Abstract
In the title compound, (C24H20P)2[Cr(CN)4(N)]·2H2O, the complex anion exhibits a square-based pyramidal geometry around the central CrV atom, which is coordinated by a nitride ligand in the apical position and by four cyanide ligands in the equatorial plane. The chromium atom is located 0.4493 (13) Å out of the plane formed by the ligating C atoms of the cyanide ligands. The water molecules of crystallization form intermolecular O—H⋯N hydrogen bonds to the N atoms of two cyanide ligands of neighbouring complex anions, forming an infinite hydrogen-bonded chain parallel to [011] of water molecules and [Cr(N)(CN)4]2− anions. The terminal nitride ligands are not engaged in intermolecular interactions.
Related literature
For related structures of nitridocyanidometalates, see: Baldas et al. (1990 ▶); Bendix et al. (1998 ▶, 2000 ▶); Britten et al. (1993 ▶); Che et al. (1989 ▶); Purcell et al. (1991 ▶); van der Westhuizen et al. (1994 ▶). For general background to CrV nitrido complexes, see: Birk & Bendix (2003 ▶).
Experimental
Crystal data
(C24H20P)2[Cr(CN)4(N)]·2H2O
M r = 884.86
Triclinic,

a = 11.996 (5) Å
b = 12.387 (5) Å
c = 16.721 (4) Å
α = 98.34 (3)°
β = 110.01 (2)°
γ = 90.52 (4)°
V = 2305.3 (15) Å3
Z = 2
Mo Kα radiation
μ = 0.36 mm−1
T = 122 K
0.12 × 0.09 × 0.06 mm
Data collection
Nonius KappaCCD area-detector diffractometer
Absorption correction: integration (Gaussian; Coppens, 1970 ▶) T min = 0.952, T max = 0.980
72047 measured reflections
8118 independent reflections
6480 reflections with I > 2σ(I)
R int = 0.061
Refinement
R[F 2 > 2σ(F 2)] = 0.039
wR(F 2) = 0.092
S = 1.10
8118 reflections
571 parameters
4 restraints
H atoms treated by a mixture of independent and constrained refinement
Δρmax = 0.41 e Å−3
Δρmin = −0.44 e Å−3
Data collection: COLLECT (Nonius, 1999 ▶); cell refinement: COLLECT; data reduction: EVALCCD (Duisenberg et al., 2003 ▶); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008 ▶); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008 ▶); molecular graphics: ORTEP-3 (Farrugia, 1997 ▶); software used to prepare material for publication: SHELXL97.
Supplementary Material
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536811002108/fj2383sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536811002108/fj2383Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Table 1. Selected geometric parameters (Å, °).
| Cr—N1 | 1.538 (2) |
| Cr—C2 | 2.066 (3) |
| Cr—C3 | 2.040 (3) |
| Cr—C4 | 2.068 (3) |
| Cr—C5 | 2.049 (3) |
| N1—Cr—C2 | 99.23 (11) |
| N1—Cr—C3 | 105.79 (11) |
| N1—Cr—C4 | 99.95 (11) |
| N1—Cr—C5 | 105.70 (11) |
Table 2. Hydrogen-bond geometry (Å, °).
| D—H⋯A | D—H | H⋯A | D⋯A | D—H⋯A |
|---|---|---|---|---|
| O1—H1A⋯N2 | 0.86 (2) | 2.23 (2) | 3.065 (3) | 167 (4) |
| O1—H1B⋯N2i | 0.85 (2) | 2.18 (2) | 3.035 (3) | 177 (4) |
| O2—H2A⋯N4 | 0.81 (2) | 2.16 (2) | 2.973 (3) | 178 (3) |
| O2—H2B⋯N4ii | 0.82 (2) | 2.22 (2) | 3.039 (3) | 173 (3) |
Symmetry codes: (i)  ; (ii)
; (ii)  .
.
Table 3. Comparative geometric parameters (Å) of cyanidonitridometalates of chromate(V) and manganate(V).
| [Cr(CN)4(N)]2−a | [Mn(CN)4(N)]2−b | [Cr(CN)5(N)]3−c | [Mn(CN)5(N)]3−c | |
|---|---|---|---|---|
| M N | 1.538 (2) | 1.507 (2) | 1.594 (9) | 1.499 (8) |
| M—Ccis | 2.040 (3)–2.068 (3) | 1.974 (2)–1.995 (2) | 2.039 (7)–2.08 (2) | 1.985 (6)–2.001 (7) |
| M—Ctrans | 2.299 (12) | 2.243 (7) | ||
| M—oopd | 0.449 | 0.436 | 0.255 | 0.222 |
supplementary crystallographic information
Comment
Crystal structures of mononuclear nitridocyanidometalates have previously been reported for technetium (Baldas et al., 1990), rhenium (Britten et al., 1993; Purcell et al., 1991), osmium (Che et al., 1989; van der Westhuizen et al., 1994). We have previously reported the syntheses and crystal stuctures of six coordinated pentacyanidonitridometalates of chromate(V) and manganate(V) (Bendix et al., 2000) as well as the synthesis of the title compound and crystal structure of the isostructural manganese compound (Bendix et al., 1998). In all the cases the nitride ligand imposes a strong trans-influence on the auxiliary ligands evidenced by a significant elongation of the M—Xtrans bond length and the displacement of the metal out of the plane spanned by cyanido-carbon atoms.
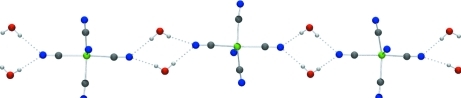
The molecular structure of the title compound is shown in Fig. 1. The CrV has a square based pyramidal coordination geometry frequently seen for CrV nitrido complexes (Birk & Bendix, 2003). The nitride occupy the apical position and the four cyanide ligands span the equatorial plane. The Cr atom is displaced 0.4493 (13) Å out of the plane spanned by the cyanido-carbon atoms. Selected geometric parameters are listed in Table 1. The water of crystallization form weak intermolecular hydrogen bonds (Table 2) to two nitrogen atoms of two cyanide ligands as depicted in Fig. 2. The complex anions and water molecules form an infinite chain. For comparison, Table 3 lists selected geometric parameters of the isostructural manganese compound and the six coordinated pentacyanidonitridometalates of chromate(V) and manganate(V).
Experimental
The title compound was prepared as previously reported (Bendix et al., 1998). A solution of [Cr(N)(CN)4]2- (0.10 g; 0.21 mmol) in water (6.5 ml) was allowed to slowly diffuse into a solution of [PPh4]Cl (0.20 g; 0.53 mmol) in water (15 ml). The pale yellow crystals that precipitated were collected by filtration, washed with water, and air-dried. Yield: 0.16 g (86%).
Refinement
H atoms on the phenyl groups were found in a difference Fourier map and were included in the refinement as constrained idealized protons riding the parent atom, with C—H = 0.95 Å. Ihe H atoms of the crystal waters were found in a difference Fourier map and were refined semi-free with a distance restraint and the Uiso equal to 1.5 times the Ueq of the parent oxygen.
Figures
Fig. 1.
A view of the molecular structure of (1) with the atom-labelling scheme. Displacement ellipsoids are drawn at the 50% probability level.
Fig. 2.
A ball-and-stick representation of the infinite chain of hydrogen bonded water molecules and [Cr(N)(CN)4]2- anions.
Crystal data
| (C24H20P)2[Cr(CN)4(N)]·2H2O | Z = 2 |
| Mr = 884.86 | F(000) = 922 |
| Triclinic, P1 | Dx = 1.275 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 11.996 (5) Å | Cell parameters from 83863 reflections |
| b = 12.387 (5) Å | θ = 1.3–27.6° |
| c = 16.721 (4) Å | µ = 0.36 mm−1 |
| α = 98.34 (3)° | T = 122 K |
| β = 110.01 (2)° | Prism, yellow |
| γ = 90.52 (4)° | 0.12 × 0.09 × 0.06 mm |
| V = 2305.3 (15) Å3 |
Data collection
| Nonius KappaCCD area-detector diffractometer | 8118 independent reflections |
| Radiation source: fine-focus sealed tube | 6480 reflections with I > 2σ(I) |
| graphite | Rint = 0.061 |
| ω and φ scans | θmax = 25.0°, θmin = 1.3° |
| Absorption correction: integration (Gaussian; Coppens, 1970) | h = −14→14 |
| Tmin = 0.952, Tmax = 0.980 | k = −14→14 |
| 72047 measured reflections | l = −19→19 |
Refinement
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.039 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.092 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.10 | w = 1/[σ2(Fo2) + (0.P)2 + 2.4326P] where P = (Fo2 + 2Fc2)/3 |
| 8118 reflections | (Δ/σ)max = 0.001 |
| 571 parameters | Δρmax = 0.41 e Å−3 |
| 4 restraints | Δρmin = −0.44 e Å−3 |
Special details
| Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes.Least-squares planes (x,y,z in crystal coordinates) and deviations from them (* indicates atom used to define plane)2.5074 (0.0102) x + 8.4452 (0.0095) y + 7.9575 (0.0134) z = 6.0445 (0.0089)* 0.1022 (0.0012) C2 * -0.1077 (0.0012) C3 * 0.1077 (0.0012) C4 * -0.1021 (0.0012) C5 0.4493 (0.0013) CrRms deviation of fitted atoms = 0.1050 |
| Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| Cr | 0.97125 (3) | 0.24895 (3) | 0.24580 (2) | 0.01723 (10) | |
| N1 | 1.03216 (19) | 0.34819 (17) | 0.31458 (13) | 0.0287 (5) | |
| C2 | 0.9557 (2) | 0.3156 (2) | 0.13640 (16) | 0.0229 (5) | |
| N2 | 0.9541 (2) | 0.35860 (19) | 0.07957 (15) | 0.0356 (6) | |
| C3 | 1.1055 (2) | 0.16264 (19) | 0.22511 (14) | 0.0194 (5) | |
| N3 | 1.17809 (19) | 0.10990 (18) | 0.21318 (14) | 0.0287 (5) | |
| C4 | 0.9671 (2) | 0.1357 (2) | 0.32438 (15) | 0.0213 (5) | |
| N4 | 0.96893 (19) | 0.07573 (18) | 0.37107 (14) | 0.0302 (5) | |
| C5 | 0.7926 (2) | 0.25946 (19) | 0.22163 (15) | 0.0202 (5) | |
| N5 | 0.6916 (2) | 0.25833 (18) | 0.20697 (14) | 0.0308 (5) | |
| P1 | 0.66573 (5) | 0.81476 (5) | 0.15855 (4) | 0.01645 (14) | |
| C11 | 0.7218 (2) | 0.88033 (19) | 0.08928 (14) | 0.0197 (5) | |
| C12 | 0.7143 (3) | 0.9925 (2) | 0.08778 (17) | 0.0309 (6) | |
| H12 | 0.6772 | 1.0353 | 0.1223 | 0.037* | |
| C13 | 0.7613 (3) | 1.0411 (2) | 0.03558 (18) | 0.0405 (7) | |
| H13 | 0.7575 | 1.1176 | 0.0351 | 0.049* | |
| C14 | 0.8135 (3) | 0.9788 (2) | −0.01559 (17) | 0.0344 (7) | |
| H14 | 0.8452 | 1.0125 | −0.0514 | 0.041* | |
| C15 | 0.8200 (2) | 0.8679 (2) | −0.01495 (16) | 0.0292 (6) | |
| H15 | 0.8559 | 0.8254 | −0.0505 | 0.035* | |
| C16 | 0.7746 (2) | 0.8180 (2) | 0.03735 (16) | 0.0257 (6) | |
| H16 | 0.7794 | 0.7415 | 0.0378 | 0.031* | |
| C21 | 0.5901 (2) | 0.91094 (18) | 0.20943 (14) | 0.0172 (5) | |
| C22 | 0.4676 (2) | 0.89460 (19) | 0.18939 (15) | 0.0209 (5) | |
| H22 | 0.4259 | 0.8310 | 0.1516 | 0.025* | |
| C23 | 0.4068 (2) | 0.9710 (2) | 0.22452 (16) | 0.0244 (6) | |
| H23 | 0.3235 | 0.9597 | 0.2109 | 0.029* | |
| C24 | 0.4674 (2) | 1.0636 (2) | 0.27944 (15) | 0.0246 (6) | |
| H24 | 0.4255 | 1.1160 | 0.3035 | 0.030* | |
| C25 | 0.5885 (2) | 1.08050 (19) | 0.29953 (15) | 0.0242 (6) | |
| H25 | 0.6293 | 1.1445 | 0.3372 | 0.029* | |
| C26 | 0.6512 (2) | 1.00486 (19) | 0.26518 (14) | 0.0201 (5) | |
| H26 | 0.7346 | 1.0166 | 0.2793 | 0.024* | |
| C31 | 0.5587 (2) | 0.70555 (18) | 0.09688 (14) | 0.0183 (5) | |
| C32 | 0.5339 (2) | 0.62444 (19) | 0.13962 (15) | 0.0225 (5) | |
| H32 | 0.5833 | 0.6205 | 0.1972 | 0.027* | |
| C33 | 0.4381 (2) | 0.5503 (2) | 0.09839 (17) | 0.0271 (6) | |
| H33 | 0.4211 | 0.4956 | 0.1276 | 0.032* | |
| C34 | 0.3670 (2) | 0.5554 (2) | 0.01483 (17) | 0.0305 (6) | |
| H34 | 0.3006 | 0.5044 | −0.0132 | 0.037* | |
| C35 | 0.3914 (2) | 0.6341 (2) | −0.02869 (17) | 0.0316 (6) | |
| H35 | 0.3425 | 0.6363 | −0.0866 | 0.038* | |
| C36 | 0.4872 (2) | 0.7097 (2) | 0.01209 (15) | 0.0243 (6) | |
| H36 | 0.5040 | 0.7640 | −0.0176 | 0.029* | |
| C41 | 0.7823 (2) | 0.75651 (19) | 0.23712 (15) | 0.0193 (5) | |
| C42 | 0.8155 (2) | 0.7932 (2) | 0.32521 (15) | 0.0237 (5) | |
| H42 | 0.7795 | 0.8538 | 0.3458 | 0.028* | |
| C43 | 0.9016 (2) | 0.7406 (2) | 0.38291 (16) | 0.0290 (6) | |
| H43 | 0.9252 | 0.7662 | 0.4430 | 0.035* | |
| C44 | 0.9530 (2) | 0.6517 (2) | 0.35352 (17) | 0.0284 (6) | |
| H44 | 1.0107 | 0.6155 | 0.3934 | 0.034* | |
| C45 | 0.9207 (2) | 0.6150 (2) | 0.26603 (17) | 0.0297 (6) | |
| H45 | 0.9566 | 0.5538 | 0.2460 | 0.036* | |
| C46 | 0.8365 (2) | 0.6671 (2) | 0.20761 (16) | 0.0276 (6) | |
| H46 | 0.8152 | 0.6423 | 0.1475 | 0.033* | |
| P2 | 0.65911 (5) | 0.34699 (5) | 0.62208 (4) | 0.01508 (13) | |
| C51 | 0.72980 (19) | 0.38164 (18) | 0.54899 (14) | 0.0158 (5) | |
| C52 | 0.7530 (2) | 0.29675 (19) | 0.49283 (14) | 0.0183 (5) | |
| H52 | 0.7296 | 0.2232 | 0.4926 | 0.022* | |
| C53 | 0.8102 (2) | 0.3205 (2) | 0.43758 (14) | 0.0217 (5) | |
| H53 | 0.8276 | 0.2629 | 0.4004 | 0.026* | |
| C54 | 0.8421 (2) | 0.4279 (2) | 0.43637 (15) | 0.0224 (5) | |
| H54 | 0.8807 | 0.4439 | 0.3980 | 0.027* | |
| C55 | 0.8178 (2) | 0.5120 (2) | 0.49107 (15) | 0.0231 (5) | |
| H55 | 0.8393 | 0.5856 | 0.4899 | 0.028* | |
| C56 | 0.7620 (2) | 0.48910 (19) | 0.54750 (14) | 0.0182 (5) | |
| H56 | 0.7459 | 0.5470 | 0.5852 | 0.022* | |
| C61 | 0.6187 (2) | 0.46906 (18) | 0.67540 (14) | 0.0179 (5) | |
| C62 | 0.4995 (2) | 0.4868 (2) | 0.66260 (15) | 0.0220 (5) | |
| H62 | 0.4386 | 0.4351 | 0.6246 | 0.026* | |
| C63 | 0.4707 (2) | 0.5803 (2) | 0.70586 (16) | 0.0287 (6) | |
| H63 | 0.3897 | 0.5925 | 0.6975 | 0.034* | |
| C64 | 0.5586 (3) | 0.6560 (2) | 0.76098 (16) | 0.0303 (6) | |
| H64 | 0.5377 | 0.7200 | 0.7901 | 0.036* | |
| C65 | 0.6773 (2) | 0.6389 (2) | 0.77403 (16) | 0.0281 (6) | |
| H65 | 0.7376 | 0.6912 | 0.8120 | 0.034* | |
| C66 | 0.7081 (2) | 0.54541 (19) | 0.73164 (15) | 0.0238 (6) | |
| H66 | 0.7893 | 0.5333 | 0.7407 | 0.029* | |
| C71 | 0.5277 (2) | 0.26263 (18) | 0.55768 (14) | 0.0169 (5) | |
| C72 | 0.4935 (2) | 0.1720 (2) | 0.58589 (16) | 0.0252 (6) | |
| H72 | 0.5418 | 0.1514 | 0.6389 | 0.030* | |
| C73 | 0.3883 (2) | 0.1116 (2) | 0.53614 (17) | 0.0314 (6) | |
| H73 | 0.3649 | 0.0494 | 0.5551 | 0.038* | |
| C74 | 0.3175 (2) | 0.1417 (2) | 0.45915 (16) | 0.0264 (6) | |
| H74 | 0.2454 | 0.1002 | 0.4256 | 0.032* | |
| C75 | 0.3512 (2) | 0.23212 (19) | 0.43086 (15) | 0.0221 (5) | |
| H75 | 0.3022 | 0.2528 | 0.3781 | 0.027* | |
| C76 | 0.4566 (2) | 0.29250 (18) | 0.47958 (14) | 0.0189 (5) | |
| H76 | 0.4804 | 0.3540 | 0.4599 | 0.023* | |
| C81 | 0.75605 (19) | 0.27572 (18) | 0.70289 (14) | 0.0164 (5) | |
| C82 | 0.8202 (2) | 0.19116 (19) | 0.67944 (16) | 0.0232 (5) | |
| H82 | 0.8129 | 0.1710 | 0.6208 | 0.028* | |
| C83 | 0.8942 (2) | 0.1368 (2) | 0.74176 (17) | 0.0276 (6) | |
| H83 | 0.9374 | 0.0789 | 0.7257 | 0.033* | |
| C84 | 0.9056 (2) | 0.1663 (2) | 0.82709 (17) | 0.0283 (6) | |
| H84 | 0.9572 | 0.1289 | 0.8696 | 0.034* | |
| C85 | 0.8427 (2) | 0.2498 (2) | 0.85098 (16) | 0.0302 (6) | |
| H85 | 0.8510 | 0.2699 | 0.9099 | 0.036* | |
| C86 | 0.7672 (2) | 0.3046 (2) | 0.78916 (15) | 0.0247 (6) | |
| H86 | 0.7233 | 0.3617 | 0.8055 | 0.030* | |
| O1 | 0.8836 (2) | 0.57434 (19) | 0.01213 (19) | 0.0611 (7) | |
| H1A | 0.910 (4) | 0.513 (2) | 0.025 (3) | 0.092* | |
| H1B | 0.931 (3) | 0.592 (3) | −0.013 (2) | 0.092* | |
| O2 | 0.85439 (16) | 0.04312 (15) | 0.49856 (12) | 0.0291 (4) | |
| H2A | 0.884 (3) | 0.052 (2) | 0.4627 (16) | 0.044* | |
| H2B | 0.906 (2) | 0.016 (2) | 0.5356 (16) | 0.044* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| Cr | 0.0185 (2) | 0.0193 (2) | 0.0159 (2) | 0.00301 (16) | 0.00804 (16) | 0.00416 (15) |
| N1 | 0.0316 (13) | 0.0278 (12) | 0.0253 (11) | −0.0007 (10) | 0.0099 (10) | 0.0000 (9) |
| C2 | 0.0230 (14) | 0.0229 (13) | 0.0254 (14) | 0.0029 (11) | 0.0108 (11) | 0.0058 (11) |
| N2 | 0.0412 (14) | 0.0395 (14) | 0.0325 (13) | 0.0045 (11) | 0.0165 (11) | 0.0162 (11) |
| C3 | 0.0204 (13) | 0.0226 (13) | 0.0177 (12) | −0.0011 (11) | 0.0089 (10) | 0.0053 (10) |
| N3 | 0.0271 (12) | 0.0321 (12) | 0.0328 (13) | 0.0064 (10) | 0.0162 (10) | 0.0088 (10) |
| C4 | 0.0174 (13) | 0.0286 (14) | 0.0214 (13) | 0.0058 (10) | 0.0106 (11) | 0.0049 (11) |
| N4 | 0.0309 (13) | 0.0384 (13) | 0.0302 (12) | 0.0106 (10) | 0.0168 (10) | 0.0174 (11) |
| C5 | 0.0258 (15) | 0.0203 (13) | 0.0199 (12) | 0.0081 (11) | 0.0128 (11) | 0.0073 (10) |
| N5 | 0.0293 (14) | 0.0351 (13) | 0.0353 (13) | 0.0121 (10) | 0.0174 (11) | 0.0132 (10) |
| P1 | 0.0173 (3) | 0.0175 (3) | 0.0153 (3) | 0.0030 (2) | 0.0064 (3) | 0.0029 (2) |
| C11 | 0.0192 (13) | 0.0224 (13) | 0.0177 (12) | 0.0014 (10) | 0.0057 (10) | 0.0053 (10) |
| C12 | 0.0474 (18) | 0.0236 (14) | 0.0283 (14) | 0.0048 (12) | 0.0206 (13) | 0.0061 (11) |
| C13 | 0.071 (2) | 0.0251 (15) | 0.0340 (16) | 0.0009 (14) | 0.0282 (16) | 0.0087 (13) |
| C14 | 0.0440 (17) | 0.0393 (17) | 0.0251 (14) | −0.0057 (13) | 0.0162 (13) | 0.0110 (12) |
| C15 | 0.0295 (15) | 0.0398 (16) | 0.0215 (13) | 0.0035 (12) | 0.0128 (12) | 0.0049 (12) |
| C16 | 0.0302 (15) | 0.0236 (13) | 0.0264 (14) | 0.0061 (11) | 0.0126 (12) | 0.0066 (11) |
| C21 | 0.0202 (13) | 0.0175 (12) | 0.0158 (11) | 0.0042 (10) | 0.0078 (10) | 0.0048 (9) |
| C22 | 0.0200 (13) | 0.0202 (13) | 0.0205 (12) | 0.0010 (10) | 0.0051 (10) | 0.0020 (10) |
| C23 | 0.0196 (13) | 0.0259 (14) | 0.0308 (14) | 0.0056 (11) | 0.0121 (11) | 0.0062 (11) |
| C24 | 0.0337 (15) | 0.0202 (13) | 0.0253 (13) | 0.0082 (11) | 0.0166 (12) | 0.0047 (11) |
| C25 | 0.0343 (15) | 0.0179 (13) | 0.0199 (13) | −0.0009 (11) | 0.0103 (11) | −0.0006 (10) |
| C26 | 0.0187 (13) | 0.0215 (13) | 0.0200 (12) | 0.0003 (10) | 0.0064 (10) | 0.0040 (10) |
| C31 | 0.0186 (12) | 0.0190 (12) | 0.0171 (12) | 0.0047 (10) | 0.0066 (10) | 0.0012 (10) |
| C32 | 0.0262 (14) | 0.0235 (13) | 0.0190 (12) | 0.0038 (11) | 0.0092 (11) | 0.0039 (10) |
| C33 | 0.0290 (15) | 0.0220 (13) | 0.0331 (15) | −0.0011 (11) | 0.0146 (12) | 0.0042 (11) |
| C34 | 0.0225 (14) | 0.0258 (14) | 0.0373 (16) | −0.0020 (11) | 0.0056 (12) | −0.0022 (12) |
| C35 | 0.0287 (15) | 0.0303 (15) | 0.0246 (14) | 0.0033 (12) | −0.0034 (12) | 0.0000 (12) |
| C36 | 0.0285 (14) | 0.0226 (13) | 0.0219 (13) | 0.0047 (11) | 0.0083 (11) | 0.0042 (11) |
| C41 | 0.0163 (12) | 0.0206 (12) | 0.0207 (12) | 0.0010 (10) | 0.0048 (10) | 0.0066 (10) |
| C42 | 0.0250 (14) | 0.0232 (13) | 0.0228 (13) | 0.0014 (11) | 0.0079 (11) | 0.0040 (11) |
| C43 | 0.0285 (15) | 0.0325 (15) | 0.0220 (13) | −0.0030 (12) | 0.0026 (12) | 0.0071 (11) |
| C44 | 0.0166 (13) | 0.0320 (15) | 0.0354 (15) | −0.0001 (11) | 0.0029 (12) | 0.0163 (12) |
| C45 | 0.0246 (14) | 0.0290 (15) | 0.0360 (16) | 0.0102 (12) | 0.0096 (12) | 0.0089 (12) |
| C46 | 0.0274 (14) | 0.0309 (15) | 0.0236 (13) | 0.0084 (12) | 0.0079 (12) | 0.0034 (11) |
| P2 | 0.0148 (3) | 0.0161 (3) | 0.0153 (3) | 0.0016 (2) | 0.0065 (2) | 0.0024 (2) |
| C51 | 0.0124 (11) | 0.0202 (12) | 0.0146 (11) | 0.0021 (9) | 0.0039 (9) | 0.0040 (9) |
| C52 | 0.0186 (12) | 0.0188 (12) | 0.0168 (12) | 0.0012 (10) | 0.0051 (10) | 0.0031 (10) |
| C53 | 0.0206 (13) | 0.0290 (14) | 0.0143 (12) | 0.0045 (11) | 0.0056 (10) | 0.0009 (10) |
| C54 | 0.0188 (13) | 0.0326 (14) | 0.0178 (12) | 0.0000 (11) | 0.0082 (10) | 0.0058 (11) |
| C55 | 0.0233 (13) | 0.0227 (13) | 0.0236 (13) | −0.0033 (11) | 0.0075 (11) | 0.0067 (11) |
| C56 | 0.0190 (12) | 0.0193 (12) | 0.0168 (12) | 0.0016 (10) | 0.0073 (10) | 0.0018 (10) |
| C61 | 0.0233 (13) | 0.0178 (12) | 0.0165 (12) | 0.0021 (10) | 0.0115 (10) | 0.0039 (10) |
| C62 | 0.0233 (13) | 0.0266 (13) | 0.0176 (12) | 0.0068 (11) | 0.0082 (11) | 0.0054 (10) |
| C63 | 0.0305 (15) | 0.0355 (15) | 0.0245 (14) | 0.0174 (12) | 0.0128 (12) | 0.0092 (12) |
| C64 | 0.0522 (19) | 0.0199 (13) | 0.0276 (14) | 0.0136 (13) | 0.0231 (14) | 0.0083 (11) |
| C65 | 0.0439 (17) | 0.0189 (13) | 0.0265 (14) | −0.0050 (12) | 0.0204 (13) | −0.0001 (11) |
| C66 | 0.0256 (14) | 0.0240 (13) | 0.0268 (14) | −0.0019 (11) | 0.0159 (12) | 0.0027 (11) |
| C71 | 0.0155 (12) | 0.0185 (12) | 0.0176 (12) | 0.0016 (10) | 0.0073 (10) | 0.0018 (10) |
| C72 | 0.0256 (14) | 0.0298 (14) | 0.0209 (13) | −0.0021 (11) | 0.0069 (11) | 0.0094 (11) |
| C73 | 0.0296 (15) | 0.0342 (15) | 0.0309 (15) | −0.0099 (12) | 0.0087 (12) | 0.0120 (12) |
| C74 | 0.0217 (14) | 0.0296 (14) | 0.0256 (14) | −0.0056 (11) | 0.0070 (11) | 0.0009 (11) |
| C75 | 0.0212 (13) | 0.0262 (13) | 0.0170 (12) | 0.0028 (11) | 0.0044 (10) | 0.0026 (10) |
| C76 | 0.0231 (13) | 0.0156 (12) | 0.0197 (12) | 0.0019 (10) | 0.0092 (11) | 0.0039 (10) |
| C81 | 0.0139 (12) | 0.0178 (12) | 0.0171 (12) | −0.0013 (9) | 0.0046 (10) | 0.0037 (9) |
| C82 | 0.0266 (14) | 0.0242 (13) | 0.0213 (13) | 0.0031 (11) | 0.0106 (11) | 0.0054 (10) |
| C83 | 0.0235 (14) | 0.0271 (14) | 0.0373 (15) | 0.0080 (11) | 0.0133 (12) | 0.0141 (12) |
| C84 | 0.0190 (13) | 0.0331 (15) | 0.0306 (15) | −0.0014 (11) | 0.0006 (11) | 0.0178 (12) |
| C85 | 0.0351 (16) | 0.0342 (15) | 0.0156 (13) | −0.0035 (13) | 0.0014 (12) | 0.0046 (11) |
| C86 | 0.0286 (14) | 0.0261 (14) | 0.0186 (13) | 0.0022 (11) | 0.0078 (11) | 0.0020 (11) |
| O1 | 0.0613 (16) | 0.0420 (13) | 0.112 (2) | 0.0201 (12) | 0.0621 (16) | 0.0316 (14) |
| O2 | 0.0262 (10) | 0.0365 (11) | 0.0308 (11) | 0.0065 (8) | 0.0147 (9) | 0.0133 (9) |
Geometric parameters (Å, °)
| Cr—N1 | 1.538 (2) | C45—H45 | 0.9500 |
| Cr—C2 | 2.066 (3) | C46—H46 | 0.9500 |
| Cr—C3 | 2.040 (3) | P2—C71 | 1.794 (2) |
| Cr—C4 | 2.068 (3) | P2—C61 | 1.798 (2) |
| Cr—C5 | 2.049 (3) | P2—C81 | 1.802 (2) |
| C2—N2 | 1.150 (3) | P2—C51 | 1.803 (2) |
| C3—N3 | 1.147 (3) | C51—C56 | 1.389 (3) |
| C4—N4 | 1.148 (3) | C51—C52 | 1.401 (3) |
| C5—N5 | 1.150 (3) | C52—C53 | 1.385 (3) |
| P1—C31 | 1.789 (3) | C52—H52 | 0.9500 |
| P1—C21 | 1.795 (2) | C53—C54 | 1.387 (3) |
| P1—C41 | 1.800 (2) | C53—H53 | 0.9500 |
| P1—C11 | 1.803 (2) | C54—C55 | 1.385 (3) |
| C11—C16 | 1.394 (3) | C54—H54 | 0.9500 |
| C11—C12 | 1.396 (3) | C55—C56 | 1.387 (3) |
| C12—C13 | 1.388 (4) | C55—H55 | 0.9500 |
| C12—H12 | 0.9500 | C56—H56 | 0.9500 |
| C13—C14 | 1.380 (4) | C61—C62 | 1.396 (3) |
| C13—H13 | 0.9500 | C61—C66 | 1.401 (3) |
| C14—C15 | 1.378 (4) | C62—C63 | 1.386 (3) |
| C14—H14 | 0.9500 | C62—H62 | 0.9500 |
| C15—C16 | 1.386 (3) | C63—C64 | 1.381 (4) |
| C15—H15 | 0.9500 | C63—H63 | 0.9500 |
| C16—H16 | 0.9500 | C64—C65 | 1.389 (4) |
| C21—C22 | 1.396 (3) | C64—H64 | 0.9500 |
| C21—C26 | 1.401 (3) | C65—C66 | 1.389 (3) |
| C22—C23 | 1.384 (3) | C65—H65 | 0.9500 |
| C22—H22 | 0.9500 | C66—H66 | 0.9500 |
| C23—C24 | 1.382 (4) | C71—C72 | 1.389 (3) |
| C23—H23 | 0.9500 | C71—C76 | 1.398 (3) |
| C24—C25 | 1.381 (4) | C72—C73 | 1.388 (4) |
| C24—H24 | 0.9500 | C72—H72 | 0.9500 |
| C25—C26 | 1.388 (3) | C73—C74 | 1.384 (4) |
| C25—H25 | 0.9500 | C73—H73 | 0.9500 |
| C26—H26 | 0.9500 | C74—C75 | 1.385 (3) |
| C31—C36 | 1.392 (3) | C74—H74 | 0.9500 |
| C31—C32 | 1.399 (3) | C75—C76 | 1.386 (3) |
| C32—C33 | 1.376 (4) | C75—H75 | 0.9500 |
| C32—H32 | 0.9500 | C76—H76 | 0.9500 |
| C33—C34 | 1.376 (4) | C81—C86 | 1.394 (3) |
| C33—H33 | 0.9500 | C81—C82 | 1.394 (3) |
| C34—C35 | 1.383 (4) | C82—C83 | 1.381 (3) |
| C34—H34 | 0.9500 | C82—H82 | 0.9500 |
| C35—C36 | 1.387 (4) | C83—C84 | 1.379 (4) |
| C35—H35 | 0.9500 | C83—H83 | 0.9500 |
| C36—H36 | 0.9500 | C84—C85 | 1.379 (4) |
| C41—C42 | 1.391 (3) | C84—H84 | 0.9500 |
| C41—C46 | 1.402 (3) | C85—C86 | 1.387 (4) |
| C42—C43 | 1.391 (4) | C85—H85 | 0.9500 |
| C42—H42 | 0.9500 | C86—H86 | 0.9500 |
| C43—C44 | 1.377 (4) | O1—H1A | 0.856 (19) |
| C43—H43 | 0.9500 | O1—H1B | 0.854 (19) |
| C44—C45 | 1.383 (4) | O2—H2A | 0.814 (17) |
| C44—H44 | 0.9500 | O2—H2B | 0.824 (17) |
| C45—C46 | 1.383 (4) | ||
| N1—Cr—C2 | 99.23 (11) | C46—C45—C44 | 120.2 (2) |
| N1—Cr—C3 | 105.79 (11) | C46—C45—H45 | 119.9 |
| N1—Cr—C4 | 99.95 (11) | C44—C45—H45 | 119.9 |
| N1—Cr—C5 | 105.70 (11) | C45—C46—C41 | 120.0 (2) |
| C3—Cr—C5 | 148.38 (10) | C45—C46—H46 | 120.0 |
| C3—Cr—C2 | 87.56 (10) | C41—C46—H46 | 120.0 |
| C5—Cr—C2 | 90.28 (10) | C71—P2—C61 | 109.65 (11) |
| C3—Cr—C4 | 84.71 (9) | C71—P2—C81 | 110.71 (11) |
| C5—Cr—C4 | 87.09 (10) | C61—P2—C81 | 108.47 (11) |
| C2—Cr—C4 | 160.64 (10) | C71—P2—C51 | 106.53 (11) |
| N2—C2—Cr | 174.6 (2) | C61—P2—C51 | 110.02 (11) |
| N3—C3—Cr | 177.0 (2) | C81—P2—C51 | 111.46 (11) |
| N4—C4—Cr | 176.8 (2) | C56—C51—C52 | 119.7 (2) |
| N5—C5—Cr | 175.7 (2) | C56—C51—P2 | 121.93 (17) |
| C31—P1—C21 | 106.70 (11) | C52—C51—P2 | 118.37 (17) |
| C31—P1—C41 | 107.08 (11) | C53—C52—C51 | 119.7 (2) |
| C21—P1—C41 | 111.23 (11) | C53—C52—H52 | 120.1 |
| C31—P1—C11 | 110.46 (11) | C51—C52—H52 | 120.1 |
| C21—P1—C11 | 109.57 (11) | C52—C53—C54 | 120.3 (2) |
| C41—P1—C11 | 111.66 (11) | C52—C53—H53 | 119.9 |
| C16—C11—C12 | 119.8 (2) | C54—C53—H53 | 119.9 |
| C16—C11—P1 | 119.24 (18) | C55—C54—C53 | 120.0 (2) |
| C12—C11—P1 | 120.99 (18) | C55—C54—H54 | 120.0 |
| C13—C12—C11 | 119.6 (2) | C53—C54—H54 | 120.0 |
| C13—C12—H12 | 120.2 | C54—C55—C56 | 120.2 (2) |
| C11—C12—H12 | 120.2 | C54—C55—H55 | 119.9 |
| C14—C13—C12 | 120.3 (3) | C56—C55—H55 | 119.9 |
| C14—C13—H13 | 119.9 | C55—C56—C51 | 120.0 (2) |
| C12—C13—H13 | 119.9 | C55—C56—H56 | 120.0 |
| C15—C14—C13 | 120.3 (2) | C51—C56—H56 | 120.0 |
| C15—C14—H14 | 119.8 | C62—C61—C66 | 120.1 (2) |
| C13—C14—H14 | 119.8 | C62—C61—P2 | 120.50 (18) |
| C14—C15—C16 | 120.3 (2) | C66—C61—P2 | 119.43 (18) |
| C14—C15—H15 | 119.9 | C63—C62—C61 | 119.4 (2) |
| C16—C15—H15 | 119.9 | C63—C62—H62 | 120.3 |
| C15—C16—C11 | 119.8 (2) | C61—C62—H62 | 120.3 |
| C15—C16—H16 | 120.1 | C64—C63—C62 | 120.7 (2) |
| C11—C16—H16 | 120.1 | C64—C63—H63 | 119.7 |
| C22—C21—C26 | 119.8 (2) | C62—C63—H63 | 119.7 |
| C22—C21—P1 | 119.34 (18) | C63—C64—C65 | 120.2 (2) |
| C26—C21—P1 | 120.78 (18) | C63—C64—H64 | 119.9 |
| C23—C22—C21 | 120.1 (2) | C65—C64—H64 | 119.9 |
| C23—C22—H22 | 120.0 | C66—C65—C64 | 120.0 (2) |
| C21—C22—H22 | 120.0 | C66—C65—H65 | 120.0 |
| C24—C23—C22 | 120.0 (2) | C64—C65—H65 | 120.0 |
| C24—C23—H23 | 120.0 | C65—C66—C61 | 119.6 (2) |
| C22—C23—H23 | 120.0 | C65—C66—H66 | 120.2 |
| C25—C24—C23 | 120.3 (2) | C61—C66—H66 | 120.2 |
| C25—C24—H24 | 119.9 | C72—C71—C76 | 120.1 (2) |
| C23—C24—H24 | 119.9 | C72—C71—P2 | 121.21 (18) |
| C24—C25—C26 | 120.7 (2) | C76—C71—P2 | 118.66 (17) |
| C24—C25—H25 | 119.7 | C73—C72—C71 | 119.6 (2) |
| C26—C25—H25 | 119.7 | C73—C72—H72 | 120.2 |
| C25—C26—C21 | 119.2 (2) | C71—C72—H72 | 120.2 |
| C25—C26—H26 | 120.4 | C74—C73—C72 | 120.4 (2) |
| C21—C26—H26 | 120.4 | C74—C73—H73 | 119.8 |
| C36—C31—C32 | 119.7 (2) | C72—C73—H73 | 119.8 |
| C36—C31—P1 | 121.13 (18) | C73—C74—C75 | 120.2 (2) |
| C32—C31—P1 | 118.33 (18) | C73—C74—H74 | 119.9 |
| C33—C32—C31 | 120.0 (2) | C75—C74—H74 | 119.9 |
| C33—C32—H32 | 120.0 | C74—C75—C76 | 120.0 (2) |
| C31—C32—H32 | 120.0 | C74—C75—H75 | 120.0 |
| C32—C33—C34 | 120.1 (2) | C76—C75—H75 | 120.0 |
| C32—C33—H33 | 120.0 | C75—C76—C71 | 119.8 (2) |
| C34—C33—H33 | 120.0 | C75—C76—H76 | 120.1 |
| C33—C34—C35 | 120.5 (2) | C71—C76—H76 | 120.1 |
| C33—C34—H34 | 119.7 | C86—C81—C82 | 119.6 (2) |
| C35—C34—H34 | 119.7 | C86—C81—P2 | 120.22 (18) |
| C34—C35—C36 | 120.1 (2) | C82—C81—P2 | 120.14 (17) |
| C34—C35—H35 | 120.0 | C83—C82—C81 | 119.8 (2) |
| C36—C35—H35 | 120.0 | C83—C82—H82 | 120.1 |
| C35—C36—C31 | 119.5 (2) | C81—C82—H82 | 120.1 |
| C35—C36—H36 | 120.2 | C84—C83—C82 | 120.4 (2) |
| C31—C36—H36 | 120.2 | C84—C83—H83 | 119.8 |
| C42—C41—C46 | 119.5 (2) | C82—C83—H83 | 119.8 |
| C42—C41—P1 | 122.46 (18) | C83—C84—C85 | 120.3 (2) |
| C46—C41—P1 | 117.92 (18) | C83—C84—H84 | 119.8 |
| C43—C42—C41 | 119.6 (2) | C85—C84—H84 | 119.8 |
| C43—C42—H42 | 120.2 | C84—C85—C86 | 120.1 (2) |
| C41—C42—H42 | 120.2 | C84—C85—H85 | 120.0 |
| C44—C43—C42 | 120.5 (2) | C86—C85—H85 | 120.0 |
| C44—C43—H43 | 119.8 | C85—C86—C81 | 119.8 (2) |
| C42—C43—H43 | 119.8 | C85—C86—H86 | 120.1 |
| C43—C44—C45 | 120.2 (2) | C81—C86—H86 | 120.1 |
| C43—C44—H44 | 119.9 | H1A—O1—H1B | 100 (4) |
| C45—C44—H44 | 119.9 | H2A—O2—H2B | 105 (3) |
Hydrogen-bond geometry (Å, °)
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1A···N2 | 0.86 (2) | 2.23 (2) | 3.065 (3) | 167 (4) |
| O1—H1B···N2i | 0.85 (2) | 2.18 (2) | 3.035 (3) | 177 (4) |
| O2—H2A···N4 | 0.81 (2) | 2.16 (2) | 2.973 (3) | 178 (3) |
| O2—H2B···N4ii | 0.82 (2) | 2.22 (2) | 3.039 (3) | 173 (3) |
Symmetry codes: (i) −x+2, −y+1, −z; (ii) −x+2, −y, −z+1.
Table 3 Comparative geometric parameters (Å) of cyanidonitridometalates of chromate(V) and manganate(V).
| [Cr(CN)4(N)]2-a | [Mn(CN)4(N)]2-b | [Cr(CN)5(N)]3-c | [Mn(CN)5(N)]3-c | |
| M≡N | 1.538 (2) | 1.507 (2) | 1.594 (9) | 1.499 (8) |
| M—Ccis | 2.040 (3)-2.068 (3) | 1.974 (2)-1.995 (2) | 2.039 (7)-2.08 (2) | 1.985 (6)-2.001 (7) |
| M—Ctrans | 2.299 (12) | 2.243 (7) | ||
| M—oopd | 0.449 | 0.436 | 0.255 | 0.222 |
Notes: (a) this work; (b) Bendix et al. (1998); (c) Bendix et al. (2000); (d) oop = out-of-plane.
Footnotes
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: FJ2383).
References
- Baldas, J., Boas, J. F., Colmanet, S. F. & Mackay, M. F. (1990). Inorg. Chim. Acta, 170, 233–239.
- Bendix, J., Deeth, R. J., Weyhermüller, T., Bill, E. & Wieghardt, K. (2000). Inorg. Chem. 39, 930–938. [DOI] [PubMed]
- Bendix, J., Meyer, K., Weyhermüller, T., Bill, E., Metzler-Nolte, N. & Wieghardt, K. (1998). Inorg. Chem. 37, 1767–1775.
- Birk, T. & Bendix, J. (2003). Inorg. Chem. 42, 7608–7615. [DOI] [PubMed]
- Britten, J. F., Lock, C. J. L. & Wei, Y. (1993). Acta Cryst. C49, 1277–1280.
- Che, C. M., Lam, H. W. & Mak, T. C. W. (1989). J. Chem. Soc. Chem. Commun. pp. 1529–1531.
- Coppens, P. (1970). Crystallographic Computing, edited by F. R. Ahmed, S. R. Hall & C. P. Huber, pp. 255–270. Copenhagen: Munksgaard.
- Duisenberg, A. J. M., Kroon-Batenburg, L. M. J. & Schreurs, A. M. M. (2003). J. Appl. Cryst. 36, 220–229.
- Farrugia, L. J. (1997). J. Appl. Cryst. 30, 565.
- Nonius (1999). COLLECT Nonius BV, Delft, The Netherlands.
- Purcell, W., Potgieter, I. Z., Damoense, L. J. & Leipolldt, J. S. (1991). Transition Met. Chem. 16, 473–475.
- Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. [DOI] [PubMed]
- Westhuizen, H. J. van der, Basson, S. S., Leipoldt, J. G. & Purcell, W. (1994). Transition Met. Chem. 19, 582–584.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536811002108/fj2383sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536811002108/fj2383Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report