Abstract
Serotonin 2C receptor (5-HT2CR) agonists administered systemically attenuate both cocaine-primed and cue-elicited reinstatement of extinguished cocaine-seeking behavior. To further elucidate the function of these receptors in addiction-like processes, this study examined the effects of microinfusing the 5-HT2CR agonist MK212 (0, 10, 30, 100 ng/side/0.2 μl) into the medial prefrontal cortex (mPFC) on cocaine self-administration and reinstatement of extinguished cocaine-seeking behavior. Male Sprague–Dawley rats were trained to self-administer cocaine (0.75 mg/kg, i.v.) paired with light and tone cues. Once responding stabilized, rats received MK212 microinfusions before tests for maintenance of cocaine self-administration. Next, extinction training to reduce cocaine-seeking behavior, defined as responses performed without cocaine reinforcement available, occurred until low extinction baselines were achieved. Rats then received MK212 microinfusions before tests for reinstatement of extinguished cocaine-seeking behavior elicited by cocaine-priming injections (10 mg/kg, i.p.) or response-contingent presentations of the cocaine-associated cues; operant responses during cocaine-primed reinstatement tests produced no consequences. MK212 microinfusions into the prelimbic and infralimbic, but not anterior cingulate, regions of the mPFC dose-dependently attenuated both cocaine-primed and cue-elicited reinstatement of extinguished cocaine-seeking behavior, but did not reliably affect cocaine self-administration. A subsequent experiment showed that the effects of MK212 (100 ng/side/0.2 μl) on reinstatement of extinguished cocaine-seeking behavior were blocked by co-administration of the 5-HT2CR antagonist SB242084 (200 ng/side/0.2 μl). MK212 administered alone into the mPFC as a drug prime produced no discernable effects on cocaine-seeking behavior. These findings suggest that stimulation of 5-HT2CRs in the mPFC attenuates the incentive motivational effects produced by sampling cocaine or exposure to drug-paired cues.
Keywords: reinstatement, serotonin, motivation, relapse, craving, addiction
INTRODUCTION
A major difficulty in treating cocaine dependence is the high incidence of relapse (O'Brien, 2005; Wallace, 1992), which may occur even after prolonged periods of abstinence. Factors contributing to relapse include incentive motivational effects produced by either sampling cocaine or exposure to drug-related cues (Davis and Smith, 1976; de Wit and Stewart, 1981). Incentive motivation for cocaine is measured in animals using the extinction/reinstatement model (de Wit and Stewart, 1981), whereby animals are initially trained to perform an operant response reinforced with cocaine, followed by extinction training during which responses produce no consequences. Responding in the absence of drug reinforcement is referred to as cocaine-seeking behavior and provides a measure of incentive motivation for cocaine. Measuring reinstatement of extinguished cocaine-seeking behavior elicited by cocaine or cocaine-associated cues provides an animal model to study the mechanisms involved in the incentive motivational effects of these stimuli (Epstein et al, 2006).
Serotonin (5-HT) systems are critical in mediating the incentive motivational effects of cocaine and cocaine-paired cues in both animals and humans (Bubar and Cunningham, 2008; Hyman et al, 2006; Rothman et al, 2008). In humans, acute 5-HT depletion reduces self-reports of craving elicited by cocaine-associated cues (Satel et al, 1995) and reduces euphoric effects of intranasal cocaine (Aronson et al, 1995). However, increasing 5-HT with the reuptake inhibitor fluoxetine also reduces cocaine-induced positive subjective effects and craving (Batki et al, 1993; Walsh et al, 1994), although others report an increase in ‘likely to use cocaine' (Harris et al, 2004). In rats, 5-HT depletion or elevation with indirect agonists attenuates reinstatement of cocaine-seeking behavior after exposure to cocaine-paired cues (Tran-Nguyen et al, 1999, 2001). In contrast, indirect agonists fail to reliably alter effects of cocaine priming (Baker et al, 2001; Burmeister et al, 2003).
The inconsistency of effects elictied by cocaine priming is likely related to the complexity of 5-HT systems. There are 14 different 5-HT receptor (5-HTR) subtypes that have diverse effects (Barnes and Sharp, 1999) and are differentially regulated (Cunningham et al, 1992; Neumaier et al, 2002; Rocha et al, 1993) and/or affected functionally (Baumann and Rothman, 1995, 1996, 1998; Darmani et al, 1992; King et al, 1993; Lee and Meltzer, 1994; Levy et al, 1992, 1994) by chronic cocaine, other indirect agonists, or 5-HT depletion. Moreover, 5-HT may produce similar or opposite effects through stimulation of different 5-HTRs. For example, actions at 5-HT2A and 5-HT2CRs have opposite effects on cocaine-seeking behavior. Agonist stimulation of 5-HT2CRs decreases cue- and cocaine-primed reinstatement of extinguished cocaine-seeking behavior (Burbassi and Cervo, 2008; Fletcher et al, 2008; Grottick et al, 2000; Neisewander and Acosta, 2007), whereas blocking 5-HT2ARs with selective antagonists decreases cue- (Burmeister et al, 2004; Filip, 2005; Nic Dhonnchadha et al, 2009) and cocaine-primed reinstatement (Filip, 2005; Fletcher et al, 2002a).
Little is known about the neural circuitry underlying the inhibitory effects of 5-HT2CR agonists on reinstatement of extinguished cocaine-seeking behavior. The medial prefrontal cortex (mPFC) is likely involved given that it is densely populated with 5-HT2CR mRNA and protein (Liu et al, 2007; López-Giménez et al, 1997; Pompeiano et al, 1994) and the neural circuitries mediating the incentive motivational effects of cocaine priming vs cues converge in the mPFC (Neisewander et al, 2000; Peters et al, 2009; Robbins and Everitt, 2002; Weiss, 2005). Furthermore, 5-HT2CRs in the mPFC mediate other stimulant-induced behavior. For instance, the 5-HT2CR agonist MK212 microinfused into the mPFC inhibits both cocaine-induced hyperactivity and discriminative-stimulus effects without altering spontaneous locomotion (Filip and Cunningham, 2003) and decreases MDMA-induced behavioral sensitization (Ramos et al, 2005).
The mPFC is anatomically and functionally heterogeneous. The ventromedial (vm) PFC, consisting of the ventral prelimbic (PrL) and infralimbic (IL) cortices, is preferentially connected with the nucleus accumbens shell (NAcsh) and core (NAcc), amygdala, and ventral tegmental area (VTA), whereas the dorsomedial PFC, consisting of the dorsal PrL and anterior cingulate cortices, is predominantly linked with the NAcc (Ding et al, 2001; Gabbott et al, 1997, 2005; McFarland and Kalivas, 2001; Zahm and Brog, 1992). Inactivation of the PrL, but not IL, cortex reduces context-, stress-, cue-, and cocaine-primed reinstatement of extinguished cocaine-seeking behavior (Capriles et al, 2003; Fuchs et al, 2005; McFarland and Kalivas, 2001; McLaughlin and See, 2003; Peters et al, 2009). However, lesions of either the PrL (Zavala et al, 2003) or IL (Tzschentke and Schmidt, 1999) cortices reduce cocaine-conditioned place preference and behavioral sensitization to cocaine. The mPFC also has a function in cocaine reinforcement as animals will self-administer cocaine directly into this region (Goeders and Smith, 1984).
This study investigated whether 5-HT2CRs located within the mPFC inhibit cue- and cocaine-primed reinstatement of extinguished cocaine-seeking behavior by microinfusing MK212 into the PrL and IL cortices before testing. Additional tests examined the behavioral (effects of intra-mPFC MK212 microinfusions on maintenance of cocaine self-administration), pharmacological (effects of co-infusion with the 5-HT2CR antagonist SB242084), and anatomical (effects of anterior cingulate cortex MK212 microinfusions) specificity of the MK212 effects.
MATERIALS AND METHODS
Animals
Male Sprague–Dawley rats (n=77) weighing 275–325 g at the time of surgery were individually housed under a 12-h reversed light/dark cycle. All husbandry and experimentation adhered to the Guide for the Care and Use of Laboratory Animals (1996).
Surgery
Catheters were implanted into jugular veins under isoflurane (2–3%) anesthesia as detailed previously (Pentkowski et al, 2009). Next, each rat was placed into a stereotaxic instrument, the scalp was incised and retracted, and the head was positioned with Bregma and Lambda at the same DV coordinate. Holes were drilled into the skull to bilaterally insert steel guide cannulae (26G, Plastics One, Roanoke, VA) into the mPFC using the following coordinates derived from previous research (Filip and Cunningham, 2003): +2.10 (Cg2) or +2.70 (PrL, IL) mm anterior to bregma, ±0.75 mm from the midline, and −3.00 mm ventral from the surface of the skull (Paxinos and Watson, 2007). The guide cannulae and metal end of the catheter were secured to the skull and anchor screws using dental acrylic. Stylets (30G, Plastics One) were placed into the guide cannulae to maintain patency. Rats were returned to their home cages for 5–6 days recovery. Catheters were flushed daily with 0.1 ml saline containing heparin sodium (70 U/ml; APP Pharmaceuticals, Schaumburg, IL), Abbokinase (20 mg/ml; ImaRx Therapeutics, Tucson, AZ), and Timentin (66.7 mg/ml; GlaxoSmithKline, Research Triangle Park, NC) to maintain patency. Proper catheter function was tested periodically by administering 0.05 ml methohexital sodium (16.7 mg/ml; JHP Pharmaceuticals, Rochester, MI), a dose that produces brief anesthetic effects only when administered i.v.
Drugs
Cocaine hydrochloride (RTI International, Research Triangle Park, NC) dissolved in saline was filtered through 0.2 μm membranes. MK212 and SB242084 (Tocris Cookson, Ellisville, MO) were dissolved in artificial cerebrospinal fluid (aCSF) and adjusted to a pH of 7.4. Injections were administered i.p. at a volume of 1 ml/kg or intracranially at a volume of 0.2 μl/side. Microinfusions were delivered over a 1-min period using bilateral injection cannulae (30G, Plastics One) connected through polyethylene 50 tubing (Becton Dickinson, Sparks, MD) to 25-μl syringes (Hamilton, Reno, NV) mounted in an infusion pump (CMA Microdialysis, North Chelmsford, MA). Injection cannulae extended exactly 1 (Cg2, PrL) or 2 (IL) mm below guide cannulae tips. Movement of an air bubble the correct distance in the injection tubing confirmed accurate infusion volume. Injection cannulae remained in place for 1 min before removal, and then the stylets were replaced.
Experimental Phases
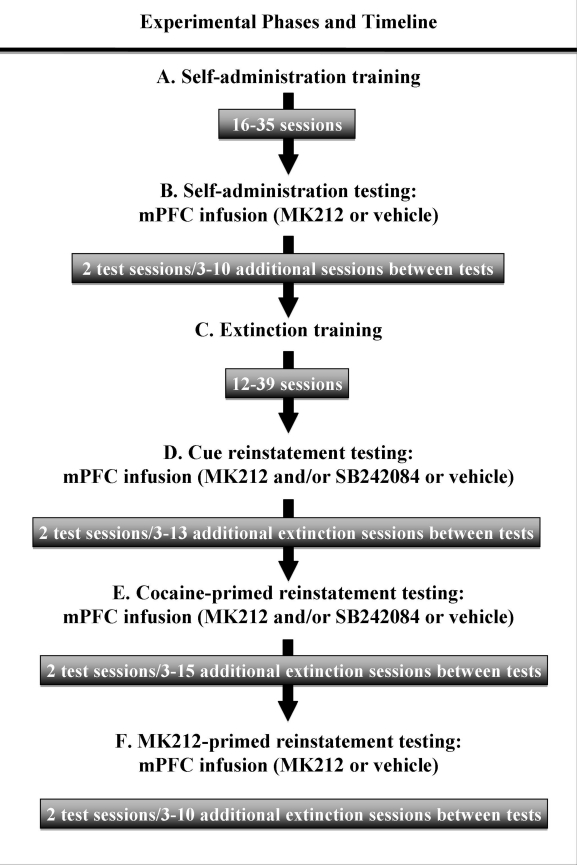
An outline of each experimental phase and a timeline for the first experiment is shown in Figure 1. Each phase is detailed below.
Figure 1.
Timeline depicting the order of experimental stages for rats included in the MK212 and MK212+SB242084 experiments: (a) self-administration training; (b) effects of MK212 on maintenance of cocaine self-administration (0.75 mg/kg/0.1 ml, i.v.); (c) extinction training; (d) effects of MK212 or MK212+SB242084 on cue-elicited reinstatement; (e) effects of MK212 or MK212+SB242084 on cocaine-primed reinstatement; and (f) effects of MK212 priming on reinstatement. The sequence of stages (d), (e), and (f) were counterbalanced for order of presentation to control for order effects. The specific number of sessions at each phase varied depending on individual performance. Across all testing phases, rats received a maximum of eight intracranial microinfusions.
Self-Administration Training
Self-administration training occurred in operant conditioning chambers (30 × 25 × 25 cm; Med Associates, St Albans, VT) equipped with active and inactive levers, a cue light 4 cm above the active lever, a tone generator (500 Hz, 10 dB above background noise), and a house light on the top center of the wall opposite the levers. Each chamber was housed within a larger ventilated sound-attenuating chamber. Infusion pumps (Med Associates) were connected to liquid swivels (Instech, Plymouth Meeting, PA) located above the chambers. Swivels were fastened to catheters through polyethylene 50 tubing encased inside metal spring leashes (Plastics One).
Rats were trained to self-administer cocaine (0.75 mg/kg/0.1 ml, i.v.) 6 days/week during 2-h sessions. Schedule completions on the active lever resulted in the simultaneous activation of the cue light and tone generator followed 1 s later by a 6-s cocaine infusion. The light and tone cues were inactivated with the termination of the infusion and the house light switched on for 20 s to signal a timeout period, during which lever presses were recorded, but produced no consequences. Inactive lever presses were recorded, but produced no consequences. To facilitate acquisition of cocaine self-administration, rats were restricted to 16 g of food/day beginning 2 days before training and then maintained on food restriction (16–22 g/day) until they progressed from a fixed ratio (FR) 1 to a variable ratio (VR) 5 schedule of reinforcement. We chose a VR5 schedule of reinforcement because this schedule produces relatively high reinstatement response rates, thereby increasing sensitivity to detect MK212-induced decreases in reinstatement of extinguished cocaine-seeking behavior (Acosta et al, 2008). After reaching criterion for 5 consecutive days on a VR5 schedule, rats were given ad libitum access to food throughout the rest of training (ie at least 5 more sessions) and testing.
Effects of MK212 on Self-Administration
After reaching a stability criterion of <10% variability in the number of cocaine infusions/session across three consecutive sessions on the VR5 schedule, without upward or downward trends, rats were assigned to MK212 dosage groups (10, 30, 100 ng/0.2 μl/side) counterbalanced for previous cocaine intake (n=6–10/group). They were tested for the effects of MK212 on self-administration of cocaine (0.75 mg/kg, i.v.), receiving an intracranial vehicle (aCSF) microinfusion before one test and their assigned MK212 dose before the other test, with order of these treatments counterbalanced. The test sessions began 1–2 min after the microinfusions, and at least three training sessions intervened test days to re-establish stable self-administration rates.
Extinction Training
Extinction training began the day after the last self-administration session and consisted of daily 1-h exposures to the self-administration environment. During extinction, rats were connected to the tethers, and active and inactive lever responses were recorded, but produced no consequences (ie the cocaine infusion pump was not activated, and the discrete light and tone cues were not presented). Responding on the active lever in the absence of cocaine reinforcement is the operational definition of cocaine-seeking behavior. Extinction training continued until response rates on the active lever declined to 20% of the highest rate observed during extinction or to <20 responses per hour. Once responses declined to criterion levels, cocaine-seeking behavior was considered to be extinguished.
Effects of MK212 on Reinstatement
After reaching the extinction criterion, rats were re-assigned to MK212 dosage groups (10, 30, 100 ng/0.2 μl/side) counterbalanced for earlier cocaine intake (n=6–8/group). Rats received the same assigned dose of MK212 throughout the reinstatement test phase. There were a total of six, 1-h reinstatement tests. The first two tests assessed MK212 effects on extinction, and the other four tests included two cue-elicited and two cocaine-primed reinstatement tests, with the order of test type counterbalanced. For each test type, rats received an intracranial microinfusion of vehicle (aCSF) before one test and their assigned dose of MK212 before the other test, with order of these treatments counterbalanced. Rats were connected to the tethers 1–2 min after the microinfusions. At least three extinction sessions intervened between test days to re-establish baseline response rates. For MK212-primed and cocaine-primed reinstatement tests, responses on the levers produced no consequences (ie no cues were presented). For cocaine-primed reinstatement tests, rats received cocaine (10 mg/kg, i.p.) primes immediately before placement into the self-administration chambers. To control for injection stress, rats were given i.p. saline injections immediately before the two extinction sessions preceding cocaine-primed reinstatement testing, and response rates during these sessions served as the extinction baseline. During cue-induced reinstatement tests, active lever responses on an FR1 schedule resulted in presentations of the stimulus complex previously paired with cocaine infusions (ie, light and tone cues, infusion pump). If a rat did not respond within the first 5 min, a non-contingent cue was presented.
Effects of SB242084 on MK212-Induced Decreases in Reinstatement
A separate cohort of rats was used to examine whether the 5-HT2CR antagonist SB242084 blocked the effects of MK212 on reinstatement. Rats were assigned to one of two groups counterbalanced for cocaine intake during self-administration: MK212/SB242084 or vehicle/SB242084 (n=8–10/group). Rats were tested twice for cue-induced reinstatement and twice for cocaine-primed reinstatement, with order of test type counterbalanced. For each test type, rats in the MK212/SB242084 group were pretreated with MK212 (100 ng/0.2 μl/side) before one test and MK212 (100 ng/0.1 μl/side)+SB242084 (200 ng/0.1 μl/side) before the other test; rats in the vehicle/SB242084 group were pretreated with vehicle before one test and SB242084 (200 ng/0.2 μl/side) before the other test. Order of respective pretreatments was counterbalanced. All other aspects of training and testing were identical to the previous experiment.
Histology
Rats were killed with sodium pentobarbital (100 mg/kg, i.p.) and then 0.2 μl/side of 1% methylene blue was infused intracranially to verify cannulae tip placements. Placements were determined from coronal sections (40 μm) under a microscope by an observer blind to experimental conditions. Sections were then thionin stained to assess possible neurotoxicity.
Statistical Analyses
Infusion and response rates were analyzed using separate ANOVAs with dosage group as a between-subjects variable, when appropriate, and test session (baseline, vehicle pretreatment, and MK212 and/or SB242084 pretreatment) as the repeated-measures factor. Post hoc Newman–Keuls tests provided subsequent pairwise comparisons. Baseline response rates for these measures were defined as the average response rate during the self-administration or extinction sessions that preceded vehicle and drug pretreatment tests. Reinstatement for both the cue- and cocaine-primed tests was operationally defined as a minimum of 10 active lever responses and at least a doubling of baseline response rate during either the vehicle (aCSF) or drug (MK212 and/or SB242084) pretreatment test session; animals that failed to meet these criteria on both test days were excluded from the analyses as ‘non-reinstaters.'
RESULTS
Histology
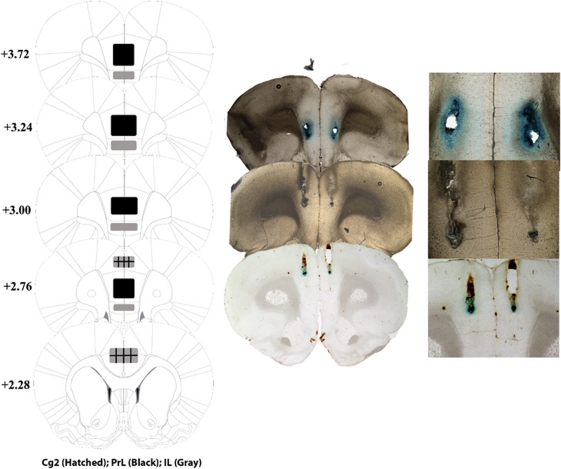
Figure 2 presents serial histological reconstructions and representative photomicrographs of injector tip placements within the subregions of the mPFC for rats included in the analyses. Three rats were excluded due to placements outside the mPFC and five were eliminated because of catheter failure. Microscopic examination of thionin-stained coronal sections indicated a lack of tissue damage aside from typical amounts sustained from cannulae implantation. Neurotoxicity, indicated by neuronal cell loss filled with gliosis, was not observed. Furthermore, the stable and consistent levels of reinstatement elicited by cocaine-priming injections and exposure to cocaine-paired cues (see below) after intracranial vehicle microinfusions are consistent with a lack of neurotoxicity.
Figure 2.
Histological reconstructions (left) presenting injector tip placements within the PrL (black), IL (gray), and Cg2 (hatched) subregions of the mPFC of rats included in the analysis; schematic representations (Paxinos and Watson, 2007) were used with permission from Elsevier. Representative photomicrographs showing methylene blue microinfusions for each mPFC subregion taken at magnifications of × 1 (middle) and × 10 (right).
Self-Administration Training
Depending on individual performance, rats tested for the effects of MK212 on cocaine reinforcement and reinstatement of extinguished cocaine-seeking behavior received a minimum of 16–35 self-administration sessions before testing and a maximum of 22–42 total sessions, including those that occurred during the training and testing phases; rats in the antagonist experiment received 26 training sessions. Total cocaine intake and the average number of infusions across the last 5 days of VR5 self-administration training did not differ across groups in either experiment, nor was there a difference in response rates (Table 1).
Table 1. Cocaine Reinforcers and Response Rates (Mean±SEM) During Self-Administration (SA) and Extinction.
| Brain region/drug | Infusions/session (last 5 days) | Total infusions |
Active lever presses/hour |
|
|---|---|---|---|---|
| Last SA day | First extinction day | |||
| PrL | ||||
| 10 ng/side MK212 (n=7) | 26.11±0.82 | 642.86±11.29 | 91.57±14.74 | 135.14±34.17 |
| 30 ng/side MK212 (n=8) | 26.43±0.94 | 602.00±38.09 | 81.56±12.92 | 82.88±10.12 |
| 100 ng/side MK212 (n=7) | 27.17±1.59 | 718.57±62.86 | 78.29±13.76 | 109.71±15.68 |
| IL | ||||
| 10 ng/side MK212 (n=6) | 23.70±1.24 | 663.33±76.27 | 64.17±6.88 | 119.18±16.13 |
| 30 ng/side MK212 (n=7) | 29.31±2.25 | 698.29±69.76 | 119.43±43.43 | 85.86±19.96 |
| 100 ng/side MK212 (n=7) | 25.17±1.26 | 687.57±47.49 | 67.64±4.91 | 137.14±14.85 |
| Cg2 | ||||
| 100 ng/side MK212 (n=7) | 27.34±2.18 | 658.00±58.50 | 67.00±4.10 | 112.00±31.57 |
| PrL/IL | ||||
| SB242084/vehicle (n=10) | 30.76±1.84 | 521.50±52.48 | 90.65±10.24 | 98.50±13.45 |
| MK212/MK212+SB242084 (n=10) | 29.66±2.01 | 496.80±40.03 | 78.80±7.88 | 97.60±11.53 |
Each i.v. cocaine infusion contained 0.75 mg/kg/0.1 ml.
Extinction Training
Depending on individual performance, rats tested for the effects of MK212 and/or SB242084 on reinstatement of extinguished cocaine-seeking behavior received a minimum of 12–39 extinction sessions before testing and a maximum of 36–68 total sessions, including those that occurred during the training and testing phases. Active lever presses decreased during extinction training, and there were no group differences on the first day of extinction (Table 1) or across the 12 days of extinction prior to reinstatement testing (data not shown).
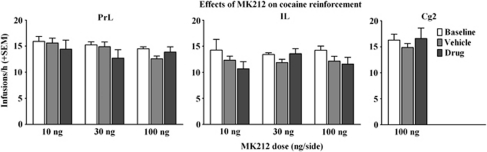
Effects of MK212 on Cocaine Self-Administration
Figure 3 illustrates the effects of intra-mPFC MK212 microinfusions on the number of cocaine infusions obtained during self-administration testing. MK212 failed to produce a dosage group by test-session interaction, but the ANOVA indicated a main effect of test session [F(2, 34)=5.4, p<0.001]. When collapsed across MK212 dosage groups, post hoc Newman–Keuls revealed a decrease in responding on both the vehicle and drug pretreatment tests relative to baseline. There was no difference between vehicle and drug groups when collapsed across dose, suggesting that these effects resulted from injection stress rather than stimulation of 5-HT2CRs.
Figure 3.
Effects of MK212 microinfused into the PrL, IL, and Cg2 subregions of the mPFC (10, 30, 100 ng/side) on the mean number of reinforcers (+SEM) obtained on a VR5 schedule of cocaine (0.75 mg/kg, i.v.) reinforcement during a 1-h test session. Baselines (white bars) represent mean infusions during the self-administration sessions preceding each test. Rats (n=6–10/group) were pretreated with vehicle (gray bars) before one test and their assigned dose of MK212 (black bars) before the other test, with order of these treatments counterbalanced. IL microinfusions produced a main effect of day regardless of MK212 dosage group. Post hoc comparisons collapsed across dosage groups indicated that the mean (+SEM) number of infusions after vehicle (12.10±0.44) or drug (12.00±0.72) was lower than that obtained during baseline (13.95±0.65; Newman–Keuls, P<0.05).
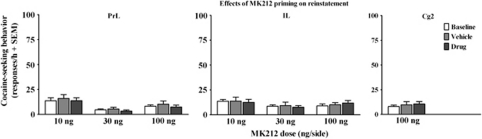
Effects of MK212 Alone on Reinstatement
Figure 4 illustrates the effects of intra-mPFC MK212 microinfusions on extinguished active lever responding. The ANOVAs of these data for each PFC subregion failed to uncover significant effects.
Figure 4.
Effects of MK212 (10, 30, 100 ng/side) microinfused alone into the PrL, IL, and Cg2 subregions of the mPFC on extinguished cocaine-seeking behavior expressed as the mean number of active lever responses (+SEM) during a 1-h test session. Baselines (white bars) represent mean responses during the extinction sessions preceding each test. Rats (n=6–8/group) were pretreated with vehicle (gray bars) before one test and their assigned dose of MK212 (black bars) before the other test, with order of these treatments counterbalanced; responses produced no scheduled consequences during testing nor did rats receive cocaine on the test day.
Effects of MK212 on Cue-Elicited Reinstatement
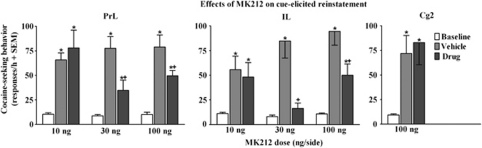
Of the 49 rats tested for cue-elicited reinstatement, 2 rats from the IL and 1 from the Cg2 group failed to meet the reinstatement criteria and were omitted. Figure 5 illustrates the effects of intra-mPFC MK212 microinfusions on active lever responding during FR1 cue-elicited reinstatement of extinguished cocaine-seeking behavior. ANOVAs of active lever responses revealed dosage group by test-session interactions for the PrL [F(4, 38)=4.1, p<0.01] and IL [F(4, 30)=3.3, p<0.05] cortices, and a main effect of day for the Cg2 [F(2, 12)=8.7, p<0.005] region. Lever presses on the inactive lever were negligible and there was no effect of MK212 in any of the mPFC subregions on inactive lever responding (data not shown). Post hoc comparisons indicated an increase in responding during the vehicle pretreatment test when response-contingent cues were available, relative to baseline when active lever responses produced no consequences (Newman–Keuls, p<0.05), indicating cue-elicited reinstatement of extinguished cocaine-seeking behavior in all groups pretreated with vehicle regardless of drug-dosage group or region. MK212 dose and region dependently decreased cue-elicited reinstatement of extinguished cocaine-seeking behavior. In both the PrL and IL cortices, the 10 ng/side dose failed to alter reinstatement as responding increased relative to baseline (Newman–Keuls, p<0.05) and did not differ from vehicle pretreatment. In the PrL cortex, rats pretreated with the 30 and 100 ng/side doses exhibited increased responding relative to baseline, but also attenuated reinstatement of responding relative to vehicle (Newman–Keuls, p<0.05). In the IL cortex, the 30 ng/side dose blocked reinstatement as responding did not differ from baseline and was decreased relative to vehicle pretreatment, whereas the 100 ng/side dose attenuated reinstatement as responding was increased relative to baseline, but was also decreased relative to vehicle pretreatment (Newman–Keuls, p<0.05). In the Cg2 region, the 100 ng/side dose failed to alter reinstatement as responding did not differ relative to vehicle pretreatment and was increased relative to baseline (Newman–Keuls, p<0.05).
Figure 5.
Effects of MK212 (10, 30, 100 ng/side) microinfused into the PrL, IL, and Cg2 subregions of the mPFC on cue-elicited reinstatement of extinguished cocaine-seeking behavior expressed as the mean number of active lever responses (+SEM) during a 1-h test session. Baselines (white bars) represent mean responses during the extinction sessions preceding each test. Rats (n=6–8/group) were pretreated with vehicle (gray bars) before one test and their assigned dose of MK212 (black bars) before the other test, with order of these treatments counterbalanced. Cues were available response contingently during the test session on an FR1 schedule. Asterisk (*) represents a difference from baseline (Newman–Keuls, p<0.05). Plus sign (+) represents a difference from vehicle pretreatment test day (Newman–Keuls, p<0.05).
Effects of MK212 on Cocaine-Primed Reinstatement
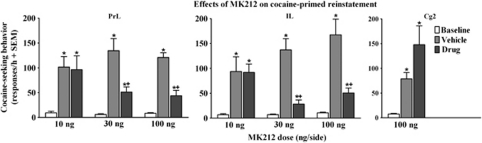
Of the 49 rats tested for cocaine-primed reinstatement, 1 rat from the IL group failed to meet the reinstatement criteria and was omitted. Figure 6 illustrates the effects of intra-mPFC MK212 microinfusions on active lever responding during the cocaine-primed (10 mg/kg, i.p.) reinstatement tests. The ANOVAs of active lever responses revealed dosage group by test-session interactions for the PrL [F(4, 38)=4.2, p<0.01] and IL [F(4, 30)=6.7, p<0.001] cortices, and a main effect of day for the Cg2 [F(2, 10)=9.3, p<0.01] region. Lever presses on the inactive lever were negligible and there was no effect of MK212 in any of the mPFC subregions on inactive lever responding (data not shown). Post hoc comparisons indicated an increase in responding on the vehicle pretreatment test when cocaine-priming injections were given, relative to baseline when saline-priming injections were administered (Newman–Keuls, p<0.05), indicating cocaine-primed reinstatement of extinguished cocaine-seeking behavior in all groups pretreated with vehicle regardless of drug dose or region. MK212 dose and region dependently decreased cocaine-primed reinstatement. In the PrL and IL cortices, the 10 ng/side dose failed to alter reinstatement as responding increased relative to baseline and did not differ from vehicle pretreatment, whereas the 30 and 100 ng/side doses attenuated reinstatement as responding increased relative to baseline, but was decreased relative to vehicle pretreatment (Newman–Keuls, p<0.05). In the Cg2 region, the 100 ng/side dose failed to alter reinstatement as responding did not differ relative to vehicle pretreatment and was increased relative to baseline (Newman–Keuls, p<0.05).
Figure 6.
Effects of MK212 (10, 30, 100 ng/side) microinfused into the PrL, IL, and Cg2 subregions of the mPFC on cocaine-primed (10 mg/kg, i.p.) reinstatement of extinguished cocaine-seeking behavior expressed as the mean number of active lever responses (+SEM) during a 1-h test session. Baselines (white bars) represent mean responses during the extinction sessions preceding each test. Rats (n=6–8/group) were pretreated with vehicle (gray bars) before one test and their assigned dose of MK212 (black bars) before the other test, with order of these treatments counterbalanced. The cocaine prime was administered after the intra-mPFC microinfusions and immediately before testing. No cues were presented during the test sessions. Asterisk (*) represents a difference from baseline (Newman–Keuls, p<0.05). Plus sign (+) represents a difference from vehicle pretreatment test day (Newman–Keuls, p<0.05).
Effects of SB242084 on MK212-Induced Decreases in Cue-Elicited Reinstatement
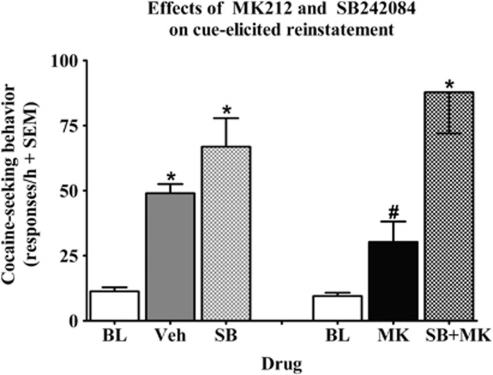
Of the 20 rats tested for cue-elicited reinstatement, 2 rats from the vehicle/SB242084 group failed to meet reinstatement criteria and were omitted. Figure 7 illustrates the effects of SB242084 on the MK212-induced decrease in active lever responding during cue-elicited reinstatement testing. The ANOVAs of active lever responses revealed drug group by test-session interactions for both the MK212/SB242084 [F(2, 18)=17.7, p<0.001] and vehicle/SB242084 [F(2, 14)=15.8, p<0.001] groups. Lever presses on the inactive lever were negligible and there was no effect of intra-mPFC microinfusions on inactive lever presses (data not shown). Post hoc comparisons indicated an increase in responding during the vehicle, SB242084, and MK212+SB242084 pretreatment test sessions when response-contingent cues were available, relative to baseline when active lever responses produced no consequences (Newman–Keuls, p<0.05), indicating cue-elicited reinstatement of extinguished cocaine-seeking behavior regardless of drug-dosage group. MK212 blocked cue-elicited reinstatement as rats receiving 100 ng/side of MK212 did not differ from their respective baselines and exhibited a reduction in active lever responses after exposure to cocaine-paired cues compared with MK212+SB242084 pretreatment (Newman–Keuls, p<0.05).
Figure 7.
Effects of SB242084 (200 ng/side) on the MK212 (100 ng/side)-induced decrease in cue-elicited reinstatement of extinguished cocaine-seeking behavior expressed as the mean number of active lever responses (+SEM) during a 1-h test session. Baselines (white bars) represent mean responses during the extinction sessions preceding each test. Rats in the vehicle/SB242084 group (n=8–10/group) were pretreated with vehicle (gray bars) before one test and SB242084 (checkered gray bars) before the other test, whereas rats in the MK212/SB242084 group were pretreated with MK212 (black bars) before one test and MK212+SB242084 (checkered black bars) before the other test, with order of these treatments counterbalanced. Cues were available response contingently on an FR1 schedule. Asterisk (*) represents a difference from baseline (Newman–Keuls, p<0.05). Pound sign (#) represents a difference from MK212+SB242084 pretreatment test day (Newman–Keuls, p<0.05).
Effects of SB242084 on MK212-Induced Decreases in Cocaine-Primed Reinstatement
Of the 20 rats tested for cocaine-primed reinstatement, 1 rat from the vehicle/SB242084 drug group failed to meet criteria and was omitted. Figure 8 illustrates the effects of SB242084 on the MK212-induced decrease in active lever responding during cocaine-primed reinstatement testing. The ANOVAs of active lever responses revealed drug-dosage group by test-session interactions for both the MK212/SB242084 [F(2, 18)=24.5, p<0.001] and vehicle/SB242084 [F(2, 16)=9.2, p<0.005] groups. Lever presses on the inactive lever were negligible and there was no effect of intra-mPFC microinfusions on inactive lever presses (data not shown). Post hoc comparisons indicated an increase in responding during the vehicle, SB242084, and MK212+SB242084 pretreatment test sessions when cocaine-priming injections were given, relative to baseline when saline-priming injections were administered (Newman–Keuls, p<0.05), indicating cocaine-primed reinstatement of extinguished cocaine-seeking behavior regardless of drug-dosage group. MK212 blocked cocaine-primed reinstatement as rats receiving 100 ng/side of MK212 did not differ from their respective baselines, and exhibited a reduction in active lever responses after cocaine-priming injections compared with MK212+SB242084 pretreatment (Newman–Keuls, p<0.05).
Figure 8.
Effects of SB242084 (200 ng/side) on the MK212 (100 ng/side)-induced decrease in cocaine-primed reinstatement of extinguished cocaine-seeking behavior expressed as the mean number of active lever responses (+SEM) during a 1-h test session. Baselines (white bars) represent mean responses during the extinction sessions preceding each test. Rats in the vehicle/SB242084 group (n=8–10/group) were pretreated with vehicle (gray bars) before one test and SB242084 (checkered gray bars) before the other test, whereas rats in the MK212/SB242084 group were pretreated with MK212 (black bars) before one test and MK212+SB242084 (checkered black bars) before the other test, with order of these treatments counterbalanced. For cocaine-primed reinstatement, the cocaine prime (10 mg/kg, i.p.) was administered immediately before testing and no cues were presented during the test sessions. Asterisk (*) represents a difference from baseline (Newman–Keuls, p<0.05). Pound sign (#) represents a difference from MK212+SB242084 pretreatment test day (Newman–Keuls, p<0.05).
DISCUSSION
The results from this study show that microinfusions of the selective 5-HT2CR agonist MK212 into the PrL and IL subregions of the mPFC dose-dependently attenuated cue-elicited and cocaine-primed reinstatement of extinguished cocaine-seeking behavior. Although other studies have evaluated the effects of intra-vmPFC MK212 microinfusions on the stimulant and discriminative stimulus effects of cocaine (Filip and Cunningham, 2003), this is the first study to show a function for mPFC 5-HT2CRs in mediating the incentive motivation for cocaine. These effects seem to be region specific as microinfusions into the neighboring Cg2 subregion of the anterior cingulate cortex did not alter cue- or cocaine-primed reinstatement. Furthermore, the reduction in cocaine-seeking behavior seems to be behaviorally specific as the number of cocaine reinforcers obtained during self-administration testing at the training dose of cocaine (0.75 mg/kg, i.v.) was not altered by MK212 microinfusions into any of the three subregions of the mPFC, although the microinfusion procedure itself produced a small decrease in self-administration rate regardless of whether drug or vehicle was infused.
MK212 binds to 5-HT2CRs with the highest affinity compared with other receptors; however, it also has affinity for 5-HT2A, 5-HT2B, and 5-HT3Rs (Cussac et al, 2002; Glennon et al, 1989; Kennett, 1993; Porter et al, 1999). To verify that the effects of MK212 on cue-elicited and cocaine-primed reinstatement were 5-HT2CR mediated, we showed that co-administration of the 5-HT2CR antagonist SB242084 blocked the agonist effects. SB242084 has a high affinity for 5-HT2CRs (pKi=9) relative to 5-HT2BRs and 5-HT2ARs, with a 150-fold selectivity over the 2A and 2B receptors (Kennett et al, 1997). The ability of SB242084 to block the effects of MK212 suggests that these effects are 5-HT2CR mediated. Moreover, because SB242084 administered alone as a drug prime did not alter cocaine-seeking behavior, it seems unlikely that stimulation of mPFC 5-HT2CRs through tonic levels of 5-HT modulates incentive motivation for cocaine. Furthermore, increasing stimulation of 5-HT2CRs above tonic levels with MK212 in the absence of a reinstating stimulus did not alter cocaine-seeking behavior, but attenuated responding elicited by cues and cocaine primes, providing evidence that increased stimulation of 5-HT2CRs within the mPFC inhibits the incentive motivational effects of these stimuli. Collectively, these results strongly suggest that the effects of intra-mPFC MK212 microinfusions on cue-elicited and cocaine-primed reinstatement of extinguished cocaine-seeking behavior are 5-HT2CR mediated.
The decrease in cocaine-seeking behavior after intra-mPFC MK212 microinfusions may have involved several mechanisms, including sensory or motor systems, learning and memory, or motivation. In our view, however, it is unlikely that learning and memory systems were responsible for the present behavioral effects, because the rats' self-administration behavior, which involves memory, was not affected. Moreover, lesions of the PrL cortex have been shown to attenuate cocaine-primed reinstatement of extinguished cocaine-conditioned place preference without altering acquisition or extinction of this behavior (Zavala et al, 2003). Earlier research has shown that microinfusions of MK212 into the vmPFC block cocaine-induced hyperactivity without altering spontaneous locomotion (Filip and Cunningham, 2003). In light of the latter findings and our results showing that MK212 did not impair operant behavior during cocaine self-administration testing, it seems unlikely that the observed effects of MK212 on cocaine-seeking behavior resulted from non-specific motor impairments. Finally, the present findings are complementary to previous research showing that MK212 microinfusions into the vmPFC attenuate the discriminative stimulus effects of cocaine (Filip and Cunningham, 2003). Although drug discrimination and reinstatement of extinguished cocaine-seeking behavior are not different behavioral expressions of a unitary neurobiological process, there is a considerable degree of overlap between reinstatement of drug-seeking behavior and drug discrimination (for a review see Spealman et al, 1999). From the above discussion, we favor the explanation that enhanced signaling through 5-HT2CRs located within the PrL and IL, but not Cg2, subregions of the mPFC attenuates cocaine-induced and cocaine-conditioned incentive motivation for cocaine.
It is important to note that the purpose of examining the effects of intra-mPFC MK212 microinfusions on cocaine self-administration was to provide a control manipulation to examine the specificity of potential effects on cocaine-seeking behavior, rather than to investigate the function of cortical 5-HT2CRs in cocaine reinforcement, per se. This study is, therefore, limited because only one dose of cocaine (0.75 mg/kg, i.v.) was examined. However, the 0.75 mg/kg, i.v. dose of cocaine that was tested falls midway on the descending limb of the cocaine dose-effect function, thus we likely had the sensitivity to detect either an increase or decrease in reinforcement rates during self-administration testing. Nevertheless, we cannot rule out the possibility that mPFC 5-HT2CRs may influence cocaine reinforcement at other doses as peripheral administration of the 5-HT2CR agonist Ro600175 reduces maintenance of cocaine self-administration, as well as break points for cocaine on a progressive ratio schedule of reinforcement (Grottick et al, 2000). Furthermore, systemic administration of the selective 5-HT2CR antagonist SB242084 increases responding for low (0.0625 and 0.125, but not 0.25 mg) doses of cocaine (Fletcher et al, 2002b), and 5-HT2CR-deficient mice self-administer more cocaine reinforcers on a progressive ratio schedule compared with wild-type controls (Rocha et al, 2002). In light of these findings, we speculate that 5-HT2CRs in regions other than the mPFC likely modulate the reinforcing effects of cocaine. For instance, microinfusions of the 5-HT2CR agonist Ro600175 into the VTA reduce the reinforcing effects of self-administered cocaine on both fixed and progressive ratios (Fletcher et al, 2004), suggesting that the VTA may be responsible for the reduction of cocaine self-administration by systemically administered 5-HT2CR agonists.
Neuroanatomically, the reduction in cocaine-seeking behavior following intra-mPFC MK212 microinfusions may have resulted from inhibition of the mesolimbic dopamine (DA) system through 5-HT2CR stimulation of a mPFC afferent pathway. A fundamental feature of addiction is the inability to self-regulate chronic drug-seeking and -taking behaviors (Jentsch and Taylor, 1999; Kalivas, 2008), and these deficits in self-regulation are associated with functional ‘hypofrontality' in the PFC (Childress et al, 1999; Goldstein and Volkow, 2002). The mPFC is densely innervated by 5-HT neurons (Van Bockstaele et al, 1993) and 5-HT2CRs provide modulatory influence over PFC function (Clemett et al, 2000; Lopez-Gimenez et al, 2001; Pompeiano et al, 1994). 5-HT2CRs within the mPFC are localized postsynaptically (Clemett et al, 2000) on both GABA interneurons (Liu et al, 2007) that form numerous synapses on glutamate pyramidal projection neurons, and on glutamatergic output neurons directly (Carr et al, 2002; Vysokanov et al, 1998), thus regulating mPFC output (Eyles et al, 2002). Pyramidal projections terminate in subcortical regions, including the VTA and NAc, which are the origin and terminal regions, respectively, of the DA mesoaccumbens pathway (Gabbott et al, 2005). Thus, stimulation of 5-HT2CRs located on mPFC GABA interneurons or glutamate projection neurons may reduce or increase, respectively, excitatory output from the mPFC to the mesoaccumbens pathway, providing a mechanism for mPFC 5-HT2CRs to regulate glutamate and DA levels in the NAc.
In support of the above hypothesis, GABA agonist microinfusions into the mPFC inhibit glutamate release in the VTA and NAc (Harte and O'Connor, 2005), whereas GABA antagonist microinfusions into mPFC increase DA release in the NAc (Karreman and Moghaddam, 1996). These effects could involve 5-HT2CR mediation as intra-mPFC iontophoretic application of mCPP, a drug possessing 5-HT2CR agonist properties, suppresses spontaneous and glutamate-activated firing of PFC neurons (Bergqvist et al, 1999). In addition, 5-HT2CR mutant mice exhibited higher baseline extracellular DA levels in the NAc (Abdallah et al, 2009) and increased cocaine-induced DA release in the NAc compared with wild-type controls (Rocha et al, 2002). In contrast, mPFC electrical stimulation increases glutamate levels in the VTA (Rossetti et al, 1998) and NAc, as well as DA release in the NAc (You et al, 1998). Thus, the results obtained from MK212 microinfusions into the mPFC may have resulted from activation of 5-HT2CRs located on GABA neurons, leading to a reduction in DA release in the NAcsh and an accompanying decrease in cocaine-seeking behavior. Importantly, GABA agonists infused into the PrL cortex block cocaine-primed reinstatement (McFarland and Kalivas, 2001), whereas GABA agonists infused into the vmPFC encompassing both the PrL and IL cortices block incubation of cue-elicited craving (Koya et al, 2009), suggesting that the effects of MK212 in the present study involve increases in GABA neurotransmission. However, the reduction in cocaine-seeking behavior after MK212 microinfusions into the IL cortex may have resulted from direct stimulation of glutamatergic projection neurons as microinfusions of the glutamate agonist AMPA into this region reduce cocaine-seeking behavior as well (Peters et al, 2008).
In conclusion, this study provides strong evidence for an inhibitory function of mPFC 5-HT2CRs in reinstatement of extinguished cocaine-seeking behavior produced by either drug-paired cues or cocaine-priming injections. Further research is needed to determine the neural circuitry through which mPFC 5-HT2CRs produce inhibitory effects on cocaine-seeking behavior. Elucidating the neural mechanisms underlying the incentive motivational effects produced by sampling cocaine and exposure to cocaine-associated cues is important for developing pharmacological treatments to help prevent cocaine craving and relapse.
Acknowledgments
We thank Valeria Routt and Natalie Peartree for their expert surgical assistance. This research was supported by NIDA grants F32DA025413 and R01DA11064.
The authors declare no conflict of interest.
References
- Abdallah L, Bonasera SJ, Hopf FW, O'Dell L, Giorgetti M, Jongsma M, et al. Impact of serotonin 2C receptor null mutation on physiology and behavior associated with nigrostriatal dopamine pathway function. J Neurosci. 2009;29:8156–8165. doi: 10.1523/JNEUROSCI.3905-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acosta JI, Thiel KJ, Sanabria F, Browning JR, Neisewander JL. Effect of schedule of reinforcement on cue-elicited reinstatement of cocaine-seeking behavior. Behav Pharmacol. 2008;19:129–136. doi: 10.1097/FBP.0b013e3282f62c89. [DOI] [PubMed] [Google Scholar]
- Aronson SC, Black JE, McDougle CJ, Scanley BE, Jatlow P, Kosten TR, et al. Serotonergic mechanisms of cocaine effects in humans. Psychopharmacology (Berl) 1995;119:179–185. doi: 10.1007/BF02246159. [DOI] [PubMed] [Google Scholar]
- Baker DA, Tran-Nguyen TL, Fuchs RA, Neisewander JL. Influence of individual differences and chronic fluoxetine treatment on cocaine-seeking behavior in rats. Psychopharmacology (Berl) 2001;155:18–26. doi: 10.1007/s002130000676. [DOI] [PubMed] [Google Scholar]
- Barnes NM, Sharp T. A review of central 5-HT receptors and their function. Neuropharmacology. 1999;38:1083–1152. doi: 10.1016/s0028-3908(99)00010-6. [DOI] [PubMed] [Google Scholar]
- Batki SL, Manfredi LB, Jacob Pd, Jones RT. Fluoxetine for cocaine dependence in methadone maintenance: quantitative plasma and urine cocaine/benzoylecgonine concentrations [see comments] J Clin Psychopharmacol. 1993;13:243–250. [PubMed] [Google Scholar]
- Baumann MH, Rothman RB. Repeated cocaine administration reduces 5-HT1A-mediated prolactin secretion in rats. Neurosci Lett. 1995;193:9–12. doi: 10.1016/0304-3940(95)11652-d. [DOI] [PubMed] [Google Scholar]
- Baumann MH, Rothman RB. Chronic cocaine exposure potentiates prolactin and head shake responses to 5-HT2 receptor stimulation in rats. Neuropharmacology. 1996;35:295–301. doi: 10.1016/0028-3908(95)00166-2. [DOI] [PubMed] [Google Scholar]
- Baumann MH, Rothman RB. Alterations in serotonergic responsiveness during cocaine withdrawal in rats: similarities to major depression in humans. Biol Psychiatry. 1998;44:578–591. doi: 10.1016/s0006-3223(98)00123-1. [DOI] [PubMed] [Google Scholar]
- Bergqvist PB, Dong J, Blier P. Effect of atypical antipsychotic drugs on 5-HT2 receptors in the rat orbito-frontal cortex: an in vivo electrophysiological study. Psychopharmacology (Berl) 1999;143:89–96. doi: 10.1007/s002130050923. [DOI] [PubMed] [Google Scholar]
- Bubar MJ, Cunningham KA. Prospects for serotonin 5-HT2R pharmacotherapy in psychostimulant abuse. Prog Brain Res. 2008;172:319–346. doi: 10.1016/S0079-6123(08)00916-3. [DOI] [PubMed] [Google Scholar]
- Burbassi S, Cervo L. Stimulation of serotonin2C receptors influences cocaine-seeking behavior in response to drug-associated stimuli in rats. Psychopharmacology (Berl) 2008;196:15–27. doi: 10.1007/s00213-007-0916-7. [DOI] [PubMed] [Google Scholar]
- Burmeister JJ, Lungren EM, Kirschner KF, Neisewander JL. Differential roles of 5-HT receptor subtypes in cue and cocaine reinstatement of cocaine-seeking behavior in rats. Neuropsychopharmacology. 2004;29:660–668. doi: 10.1038/sj.npp.1300346. [DOI] [PubMed] [Google Scholar]
- Burmeister JJ, Lungren EM, Neisewander JL. Effects of fluoxetine and d-fenfluramine on cocaine-seeking behavior in rats. Psychopharmacology (Berl) 2003;168:146–154. doi: 10.1007/s00213-002-1307-8. [DOI] [PubMed] [Google Scholar]
- Capriles N, Rodaros D, Sorge RE, Stewart J. A role for the prefrontal cortex in stress- and cocaine-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 2003;168:66–74. doi: 10.1007/s00213-002-1283-z. [DOI] [PubMed] [Google Scholar]
- Carr DB, Cooper DC, Ulrich SL, Spruston N, Surmeier DJ. Serotonin receptor activation inhibits sodium current and dendritic excitability in prefrontal cortex via a protein kinase C-dependent mechanism. J Neurosci. 2002;22:6846–6855. doi: 10.1523/JNEUROSCI.22-16-06846.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O'Brien CP. Limbic activation during cue-induced cocaine craving. Am J Psychiatry. 1999;156:11–18. doi: 10.1176/ajp.156.1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clemett DA, Punhani T, Duxon MS, Blackburn TP, Fone KC. Immunohistochemical localisation of the 5-HT2C receptor protein in the rat CNS. Neuropharmacology. 2000;39:123–132. doi: 10.1016/s0028-3908(99)00086-6. [DOI] [PubMed] [Google Scholar]
- Cunningham KA, Paris JM, Goeders NE. Chronic cocaine enhances serotonin autoregulation and serotonin uptake binding. Synapse. 1992;11:112–123. doi: 10.1002/syn.890110204. [DOI] [PubMed] [Google Scholar]
- Cussac D, Newman-Tancredi A, Quentric Y, Carpentier N, Poissonnet G, Parmentier JG, et al. Characterization of phospholipase C activity at h5-HT2C compared with h5-HT2B receptors: influence of novel ligands upon membrane-bound levels of [3H]phosphatidylinositols. Naunyn Schmiedebergs Arch Pharmacol. 2002;365:242–252. doi: 10.1007/s00210-001-0505-y. [DOI] [PubMed] [Google Scholar]
- Darmani NA, Martin BR, Glennon RA. Repeated administration of low doses of cocaine enhances the sensitivity of 5-HT2 receptor function. Pharmacol Biochem Behav. 1992;41:519–527. doi: 10.1016/0091-3057(92)90367-o. [DOI] [PubMed] [Google Scholar]
- Davis WM, Smith SG. Role of conditioned reinforcers in the initiation, maintenance and extinction of drug-seeking behavior. Pavlov J Biol Sci. 1976;11:222–236. doi: 10.1007/BF03000316. [DOI] [PubMed] [Google Scholar]
- de Wit H, Stewart J. Reinstatement of cocaine-reinforced responding in the rat. Psychopharmacology (Berl) 1981;75:134–143. doi: 10.1007/BF00432175. [DOI] [PubMed] [Google Scholar]
- Ding DC, Gabbott PL, Totterdell S. Differences in the laminar origin of projections from the medial prefrontal cortex to the nucleus accumbens shell and core regions in the rat. Brain Res. 2001;917:81–89. doi: 10.1016/s0006-8993(01)02912-2. [DOI] [PubMed] [Google Scholar]
- Epstein DH, Preston KL, Stewart J, Shaham Y. Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology (Berl) 2006;189:1–16. doi: 10.1007/s00213-006-0529-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eyles DW, McGrath JJ, Reynolds GP. Neuronal calcium-binding proteins and schizophrenia. Schizophr Res. 2002;57:27–34. doi: 10.1016/s0920-9964(01)00299-7. [DOI] [PubMed] [Google Scholar]
- Filip M. Role of serotonin (5-HT)2 receptors in cocaine self-administration and seeking behavior in rats. Pharmacol Rep. 2005;57:35–46. [PubMed] [Google Scholar]
- Filip M, Cunningham KA. Hyperlocomotive and discriminative stimulus effects of cocaine are under the control of serotonin(2C) (5-HT(2C)) receptors in rat prefrontal cortex. J Pharmacol Exp Ther. 2003;306:734–743. doi: 10.1124/jpet.102.045716. [DOI] [PubMed] [Google Scholar]
- Fletcher PJ, Chintoh AF, Sinyard J, Higgins GA. Injection of the 5-HT2C receptor agonist Ro60-0175 into the ventral tegmental area reduces cocaine-induced locomotor activity and cocaine self-administration. Neuropsychopharmacology. 2004;29:308–318. doi: 10.1038/sj.npp.1300319. [DOI] [PubMed] [Google Scholar]
- Fletcher PJ, Grottick AJ, Higgins GA. Differential effects of the 5-HT2A receptor antagonist M100907 and the 5-HT2C receptor antagonist SB242084 on cocaine-induced locomotor activity, cocaine self-administration and cocaine-induced reinstatement of responding. Neuropsychopharmacology. 2002a;27:576–586. doi: 10.1016/S0893-133X(02)00342-1. [DOI] [PubMed] [Google Scholar]
- Fletcher PJ, Grottick AJ, Higgins GA. Differential effects of the 5-HT(2A) receptor antagonist M100907 and the 5-HT(2C) receptor antagonist SB242084 on cocaine-induced locomotor activity, cocaine self-administration and cocaine-induced reinstatement of responding. Neuropsychopharmacology. 2002b;27:576–586. doi: 10.1016/S0893-133X(02)00342-1. [DOI] [PubMed] [Google Scholar]
- Fletcher PJ, Rizos Z, Sinyard J, Tampakeras M, Higgins GA. The 5-HT2C receptor agonist Ro60-0175 reduces cocaine self-administration and reinstatement induced by the stressor yohimbine, and contextual cues. Neuropsychopharmacology. 2008;33:1402–1412. doi: 10.1038/sj.npp.1301509. [DOI] [PubMed] [Google Scholar]
- Fuchs RA, Evans KA, Ledford CC, Parker MP, Case JM, Mehta RH, et al. The role of the dorsomedial prefrontal cortex, basolateral amygdala, and dorsal hippocampus in contextual reinstatement of cocaine seeking in rats. Neuropsychopharmacology. 2005;30:296–309. doi: 10.1038/sj.npp.1300579. [DOI] [PubMed] [Google Scholar]
- Gabbott PL, Dickie BG, Vaid RR, Headlam AJ, Bacon SJ. Local-circuit neurones in the medial prefrontal cortex (areas 25, 32 and 24b) in the rat: morphology and quantitative distribution. J Comp Neurol. 1997;377:465–499. doi: 10.1002/(sici)1096-9861(19970127)377:4<465::aid-cne1>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- Gabbott PL, Warner TA, Jays PR, Salway P, Busby SJ. Prefrontal cortex in the rat: projections to subcortical autonomic, motor, and limbic centers. J Comp Neurol. 2005;492:145–177. doi: 10.1002/cne.20738. [DOI] [PubMed] [Google Scholar]
- Glennon RA, Ismaiel AE, McCarthy BG, Peroutka SJ. Binding of arylpiperazines to 5-HT3 serotonin receptors: results of a structure-affinity study. Eur J Pharmacol. 1989;168:387–392. doi: 10.1016/0014-2999(89)90802-9. [DOI] [PubMed] [Google Scholar]
- Goeders NE, Smith JE. Parameters of intracranial self-administration of cocaine into the medial prefrontal cortex. NIDA Res Monogr. 1984;55:132–137. [PubMed] [Google Scholar]
- Goldstein RZ, Volkow ND. Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry. 2002;159:1642–1652. doi: 10.1176/appi.ajp.159.10.1642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grottick AJ, Fletcher PJ, Higgins GA. Studies to investigate the role of 5-HT2C receptors on cocaine- and food-maintained behavior. J Pharmacol Exp Ther. 2000;295:1183–1191. [PubMed] [Google Scholar]
- Harris DS, Batki SL, Berger SP. Fluoxetine attenuates adrenocortical but not subjective responses to cocaine cues. Am J Drug Alcohol Abuse. 2004;30:765–782. doi: 10.1081/ada-200037542. [DOI] [PubMed] [Google Scholar]
- Harte M, O'Connor WT. Evidence for a selective prefrontal cortical GABA(B) receptor-mediated inhibition of glutamate release in the ventral tegmental area: a dual probe microdialysis study in the awake rat. Neuroscience. 2005;130:215–222. doi: 10.1016/j.neuroscience.2004.08.045. [DOI] [PubMed] [Google Scholar]
- Hyman SE, Malenka RC, Nestler EJ. Neural mechanisms of addiction: the role of reward-related learning and memory. Annu Rev Neurosci. 2006;29:565–598. doi: 10.1146/annurev.neuro.29.051605.113009. [DOI] [PubMed] [Google Scholar]
- Jentsch JD, Taylor JR. Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology (Berl) 1999;146:373–390. doi: 10.1007/pl00005483. [DOI] [PubMed] [Google Scholar]
- Kalivas PW. Addiction as a pathology in prefrontal cortical regulation of corticostriatal habit circuitry. Neurotox Res. 2008;14:185–189. doi: 10.1007/BF03033809. [DOI] [PubMed] [Google Scholar]
- Karreman M, Moghaddam B. The prefrontal cortex regulates the basal release of dopamine in the limbic striatum: an effect mediated by ventral tegmental area. J Neurochem. 1996;66:589–598. doi: 10.1046/j.1471-4159.1996.66020589.x. [DOI] [PubMed] [Google Scholar]
- Kennett GA. 5-HT1C receptors and their therapeutic relevance. Curr Opin Invest Drugs. 1993;2:317–362. [Google Scholar]
- Kennett GA, Wood MD, Bright F, Trail B, Riley G, Holland V, et al. SB 242084, a selective and brain penetrant 5-HT2C receptor antagonist. Neuropharmacology. 1997;36:609–620. doi: 10.1016/s0028-3908(97)00038-5. [DOI] [PubMed] [Google Scholar]
- King GR, Joyner CM, Ellinwood EH., Jr Withdrawal from continuous or intermittent cocaine: behavioral responsivity to 5-HT1 receptor agonists. Pharmacol Biochem Behav. 1993;45:577–587. doi: 10.1016/0091-3057(93)90509-r. [DOI] [PubMed] [Google Scholar]
- Koya E, Uejima JL, Wihbey KA, Bossert JM, Hope BT, Shaham Y. Role of ventral medial prefrontal cortex in incubation of cocaine craving. Neuropharmacology. 2009;56 (Suppl 1:177–185. doi: 10.1016/j.neuropharm.2008.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee MA, Meltzer HY. Blunted oral body temperature response to MK-212 in cocaine addicts. Drug Alcohol Depend. 1994;35:217–222. doi: 10.1016/0376-8716(94)90077-9. [DOI] [PubMed] [Google Scholar]
- Levy AD, Li Q, Alvarez Sanz MC, Rittenhouse PA, Brownfield MS, Van de Kar LD. Repeated cocaine modifies the neuroendocrine responses to the 5-HT1C/5- HT2 receptor agonist DOI. Eur J Pharmacol. 1992;221:121–127. doi: 10.1016/0014-2999(92)90780-8. [DOI] [PubMed] [Google Scholar]
- Levy AD, Li Q, Van de Kar LD. Repeated cocaine exposure inhibits the adrenocorticotropic hormone response to the serotonin releaser d-fenfluramine and the 5-HT1A agonist, 8-OH-DPAT. Neuropharmacology. 1994;33:335–342. doi: 10.1016/0028-3908(94)90063-9. [DOI] [PubMed] [Google Scholar]
- Liu S, Bubar MJ, Lanfranco MF, Hillman GR, Cunningham KA. Serotonin2C receptor localization in GABA neurons of the rat medial prefrontal cortex: implications for understanding the neurobiology of addiction. Neuroscience. 2007;146:1677–1688. doi: 10.1016/j.neuroscience.2007.02.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Giménez JF, Mengod G, Palacios JM, Vilaró MT. Selective visualization of rat brain 5-HT2A receptors by autoradiography with [3H]MDL 100,907. Naunyn Schmiedebergs Arch Pharmacol. 1997;356:446–454. doi: 10.1007/pl00005075. [DOI] [PubMed] [Google Scholar]
- Lopez-Gimenez JF, Mengod G, Palacios JM, Vilaro MT. Regional distribution and cellular localization of 5-HT2C receptor mRNA in monkey brain: comparison with [3H]mesulergine binding sites and choline acetyltransferase mRNA. Synapse. 2001;42:12–26. doi: 10.1002/syn.1095. [DOI] [PubMed] [Google Scholar]
- McFarland K, Kalivas PW. The circuitry mediating cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2001;21:8655–8663. doi: 10.1523/JNEUROSCI.21-21-08655.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin J, See RE. Selective inactivation of the dorsomedial prefrontal cortex and the basolateral amygdala attenuates conditioned-cued reinstatement of extinguished cocaine-seeking behavior in rats. Psychopharmacology (Berl) 2003;168:57–65. doi: 10.1007/s00213-002-1196-x. [DOI] [PubMed] [Google Scholar]
- Neisewander JL, Acosta JI. Stimulation of 5-HT2C receptors attenuates cue and cocaine-primed reinstatement of cocaine-seeking behavior in rats. Behav Pharmacol. 2007;18:791–800. doi: 10.1097/FBP.0b013e3282f1c94b. [DOI] [PubMed] [Google Scholar]
- Neisewander JL, Baker DA, Fuchs RA, Tran-Nguyen LT, Palmer A, Marshall JF. Fos protein expression and cocaine-seeking behavior in rats after exposure to a cocaine self-administration environment. J Neurosci. 2000;20:798–805. doi: 10.1523/JNEUROSCI.20-02-00798.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumaier JF, Vincow ES, Arvanitogiannis A, Wise RA, Carlezon WA., Jr Elevated expression of 5-HT1B receptors in nucleus accumbens efferents sensitizes animals to cocaine. J Neurosci. 2002;22:10856–10863. doi: 10.1523/JNEUROSCI.22-24-10856.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nic Dhonnchadha BA, Fox RG, Stutz SJ, Rice KC, Cunningham KA. Blockade of the serotonin 5-HT2A receptor suppresses cue-evoked reinstatement of cocaine-seeking behavior in a rat self-administration model. Behav Neurosci. 2009;123:382–396. doi: 10.1037/a0014592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien CP. Anticraving medications for relapse prevention: a possible new class of psychoactive medications. Am J Psychiatry. 2005;162:1423–1431. doi: 10.1176/appi.ajp.162.8.1423. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C.2007The Rat Brain in Stereotaxic Coordinates6th edn.Academic Press/Elsevier: Amsterdam, Boston; 1 v. (unpaged)pp. [Google Scholar]
- Pentkowski NS, Acosta JI, Browning JR, Hamilton EC, Neisewander JL. Stimulation of 5-HT(1B) receptors enhances cocaine reinforcement yet reduces cocaine-seeking behavior. Addict Biol. 2009;14:419–430. doi: 10.1111/j.1369-1600.2009.00162.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, Kalivas PW, Quirk GJ. Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem. 2009;16:279–288. doi: 10.1101/lm.1041309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, LaLumiere RT, Kalivas PW. Infralimbic prefrontal cortex is responsible for inhibiting cocaine seeking in extinguished rats. J Neurosci. 2008;28:6046–6053. doi: 10.1523/JNEUROSCI.1045-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pompeiano M, Palacios JM, Mengod G. Distribution of the serotonin 5-HT2 receptor family mRNAs: comparison between 5-HT2A and 5-HT2C receptors. Brain Res Mol Brain Res. 1994;23:163–178. doi: 10.1016/0169-328x(94)90223-2. [DOI] [PubMed] [Google Scholar]
- Porter RH, Benwell KR, Lamb H, Malcolm CS, Allen NH, Revell DF, et al. Functional characterization of agonists at recombinant human 5-HT2A, 5-HT2B and 5-HT2C receptors in CHO-K1 cells. Br J Pharmacol. 1999;128:13–20. doi: 10.1038/sj.bjp.0702751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramos M, Goni-Allo B, Aguirre N. Administration of SCH 23390 into the medial prefrontal cortex blocks the expression of MDMA-induced behavioral sensitization in rats: an effect mediated by 5-HT2C receptor stimulation and not by D1 receptor blockade. Neuropsychopharmacology. 2005;30:2180–2191. doi: 10.1038/sj.npp.1300735. [DOI] [PubMed] [Google Scholar]
- Robbins TW, Everitt BJ. Limbic-striatal memory systems and drug addiction. Neurobiol Learn Mem. 2002;78:625–636. doi: 10.1006/nlme.2002.4103. [DOI] [PubMed] [Google Scholar]
- Rocha B, Di Scala G, Rigo M, Hoyer D, Sandner G. Effect of 5,7-dihydroxytryptamine lesion on mianserin-induced conditioned place aversion and on 5-hydroxytryptamine1C receptors in the rat brain. Neuroscience. 1993;56:687–693. doi: 10.1016/0306-4522(93)90365-m. [DOI] [PubMed] [Google Scholar]
- Rocha BA, Goulding EH, O'Dell LE, Mead AN, Coufal NG, Parsons LH, et al. Enhanced locomotor, reinforcing, and neurochemical effects of cocaine in serotonin 5-hydroxytryptamine 2C receptor mutant mice. J Neurosci. 2002;22:10039–10045. doi: 10.1523/JNEUROSCI.22-22-10039.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossetti ZL, Marcangione C, Wise RA. Increase of extracellular glutamate and expression of Fos-like immunoreactivity in the ventral tegmental area in response to electrical stimulation of the prefrontal cortex. J Neurochem. 1998;70:1503–1512. doi: 10.1046/j.1471-4159.1998.70041503.x. [DOI] [PubMed] [Google Scholar]
- Rothman RB, Blough BE, Baumann MH. Dual dopamine/serotonin releasers: potential treatment agents for stimulant addiction. Exp Clin Psychopharmacol. 2008;16:458–474. doi: 10.1037/a0014103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satel SL, Krystal JH, Delgado PL, Kosten TR, Charney DS. Tryptophan depletion and attenuation of cue-induced craving for cocaine. Am J Psychiatry. 1995;152:778–783. doi: 10.1176/ajp.152.5.778. [DOI] [PubMed] [Google Scholar]
- Spealman RD, Barrett-Larimore RL, Rowlett JK, Platt DM, Khroyan TV. Pharmacological and environmental determinants of relapse to cocaine-seeking behavior. Pharmacol Biochem Behav. 1999;64:327–336. doi: 10.1016/s0091-3057(99)00049-0. [DOI] [PubMed] [Google Scholar]
- Tran-Nguyen LT, Baker DA, Grote KA, Solano J, Neisewander JL. Serotonin depletion attenuates cocaine-seeking behavior in rats. Psychopharmacology (Berl) 1999;146:60–66. doi: 10.1007/s002130051088. [DOI] [PubMed] [Google Scholar]
- Tran-Nguyen LT, Bellew JG, Grote KA, Neisewander JL. Serotonin depletion attenuates cocaine seeking but enhances sucrose seeking and the effects of cocaine priming on reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 2001;157:340–348. doi: 10.1007/s002130100822. [DOI] [PubMed] [Google Scholar]
- Tzschentke TM, Schmidt WJ. Functional heterogeneity of the rat medial prefrontal cortex: effects of discrete subarea-specific lesions on drug-induced conditioned place preference and behavioural sensitization. Eur J Neurosci. 1999;11:4099–4109. doi: 10.1046/j.1460-9568.1999.00834.x. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Biswas A, Pickel VM. Topography of serotonin neurons in the dorsal raphe nucleus that send axon collaterals to the rat prefrontal cortex and nucleus accumbens. Brain Res. 1993;624:188–198. doi: 10.1016/0006-8993(93)90077-z. [DOI] [PubMed] [Google Scholar]
- Vysokanov A, Flores-Hernandez J, Surmeier DJ. mRNAs for clozapine-sensitive receptors co-localize in rat prefrontal cortex neurons. Neurosci Lett. 1998;258:179–182. doi: 10.1016/s0304-3940(98)00882-9. [DOI] [PubMed] [Google Scholar]
- Wallace BC. Treating crack cocaine dependence: the critical role of relapse prevention. J Psychoactive Drugs. 1992;24:213–222. doi: 10.1080/02791072.1992.10471641. [DOI] [PubMed] [Google Scholar]
- Walsh SL, Preston KL, Sullivan JT, Fromme R, Bigelow GE. Fluoxetine alters the effects of intravenous cocaine in humans. J Clin Psychopharmacol. 1994;14:396–407. [PubMed] [Google Scholar]
- Weiss F. Neurobiology of craving, conditioned reward and relapse. Curr Opin Pharmacol. 2005;5:9–19. doi: 10.1016/j.coph.2004.11.001. [DOI] [PubMed] [Google Scholar]
- You ZB, Tzschentke TM, Brodin E, Wise RA. Electrical stimulation of the prefrontal cortex increases cholecystokinin, glutamate, and dopamine release in the nucleus accumbens: an in vivo microdialysis study in freely moving rats. J Neurosci. 1998;18:6492–6500. doi: 10.1523/JNEUROSCI.18-16-06492.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahm DS, Brog JS. On the significance of subterritories in the ‘accumbens' part of the rat ventral striatum. Neuroscience. 1992;50:751–767. doi: 10.1016/0306-4522(92)90202-d. [DOI] [PubMed] [Google Scholar]
- Zavala AR, Weber SM, Rice HJ, Alleweireldt AT, Neisewander JL. Role of the prelimbic subregion of the medial prefrontal cortex in acquisition, extinction, and reinstatement of cocaine-conditioned place preference. Brain Res. 2003;990:157–164. doi: 10.1016/s0006-8993(03)03452-8. [DOI] [PubMed] [Google Scholar]