Abstract
In a recent human [11C]-(+)-PHNO positron emission tomography study, olanzapine, clozapine, and risperidone occupied D2 receptors in striatum (STR), but, despite their similar in vitro D2 and D3 affinities, failed to occupy D3 receptors in globus pallidus. This study had two aims: (1) to characterize the regional D2/D3 pharmacology of in vitro and ex vivo [3H]-(+)-PHNO binding sites in rat brain and (2) to compare, using [3H]-(+)-PHNO autoradiography, the ex vivo and in vitro pharmacology of olanzapine, clozapine, risperidone, and haloperidol. Using the D3-selective drug SB277011, we found that ex vivo and in vitro [3H]-(+)-PHNO binding in STR is exclusively due to D2, whereas that in cerebellar lobes 9 and 10 is exclusively due to D3. Surprisingly, the D3 contribution to [3H]-(+)-PHNO binding in the islands of Calleja, ventral pallidum, substantia nigra, and nucleus accumbens was greater ex vivo than in vitro. Ex vivo, systemically administered olanzapine, risperidone, and haloperidol, at doses occupying ∼80% D2, did not occupy D3 receptors. Clozapine, which also occupied ∼80% of D2 receptors ex vivo, occupied a smaller percentage of D3 receptors than predicted by its in vitro pharmacology. Across brain regions, ex vivo occupancy by antipsychotics was inversely related to the D3 contribution to [3H]-(+)-PHNO binding. In contrast, in vitro occupancy was similar across brain regions, independent of the regional D3 contribution. These data indicate that at clinically relevant doses, olanzapine, clozapine, risperidone, and haloperidol are D2-selective ex vivo. This unforeseen finding suggests that their clinical effects cannot be attributed to D3 receptor blockade.
Keywords: [3H]-(+)-PHNO, autoradiography, rat, dopamine D2 receptor, dopamine D3 receptor, antipsychotic drugs
INTRODUCTION
In agreement with its high in vitro affinity for both D2 and D3 receptors (Freedman et al, 1994; van Vliet et al, 2000), the agonist positron emission tomography (PET) radiotracer [11C]-(+)-PHNO is thought to label both receptor subtypes in vivo. In both human and baboon, the regional pattern of in vivo [11C]-(+)-PHNO binding is unique among D2/D3 radiotracers, with highest binding in the globus pallidus (GP) followed by ventral striatum (VS), caudate (CAU) and putamen (PUT), and substantia nigra (SN) (Ginovart et al, 2007; Narendran et al, 2006; Willeit et al, 2006). In general agreement with the distribution of D2 and D3 receptors, [11C]-(+)-PHNO binding sites in CAU and PUT are thought to represent primarily D2 receptors, whereas those in GP and SN are thought to be primarily of the D3 receptor type (Ginovart et al, 2007; Narendran et al, 2006; Willeit et al, 2006). The pharmacological dissimilarity between [11C]-(+)-PHNO binding sites in CAU/PUT and GP was first suggested by their anatomical distribution and the slower washout of the radiotracer from GP than from CAU and PUT (Ginovart et al, 2007; Willeit et al, 2006), but has since been supported by pharmacological evidence showing that [11C]-(+)-PHNO binding potential with respect to non-displaceable binding (BPND) in the GP can be blocked in a regionally selective manner by the D3-selective drugs BP897 and SB277011 in baboon (Narendran et al, 2006; Rabiner et al, 2009), and pramipexole (Graff-Guerrero et al, 2009) and ABT-925 (Graff-Guerrero et al, 2008) in human. Further support for the binding of [3H]-(+)-PHNO to both receptor subtypes is provided by mouse experiments showing that D2 receptor knockout abolishes [3H]-(+)-PHNO binding in the striatum (STR) while leaving SB277011-sensitive binding in midbrain and cerebellum lobes 9 and 10 largely intact (Rabiner et al, 2009).
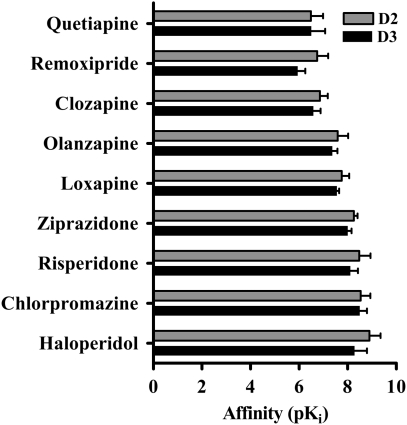
The observation that [11C]-(+)-PHNO labels both D2 and D3 receptors in vivo, coupled with the anatomical separation between regions with primarily D2 (CAU/PUT) or D3 (GP) binding sites, makes [11C]-(+)-PHNO a potentially useful radiotracer for measuring occupancy by drugs that bind to either or both receptor types. Antipsychotic drugs, which have similar affinity for D2 and D3 receptors in vitro (Figure 1), are expected to occupy comparable proportions of brain D2 and D3 receptor populations. Although it is well established that D2 receptor blockade is required for the therapeutic efficacy of these drugs (Farde et al, 1992; Kapur et al, 2000; Kapur et al, 1999; Nordstrom et al, 1993), it has been suggested that the D3 receptor may be at least partially responsible for their clinical effect (Joyce and Millan, 2005; Sokoloff et al, 1990). However, there has traditionally been no way to test the D2 vs D3 binding properties of these drugs in the living brain.
Figure 1.
In vitro affinity (pKi) of various antipsychotic drugs for cloned dopamine D2 and D3 receptors (human and rat). None of the drugs shown here display >fourfold selectivity for D2 over D3 receptors. Data are from the National Institutes of Mental Health (NIMH) Psychoactive Drug Screening Program (PDSP) Ki database located at http://pdsp.cwru.edu/pdsp.asp and references therein. The number of individual values included in the average D2 and D3 affinities, respectively, are quetiapine 17, 10; remoxipride 8, 7; clozapine 33, 20; olanzapine 19, 12; loxapine 13, 4; ziprazidone 7, 5; risperidone 24, 13; chlorpromazine 19, 10; and haloperidol 39, 22. Radioligands used in determination of Ki values were [3H]-raclopride, [3H]-nemonapride, [125I]-iodosulpiride, [3H]-spiperone, or [3H]-N-methylspiperone.
A recent [11C]-(+)-PHNO PET study in schizophrenic patients conducted at our center revealed that chronic treatment with the antipsychotic drugs olanzapine, clozapine, or risperidone, produced high levels of receptor occupancy in D2-rich CAU, PUT, and VS, but no receptor occupancy in D3-rich GP (Graff-Guerrero et al, 2009). The purpose of this study was to clarify this discrepancy by comparing ex vivo and in vitro D2 and D3 occupancy by olanzapine, clozapine, and risperidone, as well as the typical antipsychotic haloperidol, using [3H]-(+)-PHNO autoradiography in rat brain. We first describe the ex vivo and in vitro distribution of [3H]-(+)-PHNO binding in rat brain, and use the D3-selective drug SB277011 to estimate the D3 receptor contribution to the binding signal in each of the major dopaminergic regions of interest (ROIs). We then use ex vivo and in vitro [3H]-(+)-PHNO autoradiography to examine the regional receptor occupancy produced by each of the above antipsychotic drugs to clarify their D2 vs D3 binding properties.
MATERIALS AND METHODS
General
Male Sprague–Dawley rats weighing 250–275 g at the beginning of the study were housed two per cage under a 12 h light 12 h dark photocycle and were allowed unlimited access to food and water. All rats were housed in the animal facility at the Centre for Addiction and Mental Health for 1 week before use in experiments. [3H]-(+)-PHNO (two batches, 50 and 78 Ci/mmol) was purchased from PerkinElmer Life Sciences (Boston, MA). Olanzapine, risperidone, haloperidol, and clozapine were purchased from Bosche Scientific (New Brunswick, NJ), and SB277011 was a gift from Dr Bernard Le Foll at the Centre for Addiction and Mental Health. All animal experiments were conducted with approval of the Animal Ethics Committee at the Centre for Addiction and Mental Health and in accordance with the Canadian Council on Animal Care.
Drug Treatments
Antipsychotic drugs were administered chronically so as to mimic the conditions of the human PET experiment from which this study arose. High-dose chronic olanzapine (7.5 mg/kg/day; 3, 7, 14, or 21 days; n=4 per group), risperidone (4.2 mg/kg/day; 3 or 21 days; n=5 per group), haloperidol (0.3 mg/kg/day; 3 or 21 days; n=5 per group), or chronic vehicle (1% acetic acid in saline; 21 days; n=18) was administered through implanted osmotic minipumps. Antipsychotic doses were chosen in an attempt to target 90% striatal D2/D3 receptor occupancy based on the results of earlier reports (Kapur et al, 2003; McCormick et al, 2008). The concentration of antipsychotic drugs required to deliver the above doses was calculated using the predicted rat weight at the midpoint of the treatment period (assuming 7 g daily weight gain) and the known delivery rate of the osmotic minipumps (2.5 and 5 μl/h for 2ML4 and 2ML2 models, respectively). Osmotic minipumps were filled (2 ml total volume) with either vehicle or antipsychotic drug solution and implanted subcutaneously in the animals' upper back under 2.5% isoflurane anesthesia.
The combination of low potency and low solubility of clozapine relative to olanzapine, risperidone, and haloperidol limited the maximum dose that could be delivered through osmotic minipump (Kapur et al, 2003). Therefore, to achieve striatal D2/D3 receptor occupancy comparable with that of the other antipsychotics, we chose to administer clozapine acutely at a dose of 60 mg/kg, s.c. (n=12). Acute vehicle-treated rats (2% acetic acid in saline, n=6) were used as a control group.
To estimate the relative contribution of D2 vs D3 receptors to regional [3H]-(+)-PHNO binding, rats were treated acutely with the D3-selective drug SB277011 at a D3-selective dose (McCormick et al, 2008; Rabiner et al, 2009) of 10 mg/kg i.p. or with vehicle (30% β-cyclodextrin in saline), during day 7 of chronic olanzapine or chronic vehicle treatment (1% acetic acid in saline, n=5 per group).
Ex Vivo [3H]-(+)-PHNO Autoradiography
Rats were injected i.v. with ∼2 nmol of [3H]-(+)-PHNO through the tail vein either during chronic olanzapine, risperidone, or haloperidol treatment, or 30 or 60 min after acute treatment with clozapine or SB277011, respectively, and killed 60 min later by decapitation. A killing time of 60 min post-radiotracer injection was chosen because it allows for substantial clearance of non-displaceable radiotracer binding, resulting in good signal contrast between ROIs and the cerebellar cortex (CER) reference region. A blood sample was collected from the trunk of each rat for analysis of plasma antipsychotic concentrations. Whole brains were excised, rinsed in saline, and frozen at −80°C until further use. Plasma samples were sent to the clinical laboratory at the Centre for Addiction and Mental Health for analysis of antipsychotic concentrations.
Twenty μm brain sections were cut at −10°C on a Hacker Bright cryostat and thaw mounted onto microscope slides. For each brain, duplicate tissue sections were collected at the following anterior–posterior coordinates (anterior to bregma) (Paxinos and Watson, 1986) and included the following ROIs: (1) 1.6 mm, cerebral cortex (CRT), STR, nucleus accumbens (NACC), islands of Calleja (ICJ); (2) −0.3 mm, STR, ventral pallidum (VP); (3) −5.2 mm, SN; (4) −12.7 mm, CER, cerebellar lobes 9 and 10 (LOB). The brain sections were dried in a dessicator containing Drierite for at least 24 h at 4°C and exposed to Fujifilm tritium-sensitive imaging plates (model BAS TR2025) for 4 weeks. Regional tissue radioactivity was determined using a BAS 5000 Fujifilm image plate reader. Ex vivo [3H]-(+)-PHNO binding was quantified using radioactivity in the ROI and CER (as an estimate of non-displaceable binding), as the specific binding ratio, SBR=(ROI-CER)/CER. For each ROI, occupancy was calculated as:
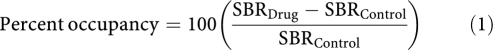 |
where SBRDrug is the SBR in an individual drug-treated animal and SBRControl is the average SBR in the control vehicle-treated group.
In Vitro [3H]-(+)-PHNO Autoradiography
Five rats were killed by decapitation and their brains removed, frozen on dry ice, and stored at −80°C until further use. For each anterior–posterior brain coordinate given above, 16 adjacent 10 μm brain sections were collected. The tissue sections were thaw mounted such that slide #1 contained the most anterior tissue section cut at each brain coordinate, slide #2 contained the next most posterior section from each coordinate, slide #3, the third most posterior, etc. For each brain, all of the 16 resulting sequential slides, therefore, contained all ROIs, but at progressively more posterior positions. In total, 80 slides were produced (5 brains, 16 slides per brain). Each of these 80 slides was then randomly assigned to one of 16 treatment groups. The treatment groups were as follows: control; 47, 180, or 380 nM olanzapine; 6, 23, or 48 nM risperidone; 4, 14, or 30 nM haloperidol; 330, 1270, or 2680 nM clozapine; 26, 100, or 210 nM SB277011. The concentrations of each drug were chosen to produce ∼70, 90, and 95% receptor occupancy (D2 for antipsychotic drugs, D3 for SB277011) based on a pKi value for each drug obtained by averaging all of the appropriate entries listed in the NIMH Psychoactive Drug Screening Program Ki database (available at http://pdsp.med.unc.edu/kidb.php) and extrapolating using a sigmoidal dose–response relationship with Hill slope=1.
Slides were incubated for 2 h at room temperature in buffer (50 mM Tris HCl, 1 mM EDTA, 1.5 mM CaCl2, 4 mM MgCl2, and NaCl either 0.6 nM) containing either 0.6 nM [3H]-(+)-PHNO alone (control) or with one of the above drug concentrations. After incubation, the slides were rinsed in ice-cold buffer (3 × 5 min), dipped for 10 s in ice-cold deionized water, and dried under a stream of room temperature air. The slides were left to dry further overnight and exposed to tritium-sensitive image plates for 3 weeks in the presence of calibrated methacrylate radioactivity standards. The image plates were scanned and regional [3H]-(+)-PHNO specific binding (SB) was calculated as the total binding in the ROI minus the average total binding in CER (as an estimate of non-displaceable binding). Effort was made to draw ROIs of the same shape, size, and location as in the ex vivo autoradiography experiments. However, in vitro [3H]-(+)-PHNO binding in the ICJ could not be reliably distinguished from binding seen in the olfactory tubercle. Therefore, in the in vitro condition, no ROIs were drawn for ICJ. For each ROI, drug occupancy was calculated in the same way as for the ex vivo experiments [ie with the substitution of SB for SBR in equation (1)].
Statistical Analysis
SBR, SB, and percent occupancy are expressed as mean±SD. For statistical comparisons, means were considered significantly different when p<0.05. Comparison of ex vivo [3H]-(+)-PHNO SBR between individual vehicle-treated groups or between treatment durations (eg 3 days vs 7 days olanzapine treatment) was performed by ANOVA followed by post hoc Bonferroni's multiple comparison test. Comparison of average [3H]-(+)-PHNO binding (SBR for ex vivo and SB for in vitro experiments) between drug-treated and control groups was performed by ANOVA followed by Dunnett's multiple comparison test. Occupancy was considered significant when the SBR or SB in the drug-treated group was significantly different from that in the respective control group. Evaluation of occupancy differences between ex vivo and in vitro conditions was performed by ANOVA followed by Bonferroni's multiple comparison test.
RESULTS
Ex Vivo [3H]-(+)-PHNO Autoradiography
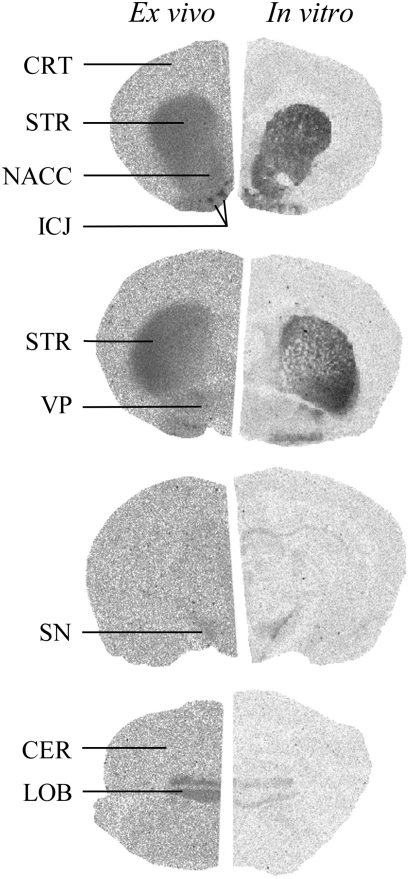
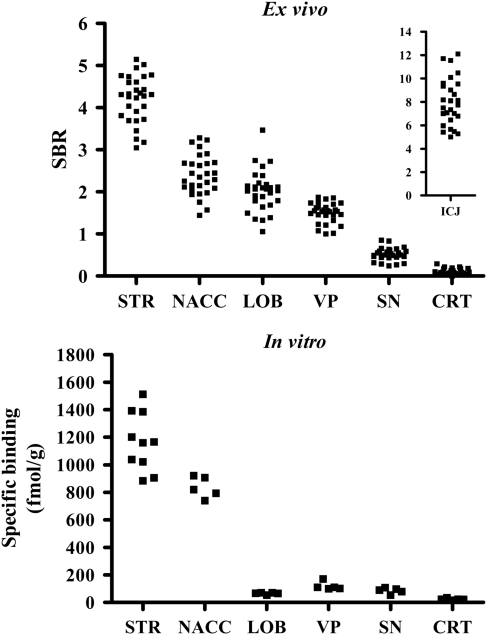
Typical ex vivo and in vitro [3H]-(+)-PHNO autoradiographs are shown in Figure 2 (left side). [3H]-(+)-PHNO SBRs were very similar between individual vehicle-treated groups and the data were, therefore, pooled to produce a single control group. Regional ex vivo [3H]-(+)-PHNO SBR in the resulting pooled vehicle-treated group is shown in Figure 3 (top panel). The highest SBR was seen in the ICJ (8.0±2.0) followed by STR (4.4±1.0), NACC (2.5±0.7), LOB (2.0±0.5), VP (1.6±0.4), and SN (0.5±0.2). The lowest SBR was seen in CRT (0.1±0.1).
Figure 2.
Typical control [3H]-(+)-PHNO autoradiographs in rat brain measured ex vivo (left) and in vitro (right). The anterior–posterior coordinate (anterior to bregma) is shown to the right of each autoradiograph. Regions of interest at each coordinate are 1.60 mm, cerebral cortex (CRT), striatum (STR), nucleus accumbens (NACC), islands of Calleja (ICJ); −0.3 mm, STR, ventral pallidum (VP); −5.2 mm, substantia nigra (SN); −12.7 mm, cerebellar cortex (CER), cerebellar lobes 9 and 10 (LOB).
Figure 3.
Regional [3H]-(+)-PHNO binding in striatum (STR), nucleus accumbens (NACC), cerebellar lobes 9 and 10 (LOB), substantia nigra (SN), and cerebral cortex (CRT), measured ex vivo in vehicle-treated rats (top) and in vitro in control brain sections (bottom). To facilitate direct visual comparison between ex vivo and in vitro [3H]-(+)-PHNO binding, only those brain regions examined in both the ex vivo and in vitro conditions are shown on the main horizontal axis. Ex vivo binding in islands of Calleja (ICJ) are shown in the inset of the top graph (note difference in scale). Note also that in vitro STR binding, as opposed to that in the other regions, was measured in two tissue sections per slide resulting in a total of 10 separate measurements.
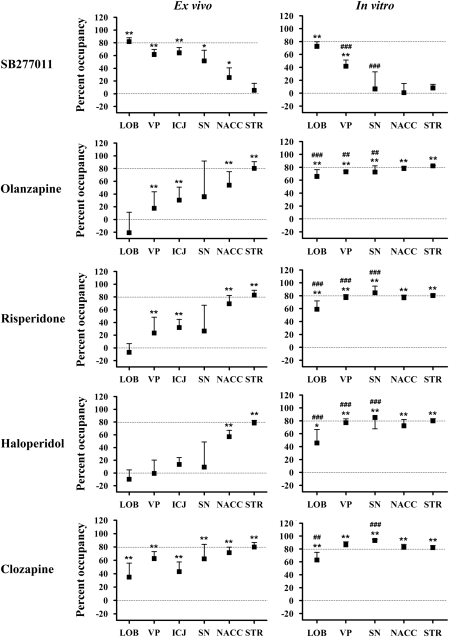
For rats treated with chronic risperidone or haloperidol, regional occupancy and plasma drug concentrations (Table 1) were similar for all treatment durations (p>0.05). We, therefore, chose to pool the data to produce a single group for each drug to increase statistical power. During chronic olanzapine treatment, plasma olanzapine concentration decreased over time as has been reported earlier (van der Zwaal et al, 2008) (Table 1), resulting in slightly reduced STR occupancy in the 14- and 21-day treatment groups (p<0.01, 74±7 and 71±8%, respectively, vs 88±7% in the 3-day treatment group). No other differences in regional occupancy were seen between olanzapine-treated groups. Consequently, we chose to also pool the chronic olanzapine data, with the understanding that this pooled group underestimates the occupancy in STR by ∼7% relative to the 3-day treatment group. Figure 4 shows ex vivo occupancy (left panels) in comparison with that measured in vitro (right panels). Treatment with chronic 7.5 mg/kg/day olanzapine plus 10 mg/kg acute SB277011 resulted in extensive receptor occupancy across all brain regions: STR, 90±3% NACC, 80±5% 83±5% SN, 89±10% ICJ, 68±14% LOB, 82±6%. Average ex vivo occupancy±SD can also be found in Supplementary Table 2.
Table 1. Antipsychotic Drug Concentrations in Blood Plasma.
| Duration (days) | Olanzapine (nM) | Haloperidol (nM) | Risperidone (nM) | Clozapine (μM) |
|---|---|---|---|---|
| Acute | — | — | — | 5.6±2.2 |
| 3 | 244±52 | 7.6±1.3 | 152±47 | — |
| 7 | 190±27 | — | — | — |
| 14 | 153±33** | — | — | — |
| 21 | 134±26** | 7.4±1.7 | 117±26 | — |
**p<0.01 vs 3-day treatment group. Dunnett's multiple comparison test.
Figure 4.
Ex vivo (left) and in vitro (right) SB277011 and antipsychotic occupancy in cerebellar lobes 9 and 10 (LOB), ventral pallidum (VP), islands of Calleja (ICJ, ex vivo condition only), nucleus accumbens (NACC), and striatum (STR). Dashed lines indicate the 0 and 80% occupancy levels. Note the similarity in STR occupancy both between antipsychotic drugs and between the ex vivo and in vitro conditions. Note also the similarity in LOB occupancy for SB277011 ex vivo vs in vitro. *p<0.05, **p<0.001 significant occupancy (ie significant reduction in [3H]-(+)-PHNO binding relative to control group); ##p<0.01, ### p<0.001 occupancy significantly different from ex vivo condition.
In Vitro [3H]-(+)-PHNO Autoradiography
Typical in vitro [3H]-(+)-PHNO autoradiographs are shown in Figure 2 (right panels) and regional in vitro SB is shown in Figure 3 (bottom panel). [3H]-(+)-PHNO SB was highest in STR (1164±129 fmol/g), followed by NACC (836±77 fmol/g), VP (118±29 fmol/g), SN (85±20 fmol/g), LOB (65±7 fmol/g), and CRT (23±7 fmol/g). Total binding in CER (used as an estimate of non-displaceable binding) was 29±7 fmol/g.
Figure 4 (right panels) shows the regional occupancy produced by the highest concentration SB277011 and each of the antipsychotic drugs in comparison with the occupancy observed ex vivo. The occupancy produced by the remaining two antipsychotic concentrations can be found in Supplementary Table 3.
DISCUSSION
Our earlier ex vivo rat experiments with [11C]-(+)-PHNO using brain dissection (McCormick et al, 2009; McCormick et al, 2008; Wilson et al, 2005) allowed the examination of [11C]-(+)-PHNO binding in large brain regions such as STR, CRT, and whole cerebellum, but not in smaller regions such as NACC, VP, and LOB. The ex vivo SBRs measured here in STR (4.4±1.0) and CRT (0.1±0.1) of vehicle-treated rats are in agreement with our earlier ex vivo measurements (McCormick et al, 2009; McCormick et al, 2008; Wilson et al, 2005). The distribution of [3H]-(+)-PHNO SBR in both striatal and non-striatal regions (Figure 3, top panel) is consistent with the known distribution of D2/D3 receptors in rat brain (Landwehrmeyer et al, 1993; Levesque et al, 1992). Our in vitro [3H]-(+)-PHNO measurements (Figure 3, bottom panel) are also consistent with the D2/D3 receptor binding pattern described in the above-cited reports and with earlier in vitro [3H]-(+)-PHNO autoradiographic data reported by Nobrega and Seeman (1994) (although the authors did not describe binding in LOB).
However, the ex vivo and in vitro distribution patterns of [3H]-(+)-PHNO reported here are quantitatively very different from one another. Visual inspection of Figure 3 suggests that these differences are due to reduced binding in D3-rich areas. Although the SBR (ex vivo) and SB (in vitro) cannot be directly compared, quantitative differences between ex vivo and in vitro [3H]-(+)-PHNO binding can be illustrated by normalization of regional binding to that in STR, which, as discussed below, is exclusively due to D2 binding and should thus be unaffected by changes in D3 binding. Relative to STR, ex vivo binding in LOB (45% of SBR in STR), VP (36% of STR), and SN (11% of STR) is significantly greater than seen in vitro (6, 10, and 7% of STR, respectively; t test, p<0.001 for LOB and VP, p<0.05 for SN), whereas no difference is seen in NACC (p>0.05). The decrease in LOB, VP, and SN binding relative to STR is also reflected in the distinctive change in regional rank order of [3H]-(+)-PHNO binding between the ex vivo (STR>NACC>LOB>VP>SN>CRT) and in vitro (STR>NACC>VP∼SN∼LOB>CRT) conditions, which would not be expected if the ex vivo vs in vitro differences in baseline binding were due to altered D2 receptor binding. Unfortunately, because ROIs could not be reliably drawn around ICJ in the in vitro condition, a direct comparison between ex vivo and in vitro binding in this region was not possible. However, the fact that the ICJ were more easily distinguishable ex vivo suggests an increased ex vivo vs in vitro signal in this region similar to that seen in LOB and VP.
Although the anatomical pattern of D2-like receptor binding is well established (Bancroft et al, 1998; Levant, 1998), the ratio of D2 to D3 receptor binding sites within individual brain region is not precisely known. Pretreatment with the ∼100-fold D3-selective drug SB277011 (Newman et al, 2005) resulted in >50% occupancy in LOB, ICJ, VP, and SN (Table 1), indicating that the D3 receptor is the major contributor to [3H]-(+)-PHNO SBR in these regions. No occupancy was seen in STR showing, as we have reported earlier (McCormick et al, 2008), that ex vivo striatal [3H]-(+)-PHNO SBR is exclusively due to D2 receptor binding in rat brain. Combined treatment with SB277011 and chronic olanzapine produce similarly large levels of occupancy in STR, NACC, SN, VP, and ICJ (∼80%), but failed to increase occupancy in LOB above that seen after SB277011 treatment alone (Table 1). Thus, LOB seems to be the only region in which [3H]-(+)-PHNO SBR is exclusively due to D3 receptor binding. Assuming that the SB277011-induced D3 receptor occupancy is similar across brain regions (∼80%), the percentage D3 contribution to [3H]-(+)-PHNO can be calculated as the ratio of SB277011 occupancy in the ROI to that in LOB. Thus, the D3 contribution to ex vivo SBR is ∼30% in NACC, ∼63% in SN, and ∼75–80% in VP and ICJ. It is likely, however, that the D3 contribution to [3H]-(+)-PHNO binding in ICJ is larger than indicated here, as the ROIs drawn around these small structures probably included some spillover from the D2-rich olfactory tubercle. The anatomical separation and relatively large size of the other regions are more amenable to accurate ROI delineation, and our reported D3 contribution in these regions is more certain.
In vitro, the same regional trend in SB277011 occupancy was seen (Figure 4). However, in vitro, SB277011 had a smaller effect on [3H]-(+)-PHNO binding in VP and SN than seen ex vivo. In addition, SB277011 had no measurable effect on in vitro [3H]-(+)-PHNO binding in the NACC, in contrast to the significant 25% NACC occupancy seen ex vivo. The estimated D3 contribution to in vitro [3H]-(+)-PHNO SB, derived from the regional effect of SB277011 treatment is ∼10% in SN and ∼57% in VP, whereas the D3 receptor does not seem to contribute to in vitro binding in NACC or STR. Thus, the D3 receptor seems to contribute more heavily to ex vivo than to in vitro [3H]-(+)-PHNO binding. This conclusion is also supported by the increased ex vivo vs in vitro [3H]-(+)-PHNO SBR in LOB, VP, and SN regions that have the highest D3 contribution. Although the mechanism of this ex vivo vs in vitro difference is unclear, possible contributing factors could include differences in levels of endogenous dopamine or the proportion of receptors in the high-affinity state. Under basal conditions, the D3 receptor has been reported to be extensively occupied by dopamine, preventing in vitro D3 binding of [125I]-iodosulpiride (Schotte et al, 1992) and [3H]-7-OH-DPAT (Schotte et al, 1996a). However, endogenous dopamine does not seem to be relevant to the current data, as the extensive dilution of dopamine into the in vitro assay volume (12 ml volume over 60 min) would be expected to increase, rather than decrease, [3H]-(+)-PHNO binding in the VP (relative to the ex vivo condition), and to have little or no effect on binding in LOB, which lacks dopaminergic innervation (Beckstead et al, 1979; Swanson, 1982). It is possible that the D3 receptor high-affinity state is selectively disrupted in vitro, resulting in lower D3 receptor [3H]-(+)-PHNO binding. However, this explanation also seems unlikely given that the D3 receptor-G protein complex responsible for high-affinity agonist binding is thought to be more stable than that of the D2 receptor (Seabrook et al, 1992; Sokoloff et al, 1990). Many other potential differences exist between our in vitro and ex vivo conditions (eg disruption of ionic gradients, membrane voltage and protein kinase/phosphatase cycles) and further investigation would be necessary to unravel their relevance to the current data. Fortunately, both ex vivo and in vitro, the pure D3 and D2 [3H]-(+)-PHNO binding signal in LOB and STR, respectively, makes these regions ideally suited for the examination of drug occupancy at these receptor types. However, in other regions, the D3 vs D2 contribution to [3H]-(+)-PHNO binding depends, at least under our experimental conditions, on whether the measurements are made in vitro or ex vivo and drug occupancy in these regions must be interpreted accordingly.
The antipsychotic drugs olanzapine (7.5 mg/kg/day), clozapine (60 mg/kg), risperidone (4.2 mg/kg/day), and haloperidol (0.3 mg/kg/day) occupied a large proportion (∼80%) of ex vivo [3H]-(+)-PHNO binding sites in STR, in agreement with their well-known D2 antagonism. However, despite their reportedly similar affinity for D2 and D3 receptors, olanzapine, risperidone, and haloperidol did not measurably occupy D3 receptors in LOB. As well, clozapine occupancy in LOB was lowest among the ROIs examined. Ex vivo antipsychotic occupancy across all regions followed a trend that was the inverse of that seen for the D3-selective drug SB277011 (Figure 3). Using in vitro [3H]-(+)-PHNO autoradiography, we confirmed that all of these drugs produce similar occupancy in STR, NACC, SN, VP, and LOB. These data provide strong pharmacological evidence that these drugs are in fact D2-selective ex vivo—a surprising finding given their in vitro pharmacological profile. Similar results have been reported by Schotte et al (1996a). Their autoradiographic experiments involved in vivo antipsychotic treatment followed by measurement of receptor occupancy using in vitro [3H]-7-OH-DPAT (D3 receptors in ICJ) or [125I]-iodosulpiride (nominally D2 receptors in NACC). They report that when administered in vivo, clozapine, olanzapine, risperidone, and haloperidol had ratios of D2 to D3 potency 2–10 times higher than when determined using in vitro competitive binding experiments, and argued that this effect was due to the in vivo inhibitory influence of endogenous dopamine on antipsychotic D3 receptor binding. However, this explanation cannot account for our observation that the antipsychotic drugs have lower potency for D3 receptors in LOB, which lacks dopaminergic innervation (Beckstead et al, 1979; Swanson, 1982), than for D2 receptors in STR, nor can it explain the inverse regional trend between antipsychotic occupancy and SB277011 occupancy. Our data are explained more completely by a model in which the in vivo D2/D3-affinity ratio for antipsychotics is greater than predicted by in vitro measurements.
Clinically, most antipsychotic drugs produce therapeutic effects at doses producing 65–80% D2 receptor occupancy (Farde et al, 1992; Kapur et al, 2000; Kapur et al, 1999; Nordstrom et al, 1993). The levels of D2 occupancy in this study are on the high end of this occupancy range (∼80%), suggesting that at clinically relevant doses, the therapeutic effects of olanzapine, risperidone, and haloperidol are not attributable to D3 receptor blockade. Clozapine was the only antipsychotic to produce significant occupancy in LOB, suggesting that its selectivity for D2 over D3 receptors in living brain is less than that of the other antipsychotics. This does not seem to be a consequence of the fact that clozapine was administered acutely rather than chronically, as the duration of treatment had little effect on regional occupancy produced by the other antipsychotics. Nevertheless, the clinical importance of the D3 receptor to clozapine's clinical effect is likely small, as the D2 occupancy of therapeutic clozapine (20–60%) (Kapur et al, 1999; Nordstrom et al, 1995) is typically less than for the other antipsychotic, indicating that clozapine's D3 receptor occupancy in the clinical setting is likely also much lower than the 35% occupancy seen here. Indeed the recent PET study by our group in schizophrenic patients found that chronic clozapine treatment, while producing ∼50% occupancy in STR, did not reduce [11C]-(+)-PHNO binding in the GP (Graff-Guerrero et al, 2009).
The current data point to the importance of in vivo or ex vivo experiments in elucidating the mechanism of action of therapeutic agents within the brain. As we show here, in vitro pharmacological measurement, although invaluable in drug and radiotracer development, can sometimes lead to erroneous assumptions regarding in vivo drug action. Ex vivo [3H]-(+)-PHNO autoradiography is a powerful technique for determining, with high anatomical resolution, the interaction of drugs with both D2 (STR) and D3 (LOB) receptors in the living brain. In particular, ex vivo autoradiography has two major strengths for preclinical drug evaluation in rat or other small research animals. First, unlike small animal PET experiments, ex vivo autoradiography does not require the use of anesthesia, which can confound interpretation of radiotracer binding results (Tsukada et al, 2000; Tsukada et al, 2008). Second, the spatial resolution of autoradiography is very high in comparison with either ex vivo dissection experiments or in vivo small animal PET experiments. The major limitation of this technique, however, is that, barring the use of prohibitively large numbers of animals, it is not easily amenable to generation of multiple time point data that can be analyzed using quantitative radiotracer pharmacokinetic modeling techniques. For dopamine receptor radiotracers, pharmacokinetic modeling is typically used to derive BPND. This equilibrium parameter, which expresses the ratio of specific to non-displaceable radiotracer distribution volumes, is commonly used as an index of receptor availability. The ratio methods, such as that used to generate our ex vivo SBR (equivalent to STR/CER—1), are common non-kinetic methods that use a non-equilibrium ratio of ROI to reference tissue radiotracer concentrations to estimate BPND. The two most common ratio methods are the transient equilibrium and late time point methods. The transient equilibrium method uses the ratio of ROI to reference tissue radiotracer concentrations at a time point corresponding to peak SB (so-called transient equilibrium) (Carson et al, 1993; Ginovart et al, 2001; Ito et al, 1998). At this particular time point, the SBR is mathematically equivalent to BPND. The late time point method, which is used in this study, instead uses the ratio of ROI to reference tissue concentrations at some time after transient equilibrium has been reached (∼30 min for [11C]-(+)-PHNO in rat) (Ginovart et al, 2006; Ginovart et al, 2001; Ito et al, 1998; Lammertsma et al, 1996). Relative to the transient equilibrium method (Carson et al, 1993; Ginovart et al, 2001; Ito et al, 1998), the late time point method can result in overestimate of BPND (Ito et al, 1998; Lammertsma et al, 1996), the magnitude of this bias related to the regional kinetics of the radiotracer. This may have bearing on interpretation of the current results. In human, the kinetics of [11C]-(+)-PHNO binding in the D2-rich dorsal STR are very different from those in the D3-rich GP (Ginovart et al, 2007). If such regional kinetic differences were to exist in rat, they could result in greater overestimate of SBR in D3-rich vs D2-rich regions, possibly helping to explain some of the quantitative difference between our ex vivo and in vitro results. Inspection of published human [11C]-(+)-PHNO PET kinetic data (Ginovart et al, 2007), however, suggests exactly the opposite; that an SBR calculated using a late time point (60 min.) overestimates BPND to a larger extent in D2-rich CAU–PUT than in the D3-rich GP. However, in the absence of detailed regional [3H]-(+)-PHNO kinetic data in rat, it is impossible to make firm statements regarding the influence of regional kinetics on the current data.
In general, ratio methods can be expected to result in reliable quantification of inter-group differences in radiotracer binding and receptor occupancy provided that no large differences in radiotracer delivery exist between groups (Fujita, 2001; Ginovart et al, 2001; Ito et al, 1998). Such delivery differences, typically attributable to inter-group differences in cerebral blood flow, can alter ROI-to-reference tissue ratios and thus confound interpretation of group differences in radiotracer binding. To the authors' knowledge, antipsychotic drugs are not known to cause changes in cerebral blood flow large enough to explain our results (Yildiz et al, 2000). Further, supporting the validity of the ratio method used here is the general agreement between the current findings and the results of our earlier [11C]-(+)-PHNO PET study of antipsychotic occupancy in human subjects (Graff-Guerrero et al, 2009) using the more kinetically robust simplified reference tissue model, which has been validated for [11C]-(+)-PHNO (Ginovart et al, 2007). Nevertheless, given the methodological limitations of our late time point ratio method (eg sensitivity to blood flow changes and regional kinetic differences), the current findings should be confirmed using multiple time point ex vivo experiments or in vivo using small animal PET.
Another methodological limitation worth considering is the fact that receptor binding in our ex vivo experiments occurred at body temperature of ∼37°C, whereas our in vitro binding studies were conducted at room temperature (∼20°C). The ex vivo vs in vitro differences in antipsychotic occupancy observed here could theoretically be explained by a selective decrease in antipsychotic affinity for the D3 receptor with increasing temperature (ie greater D2 receptor selectivity in our ex vivo experiments). However, close examination of the in vitro literature provides no support for this hypothesis, as all of the antipsychotics examined here also have similar in vitro D2 and D3 affinity at 37°C, with none displaying >4.4-fold selectivity for either receptor subtype (Kongsamut et al, 1996; Leopoldo et al, 2002; Schotte et al, 1996b; Tang et al, 1994).
In addressing the limitations of our study, it is worthwhile also to consider whether our relatively large injected [3H]-(+)-PHNO dose (∼5.7 nmol/kg vs 0.3–3 nmol/kg in our earlier studies) falls within the tracer dose range (commonly defined by receptor occupancy not exceeding 5%). Within this dose range, our ex vivo SBR (a saturable signal) should be relatively insensitive to changes in injected mass, as opposed, for example, to a dose range surrounding the EC50 in which small changes in injected dose would result in large reductions in the SBR. Our striatal SBR of 4.5 is identical to that in our earlier reports, which used 2- to 10-fold lower injected mass (McCormick et al, 2009; McCormick et al, 2008; and unpublished data), suggesting that the tracer dose range for the D2 receptor extends at least as high as the current injected dose of 5.7 nmol/kg. Second, exceeding the tracer dose range would result in an underestimate of antipsychotic D2 receptor occupancy. We find, however, that our current striatal clozapine D2 occupancy (80±7%) is very similar to that of our earlier report (80±8%) in which a twofold lower injected [3H]-(+)-PHNO dose was used (McCormick et al, 2008). [3H]-(+)-PHNO has been reported to have 30- to 45-fold higher in vitro affinity for the D3 receptor than the D2 receptor (Freedman et al, 1994; van Vliet et al, 2000) suggesting that a lower injected dose may be required to fulfill tracer dose conditions for D3 compared with D2. In the absence of reports comparing the in vivo affinity of [3H]-(+)-PHNO, it remains possible that the tracer dose range has been exceeded with respect to the D3 receptor, consequently presenting the possibility of underestimated drug D3 receptor occupancy. However, it seems unlikely that this alone could account for such a large discrepancy between our ex vivo D2 and D3 occupancies (80 vs 0%), especially given the general agreement of our results with those of our recent PET study in human subjects using an injected [11C]-(+)-PHNO dose of about 0.1 nmol/kg, ∼60-fold lower than in this study (Graff-Guerrero et al, 2009). The greater in vitro affinity of (+)-PHNO for D3 vs D2 receptors implies that exceeding tracer concentration in our in vitro experiments would result in a greater underestimate of drug occupancy at D3 vs D2 receptors. Thus, if we have exceeded tracer dose in vitro, the ‘true' antipsychotic D3 receptors occupancy will be numerically closer to, or even exceeding, that of D2 receptors. Thus, we do not feel that this would change the major conclusion of our paper (ie there is a major discrepancy between ex vivo and in vitro measures of antipsychotic occupancy).
In conclusion, this study shows that the antipsychotic drugs olanzapine, clozapine, risperidone, and haloperidol do not occupy the D3 receptor as measured ex vivo. This is in contrast to our in vitro measurements showing similar occupancy at both receptor types, and to the large body of literature indicating similar in vitro affinity of these drugs for D2 and D3 receptors. These findings corroborate and clarify the results of a recent human [11C]-(+)-PHNO PET study conducted at our center showing that, despite significant occupancy in STR, olanzapine, clozapine, and risperidone did not reduce [11C]-(+)-PHNO D3 receptor binding in GP. Furthermore, the data suggest that the therapeutic effects of olanzapine, clozapine, risperidone, and haloperidol are not attributable to blockade of the D3 receptor.
Acknowledgments
We thank Drs Gary Remington, Jeffrey Meyer, and Neil Vasdev for valuable discussion, and Dr Bernard Le Foll for his generous donation of the D3-selective drug SB277011. This work was financially supported by the Canadian Institutes of Health Research (grant numbers MOP 74702 and MOP 44051).
Patrick N McCormick, Alan A Wilson, José Nobrega, and Roger Raymond have no conflicts of interest to disclose. Ariel Graff-Guerrero has received professional services compensation from Abbott Laboratories, grant support from Janssen, and speaker compensation from Eli Lilly. Shitij Kapur has received grant support from AstraZeneca, Bristol-Myers Squibb, and Glaxo Smith Kline, and has acted as consultant/scientific advisor or received speaker compensation from AstraZeneca, Bioline, Bristol-Meyers Squibb, Eli Lilly, Janssen, Lundbeck, Otsuka, Organon, Pfizer, Servier, and Solvay Wyeth.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Bancroft GN, Morgan KA, Flietstra RJ, Levant B. Binding of [3H]PD 128907, a putatively selective ligand for the D3 dopamine receptor, in rat brain: a receptor binding and quantitative autoradiographic study. Neuropsychopharmacology. 1998;18:305–316. doi: 10.1016/S0893-133X(97)00162-0. [DOI] [PubMed] [Google Scholar]
- Beckstead RM, Domesick VB, Nauta WJ. Efferent connections of the substantia nigra and ventral tegmental area in the rat. Brain Res. 1979;175:191–217. doi: 10.1016/0006-8993(79)91001-1. [DOI] [PubMed] [Google Scholar]
- Carson RE, Channing MA, Blasberg RG, Dunn BB, Cohen RM, Rice KC, et al. Comparison of bolus and infusion methods for receptor quantitation: application to [18F]cyclofoxy and positron emission tomography. J Cereb Blood Flow Metab. 1993;13:24–42. doi: 10.1038/jcbfm.1993.6. [DOI] [PubMed] [Google Scholar]
- Farde L, Nordstrom AL, Wiesel FA, Pauli S, Halldin C, Sedvall G. Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side effects. Arch Gen Psychiatry. 1992;49:538–544. doi: 10.1001/archpsyc.1992.01820070032005. [DOI] [PubMed] [Google Scholar]
- Freedman SB, Patel S, Marwood R, Emms F, Seabrook GR, Knowles MR, et al. Expression and pharmacological characterization of the human D3 dopamine receptor. J Pharmacol Exp Ther. 1994;268:417–426. [PubMed] [Google Scholar]
- Fujita M. In vivo receptor imaging with PET and SPET-pitfalls in quantification. Int Rev Psychiatry. 2001;13:34–39. [Google Scholar]
- Ginovart N, Galineau L, Willeit M, Mizrahi R, Bloomfield PM, Seeman P, et al. Binding characteristics and sensitivity to endogenous dopamine of [11C]-(+)-PHNO, a new agonist radiotracer for imaging the high-affinity state of D2 receptors in vivo using positron emission tomography. J Neurochem. 2006;97:1089–1103. doi: 10.1111/j.1471-4159.2006.03840.x. [DOI] [PubMed] [Google Scholar]
- Ginovart N, Willeit M, Rusjan P, Graff A, Bloomfield PM, Houle S, et al. Positron emission tomography quantification of [11C]-(+)-PHNO binding in the human brain. J Cereb Blood Flow Metab. 2007;27:857–871. doi: 10.1038/sj.jcbfm.9600411. [DOI] [PubMed] [Google Scholar]
- Ginovart N, Wilson AA, Meyer JH, Hussey D, Houle S. Positron emission tomography quantification of [11C]-DASB binding to the human serotonin transporter: modeling strategies. J Cereb Blood Flow Metab. 2001;21:1342–1353. doi: 10.1097/00004647-200111000-00010. [DOI] [PubMed] [Google Scholar]
- Graff-Guerrero A, Abi-Saab W, Redden L, Katz D, Houle S, O'Neill A, et al. First demonstration of D3 occupancy in humans: blockade of [11C]-(+)-PHNO PET by ABT-92. Int J Neuropsychopharmacol. 2008;11 (Suppl. S1:223. [Google Scholar]
- Graff-Guerrero A, Mamo D, Shammi CM, Mizrahi R, Marcon H, Barsoum P, et al. The effect of antipsychotics on the high-affinity state of D2 and D3 receptors: a positron emission tomography study with [11C]-(+)-PHNO. Arch Gen Psychiatry. 2009;66:606–615. doi: 10.1001/archgenpsychiatry.2009.43. [DOI] [PubMed] [Google Scholar]
- Ito H, Hietala J, Blomqvist G, Halldin C, Farde L. Comparison of the transient equilibrium and continuous infusion method for quantitative PET analysis of [11C]raclopride binding. J Cereb Blood Flow Metab. 1998;18:941–950. doi: 10.1097/00004647-199809000-00003. [DOI] [PubMed] [Google Scholar]
- Joyce JN, Millan MJ. Dopamine D3 receptor antagonists as therapeutic agents. Drug Discov Today. 2005;10:917–925. doi: 10.1016/S1359-6446(05)03491-4. [DOI] [PubMed] [Google Scholar]
- Kapur S, VanderSpek SC, Brownlee BA, Nobrega JN. Antipsychotic dosing in preclinical models is often unrepresentative of the clinical condition: a suggested solution based on in vivo occupancy. J Pharmacol Exp Ther. 2003;305:625–631. doi: 10.1124/jpet.102.046987. [DOI] [PubMed] [Google Scholar]
- Kapur S, Zipursky R, Jones C, Shammi CS, Remington G, Seeman P. A positron emission tomography study of quetiapine in schizophrenia: a preliminary finding of an antipsychotic effect with only transiently high dopamine D2 receptor occupancy. Arch Gen Psychiatry. 2000;57:553–559. doi: 10.1001/archpsyc.57.6.553. [DOI] [PubMed] [Google Scholar]
- Kapur S, Zipursky RB, Remington G. Clinical and theoretical implications of 5-HT2 and D2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. Am J Psychiatry. 1999;156:286–293. doi: 10.1176/ajp.156.2.286. [DOI] [PubMed] [Google Scholar]
- Kongsamut S, Roehr JE, Cai J, Hartman HB, Weissensee P, Kerman LL, et al. Iloperidone binding to human and rat dopamine and 5-HT receptors. Eur J Pharmacol. 1996;317:417–423. doi: 10.1016/s0014-2999(96)00840-0. [DOI] [PubMed] [Google Scholar]
- Lammertsma AA, Bench CJ, Hume SP, Osman S, Gunn K, Brooks DJ, et al. Comparison of methods for analysis of clinical [11C]raclopride studies. J Cereb Blood Flow Metab. 1996;16:42–52. doi: 10.1097/00004647-199601000-00005. [DOI] [PubMed] [Google Scholar]
- Landwehrmeyer B, Mengod G, Palacios JM. Differential visualization of dopamine D2 and D3 receptor sites in rat brain. A comparative study using in situ hybridization histochemistry and ligand binding autoradiography. Eur J Neurosci. 1993;5:145–153. doi: 10.1111/j.1460-9568.1993.tb00480.x. [DOI] [PubMed] [Google Scholar]
- Leopoldo M, Berardi F, Colabufo NA, De Giorgio P, Lacivita E, Perrone R, et al. Structure-affinity relationship study on N-[4-(4-arylpiperazin-1-yl)butyl]arylcarboxamides as potent and selective dopamine D(3) receptor ligands. J Med Chem. 2002;45:5727–5735. doi: 10.1021/jm020952a. [DOI] [PubMed] [Google Scholar]
- Levant B. Differential distribution of D3 dopamine receptors in the brains of several mammalian species. Brain Res. 1998;800:269–274. doi: 10.1016/s0006-8993(98)00529-0. [DOI] [PubMed] [Google Scholar]
- Levesque D, Diaz J, Pilon C, Martres MP, Giros B, Souil E, et al. Identification, characterization, and localization of the dopamine D3 receptor in rat brain using 7-[3H]hydroxy-N,N-di-n-propyl-2-aminotetralin. Proc Natl Acad Sci USA. 1992;89:8155–8159. doi: 10.1073/pnas.89.17.8155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick PN, Kapur S, Reckless GE, Wilson AA. Ex vivo [11C]-(+)-PHNO binding is unchanged in animal models displaying increased high-affinity states of the D2 receptor in vitro. Synapse. 2009;63:998–1009. doi: 10.1002/syn.20671. [DOI] [PubMed] [Google Scholar]
- McCormick PN, Kapur S, Seeman P, Wilson AA. Dopamine D2 receptor radiotracers [11C](+)-PHNO and [3H]raclopride are indistinguishably inhibited by D2 agonists and antagonists ex vivo. Nucl Med Biol. 2008;35:11–17. doi: 10.1016/j.nucmedbio.2007.08.005. [DOI] [PubMed] [Google Scholar]
- Narendran R, Slifstein M, Guillin O, Hwang Y, Hwang DR, Scher E, et al. Dopamine (D2/3) receptor agonist positron emission tomography radiotracer [11C]-(+)-PHNO is a D3 receptor preferring agonist in vivo. Synapse. 2006;60:485–495. doi: 10.1002/syn.20325. [DOI] [PubMed] [Google Scholar]
- Newman AH, Grundt P, Nader MA. Dopamine D3 receptor partial agonists and antagonists as potential drug abuse therapeutic agents. J Med Chem. 2005;48:3663–3679. doi: 10.1021/jm040190e. [DOI] [PubMed] [Google Scholar]
- Nobrega JN, Seeman P. Dopamine D2 receptors mapped in rat brain with [3H](+)PHNO. Synapse. 1994;17:167–172. doi: 10.1002/syn.890170305. [DOI] [PubMed] [Google Scholar]
- Nordstrom AL, Farde L, Nyberg S, Karlsson P, Halldin C, Sedvall G. D1, D2, and 5-HT2 receptor occupancy in relation to clozapine serum concentration: a PET study of schizophrenic patients. Am J Psychiatry. 1995;152:1444–1449. doi: 10.1176/ajp.152.10.1444. [DOI] [PubMed] [Google Scholar]
- Nordstrom AL, Farde L, Wiesel FA, Forslund K, Pauli S, Halldin C, et al. Central D2-dopamine receptor occupancy in relation to antipsychotic drug effects: a double-blind PET study of schizophrenic patients. Biol Psychiatry. 1993;33:227. doi: 10.1016/0006-3223(93)90288-o. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. Academic Press: New York; 1986. [Google Scholar]
- Rabiner E, Slifstein M, Nobrega J, Plisson C, Huiban M, Raymond R, et al. In vivo quantification of regional dopamine-D3 receptor binding potential of (+)-PHNO: studies in non-human primates and transgenic mince. Synapse. 2009;63:782–793. doi: 10.1002/syn.20658. [DOI] [PubMed] [Google Scholar]
- Schotte A, Janssen PF, Bonaventure P, Leysen JE. Endogenous dopamine limits the binding of antipsychotic drugs to D3 receptors in the rat brain: a quantitative autoradiographic study. Histochem J. 1996a;28:791–799. doi: 10.1007/BF02272152. [DOI] [PubMed] [Google Scholar]
- Schotte A, Janssen PF, Gommeren W, Luyten WH, Van Gompel P, Lesage AS, et al. Risperidone compared with new and reference antipsychotic drugs: in vitro and in vivo receptor binding. Psychopharmacology (Berl) 1996b;124:57–73. doi: 10.1007/BF02245606. [DOI] [PubMed] [Google Scholar]
- Schotte A, Janssen PF, Gommeren W, Luyten WH, Leysen JE. Autoradiographic evidence for the occlusion of rat brain dopamine D3 receptors in vivo. Eur J Pharmacol. 1992;218:373–375. doi: 10.1016/0014-2999(92)90196-b. [DOI] [PubMed] [Google Scholar]
- Seabrook GR, Patel S, Marwood R, Emms F, Knowles MR, Freedman SB, et al. Stable expression of human D3 dopamine receptors in GH4C1 pituitary cells. FEBS Lett. 1992;312:123–126. doi: 10.1016/0014-5793(92)80918-7. [DOI] [PubMed] [Google Scholar]
- Sokoloff P, Giros B, Martres MP, Bouthenet ML, Schwartz JC. Molecular cloning and characterization of a novel dopamine receptor (D3) as a target for neuroleptics. Nature. 1990;347:146–151. doi: 10.1038/347146a0. [DOI] [PubMed] [Google Scholar]
- Swanson LW. The projections of the ventral tegmental area and adjacent regions: a combined fluorescent retrograde tracer and immunofluorescence study in the rat. Brain Res Bull. 1982;9:321–353. doi: 10.1016/0361-9230(82)90145-9. [DOI] [PubMed] [Google Scholar]
- Tang L, Todd RD, Heller A, O'Malley KL. Pharmacological and functional characterization of D2, D3 and D4 dopamine receptors in fibroblast and dopaminergic cell lines. J Pharmacol Exp Ther. 1994;268:495–502. [PubMed] [Google Scholar]
- Tsukada H, Harada N, Nishiyama S, Ohba H, Sato K, Fukumoto D, et al. Ketamine decreased striatal [11C]raclopride binding with no alterations in static dopamine concentrations in the striatal extracellular fluid in the monkey brain: multiparametric PET studies combined with microdialysis analysis. Synapse. 2000;37:95–103. doi: 10.1002/1098-2396(200008)37:2<95::AID-SYN3>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- Tsukada H, Ohba H, Harada N, Kakiuchi T. Effects of anesthesia on kinetics of [11C]MNPA and its response to methamphetamine in the monkey brain. Neuroimage. 2008;41:T40. [Google Scholar]
- van der Zwaal EM, Luijendijk MC, Adan RA, la Fleur SE. Olanzapine-induced weight gain: chronic infusion using osmotic minipumps does not result in stable plasma levels due to degradation of olanzapine in solution. Eur J Pharmacol. 2008;585:130–136. doi: 10.1016/j.ejphar.2007.11.078. [DOI] [PubMed] [Google Scholar]
- van Vliet LA, Rodenhuis N, Dijkstra D, Wikstrom H, Pugsley TA, Serpa KA, et al. Synthesis and pharmacological evaluation of thiopyran analogues of the dopamine D3 receptor-selective agonist (4aR,10bR)-(+)-trans-3,4,4a,10b-tetrahydro-4-n-propyl-2H,5H [1]b enzopyrano[4,3-b]-1,4-oxazin-9-ol (PD 128907) J Med Chem. 2000;43:2871–2882. doi: 10.1021/jm0000113. [DOI] [PubMed] [Google Scholar]
- Willeit M, Ginovart N, Kapur S, Houle S, Hussey D, Seeman P, et al. High-affinity states of human brain dopamine D2/3 receptors imaged by the agonist [11C]-(+)-PHNO. Biol Psychiatry. 2006;59:389–394. doi: 10.1016/j.biopsych.2005.09.017. [DOI] [PubMed] [Google Scholar]
- Wilson AA, McCormick P, Kapur S, Willeit M, Garcia A, Hussey D, et al. Radiosynthesis and evaluation of [11C]-(+)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-naphtho[1,2-b][1,4]oxazin-9 -ol as a potential radiotracer for in vivo imaging of the dopamine D2 high-affinity state with positron emission tomography. J Med Chem. 2005;48:4153–4160. doi: 10.1021/jm050155n. [DOI] [PubMed] [Google Scholar]
- Yildiz A, Eryilmaz M, Gungor F, Erkilic M, Karayalcin B. Regional cerebral blood flow in schizophrenia before and after neuroleptic medication. Nucl Med Commun. 2000;21:1113–1118. doi: 10.1097/00006231-200012000-00004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.