Abstract
Many receptor systems initiate cell signaling through ligand-induced receptor aggregation. For bivalent ligands binding to mono- or bivalent receptors, a plot of the equilibrium concentration of receptors in aggregates versus the log of the free ligand concentration, the cross-linking curve, is symmetric and bell-shaped. However, steady state cellular responses initiated through receptor cross-linking may have a different dependence on ligand concentration than the aggregated receptors that initiate and maintain these responses. We illustrate by considering the activation of the protein kinase Syk that rapidly occurs after high affinity receptors for IgE, FcεRI, are aggregated on the surface of mast cells and basophils. Using a mathematical model of Syk activation we investigate two effects, one straightforward and one less so, that result in Syk activation not qualitatively following the cross-linking curve. Model predictions show that if the mechanism by which Syk is fully activated involves the transphosphorylation of Syk by Syk, then Syk activation curves can be either bell-shaped or double humped, depending on the cellular concentrations of Syk and FcεRI. The model also predicts that the Syk activation curve can be nonsymmetric with respect to the ligand concentration. The cell can exhibit differential Syk activation at two different ligand concentrations that produce identical distributions of receptor aggregates that form and dissociate at the same rates. We discuss how, even though it is only receptor aggregates that trigger responses, differences in total ligand concentration can lead to subtle kinetic effects that yield qualitative differences in the levels of Syk activation.
Keywords: BIOCHEMICAL REACTION NETWORK, BIOCHEMICAL SYSTEMS, CELL SIGNALING PATHWAY
1 Introduction
Mast cells and basophils play major roles in allergic responses of the immediate type [1]. Allergic individuals produce IgE that is specific for the multivalent foreign molecules (allergens) that trigger their allergic responses. IgE binds with high affinity through its constant region to a monovalent receptor, FcεRI, that is expressed on the surface of basophils and mast cells and that mediates much of mast cell activation in allergic reactions. Upon exposure to the allergen, IgE is induced to aggregate on the surfaces of these cells. The bridging of multiple IgEs by the allergen excites a signaling network that results in the release of preformed granules and newly synthesized mediators of anaphylaxis [2].
FcεRI is a tetrameric complex composed of an α-chain that binds IgE to form a long lived complex, a β-chain and two disulfide-linked γ-chains [3]. The events immediately following aggregation of FcεRI result in the recruitment of the protein tyrosine kinase (PTK) Syk from the cytosol to the cell membrane through attachment to the FcεRI γ-chain and its subsequent activation. Details are reviewed in [4, 5]. Briefly, the β and γ chains each contain single immunoreceptor tyrosine-based activation motifs (ITAMs) that become phosphorylated upon aggregation by Lyn, a Src family PTK. Lyn is anchored to the inner layer of the plasma membrane and constitutively associates with the unphosphorylated β-chain. The phosphorylated β and γ ITAMs are docking sites for Lyn and Syk respectively. Syk becomes partially activated when it binds through its two Src homology two (SH2) domains to the doubly phosphorylated γ ITAM [6]. Full activation of Syk is achieved when two adjacent tyrosines in its activation loop are phosphorylated [7]. Activated Syk in mast cells phosphorylates the adaptor proteins LAT (Linker for Activation of T cells) and NTAL (Non-T-cell Activation Linker) that function as scaffolds, organizing other signaling proteins that are responsible for signaling events further downstream [8].
A recent survey by McGlashan [9] reports a broad distribution of Syk expression levels in human basophils, ranging from 5000 to 60,000 molecules per cell. MacGlashan and co-workers have observed that a low expression level of Syk attenuates the calcium response [10], an event which is more proximal to Syk activation in the signaling cascade than histamine release. For human basophils, maximal histamine release shows a strong correlation with Syk activation [9]. Although many of the biochemical reactions leading from Syk activation to histamine release have been elucidated, a complete model still eludes us. A mathematical model of Syk activation consisting of 354 chemical species and the reactions among them, has been developed by Faeder et al. [4]. Here we use the model to investigate the effects of the FcεRI and Syk expression levels on the extent of Syk activation. In particular, we use it to predict the extent of Syk activation as a function of the concentration of the allergen, for given FcεRI and Syk concentrations, and for a given set of kinetic parameters characterizing binding, unbinding, phosphorylation and dephosphorylation events.
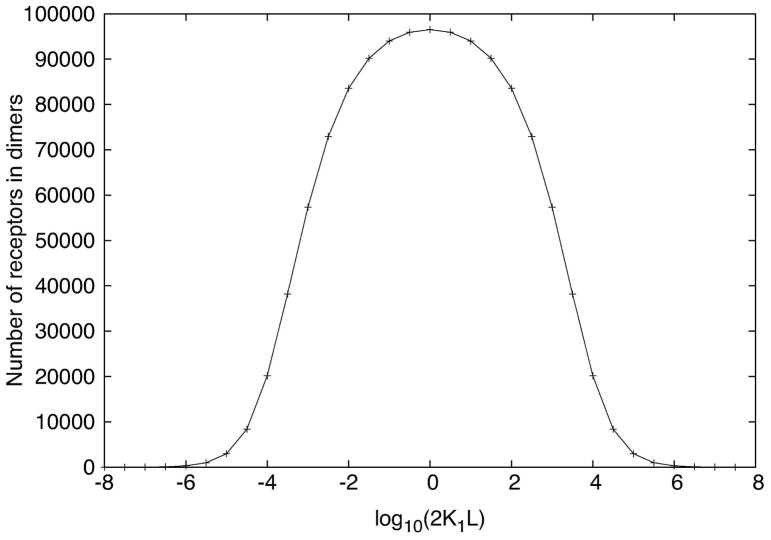
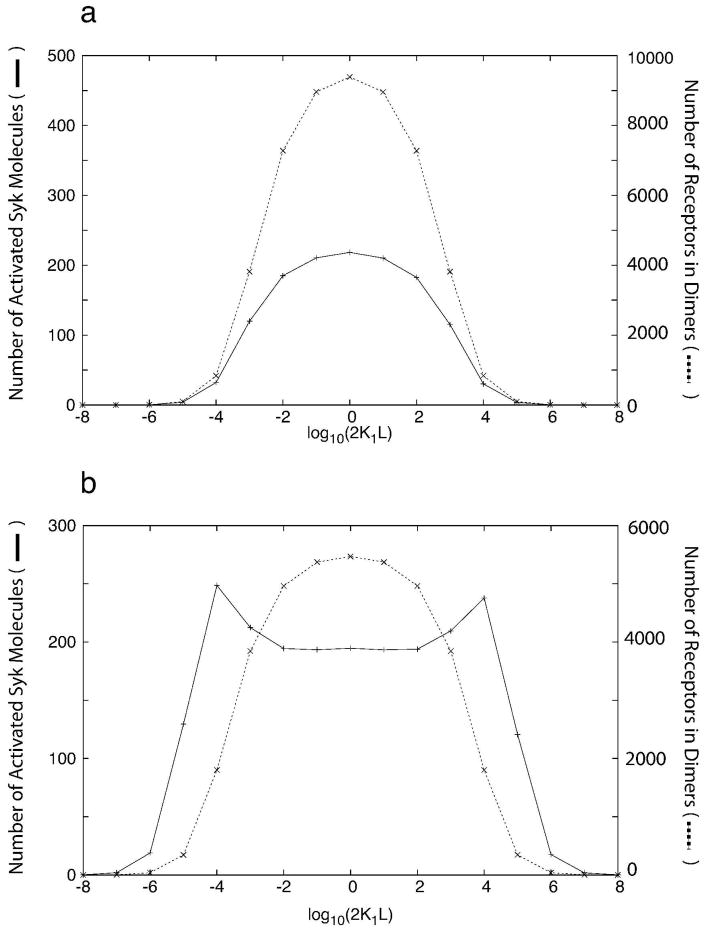
Naively, one might expect that the variation of Syk activation with the equilibrium allergen concentration should be qualitatively similar to the variation in the number of cross-linked FcεRI receptors. For bivalent allergens interacting with IgE, the receptor cross-linking curve is bell-shaped [11, 12] and symmetric as shown in Fig. 1. We define the Syk activation curve as a plot of the number of fully activated Syk molecules in a cell versus the log of the equilibrium free allergen concentration. For many physiological Syk expression levels the predicted Syk activation curve follows the receptor cross-linking curve. However, as we will see, Faeder et al.’s model predicts that for some physiological Syk expression levels, the Syk activation curve is bimodal. In the model, full activation of a Syk molecule requires its transphosphorylation by a second Syk molecule, the two Syk molecules being bound to different receptors in a receptor dimer [7]. Hence, the model predicts that for limiting Syk concentration, an excess of cross-linked receptors over Syk molecules over a range of free equilibrium allergen concentrations can occur and result in high-dose inhibition of Syk activation [13]. At other allergen concentrations, the extent of cross-linking is lower so that there is enough Syk for two Syk to be in the same receptor dimer. This predicted differential behavior at different allergen concentrations gives rise to the variable shape (unimodal/multimodal) of the Syk activation curve [13].
Figure 1.
Receptor cross-linking (dimerization) curve fork+1 = 2.5 × 107 M−1 s−1; k+2 = 8 × 10−9cm2s−1; k−1 = 0.316(10−0.5) s−1, k−2 = 0.0316(10−1.5) s−1. The receptor aggregation is maximum at L = 1/2K1, i.e. at log10(2K1L) = 0, where K1 = k+1/k−1 is the equilibrium constant for the extracellular bivalent ligand, present at concentration L, for binding to the Fab part of the IgE.
In light of the transphosphorylation mechanism of Syk activation in the model, it is not surprising that for some values of the cellular Syk and receptor concentrations, a bell-shaped receptor cross-linking curve can give rise to a bimodal Syk activation curve. What is surprising is that the model of Faeder et al. [4] predicts that a symmetric receptor cross-linking curve can give rise to an asymmetric Syk activation curve. Consider two values of the bivalent allergen concentration in Fig. 1 that yield equal concentrations of receptor dimers. At these two concentrations, the dimers form and break up at the same rates at equilibrium. Nonetheless, we will see that in the steady state the corresponding Syk activation curve (Fig. 6a) is predicted to be asymmetric. This asymmetry implies that in addition to the receptor dimer concentration, the total external ligand concentration influences the level of Syk activation. How this asymmetry arises in the steady state is a major focus of the paper.
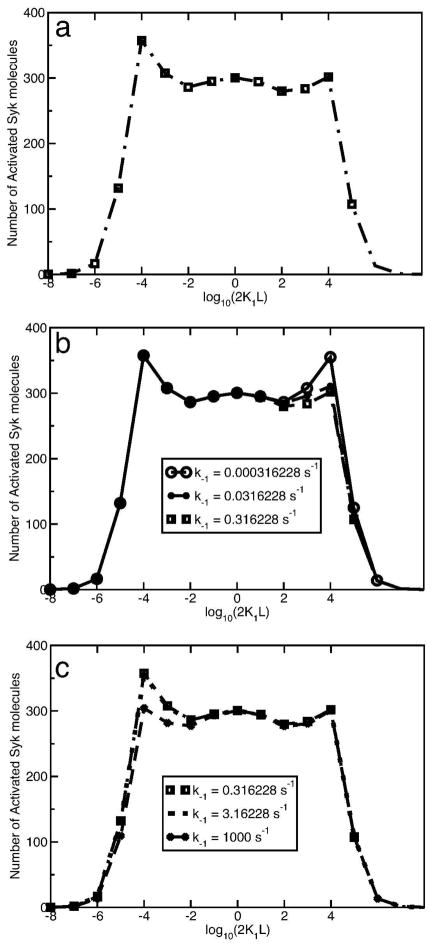
Figure 6.
(a) Syk Activation curve for k+1 = 2.5 × 107 M−1 s−1; k+2 = 8 × 10−9cm2s−1; k−1 = 0.316(10−0.5) s−1; k−2 = 0.0316(10−1.5)s−1; (b)–(c) Syk Activation curves for k+2 = 8 × 10−9cm2s−1; k−2 = 0.0316(10−1.5)s−1; (b)The k+1 and k−1 values are varied such that K1 = k+1/k−1 is fixed at 7.9 × 107 M−1. The legends show the k−1 values used and the k+1 values are accordingly adjusted so that K1 remains fixed. For parts (a), (b) and (c), [Syk]tot = 7 × 103 per cell; [Lyn]tot = 105 per cell; [FcεRI]tot = 105 per cell.
2 Mathematical model
2.1 Components
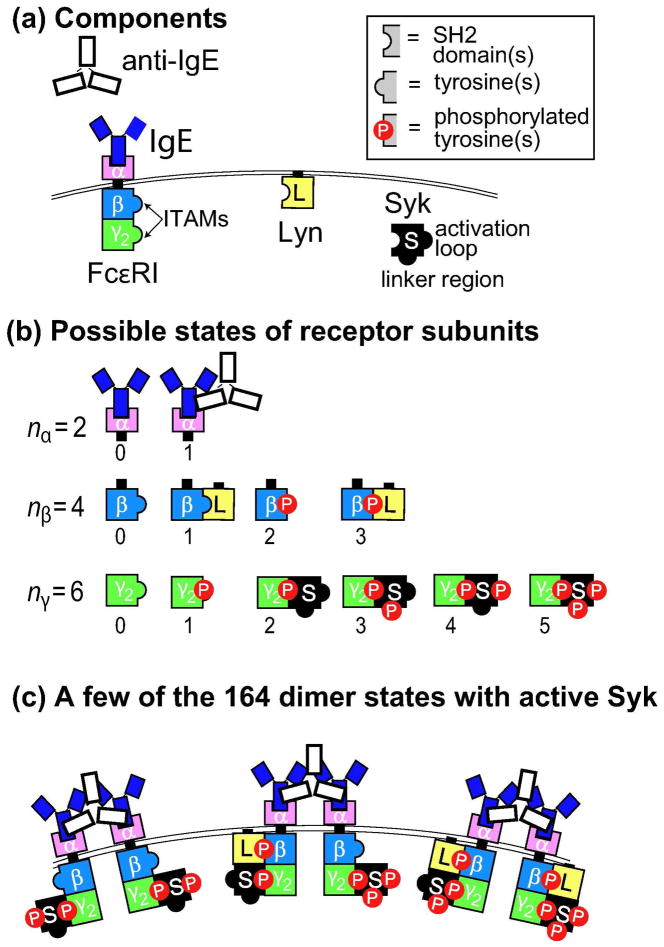
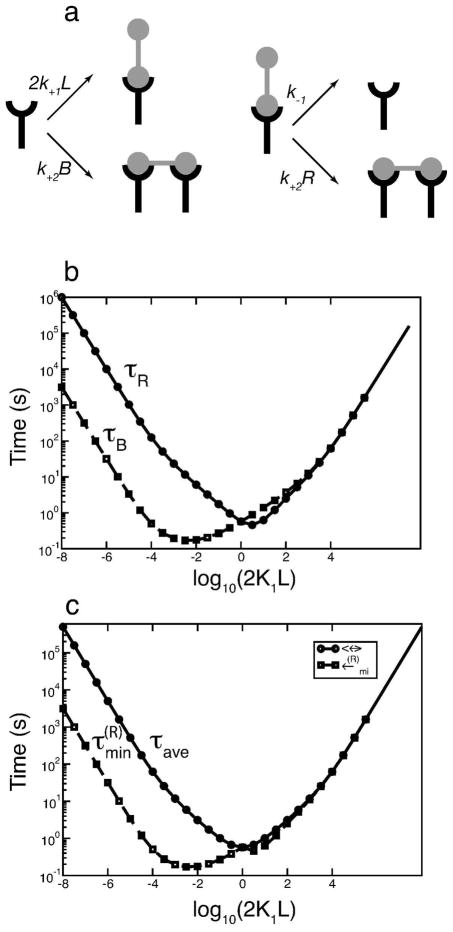
We briefly review the mathematical model of Faeder et al. [4] for the early signaling events initiated by ligand-induced aggregation of FcεRI-IgE complexes on mast cells and basophils. The model is summarized in Fig. 2. For the simulations that follow, we take as the external stimulus a symmetric bivalent ligand, such as a monoclonal anti-IgE, that cross-links two IgE molecules, each bound to a FcεRI receptor. We assume that aggregates of receptors larger than dimers cannot form. The tetrameric FcεRI is modeled as three subunits, with the disulphide-bonded pair of γ chains treated as one unit. The extracellular region of the α chain binds to the Fc portion of IgE. The binding of IgE to the α subunit of the FcεRI receptor is not explicitly considered as its half-life for the dissociation of the IgE-FcεRI complex is much longer than the processes we consider [14]. We therefore ignore dissociation and treat the IgE-FcεRI complex as a single unit which we refer to as the receptor.
Figure 2.
Components and states of the model, modified from of Faeder et al. [4]. The bivalent ligand is a monoclonal anti-IgE that can dimerize receptors.
The β subunit and the γ dimer contain the ITAMs that, upon phosphorylation, become binding sites for the SH2 domains of the two kinases, Lyn and Syk. A simplifying assumption of the model [4] is that multiple tyrosine residues on receptor subunits and Syk are treated as single units of phosphorylation, as indicated in Fig. 2a. For example, the separate tyrosines in the β ITAM are lumped together, as are those in the γ ITAM, so that in the model an ITAM is either phosphorylated or unphosphorylated. Sites of Syk tyrosine phosphorylation are lumped into two units: the activation loop, which is phosphorylated by Syk, and the linker region, which is phosphorylated by Lyn. Fig. 2b shows the states of the β chain and the lumped γ chains included in the model. Each unit can be phosphorylated or unphosphorylated, and can be associated with a kinase in any of several states. In the model, each β chain and γ dimer can bind only a single kinase molecule at a time.
The model permits a large number of receptor states (Figs. 2b and 2c). Since the state of each subunit is independent of the states of the other subunits, the total number of monomer states is nαnβnγ = 48. When a monovalent ligand is present as well as a bivalent ligand, the number of monomeric states increases to 72. In a dimer, each subunit must be engaged with the ligand, so the total number of dimer states is nβnγ (nβnγ + 1)/2 = 300. In addition, there are six nonreceptor states, free ligand, free Lyn, and Syk in each of its four possible states of phosphorylation, to give a total of 354 distinct chemical species in the model. The model of Faeder et al. [4] is composed of 354 chemical species and the chemical reactions that connect them. (For details of the chemical reactions see Fig. 2 in Faeder et al. [4].)
2.2 Network structure and parameters
The model requires as input the rate constants associated with each of the reactions, as well as the initial concentrations of each of the components, which are specific to the cell type being modeled. In the current investigation we model the Syk dose-response curves in human basophils. A typical human basophil expresses about 100,000 FcεRI, although there is much variation in the number of expressed FcεRI between basophils from allergic and non-allergic individuals. There is also considerable variation in Syk expression levels, that can vary between 5,000 and 60,000 per cell. Experimental observations [9] suggest that the median Lyn expression level in human basophils is about 100,000 molecules per cell.
We consider a ligand that differs from that used by Faeder et al. [4] in that it binds reversibly to the receptor. Other than a different bivalent ligand, the current reaction network is the same as in Ref. [4]. Lyn can associate with a receptor in a dimer in two possible ways, weakly with the unphosphorylated β chain of the receptor and strongly to the phosphorylated β ITAM. Syk associates with FcεRI through an interaction between its tandem SH2 domains and the doubly phosphorylated γ ITAM. Lyn, associated with a receptor aggregate, can transphosphorylate the β and γ ITAMs on an adjacent receptor. In the model, all Lyn molecules available to the receptor are in an active form. The available Lyn is considerably less than the total cellular Lyn. Syk is phosphorylated in the model by either Lyn or Syk through transphosphorylation. Lyn phosphorylates Syk tyrosines located in the linker region, while Syk phosphorylates the activation loop tyrosines. Although Lyn may be responsible in vivo for a small portion of the Syk activation loop phosphorylation, full Syk activation loop phosphorylation requires kinase-active Syk [15]. The parameters values we use in our model to characterize the kinetics of these reactions were either determined directly, determined from data fitting, or estimated from published results, as discussed in Faeder et al. [4]
In the model, phosphorylated units that are not protected through association with an SH2 domain can be dephosphorylated with a common rate constant, d, termed the intrinsic rate constant for dephosphorylation. Dephosphorylation is blocked when an SH2 domain is associated with the phosphorylated site. At long times, the model system goes to a steady state, not an equilibrium. In the model, tyrosines are constantly being phosphorylated and dephosphorylated. As a result, maintaining a constant level of phosphorylation requires a constant input of energy. Since the modifications of the intracellular domains of the receptors do not influence the extracellular binding, the distribution of ligand-receptor aggregates goes to the same equilibrium as would be obtained if no chemical modifications occurred.
2.3 Network generation and simulation
The binding and reaction rules and their associated rate constants were specified in the syntax of the second-generation version of BioNetGen [16, 17], BioNetGen2, which uses graph theoretic methods to automatically generate the associated network of kinetic balances (ordinary differential equations). The open-source software (available through http://bionetgen.org) uses standard numerical algorithms to solve the generated system of differential equations and obtain the time courses of all the species until the system attains a steady state. In our simulations, all the kinetic parameters except the ones related to ligand binding and unbinding are taken from Ref. 4, where a complete discussion of how the parameters were obtained is given.
3 Results
We now examine the Syk activation curve for different sets of receptor and Syk expression levels, keeping the available Lyn fixed at 105 per cell. In our simulations, the cross-linking of the receptors is caused by a bivalent ligand that recognizes only one binding site on each receptor so that the only receptor aggregates that can form are dimers. At equilibrium the cross-linking curve is always symmetric and bell shaped [11].
3.1 Relative expression of Syk and FcεRI shape the activation curve for Syk
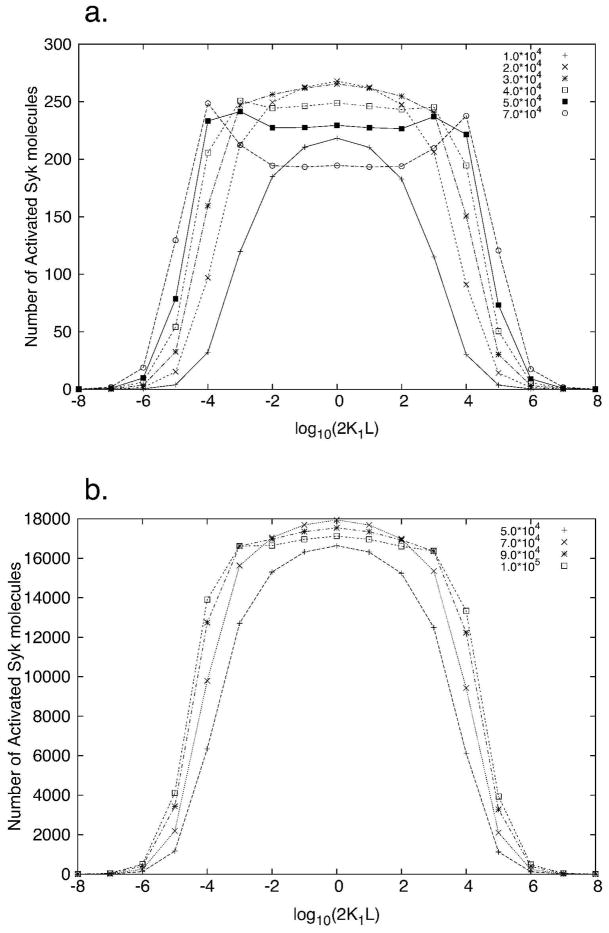
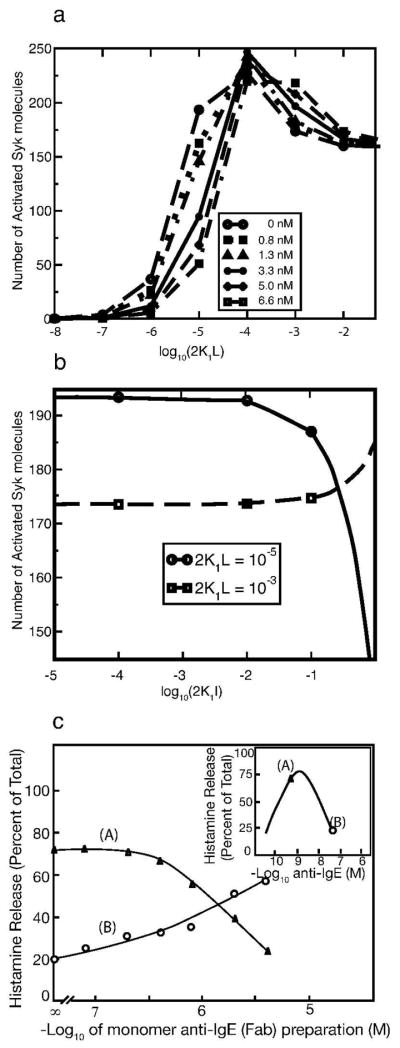
Simulations show that the shape of the Syk activation curve depends on the concentrations of Syk and FcεRI receptors. The number of FcεRI per human basophils varies from 103 to 106 with a typical value of about 100,000 FcεRI [9]. The corresponding Syk concentration ranges from 5 × 103 to 6 × 104 per cell [9]. At the lower end of the range (5 × 103 Syk per cell), a receptor population of 7 × 104 per cell results in a predicted bimodal Syk activation curve (Fig. 3a). At the higher end (6 × 104 Syk per cell), the same level of receptors results in a bell-shaped Syk activation curve (Fig. 3b).
Figure 3.
Syk Activation curves fork+1 = 2.5 × 106 M−1s−1, k+2 = 8 × 10−9 cm2s−1, k−1 = k−2 = 0.01 s−1, and [Lyn]tot = 105 per cell. Each curve corresponds to a different number of receptors per cell given in the legend. The number of activated Syk molecules on the y axis are given in numbers per cell. (a) [Syk]tot = 5 × 103 per cell; (b) [Syk]tot = 6 × 104 per cell. The number of activated Syk molecules on the y axis are the numbers per cell. In (a) the ratio of total receptors to total Syk varies from 2–14 while in (b) the same ratio varies from 0.83–1.67.
In the model, full activation of a Syk molecule requires its transphosphorylation by a second Syk, which occurs when two Syk molecules are bound to different receptors cross-linked by a bivalent ligand. We refer to a receptor dimer with two bound Syk molecules, one on each receptor, as a Syk dimer. Formation of Syk dimers is essential for Syk activation. Double-humped Syk activation curves occur (Fig. 3a) when the concentration of receptor dimers exceeds the threshold at which the number of Syk dimers becomes a decreasing function of receptor dimer concentration.
Studies of histamine release dose response curves (log-log plots of the fraction of a cell’s total histamine released in a given time versus the concentration of ligand the cell is exposed to) lead to the definition of two types of inhibition of histamine release. In Type I inhibition, commonly known as antigen excess inhibition, histamine release is inhibited when receptor crosslinks are decreased. In the more interesting Type II inhibition, histamine release is inhibited when receptor crosslinks are increased. This type of inhibition was first observed by Becker et al. [18] who exposed human basophils to anti-IgE and found that inhibition of histamine release was correlated with increased aggregation of IgE. (The terms Type I and Type II inhibition were introduced by Dembo and Goldstein [19] who noted a correlation between the type of inhibition and the type of desensitization human basophils undergo after exposure to multivalent ligands.) Applying the same terminology for Syk activation, Type I inhibition occurs in our model for parameter regimes in which the number of Syk molecules is comparable to, or exceeds, the number of receptors in dimers, so that the dose-response curve (Syk activation curve) follows the receptor cross-linking curve at all the ligand concentrations (Fig. 4a). This type of inhibition occurs because at sufficiently high ligand concentrations, the binding sites on IgE are blocked by the ligand, reducing the number of receptor cross-links. Type II inhibition occurs in our model system when there is an excess of FcεRI-bound IgE dimers compared to the number of available Syk molecules, and is characterized by decreasing signaling response (Syk activation) at ligand concentrations where receptor cross-linking is increasing. The reduction in Syk activation with increasing ligand concentration near the center of the Syk activation curve in Fig 4b is a Type II inhibition. Although we used a bivalent ligand in our simulations to illustrate the two types of inhibition, the valence of the ligand should not be critical as long as it is multivalent.
Figure 4.
Comparison of receptor cross-linking and Syk activation curves to determine the type of antigen excess inhibition. k+1 = 2.5 × 106M−1s−1, k+2 = 8 × 10−9 cm2s−1, k−1 = k−2 = 0.01 s−1; [Syk]tot = 5 × 103 per cell; [Lyn]tot = 105 per cell. (a) [FcεRI]tot = 1 × 104. The dashed line represents (Number of receptors in dimers)/20.0 and the solid line represents the number of activated Syk molecules. (b) [FcεRI]tot = 7 × 104. The dashed line represents (Number of receptors in dimers)/250.0 and the solid line represents the number of activated Syk molecules.
3.2 Effect of monovalent ligands on Syk activation
A way to distinguish between Type I and Type II inhibition experimentally is by comparing responses to anti-IgE in the presence and absence of Fab fragments [20, 21]. Adding monovalent ligand that competes for the same binding site as a multivalent ligand will always reduce the number of cross-links. Thus, if the response increases in the presence of monovalent ligand, the inhibition is Type II, while if it decreases it is of Type I. To demonstrate, we have incorporated monovalent ligand into the model, with the monovalent ligand having the same rate constants for binding and dissociation as a single anti-IgE Fab site. The reference case we use for studying the effects of adding monovalent ligands on the signaling response, measured by Syk activation in our model, is characterized by the parameter values k−1 = k−2 = 0.01s−1, k+1 = 2.5 × 106 M−1 s−1, k+2 = 8 × 10−9 molecule−1 cm2 s−1 and total Syk, Lyn and FcεRI numbers per cell of 5 × 103, 1 × 105 and 1 × 105 respectively. We use this parameter set because it yields a bimodal Syk activation curve in the absence of the monovalent ligand. We add monovalent ligand to a maximum concentration of 6.6 nM which corresponds to 4 × 106 molecules per cell. We consider the effects of adding monovalent ligands on Syk activation at two bivalent ligand concentrations, 2 × 10−5 nM and 2 × 10−3 nM, given by 2K1L values of 10−5 and 10−3 respectively. Fig. 5 shows that adding monovalent IgE ligand in increasing concentrations leads to a gradual lowering in the Syk activation level at 2K1L = 10−5 (Type I inhibition). In contrast, adding monovalent ligand in increasing concentrations at 2K1L = 10−3 results in a gradual increase of the Syk activation level (Type II inhibition).
Figure 5.
(a)The effect of adding different concentrations of a monovalent IgE ligand on the extent of Syk activation at 2K1L = 10−5 (Syk activation increases with 2K1L) and at 2K1L = 10−3 (Syk activation decreases with 2K1L). The concentration of monovalent inhibitor (Fab fragments when the ligand is anti-IgE) present in the simulation is given in the figure. (b) The variation of the extent of Syk activation as a function of the concentration per cell of a monovalent inhibitor, I, at two ligand concentrations given by 2K1L = 10−5 and 2K1L = 10−3. The ligand-receptor binding parameters used are k+1 = 2.5 × 106 M−1s−1, k+2 = 8 × 10−9 cm2s−1, k−1 = k−2 = 0.01s−1. [Syk]tot = 5 × 103 per cell; [Lyn]tot = 105 per cell; [FcεRI]tot = 105 per cell. (c) Modified Figure 1 from Magro and Alexander [20]. Plot of histamine release due to 5 × 10−10 M anti-IgE (A) and 5 × 10−8 M (B) incubated with increasing concentrations of monomer anti-IgE (Fab). The insert shows the positioning on the dose response curve of the two anti-IgE concentrations.
On plotting the Syk activation level as a function of the added monovalent ligand concentration for two values 10−5 and 10−3 of 2K1L, we obtain Fig. 5b which is qualitatively similar to the plot of histamine release as a function of the amount of added monomer anti-IgE (Fab) in Fig. 1 of Ref [20] as shown in Fig. 5c. The similarity between the predicted variation of Syk activation and the experimentally observed variation of histamine release [20,22] with added monomer concentration suggests one possible mechanism for Type II inhibition.
3.3 A symmetric cross-linking curve can lead to an asymmetric Syk activation curve
For the ligand-receptor binding parameters in Table 1, and FcεRI and Syk concentrations of 1 × 105 and 7 × 103 per cell respectively, we observe a Syk activation curve with two prominent maxima at 2K1L = 10−4 and 2K1L = 104 (Fig. 6a). This parameter set is used because it accentuates the asymmetry between the two maxima. The extent of receptor cross-linking at these two ligand concentrations are equal. The mean lifetime of the receptor dimer is independent of the ligand concentration, which implies that kinetic proofreading effects at these two ligand concentrations should be identical. Still, in the steady state, the model predicts that there is greater Syk activation at the lower ligand concentration (2K1L = 10−4), than at the higher (2K1L = 104).
Table 1.
Ligand-receptor binding parameters which accentuate the asymmetry in the Syk activation curves. Other rate constants used in our calculations are taken from Ref. 4. The bimolecular rate constants k+1 and k+2 are divided by the inverse cell density (1.0 × 10−6 ml) and the cell surface area (8.0 × 10−6 cm2) to convert them to unimolecular rate constants.
| Parameter | Description | Value(s) |
|---|---|---|
| k+1 | ligand-receptor binding forward rate constant | 2.5 × 107M−1s−1 |
| k−1 | ligand-receptor binding reverse rate constant | 0.316(10−0.5)s−1 |
| k+2 | ligand-receptor cross-linking forward rate constant | 8.0 × 10−9 cm2mole−1s−1 |
| k−2 | ligand-receptor cross-linking reverse rate constant | 0.0316(10−1.5)s−1 |
Even though all signaling events are initiated by receptor aggregation, it appears that the concentrations of free and bound receptors which are not in aggregates influence signaling. To investigate the origin of the asymmetry, we vary both the the ligand-IgE association (k+1) and dissociation (k−1) constants, such that the corresponding equilibrium constant K1, and therefore the equilibrium cross-linking curve, remains unchanged. If the ligand-IgE dissociation constant is gradually reduced from 0.3 (10−0.5) to 0.0003 (10−3.5)s−1, the Syk activation at 2K1L = 10−4 remains practically unaltered, whereas the Syk activation at 2K1L = 104 increases and approaches the extent of Syk activation at 2K1L = 10−4 (Fig. 6b). On the other hand, if the ligand-IgE dissociation constant is increased from 0.3 to 103 s−1, the Syk activation at 2K1L = 104 remains unchanged and that at 2K1L = 10−4 decreases and approaches the Syk activation level at 2K1L = 104 for k−1 = 0.3 s−1 (Fig. 6c). In order to determine the source of asymmetry in the Syk activation curve, we consider a pair of reduced models, which are discussed in the following section.
3.4 Reduced models of Syk binding to cross-linked receptors
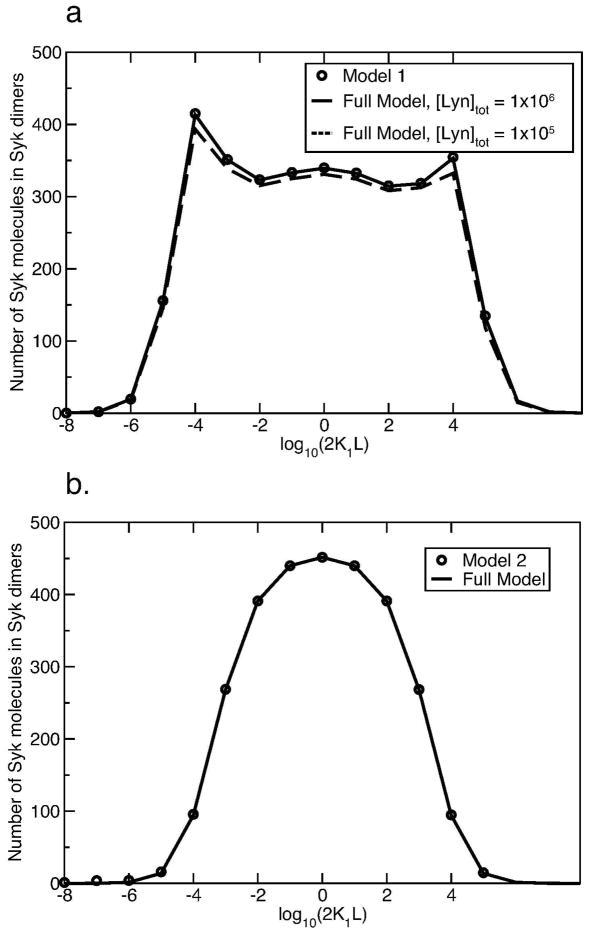
We consider two reduced models, 1 and 2, where the kinetic effects involving Lyn and the β subunit of the receptor have been eliminated. Both models correspond to the limit where the available Lyn is in large excess. In model 1, cross-linked receptors transphosphorylate each other on the γ subunit with a transphosphorylation rate that is the same as that of Lyn bound through it SH2 domain to the phosphorylated β subunit of the receptor. As in the full model, bound Syk protects the γ subunit from being dephosphorylated. The rate of dephosphorylation of unprotected phosphorylated γ subunits is the same as in the full model. As seen in Fig. 7a, model 1 exhibits an asymmetry in Syk dimer formation that approaches.
Figure 7.
Comparison of number of Syk dimers per cell obtained using reduced model 1 and 2, with the corresponding numbers obtained using the full model. [Syk]tot = 7 × 103 per cell; [FcεRI]tot = 105 per cell. We use the same ligand-receptor and Syk-p γ binding and unbinding parameters as in Table 2 for both the reduced models and the full model. (a) We consider two different total cellular concentrations of Lyn in the full model, [Lyn]tot = 105 and [Lyn]tot = 106 per cell. (b) Comparison of reduced model 2 to the full model when Lyn is in large excess and the rate of dephosphorylation is negligible ([Lyn]tot = 108 per cell, k−p = 10−4s−1).
In model 2, in addition to assuming Lyn is in excess, all receptor γ subunits are always phosphorylated, thereby eliminating the kinetic effects of receptor phosphorylation. This corresponds to the additional limit that the rate of receptor dephosphorylation goes to zero. For example, if in the full model the available Lyn is increased from 105 to 108 per cell and the rate constant for dephosphorylation reduced from 20 s−1 to 10−4 s−1, then a plot of Syk dimer formation for the full model and model 2 are identical (Fig. 7b). When this second assumption is made, the asymmetry in the Syk dimer curve is eliminated, indicating that the kinetics of phosphorylation and dephosphorylation of the γ subunit plays a role in creating the asymmetry.
3.5 Asymmetry in Syk activation originates from asymmetry in Syk dimer formation
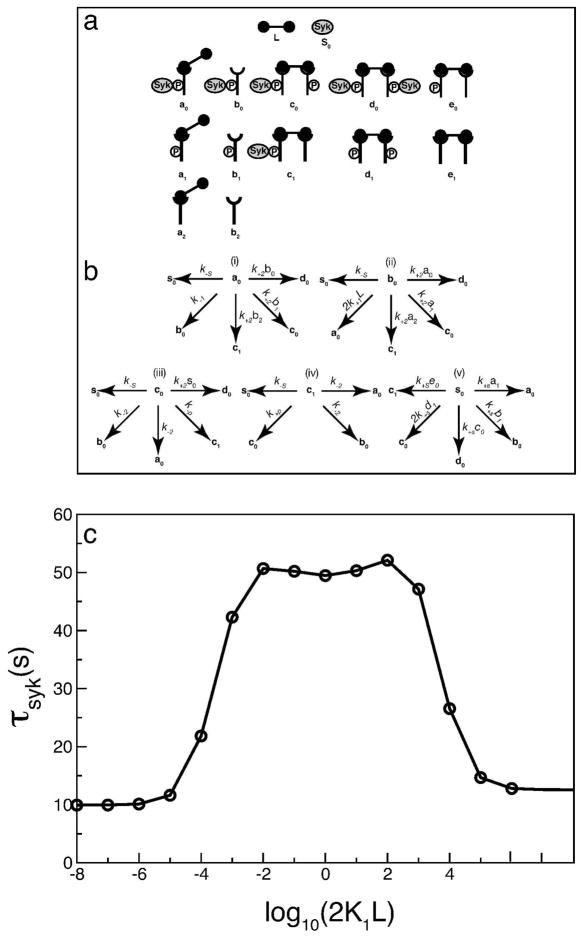
Since model 1 captures the asymmetry in Syk activation predicted by the full model, we use it to analyze the source of asymmetry in Syk dimer formation. As shown in Fig. 8b for model 1, one can form a Syk dimer (d0) in a number ways. We have obtained expressions for the mean time required by any particular Syk molecule to end up in a Syk dimer, starting as either cytosolic Syk or as part of a complex with other molecule(s).(See Appendix.) This mean time τSyk is not symmetric with respect to reflection about 2K1L = 1. The mean time at 2K1L = 10−4 is less than its counterpart at 2K1L = 104, as shown in Fig 8c.
Figure 8.
(a) Schematic representation of the different Syk containing species in the reduced model 1. We tag a particular Syk molecule and calculate the mean time required by this Syk molecule to be incorporated into a Syk dimer (d0). (b) (i–v) shows the different routes by which a tagged Syk molecule can be transferred from one species to another. (c) Mean time τSyk taken by any particular Syk molecule to be incorporated into a Syk dimer, starting as cytosolic Syk (s0) or as other specie(s) (a0, b0, c0, c1) in (a). The rate parameters are taken from Table. 2. [Syk]tot = 7 × 103 per cell; [FcεRI]tot = 105 per cell.
3.6 Asymmetry in Syk activation depends on the receptor-ligand dissociation constant
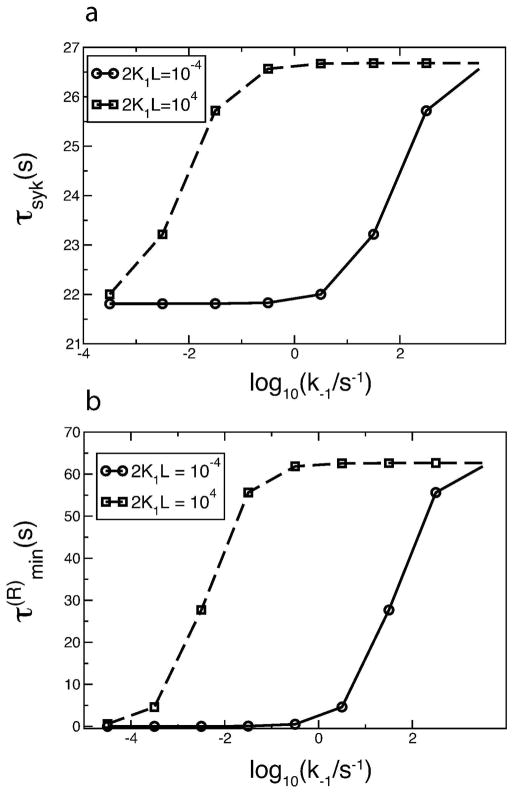
In Fig. 9a, we vary k−1 and k+1 keeping their ratio K1 fixed, and calculate the average time τSyk required by a Syk to end up in a Syk dimer. (See Appendix for the method used to calculate τSyk.) At k−1 = 10−3.5 s−1, the τSyk values at 2K1L = 10−4 and 2K1L = 104 are close to each other, so that there is little difference in the extent of Syk activation at these two ligand concentrations as was seen in Fig. 6b. As k−1 is increased from 10−3.5 s−1, τSyk at 2K1L = 10−4 remains unchanged while τSyk at 2K1L = 104 increases. This is consistent with our results from the full model, that Syk activation at 2K1L = 10−4 is essentially unchanged, while Syk activation at 2K1L = 104 decreases as k−1 is increased and K1 is kept fixed (Fig. 6b). The difference between the τSyk values at the two ligand concentrations increases, as does the asymmetry in Syk activation obtained using the full model. When k−1 is increased beyond 0.316 (10−0.5) s−1, τSyk at 2K1L = 10−4 increases while τSyk at 2K1L = 104 reaches a plateau. As a result, the difference in the τSyk values at these two ligand concentrations decreases with increasing k−1. As k−1 is increased to 103 s−1, τSyk at both 2K1L = 10−4 and 2K1L = 104 approaches the same value. This is the same trend exhibited by Syk activation in the full model (Fig. 6c). The strong correlation between τSyk from reduced model 1 and Syk activation in the full model supports the idea that the asymmetry in the Syk activation profile originates from the asymmetry in τSyk.
Figure 9.
Variation withk−1 of (a) average time (τSyk) required by any particular Syk molecule to end up in a Syk dimer and (b) minimum time ( ) required by a receptor to be incorporated into a receptor dimer, at ligand concentrations given by 2K1L = 10−4 and 2K1L = 104. The k−1 and k+1 are varied in such a way that K1 = k+1/k−1 remains fixed at 7.9 × 107 M−1. Other rate constants are taken from Table 2. [Syk]tot = 7 × 103 per cell; [FcεRI]tot = 105 per cell.
3.7 Mechanism for asymmetry
The question remains, how can the kinetics of ligand binding, which occurs on the outside of the cell, give rise to an asymmetry in the Syk activation curve, when the receptor cross-linking curve is symmetric? To answer the question we considered two receptors, one free and one bound. These receptors might, for example, have come from a dimer when it dissociated and their cytoplasmic domains may be associated with Syk or Lyn or both. In the Appendix we show how to calculate two quantities at equilibrium, τR and τB, the mean times respectively of an unbound receptor to enter into an aggregate and for a bound receptor that is not cross-linked to a second receptor to enter into an aggregate (Fig. 10a). In Fig. 10b we show these mean times as a function of the bivalent ligand concentration. Neither are symmetric, nor is their minimum, although their sum is symmetric (Fig. 10c). Monovalent binding is responsible for the asymmetry. Consider the mean times at two free ligand concentrations, L+ and L−, where log(2K1L+) = −log(2K1L−). At L−, the bound receptor returns to a dimer more rapidly than the unbound receptor because it is present in the lowest concentration, and so it more rapidly finds a partner to crosslink. At L+ the opposite is true. The asymmetry arises in the minimum time for a receptor to reform a dimer, , because the free ligand concentration has an asymmetric effect on the lifetime of the minority receptor (the receptor at lowest concentration). At high ligand concentrations, minority receptors (unbound) are rapidly converted to bound receptors giving them less time to enter into a dimer. Thus, they may have to undergo interconversion between unbound and bound forms many times before forming a dimer. At low ligand concentrations, the initial conversion of the minority receptors (bound) to the unbound form is independent of the ligand concentration, depending only on the rate of dissociation.
Figure 10.
(a) Rate constants that characterize the transitions that a free receptor and a bound receptor not in an aggregate can undergo. (b) The mean times for a free receptor (τR) and a bound receptor (τB) to find a partner and form a dimer as a function of the bivalent ligand concentration. (c) The mean τave of τR and τB and the minimum of τR and τB as a function of the ligand concentration. In parts (b) and (c), the ligand receptor binding and cross-linking parameters are taken from Table 2 and [FcεRI]tot = 105 per cell.
In Fig. 9b we keep the equilibrium constant K1 = k+1/k−1 fixed and, for the same set of parameters as in Fig. 9a, vary k−1. We observe that the dependence of on log10(k−1/s−1) at the two ligand concentrations given by 2K1L = 10−4 and 2K1L = 104 is qualitatively similar to that of τSyk. Since receptor dimerization is upstream from, and necessary for Syk activation, we infer that the dependence of τSyk on k−1 results from the corresponding dependence of on k−1.
4 Discussion
On mast cells and basophils, mutivalent ligands (allergens) bind to, and aggregate, IgE complexed with FcεRI. We have used a model [4] of the early cell signaling events (seconds to minutes) mediated by the dimerization of IgE-FcεRI complexes to study the dependence of the activation of Syk on the cellular concentrations of Syk and IgE-FcεRI complexes. In the model, full activation of Syk can only occur when two Syk molecules are bound to a receptor dimer formed by a bivalent ligand. A Syk molecule becomes fully activated when it is transphosphorylated on its activation loop tyrosines (Tyr518 and Tyr519 in murine Syk) by a Syk bound to the adjacent receptor in the dimer. Using antibodies specific for phosphorylated tyrosines in the activation loop of Syk, Zhang et al. [7] showed that upon FcεRI aggregation, these tyrosines became phosphorylated and the phosphorylation depended on the kinase activity of Syk. Further, the presence of these activation loop tyrosines was necessary for Syk-mediated propagation of FcεRI signaling [23]. Syk is capable of phosphorylating its activation loop tyrosines both in vitro [7, 24] and in vivo [7], but it is still unresolved whether transphosphorylation (phosphorylation of one Syk molecule by another in the same complex) or autophosphorylation (phosphorylation of a Syk molecule by the kinase domain of the same molecule) is the mechanism by which the Syk activation loop tyrosines are phosphorylated in mast cells and basophils.
If the mechanism of Syk activation is Syk transphosphorylation of Syk, as our model postulates [4], the Syk activation curve can be either bell-shaped or double-humped, depending on the cellular concentrations of Syk and IgE-FcεRI complexes (see Figure 3). If the mechanism of Syk activation is through autophosphorylation the model predicts that only a bell-shaped Syk activation curve is possible. In the model it is assumed that because of steric hindrance only one Syk molecule can bind per receptor, even though there are two disulfide-linked γ-chains that, when fully phosphorylated, present two binding sites for Syk. If two Syk can be bound simultaneously to the same receptor on two separate γ-chains, and if they can transphosphorylate each other to achieve full Syk activation, the prediction is still that Syk activation curves can be either bell-shaped or double humped depending on the ratio of the receptor to Syk concentrations.
Unfortunately, at present there are no experimentally-determined Syk activation curves to help us distinguish among possible mechanisms for the phosphorylation of the Syk activation loop tyrosines. If we look further downstream than Syk activation at histamine release from basophils, which requires kinase active Syk, the data are consistent with the Syk transphosphorylation mechanism. For example, although most histamine release curves show only a single maximum, double-humped histamine release curves have been observed [25, 26]. (Histamine release curves are log-log plots of the fraction of a cell’s total histamine that is released in a given time versus the concentration of ligand the cell is exposed to.) In addition, for ligand concentrations where receptor aggregate formation is high and histamine release low, the addition of monovalent hapten, which reduces the number of receptors in dimers, increases histamine release [20, 21]. In our simulations the same manipulation increases Syk activation (Figure 5). To study the properties of Syk activation we performed simulations over a range of ligand concentrations for different Syk and receptor concentrations, and we found that even though the receptor cross-linking curve was symmetric with respect to the log of the ligand concentration in the steady state (Figure 1), the Syk activation curve often was not. This was easiest to see when the Syk activation curve was doubled-humped (Figure 6). The asymmetry in the steady state occured when, at two different ligand concentrations where the total number of receptors in dimers and the lifetime of a receptor in a dimer was identical, the levels of Syk activation differed. This was puzzling since all that differed on the cell surface at the two ligand concentrations was the number of receptors bound monovalently. Why should this matter since basophils and mast cells don’t respond to monovalently bound ligand? The answer is subtle and concerns the competition between kinetic proofreading, that activation requires the receptor to undergo multiple binding and chemical modification events while the receptor is in an aggregate, and serial engagement, that ligands can interact with multiple receptors before dissociating from the cell surface.
When a receptor dimer breaks up, the two receptors will diffuse away from each other, one with a ligand attached and one free. If Syk or Lyn is bound to the receptors, these kinases will dissociate with halflives of about ten seconds, leaving behind unprotected ITAM phosphotyrosines to be rapidly dephosphorylated. The result is the return of the receptors to their basal state. If, however, a receptor can re-form a dimer before it has been transformed back to its basal state, it can partially negate the effects of kinetic proofreading. The asymmetry in the Syk activation curve arises because the minimum time required by a receptor to form a dimer depends on the free ligand concentration (Figure 10). At low ligand concentration, and therefore low numbers of bound receptors, a singly bound receptor will collide with many free receptors and have ample opportunity to rapidly form a dimer. At high ligand concentration, and therefore low numbers of free receptors, a free receptor will have many bound receptors to collide with, but, because the free ligand concentration is high, the free receptor may bind a ligand from solution before it has time to form a dimer. Thus, at low ligand concentration re-formation of dimers is more effective than at high ligand concentration at reducing the effects of kinetic proofreading. In our simulations the double-humped Syk activation curve (Figure 6) shows Syk activation higher for the first maximum (at the lower ligand concentration) than at the second maximum (at the higher ligand concentration) where the extent of receptor cross-linking is the same at both maxima.
To further probe the underlying mechanism of the asymmetry in the Syk activation curve we considered the model in the limit when Lyn is in large excess and γ-ITAM phosphorylation follows immediately upon dimer formation. In this limit simulations again showed an asymmetry in the double-humped Syk activation curve (Fig. 7a). Since for full Syk activation to occur a Syk dimer must form (Syk-FcεRI-ligand-FcεRI-Syk), we calculate the mean time for a Syk molecule that is not in a Syk dimer to be incorporated into a Syk dimer. The time for this to occur showed a similar asymmetry as that of the minimum time for a receptor to enter a dimer. It appears that the asymmetry in the Syk activation curve is a manifestation of the underlying competition that is constantly occurring between kinetic proofreading and serial engagement in signaling cascades triggered by multivalent ligands aggregating cell surface receptors.
The asymmetry we observed in simulations of Syk activation curves was always small. Although the explanation of the effect is interesting, whether it is ever important in cell signaling is an open question. Since amplifications of cell signals can be highly nonlinear, it is possible that small differences in the concentration of activated kinases early in a signaling cascade can have profound effects on later cell responses. A major function of activated Syk in mast cells and basophils is to phosphorylate a set of tyrosines on the transmembrane adapter protein LAT (linker for the activation of T cells). LAT’s three distal tyrosines, when phosphorylated, all bind the adapter protein Grb2, and Grb2 mediates the aggregation of LAT [27]. The size of the LAT aggregates that form depends strongly on whether or not all three of the Grb2 binding site tyrosines are phosphorylated [28]. Small differences in the amount of activated Syk might lead to large differences in the distribution of LAT aggregates that form downstream of Syk.
Acknowledgments
This work was supported by NIH Grant R37-GM035556 and by the Department of Energy through contract W-7405-ENG-36. The authors have no financial conflict of interest.
Appendix
Mean time for a Syk molecule to enter into a Syk dimer
We calculate for model 1, the mean times taken by a particular (tagged) Syk molecule to end up in a Syk dimer (Syk-FcεRI-Ligand-FcεRI-Syk) starting as either free Syk or as part of a Syk containing complex other than the Syk dimer. In Fig. 8a, we assign notations to different Syk containing species in the reduced model. The mean time taken to form a dimer from species i is given by τi. The kinetic parameters used are described in Table 2. We define the mean time of incorporation of the tagged Syk molecule in species d0, starting from species i, in the following fashion. This mean time is divided into two parts, the first part corresponding to the mean time of transition from species i to all other species containing the tagged Syk molecule. This is the inverse of the sum of rates of all processes that transform species i to the other species. The second part represents the mean remaining time to incorporate the tagged Syk in the Syk dimer species d0. In terms of the species from Fig. 8a and the kinetic parameters from Table 2, the mean times of transition from species a0, b0, c0, c1 and s0 respectively.
Table 2.
Kinetic parameters used in reduced model with receptor γ phosphorylation-dephosphorylation. The bimolecular rate constants are divided by the inverse cell density (1.0 × 10−6 ml) and the cell surface area (8.0 × 10−6 cm2) to convert them to unimolecular rate constants.
| Parameter | Description | Value(s) |
|---|---|---|
| k+1 | ligand-receptor binding forward rate constant | 2.5 × 107M−1s−1 |
| k−1 | ligand-receptor binding reverse rate constant | 0.316(10−0.5)s−1 |
| k+2 | ligand-receptor cross-linking forward rate constant | 8.0 × 10−9 cm2mole−1s−1 |
| k−2 | ligand-receptor cross-linking reverse rate constant | 0.0316(10−1.5)s−1 |
| k+gP | receptor γ phosphorylation forward rate constant | 3.0 s−1 |
| k−gP | receptor γ dephosphorylation rate constant | 20s−1 |
| k+S | Syk-phosphorylated receptor γ binding forward rate constant | 6.0 × 10−5 molecule−1s−1 |
| k−S | Syk-phosphorylated receptor γ reverse rate constant | 0.13s−1 |
| (1) |
| (2) |
| (3) |
| (4) |
| (5) |
In terms of the above quantitites, τi can be defined as follows.
| (6) |
| (7) |
| (8) |
| (9) |
| (10) |
The above equations are solved for τa0, τb0, τc0, τc1 and τs0 using parameters from Table 2 for different L values corresponding to log10(2K1L) values from −8 to 8. The expression levels of Syk and FcεRI receptor per cell are taken as 7 × 103 and 105 respectively. The mean time for any tagged Syk molecule to be incorporated into a Syk dimer (d0) is given by
| (11) |
where
| (12) |
and [Syk]tot = a0 + b0 + c0 + c1 + 2d0 + s0. The quantity τSyk is plotted as a function of log10(2K1L) in Fig. 8.
Mean time for a receptor to form a receptor dimer
We wish to calculate two quantities at equilibrium, τR, the mean time for an unbound receptor to enter into an aggregate and τB, the mean time for a bound receptor that is not in an aggregate to enter into an aggregate. We consider the case where the receptor is monovalent and the ligand is bivalent. There are two possible transitions an unbound receptor can undergo. It can interact with a bound receptor to form a dimer or it can bind to a ligand in solution to become a bound receptor. Similarly there are two possible transitions a bound receptor that is not in an aggregate can undergo. It can interact with a free receptor to form a dimer or it can dissociate from its ligand and become a free receptor. These transitions are illustrated in Fig. 10a where the rates of transition are given.
The mean times τR and τB will be found by solving two linear algebraic equations that are functions of the equilibrium concentrations of the free receptors, R, and the bound receptors not in dimers, B. At equilibrium these concentrations are calculated from the single site equilibrium constants for binding and crosslinking, K1 and K2 respectively, and the initial receptor and ligand concentrations, RT and LT. However, for our purposes expressing the results interms of the free ligand concentration is more useful.
If there are RT total receptors on the cell surface and there are no processes that remove or add new receptors the the total number of receptors is conserved and
| (13) |
where D is the concentration of receptor dimers. Defining r = R/RT as the fraction of free receptors Eq. (13) becomes
| (14) |
It is straight forward to show that w, the fraction of receptors not in dimers is given by
| (15) |
where
| (16) |
It follows that
| (17) |
| (18) |
We next write down the equations for the mean times at equilibrium.
| (19) |
| (20) |
The first term in each equation is the mean time to make a transition, while the second term is the mean remaining time after the first transition until the receptor is incorporated into a dimer. We define the following quantities:
| (21) |
| (22) |
The ts are mean times to make a transition while the ps are probabilities of making specific transitions. At equilibrium p1 = p2, because the probability of a ligand dissociating from a bound receptor is equal to the probability of a free receptor binding a ligand. Solving Eqs. (19) and (20) we have
| (23) |
| (24) |
References
- 1.Rivera J, Gilfillan AM. Molecular regulation of mast cell activation. J Allergy Clin Immunol. 2006;117:1214–1225. doi: 10.1016/j.jaci.2006.04.015. [DOI] [PubMed] [Google Scholar]
- 2.MacGlashan D., Jr IgE and FcεRI regulation. Ann N Y Acad Sci. 2005;1050:73–88. doi: 10.1196/annals.1313.009. [DOI] [PubMed] [Google Scholar]
- 3.Blank U, Ra C, Miller L, White K, Metzger H, Kinet JP. Complete structure and expression in transfected cells of high affinity IgE receptor. Nature. 1989;337:187–189. doi: 10.1038/337187a0. [DOI] [PubMed] [Google Scholar]
- 4.Faeder JR, Hlavacek WS, Reischl I, Blinov ML, Metzger H, Redondo A, et al. Investigation of Early Events in FcεRI-Mediated Signaling Using a Detailed Mathematical Model. J Immunol. 2003;170:3769–3781. doi: 10.4049/jimmunol.170.7.3769. [DOI] [PubMed] [Google Scholar]
- 5.Kambayashi T, Koretzky GA. Proximal signaling events in FcεRI-mediated mast cell activation. J Allergy Clin Immunol. 2007;119:544–552. doi: 10.1016/j.jaci.2007.01.017. [DOI] [PubMed] [Google Scholar]
- 6.Shiue L, Zoller MJ, Brugge JM. Syk is activated by phosphotyrosine-containing peptides representing the tyrosine-based activation motifs of the high affinity receptor for IgE. J Biol Chem. 1995;270:10498–10502. doi: 10.1074/jbc.270.18.10498. [DOI] [PubMed] [Google Scholar]
- 7.Zhang J, Billingsley ML, Kincaid RL, Sirgaganiam RP. Phosphorylation of Syk activation loop tyrosines is essential for Syk function: an in vivo study using a specific anti-Syk activation loop phosphotyrosine antibody. J Biol Chem. 2000;275:35442–35447. doi: 10.1074/jbc.M004549200. [DOI] [PubMed] [Google Scholar]
- 8.Rivera J. NTAL/LAB and LAT: a balancing act in mast-cell activation and function. Trends in Immunol. 2005;26:119–122. doi: 10.1016/j.it.2005.01.001. [DOI] [PubMed] [Google Scholar]
- 9.MacGlashan DW., Jr Relationship between spleen tyrosine kinase and phosphatidylinositol 5′ phosphatase expression and secretion from human basophils in the general population. J Allergy Clin Immunology. 2007;119:626–633. doi: 10.1016/j.jaci.2006.09.040. [DOI] [PubMed] [Google Scholar]
- 10.MacGlashan DW, Jr, Lavens-Phillips S. Characteristics of the free cytosolic calcium timelag following IgE-mediated stimulation of human basophils: significance for the non-releasing basophil phenotype. J Leukoc Biol. 2001;69:224–232. [PubMed] [Google Scholar]
- 11.Dembo M, Goldstein B. Theory of Equilibrium Binding of Symmetric Bivalent Haptens to Cell Surface Antibody: Application to Histamine Release from Basophils. J Immunol. 1978;121:345–353. [PubMed] [Google Scholar]
- 12.MacGlashan DW, Jr, Dembo M, Goldstein B. Test of a Theory Relating to the Cross-Linking of IgE Antibody on the Surface of Human Basophils. J Immunol. 1985;135:4129–4134. [PubMed] [Google Scholar]
- 13.Hlavacek WS, Faeder JR, Blinov ML, Perelson AS, Goldstein B. The Complexity of Complexes in Signal Transduction. Biotechnolgy and Bioengineering. 2003;84:783–794. doi: 10.1002/bit.10842. [DOI] [PubMed] [Google Scholar]
- 14.Kulczycki AJ, Metzger H. The interaction of IgE with rat basophilic leukemia cells. II. Quantitative aspects of the binding reaction. J Exp Med. 1974;140:1676–1695. doi: 10.1084/jem.140.6.1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hong JJ, Yankee TM, Harrison ML, Geahlen RL. Regulation of signaling in B cells through the phosphorylation of Syk on linker-region tyrosines. A mechanism for negative signaling by the Lyn tyrosine kinase. J Biol Chem. 2002;277:31703–31714. doi: 10.1074/jbc.M201362200. [DOI] [PubMed] [Google Scholar]
- 16.Blinov ML, Faeder JR, Goldstein B, Hlavacek WS. BioNetGen: Software for rule-based modeling of signal transduction based on the interactions of molecular domains. Bioinformatics. 2004;20:3289–3292. doi: 10.1093/bioinformatics/bth378. [DOI] [PubMed] [Google Scholar]
- 17.Faeder JR, Blinov ML, Hlavacek WS. Rule-based modeling of biochemical systems with BioNetGen. Methods Mol Biol. 2009;500:113–167. doi: 10.1007/978-1-59745-525-1_5. [DOI] [PubMed] [Google Scholar]
- 18.Becker KE, Ishizaka T, Metzger H, Ishizaka K, Grimley PM. Surface IgE on human basophils during histamine release. J Exp Med. 1973;138:394–409. doi: 10.1084/jem.138.2.394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dembo M, Goldstein B. A model of cell activation and desesitization by surface immunoglobin: the case of histamine release from human basophils. Cell. 1980;22:59–67. doi: 10.1016/0092-8674(80)90154-3. [DOI] [PubMed] [Google Scholar]
- 20.Magro AM, Alexander A. Histamine Release: In vitro studies of the inhibitory region of the dose-response curve. J Immunol. 1974;112:1762–1765. [PubMed] [Google Scholar]
- 21.Magro AM, Bennich H. Concanavalin A induced histamine release from human basophils in vitro. Immunology. 1977;33:51–58. [PMC free article] [PubMed] [Google Scholar]
- 22.Goldstein B, Wofsy C. Aggregation of Cell Surface Receptors. Lectures on Mathematics in the Life Sciences. 1994;24:109–135. [Google Scholar]
- 23.Zhang J, Kimura T, Siraganian RP. Mutations in the activation loop tyrosines of protein tyrosine kinase Syk abrogate intracellular signaling but not kinase activity. J Immunol. 1998;161:4366–4374. [PubMed] [Google Scholar]
- 24.Tsang E, Giannetti A, Shaw D, Dinh M, Tse JKY, Gandhi S, et al. Molecular mechanism of the Syk activation switch. J Biol Chem. 2008;283:32650–32659. doi: 10.1074/jbc.M806340200. [DOI] [PubMed] [Google Scholar]
- 25.DeLIsi C, Siriganian RP. Receptor cross-linking and histamine release: II Interpretation of and analysis of anomalous dose response patterns. J Immunol. 1979;122:2293–2299. [PubMed] [Google Scholar]
- 26.Weyer A, Dandeu JP, Marhand F, David B. In vitro histamine release from human basohils triggered by a purified allergen from Dermatophagoides Farinac: Bimodel aspect of the dose response curve. Ann Immunol Inst Pasteur. 1982:133. [PubMed] [Google Scholar]
- 27.Houtman JC, Yamaguchi H, Barda-Saad M, Braiman A, Bowden B, Appella E, et al. Oligomerization of signaling complexes by the multipoint binding of GRB2 to both LAT and SOS1. Nat Struct Mol Biol. 2006;13:798–805. doi: 10.1038/nsmb1133. [DOI] [PubMed] [Google Scholar]
- 28.Nag A, Monine M, Faeder JR, Goldstein B. Aggergation of membrane proteins by cytosloic cross-linkers: Theory and simulation of the Lat-Grb2-Sos1 system. Biophys J. 2009;96:2604–2623. doi: 10.1016/j.bpj.2009.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]