Abstract
Objectives
To explore 1) Whether arthritis associates with poorer self-efficacy and motivation for, and participation in, two specific types of physical activity (PA): Endurance training (ET) and strength training (ST), and 2) If the added burden of diabetes contributes to a further reduction in these PA determinants and types.
Methods
Self-efficacy and motivation for exercise and minutes per week of ET and ST were measured in 347 older Veterans enrolled in a home-based PA counseling intervention. Regression analyses were used to compare high versus low self-efficacy and motivation and PA minutes in persons without arthritis, with arthritis alone, and with arthritis plus diabetes.
Results
Persons with arthritis alone reported lower self-efficacy for ET and ST than those without arthritis (Odds ratio[OR]ET 0.71 (0.39,1.20); ORST 0.69 (0.39,1.20)). A further reduction in self-efficacy for these two types of PA was observed for those with both arthritis and diabetes (ORET 0.65 (0.44,0.92); ORST 0.64 (0.44,0.93); trend P<0.001). There was no trend towards a reduction in motivation for PA in those with arthritis alone or arthritis and diabetes. Persons with arthritis exhibited higher motivation for ET than those without arthritis (ORET 1.85 (1.12,3.33). There were no significant differences between the three groups in minutes of ET (P=0.93), but persons with arthritis plus diabetes reported significantly less ST compared to individuals with arthritis only (P=0.03).
Conclusions
Despite reduced self-efficacy for ET and ST and less ST in older persons with arthritis, motivation for both PA types remains high, even in the presence of diabetes.
Keywords: exercise self-efficacy, co-morbidity, strength training
Estimates suggest that arthritis affects nearly 60% of persons 65 years and older[1]. While the recommended management of arthritis includes both pharmacologic and nonpharmacologic approaches, such as physical activity[2], the presence of co-morbidities increases complexity for arthritis-related health professionals who are recommending and prescribing exercise programs. With the steep rise in rates of type 2 diabetes[3] and the role of exercise in the management of both of these conditions, there is an imperative to increase understanding about the impact of diabetes on arthritis management, including physical activity.
In the general population and in persons with arthritis, one determinant of physical activity consistently reported is self-efficacy, and persons with arthritis typically report poorer self-efficacy for exercise [4-7]. A central tenet of Bandura's Social Cognitive Theory[8], self-efficacy reflects individual capabilities to complete a specific task or goal such that self-efficacy for exercise reflects an individual's confidence in their ability to perform regular exercise. Similarly, motivation for exercise, a cognitive process in which a person becomes motivated or demotivated by virtue of comparing individual ability for the task and the importance of the expected outcome[9, 10], is also an important predictor of increased physical activity[11]. Social Cognitive Theory suggests that efficacy beliefs play a central role in the relationship between behavior and motivation, with efficacy beliefs determining the type of behavior, the amount of effort expended, and individual level of perseverance [8, 12].
In light of the newly released US physical activity guidelines that recommend regular physical activity comprised of both endurance and strength training for older adults with arthritis and/or diabetes[13], it is important to know more about how older adults approach these modes of exercise. To date, little research has examined older adults' beliefs regarding their ability to succeed at, or their motivation to participate in, endurance or strength training [14]. Examining older adults' efficacy beliefs and motivation for endurance and strength training will inform the development of interventions to increase these recommended behaviors among older adults with chronic conditions.
In this study, our objective was to explore whether arthritis contributes to poorer self-efficacy and motivation for and less participation in endurance training and strength training, and if the added burden of diabetes contributes to a further reduction in these determinants and types of physical activity. We hypothesized that, as compared to persons without arthritis, older adults with arthritis would exhibit poorer self-efficacy and motivation for both types of physical activity, and that individuals with both arthritis and diabetes would display even poorer self-efficacy and motivation for both endurance and strength training. Further, we hypothesized that as compared to persons without arthritis, older adults with arthritis would engage in less endurance and strength training physical activity than older adults without arthritis that individuals with both arthritis and diabetes would report even lower levels of these types of physical activity compared to those with arthritis alone.
Methods
Design and Subjects
This is a secondary analysis of a data set collected as part of a clinical trial assessing the impact of home-based counseling on exercise. Participants were enrolled in Veterans Life Study (Learning to Improve Fitness and Function in Elders)[15], a home-based physical activity counseling intervention directed at walking and lower extremity strength training, including the use of elastic bands. Inclusion criteria included age≥70 years, ability to walk 30 feet without human assistance, engaging in less than 150 minutes per week of physical activity. Exclusion criteria included a medical chart diagnosis of chronic pain that prevented exercise, a terminal diagnosis, unstable angina, a history of ventricular tachycardia, chronic obstructive lung disease requiring two hospitalizations in the last 12 months, uncontrolled hypertension, stroke with moderate to severe aphasia, a diagnosis of a mental or behavioral disorder, active substance abuse, dementia, and severe hearing or visual loss. The Durham Veterans Affairs Medical Center Institutional Review Board approved the protocol, and informed consent was obtained from each participant.
Of the original Life participants (n= 400), this report compared participants at baseline who had no arthritis or diabetes (n=85), arthritis only (n=178), and arthritis plus diabetes (n=84). Since the main objective of this analysis was to evaluate the contribution of diabetes to poorer efficacy beliefs in persons with arthritis, persons with diabetes only (n=53) were not included in this analysis.
Measures
Age and gender were obtained from the computerized medical record system while race and education were based on self-report at baseline. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. General health, physical function, and vitality were assessed with respective subscales of the Short Form Health Survey (SF-36; [16]).
Arthritis and diabetes were determined via self-report at baseline with a modified version of the Older Americans Resources and Services (OARS) co-morbidity index[17]. The OARS asks if participants have one or more of thirty-five specific diseases or conditions, including “arthritis and rheumatism” and “diabetes,” and respondents indicate “yes,” “no,” or “don't know.”
Using the current physical activity guidelines as a reference[18] and adapted from validated measures of self-efficacy[19], self-efficacy for endurance training was assessed by asking participants, “How sure are you that you could walk or do another type of endurance exercise for 30 minutes or more on five or more days per week? The 30 minutes does not have to be done all at the same time.” Self-efficacy for strength training was assessed by asking participants, “How sure are you that you could do exercises for 15 minutes, three days a week to make your legs stronger? The strength training exercises could be as easy as using elastic exercise bands.”
Similarly, motivation for exercise was assessed by asking participants, “How much do you want to walk or do another type of endurance exercise for 30 minutes on five or more days of the week? The 30 minutes does not have to be done all at the same time,” to assess endurance motivation and “How much do you want to do exercises for 15 minutes, three days a week, to make your legs stronger? The strength exercises could be as easy as using elastic bands” to assess motivation for strength training. Both the efficacy and motivation scales used a 5-point scale from (1) not at all to (5) extremely, with higher scores reflecting greater efficacy and greater motivation.
Participants who answered “very” (4) or “extremely sure” (5) on the self-efficacy items were dichotomized as high self-efficacy and those who answered “somewhat sure” (3), “a little sure” (2), or “not at all sure” (1) as low self-efficacy. Patients who answered “very much” (4) or “completely” (5) on the motivation items were dichotomized as high motivation and those who answered “somewhat” (3), “a little bit” (2), or “not at all” (1) as low motivation.
Amounts of reported physical activity were determined using the Community Health Activities Model Program for seniors (CHAMPS)[20], a self-report questionnaire designed specifically for use among older adults, that assesses average weekly frequency and duration of social and physical activities typically performed by older adults.
Data Analysis
Baseline demographic characteristics and amounts of reported physical activity were compared with bivariate analyses of variance and Chi-squared goodness-of-fit statistics. Logistic regression was used to evaluate the impact of arthritis and diabetes on the dichotomized measures of self-efficacy and motivation for physical activity. Similarly, linear modeling was used to determine if minutes of endurance and strength training physical activity differed for those with no arthritis, arthritis only, and arthritis plus diabetes. In an exploratory fashion, if an omnibus test of group (no arthritis; arthritis only; arthritis plus diabetes), as it was associated with the measure of physical activity or self-efficacy, was significant at P<0.10 (defined a priori as part of the analytic plan), pair-wise comparisons were made between each group. Statistical significance for pair-wise comparisons and trend tests was established as P<0.05. The tendency for self-efficacy to decrease with the addition of arthritis and diabetes was also assessed with a 1 degree of freedom trend test over the 3 ordered groups. All analyses were adjusted for baseline measures of BMI, race, age, and education.
Results
We have previously reported the demographic characteristics of this Veteran population[15, 21], which is comprised predominantly of Caucasian men with an average age of 78. For this subset of VA LIFE participants, there were no significant differences in age, race, BMI, education, or self-reported baseline physical activity when compared to those not included in this analysis (P>0.05 for all). Demographic and baseline physical activity by arthritis and diabetes diagnoses are indicated in Tables 1 and 2, respectively.
Table 1.
Baseline Characteristics
| No Arthritis or Diabetes n =85 |
Arthritis alone n =178 |
Arthritis + Diabetes n =84 |
Omnibus P value |
|
|---|---|---|---|---|
| Age – years ± SD | 77.7 ± 5.5 | 77.7 ± 5.1 | 77.3 ± 4.1 | 0.83 |
| Race - Caucasian (%) | 76.5 | 80.3 | 72.6 | 0.37 |
| BMI (weight/height2) - mean ± SD | 28.2 ± 4.6 | 28.6 ± 4.4 | 30.4 ± 4.9†¶ | 0.003 |
| Education - No college (%) | 36.5 | 43.8 | 57.1† | 0.02 |
| SF-Health Quality of Life (% Excellent, V. Good, Good) | 85.9 | 79.8 | 64.3 † | 0.009 |
| SF-Physical Function (mean±SD) | 75.2 ± 19.4 | 64.2 ± 21.8† | 57.0 ± 24.5† | <0.0001 |
| SF-Vitality (higher score means more vitality) (mean±SD) | 65.0 ± 21.0 | 55.6 ± 18.7† | 49.3 ± 22.4†¶ | <0.0001 |
P-value not estimable
P value < 0.05 for pairwise comparison with no arthritis group.
P value < 0.05 for pairwise comparison with arthritis only group.
Statistical comparisons for SF36 subscale measures were adjusted for age, race, BMI, and education.
Table 2.
Comparison of Self-Efficacy, Motivation, and Physical Activity in Persons with No Arthritis, Arthritis Alone, and Arthritis Plus Diabetes.
| No Arthritis or Diabetes n =85 |
Arthritis alone n =178 |
Arthritis + Diabetes n =84 |
Omnibus P value |
|
|---|---|---|---|---|
| Endurance | ||||
| Training Self- Efficacy |
||||
| Not at all sure | 5.9 | 6.7 | 10.7 | |
| A little sure | 8.2 | 21.9 | 27.4 | |
| Somewhat sure | 25.9 | 21.9 | 26.2 | |
| Very sure | 38.8 | 39.9 | 26.2 | |
| Extremely sure | 18.8 | 9.0 | 8.3 | 0.03 |
| Don't know/missing | 2.4 | 0.6 | 1.2 | |
| 57.7 | 48.9 | 34.5† | ||
| Very sure and Extremely sure (%) |
||||
|
| ||||
| Strength Training | ||||
| Self-Efficacy | ||||
| Not at all sure | 3.5 | 2.3 | 8.3 | |
| A little sure | 10.6 | 10.11 | 20.2 | |
| Somewhat sure | 16.5 | 27.5 | 26.2 | |
| Very sure | 44.7 | 42.7 | 35.7 | |
| Extremely sure | 23.5 | 16.9 | 8.3 | 0.01 |
| Don't know/missing | 1.2 | 0.6 | 1.2 | |
| 59.6 | 44.1† | |||
| Very sure and Extremely sure (%) |
68.2 | |||
|
| ||||
| Endurance | ||||
| Training | ||||
| Motivation | ||||
| Not at all | 8.2 | 2.8 | 2.4 | |
| A little bit | 15.3 | 14.6 | 9.5 | |
| Somewhat | 32.9 | 24.2 | 34.5 | |
| Very much | 36.5 | 48.9 | 45.2 | 0.06 |
| Completely | 5.9 | 9.0 | 7.1 | |
| Don't know | 1.2 | 0.6 | 1.2 | |
| Very much and Completely (%) |
42.4 | 57.9† | 52.4¶ | |
|
| ||||
| Strength Training | ||||
| Motivation | ||||
| Not at all | 10.6 | 1.7 | 3.6 | |
| A little bit | 5.9 | 5.1 | 3.6 | |
| Somewhat | 20.0 | 27.0 | 31.0 | |
| Very much | 47.1 | 56.2 | 48.8 | 0.46 |
| Completely | 15.3 | 9.6 | 11.9 | |
| Don't know/missing | 1.2 | 0.6 | 1.2 | |
| Very much and Completely (%) |
62.4 | 65.7 | 60.7 | |
|
| ||||
| Moderate endurance exercise activity (min/w) − mean ± SD |
41.7 ± 78.8 | 35.4 ± 93.0 | 32.3 ± 76.7 | 0.93 |
|
| ||||
| Moderate strength training activity (min/w) − mean ± SD |
19.7 ± 44.0 | 28.4 ± 62.3 | 12.3 ± 30.1¶ | 0.07 |
P value < 0.05 for pairwise comparison with no arthritis group.
P value < 0.05 for pairwise comparison with arthritis only group.
Statistical comparisons for all outcomes were adjusted for age, race, BMI, and education.
Self-efficacy for Endurance Training
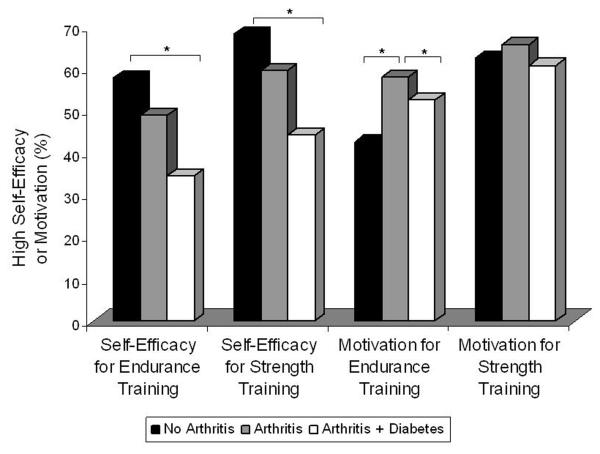
The omnibus test of group (no arthritis; arthritis only; arthritis plus diabetes), as it was associated with self-efficacy for endurance training was significant, indicating group differences in self-efficacy for endurance training (P=0.03; Table 2). As seen in Figure 1, compared to persons without arthritis, persons with arthritis reported poorer self-efficacy for endurance training (58% versus 49% high self-efficacy respectively, pairwise P=0.16; adjusted OR for high self-efficacy 0.71 (0.39, 1.20)). As expected, self-efficacy for endurance training was even poorer among those with both arthritis and diabetes (35% high self-efficacy) compared to the no arthritis group (pairwise P=0.02; adjusted OR for high self-efficacy 0.65 (0.44, 0.92)) and the arthritis alone group (pairwise P=0.81; adjusted OR for high self-efficacy 0.97 (0.69, 1.26)). Using a trend test, there was a significant decline in self-efficacy for endurance training with the added burdens of arthritis and arthritis plus diabetes (P=0.009).
Figure 1. Impact of Arthritis and Diabetes on Self-Efficacy and Motivation for Physical Activity.
Persons with no arthritis (n=85), arthritis only (n=178), and arthritis plus diabetes (n=84) indicated self-efficacy and motivation for moderate intensity endurance and strength training using a 5-point scale with higher scores reflecting greater efficacy and greater motivation . Responses were dichotomized into high and low self-efficacy as described in Methods. Group differences were compared with logistic regression. Asterisks indicate P<0.05 for the post-hoc pair-wise comparisons.
Self-efficacy for Strength Training
A similar pattern was seen in self-efficacy for strength training. The omnibus test of group (no arthritis; arthritis only; arthritis plus diabetes), as it was associated with self-efficacy for strength training, was significant, indicating group differences in self-efficacy for strength training (P=0.01; Table 2). When compared to persons without arthritis, persons with arthritis reported poorer self-efficacy for strength training (68% versus 60% high self-efficacy respectively, pairwise P=0.18; adjusted OR for high self-efficacy 0.69 (0.39, 1.20)). Among those with the added burden of diabetes, average reported self-efficacy for strength training was significantly lower compared to the no arthritis group (44% versus 68% high self-efficacy respectively, pairwise P=0.02; adjusted OR for high self-efficacy 0.64 (0.44, 0.93)) and significantly lower compared to the arthritis alone group (pairwise P=0.66; adjusted OR for high self-efficacy 0.93 (0.69, 1.26)). The trend test indicated a significant decline in self-efficacy for strength training with the added co-morbidities of arthritis and arthritis plus diabetes (P=0.004).
Motivation for Endurance Training
For motivation for endurance training, the omnibus test of group (no arthritis; arthritis only; arthritis plus diabetes) was borderline significant, suggesting group differences in motivation for strength training (P=0.06; Table 2). In contrast to our expectations for reduced motivation in those with chronic conditions, persons with arthritis reported significantly higher motivation for endurance training than persons without arthritis (58% versus 42% high motivation respectively, pairwise P=0.02; adjusted OR for high motivation 1.85 (1.12, 3.33)). In persons with arthritis and diabetes, motivation for strength training was not significantly different than the no arthritis group (52% versus 42% high self-efficacy respectively, pairwise P=0.06; adjusted OR for high motivation 1.41 (0.99, 2.0)) but was significantly less than those with arthritis alone (58% high motivation, pairwise P=0.04; adjusted OR for high motivation 0 .76 (0.55, 0.98)). Similarly, there was no trend towards less motivation for endurance training with the added burdens of arthritis and diabetes (P=0.25).
Motivation for Strength Training
Contrary to our expectation, more than 60% of participants in all groups reported high motivation for strength training, and no significant differences in level of motivation for strength training were observed between groups; suggesting no significant effect of disease state on motivation for strength training (omnibus P=0.46). As with motivation for endurance training, there was no evidence for a trend towards less motivation for strength training with arthritis or the combination of arthritis and diabetes (P=0.46)
Physical Activity Amounts
While there were no significant differences between the three groups in minutes of endurance training (omnibus P=0.93), persons with both arthritis and diabetes reported significantly less strength training compared to individuals with only arthritis (12±30 versus 28±62 minutes per week respectively, P=0.03; see Table 2).
Discussion
The results of this study suggest a complex relationship between self-efficacy, motivation, physical activity, and arthritis in older adults. In a predominantly male Veteran population, we found reductions in self-efficacy for both endurance and strength training in persons with arthritis and further reductions in those with both arthritis and diabetes. In contrast and while preliminary studies of motivation indicate a decline in motivation for exercise with increasing age [22], our analyses demonstrated high levels of motivation for both endurance and strength training exercise. Moreover, these high levels of motivation were present even among those with arthritis only and those who suffer from both arthritis and diabetes.
Although previous investigations have focused on self-efficacy for exercise within subgroups including persons with arthritis[5, 23], at-risk for diabetes[24], and older individuals[25], to our knowledge, this study is the first to compare self-efficacy for specific types of physical activity, endurance and strength training, in persons with and without arthritis and comorbidities. The influence of self-efficacy expectations on behavior is magnified in times of great challenge, such as in the face of chronic and co-morbid conditions and during the initial stages of exercise adoption [26]. In this group of older, predominantly male Veterans, we observed that persons with arthritis alone had lower self-efficacy for endurance training than those without arthritis, and that persons with both arthritis and diabetes had further reductions in self-efficacy. Similarly, self-efficacy for strength training was lower in persons with arthritis alone and further reduced in those with both arthritis and diabetes. These findings suggest that while the presence of arthritis negatively influences an individual's confidence in their ability to participate in endurance and strength training activities, having a co-morbid condition of diabetes contributes to further decline.
While this further decline in self-efficacy is not surprising and might be expected with any co-morbidity that provides additional barriers to physical activity, the lower levels of self-efficacy for both endurance and strength training demonstrated among individuals with arthritis with and without diabetes are of concern to health care providers as low efficacy is significantly associated with decreased physical activity [27, 28]. Unlike the chronic diseases that these individuals live with which are relatively stable, self-efficacy is a modifiable construct that can be enhanced through experience, social and verbal persuasion, and education relative to the body's responses to exercise [12]. In order to be effective, exercise counseling by physicians should target components of self-efficacy and include 1) presenting the exercise plan as a formal prescription rather than a general plan [29, 30], 2) developing a specific exercise plan which includes recommendations for mode, frequency, duration, and intensity of exercise in the place of more general advice about exercise, and 3) following up with patients regarding progress and barriers.
Perhaps, more notable than the declines in self-efficacy was the maintenance of motivation observed for these groups. These findings sharply contradicted our hypothesis that older adults with arthritis and diabetes would exhibit lower motivation. Additionally, these results suggest that these individuals do not perpetuate inactivity and diseases related to inactivity because of a lack of interest in physical activity. Our results warrant recognition by health professionals by suggesting that older persons, especially older men, with arthritis recognize that exercise is important for their health and possess motivation to engage in active behavior. Since motivation for exercise has been identified as an important determinant of successful incorporation of regular physical activity into daily life[11], the finding of preserved motivation for physical activity in persons with arthritis is extremely promising.
While motivation was maintained overall, there were differential trends observed between motivation for endurance exercise and motivation for strength exercise. Although no significant group differences were reported between the three groups for strength training motivation, older adults with arthritis were significantly more motivated to participate in endurance training than were older adults without arthritis. At first glance, these results might appear antithetical; however this trend may reflect general practitioners' tendencies to advise patients with chronic conditions (such as arthritis and diabetes and as exhibited by poorer health quality of life, physical function and vitality in these groups) to exercise, whereas more healthy patients less commonly receive such advice [31-33]. Indeed, perhaps raising awareness about the benefits of exercise, specifically endurance exercise, has a motivating effect on patients. In contrast, the high levels of motivation for strength training across all three study groups might be attributable, in part, to the suspected low prevalence by which physicians counsel patients about the benefits of strength training. As a result, this equates to similar levels of information (or lack thereof) between healthy and at-risk patients concerning strength training and its associated health benefits [34, 35]. Given the purported success of provider counseling for enhancing motivation for endurance training [36], efforts to improve knowledge of current physical activity guidelines and the utility of individualized exercise programs among health care professionals appear warranted for strength training.
Consistent with a social cognitive perspective, our results suggest that being motivated, while necessary, is not sufficient to drive behavior. Indeed, social cognitive theory and theories of motivation posit that the effects of motivation on physical activity behavior are regulated by efficacy cognitions [7, 11]. That is, changes in physical activity are expected among individuals who are motivated to initiate exercise and who also have the efficacy to perform well and maintain the behavior in the face of barriers. The attrition rates among those initiating an exercise program (50%) [23] emphasize that motivation in and of itself is not the sole determinant of behavior. As this study is cross-sectional, we are unable to speak to the predictive validity of the proposed mediation model. Future studies which assess these patterns of relationships using longitudinal data are needed.
This report is not without limitations. Most importantly, Veterans Life was not specifically designed to determine the effect of diabetes on self-efficacy for physical activity in persons with arthritis, and a significant selection bias for individuals more likely to be motivated for exercise training is likely. However, such a selection bias should be similar for those with and without arthritis and would not account for the differences noted here between persons without arthritis, with arthritis, and with arthritis plus diabetes. Additionally, we did not assess arthritis severity in this investigation, and are unable to comment as to whether greater arthritis severity in those with diabetes might account for poorer self-efficacy for both types of exercise. Additional studies where both arthritis severity and self-efficacy for specific types of exercise might yield more insight into this potential contributor to poorer self-efficacy in persons with arthritis. Also, our findings are limited by the use of a predominantly male, Veteran population, and our reliance on self-report assessments of arthritis, diabetes, and physical activity are limitations that should be considered in future studies. We also recognize that our results would be strengthened in the presence of multiple-item measures of self-efficacy and motivation. Such measures are more apt to distinguish between individuals who differ in their perceived efficacy for higher or lower level demands. Finally, this investigation was designed to be exploratory, and as such is susceptible to possible type-I errors associated with multiple testing. Future confirmatory investigations in mixed-gender populations using more detailed assessments of exercise self-efficacy, objective measures of physical activity, and physician confirmation of arthritis and diabetes are needed.
In conclusion, this study underscores the importance of recognizing arthritis and diabetes as a common geriatric co-morbidity that significantly impacts management, in particular behavioral modifications. Our results suggest that patients are cognizant of the benefits of physical activity, as evidenced by the high levels of motivation. However, it appears that patients are not adequately armed with information and/or experience to instill feelings of confidence that they can, in fact, change from a sedentary lifestyle to an active lifestyle; resulting in a disconnect between cognitive motivation and behavior. In light of current recommendations for individuals with arthritis and/or diabetes to regularly engage in both endurance and strength training activities, future efforts to enhance efficacy expectations for these specific modes of exercise, and consequently increase activity behavior, are needed. Additionally, efforts to enhance motivation for exercise among patients without chronic conditions as a means of disease prevention appear warranted.
Acknowledgements
We would like to recognize the significant contributions of the rest of the Project Life research team. We are also grateful to the willing participation of the primary care physicians, geriatricians, and Veterans at the Durham VA Medical Center. This project was supported by NIH/NIAP30 AGO28716-01 and VA Rehabilitation and Research Development Service grant E3386R. Dr. Huffman was supported by the ACR-REF/ASP Junior Career Development Award in Geriatric Medicine funded via Atlantic Philanthropies, ACR-REF, John A. Hartford Foundation and ASP as well as NIH/NIAMS K23AR054904. The views expressed here are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs.
References
- 1.Centers for Disease Control and Prevention (CDC) Public health and aging: projected prevalence of self-reported arthritis or chronic joint symptoms among persons aged >65 years--United States, 2005-2030. MMWR Morb Mortal Wkly Rep. 2003 May 30;52(21):489–91. [PubMed] [Google Scholar]
- 2.Physical Activity Guidelines Advisory Committee . Physical Activity Guidelines Advisory Committee Report, 2008. US Department of Health and Human Services; Washington, DC: 2008. [DOI] [PubMed] [Google Scholar]
- 3.King H, Aubert RE, Herman WH. Global burden of diabetes, 1995-2025: prevalence, numerical estimates, and projections. Diabetes Care. 1998 Sep;21(9):1414–31. doi: 10.2337/diacare.21.9.1414. [DOI] [PubMed] [Google Scholar]
- 4.Trost SG, Owen N, Bauman AE, Sallis JF, Brown W. Correlates of adults' participation in physical activity: review and update. Med Sci Sports Exerc. 2002 Dec;34(12):1996–2001. doi: 10.1097/00005768-200212000-00020. [DOI] [PubMed] [Google Scholar]
- 5.Rejeski WJ, Ettinger WH, Jr., Martin K, Morgan T. Treating disability in knee osteoarthritis with exercise therapy: a central role for self-efficacy and pain. Arthritis Care Res. 1998 Apr;11(2):94–101. doi: 10.1002/art.1790110205. [DOI] [PubMed] [Google Scholar]
- 6.Maly MR, Costigan PA, Olney SJ. Self-efficacy mediates walking performance in older adults with knee osteoarthritis. J Gerontol A Biol Sci Med Sci. 2007 Oct;62(10):1142–6. doi: 10.1093/gerona/62.10.1142. [DOI] [PubMed] [Google Scholar]
- 7.de Jong Z, Munneke M, Jansen LM, Ronday K, van Schaardenburg DJ, Brand R, et al. Differences between participants and nonparticipants in an exercise trial for adults with rheumatoid arthritis. Arthritis Rheum. 2004 Aug 15;51(4):593–600. doi: 10.1002/art.20531. [DOI] [PubMed] [Google Scholar]
- 8.Bandura A. Health promotion by social cognitive means. Health Educ Behav. 2004 Apr;31(2):143–64. doi: 10.1177/1090198104263660. [DOI] [PubMed] [Google Scholar]
- 9.Bandura A. Self-efficacy: The exercise of control. W.H. Freeman and Company; New York: 1997. [Google Scholar]
- 10.Roberts GC. Understanding the dynamics of motivation in physical activity: The influence of achievement goals on motivational processes. In: Roberts GC, editor. Advances in motivation in sport and exercise. Human Kinetics; Champaign, IL: 2001. pp. 1–50. [Google Scholar]
- 11.Veenhof C, van Hasselt TJ, Koke AJ, Dekker J, Bijlsma JW, van den Ende CH. Active involvement and long-term goals influence long-term adherence to behavioural graded activity in patients with osteoarthritis: a qualitative study. Aust J Physiother. 2006;52(4):273–8. doi: 10.1016/s0004-9514(06)70007-1. [DOI] [PubMed] [Google Scholar]
- 12.Bandura A. Social foundations of thought and action: A social cognitive theory. Prentice-Hall; Englewood Cliffs, NJ: 1986. [Google Scholar]
- 13.US Department of Health and Human Services Physical Activity Guidelines for Americans. 2008 www.health.gov/PAGuidelines; 2008.
- 14.Dacey M, Baltzell A, Zaichkowsky L. Older adults' intrinsic and extrinsic motivation toward physical activity. Am J Health Behav. 2008 Nov-Dec;32(6):570–82. doi: 10.5555/ajhb.2008.32.6.570. [DOI] [PubMed] [Google Scholar]
- 15.Morey MC, Peterson MJ, Pieper CF, Sloane R, Crowley GM, Cowper PA, et al. The Veterans Learning to Improve Fitness and Function in Elders Study: a randomized trial of primary care-based physical activity counseling for older men. J Am Geriatr Soc. 2009 Jul;57(7):1166–74. doi: 10.1111/j.1532-5415.2009.02301.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ware JE, Jr., Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992 Jun;30(6):473–83. [PubMed] [Google Scholar]
- 17.Fillenbaum G. Multidimensional Functional Assessment of Older Adults. Lawrence Erlbaum Associates; Hillsdale, NJ: 1988. [Google Scholar]
- 18.US Department of Health and Human Services . Physical activity and health: A report of the Surgeon General. In: National Center for Chronic Disease Prevention and Health Promotion, editor. US Department of Health and Human Services Centers for Disease Control and Prevention. Atlanta, GA: 1996. [Google Scholar]
- 19.McAuley E. Self-efficacy and the maintenance of exercise participation in older adults. J Behav Med. 1993 Feb;16(1):103–13. doi: 10.1007/BF00844757. [DOI] [PubMed] [Google Scholar]
- 20.Stewart AL, Mills KM, Sepsis PG, King AC, McLellan BY, Roitz K, et al. Evaluation of CHAMPS, a physical activity promotion program for older adults. Ann Behav Med. 1997 Fall;19(4):353–61. doi: 10.1007/BF02895154. [DOI] [PubMed] [Google Scholar]
- 21.Morey MC, Peterson MJ, Pieper CF, Sloane R, Crowley GM, Cowper P, et al. Project LIFE-Learning to Improve Fitness and Function in Elders: Methods, design, and baseline characteristics of randomized trial. J Rehabil Res Dev. 2008;45(1):31–42. doi: 10.1682/jrrd.2007.03.0044. [DOI] [PubMed] [Google Scholar]
- 22.Frederick-Recascino CM. Self-determination theory and participation motivation research in the sport and exercise domain. In: Deci EL, Ryan RM, editors. Handbook of self-determination research. University of Rochester Press; Rochester, NY: 2002. pp. 277–94. [Google Scholar]
- 23.Hughes SL, Seymour RB, Campbell RT, Huber G, Pollak N, Sharma L, et al. Long-term impact of Fit and Strong! on older adults with osteoarthritis. Gerontologist. 2006 Dec;46(6):801–14. doi: 10.1093/geront/46.6.801. [DOI] [PubMed] [Google Scholar]
- 24.Delahanty LM, Conroy MB, Nathan DM. Psychological predictors of physical activity in the diabetes prevention program. J Am Diet Assoc. 2006 May;106(5):698–705. doi: 10.1016/j.jada.2006.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harris TJ, Owen CG, Victor CR, Adams R, Cook DG. What factors are associated with physical activity in older people, assessed objectively by accelerometry? Br J Sports Med. 2008 May 16; doi: 10.1136/bjsm.2008.048033. [DOI] [PubMed] [Google Scholar]
- 26.McAuley E, Blissmer B. Self-efficacy determinants and consequences of physical activity. Exerc Sport Sci Rev. 2000 Apr;28(2):85–8. [PubMed] [Google Scholar]
- 27.McAuley E, Morris KS, Motl RW, Hu L, Konopack JF, Elavsky S. Long-term follow-up of physical activity behavior in older adults. Health Psychol. 2007 May;26(3):375–80. doi: 10.1037/0278-6133.26.3.375. [DOI] [PubMed] [Google Scholar]
- 28.McAuley E, Hall KS, Motl RW, White SM, Wojcicki TR, Hu L, et al. Trajectory of Declines in Physical Activity in Community-Dwelling Older Women: Social Cognitive Influences. J Gerontol B Psychol Sci Soc Sci. 2009 Jun 15; doi: 10.1093/geronb/gbp049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fontaine KR, Bartlett SJ, Heo M. Are health care professionals advising adults with arthritis to become more physically active? Arthritis Rheum. 2005 Apr 15;53(2):279–83. doi: 10.1002/art.21073. [DOI] [PubMed] [Google Scholar]
- 30.Weidinger KA, Lovegreen SL, Elliott MB, Hagood L, Haire-Joshu D, McGill JB, et al. How to make exercise counseling more effective: lessons from rural America. J Fam Pract. 2008 Jun;57(6):394–402. [PubMed] [Google Scholar]
- 31.Eakin E, Brown W, Schofield G, Mummery K, Reeves M. General practitioner advice on physical activity--who gets it? Am J Health Promot. 2007 Mar-Apr;21(4):225–8. doi: 10.4278/0890-1171-21.4.225. [DOI] [PubMed] [Google Scholar]
- 32.Sinclair J, Lawson B, Burge F. Which patients receive advice on diet and exercise? Do certain characteristics affect whether they receive such advice? Can Fam Physician. 2008 Mar;54(3):404–12. [PMC free article] [PubMed] [Google Scholar]
- 33.Wee CC, McCarthy EP, Davis RB, Phillips RS. Physician counseling about exercise. JAMA. 1999 Oct 27;282(16):1583–8. doi: 10.1001/jama.282.16.1583. [DOI] [PubMed] [Google Scholar]
- 34.Eakin E, Brown W, Schofield G, Mummery K, Reeves M. General practitioner advice on physical activity--who gets it? American Journal of Health Promotion. 2007;21(4):225–8. doi: 10.4278/0890-1171-21.4.225. [DOI] [PubMed] [Google Scholar]
- 35.Sinclair J, Lawson B, Burge F. Which patients receive advice on diet and exercise? Do certain characteristics affect whether they receive such advice? Canadian Family Physician. 2008;54(3):404–12. [PMC free article] [PubMed] [Google Scholar]
- 36.Weidinger KA, Lovegreen SL, Elliott MB, Hagood L, Haire-Joshu D, McGill JB, et al. How to make exercise counseling more effective: lessons from rural America. The Journal of Family Practice. 2008;57(6):394–402. [PubMed] [Google Scholar]