Summary
Proapoptotic Bcl-2 family members have been proposed to play a central role in regulating apoptosis. However, mice lacking bax display limited phenotypic abnormalities. As presented here, bak–/– mice were found to be developmentally normal and reproductively fit and failed to develop any age-related disorders. However, when Bak-deficient mice were mated to Bax-deficient mice to create mice lacking both genes, the majority of bax–/–bak–/– animals died perinatally with fewer than 10% surviving into adulthood. bax–/–bak–/– mice displayed multiple developmental defects, including persistence of interdigital webs, an imperforate vaginal canal, and accumulation of excess cells within both the central nervous and hematopoietic systems. Thus, Bax and Bak have overlapping roles in the regulation of apoptosis during mammalian development and tissue homeostasis.
Introduction
Programmed cell death (apoptosis) plays a critical role in the regulation of development and tissue homeostasis. In addition, apoptosis plays an important role in the elimination of damaged or infected cells (Vaux and Korsmeyer, 1999). Two major apoptotic pathways have been identified (Strasser et al., 1995; Green, 2000). The extrinsic cell death pathway provides multicellular organisms a means to eliminate cells through instruction. In the extrinsic pathway, apoptosis is initiated through ligand binding to cell surface receptors expressed on the cell that will subsequently die. In contrast, a cell-intrinsic death pathway has been defined in which apoptosis is initiated within the cell.
The best characterized of the receptors involved in the extrinsic death pathway are members of the TNF receptor family, such as Fas and TNF-R1 (Nagata, 1997). Once engaged by ligand, these receptors initiate the formation of a death-inducing signaling complex, one essential component of which is the adaptor molecule FADD. FADD in turn recruits Caspase 8. Upon recruitment to the signaling complex, Caspase 8 undergoes autoproteolytic activation. Once activated, Caspase 8 can initiate both the activation of additional caspases and the degradation of intracellular substrates. These events result in the orderly degradation of intracellular contents and cell death.
The cell-intrinsic apoptotic pathway is initiated when the outer mitochondrial membrane loses its integrity, resulting in the release of cytochrome c and other apoptotic regulatory proteins into the cytosol (Green, 2000). In the cytosol, released cytochrome c binds to Apaf-1. Apaf-1 in the presence of cytochrome c and dATP recruits Caspase 9 to form a multimeric complex. Proteolytic activation of Caspase 9 in turn leads to the initiation of a caspase cascade that includes Caspase 3, resulting in the morphologic features of apoptotic cell death.
Bcl-2 proteins are evolutionarily conserved regulators of apoptosis (Adams and Cory, 1998). The Bcl-2 family is composed of both proapoptotic and anti-apoptotic family members. The best characterized anti-apoptotic proteins, Bcl-2 and Bcl-xL, appear to directly or indirectly preserve the integrity of the outer mitochondrial membrane, thus preventing cytochrome c release and cell death initiation through the cell intrinsic death pathway (Kluck et al., 1997; Vander Heiden et al., 1997; Yang et al., 1997). In contrast, the proapoptotic family members Bax, Bid, and Bak have been shown to promote cyto-chrome c release (Jurgensmeier et al., 1998; Li et al., 1998; Luo et al., 1998; Kluck et al., 1999; Wei et al., 2000). Mice lacking Bcl-2, Bcl-xL, Bax, or Bid have been reported (Nakayama et al., 1993; Veis et al., 1993; Knudson et al., 1995; Motoyama et al., 1995; Yin et al., 1999). Bcl-2-deficient mice demonstrate defects in renal development and undergo premature loss of lymphoid cells and melanocytes, resulting in immunodeficiency and loss of hair pigmentation, respectively (Nakayama et al., 1993; Veis et al., 1993). Animals deficient in Bcl-xL die during embryogenesis, displaying a massive increase in apoptosis in developing postmitotic cells of the embryonic spinal cord, brain stem, and dorsal root ganglia (Motoyama et al., 1995). Extensive apoptosis is also observed within the liver and hematopoietic systems.
Animals lacking the proapoptotic family member Bax are viable, demonstrating that Bax is not required for development (Knudson et al., 1995). However, male bax–/– mice are sterile due to a developmental arrest in spermatogenesis that is associated with a paradoxical increase in the death of spermatogonia. Bax-deficient animals also have increased numbers of sympathetic and motor neurons. During in vitro culture, sympathetic neurons from Bax-deficient animals are resistant to cell death upon trophic factor deprivation.
Although Bcl-2 proteins are thought primarily to be regulators of the intrinsic apoptotic pathway, recent evidence suggests that proapoptotic Bcl-2 family members can act to promote cell death in the extrinsic pathway as well. Bid has been shown to be a substrate for Caspase 8 (Li et al., 1998; Luo et al., 1998). Caspase 8 cleavage of Bid results in translocation of truncated Bid to the mitochondria where it promotes cytochrome c release. Surprisingly, Bid-deficient mice are viable without obvious developmental abnormalities (Yin et al., 1999). However, bid–/– mice display decreased hepatocellular death in response to in vivo administration of an anti-Fas antibody.
Bak represents another proapoptotic Bcl-2 family member that is closely related to Bax and is roughly comparable to Bax as assayed by in vitro systems (Chittenden et al., 1995; Orth and Dixit, 1997; Tao et al., 1997). In addition, Bak is expressed in essentially all organs, suggesting that it may be a regulator of apoptosis in multiple cell types (Farrow et al., 1995; Kiefer et al., 1995; Krajewski et al., 1996). To investigate this possibility, we generated Bak-deficient mice by gene targeting. Bak-deficient mice failed to demonstrate observable developmental abnormalities. Both male and female animals were capable of reproduction. Bak-deficient mice did not develop any reproducible abnormalities as they aged, and cells isolated from these mice behaved normally when apoptosis was initiated through either the cell-intrinsic or cell-extrinsic pathways. These data suggest that either Bak is not an essential regulator of apoptosis or the function of Bak is redundant with that of other broadly expressed proapoptotic Bcl-2 family members.
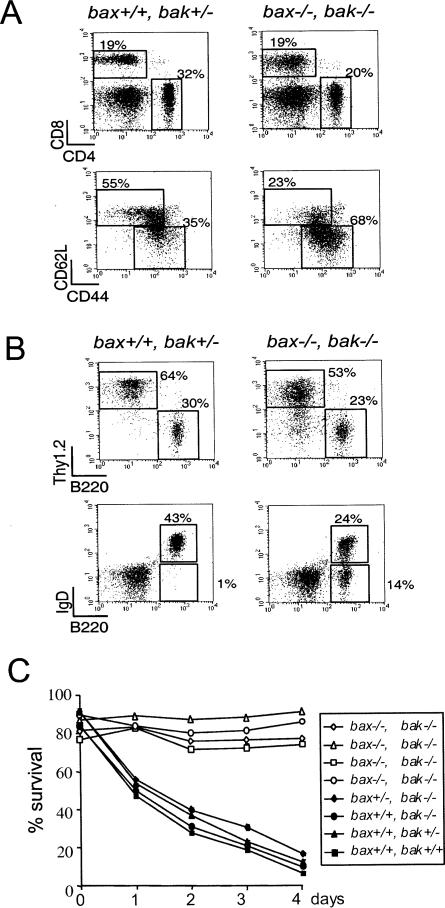
Since Bax also displays a widespread tissue distribution and bax–/– mice have relatively limited phenotypic abnormalities (Oltvai et al., 1993; Knudson et al., 1995), we undertook to breed bax–/–bak–/– mice. The majority of bax–/–bak–/– mice died perinatally with fewer than 10% of the animals reaching adulthood. bax–/–bak–/– adult mice displayed multiple phenotypic abnormalities. Adult bax–/–bak–/– mice retained interdigital webs throughout life, and females had imperforate vaginas. The animals displayed multiple neurologic abnormalities, including deafness, circling behavior, and persistence of a large number of undifferentiated cells in the periventricular progenitor zones. The hematopoietic system was also grossly abnormal, with increased numbers of hematopoietic progenitors and elevated numbers of granulocytes and lymphocytes. Progressive accumulation of mature B and T cells led to massive enlargement of the spleen and lymph nodes and infiltration of parenchymal organs. In in vitro culture, cells from bax–/–bak–/– mice were resistant to death induced by neglect or irradiation but could initiate apoptosis in response to Fas cross-linking. Together, these data suggest that Bax and Bak represent redundant proteins involved in the regulation of apoptosis. Simultaneous deficiency in both Bak and Bax results in abnormalities associated with the accumulation of cells that normally die either during development or during the regulation of tissue homeostasis.
Results
Generation of bak–/– Mice
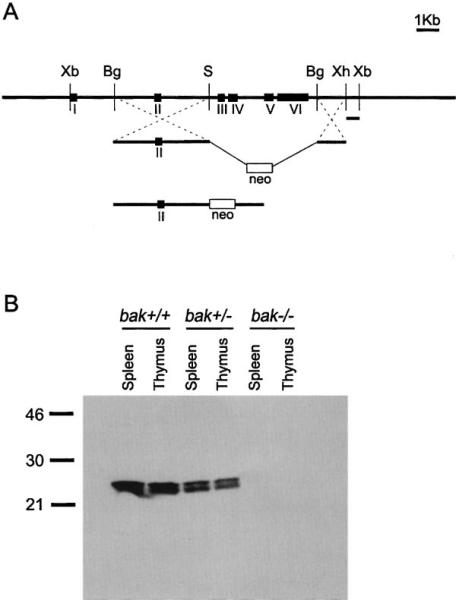
A targeting construct was made that eliminated exons III–VI, which encode the Bcl-2 homology (BH) domains 1–3 of the bak gene (Figure 1A). The linearized construct was electroporated into R1 ES cells, and transfectants were selected by G418 resistance. Four independent homologous recombinants were obtained, confirmed by Southern blotting, and injected into C57BL/6 blastocysts. Chimeric mice were identified and crossed with C57BL/6 mice to generate heterozygous Bak mutants. Heterozygous mice were crossed to generate homozygous Bak-deficient (bak–/–) mice. Of 77 offspring from initial heterozygote matings, 23 were wild type, 30 were heterozygous, and 24 were homozygous mutants. A lack of Bak expression in the homozygous mutants was confirmed by Western blot analysis (Figure 1B). The bak–/– mice appeared outwardly normal. Autopsy and microscopic evaluation of multiple tissues failed to reveal any significant lesions. Assays for cell survival in response to a variety of apoptotic stimuli revealed no statistical differences in in vitro survival of bak–/– cells in comparison to wild-type controls. Both bak–/– males and females were fertile and bred well. In over one year of observation, bak–/– animals have not developed any reproducible age-related disorders.
Figure 1. Generation of bak–/– Mice.
(A) The genomic organization of the bak gene, targeting strategy and the final targeting construct is shown. Restriction enzyme sites shown are (Xb), XbaI; (Bg), BglII; (S), SalI; and (Xh), XhoI.
(B) Western blot analysis of whole cell lysates from spleen and thymus of bak+/+, bak+/–, and bak–/– mice.
Generation of bax–/–bak–/– Mice
The above data suggested that either Bak represents a nonessential gene or that the function of Bak is redundant with other proapoptotic Bcl-2 family members. To investigate this latter possibility, we sought to determine whether breeding Bak and Bax mutations together would unmask more significant phenotypic abnormalities than those observed in either single deficient animal. To generate bax–/–bak–/– animals, bak+/– mice were crossed with bax+/– mice. Offspring that were bax+/–bak+/– were identified and interbred. Genotyping at weaning of offspring from these crosses revealed that bax–/–bak–/– animals were obtained at less than 10% of the expected Mendelian frequency. Furthermore, prior to weaning a number of dead pups were identified in these litters. Genotyping of these dead animals showed that many were bax–/–bak–/–. This suggested that bax–/–bak–/– animals were dying in the perinatal period. To assess this further, breeding pairs of bax+/–bak–/– animals were established and the offspring of six sequential litters were sacrificed at birth. Of 37 mice produced by intercrossing bax+/–bak–/– mice, 8 were found to be bax+/+bak–/–, 23 were bax+/–bak–/–, and 6 were bax–/–bak–/–. Careful observation of additional litters from these matings revealed individual animals that failed to nurse appropriately and which were frequently found isolated from their siblings and mother in the cage. Genotyping of these animals demonstrated that they were bax–/–bak–/– mice. Although frequent observation allowed us to reproducibly observe these animals prior to death, the majority died within 48 hr of birth.
Persistence of Interdigital Tissue in Adult bax–/–bak–/– Mice
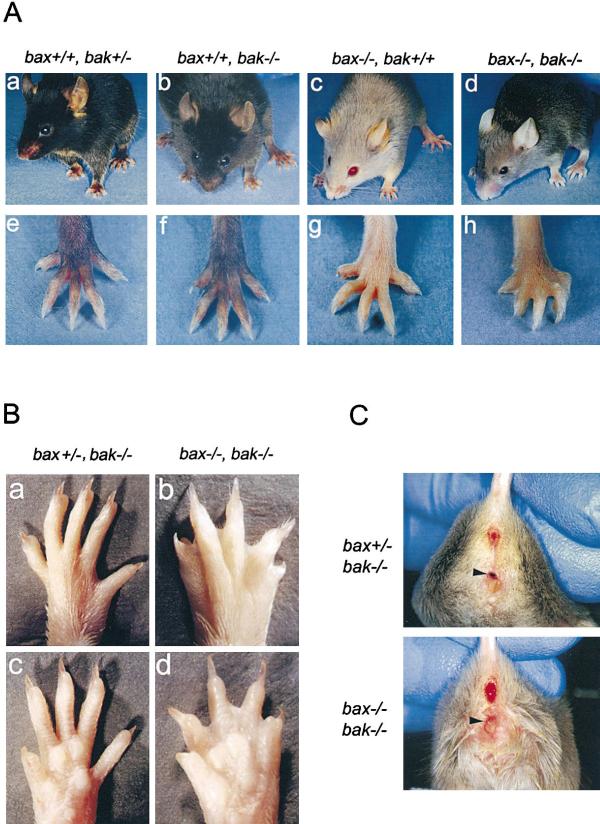
A significant number of bax–/–bak–/– mice survived into adulthood, and these adult animals were characterized in detail at 12–16 weeks of age. Adult bax–/–bak–/– mice displayed several phenotypic abnormalities upon gross examination, the most striking of which is that the bax–/–bak–/– mice retained interdigital webs on both their fore and rear paws, as shown in Figure 2A. bax+/+bak+/–, bax+/+bak–/–, and bax–/–bak+/+ mice did not have interdigital webs. In Figure 2B, the dorsal and palmar views of paws from a bax+/–bak–/– mouse and from a bax–/–bak–/– mouse are shown. The presence of just one copy of wild-type bax completely eliminated interdigital webbing. A single wild-type copy of bak also eliminated the webbing (data not shown).
Figure 2. Gross Anatomical Phenotypes of bax–/–bak–/– Mice.
(A) Photographs of bax+/+bak+/– (a and e), bax+/+bak–/– (b and f), bax–/–bak+/+ (c and g), and bax–/–bak–/– (d and h) mice.
(B) Dorsal and ventral views of paws from bax+/–bak–/– (a and c) and bax–/–bak–/– (b and d).
(C) Photographs of vaginal openings from bax+/–bak–/– and bax–/–bak–/– mice. Arrows point to external vaginal region.
Failure to Develop an External Vaginal Introitis
A second visible abnormality of bax–/–bak–/– mice was that all adult females had an imperforate vagina (Figure 2C). The presence of an imperforate vagina was not observed in either bax–/– or bak–/– mice (data not shown). Again, the presence of a single wild-type allele of either bax or bak was sufficient to reproducibly eliminate this anatomic abnormality (Figure 2C and data not shown).
Neurological Abnormalities
bax–/–bak–/– mice were all unresponsive to auditory stimulation. In addition, most double mutant mice displayed circling behavior when exposed to external stress. External stress, such as cage changing, also appeared to elicit seizure activity in a number of bax–/–bak–/– mice. However, the exencephaly and cranial bone defects that have been observed at birth in Caspase 9–/– and/or Apaf-1–/– (Cecconi et al., 1998; Kuida et al., 1998; Yoshida et al., 1998; Honarpour et al., 2000) mice were not observed in bax–/–bak–/– mice.
Histological Analysis of Brain Tissue from bax–/–bak–/– Mice
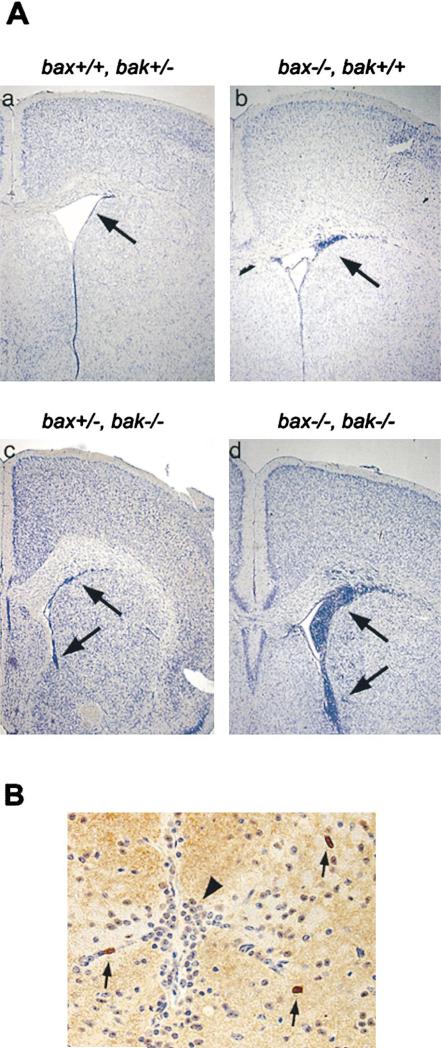
To assess the central nervous system of bax–/–bak–/– mice, brains were dissected from these mice as well as control littermates with other genotypes. Overall, brains of the bax–/–bak–/– mutant mice were larger than those of other genotypes examined (data not shown). Upon histological analysis, there appeared to be a generalized increase in the number of neurons in multiple regions of the brain. However, the most striking abnormality of the bax–/–bak–/– brains was the large accumulation of small neuronal cells with dense chromatin staining observed in the periventricular region (Figure 3A). Similar accumulations of such cells are found during normal embryogenesis in the same region (equivalent to the ganglionic eminence). This region is known to be populated by neural stem cells and an area where during terminal division some daughter cells differentiate into postmitotic neurons while other daughter cells undergo programmed cell death (Bayer and Altman, 1991; Clarke et al., 2000). In adult bax–/–bak–/– mice, similar populations of small cells were also found in other locations where neural stem cells are commonly found, including the hippocampus, cerebellum, and olfactory bulb (data not shown). In addition, similar accumulations of small cells were found in regions not commonly known to contain neural stem cells in adult mice, such as the dorsal mid-line of the mesencephalon (Figure 3B). A gene dosage effect for this phenotype was evident as both single bax–/– and single bak–/– brains displayed discrete accumulations of small cells in the periventricular area (Figure 3A). However, bax–/–bak–/– mice had a dramatic increase in both the number and distribution of these cells when compared to animals lacking either Bax or Bak alone.
Figure 3. Periventricular Accumulations of Small Neuronal Cells in the Brains of bax–/–bak–/– Mice.
(A) Histological sections of brain tissue from bax+/+bak+/– (a), bax–/–bak+/+ (b), bax+/–bak–/– (c), and bax–/–bak–/– (d) mice at 25× magnification. Each set is at approximately the same level and includes the anterior basal ganglia and the corpus callosum. Arrow points to the periventricular region in all sections.
(B) Histological section of the dorsal midline of the mesencephalon from bax–/–bak–/– mouse at a 400× magnification, stained with anti-CD45RB. Arrows point to CD45+ cells. Arrowhead points to abnormal accumulation of small neuronal cells.
Given the hematopoietic defects seen elsewhere in the bax–/–bak–/– mice (see below), we explored the possibility that the ectopic cells in the CNS were of hematopoietic origin. Tissue sections from bax–/–bak–/– brains were stained with anti-CD45, a marker expressed on a broad spectrum of hematopoietic cells. While immunoreactive CD45+ hematopoietic cells were identified within blood vessels, the small cell population in the neural parenchyma of bax–/–bak–/– mice was CD45– (Figure 3B).
Absence of Developmental Defects in Other Parenchymal Organs
Despite the abnormalities observed in the central nervous system and reproductive systems of bax–/–bak–/– mice, most other organs appeared to be formed normally during development. No gross anatomical or histologic abnormalities were observed in the kidney, liver, lung, heart, pancreas, or bladder in either bak–/– or bax–/– bak–/– animals. To confirm that these internal organs were also functioning at physiologic levels, standard blood chemistries were determined for bax–/–bak–/– mice and appropriate littermate controls. No consistent abnormalities in electrolytes, blood glucose, renal function, or hepatic function tests were found in either bak–/– or bax–/–bak–/– animals (data not shown). bax–/– mice display defective spermatogenesis that is associated with a developmental arrest of spermatogonia. Histology of the testes from bax–/–bak–/– male mice was indistinguishable from that observed in bax–/– littermates (data not shown).
Increased Numbers of Myeloid and Lymphoid Cells in bax–/–bak–/– Animals
Blood cell counts revealed markedly elevated white blood cell counts in bax–/–bak–/– mice (Table 1). Statistically significant increases in both myeloid cells and lymphoid cells were present. In addition, bax–/–bak–/– animals displayed mild anemia and thrombocytopenia that were not observed in either bax–/– or bak–/– animals. Hematopoietic colony assays revealed consistent and statistically significant increases in the number of myeloid colony forming units (CFU)/106 bone marrow cells (Table 2). A gene dosage effect also appeared to be present as the total numbers of CFU increased in rough proportion to the total number of mutant bak and bax alleles present. Mild elevations in erythroid and megakaryocyte CFU were also observed.
Table 1.
Hematological Profile of bax-/-bak-/- and Control Mice
| Genotype | WBC | Neutrophils | Lymphocytes | RBC |
|---|---|---|---|---|
| bax-/-bak-/- | ||||
| 1 | 15.3 | 4.1 | 10.0 | 7.4 |
| 2 | 24.2 | 2.5 | 20.4 | 4.6 |
| 3 | 28.7 | 2.0 | 26.0 | 9.5 |
| 4 | 39.2 | 5.1 | 31.9 | 8.8 |
| bax+/-bak-/- | ||||
| 1 | 11.1 | 3.7 | 5.5 | 10.1 |
| 2 | 8.6 | 1.3 | 6.5 | 9.5 |
| bax+/+bak-/- | ||||
| 1 | 3.7 | 1.2 | 2.4 | 9.8 |
| 2 | 4.6 | 0.7 | 3.6 | 9.8 |
| bax+/+bak+/- | ||||
| 1 | 6.4 | 1.6 | 4.7 | 9.8 |
| 2 | 5.7 | 1.3 | 4.2 | 8.8 |
| bax+/+bak+/+ | ||||
| 1 | 4.7 | 0.7 | 3.8 | 9.9 |
| 2 | 4.8 | 0.5 | 4.0 | 9.3 |
| 3 | 4.3 | 1.3 | 2.9 | 10.3 |
The number of white blood cells (WBC), neutrophils, lymphocytes (× 103/μl), and red blood cells (RBC, × 106/μl) was determined using a Hemavet 1500 apparatus.
Table 2.
Bone Marrow Hematopoietic Progenitor Assay
| Genotype | Myeloid | Erythroid/Meg | Total |
|---|---|---|---|
| bax-/-bak-/- | |||
| 1 | 297 | 98 | 395 |
| 2 | 250 | 93 | 343 |
| 3 | 302 | 88 | 390 |
| bax +/- bak -/- | 208 | 111 | 319 |
| bax +/+ bak -/- | 154 | 104 | 258 |
| bax+/+bak+/- | 172 | 74 | 246 |
| bax+/+bak+/+ | 144 ± 12 | 68 ± 5 | 211 ± 7 |
Bone marrow cells were isolated from bax-/-bak-/- and control mice and cultured in complete methylcellulose medium for 5 days. CFU were scored based on colony morphology and cytological staining. Myeloid CFU were a combination of macrophage, granulocyte-erythrocyte-megakaryocyte-macrophage, granulocyte-macrophage, and granulocyte CFU. Likewise, the erythroid and megakaryocyte CFU were combined. For the wild-type group, data is expressed as the mean ± standard deviation.
Massive Lymphoid Accumulation in bax–/–bak–/– Mice
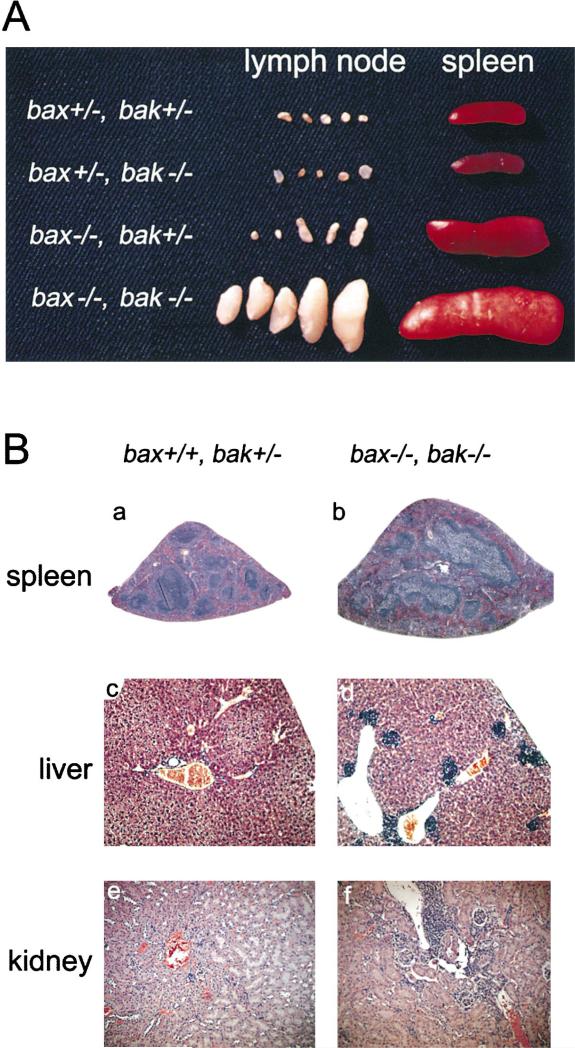
The majority of the increase in the white blood cell count of bax–/–bak–/– animals was due to an elevation in the number of lymphocytes. bax–/–bak–/– mice had increased numbers of lymphocytes not only within the circulation but also within the peripheral lymphoid organs. As described previously (Knudson et al., 1995), animals deficient in Bax had spleens and lymph nodes that were slightly enlarged over wild-type mice (Figure 4A). However, bax–/–bak–/– mice have a significant further enlargement of the peripheral lymphoid organs. Dramatic splenomegaly was observed in all bax–/–bak–/– mutant mice examined, and Figure 4B shows a histologic section of the spleen from a bax–/–bak–/– mouse. The bax–/–bak–/– spleen had an expanded red pulp region that, by histological analysis, contained a large increase in the number of plasma cells and histiocytes. A pronounced hyperplasia of the white pulp was also observed.
Figure 4. Splenomegaly, Lymphadenopathy, and Lymphoid Infiltration of Peripheral Organs in bax–/–bak–/– Mice.
(A) Photograph of spleens and lymph nodes from bax–/–bak–/– and control mice.
(B) Tissue sections of spleens (a and b) at 2× magnification, livers (c and d) at 10× magnification, and kidneys (e and f) at 10× magnification from bax+/+bak+/– (a, c, and e) and bax–/–bak–/– (b, d, and f) mice.
In addition to lymphoid accumulation in the peripheral lymphoid organs, lymphocytic infiltration of parenchymal organs was also observed. Lymphocyte infiltration in the liver was periportal as well as periarterial (Figure 4B). The kidney also displayed perivascular lymphocytic infiltration. Several older bax–/–bak–/– mice also displayed varying degrees of glomerulonephropathy characterized by hypercellularity.
Lymphocytes that Accumulated in bax–/–bak–/– Mice Had a Memory Cell Phenotype
To determine the phenotypic properties of the lymphocytes that accumulate in bax–/–bak–/– mice, lymphocytes from the peripheral lymph nodes and spleens were phenotyped by flow cytometry. Lymph nodes from both control and bax–/–bak–/– animals had similar frequencies of CD4 and CD8 T cells (Figure 5A). However, the T cells that accumulate in the bax–/–bak–/– animals were skewed toward a memory cell phenotype. The T cells were small in size as measured by forward angle light scatter, lacked expression of activation markers such as CD25, had increased expression of the memory cell marker, CD44, and decreased expression of the peripheral addressin, CD62L (Figure 5A). The percentages of B cells were also found to be similar in bax–/–bak–/– lymph nodes as compared to control lymph nodes. However, the B220+ B cells present in the lymph node were skewed toward a B220bright IgD– phenotype, indicating the presence of an increased number of class-switched or memory B cells (Figure 5B). Similar data were obtained in spleen (data not shown). T and B cells isolated from either bax–/– or bak–/– single-deficient mice did not display increased percentages of cells with a memory phenotype.
Figure 5. Peripheral Lymphocytes that Accumulate in bax–/–bak–/– Mice Are Enriched in Memory Cells and Fail to Die by Neglect Peripheral lymphocytes from lymph node cells were analyzed by flow cytometry.
(A and B) CD4 and CD8 expression (A) and Thy1.2 and B220 expression (B). Gated T cells were also analyzed for the expression of CD44 and CD62L. Gated B cells were also analyzed for expression of IgD.
(C) Peripheral lymphocytes from spleens of bax–/–bak–/– (open symbols) and control mice (closed symbols) were cultured for 4 days. Cells were stained with PI and subjected to FACS analysis at the indicated times. Samples were assayed in triplicate.
Lymphocytes from bax–/–bak–/– Mice Were Resistant to Death by Neglect
Of the defects observed in adult bax–/–bak–/– mice, only the lymphocyte accumulation was not also observed in animals sacrificed at birth. Thus, the combined deficiency of Bax and Bak led to the accumulation of lymphocytes as the animals aged. The increase in lymphocyte number could occur either as a consequence of increased production or increased survival. The small size and lack of expression of activation markers such as CD25 on lymphocytes in both lymphoid and nonlymphoid organs of the bax–/–bak–/– animals suggest that the cells accumulate as a result of a defect in apoptosis. To address this issue, splenocytes from bax–/–bak–/– and control mice were isolated, and cell survival in suspension culture was analyzed over the ensuing 4 days. Figure 5C shows the percentage of live cells as determined by propidium iodide staining at daily intervals. Splenocytes from bax–/–bak–/– mice showed markedly enhanced survival, demonstrating a defect in death by neglect. In contrast, cells from Bak-deficient animals and animals expressing both Bax and Bak underwent apoptosis when placed in single cell culture. The ability of bax–/– lymphoid cells to undergo death by neglect has been previously analyzed and found to be similar to wild-type mice (Knudson, et al., 1995 and data not shown).
Impaired Radiation- and Etoposide-Induced but Normal Fas-Induced Death in bax–/–bak–/– Mice
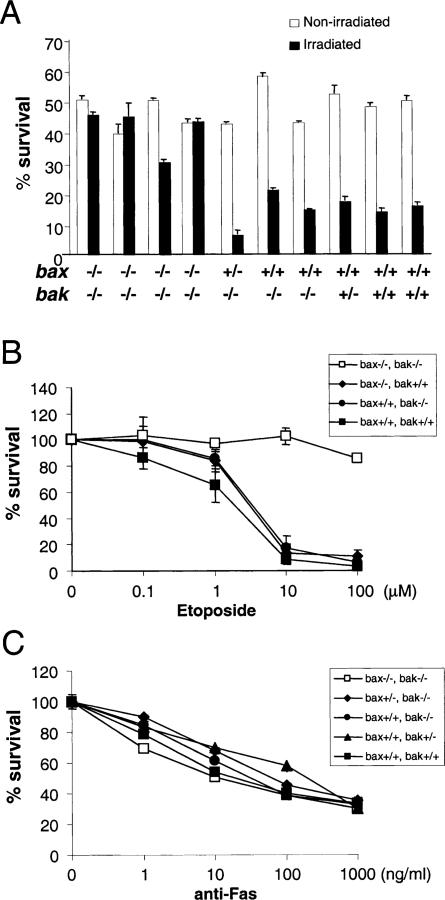
One of the classic assays for apoptosis is the radiation-induced death of thymocytes (Wyllie, 1980). Irradiation of thymocytes has been shown to initiate apoptosis in a p53-dependent fashion (Clarke et al., 1993; Lowe et al., 1993). Cell death appears to result from activation of the cell-intrinsic pathway involving mitochondria (Kuida et al., 1998). Neither bax–/– nor bak–/– animals displayed defects in radiation-induced apoptosis (Figure 6A and Knudson et al., 1995). Thymocytes from bax–/–bak–/– animals displayed enhanced survival compared to littermate controls when exposed to gamma irradiation. Thymocyte cell death following treatment with the chemotherapeutic agent etoposide is also reported to be p53-dependent. Thymocytes from bax–/–bak–/– were found to be resistant to treatment with etoposide (Figure 6B), whereas bax–/– and bak–/– single deficient thymocytes were sensitive to this treatment. In contrast, when thymocytes from bax–/–bak–/– and control mice were subjected to cross-linking of the Fas receptor, bax–/–bak–/– thymocytes showed sensitivity similar to that of the control thymocytes (Figure 6C). This indicates that, at least in this tissue, the cell-extrinsic pathway is not dependent on the proapoptotic activity of either Bax or Bak.
Figure 6. Thymocyte Death Assays in bax–/–bak–/– Mice.
(A) Thymocytes from bax–/–bak–/– mice and control mice were treated with 500 rad of γ irradiation (closed bars) or left untreated (open bars). Cells were stained with PI and subjected to FACS analysis after 24 hr of culture. Mean and standard deviations of triplicate samples from each mouse are presented.
(B) Thymocytes were cultured with the indicated amounts of etoposide. Cells were stained with PI and subjected to FACS analysis after 24 hr of culture. Mean and standard deviations of triplicate samples are presented.
(C) Thymocytes were cultured with indicated amounts of anti-Fas antibody. Cells were stained with PI and subjected to FACS analysis after 24 hr of culture. Mean and standard deviations of triplicate samples are presented.
Discussion
The phenotype of bax–/–bak–/– mice provides a clear demonstration of the importance of proapoptotic Bcl-2 proteins in the regulation of both mammalian development and tissue homeostasis. The combined deficiency of Bax and Bak has a significantly more severe and diverse phenotype than deficiency in either gene alone. Bak deficiency alone has no demonstrable phenotype that we have as yet been able to uncover. Bax-deficient animals are viable and their major developmental defect is male sterility that is associated with a developmental arrest of the spermatogonia compartment with a resultant increase in apoptotic germ cell death (Knudson et al., 1995). Although slight increases in sympathetic neurons, motor neurons, and lymphocytes have been described in the absence of Bax, bax–/– mice appear to be behaviorally normal, they fail to develop any significant lymphoproliferative complications, and the females are capable of reproduction (Knudson et al., 1995; Deckwerth et al., 1996). In contrast, bax–/–bak–/– animals had significant disruptions in the development of the digits, female reproductive tract, and the central nervous system. bax–/–bak–/– mice appear to have increased number of hematopoietic precursors and develop a progressive accumulation of mature lymphocytes. Lymphocytes that accumulate in bax–/–bak–/– mice displayed defects in death by neglect and were also resistant to radiation- and etoposide-induced apoptosis. However, the ability of bax–/–bak–/– thymocytes to initiate apoptosis in response to Fas signal transduction was comparable to that of wild-type thymocytes. Together, the data indicate that Bax and Bak have largely overlapping roles in regulating cell death initiated by activators of the intrinsic cell death pathway. However, at least in some tissues, the extrinsic cell death pathway can initiate apoptosis in the absence of both Bax and Bak.
The phenotype of bax–/–bak–/– mice overlaps the phenotypes described for mice lacking individual genes involved in the intrinsic cell death pathway. In fact, the bax–/–bak–/– mice in most respects have a more severe phenotype than those described for Apaf-1–/– or Caspase 9–/– mice (Cecconi et al., 1998; Kuida et al., 1998; Yoshida et al., 1998; Honarpour et al., 2000). The accumulation of lymphoid tissue and the failure of vaginal development observed in bax–/–bak–/– mice has not been described in adult Apaf-1–/– or Caspase 9–/– mice. The resorption of interdigital webs was completed during embryogenesis in both Apaf-1–/– and Caspase 9–/– mice. In addition, the adult mice deficient in Caspase 9 or Apaf-1 that have been analyzed do not demonstrate any morphologic abnormalities in their central nervous systems. In contrast, all of the bax–/–bak–/– mice that we have analyzed demonstrate persistence of increased numbers of neuronal cells in the periventricular progenitor regions.
Like Apaf-1–/– and Caspase 9–/– mice, the majority of bax–/–bak–/– mice died perinatally. However, unlike the Caspase 9–/– and Apaf-1–/– mice, we have not seen any evidence of exencephaly in the bax–/–bak–/– animals, nor have the defects in cranial bone formation reported for Apaf-1–/– mice been observed in the bax–/–bak–/– mice. Caspase 9–/– animals have been reported to have obstruction of the ventricles as a result of expansion in the size of the periventricular progenitor zones (Kuida et al., 1998). Although the periventricular progenitor zones persist into adulthood in the bax–/–bak–/– mice, hydrocephalus does not appear to be an explanation for the perinatal death of bax–/–bak–/– animals. In the bax–/–bak–/– mice, death seemed to occur because of a failure of the newborn animals to nurse. bax–/–bak–/– pups were frequently found still alive but neglected by their mother and separated from the rest of their litter. Attempts at foster mothering these animals have failed, suggesting that the bax–/–bak–/– animals have a defect in suckling or social behavior. This could result from the neuronal developmental abnormalities such as those that lead to the loss of response to auditory stimulation and the circling behavior observed in adults. These abnormalities are commonly seen when the development of neuroepithelial structures is abnormal (Alagramam et al., 1999). Abnormalities with optic and inner ear development have been reported in Apaf-1–/– mice (Honarpour et al., 2000). Alternatively, abnormalities in cranial bone formation can also result in hearing and behavioral abnormalities, and this might result from a milder form of the cranioschisis seen in Apaf-1–/– mice (Yoshida et al., 1998). Regardless, suckling is a complex behavioral trait, and the failure of bax–/–bak–/– mice to suckle effectively does not appear to have a simple, readily apparent mechanistic basis.
The developmental and homeostatic defects in the bax–/–bak–/– mice indicate that Bax and Bak are redundant regulators of the intrinsic cell death pathway. This redundancy of function is not observed in the other genes involved in the intrinsic cell death pathway, including Apaf-1, Caspase 9, and Caspase 3. The failure of bax–/–bak–/– mice to develop the exencephaly observed in Apaf-1–/– and Caspase 9–/– mice could result from additional redundancy among proapoptotic Bcl-2 family members in developing neural tissue or from compensatory changes in the expression of anti-apoptotic Bcl-2 family members within the central nervous system. Alternatively, lack of exencephaly may result from the ability of Apaf-1 and Caspase 9 to act as amplifiers of the cell death decision downstream of mitochondrial cytochrome c release and somehow compensate for the lack of Bax and Bak during the developmental elimination of excess neural cells.
The ability of some Apaf-1–/– and Caspase 9–/– mice to develop normally has been interpreted to suggest that there must be redundant apoptotic pathways that serve to cause the elimination of interdigital webs, remove excess neuroprogenitors, and prevent the accumulation of lymphoid cells (Kuida et al., 1998; Honarpour et al., 2000). The fact that these excess accumulations of cells persist in bax–/–bak–/– mice suggests that these additional mediators of cell death are dependent on Bax and/or Bak function. It has recently been described that other proteins in addition to cytochrome c can be released from mitochondria that can promote apoptosis. These include AIF, which upon release appears to translocate to the nucleus and promote DNA degradation (Susin et al., 1999), and Smac/DIABLO, which can block the ability of IAP proteins to act as caspase inhibitors (Du et al., 2000; Verhagen et al., 2000). The Bax- and/or Bak-dependent release of additional proapoptotic proteins such as AIF and Smac/DIABLO from mitochondria could potentially induce cell death in the absence of a functional cytochrome c/Apaf-1/Caspase 9 complex.
Bax and Bak have also been proposed as important mediators of the cell-extrinsic death pathway, acting as molecules that interact with the caspase substrate, Bid, to induce cytochrome c release from mitochondria (Eskes et al., 2000; Wei et al., 2000). At least in developing thymocytes, neither Bax nor Bak is required for Fas-induced death. Fas signal transduction also induces thymocyte death in Apaf-1–/– and Caspase 9–/– mice (Kuida et al., 1998; Yoshida et al., 1998). Thus, communication between the extrinsic and intrinsic pathways is not essential for Fas-mediated death in all tissues. Alternatively, additional proapoptotic Bcl-2-related proteins may also interact with Bid to carry out this function.
In summary, these data demonstrate the importance of proapoptotic Bcl-2 family members in the developmental involution of interdigital webs and of neuronal cells in the periventricular progenitor zones. The abnormal development of the female reproductive tract and the absence of hearing support previous studies that suggested that the function of Bcl-2 family genes may be important in the development of these complex tissues (Ishii et al., 1996; Rodriguez et al., 1997). Finally, the progressive accumulation of lymphoid cells demonstrates the importance of these genes in the control of tissue homeostasis in adults. A common feature of many of the excess cells that accumulate in the bax–/–bak–/– mice is that these are cells thought to be eliminated in normal animals as a result of the loss of cell survival signaling. Cell death induced by growth factor withdrawal (death by neglect) can be inhibited in multiple tissues by targeted transgene expression of anti-apoptotic Bcl-2 proteins. The data presented here demonstrate that death by neglect can also be impaired by the simultaneous deficiency in two proapoptotic Bcl-2 family members. Unlike other central apoptotic proteins, Bax and Bak appear to play overlapping roles in regulating the elimination of excess cells during both embryonic development and tissue homeostasis.
Experimental Procedures
Generation of Knockout Mice
The genomic organization for the bak mouse gene has been described (Ulrich et al., 1997). A targeting construct was made that replaced exons III–VI with the 1.2 kb Xho I–Bam HI fragment of pMCIneo Poly(A) (Stratagene, La Jolla, CA). This deletion removed 235 aa from the 254 aa coding region of Bak, including the entire BH1, BH2, and BH3 domains. The 59 flank of the targeting construct contained the 4.3 kb Bgl II–SalI fragment, and the 39 flank contained the 1.2 kb BglII–XhoI fragment, as depicted in Figure 1A. The bak targeting construct was electroporated into R1 ES cells (Nagy et al., 1993). Electroporation, selection, and maintenance of ES cells have been previously described (Shiels et al., 2000). The resulting expanded ES cell clones were screened for homologous recombination by Southern blotting. Four independent ES cell lines were identified and injected as described (Shiels et al., 2000). The generation of Bax mutant mice has been previously described (Knudson et al., 1995). bax–/–bak–/– mice were generated from matings of bax+/–bak+/– parents. These parental mice were generated from outcrosses of B6.129X1 (N9) bax+/– mice mated with outcrosses of (129S3 × 129X1) × C57BL/6 bak+/– mice. Offspring were genotyped and bax+/–bak+/– mice set up in breeding pairs. In more recent breedings, set up to maximize for the yield of double mutant mice, breedings were also set up with mice that were bax+/–bak–/–. Mice were genotyped by PCR of DNA samples from tail snips. The wild-type bak allele was identified using sense primer 5′-GGTGTCCACACTAGAGAACTACTC and anti-sense primer 5′-GAGCCATGAAGATGTTTAGC. The mutant bak allele was identified using a neomycin gene sense primer (5′-GCCCGGTTCTTTTTGTCAAGACCGA). The anti-sense primer for the mutant bak allele was the same as that one used for the wild-type allele. The wild-type bax allele was identified using sense primer 5′-TGATCAGAACCATCATG and anti-sense primer 5′-GTTGACCAGAGTGGCGTAGG. To detect the bax null allele, a neomycin gene-specific primer (5′-CCGCTTCCATTGCTCAGCGG) was used in combination with the wild-type bax anti-sense primer described above.
Western Blot Analysis
Single cell suspensions from thymus and spleen of mice were prepared and lysed in RIPA buffer (10 mM Tris [pH 8], 140 mM NaCl, 1% deoxycholate, 0.1% SDS, 1% Triton X-100, 0.025% NaN3 with the addition of a protease inhibitor cocktail [Roche, Indianapolis, IN]). The protein concentration of each lysate was determined by a colorimetric assay (BCA Protein Assay, Pierce, Rockford, IL), and 20 mg of total protein was resolved on a 12% precast SDS–polyacrylamide gel (Novex, San Diego, CA). After transfer to nitrocellu-lose using standard techniques, the Western blot was probed with a polyclonal anti-Bak antibody (Upstate Biotechnology, Lake Placid, NY).
Gross and Histopathological Analysis
After euthanasia, heart blood was collected and analyzed in a Hemavet 1500 apparatus (CDC Technologies Inc., Oxford, CT). A variety of tissues were harvested and fixed in 10% phosphate-buffered formalin (ACCRA Lab, Swedesboro, NJ). Brains were freed from the skull bone before fixation. Tissues were embedded, sectioned, mounted, and stained with hematoxylin-eosin using standard techniques. Brains were sectioned coronally and stained with cresyl violet. Photographs of slides were taken using an ES 800 Nikon microscope and a MicroMAX-5MHz-1300Y digital camera (Princeton Instruments, Inc. Trenton NJ) and analyzed with the MetaMorph Imaging System (Universal Imaging Corporation, West Chester, PA). Single cell suspensions were also made from lymphoid tissues for further analysis. For immunohistochemistry, 10 μm thick sections were deparaffinized, and immunohistochemistry was performed using standard protocols (Kendler and Golden, 1996). The primary antibody was anti-CD45RB (PharMingen, San Diego, CA), which recognizes B cells, most subsets of T cells, macrophages, and dendritic cells, used at 1 μg/ml.
Flow Cytometry
Flow cytometry was done on a FACS Calibur, and data was analyzed using Cell Quest software (Becton Dickinson; Mountain View, CA). Antibodies used include: CD4-CyChrome (clone H129.19; Phar-Mingen, San Diego, CA); CD8-APC (clone 53–6.7; PharMingen); CD44-CyChrome (clone IM7; PharMingen); CD62L-APC (clone MEL-14; PharMingen); B220-APC (clone RA3–6B2; PharMingen); Thy1.2-PE (clone OX-7; PharMingen); and IgD-FITC (clone 11–26c.2a; Phar-Mingen). Cell viabilities were determined by propidium iodide exclusion (PI; Molecular Probes, Eugene, OR).
Lymphocyte Death Assays
Splenocytes were isolated from mice with different genotypes. 1 × 105 cells were plated in each well of 96-well microtiter plates. Cells were cultured for 0, 1, 2, 3, or 4 days in RPMI 1640 medium supplemented with 10% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin, 4 mM L-glutamine, 5.5 × 10–5 M β-mercaptoethanol, and 10 mM Hepes solution. Thymocyte death assay was performed as described (Kuida et al., 1998). Briefly, thymocytes were isolated from mice with different genotypes. One million cells were treated with anti-Fas antibody (Jo2, PharMingen) plus 30 μg/ml cycloheximide (Sigma), γ radiation (500 rad, Gammacell), indicated doses of etopo-side (Clontech, Palo Alto, CA) or complete medium only. Cell viabilities were determined by PI exclusion.
Bone Marrow Hematopoietic Progenitor Assays
Bone marrow was harvested as described (Adelman et al., 1999). Briefly, the femur and tibia were dissected free from the surrounding muscle and tendons. Marrow was expunged with PBS (without Ca2+ or Mg2+), supplemented with 2% FBS and 1% pen/strep. Red cells were lysed and the remaining cells were counted using a Coulter Z2 Counter. 2 × 104 cells were plated in “complete” methylcellulose containing 15% FBS, 50 ng/ml rmSCF, 10 ng/ml rmIL-3, 10 ng/ml rhIL-6, 3 U/ml rhEPO, 10 μg/ml bovine insulin, 200 μg/ml human transferrin, 1% BSA, and 10–4 M β-ME (Stem Cell Technologies). Cultures were set up in triplicate and cultured at 37°C in air plus 5% CO2 for 5 days. Colony forming units (CFU) were scored based on colony morphology and cytological staining. CFU-GEMM, CFU-M, CFU-GM, and CFU-G were scored as myeloid colonies; CFU-E were scored as erythroid colonies; and CFU-Meg were scored as megakaryocyte colonies.
Acknowledgments
We thank the other members of the laboratories for helpful discussions and critique of the manuscript. We thank Barbi Judd for expert technical assistance and Susan Kerns for expert editorial assistance. This work was supported by grants from the National Institutes of Health, the Howard Hughes Medical Institute, and The Abramson Family Cancer Research Institute.
References
- Adams JM, Cory S. The Bcl-2 protein family: arbiters of cell survival. Science. 1998;281:1322–1326. doi: 10.1126/science.281.5381.1322. [DOI] [PubMed] [Google Scholar]
- Adelman DM, Maltepe E, Simon MC. Multilineage embryonic hematopoiesis requires hypoxic ARNT activity. Genes Dev. 1999;13:2478–2483. doi: 10.1101/gad.13.19.2478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alagramam KN, Kwon HY, Cacheiro NL, Stubbs L, Wright CG, Erway LC, Woychik RP. A new mouse insertional mutation that causes sensorineural deafness and vestibular defects. Genetics. 1999;152:1691–1699. doi: 10.1093/genetics/152.4.1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer SA, Altman J. Neocortical development. Raven Press; New York: 1991. [Google Scholar]
- Cecconi F, Alvarez-Bolado G, Meyer BI, Roth KA, Gruss P. Apaf1 (CED-4 homolog) regulates programmed cell death in mammalian development. Cell. 1998;94:727–737. doi: 10.1016/s0092-8674(00)81732-8. [DOI] [PubMed] [Google Scholar]
- Chittenden T, Harrington EA, O'Connor R, Flemington C, Lutz RJ, Evan GI, Guild BC. Induction of apoptosis by the Bcl-2 homologue Bak. Nature. 1995;374:733–736. doi: 10.1038/374733a0. [DOI] [PubMed] [Google Scholar]
- Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML, Wyllie AH. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature. 1993;362:849–852. doi: 10.1038/362849a0. [DOI] [PubMed] [Google Scholar]
- Clarke DL, Johansson CB, Wilbertz J, Veress B, Nilsson E, Karlstrom H, Lendahl U, Frisen J. Generalized potential of adult neural stem cells. Science. 2000;288:1660–1663. doi: 10.1126/science.288.5471.1660. [DOI] [PubMed] [Google Scholar]
- Deckwerth TL, Elliott JL, Knudson CM, Johnson EM, Snider WD, Korsmeyer SJ. BAX is required for neuronal death after trophic factor deprivation and during development. Neuron. 1996;17:401–411. doi: 10.1016/s0896-6273(00)80173-7. [DOI] [PubMed] [Google Scholar]
- Du C, Fang M, Li Y, Li L, Wang X. Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell. 2000;102:33–42. doi: 10.1016/s0092-8674(00)00008-8. [DOI] [PubMed] [Google Scholar]
- Eskes R, Desagher S, Antonsson B, Martinou JC. Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol. Cell. Biol. 2000;20:929–935. doi: 10.1128/mcb.20.3.929-935.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farrow SN, White JH, Martinou I, Raven T, Pun KT, Grinham CJ, Martinou JC, Brown R. Cloning of a bcl-2 homologue by interaction with adenovirus E1B 19K. Nature. 1995;374:731–733. doi: 10.1038/374731a0. Erratum: Nature 375(6530), 1995. [DOI] [PubMed] [Google Scholar]
- Green DR. Apoptotic pathways: paper wraps stone blunts scissors. Cell. 2000;102:1–4. doi: 10.1016/s0092-8674(00)00003-9. [DOI] [PubMed] [Google Scholar]
- Honarpour N, Du C, Richardson JA, Hammer RE, Wang X, Herz J. Adult Apaf-1-deficient mice exhibit male infertility. Dev. Biol. 2000;218:248–258. doi: 10.1006/dbio.1999.9585. [DOI] [PubMed] [Google Scholar]
- Ishii N, Wanaka A, Ohno K, Matsumoto K, Eguchi Y, Mori T, Tsujimoto Y, Tohyama M. Localization of bcl-2, bax, and bcl-x mRNAs in the developing inner ear of the mouse. Brain Res. 1996;726:123–128. [PubMed] [Google Scholar]
- Jurgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC. Bax directly induces release of cytochrome c from isolated mitochondria. Proc. Natl. Acad. Sci. USA. 1998;95:4997–5002. doi: 10.1073/pnas.95.9.4997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kendler A, Golden JA. Progenitor cell proliferation outside the ventricular and subventricular zones during human brain development. J. Neuropathol. Exp. Neurol. 1996;55:1253–1258. doi: 10.1097/00005072-199612000-00009. [DOI] [PubMed] [Google Scholar]
- Kiefer MC, Brauer MJ, Powers VC, Wu JJ, Umansky SR, Tomei LD, Barr PJ. Modulation of apoptosis by the widely distributed Bcl-2 homologue Bak. Nature. 1995;374:736–739. doi: 10.1038/374736a0. [DOI] [PubMed] [Google Scholar]
- Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science. 1997;275:1132–1136. doi: 10.1126/science.275.5303.1132. [DOI] [PubMed] [Google Scholar]
- Kluck RM, Esposti MD, Perkins G, Renken C, Kuwana T, Bossy-Wetzel E, Goldberg M, Allen T, Barber MJ, Green DR, Newmeyer DD. The pro-apoptotic proteins, Bid and Bax, cause a limited permeabilization of the mitochondrial outer membrane that is enhanced by cytosol. J. Cell Biol. 1999;147:809–822. doi: 10.1083/jcb.147.4.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knudson CM, Tung KS, Tourtellotte WG, Brown GA, Korsmeyer SJ. Bax-deficient mice with lymphoid hyperplasia and male germ cell death. Science. 1995;270:96–99. doi: 10.1126/science.270.5233.96. [DOI] [PubMed] [Google Scholar]
- Krajewski S, Krajewska M, Reed JC. Immunohistochemical analysis of in vivo patterns of Bak expression, a proapoptotic member of the Bcl-2 protein family. Cancer Res. 1996;56:2849–2855. [PubMed] [Google Scholar]
- Kuida K, Haydar TF, Kuan CY, Gu Y, Taya C, Karasuyama H, Su MS, Rakic P, Flavell RA. Reduced apoptosis and cytochrome c–mediated caspase activation in mice lacking caspase 9. Cell. 1998;94:325–337. doi: 10.1016/s0092-8674(00)81476-2. [DOI] [PubMed] [Google Scholar]
- Li H, Zhu H, Xu CJ, Yuan J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell. 1998;94:491–501. doi: 10.1016/s0092-8674(00)81590-1. [DOI] [PubMed] [Google Scholar]
- Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature. 1993;362:847–849. doi: 10.1038/362847a0. [DOI] [PubMed] [Google Scholar]
- Luo X, Budihardjo I, Zou H, Slaughter C, Wang X. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell. 1998;94:481–490. doi: 10.1016/s0092-8674(00)81589-5. [DOI] [PubMed] [Google Scholar]
- Motoyama N, Wang F, Roth KA, Sawa H, Nakayama K, Negishi I, Senju S, Zhang Q, Fujii S, et al. Massive cell death of immature hematopoietic cells and neurons in Bcl-x- deficient mice. Science. 1995;267:1506–1510. doi: 10.1126/science.7878471. [DOI] [PubMed] [Google Scholar]
- Nagata S. Apoptosis by death factor. Cell. 1997;88:355–365. doi: 10.1016/s0092-8674(00)81874-7. [DOI] [PubMed] [Google Scholar]
- Nagy A, Rossant J, Nagy R, Abramow-Newerly W, Roder JC. Derivation of completely cell culture–derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA. 1993;90:8424–8428. doi: 10.1073/pnas.90.18.8424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama K, Negishi I, Kuida K, Shinkai Y, Louie MC, Fields LE, Lucas PJ, Stewart V, Alt FW, et al. Disappearance of the lymphoid system in Bcl-2 homozygous mutant chimeric mice. Science. 1993;261:1584–1588. doi: 10.1126/science.8372353. [DOI] [PubMed] [Google Scholar]
- Oltvai ZN, Milliman CL, Korsmeyer SJ. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell. 1993;74:609–619. doi: 10.1016/0092-8674(93)90509-o. [DOI] [PubMed] [Google Scholar]
- Orth K, Dixit VM. Bik and Bak induce apoptosis downstream of CrmA but upstream of inhibitor of apoptosis. J. Biol. Chem. 1997;272:8841–8844. doi: 10.1074/jbc.272.14.8841. [DOI] [PubMed] [Google Scholar]
- Rodriguez I, Araki K, Khatib K, Martinou JC, Vassalli P. Mouse vaginal opening is an apoptosis-dependent process which can be prevented by the overexpression of Bcl2. Dev. Biol. 1997;184:115–121. doi: 10.1006/dbio.1997.8522. [DOI] [PubMed] [Google Scholar]
- Shiels H, Li X, Schumacker PT, Maltepe E, Padrid PA, Sperling A, Thompson CB, Lindsten T. TRAF4 deficiency leads to tracheal malformation with resulting alterations in airflow to the lungs. Am. J. Pathol. 2000;157:679–688. doi: 10.1016/S0002-9440(10)64578-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strasser A, Harris AW, Huang DCS, Krammer PH, Cory S. Bcl-2 and Fas/APO-1 regulate distinct pathways to lymphocyte apoptosis. EMBO J. 1995;14:6136–6147. doi: 10.1002/j.1460-2075.1995.tb00304.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler M, et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature. 1999;397:441–446. doi: 10.1038/17135. [DOI] [PubMed] [Google Scholar]
- Tao W, Kurschner C, Morgan JI. Modulation of cell death in yeast by the Bcl-2 family of proteins. J. Biol. Chem. 1997;272:15547–15552. doi: 10.1074/jbc.272.24.15547. [DOI] [PubMed] [Google Scholar]
- Ulrich E, Kauffmann-Zeh A, Hueber AO, Williamson J, Chittenden T, Ma A, Evan G. Gene structure, cDNA sequence, and expression of murine Bak, a proapoptotic Bcl-2 family member. Genomics. 1997;44:195–200. doi: 10.1006/geno.1997.4858. [DOI] [PubMed] [Google Scholar]
- Vander Heiden MG, Chandel NS, Williamson EK, Schumacker PT, Thompson CB. Bcl-xL regulates the membrane potential and volume homeostasis of mitochondria. Cell. 1997;91:627–637. doi: 10.1016/s0092-8674(00)80450-x. [DOI] [PubMed] [Google Scholar]
- Vaux DL, Korsmeyer SJ. Cell death in development. Cell. 1999;96:245–254. doi: 10.1016/s0092-8674(00)80564-4. [DOI] [PubMed] [Google Scholar]
- Veis DJ, Sorenson CM, Shutter JR, Korsmeyer SJ. Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell. 1993;75:229–240. doi: 10.1016/0092-8674(93)80065-m. [DOI] [PubMed] [Google Scholar]
- Verhagen AM, Ekert PG, Pakusch M, Silke J, Connolly LM, Reid GE, Moritz RL, Simpson RJ, Vaux DL. Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins. Cell. 2000;102:43–53. doi: 10.1016/s0092-8674(00)00009-x. [DOI] [PubMed] [Google Scholar]
- Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M, Thompson CB, Korsmeyer SJ. tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev. 2000;14:2060–2071. [PMC free article] [PubMed] [Google Scholar]
- Wyllie AH. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature. 1980;284:555–556. doi: 10.1038/284555a0. [DOI] [PubMed] [Google Scholar]
- Yang J, Liu X, Bhalla K, Kim CN, Ibrado AM, Cai J, Peng TI, Jones DP, Wang X. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science. 1997;275:1129–1132. doi: 10.1126/science.275.5303.1129. [DOI] [PubMed] [Google Scholar]
- Yin XM, Wang K, Gross A, Zhao Y, Zinkel S, Klocke B, Roth KA, Korsmeyer SJ. Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature. 1999;400:886–891. doi: 10.1038/23730. [DOI] [PubMed] [Google Scholar]
- Yoshida H, Kong YY, Yoshida R, Elia AJ, Hakem A, Hakem R, Penninger JM, Mak TW. Apaf1 is required for mitochondrial pathways of apoptosis and brain development. Cell. 1998;94:739–750. doi: 10.1016/s0092-8674(00)81733-x. [DOI] [PubMed] [Google Scholar]