Abstract
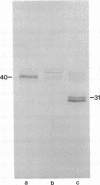
Hydrophobic surfactant-associated protein of Mr 6000-14,000 was isolated from ether/ethanol or chloroform/methanol extracts of mammalian pulmonary surfactant. Automated Edman degradation in a gas-phase sequencer showed the major N-terminus of the human low molecular weight protein to be Phe-Pro-Ile-Pro-Leu-Pro-Tyr-Cys-Trp-Leu-Cys-Arg-Ala-Leu-. Because of the N-terminal phenylalanine, the surfactant protein was designated SPL(Phe). Antiserum generated against hydrophobic surfactant protein(s) from bovine pulmonary surfactant recognized protein of Mr 6000-14,000 in immunoblot analysis and was used to screen a lambda gt11 expression library constructed from adult human lung poly(A)+ RNA. This resulted in identification of a 1.4-kilobase cDNA clone that was shown to encode the N-terminus of the surfactant polypeptide SPL(Phe) (Phe-Pro-Ile-Pro-Leu-Pro-) within an open reading frame for a larger protein. Expression of a fused beta-galactosidase-SPL(Phe) gene in Escherichia coli yielded an immunoreactive Mr 34,000 fusion peptide. Hybrid-arrested translation with this cDNA and immunoprecipitation of [35S]methionine-labeled in vitro translation products of human poly(A)+ RNA with a surfactant polyclonal antibody resulted in identification of a Mr 40,000 precursor protein. Blot hybridization analysis of electrophoretically fractionated RNA from human lung detected a 2.0-kilobase RNA that was more abundant in adult lung than in fetal lung. The larger RNA and translation product indicates that SPL(Phe) is derived by proteolysis of a large polypeptide precursor. The amino acid sequence of the predicted protein, beginning Phe-Pro-Ile-Pro-Leu-Pro-Try-, comprises a hydrophobic peptide that is a major protein component of surfactant lipid extracts used successfully to treat hyaline membrane disease in newborn infants. These proteins, and specifically SPL(Phe), may therefore be useful for synthesis of replacement surfactants for treatment of hyaline membrane disease in newborn infants or of other surfactant-deficient states.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Claypool W. D., Wang D. L., Chander A., Fisher A. B. An ethanol/ether soluble apoprotein from rat lung surfactant augments liposome uptake by isolated granular pneumocytes. J Clin Invest. 1984 Sep;74(3):677–684. doi: 10.1172/JCI111483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enhorning G., Shennan A., Possmayer F., Dunn M., Chen C. P., Milligan J. Prevention of neonatal respiratory distress syndrome by tracheal instillation of surfactant: a randomized clinical trial. Pediatrics. 1985 Aug;76(2):145–153. [PubMed] [Google Scholar]
- FOLCH J., LEES M., SLOANE STANLEY G. H. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957 May;226(1):497–509. [PubMed] [Google Scholar]
- Fujiwara T., Maeta H., Chida S., Morita T., Watabe Y., Abe T. Artificial surfactant therapy in hyaline-membrane disease. Lancet. 1980 Jan 12;1(8159):55–59. doi: 10.1016/s0140-6736(80)90489-4. [DOI] [PubMed] [Google Scholar]
- King R. J., Klass D. J., Gikas E. G., Clements J. A. Isolation of apoproteins from canine surface active material. Am J Physiol. 1973 Apr;224(4):788–795. doi: 10.1152/ajplegacy.1973.224.4.788. [DOI] [PubMed] [Google Scholar]
- Kwong M. S., Egan E. A., Notter R. H., Shapiro D. L. Double-blind clinical trial of calf lung surfactant extract for the prevention of hyaline membrane disease in extremely premature infants. Pediatrics. 1985 Oct;76(4):585–592. [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Messing J., Crea R., Seeburg P. H. A system for shotgun DNA sequencing. Nucleic Acids Res. 1981 Jan 24;9(2):309–321. doi: 10.1093/nar/9.2.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Ohtsuka E., Matsuki S., Ikehara M., Takahashi Y., Matsubara K. An alternative approach to deoxyoligonucleotides as hybridization probes by insertion of deoxyinosine at ambiguous codon positions. J Biol Chem. 1985 Mar 10;260(5):2605–2608. [PubMed] [Google Scholar]
- Phizackerley P. J., Town M. H., Newman G. E. Hydrophobic proteins of lamellated osmiophilic bodies isolated from pig lung. Biochem J. 1979 Dec 1;183(3):731–736. doi: 10.1042/bj1830731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rooney S. A. The surfactant system and lung phospholipid biochemistry. Am Rev Respir Dis. 1985 Mar;131(3):439–460. doi: 10.1164/arrd.1985.131.3.439. [DOI] [PubMed] [Google Scholar]
- Ross G. F., Meuth J., Ohning B., Kim Y., Whitsett J. A. Purification of canine surfactant-associated glycoproteins A. Identification of a collagenase-resistant domain. Biochim Biophys Acta. 1986 Mar 28;870(2):267–278. doi: 10.1016/0167-4838(86)90231-1. [DOI] [PubMed] [Google Scholar]
- Ross G. F., Notter R. H., Meuth J., Whitsett J. A. Phospholipid binding and biophysical activity of pulmonary surfactant-associated protein (SAP)-35 and its non-collagenous COOH-terminal domains. J Biol Chem. 1986 Oct 25;261(30):14283–14291. [PubMed] [Google Scholar]
- Segrest J. P., Jackson R. L., Morrisett J. D., Gotto A. M., Jr A molecular theory of lipid-protein interactions in the plasma lipoproteins. FEBS Lett. 1974 Jan 15;38(3):247–258. doi: 10.1016/0014-5793(74)80064-5. [DOI] [PubMed] [Google Scholar]
- Suzuki Y. Effect of protein, cholesterol, and phosphatidylglycerol on the surface activity of the lipid-protein complex reconstituted from pig pulmonary surfactant. J Lipid Res. 1982 Jan;23(1):62–69. [PubMed] [Google Scholar]
- Takahashi A., Fujiwara T. Proteolipid in bovine lung surfactant: its role in surfactant function. Biochem Biophys Res Commun. 1986 Mar 13;135(2):527–532. doi: 10.1016/0006-291x(86)90026-4. [DOI] [PubMed] [Google Scholar]
- Tanaka Y., Takei T., Kanazawa Y. Lung surfactants. II. Effects of fatty acids, triacylglycerols and protein on the activity of lung surfactant. Chem Pharm Bull (Tokyo) 1983 Nov;31(11):4100–4109. doi: 10.1248/cpb.31.4100. [DOI] [PubMed] [Google Scholar]
- White R. T., Damm D., Miller J., Spratt K., Schilling J., Hawgood S., Benson B., Cordell B. Isolation and characterization of the human pulmonary surfactant apoprotein gene. 1985 Sep 26-Oct 2Nature. 317(6035):361–363. doi: 10.1038/317361a0. [DOI] [PubMed] [Google Scholar]
- Whitsett J. A., Hull W. M., Ohning B., Ross G., Weaver T. E. Immunologic identification of a pulmonary surfactant-associated protein of molecular weight = 6000 daltons. Pediatr Res. 1986 Aug;20(8):744–749. doi: 10.1203/00006450-198608000-00009. [DOI] [PubMed] [Google Scholar]
- Whitsett J. A., Hull W., Ross G., Weaver T. Characteristics of human surfactant-associated glycoproteins A. Pediatr Res. 1985 May;19(5):501–508. doi: 10.1203/00006450-198505000-00018. [DOI] [PubMed] [Google Scholar]
- Whitsett J. A., Ohning B. L., Ross G., Meuth J., Weaver T., Holm B. A., Shapiro D. L., Notter R. H. Hydrophobic surfactant-associated protein in whole lung surfactant and its importance for biophysical activity in lung surfactant extracts used for replacement therapy. Pediatr Res. 1986 May;20(5):460–467. doi: 10.1203/00006450-198605000-00016. [DOI] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1194–1198. doi: 10.1073/pnas.80.5.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Yeast RNA polymerase II genes: isolation with antibody probes. Science. 1983 Nov 18;222(4625):778–782. doi: 10.1126/science.6356359. [DOI] [PubMed] [Google Scholar]
- Yu S. H., Possmayer F. Reconstitution of surfactant activity by using the 6 kDa apoprotein associated with pulmonary surfactant. Biochem J. 1986 May 15;236(1):85–89. doi: 10.1042/bj2360085. [DOI] [PMC free article] [PubMed] [Google Scholar]