FJ, Leufkens AM, Peeters PH, Rodriguez L, Bonet C, Sanchez MJ, Dorronsoro M, Navarro C, Barricarte A, Palmqvist R, Hallmans G, Khaw KT, Wareham N, Allen NE, Spencer E, Romaguera D, Norat T, Pischon T. Circulating C-reactive protein concentrations and risks of colon and rectal cancer: a nested case-control study within the European Prospective Investigation into Cancer and Nutrition. Am J Epidemiol. 2010;172:407–18. doi: 10.1093/aje/kwq135. [DOI] [PubMed] [Google Scholar]
- 21.Siemes C, Visser LE, Coebergh JW, Splinter TA, Witteman JC, Uitterlinden AG, Hofman A, Pols HA, Stricker BH. C-reactive protein levels, variation in the C-reactive protein gene, and cancer risk: the Rotterdam Study. J Clin Oncol. 2006;24:5216–22. doi: 10.1200/JCO.2006.07.1381. [DOI] [PubMed] [Google Scholar]
- 22.Heikkila K, Harris R, Lowe G, Rumley A, Yarnell J, Gallacher J, Ben-Shlomo Y, Ebrahim S, Lawlor DA. Associations of circulating C-reactive protein and interleukin-6 with cancer risk: findings from two prospective cohorts and a meta-analysis. Cancer Causes Control. 2009;20:15–26. doi: 10.1007/s10552-008-9212-z. [DOI] [PubMed] [Google Scholar]
- 23.Allin KH, Bojesen SE, Nordestgaard BG. Baseline C-reactive protein is associated with incident cancer and survival in patients with cancer. J Clin Oncol. 2009;27:2217–24. doi: 10.1200/JCO.2008.19.8440. [DOI] [PubMed] [Google Scholar]
- 24.Ito Y, Suzuki K, Tamakoshi K, Wakai K, Kojima M, Ozasa K, Watanabe Y, Kawado M, Hashimoto S, Suzuki S, Tokudome S, Toyoshima H, Hayakawa N, Kato K, Watanabe M, Ohta Y, Maruta M, Tamakoshi A. Colorectal cancer and serum C-reactive protein levels: a case-control study nested in the JACC Study. J Epidemiol. 2005;15 (Suppl 2):S185–9. doi: 10.2188/jea.15.S185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Allin KH, Nordestgaard BG, Zacho J, Tybjaerg-Hansen A, Bojesen SE. C-reactive protein and the risk of cancer: a mendelian randomization study. J Natl Cancer Inst. 2010;102:202–6. doi: 10.1093/jnci/djp459. [DOI] [PubMed] [Google Scholar]
- 26.Zhang SM, Buring JE, Lee IM, Cook NR, Ridker PM. C-reactive protein levels are not associated with increased risk for colorectal cancer in women. Ann Intern Med. 2005;142:425–32. doi: 10.7326/0003-4819-142-6-200503150-00008. [DOI] [PubMed] [Google Scholar]
- 27.Hankinson SE, Willett WC, Manson JE, Hunter DJ, Colditz GA, Stampfer MJ, Longcope C, Speizer FE. Alcohol, height, and adiposity in relation to estrogen and prolactin levels in postmenopausal women. J Natl Cancer Inst. 1995;87:1297–302. doi: 10.1093/jnci/87.17.1297. [DOI] [PubMed] [Google Scholar]
- 28.Hunter DJ, Hankinson SE, Hough H, Gertig DM, Garcia-Closas M, Spiegelman D, Manson JE, Colditz GA, Willett WC, Speizer FE, Kelsey K. A prospective study of NAT2 acetylation genotype, cigarette smoking, and risk of breast cancer. Carcinogenesis. 1997;18:2127–32. doi: 10.1093/carcin/18.11.2127. [DOI] [PubMed] [Google Scholar]
- 29.Stampfer MJ, Willett WC, Speizer FE, Dysert DC, Lipnick R, Rosner B, Hennekens CH. Test of the National Death Index. Am J Epidemiol. 1984;119:837–839. doi: 10.1093/oxfordjournals.aje.a113804. [DOI] [PubMed] [Google Scholar]
- 30.Rich-Edwards JW, Corsano KA, Stampfer MJ. Test of the National Death Index and Equifax Nationwide Death Search. Am J Epidemiol. 1994;140:1016–9. doi: 10.1093/oxfordjournals.aje.a117191. [DOI] [PubMed] [Google Scholar]
- 31.Pai JK, Curhan GC, Cannuscio CC, Rifai N, Ridker PM, Rimm EB. Stability of novel plasma markers associated with cardiovascular disease: processing within 36 hours of specimen collection. Clin Chem. 2002;48:1781–4. [PubMed] [Google Scholar]
- 32.Chan AT, Ogino S, Fuchs CS. Aspirin use and survival after diagnosis of colorectal cancer. JAMA. 2009;302:649–58. doi: 10.1001/jama.2009.1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ridker PM, Hennekens CH, Rifai N, Buring JE, Manson JE. Hormone replacement therapy and increased plasma concentration of C-reactive protein. Circulation. 1999;100:713–6. doi: 10.1161/01.cir.100.7.713. [DOI] [PubMed] [Google Scholar]
- 34.Durrleman S, Simon R. Flexible regression models with cubic splines. Stat Med. 1989;8:551–61. doi: 10.1002/sim.4780080504. [DOI] [PubMed] [Google Scholar]
- 35.Baba Y, Nosho K, Shima K, Freed E, Irahara N, Philips J, Meyerhardt JA, Hornick JL, Shivdasani RA, Fuchs CS, Ogino S. Relationship of CDX2 loss with molecular features and prognosis in colorectal cancer. Clin Cancer Res. 2009;15:4665–73. doi: 10.1158/1078-0432.CCR-09-0401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Karlson EW, Chang SC, Cui J, Chibnik LB, Fraser PA, De Vivo I, Costenbader KH. Gene-environment interaction between HLA-DRB1 shared epitope and heavy cigarette smoking in predicting incident rheumatoid arthritis. Ann Rheum Dis. 2009;69:54–60. doi: 10.1136/ard.2008.102962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rothman KJ, Greenland S, Lash TL. Modern epidemiology. Wolters Kluwer Health/Lippincott Williams & Wilkins; 2008. [Google Scholar]
- 38.Lundberg M, Fredlund P, Hallqvist J, Diderichsen F. A SAS program calculating three measures of interaction with confidence intervals. Epidemiology. 1996;7:655–6. [PubMed] [Google Scholar]
- 39.Andersson T, Alfredsson L, Kallberg H, Zdravkovic S, Ahlbom A. Calculating measures of biological interaction. Eur J Epidemiol. 2005;20:575–9. doi: 10.1007/s10654-005-7835-x. [DOI] [PubMed] [Google Scholar]
- 40.Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med. 2000;342:836–43. doi: 10.1056/NEJM200003233421202. [DOI] [PubMed] [Google Scholar]
- 41.Naugler WE, Sakurai T, Kim S, Maeda S, Kim K, Elsharkawy AM, Karin M. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science. 2007;317:121–4. doi: 10.1126/science.1140485. [DOI] [PubMed] [Google Scholar]
- 42.Shai I, Schulze MB, Manson JE, Rexrode KM, Stampfer MJ, Mantzoros C, Hu FB. A prospective study of soluble tumor necrosis factor-alpha receptor II (sTNF-RII) and risk of coronary heart disease among women with type 2 diabetes. Diabetes Care. 2005;28:1376–82. doi: 10.2337/diacare.28.6.1376. [DOI] [PubMed] [Google Scholar]
- 43.Diez-Ruiz A, Tilz GP, Zangerle R, Baier-Bitterlich G, Wachter H, Fuchs D. Soluble receptors for tumour necrosis factor in clinical laboratory diagnosis. Eur J Haematol. 1995;54:1–8. doi: 10.1111/j.1600-0609.1995.tb01618.x. [DOI] [PubMed] [Google Scholar]
- 44.DeRijk R, Michelson D, Karp B, Petrides J, Galliven E, Deuster P, Paciotti G, Gold PW, Sternberg EM. Exercise and circadian rhythm-induced variations in plasma cortisol differentially regulate interleukin-1 beta (IL-1 beta), IL-6, and tumor necrosis factor-alpha (TNF alpha) production in humans: high sensitivity of TNF alpha and resistance of IL-6. J Clin Endocrinol Metab. 1997;82:2182–91. doi: 10.1210/jcem.82.7.4041. [DOI] [PubMed] [Google Scholar]
- 45.Ohgushi M, Taniguchi A, Fukushima M, Nakai Y, Kuroe A, Ohya M, Nagasaka S, Taki Y, Yoshii S, Matsumoto K, Yamada Y, Inagaki N, Seino Y. Soluble tumor necrosis factor receptor 2 is independently associated with pulse wave velocity in nonobese Japanese patients with type 2 diabetes mellitus. Metabolism. 2007;56:571–7. doi: 10.1016/j.metabol.2006.12.007. [DOI] [PubMed] [Google Scholar]
- 46.Hotamisligil GS, Arner P, Atkinson RL, Spiegelman BM. Differential regulation of the p80 tumor necrosis factor receptor in human obesity and insulin resistance. Diabetes. 1997;46:451–5. doi: 10.2337/diab.46.3.451. [DOI] [PubMed] [Google Scholar]
- 47.Aderka D. The potential biological and clinical significance of the soluble tumor necrosis factor receptors. Cytokine Growth Factor Rev. 1996;7:231–40. doi: 10.1016/s1359-6101(96)00026-3. [DOI] [PubMed] [Google Scholar]
- 48.de Visser KE, Eichten A, Coussens LM. Paradoxical roles of the immune system during cancer development. Nat Rev Cancer. 2006;6:24–37. doi: 10.1038/nrc1782. [DOI] [PubMed] [Google Scholar]
- 49.Slattery ML, Samowitz W, Hoffman M, Ma KN, Levin TR, Neuhausen S. Aspirin, NSAIDs, and colorectal cancer: possible involvement in an insulin-related pathway. Cancer Epidemiol Biomarkers Prev. 2004;13:538–45. [PubMed] [Google Scholar]
- 50.Kim S, Keku TO, Martin C, Galanko J, Woosley JT, Schroeder JC, Satia JA, Halabi S, Sandler RS. Circulating levels of inflammatory cytokines and risk of colorectal adenomas. Cancer Res. 2008;68:323–8. doi: 10.1158/0008-5472.CAN-07-2924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ho GY, Xue X, Cushman M, McKeown-Eyssen G, Sandler RS, Ahnen DJ, Barry EL, Saibil F, Bresalier RS, Rohan TE, Baron JA. Antagonistic Effects of Aspirin and Folic Acid on Inflammation Markers and Subsequent Risk of Recurrent Colorectal Adenomas. J Natl Cancer Inst. 2009 doi: 10.1093/jnci/djp346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Baron JA, Cole BF, Sandler RS, Haile RW, Ahnen D, Bresalier R, McKeown-Eyssen G, Summers RW, Rothstein R, Burke CA, Snover DC, Church TR, Allen JI, Beach M, Beck GJ, Bond JH, Byers T, Greenberg ER, Mandel JS, Marcon N, Mott LA, Pearson L, Saibil F, van Stolk RU. A randomized trial of aspirin to prevent colorectal adenomas. N Engl J Med. 2003;348:891–9. doi: 10.1056/NEJMoa021735. [DOI] [PubMed] [Google Scholar]
- 53.Flossmann E, Rothwell PM. Effect of aspirin on long-term risk of colorectal cancer: consistent evidence from randomised and observational studies. Lancet. 2007;369:1603–13. doi: 10.1016/S0140-6736(07)60747-8. [DOI] [PubMed] [Google Scholar]
- 54.Platz EA, Sutcliffe S, De Marzo AM, Drake CG, Rifai N, Hsing AW, Hoque A, Neuhouser ML, Goodman PJ, Kristal AR. Intra-individual variation in serum C-reactive protein over 4 years: an implication for epidemiologic studies. Cancer Causes Control. 2010;21:847–51. doi: 10.1007/s10552-010-9511-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Clarke R, Shipley M, Lewington S, Youngman L, Collins R, Marmot M, Peto R. Underestimation of risk associations due to regression dilution in long-term follow-up of prospective studies. Am J Epidemiol. 1999;150:341–53. doi: 10.1093/oxfordjournals.aje.a010013. [DOI] [PubMed] [Google Scholar]
- 56.Chan AT, Giovannucci EL, Schernhammer ES, Colditz GC, Hunter DJ, Willett WC, Fuchs CS. A prospective study of aspirin use and the risk of colorectal adenoma. Ann Intern Med. 2004;140:157–166. doi: 10.7326/0003-4819-140-3-200402030-00006. [DOI] [PubMed] [Google Scholar]
- 57.Chan AT, Manson JE, Feskanich D, Stampfer MJ, Colditz GA, Fuchs CS. Long-term aspirin use and mortality in women. Arch Intern Med. 2007;167:562–72. doi: 10.1001/archinte.167.6.562. [DOI] [PubMed] [Google Scholar]
- 58.Chan AT, Manson JE, Albert CM, Chae CU, Rexrode KM, Curhan GC, Rimm EB, Willett WC, Fuchs CS. Nonsteroidal antiinflammatory drugs, acetaminophen, and the risk of cardiovascular events. Circulation. 2006;113:1578–1587. doi: 10.1161/CIRCULATIONAHA.105.595793. [DOI] [PubMed] [Google Scholar]
- 59.Huang ES, Strate LL, Ho WW, Lee SS, Chan AT. Long-term use of aspirin and the risk of gastrointestinal bleeding in a prospective, population-based cohort. Gastroenterology. 2010;138:S64–64. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.