Abstract
Necrotizing enterocolitis (NEC) is the most common intestinal emergency among premature infants. Risk factors in premature infants include immature intestinal immunity and an intestinal microbiota dominated by hospital-acquired bacteria. Some probiotics have been shown to decrease the incidence of NEC in premature infants. Among term infants, NEC is rare. However, among term infants with cyanotic congenital heart disease (CCHD), the incidence of NEC is similar to that of premature infants but with even greater mortality rates. Mechanisms by which NEC occurs in term infants with CCHD are unknown. Of central interest is the potential role of changes in the intestinal microbiota and whether these can be modified with probiotic bacteria; accordingly, we review the literature, propose hypotheses and present the rationale for future studies involving preliminary probiotic clinical trials.
Key words: intestinal microbiota, probiotic, bifidobacterium, cyanotic congenital heart disease, necrotizing enterocolitis
Introduction
Necrotizing enterocolitis (NEC) is the most common and destructive intestinal disease in premature infants, but rare and sporadic in term infants. Term infants with cyanotic congenital heart disease (CCHD) have a disturbingly high incidence of NEC. While the pathophysiology of NEC in premature infants has been much studied, processes by which NEC develops in term infants remain unclear. The most promising development in the prevention of NEC in premature infants over the last decade has been the use of probiotics. In this manuscript, we briefly review what is known about intestinal microbiota, NEC and probiotics in premature infants; with this background we then propose hypotheses and present the rationale for future clinical trials of probiotics in term infants with CCHD.
Cyanotic Congenital Heart Disease (CCHD): The ‘Blue Baby Syndrome’ Historical Perspective
In 1944 at the Johns Hopkins Hospital, Blalock and Thomas successfully performed an experimental operation to improve pulmonary blood flow in a cyanotic (blue) baby with tetralogy of Fallot, a common form of CCHD.1 Their surgical shunt technique consisted of anastomosis of the subclavian artery to the pulmonary artery. This pioneering work paved the way for the development of surgical interventions for other forms of CCHD. Fortunately, over 65 years after that first life-saving operation, pediatric cardiothoracic surgery and cardiopulmonary bypass procedures for the early definitive correction of tetralogy of Fallot have mortality rates of 3% or less and surgeries for all but the most severe forms of CCHD are generally successful.2 The inspiring history and evolution of this field have been well summarized.3,4
Common Forms of CCHD Include:
Left-sided obstructive lesions
Critical aortic stenosis
Coarctation of the aorta
Interrupted aortic arch
Right-sided obstructive lesions
Tetralogy of Fallot
Critical pulmonic stenosis
Ebstein's anomaly
Transposition of the great arteries
Single ventricle physiology
Hypoplastic left heart
Double outlet right ventricle
Tricuspid atresia
Pulmonary atresia with intact ventricular septum
Truncus arteriosus
Total anomalous pulmonary venous return
Premature Infants and Necrotizing Enterocolitis
With the development of neonatology and the increased survival of small premature infants, NEC has become the most common gastrointestinal emergency in the premature infant. The incidence of NEC is 3–10% in infants with birth weight less than 1,500 grams.5 This amounts to 10,000 infants diagnosed with NEC annually in the United States with peak incidence occurring 2–6 weeks after birth in premature infants.6 Death rates for NEC in premature infants vary depending on the level of prematurity and concurrent medical conditions, but several reports range from 9–30%.7,8 Mortality rates are highest in the most premature infants and in those forms of NEC characterized by full-thickness necrosis and cardiovascular complications.9
Previous reviews have detailed the complex pathophysiologic mechanisms of NEC in premature infants.10,11 In brief, the primary event of NEC in the premature infant appears to involve opportunistic pathogenic bacteria and translocation of bacterial antigens from the intestinal lumen triggering a systemic inflammatory cascade, intestinal necrosis and often cardiovascular instability. The premature infant appears to be at increased risk due to two major factors: (1) gut immaturity—the barrier function and innate immune system of the premature intestines are immature; and (2) gut dysbiosis—the premature infant becomes colonized with an intestinal microbiota that differs markedly from that of the healthy term infant. These differences are due in part to immature intestinal immunity12 as well as the frequent use of antibiotics, the presence of indwelling feeding tubes, lack of contact with the maternal microbiota and prolonged hospital stays.
Fecal microbiota analyses by conventional culturing techniques demonstrate that the intestinal microbiota of premature infants, compared to healthy term infants, show delayed colonization of beneficial Gram-positive bacteria and increased colonization of pathogenic and potentially pathogenic Gram-negative and Gram-positive bacteria.13 A culture-independent 16S ribosomal RNA gene-based study revealed that preterm infants that developed NEC, compared to preterm infants that did not, had less community diversity, greater abundance of Gammaproteobacteria, and had received more days of antibiotics prior to the development of NEC.14
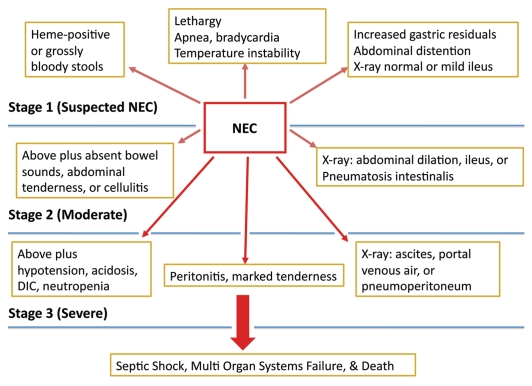
Clinical staging of NEC is generally based on modified Bell criteria (Fig. 1).15 NEC is usually managed conservatively with cessation of enteral feedings, continuous gastric suctioning and broad-spectrum intravenous antibiotics, however, 20–40% of infants undergo surgery, most commonly due to evidence of intestinal perforation. These infants often require resection of large segments of necrotic bowel leading to long-term difficulties with feeding intolerance and poor growth and abnormal neurodevelopment.16 In spite of decades of research, the incidence and mortality rates of NEC in the US have not decreased, due to the rapid onset and progression of this disease and the lack of successful interventions and prevention strategies.17,18
Figure 1.
Modified Bell criteria for clinical staging of necrotizing enterocolitis (NEC).15
Several promising prevention strategies have been explored to date in premature infants. Feeding of human milk either from the infant's mother19 or from screened donors20 decreases the risk of NEC. Enteral antibiotics21 and enteral infusions of HCl22 have been shown to prevent NEC, but concerns of safety and feasibility have limited further exploration of these modalities. Epidermal growth factor and heparin-binding epidermal growth factor consistently prevent NEC in animal models23 but have not been studied in premature infants. Probiotics are the most promising intervention to date in this population.24–26 Routine use of probiotics for premature infants in the US is not currently recommended due to uncertainty regarding dosage, organism and safety.27,28
Term Infants, NEC and CCHD
Epidemiology.
NEC among term infants has mostly been reported in case studies and case series from single institutions, therefore determining the incidence in this group is challenging. The most comprehensive estimate comes from a review of 29 cases of NEC in term infants in Australia, representing one case of NEC for every 20,000 live term births.29 Several authors have estimated that 10% of all NEC cases occur in term infants.30,31
Risk factors. Predisposing factors for NEC in these term infants include CCHD,32 polycythemia treated with partial exchange transfusion,33 Hirschsprung's disease,34 endocrine disease,29 early onset bacterial sepsis, hypotension, perinatal asphyxia, hypoglycemia, respiratory distress syndrome, protracted diarrhea, pre-eclampsia, cocaine abuse during pregnancy, cows' milk allergy and anti-C Rhesus incompatibility.35,36 In the two largest case series of term infants with NEC, the number of infants with CCHD and NEC were similar: 10 of 29 infants in Australia29 and 8 of 30 infants in Utah.35
Among term infants with CCHD, the incidence of NEC has been reported to be 3–7%, which is similar to the incidence among premature infants.32,37 Mortality rates among infants with CCHD who develop NEC, however, are even greater than in premature infants31,38 with one study reporting mortality as distressingly high as 71%.39 The highest risk seems to be in infants with (1) hypoplastic left heart,40 where the heart's left side (aorta, aortic valve, left ventricle and mitral valve) is underdeveloped and (2) truncus arteriosus,41 which is characterized by a large ventricular septal defect over which a large, single great blood vessel (truncus) gives rise to both the pulmonary arteries and the aorta.32
Proposed gut pathophysiology.
The exact mechanisms by which NEC develops in infants with CCHD are unknown. In term infants, intestinal function and immunity is not completely mature. In addition, in term infants with CCHD it is likely that both the acute mesenteric blood-flow reduction (hypoperfusion) and chronic cyanosis (hypoxia) may alter the development of the gut microbiota and the integrity of the gut barrier, increasing risk for NEC. Additional factors may include alterations in coagulation, apoptosis, peristalsis and motility, nutrient and water absorptive balances, tissue oxidative metabolism, cellular reduction-oxidation potential and regulation of reactive oxygen species generation and inflammatory responses.30,42–44 It is likely that, as in premature infants, disruption of the mucin layer and/or the epithelial monolayer separating the intestinal lumen from the lamina propria results in translocation of intestinal microbial products such as lipopolysaccharide (LPS), flagella, peptidoglycans, nucleic acids and other antigens, triggering local and systemic inflammation, intestinal necrosis and cardiovascular instability.30,44 The effect of persistent T-cell hyperactivation may also contribute to the pathologic outcome as seen in other microbial translocation-based diseases such as HIV/AIDS.45
The care of term infants with CCHD may also play a signifi- cant role in the development of NEC in this fragile population including: prolonged hospital stays and exposure to nosocomial microbes, delays in feeding, decreased rates of human milk feeding, poor nutritional status, courses of broad-spectrum antibiotics,14 and the inflammation and stress associated with cardiopulmonary bypass and cardiac surgery.30
Of central interest is the role of changes in the microbial community structure and function within the gut among these term infants with CCHD and whether any observed changes can be modified with probiotic bacteria.
Animal study insights. Animal models of NEC have predominantly been developed to study the immature gut (simulating the premature infant). In these models, the role of the innate and adaptive immune response in NEC has been well characterized including recognition of bacterial antigens by Toll-like receptors (TLRs), particularly TLR-2 and -4, leading to activation of transcription factors (e.g., NFκB), resulting in increased secretion of pro-inflammatory chemokines and cytokines (e.g., TNFα and numerous interleukins) as well as platelet activating factor.43,46 It is presumed that similar increases in pro-inflammatory signaling cascades occur in NEC in term infants with CCHD, though this has not been well studied.
The intestines are highly susceptible to hypoperfusion-induced pathologies due to a high critical oxygen requirement and the mucosal micro-circulatory network. An intriguing animal model that sheds some light on NEC in the term infant with CCHD is the adult rat hemorrhagic shock and resuscitation (HS/R) model. This model creates acute micro-circulatory mesenteric hypoperfusion resulting in intestinal mucosal injury. Interestingly, intravenous heparin-binding epidermal growth factor treatment attenuated injury of the intestinal mucosa and increased intestinal restitution. This protection occurs, at least in part, by reversing post-HS/R mesenteric vasoconstriction leading to significantly increased villous micro-circulatory blood flow. Multiple vasodilatory mechanisms may mediate these processes including the release of nitric oxide by endothelial cells and a reduction of the polymorphonuclear leukocyte/endothelial cell interactions associated with the inflammatory response.47 Changes in the intestinal microbiota in this model have not been reported. Acute hypoxia appeared to have no effect on microbiota colonization and translocation in the newborn rabbit; however, this study used only aerobic culture techniques.48 The impact of chronic hypoxia on the integrity of the mucosal barrier, on gut permeability and microbial translocation, and on the molecular genetic architecture of gut microbial communities, has not been determined.
Intestinal Microbiota and Probiotics: Potential Role in Term Infants with CCHD
Advances in the study of human microbial ecology.
The development of culture-independent methods to identify, examine and explore microbes based on molecular sequence analysis49–51 has dramatically increased momentum in this area of investigation and indeed made this “an exciting time to study gastrointestinal microbiology.”52 The recently published draft of the International MetaHIT (Metagenomics of the Human Intestinal Tract) project created a gene catalogue of the human gut microbiota (the microbiome).53 This is one of many large scale studies of the human microbial ecosystem, including the NIH Human Microbiome Project,54 which are defining the role of both individual pathogens55 and entire microbial communities14 in the development of diseases such as NEC.
The gut microbiota of term infants.
The fetal gut is generally believed to be sterile until rupture of the fetal membranes. Microbial colonization of the healthy term newborn gut progresses quickly with the initial wave of colonists arriving predominantly from the maternal genito-urinary tract followed by a more stable population. Mode of delivery and exposure to antibiotics influence the intestinal microbiota briefly, but then a stable fairly simple community is established which is mostly influenced by feeding (breast vs. formula) and other environmental factors. There is significant inter-individual variation at lower-taxonomic levels (genus and species/strain) in the healthy term infant, but at the higher-taxonomic phylum-level, the rank abundance of this community is dominated mostly by members of the phyla Proteobacteria, Firmicutes and Actinobacteria (mixed results) with little from the Bacteroidetes or other phyla. This community does not shift significantly until the introduction of solid foods at 4 to 8 months of age, at which time diversity increases and leads to a gut microbiota composition similar to the adult, dominated by the phyla Firmicutes and Bacteroidetes with a small fraction allotted to the Proteobacteria, Actinobacteria and other phyla, by 2 years of age.
The gut microbiota of term infants with CCHD.
We found no studies that characterize the intestinal microbiota of the term infant with CCHD in either the NIH-National Library of Medicine (Pubmed) database or the ClinicalTrials.gov registry. The human intestinal microbiota is critical to homeostasis and particularly relevant to infants with CCHD due to many co-host functioning processes including: regulation of angiogenesis in the gut microvascular network, maintenance of proper reductive- to oxidative-stress and anti- to pro-inflammatory balances, epithelial turnover and genetic/epigenetic regulation, intestinal pH and motility, synthesis of micronutrients like vitamin K and several of the B vitamins, regulation of bile salt and lipid metabolism and fat deposition, nitrogen metabolism, fermentation of polysaccharides and absorption of digestible microbial products, absorption of electrolytes and trace minerals and bioenergetic/thermodynamic regulation.56,57 In addition, gut microbial communities can protect the host and the ecosystem's vital diversity through competition with opportunist colonization as well as through interfering with receptor binding and virulence factor production.58,59
Growth. The specific role of intestinal microbiota in energy regulation has particular relevance in term infants with CCHD, who are at high risk for poor growth. Gut microbes participate in normal digestion and metabolism of nutrients even during periods of nutrient deprivation.60 Conventionally reared mice have a 40% higher body fat content than germ-free mice even though the latter consume more food. When the distal gut microbiota from normal mice is transplanted into germ free mice, there is a 60% increase in body fat within 2 weeks without any increase in food consumption or obvious difference in energy expenditure.61,62 Microbes generate short-chain fatty acids and ferment indigestible polysaccharides to digestible monosaccharides increasing the amount of energy extracted from the diet. Metagenomic studies of genetically obese mice and humans demonstrate correlations between obesity and an ‘energy-harvesting’ gut microbiome composition.63 Conversely dietary manipulation can rapidly alter the gut microbiome composition despite host colonization history64 and independent of obesity.65 Since term infants with CCHD, like premature infants, are at high risk for poor growth, they require maximal caloric intake to ensure adequate weight and length gains. Furthermore, growth is a significant challenge for infants with CCHD even in the context of very high caloric intake. In a recent study in Egypt, 18 infants with CCHD (ten with tetralogy of Fallot, four with tricuspid atresia and four with pulmonary atresia), in comparison to ageand sex-matched healthy controls, had a mean weight and height at 46 and 81%, respectively, of their counterparts.66
Chronic hypoxia also influences growth. In a lamb model of chronic hypoxia, the pulmonary artery was partially occluded and an atrial septostomy performed to create an aortic oxygen saturation of 60–74%. After two weeks of stable chronic hypoxia, somatic growth in this model was decreased to 60% of control lambs and intestinal length and weight were unchanged. Specific activity of the digestive enzyme lactase, the principal disaccharidase of the infant lamb intestine, and total small intestinal contents of lactase, were significantly curtailed in the hypoxemic animals.67 The intestinal microbiota was not analyzed in this study. In a mouse model of chronic hypoxia (11% oxygen for 4 weeks), somatic weight gain was decreased to 70% of controls, heart weight increased, but weights of liver, kidney and brain decreased compared to controls. Intestinal weights and lengths and the composition of the intestinal microbiota were not reported.68 Similar studies have been performed in rats with similar results.69,70
Attempts to manipulate the intestinal microbiota with probiotics to improve growth in premature infants have been successful in some populations but not in others.71,72 Similar studies in term infants with CCHD have not yet been attempted.
Probiotics for term infants with CCHD.
The joint Food and Agriculture Organization of the United Nations/World Health Organization defines probiotics as “live microorganisms, which when administered in adequate amounts, confer a health benefit on the host”.73 These products can be sub-categorized to include dietary supplements, medical foods or designer probiotics that are genetically altered.74 Probiotics have been shown to provide diverse human health benefits,75–78 particularly in the pediatric population.79–81 Clinical recommendations regarding the use of probiotics in term and pre-term infants remain unclear due to the extensive array of probiotic products and doses administered. Clinical trials in the neonatal and pediatric population have included single organisms and combinations, most commonly lactobacilli, bifidobacteria, streptococci, enterococci, clostridia, species of the genera Bacillus, E. coli and the yeast Saccharomyces boulardii.82,83
Many probiotic species administered are already inhabitants of the healthy gut and their function when ingested is similar to the homeostatic functions of the ‘normal’ microbiota. However, some probiotic species have originated very specific and unique metabolic capabilities, possibly in co-evolutionary response to persistent selective pressures exerted by host biology and ingested matter. A compelling example in the neonate is the development of specific enzymes (sialidase and fucosidase) in the genome of Bifidobacterium longum ssp. infantis which allow deconstruction and consumption of human milk oligosaccharides.84
Potential mechanisms. In vitro and animal studies suggest several mechanisms of action of probiotics: competitive inhibition of pathogen-adhesion receptor-binding sites, growth promotion of partnering commensals and mutualists, induction of transforming growth factor-β and modulation of cellular apoptosis/proliferation, decreased expression of pro-inflammatory cytokines, increased expression of anti-inflammatory cytokines, secretion of enzymes and beneficial metabolites, modulation of cellular redox potential, functional enhancement of epithelial tight junctions, induction of intestinal mucin production, signaling interactions with cell surface molecules invoking cytoprotective and immune responses (with local and systemic effects), increased expression of innate immune antimicrobial peptides, and possibly even evoking neuro-endocrine and hormonal responses involved in growth and development.57,78,85–88
Probiotics and cardiovascular health.
In adults, a potential role of intestinal microbiota and probiotics in prevention/treatment of atherosclerosis and coronary artery disease has been proposed.86,89–91 Animal trials of probiotics in coronary heart disease have focused mostly on lowering serum lipoprotein cholesterol.92 A rat model of heart attacks or myocardial infarctions (MI) demonstrated that a combination of two probiotics prior to and following the induced MI had less post-MI apoptosis in certain cerebral regions (e.g., limbic system) than the controls. The proposed mechanism is a decrease in circulating pro-inflammatory cytokines as a result of probiotic ingestion.93
Unanswered Questions and Hypothesis
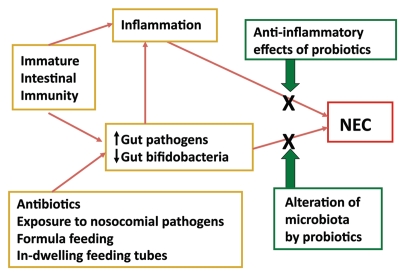
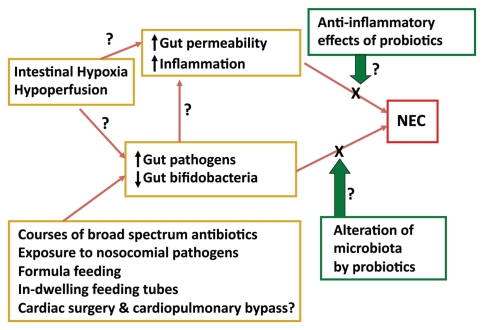
Many questions remain unanswered regarding the use of probiotics in high-risk term infants with CCHD. Figure 2 summarizes some of the proposed mechanisms of action of probiotics in preventing NEC in premature infants. The following questions remain unanswered in term infants with CCHD (Fig. 3):
How does the intestinal microbiota of the term infant with CCHD differ from that of the healthy breast-fed term infant?
Does the administration of probiotics change the intestinal microbiota of the term infant with CCHD?
Does administration of probiotics to term infants with CCHD change fecal or plasma levels of pro-inflammatory Gram-negative bacterial antigens (e.g., LPS) or cytokines?
Does feeding human milk to infants with CCHD decrease the risk of NEC?
Does administration of probiotics and/or prebiotics to infants with CCHD decrease the risk of NEC?
Figure 2.
Proposed mechanisms for prevention of NEC by probiotics in premature infants.
Figure 3.
Unanswered questions regarding NEC onset in term infants with cyanotic congenital heart disease (CCHD) and the potential role of probiotics.
Future studies could include experimental investigation of the following overarching hypotheses: CCHD alters the intestinal microbiota composition of term infants increasing the risk of NEC and prophylactic administration of probiotics alters the intestinal microbiota in this high-risk population decreasing the NEC risk. Pilot probiotic clinical trials in infants with CCHD could include analysis of fecal microbiota and fecal LPS levels with comparisons made between the probiotic group and the placebo group and between the placebo group and age- and sex-matched healthy control infants. Such studies would be essential to determine feasibility and sample size for a larger multi-center trial of NEC prevention. Parallel animal studies to elucidate the impact of chronic hypoxia on the development of the intestinal microbiota as well as intestinal permeability and immune function would also be of interest.
Potential Barriers to Research Progress
Guidelines to designing, conducting, publishing and communicating results of clinical trials involving probiotic applications in humans have recently been published.94 The following concerns are of particular relevance in the context of infants with CCHD.
Safety.
The safety of probiotics has been extensively reviewed.95,96 Probiotics have been added to infant formulas in Europe and Asia for many years without evidence of harm.97 Probiotic containing formulas are now available in North America. Potential safety concerns include the following:
Potential for systemic infection including septicemia and endocarditis from probiotic species. Rare cases of bacteremia and fungemia have been reported, particularly for species of lactobacilli98 and Saccharomyces.99 However population studies in countries with high probiotic use have not shown any increased risk of bacteremia or endocarditis due to probiotics.100,101 Probiotics appear to actually decrease rather than increase bacterial translocation in both animal models102,103 and humans.104
Antibiotic resistance. Testing of 41 probiotic supplements and foods demonstrated high sensitivity to chloramphenicol, tetracycline, ampicillin, amoxicillin/clavulanic acid, cephalothin and imipenem, moderate sensitivity to vancomycin, rifampicin, streptomycin, bacitracin and erythromycin and high levels of resistance to ciprofloxacin, amikacin, trimethoprim/sulphamethoxazole and gentamicin.105 Bifidobacteria appear to have minimal antibiotic resistance. Twenty-six strains of B. breve showed susceptibility to all antibiotics tested (17 total) except streptomycin.106
Purity and viability of commercial probiotic products. Several studies have demonstrated that labeling of commercial probiotic products is often inaccurate. Some commercial products contain organisms not listed and others do not contain the specific strain listed.107 In addition, stability of probiotics changes over time so that it is challenging to be certain of the dose provided. Ideally, strains with a known genomic sequence and sensitivity pattern should be used84 and regular testing of viability performed.
Onset of disease.
The onset of NEC in term infants is very early (average 4 days of life). This may not apply to infants with CCHD, as most of these infants develop NEC post-operatively. Early onset of NEC suggests that attempts to change the intestinal microbiota and thereby prevent this disease may not be effective.
Potential confounders.
Infants with CCHD often receive prolonged courses of antibiotics, which may negate any positive effects of the probiotic administered. In addition, infant feeding patterns have a significant effect on the intestinal microbiota. Individual institution protocols for care may also impact outcomes.
Specimen collection.
Stool has often been assumed to represent the microbiota of more distal intestinal sites (e.g., ileum to anus) and, therefore, may not provide an accurate assessment of the microbiota inhabiting more proximal sites. In addition, stool specimens can also dominantly represent allochthonous and transient species (also called liquid-phase microbes), as opposed to the autochthonous and adherent biofilms (also called particle-phase microbes) interacting directly with the host gut wall.108 The abundance and diversity of these species may be underestimated in fecal surveys as shown by mucosal biopsy-specific detection of microbes109 implicated in other inflammatory bowel diseases. Qualitatively, spatial and temporal studies of human intestinal ecology as well as comparative studies of intestinal sample collection/preparation methods produce differing results. Standardization of sampling and laboratory approaches will add clarity to this complex field.
Conclusion
Necrotizing enterocolitis is a devastating complication in term neonates with cyanotic congenital heart disease, often resulting in great morbidity and mortality among these high risk ‘blue babies’. Whether intestinal hypoxia and hypoperfusion alters the intestinal microbiota increasing the risk of NEC is unknown. Whether administration of probiotics to these infants alters their intestinal microbiota, decreases the risk of NEC, and/or improves weight gain also is unknown. If a simple preventive approach, like probiotic therapy, were effective, this would be a significant breakthrough and shift in the treatment paradigm for these high risk infants.
Acknowledgements
C.L.E. receives support from the Children's Miracle Network Competitive Research Grant, the UC Davis Professors for the Future Predoctoral Fellowship, from the UC Davis Clinical & Translational Science Center (CTSC)'s NIH-sponsored T32 Predoctoral Clinical Research Training Program (NIH-NCRR & Roadmap Grant # ULI RR024146), and from J.C.R.
J.C.R. receives support from NIH NHLBI 55067 and the Richard A. and Nora Eccles Harrison Endowed Chair in Diabetes Research.
M.A.U. receives support from the NIH (HD059127).
Abbreviations
- NEC
necrotizing enterocolitis
- CCHD
cyanotic congenital heart disease
- LPS
lipopolysaccharide
- TLR
toll-like receptor
Footnotes
Previously published online: www.landesbioscience.com/journals/gutmicrobes/gutmicrobes/14077
References
- 1.Blalock A, Taussig HB. Landmark article May 19, 1945: The surgical treatment of malformations of the heart in which there is pulmonary stenosis or pulmonary atresia. JAMA. 1984;27(251):2123–2138. doi: 10.1001/jama.251.16.2123. By Blalock A, Taussig HB. [DOI] [PubMed] [Google Scholar]
- 2.Pigula FA, Khalil PN, Mayer JE, del Nido PJ, Jonas RA. Repair of tetralogy of Fallot in neonates and young infants. Circulation. 1999;100:157–161. doi: 10.1161/01.cir.100.suppl_2.ii-157. [DOI] [PubMed] [Google Scholar]
- 3.Murphy AM, Cameron DE. The Blalock-Taussig-Thomas collaboration: a model for medical progress. JAMA. 2008;300:328–330. doi: 10.1001/jama.300.3.328. [DOI] [PubMed] [Google Scholar]
- 4.Williams JA, Bansal AK, Kim BJ, Nwakanma LU, Patel ND, Seth AK, et al. Two thousand Blalock-Taussig shunts: a six-decade experience. Ann Thorac Surg. 2007;84:2070–2075. doi: 10.1016/j.athoracsur.2007.06.067. [DOI] [PubMed] [Google Scholar]
- 5.Lin PW, Nasr TR, Stoll BJ. Necrotizing enterocolitis: recent scientific advances in pathophysiology and prevention. Semin Perinatol. 2008;32:70–82. doi: 10.1053/j.semperi.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 6.Hunter CJ, Podd B, Ford HR, Camerini V. Evidence vs. experience in neonatal practices in necrotizing enterocolitis. J Perinatol. 2008;28:9–13. doi: 10.1038/jp.2008.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lin PW, Stoll BJ. Necrotising enterocolitis. Lancet. 2006;368:1271–1283. doi: 10.1016/S0140-6736(06)69525-1. [DOI] [PubMed] [Google Scholar]
- 8.Stoll BJ. Epidemiology of necrotizing enterocolitis. Clin Perinatol. 1994;21:205–218. doi: 10.1016/S0095-5108(18)30341-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Blakely ML, Lally KP, McDonald S, Brown RL, Barnhart DC, Ricketts RR, et al. Subcommittee of the NICHD Neonatal Research Network, author. Postoperative outcomes of extremely low birth-weight infants with necrotizing enterocolitis or isolated intestinal perforation: a prospective cohort study by the NICHD Neonatal Research Network. Ann Surg. 2005;241:984–989. doi: 10.1097/01.sla.0000164181.67862.7f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Emami CN, Petrosyan M, Giuliani S, Williams M, Hunter C, Prasadarao NV, et al. Role of the host defense system and intestinal microbial flora in the pathogenesis of necrotizing enterocolitis. Surg Infect (Larchmt) 2009;10:407–417. doi: 10.1089/sur.2009.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Neu J, Mshvildadze M, Mai V. A roadmap for understanding and preventing necrotizing enterocolitis. Curr Gastroenterol Rep. 2008;10:450–457. doi: 10.1007/s11894-008-0084-x. [DOI] [PubMed] [Google Scholar]
- 12.Salzman NH, Hung K, Haribhai D, Chu H, Karlsson-Sjöberg J, Amir E, et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat Immunol. 2010;11:76–83. doi: 10.1038/ni.1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Westerbeek EA, van den Berg A, Lafeber HN, Knol J, Fetter WP, van Elburg RM. The intestinal bacterial colonisation in preterm infants: a review of the literature. Clin Nutr. 2006;25:361–368. doi: 10.1016/j.clnu.2006.03.002. [DOI] [PubMed] [Google Scholar]
- 14.Wang Y, Hoenig JD, Malin KJ, Qamar S, Petrof EO, Sun J, et al. 16S rRNA gene-based analysis of fecal microbiota from preterm infants with and without necrotizing enterocolitis. ISME J. 2009;3:944–954. doi: 10.1038/ismej.2009.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Walsh MC, Kliegman RM. Necrotizing enterocolitis: treatment based on staging criteria. Pediatr Clin North Am. 1986;33:179–201. doi: 10.1016/S0031-3955(16)34975-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vohr BR, Wright LL, Dusick AM, Mele L, Verter J, Steichen JJ, et al. Neurodevelopmental and functional outcomes of extremely low birth weight infants in the National Institute of Child Health and Human Development Neonatal Research Network 1993–1994. Pediatrics. 2000;105:1216–1226. doi: 10.1542/peds.105.6.1216. [DOI] [PubMed] [Google Scholar]
- 17.Stout G, Lambert DK, Baer VL, Gordon PV, Henry E, Wiedmeier SE, et al. Necrotizing enterocolitis during the first week of life: a multicentered case-control and cohort comparison study. J Perinatol. 2008;28:556–560. doi: 10.1038/jp.2008.36. [DOI] [PubMed] [Google Scholar]
- 18.Grave GD, Nelson SA, Walker WA, Moss RL, Dvorak B, Hamilton FA, et al. New therapies and preventive approaches for necrotizing enterocolitis: report of a research planning workshop. Pediatr Res. 2007;62:510–514. doi: 10.1203/PDR.0b013e318142580a. [DOI] [PubMed] [Google Scholar]
- 19.Sisk PM, Lovelady CA, Dillard RG, Gruber KJ, O'Shea TM. Early human milk feeding is associated with a lower risk of necrotizing enterocolitis in very low birth weight infants. J Perinatol. 2007;27:428–433. doi: 10.1038/sj.jp.7211758. [DOI] [PubMed] [Google Scholar]
- 20.Boyd CA, Quigley MA, Brocklehurst P. Donor breast milk versus infant formula for preterm infants: systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2007;92:169–175. doi: 10.1136/adc.2005.089490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bury RG, Tudehope D. Enteral antibiotics for preventing necrotizing enterocolitis in low birthweight or preterm infants. Cochrane Database Syst Rev. 2001:405. doi: 10.1002/14651858.CD000405. [DOI] [PubMed] [Google Scholar]
- 22.Carrion V, Egan EA. Prevention of neonatal necrotizing enterocolitis. J Pediatr Gastroenterol Nutr. 1990;11:317–323. doi: 10.1097/00005176-199010000-00006. [DOI] [PubMed] [Google Scholar]
- 23.Dvorak B. Milk epidermal growth factor and gut protection. J Pediatr. 2010;156:31–35. doi: 10.1016/j.jpeds.2009.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Deshpande G, Rao S, Patole S. Probiotics for prevention of necrotising enterocolitis in preterm neonates with very low birthweight: a systematic review of randomised controlled trials. Lancet. 2007;369:1614–1620. doi: 10.1016/S0140-6736(07)60748-X. [DOI] [PubMed] [Google Scholar]
- 25.Alfaleh K, Bassler D. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst Rev. 2008:5496. doi: 10.1002/14651858.CD005496.pub2. [DOI] [PubMed] [Google Scholar]
- 26.Lin HC, Hsu CH, Chen HL, Chung MY, Hsu JF, Lien RI, et al. Oral probiotics prevent necrotizing enterocolitis in very low birth weight preterm infants: a multicenter, randomized, controlled trial. Pediatrics. 2008;122:693–700. doi: 10.1542/peds.2007-3007. [DOI] [PubMed] [Google Scholar]
- 27.Caplan MS. Probiotic and prebiotic supplementation for the prevention of neonatal necrotizing enterocolitis. J Perinatol. 2009;29:2–6. doi: 10.1038/jp.2009.21. [DOI] [PubMed] [Google Scholar]
- 28.Walker WA, Sherman P, Shneider BL, Cohen M, Barnard J. State of research in pediatric gastroenterology, hepatology and nutrition: 2010 and beyond. Gastroenterology. 2010;138:411–416. doi: 10.1053/j.gastro.2009.12.034. [DOI] [PubMed] [Google Scholar]
- 29.Bolisetty S, Lui K, Oei J, Wojtulewicz J. A regional study of underlying congenital diseases in term neonates with necrotizing enterocolitis. Acta Paediatrica. 2000;89:1226–1230. doi: 10.1080/080352500750027619. [DOI] [PubMed] [Google Scholar]
- 30.Giannone PJ, Luce WA, Nankervis CA, Hoffman TM, Wold LE. Necrotizing enterocolitis in neonates with congenital heart disease. Life Sci. 2008;82:341–347. doi: 10.1016/j.lfs.2007.09.036. [DOI] [PubMed] [Google Scholar]
- 31.Ostlie DJ, Spilde TL, St. Peter SD, Sexton N, Miller KA, Sharp RJ, et al. Necrotizing enterocolitis in full-term infants. J Pediatr Surg. 2003;38:1039–1042. doi: 10.1016/s0022-3468(03)00187-8. [DOI] [PubMed] [Google Scholar]
- 32.McElhinney DB, Hedrick HL, Bush DM, Pereira GR, Stafford PW, Gaynor JW. Necrotizing enterocolitis in neonates with congenital heart disease: risk factors and outcomes. Pediatrics. 2000;106:1080–1087. doi: 10.1542/peds.106.5.1080. [DOI] [PubMed] [Google Scholar]
- 33.Ozek E, Soll R, Schimmel MS. Partial exchange transfusion to prevent neurodevelopmental disability in infants with polycythemia. Cochrane Database Syst Rev. 2010:5089. doi: 10.1002/14651858.CD005089.pub2. [DOI] [PubMed] [Google Scholar]
- 34.Raboei EH. Necrotizing enterocolitis in full-term neonates: is it aganglionosis? Eur J Pediatr Surg. 2009;19:101–104. doi: 10.1055/s-0029-1202771. [DOI] [PubMed] [Google Scholar]
- 35.Lambert DK, Christensen RD, Henry E, Besner GE, Baer VL, Wiedmeier SE, et al. Necrotizing enterocolitis in term neonates: data from a multihospital health-care system. J Perinatol. 2007;27:437–443. doi: 10.1038/sj.jp.7211738. [DOI] [PubMed] [Google Scholar]
- 36.Maayan-Metzger A, Itzchak A, Mazkereth R, Kuint J. Necrotizing enterocolitis in full-term infants: casecontrol study and review of the literature. J Perinatol. 2004;24:494–499. doi: 10.1038/sj.jp.7211135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leung MP, Chau KT, Hui PW, Tam AY, Chan FL, Lai CL, et al. Necrotizing enterocolitis in neonates with symptomatic congenital heart disease. J Pediatr. 1988;113:1044–1046. doi: 10.1016/s0022-3476(88)80580-8. [DOI] [PubMed] [Google Scholar]
- 38.Yang YH, Yau KI, Wu MH, Tang JR. Cyanotic congenital heart disease and necrotizing enterocolitis: report of three cases. Acta Paediatrica Taiwanica. 1999;40:348–350. [PubMed] [Google Scholar]
- 39.Cheng W, Leung MP, Tam PK. Surgical intervention in necrotizing enterocolitis in neonates with symptomatic congenital heart disease. Pediatr Surg Int. 1999;15:492–495. doi: 10.1007/s003830050647. [DOI] [PubMed] [Google Scholar]
- 40.American Heart Association, author. Common Congenital Cardiovascular Defects. Single-Ventricle Defects. [1 February 2010]. http://www.americanheart.org/presenter.jhtml?identifier=11072#HLHS.
- 41.American Heart Association, author. Common Congenital Cardiovascular Defects. Truncus Arteriosus. [1 February 2010]. http://www.americanheart.org/presenter.jhtml?identifier=11073.
- 42.Abreu MT. The Ying and Yang of bacterial signaling in necrotizing enterocolitis. Gastroenterology. 2010;138:39–43. doi: 10.1053/j.gastro.2009.11.031. [DOI] [PubMed] [Google Scholar]
- 43.Frost BL, Jilling T, Caplan MS. The importance of proinflammatory signaling in neonatal necrotizing enterocolitis. Semin Perinatol. 2008;32:100–106. doi: 10.1053/j.semperi.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Anand RJ, Leaphart CL, Mollen KP, Hackam DJ. The role of the intestinal barrier in the pathogenesis of necrotizing enterocolitis. Shock. 2007;27:124–133. doi: 10.1097/01.shk.0000239774.02904.65. [DOI] [PubMed] [Google Scholar]
- 45.Jiang W, Lederman MM, Hunt P, Sieg SF, Haley K, Rodriguez B, et al. Plasma levels of bacterial DNA correlate with immune activation and the magnitude of immune restoration in persons with antiretroviral-treated HIV infection. J Infect Dis. 2009;199:1177–1185. doi: 10.1086/597476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chan KL, Wong KF, Luk JM. Role of LPS/CD14/TLR4-mediated inflammation in necrotizing enterocolitis: pathogenesis and therapeutic implications. World J Gastroenterol. 2009;15:4745–4752. doi: 10.3748/wjg.15.4745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.El-Assal ON, Radulescu A, Besner GE. Heparin-binding EGF-like growth factor preserves mesenteric microcirculatory blood flow and protects against intestinal injury in rats subjected to hemorrhagic shock and resuscitation. Surgery. 2007;142:234–242. doi: 10.1016/j.surg.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 48.Urao M, Teitelbaum DH, Drongowski RA, Coran AG. The association of gut-associated lymphoid tissue and bacterial translocation in the newborn rabbit. J Pediatr Surg. 1996;31:1482–1487. doi: 10.1016/s0022-3468(96)90160-8. [DOI] [PubMed] [Google Scholar]
- 49.Zaneveld JR, Lozupone C, Gordon JI, Knight R. Ribosomal RNA diversity predicts genome diversity in gut bacteria and their relatives. Nucleic Acids Res. 2010;38:3869–3879. doi: 10.1093/nar/gkq066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wu D, Hugenholtz P, Mavromatis K, Pukall R, Dalin E, Ivanova NN. A phylogeny-driven genomic encyclopedia of bacteria and archaea. Nature. 2009;462:1056–1060. doi: 10.1038/nature08656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Weng L, Rubin EM, Bristow J. Application of sequence-based methods in human microbial ecology. Genome Res. 2006;16:316–322. doi: 10.1101/gr.3676406. [DOI] [PubMed] [Google Scholar]
- 52.Hecht G, Riedmann EM. Exciting times for gastrointestinal microbiology. Gut Microbes. 2010;1:1–2. doi: 10.4161/gmic.1.1.10544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464:59–65. doi: 10.1038/nature08821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Peterson J, Garges S, Giovanni M, McInnes P, Wang L, et al. The NIH human microbiome project. Genome Res. 2009;19:2317–2323. doi: 10.1101/gr.096651.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dittmar E, Beyer P, Fischer D, Schäfer V, Schoepe H, Bauer K, et al. Necrotizing enterocolitis of the neonate with Clostridium perfringens: diagnosis, clinical course and role of alpha toxin. Eur J Pediatr. 2008;167:891–895. doi: 10.1007/s00431-007-0614-9. [DOI] [PubMed] [Google Scholar]
- 56.DiBaise JK, Zhang H, Crowell MD, Krajmalnik-Brown R, Decker GA, Rittmann BE. Gut microbiota and its possible relationship with obesity. Mayo Clin Proc. 2008;83:460–469. doi: 10.4065/83.4.460. [DOI] [PubMed] [Google Scholar]
- 57.Neish AS. Microbes in gastrointestinal health and disease. Gastroenterology. 2009;136:65–80. doi: 10.1053/j.gastro.2008.10.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hodges K, Gill R. Infectious diarrhea: Cellular and molecular mechanisms. Gut Microbes. 2010;1:4–21. doi: 10.4161/gmic.1.1.11036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Santos RL, Raffatellu M, Bevins CL, Adams LG, Tükel C, Tsolis RM, et al. Life in the inflamed intestine, Salmonella style. Trends Microbiol. 2009;17:498–506. doi: 10.1016/j.tim.2009.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Crawford PA, Crowley JR, Sambandam N, Muegge BD, Costello EK, Hamady M, et al. Regulation of myocardial ketone body metabolism by the gut microbiota during nutrient deprivation. Proc Natl Acad Sci USA. 2009;106:11276–11281. doi: 10.1073/pnas.0902366106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Backhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci USA. 2004;101:15718–15723. doi: 10.1073/pnas.0407076101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Backhed F, Manchester JK, Semenkovich CF, Gordon JI. Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc Natl Acad Sci USA. 2007;104:979–984. doi: 10.1073/pnas.0605374104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444:1022–1023. doi: 10.1038/4441022a. [DOI] [PubMed] [Google Scholar]
- 64.Turnbaugh PJ, Ridaura VK, Faith JJ, Rey FE, Knight R, Gordon JI. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med. 2009;1:6–14. doi: 10.1126/scitranslmed.3000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hildebrandt MA, Hoffmann C, Sherrill-Mix SA, Keilbaugh SA, Hamady M, Chen YY, et al. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology. 2009;137:1716–1724. doi: 10.1053/j.gastro.2009.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kandil ME, Elwan A, Hussein Y, Kandeel W, Rasheed M. Ghrelin levels in children with congenital heart disease. J Trop Pediatr. 2009;55:307–312. doi: 10.1093/tropej/fmp012. [DOI] [PubMed] [Google Scholar]
- 67.Bernstein D, Bell JG, Kwong L, Castillo RO. Alterations in postnatal intestinal function during chronic hypoxemia. Pediatr Res. 1992;31:234–238. doi: 10.1203/00006450-199203000-00008. [DOI] [PubMed] [Google Scholar]
- 68.Farahani R, Kanaan A, Gavrialov O, Brunnert S, Douglas RM, Morcillo P, et al. Differential effects of chronic intermittent and chronic constant hypoxia on postnatal growth and development. Pediatr Pulmonol. 2008;43:20–28. doi: 10.1002/ppul.20729. [DOI] [PubMed] [Google Scholar]
- 69.Moromisato DY, Moromisato MY, Zanconato S, Roberts CT., Jr. Effect of hypoxia on lung, heart and liver insulin-like growth factor-I gene and receptor expression in the newborn rat. Crit Care Med. 1996;24:919–924. doi: 10.1097/00003246-199606000-00008. [DOI] [PubMed] [Google Scholar]
- 70.Mortola JP, Xu LJ, Lauzon AM. Body growth, lung and heart weight and DNA content in newborn rats exposed to different levels of chronic hypoxia. Can J Physiol Pharmacol. 1990;68:1590–1594. doi: 10.1139/y90-242. [DOI] [PubMed] [Google Scholar]
- 71.Kitajima H, Sumida Y, Tanaka R, Yuki N, Takayama H, Fujimura M. Early administration of Bifidobacterium breve to preterm infants: randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 1997;76:101–107. doi: 10.1136/fn.76.2.f101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Underwood MA, Salzman NH, Bennett SH, Barman M, Mills DA, Marcobal A, et al. A randomized placebo-controlled comparison of 2 prebiotic/probiotic combinations in preterm infants: impact on weight gain, intestinal microbiota and fecal short-chain fatty acids. J Pediatr Gastroenterol Nutr. 2009;48:216–225. doi: 10.1097/MPG.0b013e31818de195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Joint Food and Agriculture Organization of the United Nations/World Health Organization expert consultation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria, October 2001. [27 January 2009]. http://www.who.int/foodsafety/publications/fs_management/en/probiotics.pdf.
- 74.Sanders ME. How do we know when something called “probiotic” is really a probiotic? A guideline for consumers and health care professionals. Functional Food Reviews. 2009;1:3–12. doi: 10.2310/6180.2009.00002. [DOI] [Google Scholar]
- 75.Kalliomäki M, Antoine JM, Herz U, Rijkers GT, Wells JM, Mercenier A. Guidance for substantiating the evidence for beneficial effects of probiotics: prevention and management of allergic diseases by probiotics. J Nutr. 2010;140:713–721. doi: 10.3945/jn.109.113761. [DOI] [PubMed] [Google Scholar]
- 76.Haller D, Antoine JM, Bengmark S, Enck P, Rijkers GT, Lenoir-Wijnkoop I. Guidance for substantiating the evidence for beneficial effects of probiotics: probiotics in chronic inflammatory bowel disease and the functional disorder irritable bowel syndrome. J Nutr. 2010;140:690–697. doi: 10.3945/jn.109.113746. [DOI] [PubMed] [Google Scholar]
- 77.Wolvers D, Antoine JM, Myllyluoma E, Schrezenmeir J, Szajewska H, Rijkers GT. Guidance for substantiating the evidence for beneficial effects of probiotics: prevention and management of infections by probiotics. J Nutr. 2010;140:698–712. doi: 10.3945/jn.109.113753. [DOI] [PubMed] [Google Scholar]
- 78.Rabot S, Rafter J, Rijkers GT, Watzl B, Antoine JM. Guidance for substantiating the evidence for beneficial effects of probiotics: impact of probiotics on digestive system metabolism. J Nutr. 2010;140:677–689. doi: 10.3945/jn.109.113738. [DOI] [PubMed] [Google Scholar]
- 79.Chen CC, Kong MS, Lai MW, Chao HC, Chang KW, Chen SY, et al. Probiotics have clinical, microbiologic and immunologic efficacy in acute infectious diarrhea. Pediatr Infect Dis J. 2010;29:135–138. doi: 10.1097/inf.0b013e3181b530bf. [DOI] [PubMed] [Google Scholar]
- 80.Savino F, Pelle E, Palumeri E, Oggero R, Miniero R. Lactobacillus reuteri (American Type Culture Collection Strain 55730) versus simethicone in the treatment of infantile colic: a prospective randomized study. Pediatrics. 2007;119:124–130. doi: 10.1542/peds.2006-1222. [DOI] [PubMed] [Google Scholar]
- 81.Osborn DA, Sinn JK. Probiotics in infants for prevention of allergic disease and food hypersensitivity. Cochrane Database Syst Rev. 2007:6475. doi: 10.1002/14651858.CD006475.pub2. [DOI] [PubMed] [Google Scholar]
- 82.Alfaleh K, Anabrees J, Bassler D. Probiotics reduce the risk of necrotizing enterocolitis in preterm infants: a meta-analysis. Neonatology. 2010;97:93–99. doi: 10.1159/000235684. [DOI] [PubMed] [Google Scholar]
- 83.Sethi T. Probiotics in pediatric care. Explore. 2009;5:245–249. doi: 10.1016/j.explore.2009.05.008. [DOI] [PubMed] [Google Scholar]
- 84.Sela DA, Chapman J, Adeuya A, Kim JH, Chen F, Whitehead TR, et al. The genome sequence of Bifidobacterium longum subsp. infantis reveals adaptations for milk utilization within the infant microbiome. Proc Natl Acad Sci USA. 2008;105:18964–18969. doi: 10.1073/pnas.0809584105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lebeer S, Vanderleyden J, De Keersmaecker SC. Host interactions of probiotic bacterial surface molecules: comparison with commensals and pathogens. Nat Rev Microbiol. 2010;8:171–184. doi: 10.1038/nrmicro2297. [DOI] [PubMed] [Google Scholar]
- 86.Lin PW, Myers LE, Ray L, Song SC, Nasr TR, Berardinelli AJ, et al. Lactobacillus rhamnosus blocks inflammatory signaling in vivo via reactive oxygen species generation. Free Radic Biol Med. 2009;47:1205–1211. doi: 10.1016/j.freeradbiomed.2009.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ventura M, O'Flaherty S, Claesson MJ, Turroni F, Klaenhammer TR, van Sinderen D, et al. Genome-scale analyses of health-promoting bacteria: probiogenomics. Nat Rev Microbiol. 2009;7:61–71. doi: 10.1038/nrmicro2047. [DOI] [PubMed] [Google Scholar]
- 88.Lin PW, Nasr TR, Berardinelli AJ, Kumar A, Neish AS. The probiotic Lactobacillus GG may augment intestinal host defense by regulating apoptosis and promoting cytoprotective responses in the developing murine gut. Pediatr Res. 2008;64:511–516. doi: 10.1203/PDR.0b013e3181827c0f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Bäckhed F, Crawford PA. Coordinated regulation of the metabolome and lipidome at the host-microbial interface. Biochimica Et Biophysica Acta. 2010;1801:240–245. doi: 10.1016/j.bbalip.2009.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kumar A, Wu H, Collier-Hyams LS, Kwon YM, Hanson JM, Neish AS. The bacterial fermentation product butyrate influences epithelial signaling via reactive oxygen species-mediated changes in cullin-1 neddylation. J Immunol. 2009;182:538–546. doi: 10.4049/jimmunol.182.1.538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Fava F, Lovegrove JA, Gitau R, Jackson KG, Tuohy KM. The gut microbiota and lipid metabolism: implications for human health and coronary heart disease. Curr Med Chem. 2006;13:3005–3021. doi: 10.2174/092986706778521814. [DOI] [PubMed] [Google Scholar]
- 92.de Roos NM, Katan MB. Effects of probiotic bacteria on diarrhea, lipid metabolism and carcinogenesis: a review of papers published between 1988 and 1998. Am J Clin Nutr. 2000;71:405–411. doi: 10.1093/ajcn/71.2.405. [DOI] [PubMed] [Google Scholar]
- 93.Girard SA, Bah TM, Kaloustian S, Lada-Moldovan L, Rondeau I, Tompkins TA, et al. Lactobacillus helveticus and Bifidobacterium longum taken in combination reduce the apoptosis propensity in the limbic system after myocardial infarction in a rat model. Br J Nutr. 2009;102:1420–1425. doi: 10.1017/S0007114509990766. [DOI] [PubMed] [Google Scholar]
- 94.Shane AL, Cabana MD, Vidry S, Merenstein D, Hummelen R, Ellis CL, et al. Guide to designing, conducting, publishing and communicating results of clinical studies involving probiotic applications in human participants. Gut Microbes. 2010;1:243–253. doi: 10.4161/gmic.1.4.12707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sanders ME, Akkermans LMA, Haller D, Hammerman C, Heimbach JT, Hörmannsperger G, et al. Safety assessment of probiotics for human use. Gut Microbes. 2010;1:164–185. doi: 10.4161/gmic.1.3.12127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kukkonen K, Savilahti E, Haahtela T, Juntunen-Backman K, Korpela R, Poussa T, et al. Long-term safety and impact on infection rates of postnatal probiotic and prebiotic (synbiotic) treatment: randomized, doubleblind, placebo-controlled trial. Pediatrics. 2008;122:8–12. doi: 10.1542/peds.2007-1192. [DOI] [PubMed] [Google Scholar]
- 97.Vlieger AM, Robroch A, van Buuren S, Kiers J, Rijkers G, Benninga MA, et al. Tolerance and safety of Lactobacillus paracasei ssp. paracasei in combination with Bifidobacterium animalis ssp. lactis in a prebiotic-containing infant formula: a randomised controlled trial. Br J Nutr. 2009;102:869–875. doi: 10.1017/S0007114509289069. [DOI] [PubMed] [Google Scholar]
- 98.Cannon JP, Lee TA, Bolanos JT, Danziger LH. Pathogenic relevance of Lactobacillus: a retrospective review of over 200 cases. Eur J Clin Microbiol Infect Dis. 2005;24:31–40. doi: 10.1007/s10096-004-1253-y. [DOI] [PubMed] [Google Scholar]
- 99.Lherm T, Monet C, Nougière B, Soulier M, Larbi D, Le Gall C, et al. Seven cases of fungemia with Saccharomyces boulardii in critically ill patients. Intensive Care Med. 2002;28:797–801. doi: 10.1007/s00134-002-1267-9. [DOI] [PubMed] [Google Scholar]
- 100.Sullivan A, Nord CE. Probiotic lactobacilli and bacteraemia in Stockholm. Scand J Infect Dis. 2006;38:327–331. doi: 10.1080/00365540500449826. [DOI] [PubMed] [Google Scholar]
- 101.Salminen MK, Tynkkynen S, Rautelin H, Saxelin M, Vaara M, Ruutu P, et al. Lactobacillus bacteremia during a rapid increase in probiotic use of Lactobacillus rhamnosus GG in Finland. Clin Infect Dis. 2002;35:1155–1160. doi: 10.1086/342912. [DOI] [PubMed] [Google Scholar]
- 102.Yamazaki S, Machii K, Tsuyuki S, Momose H, Kawashima T, Ueda K. Immunological responses to monoassociated Bifidobacterium longum and their relation to prevention of bacterial invasion. Immunology. 1985;56:43–50. [PMC free article] [PubMed] [Google Scholar]
- 103.Abe F, Muto M, Yaeshima T, Iwatsuki K, Aihara H, Ohashi Y, et al. Safety evaluation of probiotic bifidobacteria by analysis of mucin degradation activity and translocation ability. Anaerobe. 2010;16:131–136. doi: 10.1016/j.anaerobe.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 104.McNaught CE, Woodcock NP, MacFie J, Mitchell CJ. A prospective randomised study of the probiotic Lactobacillus plantarum 299V on indices of gut barrier function in elective surgical patients. Gut. 2002;51:827–831. doi: 10.1136/gut.51.6.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Liu C, Zhang ZY, Dong K, Yuan JP, Guo XK. Antibiotic resistance of probiotic strains of lactic acid bacteria isolated from marketed foods and drugs. Biomed Environ Sci. 2009;22:401–412. doi: 10.1016/S0895-3988(10)60018-9. [DOI] [PubMed] [Google Scholar]
- 106.Kiwaki M, Sato T. Antimicrobial susceptibility of Bifidobacterium breve strains and genetic analysis of streptomycin resistance of probiotic B. breve strain Yakult. Int J Food Microbiol. 2009;134:211–215. doi: 10.1016/j.ijfoodmicro.2009.06.011. [DOI] [PubMed] [Google Scholar]
- 107.Marcobal A, Underwood MA, Mills DA. Rapid determination of the bacterial composition of commercial probiotic products by terminal restriction fragment length polymorphism analysis. J Pediatr Gastroenterol Nutr. 2008;46:608–611. doi: 10.1097/MPG.0b013e3181660694. [DOI] [PubMed] [Google Scholar]
- 108.Walker AW, Duncan SH, Harmsen HJ, Holtrop G, Welling GW, et al. The species composition of the human intestinal microbiota differs between particle-associated and liquid phase communities. Environ Microbiol. 2008;10:3275–3283. doi: 10.1111/j.1462-2920.2008.01717.x. [DOI] [PubMed] [Google Scholar]
- 109.Hartman AL, Lough DM, Barupal DK, Fiehn O, Fishbein T, Zasloff M, et al. Human gut microbiome adopts an alternative state following small bowel transplantation. Proc Natl Acad Sci USA. 2009;106:17187–17192. doi: 10.1073/pnas.0904847106. [DOI] [PMC free article] [PubMed] [Google Scholar]