Abstract
Liver cirrhosis is often complicated by an impaired renal excretion of water and sodium. Diuretics tend to further deteriorate renal function. It is unknown whether chronic selective adenosine A1 receptor blockade, via inhibition of the hepatorenal reflex and the tubuloglomerular feedback, might exert diuretic and natriuretic effects without a reduction of the glomerular filtration rate. In healthy animals intravenous treatment with the novel A1 receptor antagonist SLV329 resulted in a strong dose-dependent diuretic (up to 3.4-fold) and natriuretic (up to 13.5-fold) effect without affecting creatinine clearance. Male Wistar rats with thioacetamide-induced liver cirrhosis received SLV329, vehicle or furosemide for 12 weeks. The creatinine clearance of cirrhotic animals decreased significantly (−36.5%, p<0.05), especially in those receiving furosemide (−41.9%, p<0.01). SLV329 was able to prevent this decline of creatinine clearance. Mortality was significantly lower in cirrhotic animals treated with SLV329 in comparison to animals treated with furosemide (17% vs. 54%, p<0.05). SLV329 did not relevantly influence the degree of liver fibrosis, kidney histology or expression of hepatic or renal adenosine receptors. In conclusion, chronic treatment with SLV329 prevented the decrease of creatinine clearance in a rat model of liver cirrhosis. Further studies will have to establish whether adenosine A1 receptor antagonists are clinically beneficial at different stages of liver cirrhosis.
Introduction
Liver cirrhosis is often complicated by an impaired renal capacity of maintaining water and sodium balance. Splanchnic arterial vasodilatation due to an increased release of endogenous vasodilators leads to compensatory activation of the endogenous vasoconstrictor systems: the sympathetic nervous system, the renin-angiotensin system and the non-osmotic release of vasopressin [1]. This causes renal sodium/water retention and renal vasoconstriction eventually leading to the hepatorenal syndrome. In many patients, water retention can be controlled by sodium restriction and diuretics. However, diuretic therapy often entails deterioration of renal function [1]. There is an urgent clinical need for alternative pharmacological approaches.
The adenosine system with its four receptors (A1, A2A, A2B and A3) is involved in several key functions of both liver and kidneys [2]. Decreasing the portal flow, as it is the case in cirrhosis, results in an activation of the hepatic adenosine system [3]. Adenosine, via A1 receptors (A1R), serves as a mediator for triggering the hepatorenal reflex leading to renal water and sodium retention [4], [5]. However, the exact localization of adenosine receptors within the liver remains unclear. In the kidney A1R are highly expressed in the preglomerular microcirculation, but also on the proximal tubules and other renal structures [6]. Short-term infusion of selective A1R antagonists inhibits the tubuloglomerular feedback and causes diuresis and natriuresis [7], [8]. Blockade of hepatic and renal A1R could therefore provide a new therapeutic option in conditions with sodium and water retention such as liver cirrhosis [9].
Thioacetamide has been widely used to induce chronic liver injury in animal models since it mimics the human disease closely. Rats treated with thioacetamide present with typical cirrhotic liver damage and an impaired ability of excreting sodium and water [10].
The first part of the present study evaluated the receptor binding affinity of the novel adenosine A1R antagonist SLV329 - a pyrimidine derivative - and confirmed its in vivo action on diuresis and natriuresis in healthy animals. As main hypothesis of this study, we tested whether SLV329 might exert diuretic effects without impairing renal function in animals with thioacetamide-induced liver cirrhosis.
Results
In vitro selectivity profile of SLV329
In receptor binding experiments using cloned human receptors SLV329 behaved as a potent (pKi 9.2) and selective A1R ligand. The affinity of SLV329 for the other adenosine receptors was at least 100-fold lower (Table 1). The receptor-binding affinities and enzyme inhibitory properties of SLV329 were evaluated in a series of 94 receptors and 6 phosphodiesterases including adenosine transporters and a wide range of adrenergic, muscarinic, nicotinic, dopaminergic, serotoninergic, histaminergic, glutamatergic, opioid, angiotensin, bradykinin and neuropeptide receptors as well as ion channels and transporter sites. Only the significant affinities of SLV329 are summarized in Table 1. Apart from adenosine receptors, significant binding was measured only for the high-affinity rolipram-binding site on phosphodiesterase 4. This effect was consistent with the inhibition of this enzyme by SLV329. However, phosphodiesterase 4 inhibition was much less potent than A1R binding. The ratio of the Ki value for phosphodiesterase 4 inhibition by SLV329 to that of the A1R binding was 3170. The activities on other phosphodiesterases were at least 10-fold lower.
Table 1. Receptor binding affinities and enzyme inhibitory properties of SLV329.
| Assay | Cell/tissue | Ligand | SLV329 Affinity |
| Adenosine A1 | CHO cells | 3H-DPCPX | 9.2±0.2 |
| Adenosine A2A | HEK293 cells | 3H-CGS21680 | 7.2±0.2 |
| Adenosine A3 | HEK293 cells | 3H-AB-MECA | 6.9±0.1 |
| Adenosine A2B | HEK293 cells | 3H-DPCPX | 6.3±0.1 |
| Phosphodiesterase 4 rolipram binding | Total brain | 3H-Rolipram | 6.1±0.2 |
| Phosphodiesterase 4 enzyme | U-937 cells | 3H-cAMP | 5.4±0.1 |
| Phosphodiesterase 6 enzyme | Retina | 3H-cAMP | 4.3±0.1 |
Only significant affinities and enzyme inhibition of SLV329 are shown.
Cells and tissues used to obtain receptors/enzymes and radioactive ligands of the respective assays are listed. Cells and tissues were provided by Cerep (Celle l'Evescault, France)
Results are expressed as pKi for radioligand affinity assays, and as pIC50 for enzyme inhibition. Mean ± standard deviation of at least 3 determinations.
The half-maximally inhibitory concentration (IC50) value of SLV329 is 3.2 µg/l.
Effects of SLV329 on renal function in healthy rats
Five different intravenous doses of SLV329 were tested in healthy anesthetized rats. Table 2 shows that SLV329 treatment resulted in a marked dose-dependent increase of diuresis by up to 3.4-fold and sodium excretion by up to 13.5-fold compared to the time-matched vehicle control group (both p<0.0001). The half-maximally effective concentration (EC50) values for diuresis and natriuresis were calculated to be 26 µg/l and 17 µg/l respectively. In contrast, the rate of potassium excretion was only modestly affected (up to only 1.7-fold vs. vehicle). The creatinine clearance was not affected by SLV329, even at the highest dose. SLV329 caused a dose-dependent increase in urinary adenosine excretion by up to 4.2-fold.
Table 2. Effects of different doses of SLV329 in anesthetized rats.
| SLV329 | bolus (µg/kg) | Vehicle | 18 | 54 | 180 | 540 | 945 |
| infusion (µg/kg*min) | Vehicle | 0.37 | 1.1 | 3.7 | 11 | 19.3 | |
| Plasma SLV329 (µg/l) | 0±0 | 11±2.6 | 31±4.4 | 54±12 | 86±21 | 182±63 | |
| Diuresis (fold vs. vehicle) | 1±0.1 | 1.4±0.1* | 2.5±0.3**** | 2.3±0.3*** | 3.4±0.4**** | 2.8±0.4**** | |
| Diuresis (ml/kg*h) | 1±0.1 | 1.4±0.1* | 2.5±0.3**** | 2.3±0.3*** | 3.4±0.4**** | 2.8±0.4**** | |
| Natriuresis (fold vs. vehicle) | 1±0.2 | 5.0±0.8*** | 7.6±0.9**** | 7.9±1.3**** | 13.5±1.3**** | 8.3±1.4**** | |
| Natriuresis (µg/kg*h) | 65±16 | 324±52*** | 497±62**** | 518±84**** | 882±87**** | 544±94**** | |
| Kaliuresis (fold vs. vehicle) | 1±0.1 | 1.6±0.2 | 1.4±0.1 | 1.7±0.1** | 1.3±0.1 | 1.6±0.2 | |
| Kaliuresis (µg/kg*h) | 106±13 | 167±21 | 146±13 | 175±16** | 136±12 | 170±22 | |
| Creatinine clearance (ml/min) | 1.9±0.3 | 3.2±1.1 | 2.0±0.3 | 1.6±0.2 | 2.6±0.8 | 2.4±0.7 | |
| Adenosine excretion (nmol/h) | 2.4±0.7 | 3.1±0.4* | 4.9±0.8** | 5.9±1.5* | 10.2±2.3*** | 9.8±1.9*** | |
| Body weight (g) | 363±3 | 360±2 | 356±3 | 362±2 | 364±2 | 357±2 | |
| Systolic blood pressure (mmHg) | 107±6 | 108±2 | 104±3 | 110±5 | 113±7 | 116±6 | |
| Heart rate (beats/min) | 313±19 | 317±23 | 328±21 | 350±10 | 294±23 | 301±30 | |
A bolus of SLV329 followed by an intravenous infusion over 3 hours produces a strong dose-dependent stimulation of renal water, sodium and adenosine excretion without any effect on creatinine clearance. Diuresis and electrolyte excretion is expressed as fold stimulation vs. time-matched vehicle controls.
Data are means ± standard error of the mean.
n = 9-13 animals per dose.
*p<0.05,
**p<0.01,
***p<0.001,
****p<0.0001 vs. time-matched vehicle controls.
Effects of SLV329 on renal function in thioacetamide-induced liver cirrhosis
Descriptive data, results from histological evaluation and Western blots are summarized in Table 3. Representative immunoblots for hepatic and renal adenosine receptors are shown in Figure 1. The results of plasma and urine analyses are shown in Table 4.
Table 3. Effects of treatment with furosemide and SLV329 in rats with and without liver cirrhosis.
| Group | Con | Con+Fur | Con+SLV | Cir | Cir+Fur | Cir+SLV |
| Bodyweight week 0 (g) | 358±6 | 344±10 | 356±11 | 351±6 | 354±6 | 352±5 |
| Bodyweight week 8 (g) | 443±11 | 433±13 | 439±15 | 333±8*** | 345±6*** | 335±5*** |
| Bodyweight week 16 (g) | 464±11 | 448±10 | 479±16 | 299±9*** | 263±13*** | 296±9*** # |
| Water intake week 0–8 (ml/kg*d) | 41±4 | 47±4 | 40±5 | 49±2* | 46±1 | 50±2 |
| Water intake week 8–16 (ml/kg*d) | 65±2 | 69±2 | 70±4 | 44±2*** | 52±2*** § | 50±2** § |
| Water intake week 11 (ml/kg*d) | 63±4 | 76±5§ | 72±3§§ | 48±2** | 52±2** | 54±4** § |
| Systolic BP week 0 (mmHg) | 124±2 | 126±6 | 130±3 | 126±1 | 125±2 | 127±2 |
| Systolic BP week 12 (mmHg) | 122±3 | 119±5 | 134±7§ | 110±5 | 118±5 | 118±3* |
| Liver weight (g) | 12.5±0.5 | 11.9±0.5 | 13.2±0.7 | 13.9±0.5 | 13.4±0.5* | 14.1±0.3 |
| Kidney weight (g) | 3.1±0.1 | 3.0±0.1 | 3.2±0.1 | 3.1±0.1 | 3.0±0.1 | 2.9±0.1 |
| Liver interstitial fibrosis (%) | 0.3±0.1 | 0.4±0.1 | 0.9±0.3 | 8.6±1.1*** | 5.2±1.1*** | 8.6±1.4*** |
| Kidney interstitial fibrosis (%) | 3.0±0.3 | 3.4±0.6 | 3.5±0.6 | 2.9±0.3 | 3.1±0.6 | 3.2±0.4 |
| Kidney media/lumen ratio | 3.3±0.4 | 3.1±0.2 | 2.6±0.3 | 2.6±0.2 | 2.8±0.2 | 3.0±0.3 |
| Glomerulosclerosis index (0–4) | 1.8±0.04 | 1.8±0.1 | 1.6±0.1 | 1.7±0.05 | 1.7±0.1 | 1.8±0.1* |
| Liver A1 receptor | 1.0±0.1 | 1.0±0.2 | 1.1±0.1 | 0.8±0.1 | 1.0±0.1 | 1.0±0.1 |
| Liver A2A receptor | 1.0±0.2 | 1.2±0.1 | 0.9±0.1 | 0.7±0.1 | 0.8±0.1 | 0.8±0.1 |
| Liver A2B receptor | 1.0±0.2 | 1.0±0.3 | 0.7±0.1 | 0.4±0.1* | 0.6±0.1 | 0.6±0.1 |
| Liver A3 receptor | 1.0±0.2 | 0.7±0.1 | 0.9±0.2 | 0.8±0.2 | 1.0±0.2 | 0.8±0.1 |
| Kidney A1 receptor | 1.0±0.03 | 1.0±0.2 | 0.6±0.1§§ | 0.8±0.1* | 0.8±0.1 | 0.7±0.1 |
| Kidney A2A receptor | 1.0±0.1 | 0.9±0.2 | 1.0±0.1 | 0.9±0.2 | 1.2±0.2 | 1.2±0.1 |
| Kidney A2B receptor | 1.0±0.1 | 0.9±0.2 | 1.4±0.2 | 1.3±0.2 | 1.3±0.1 | 1.0±0.2 |
| Kidney A3 receptor | 1.0±0.1 | 0.7±0.2 | 0.8±0.2 | 0.7±0.1 | 1.2±0.2 | 0.9±0.3 |
Con, control; Cir, liver cirrhosis; Fur, furosemide; SLV, SLV329, BP, blood pressure.
Results of adenosine receptors from Western blots are expressed as fold expression vs. Con.
Data are means ± standard error of the mean. n = 8–14 per group.
*p<0.05,
**p<0.01,
***p>0.001 vs. Con group with same treatment,
p<0.05,
p<0.01 vs. same group without Fur or SLV treatment,
p<0.05 vs. same group with Fur treatment.
Figure 1. Representative immunoblots for hepatic and renal adenosine A1, A2A, A2B and A3 receptors.
Beta-actin was used for normalization before statistical analysis. Con, control; Cir, liver cirrhosis; Fur, furosemide; SLV, SLV329; kDa, kilodalton.
Table 4. Effects of treatment with furosemide and SLV329 on plasma and urine parameters in rats with and without liver cirrhosis.
| Group | Con | Con+Fur | Con+SLV | Cir | Cir+Fur | Cir+SLV |
| ALT week 8 (U/l) | 26±3 | 36±5 | 29±4 | 41±6 | 27±3* § | 31±4 |
| ALT week 16 (U/l) | 55±4 | 48±2 | 47±4 | 73±9 | 66±11 | 53±7 |
| Bilirubin week 8 (µmol/l) | 1.3±0.1 | 1.5±0.3 | 1.4±0.2 | 7.3±0.7*** | 6.0±0.8*** | 5.7±0.9*** |
| Bilirubin week 16 (µmol/l) | 1.2±0.3 | 1.5±0.4 | 1.3±0.3 | 16±1.8*** | 22±2.9*** | 12±2.0*** # |
| Albumin week 8 (g/l) | 31±0.4 | 32±0.4 | 31±0.2# | 31±0.5 | 32±0.5 | 31±0.4 |
| Albumin week 16 (g/l) | 29±0.5 | 29±0.7 | 28±0.3# | 26±0.6** | 25±1.0* | 26±0.5*** |
| Creatinine week 8 (µmol/l) | 52±2 | 54±3 | 54±3 | 55±2 | 52±2 | 49±1§ |
| Creatinine week 16 (µmol/l) | 60±1 | 60±2 | 55±1§§ # | 51±2* | 47±2** | 45±2** § |
| Urine volume week 8 (ml/d) | 51±6 | 59±4 | 52±8 | 58±3 | 61±3 | 65±5 |
| Urine volume week 16 (ml/d) | 71±8 | 75±3 | 67±9 | 67±8 | 58±6 | 69±7 |
| Na+ excretion week 8 (mmol/d) | 3.2±0.5 | 3.1±0.2 | 2.9±0.5 | 3.6±0.5 | 3.7±0.4 | 2.9±0.3 |
| Na+ excretion week 16 (mmol/d) | 3.7±0.6 | 5.5±0.5 | 4.4±0.6 | 4.7±0.6 | 3.8±0.9 | 4.1±0.3 |
| K+ excretion week 8 (mmol/d) | 4.4±0.6 | 5.0±0.5 | 4.3±0.5 | 4.4±0.5 | 4.8±0.4 | 4.3±0.4 |
| K+ excretion week 16 (mmol/d) | 7.6±0.6 | 7.7±0.8 | 6.7±0.7 | 6.2±0.6 | 5.0±0.7 | 7.0±0.4 |
Con, control; Cir, liver cirrhosis; Fur, furosemide; SLV, SLV329.
In week 0, there were no significant differences between the groups, except for plasma creatinine (Con+Fur 46±6 vs. Cir+Fur 53±5 µmol/l; p<0.05) and urine volume (Con+Fur 70±16 vs. Cir+Fur 58±6 ml/d; p<0.05).
Data are means ± standard error of the mean. n = 8–14 per group.
*p<0.05,
**p<0.01,
***p>0.001 vs. Con group with same treatment,
p<0.05,
p<0.01 vs. same group without Fur or SLV treatment,
p<0.05 vs. same group with Fur treatment.
Livers of the thioacetamide-treated rats showed macroscopic signs of nodular cirrhosis and tended to be heavier than livers from control groups. While liver fibrosis was seen in thioacetamide-treated rats (p<0.001), kidney histology showed no relevant alterations in any of the groups. During the development of liver cirrhosis bilirubin rose markedly, whereas there was a significant decrease of plasma albumin. There was no relevant amount of ascites in any of the cirrhotic groups.
Bodyweight (p<0.001) and water intake (p<0.01) was lower in rats treated with thioacetamide as compared to control animals. Furosemide and SLV329 caused an increase of mean water intake (probably reflecting increased diuresis), particularly at the beginning of the treatment (e.g. week 11, p<0.05; Table 3).
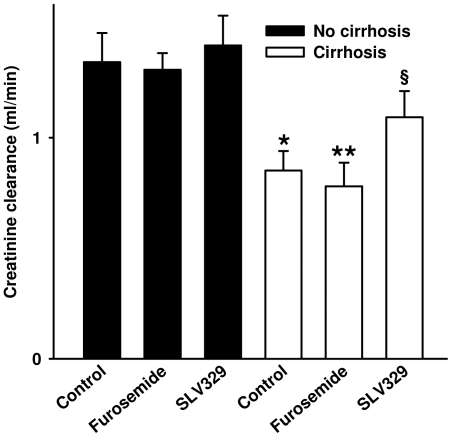
Plasma creatinine was reduced in all cirrhotic animals, most probably due to reduced muscle mass because of wasting. Plasma creatinine was significantly lower in the SLV329-treated groups, in spite of higher mean body weight. The creatinine clearance was significantly reduced in cirrhotic animals compared to controls (−36,5%, p<0.05; Figure 2). This was even slightly more pronounced in the group treated with furosemide (−41.9%, p<0.01 compared to controls). In contrast, the creatinine clearance of the cirrhotic group treated with SLV329 did not differ significantly from any of the non-cirrhotic groups.
Figure 2. Creatinine clearance (week 16) in rats with and without liver cirrhosis.
There were no significant differences between the groups in week 0 and week 8. In week 16 creatinine clearance of the cirrhotic SLV329 group was not significantly different from any of the non-cirrhotic groups. Data are means ± standard error of the mean. n = 8–10 per group (week 16). *p = 0.05, **p<0.01 vs. non-cirrhotic control rats. §p = 0.07 vs. cirrhotic animals with furosemide treatment.
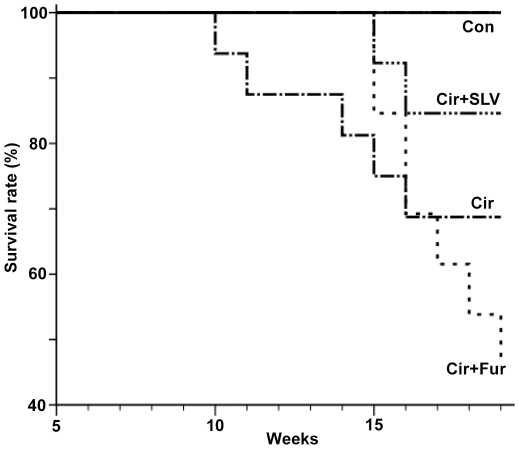
The reduction of mortality in cirrhotic animals treated with SLV329 compared to untreated cirrhotic animals did not reach statistical significance. However, mortality was significantly lower compared to cirrhotic animals treated with furosemide (17% vs. 54%, p<0.05; Figure 3).
Figure 3. Kaplan-Meier mortality chart of rats with liver cirrhosis (Cir; mortality 5/14), cirrhotic rats with furosemide treatment (Cir+Fur; 7/13), cirrhotic rats with SLV329 treatment (Cir+SLV; 2/12) and all control groups without liver cirrhosis (Con; 0/24).
p<0.05 for Cir+Fur vs. Cir+SLV (log-rank test).
In the Western blot analyses hepatic A2R seemed to be reduced in cirrhotic rats. This difference was statistically significant for A2AR and A2BR when testing all cirrhotic rats vs. all controls (p<0.01). Treatment with SLV329 reduced A1R expression in renal tissue of non-cirrhotic animals (p<0.01).
Mean plasma concentrations of SLV329 were 49±8 µg/l on day 10 after the beginning of SLV329 treatment and 54±13 µg/l after 4 weeks in 6 random animals.
Heart rate, lipase, creatine kinase and urinary protein excretion were not different between the groups at any time (data not shown).
Discussion
The present study was set out to investigate the receptor binding affinity of the novel A1R antagonist SLV329, to evaluate its in vivo effects on diuresis and natriuresis in healthy animals and to find out whether it exerts beneficial effects in an animal model of thioacetamide-induced liver cirrhosis.
Receptor binding experiments showed that SLV329 behaves as a potent and selective A1R ligand in vitro.
Intravenous treatment of healthy rats with SLV329 resulted in a strong dose-dependent diuretic and natriuretic effect, whereas the effect on kaliuresis was relatively small and the creatinine clearance remained unchanged. This is in line with effects that have been reported in a variety of rat strains including Wistar rats [11]–[13].
The ability of diuretics to prevent sodium reabsorption results in an increased delivery of electrolytes to the distal tubule. This leads, in turn, to an augmented release of adenosine, which may activate A1R in afferent arterioles [14]. By blocking those receptors, A1R antagonists cause an uncoupling of the tubuloglomerular feedback which may at least partly explain the elevated concentrations of adenosine measured in the urine of animals treated with SLV329 [15], [16]. However, urinary excretion of paracrine mediators do not necessarily reflect their local tissue concentrations. Alternatively, blockade of A1R might result in elevated intracellular cyclic adenosine monophosphate levels and release in the kidney, which will eventually lead to increased extracellular adenosine concentrations due to cyclic adenosine monophosphate degradation [17]–[19]. However, in spite of the observed increase in urinary adenosine excretion, SLV329 did not decrease the rate of creatinine clearance, even at the highest dose.
Previous studies demonstrated that a single application of an A1R antagonist causes an increase of renal sodium and water excretion in animals and patients with liver cirrhosis without affecting the glomerular filtration rate [20], [21]. As a next step towards a possible clinical application, the present study investigated for the first time the effects of a chronic application of a selective A1R antagonist on kidney function and mortality starting at an early stage of liver cirrhosis. This proof-of-concept experiment included a low-dose furosemide-treated group because loop diuretics are often applied in cirrhotic patients and tend to deteriorate renal function. However, monotherapy with a loop diuretic is of course not the typical clinical situation at an early stage of liver cirrhosis without severe water retention. In the present study the creatinine clearance, used as a surrogate for the glomerular filtration rate, was significantly reduced in cirrhotic animals, especially in those receiving furosemide. In contrast, the A1R antagonist SLV329 was able to prevent this decline of creatinine clearance. The reduction of mortality in cirrhotic animals treated with SLV329 in comparison to vehicle treatment was not statistically significant. However, mortality was significantly lower in cirrhotic animals treated with SLV329 in comparison to animals treated with furosemide (17% vs. 54%). It is a limitation of this study that creatinine clearance was used instead of inulin clearance. Inulin clearance was not used because mortality would have increased further due to additional anesthesia. Creatinine clearance is influenced by muscle mass, liver function and tubular secretion of creatinine. However, there were no significant differences of neither body weight nor liver function between cirrhotic animals with and without SLV329 treatment. Tubular secretion of creatinine increases with declining glomerular filtration rate. Thus, the glomerular filtration rate of untreated cirrhotic animals and those treated with furosemide will be even lower than the creatinine clearance suggests, making the difference to the group treated with SLV329 even larger.
Mean SLV329 plasma concentrations of 49–54 µg/l were actually reached by chronic application in the cirrhosis model. This plasma concentration was able to cause strong short-term effects on diuresis and saliuresis when administered intravenously as shown in Table 2. A steady-state is usually reached in the course of long-term diuretic therapy by means of compensatory mechanisms of tubular reabsorption [22]. This explains why the 24-hour urine volume and electrolyte excretion in week 16 is not different between the groups of the cirrhosis model. However, when looking at mean water intake over 8 weeks, as a putative surrogate of diuresis, the effects of furosemide and SLV329 can be detected. It would have been interesting to evaluate fractional sodium excretion and free water clearance. This was not done due to limited plasma quantities.
SLV329 does not affect the expression of hepatic or renal adenosine receptors in cirrhotic animals. The expression of A2R seems to be reduced in cirrhotic animals, independent of the treatment group. As yet, there are no reports on the expression of A2R in cirrhotic liver tissue. However, it is known, that hepatic A2R play an active role in the pathogenesis of hepatic fibrosis [23]. It is a limitation of this study that adenosine receptor expression was evaluated only in liver and kidney homogenates by Western blot. Immunohistochemistry or radioligand binding experiments might give further detailed information.
The beneficial effects of SLV329 in the cirrhosis model cannot be explained by morphological effects since SLV329 did not relevantly influence the degree of liver fibrosis, kidney histology or expression of hepatic or renal adenosine receptors. Animal studies suggest that liver cirrhosis activates the hepatorenal reflex via A1R, leading to renal water and sodium retention [24], [25]. Animal and human studies suggest that a resetting of the tubuloglomerular feedback contributes to the pathophysiology of kidney impairment in liver cirrhosis [26]. Thus, inhibition of both the hepatorenal reflex and the tubuloglomerular feedback might explain the higher rate of creatinine clearance in the animals treated with SLV329. In addition to the effect on creatinine clearance, yet unknown effects of SLV329 might contribute to the reduction of mortality. As an addition to the present study it would be interesting to study liver cirrhosis in the established murine A1R knockout model.
The ability of A1R antagonists to induce diuresis and natriuresis while not compromising glomerular filtration rate has become an attractive therapeutic option for the treatment of other fluid retention disorders, e.g. in kidney disease and heart failure, especially in conditions associated with diuretic resistance [27]. The preexisting experience with this class of drugs for other indications (including a large phase 3 trial) might facilitate the future translation of the results of this study to clinical application [28].
In conclusion, this study described some pharmacodynamic characteristics of the novel adenosine A1R-specific antagonist SLV329 and demonstrated its long-term safety and efficacy in an animal model of liver cirrhosis. Chronic SLV329 treatment starting at an early stage of liver cirrhosis prevented the decrease of creatinine clearance. Further studies will have to evaluate, whether SLV329 or other A1R antagonists are clinically beneficial at different stages of liver cirrhosis, either as an add-on to aldosterone antagonists or in combination with loop diuretics.
Materials and Methods
Receptor binding and enzyme assays
Receptor binding affinities as well as enzyme inhibitory properties of the new compound SLV329 were evaluated in a series of 94 receptors and 6 phosphodiesterases as described previously [29]. All cells and tissues needed for the assays were provided by Cerep (Celle l'Evescault, France).
Receptor binding assays were conducted as follows: after incubation of SLV329 with a receptor preparation and its radioactive ligand, the receptor preparations were rapidly filtered under vacuum through glass fiber filters, the filters were washed extensively with an ice-cold buffer using a harvester. Bound radioactivity was measured by scintillation counting using a liquid scintillation cocktail. Enzyme assays were carried out as follows: after incubation of SLV329 with an enzyme preparation and its radioactive substrate, radioactivity of the enzyme product was measured by scintillation counting using a liquid scintillation cocktail. Testing was done at a 3-log concentration range around a predetermined half-maximally inhibitory concentration (IC50) for the respective assay. The highest concentration tested for primes was 10 µM in receptor binding and 100 µM for enzyme assays. If no significant receptor binding or enzyme inhibition was detected at those concentrations SLV329 was considered to be inactive. Results were calculated as percentage of control values (enzyme assays) or for receptor binding assays as percentage of total ligand binding and that of nonspecific binding per concentration of SLV329. From the concentration-displacement curves IC50 values were determined by nonlinear regression analysis using Hill equation curve fitting. The inhibition constants (Ki) were calculated from the Cheng-Prusoff equation Ki = IC50/(1+L/Kd), where L is the concentration of radioligand in the assay and Kd the affinity of the radioligand for the receptor. Results were expressed as mean pKi values ± standard deviation (SD) of at least three separate experiments.
Effects of SLV329 on renal function in healthy rats
All animal experiments of this study were conducted in strict accordance with the European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes (ETS123) and the German law on animal welfare and all efforts were made to minimize suffering. The study protocol was approved by the Niedersächsisches Landesamt für Verbraucherschutz und Lebensmittelsicherheit (Bezirksregierung Hannover, approval number 509.6.42502-04/875).
Male Sprague-Dawley Crl:CD(SD)BR rats (150–170 g) were fasted overnight. The rats were anesthetized with 80 mg/kg thiobutabarbital intraperitoneally; additional doses of 40 mg/kg were given 2.5 h and 5 hours later. Catheters were placed in one jugular vein (for SLV329 or vehicle administration), one carotid artery (for blood sampling and blood pressure measurements), and the bladder. The rats were kept on a heated table to maintain body temperature at 37°C. After an equilibration period of 30 min, urine was sampled for a period of 3 h, then the animals received vehicle or SLV329 as follows: a slowly applied (30–60 s) loading bolus of 18, 54, 180, 540, and 945 µg/kg of SLV329 (in 0.45 ml/kg), followed by a continuous intravenous infusion at a rate of 0.37, 1.1, 3.7, 11, and 19.3 µg/kg SLV329 per minute (in 9.3 µl/kg per minute) for three hours. Urine and blood samples were collected 2 min before the SLV329 bolus and after three hours of SLV329 infusion. 5-sulfosalicylic acid was added to the urine aliquots for adenosine measurements at a final concentration of 8 g/l before freezing. Electrolytes and creatinine in plasma and urine were measured by standard automated analyzer by Medizinisches Labor Hannover (Hannover, Germany). Urine adenosine concentrations were quantified by high-pressure liquid chromatography using a MZ nucleosil C18 column (125×4 mm, 10 µm) with ultraviolet-detection (Immundiagnostik, Bensheim, Germany). Quantification of SLV329 plasma concentrations was performed after solid phase extraction using a validated reversed phase high-pressure liquid chromatography method with MS/MS-detection (Sciex Api 3000, Perkin Elmer, Waltham, MA, USA). The data describing the concentration-dependence of the diuretic and natriuretic effects of SLV329 were fitted to estimate the half-maximally effective concentration (EC50) using Prism 5.00 (GraphPad Software Inc., San Diego, CA, USA).
Effects of SLV329 in thioacetamide-induced liver cirrhosis
The study protocol was approved by the Landesamt für Gesundheit und Soziales, Berlin (approval number G0163/06). Male Wistar rats (250–300 g) were maintained under controlled conditions (20±2°C, 12 h light/dark cycle) and kept on a standard diet (0.2% sodium) with water ad libitum.
Animals were divided into 6 groups:
Controls (Con; n = 8)
Controls with furosemide treatment (Con+Fur; n = 8)
Controls with SLV329 treatment (Con+SLV; n = 8)
Cirrhosis (Cir; n = 14)
Cirrhosis with furosemide treatment (Cir+Fur; n = 13)
Cirrhosis with SLV329 treatment (Cir+SLV; n = 12)
Liver cirrhosis was induced by oral administration of thioacetamide via drinking water for 18 weeks. The initial concentration was 0.03%. This concentration was modified weekly according to weight changes in response to thioacetamide. The concentration was increased/reduced by 0.015% (absolute) in case of weight gain/loss >25 g per week. Treatment with furosemide or SLV329 was started in week 8, since it is known that liver cirrhosis develops by then [30]. Thioacetamide concentrations remained unchanged from week 8 until week 12. In week 12 the concentration was increased by 0.015% in all animals receiving thioacetamide. The resulting concentration was given until week 18. Furosemide was injected intraperitoneally (7.5 mg/kg) thrice weekly (always between 8 and 10 a.m.) in the respective groups starting from week 8 until study end. Also starting from week 8 the respective groups received a standard rat chow formulated with SLV329. Chow was obtained from Altromin (Lage, Germany) in several concentrations (0.0075%, 0.014%, and 0.05% SLV329) and fed to the rats according to food intake. The target concentration was 5 mg/kg per day. Subsequently calculated mean intakes of the groups were Con+SLV 5.4 mg/kg*d and Cir+SLV 5.1 mg/kg*d. Blood was taken from the retro-orbital vein plexus 10 days and 4 weeks after the beginning of SLV329 treatment in 6 random animals to measure the plasma concentration of SLV329. The samples were analyzed as described above.
Body weight, water and food intake were measured weekly. Systolic blood pressure and heart rate were measured in week 0 and 12 by tail plethysmography as described previously.[31] All animals were placed in metabolism cages in week 0, 8 and 16. Blood was taken from the retro-orbital vein plexus. The animals were sacrificed after week 20 and liver and kidneys were excised. Kidney samples were embedded in paraffin, cut into 3 µm sections and submitted to periodic acid-Schiff, elastica and sirius red staining. The extent of glomerulosclerosis, the renal media/lumen ratio and renal interstitial fibrosis was determined as described previously [32]. The extent of liver fibrosis was analyzed as in the kidneys.
Alanine aminotransferase, bilirubin, albumin, creatinine, lipase and creatine kinase were measured in plasma using commercially available assays (ABX Pentra 400, Horiba Medical, Montpellier, France). Creatinine, sodium, potassium and protein were measured in urine also using ABX Pentra 400.
Western blot
Western blot was performed in liver and kidney tissue for A1, A2A, A2B and A3 receptors using a method based on a previous publication [33]. In brief, snap frozen liver and kidney samples were pulverized in liquid nitrogen and dissolved in lysis buffer. After incubation at room temperature for 10 min, the suspension was centrifuged (16.200 g, 20°C, 45 min). Protein concentrations were determined in the supernatant using the Bradford method. Supernatant and Bradford solution (40 µl each) were mixed in a well of a microtiter plate and incubated for 10 min at room temperature on a shaker. Extinction was then measured photometrically at 595 nm. The protein concentration of each sample was calculated according to a standard dilution series. The samples were diluted with lysis buffer (5 g/l) to assure equal loading. Samples (23 µg protein per lane) were separated by sodium dodecylsulfate polyacrylamide gel electrophoresis (10%, 80 V for 30 min, then 110 V) and semi-dry-blotted (1,5 mA/cm2, 60 min) onto nitrocellulose membranes. Ponceau staining of membranes confirmed equal loading of proteins. Membranes were blocked with 5% skim milk and incubated overnight with primary rabbit antibodies directed against A1R (0.025%, Sigma-Aldrich, Munich, Germany), A2AR (0.1%, Millipore, Schwalbach, Germany), A2BR (0.1%, Millipore), A3R (0.2%, Millipore), and beta-actin for normalization (0.0025%, Sigma-Aldrich). The specificity of the antibodies has been documented by the manufacturers and was not tested again in this study. After extensive washing blots were incubated with a horseradish peroxidase-linked anti-rabbit IgG (60 min, 0.0001%, Santa Cruz Biotechnology, Santa Cruz, CA, USA). Immunoreactive bands were detected using an enhanced chemiluminescence system and were subsequently quantified with the AlphaEaseFC software (Alpha Innotech, San Leandro, CA, USA). The values thus obtained were corrected for different conditions between single runs, using a standard dilution series that was run on each gel. Results of adenosine receptors were then normalized to beta-actin.
Statistical analysis
Data was analyzed with SPSS 17.0 (SSPS Inc., Chicago, IL, USA). The nonparametric Kruskal-Wallis and the Mann-Whitney-U test were used to detect significant differences between groups of interest. Mortality rates were estimated by the Kaplan-Meier method and compared by log-rank test.
Footnotes
Competing Interests: The authors have read the journal's policy and have the following conflicts: Berthold Hocher was an employee of Solvay Pharmaceuticals at the time of the study. Dieter Ziegler and Yvan Fischer are employees of Abbott Products GmbH. Solvay Pharmaceuticals (now Abbott Products GmbH) financially supported the study. Abbott Products GmbH holds the patent of SLV329 and is developing this compound. This does not alter the authors' adherence to all the PLoS ONE policies on sharing data and materials, as detailed online in the guide for authors.
Funding: This study was partially supported by a grant of the Dr. Werner Jackstädt-Stiftung to Markus Alter. This funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. This study was also supported by Solvay Pharmaceuticals (now Abbott Products GmbH). Dieter Ziegler and Yvan Fischer are employees of Abbott Products GmbH and played a role in study design, data collection and analysis, decision to publish and preparation of the manuscript. The study design was set up together with Solvay Pharmaceuticals. Solvay Pharmaceuticals agreed to publish the manuscript.
References
- 1.Gines P, Schrier RW. Renal failure in cirrhosis. N Engl J Med. 2009;361:1279–1290. doi: 10.1056/NEJMra0809139. [DOI] [PubMed] [Google Scholar]
- 2.Linden J. Molecular approach to adenosine receptors: receptor-mediated mechanisms of tissue protection. Annu Rev Pharmacol Toxicol. 2001;41:775–787. doi: 10.1146/annurev.pharmtox.41.1.775. [DOI] [PubMed] [Google Scholar]
- 3.Ming Z, Smyth DD, Lautt WW. Decreases in portal flow trigger a hepatorenal reflex to inhibit renal sodium and water excretion in rats: role of adenosine. Hepatology. 2002;35:167–175. doi: 10.1053/jhep.2002.30425. [DOI] [PubMed] [Google Scholar]
- 4.Ming Z, Smyth DD, Lautt WW. Decreases in portal flow trigger a hepatorenal reflex to inhibit renal sodium and water excretion in rats: role of adenosine. Hepatology. 2002;35:167–175. doi: 10.1053/jhep.2002.30425. [DOI] [PubMed] [Google Scholar]
- 5.Ming Z, Lautt WW. Intrahepatic adenosine-mediated activation of hepatorenal reflex is via A1 receptors in rats. Can J Physiol Pharmacol. 2006;84:1177–1184. doi: 10.1139/y06-063. [DOI] [PubMed] [Google Scholar]
- 6.Jackson EK, Zhu C, Tofovic SP. Expression of adenosine receptors in the preglomerular microcirculation. Am J Physiol Renal Physiol. 2002;283:F41–F51. doi: 10.1152/ajprenal.00232.2001. [DOI] [PubMed] [Google Scholar]
- 7.Wilcox CS, Welch WJ, Schreiner GF, Belardinelli L. Natriuretic and diuretic actions of a highly selective adenosine A1 receptor antagonist. J Am Soc Nephrol. 1999;10:714–720. doi: 10.1681/ASN.V104714. [DOI] [PubMed] [Google Scholar]
- 8.Vallon V, Muhlbauer B, Osswald H. Adenosine and kidney function. Physiol Rev. 2006;86:901–940. doi: 10.1152/physrev.00031.2005. [DOI] [PubMed] [Google Scholar]
- 9.Gottlieb SS, Brater DC, Thomas I, Havranek E, Bourge R, et al. BG9719 (CVT-124), an A1 adenosine receptor antagonist, protects against the decline in renal function observed with diuretic therapy. Circulation. 2002;105:1348–1353. doi: 10.1161/hc1102.105264. [DOI] [PubMed] [Google Scholar]
- 10.Ming Z, Fan YJ, Yang X, Lautt WW. Blockade of intrahepatic adenosine receptors improves urine excretion in cirrhotic rats induced by thioacetamide. J Hepatol. 2005;42:680–686. doi: 10.1016/j.jhep.2004.12.023. [DOI] [PubMed] [Google Scholar]
- 11.Miracle CM, Rieg T, Blantz RC, Vallon V, Thomson SC. Combined effects of carbonic anhydrase inhibitor and adenosine A1 receptor antagonist on hemodynamic and tubular function in the kidney. Kidney Blood Press Res. 2007;30:388–399. doi: 10.1159/000108625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wilcox CS, Welch WJ, Schreiner GF, Belardinelli L. Natriuretic and diuretic actions of a highly selective adenosine A1 receptor antagonist. J Am Soc Nephrol. 1999;10:714–720. doi: 10.1681/ASN.V104714. [DOI] [PubMed] [Google Scholar]
- 13.Knight RJ, Bowmer CJ, Yates MS. The diuretic action of 8-cyclopentyl-1,3-dipropylxanthine, a selective A1 adenosine receptor antagonist. Br J Pharmacol. 1993;109:271–277. doi: 10.1111/j.1476-5381.1993.tb13564.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vallon V, Muhlbauer B, Osswald H. Adenosine and kidney function. Physiol Rev. 2006;86:901–940. doi: 10.1152/physrev.00031.2005. [DOI] [PubMed] [Google Scholar]
- 15.Wilcox CS, Welch WJ, Schreiner GF, Belardinelli L. Natriuretic and diuretic actions of a highly selective adenosine A1 receptor antagonist. J Am Soc Nephrol. 1999;10:714–720. doi: 10.1681/ASN.V104714. [DOI] [PubMed] [Google Scholar]
- 16.Vallon V, Muhlbauer B, Osswald H. Adenosine and kidney function. Physiol Rev. 2006;86:901–940. doi: 10.1152/physrev.00031.2005. [DOI] [PubMed] [Google Scholar]
- 17.Jackson EK, Mi Z, Dubey RK. The extracellular cAMP-adenosine pathway significantly contributes to the in vivo production of adenosine. J Pharmacol Exp Ther. 2007;320:117–123. doi: 10.1124/jpet.106.112748. [DOI] [PubMed] [Google Scholar]
- 18.Stanley AJ, Forrest EH, Dabos K, Bouchier IA, Hayes PC. Natriuretic effect of an adenosine-1 receptor antagonist in cirrhotic patients with ascites. Gastroenterology. 1998;115:406–411. doi: 10.1016/s0016-5085(98)70207-0. [DOI] [PubMed] [Google Scholar]
- 19.Toya Y, Umemura S, Iwamoto T, Hirawa N, Kihara M, et al. Identification and characterization of adenosine A1 receptor-cAMP system in human glomeruli. Kidney Int. 1993;43:928–932. doi: 10.1038/ki.1993.130. [DOI] [PubMed] [Google Scholar]
- 20.Ming Z, Fan YJ, Yang X, Lautt WW. Blockade of intrahepatic adenosine receptors improves urine excretion in cirrhotic rats induced by thioacetamide. J Hepatol. 2005;42:680–686. doi: 10.1016/j.jhep.2004.12.023. [DOI] [PubMed] [Google Scholar]
- 21.Stanley AJ, Forrest EH, Dabos K, Bouchier IA, Hayes PC. Natriuretic effect of an adenosine-1 receptor antagonist in cirrhotic patients with ascites. Gastroenterology. 1998;115:406–411. doi: 10.1016/s0016-5085(98)70207-0. [DOI] [PubMed] [Google Scholar]
- 22.Reyes AJ. Renal excretory profiles of loop diuretics: consequences for therapeutic application. J Cardiovasc Pharmacol. 1993;22(Suppl 3):S11–S23. doi: 10.1097/00005344-199322003-00003. [DOI] [PubMed] [Google Scholar]
- 23.Chan ES, Montesinos MC, Fernandez P, Desai A, Delano DL, et al. Adenosine A(2A) receptors play a role in the pathogenesis of hepatic cirrhosis. Br J Pharmacol. 2006;148:1144–1155. doi: 10.1038/sj.bjp.0706812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ming Z, Smyth DD, Lautt WW. Decreases in portal flow trigger a hepatorenal reflex to inhibit renal sodium and water excretion in rats: role of adenosine. Hepatology. 2002;35:167–175. doi: 10.1053/jhep.2002.30425. [DOI] [PubMed] [Google Scholar]
- 25.Ming Z, Lautt WW. Intrahepatic adenosine-mediated activation of hepatorenal reflex is via A1 receptors in rats. Can J Physiol Pharmacol. 2006;84:1177–1184. doi: 10.1139/y06-063. [DOI] [PubMed] [Google Scholar]
- 26.Sansoe G, Silvano S, Mengozzi G, Smedile A, Touscoz G, et al. Loss of tubuloglomerular feedback in decompensated liver cirrhosis: physiopathological implications. Dig Dis Sci. 2005;50:955–963. doi: 10.1007/s10620-005-2671-0. [DOI] [PubMed] [Google Scholar]
- 27.Hocher B. Adenosine A1 receptor antagonists in clinical research and development. Kidney Int. 2010;78:438–445. doi: 10.1038/ki.2010.204. [DOI] [PubMed] [Google Scholar]
- 28.Hocher B. Adenosine A1 receptor antagonists in clinical research and development. Kidney Int. 2010;78:438–445. doi: 10.1038/ki.2010.204. [DOI] [PubMed] [Google Scholar]
- 29.Kalk P, Eggert B, Relle K, Godes M, Heiden S, et al. The adenosine A1 receptor antagonist SLV320 reduces myocardial fibrosis in rats with 5/6 nephrectomy without affecting blood pressure. Br J Pharmacol. 2007;151:1025–1032. doi: 10.1038/sj.bjp.0707319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ikejima K, Takei Y, Honda H, Hirose M, Yoshikawa M, et al. Leptin receptor-mediated signaling regulates hepatic fibrogenesis and remodeling of extracellular matrix in the rat. Gastroenterology. 2002;122:1399–1410. doi: 10.1053/gast.2002.32995. [DOI] [PubMed] [Google Scholar]
- 31.Quaschning T, Voss F, Relle K, Kalk P, Vignon-Zellweger N, et al. Lack of endothelial nitric oxide synthase promotes endothelin-induced hypertension: lessons from endothelin-1 transgenic/endothelial nitric oxide synthase knockout mice. J Am Soc Nephrol. 2007;18:730–740. doi: 10.1681/ASN.2006050541. [DOI] [PubMed] [Google Scholar]
- 32.Pfab T, Thone-Reineke C, Theilig F, Lange I, Witt H, et al. Diabetic endothelin B receptor-deficient rats develop severe hypertension and progressive renal failure. J Am Soc Nephrol. 2006;17:1082–1089. doi: 10.1681/ASN.2005080833. [DOI] [PubMed] [Google Scholar]
- 33.Jackson EK, Zhu C, Tofovic SP. Expression of adenosine receptors in the preglomerular microcirculation. Am J Physiol Renal Physiol. 2002;283:F41–F51. doi: 10.1152/ajprenal.00232.2001. [DOI] [PubMed] [Google Scholar]