Abstract
Inflammatory bowel disease (IBD) and colorectal cancer (CRC) are common health problems worldwide. Tumor necrosis factor (TNF) is a type of cytokine that induces inflammation and inhibits tumorigenesis. Several studies have assessed the relationship between the polymorphism of TNF-α-308 G>A and the susceptibility to IBD and CRC; however, the results have been controversial. In addition, the hypothesis whether the increased risk of CRC in IBD patients could be partly ascribed to the polymorphism of TNF-α-308 G>A was unclear. Therefore, we conducted this meta-analysis to confirm these associations. Pooled odd ratios (ORs) and 95% confidence intervals (95% CIs) were calculated on the basis of data from 14, 18, and 7 studies from a total of 27 studies for the associations between the polymorphism of TNF-α-308 G>A and ulcerative colitis, Crohn's disease (CD) and CRC. In Europeans, the AA genotype increased the risk of ulcerative colitis (UC) (OR, 2.041; 95% CI, 1.261–3.301) and CD (OR, 1.730; 95% CI, 1.168–2.564) significantly, without obvious heterogeneity and publication bias. Meanwhile, the GA genotype increased the risk of UC in Asians (OR, 2.360; 95% CI, 1.269–4.390) significantly. However, no significant association was observed for CRC in any ethnic population. The results of this meta-analysis suggested that the polymorphism of TNF-α-308 G>A participates in modifying the susceptibility to UC and CD in Europeans and Asians. The increased risk of CRC in IBD patients should be clarified as the combined effects of polymorphisms in TNF-α and other cytokines, and the interaction with environmental factors, in future studies.
Keywords: tumor necrosis factor-α, Crohn's disease, ulcerative colitis, inflammatory bowel disease, colorectal cancer
INTRODUCTION
Inflammatory bowel disease (IBD) refers to two chronic diseases caused by an inflammatory condition of the intestine: ulcerative colitis (UC) and Crohn's disease (CD). It has been reported that age-standardized incidence rates were 1.85 per 100 000 person-years for UC and 0.70 per 100 000 person-years for CD worldwide.1 Several observational studies suggested that patients with long-standing UC or CD have an increased risk of developing colorectal cancer (CRC).2, 3 A meta-analysis conducted by Eaden et al4 also pointed out that the susceptibility to CRC for any UC patient was increased significantly with an odds ratio(OR) of 3.7 and 95% confidence interval (95% CI) of 3.2–4.2%.
Until now, the pathogenic mechanism of IBD was uncertain. Extensive studies have suggested that IBD is a multifactorial disease that has arisen from both environmental risk factors and genetic susceptibility.5, 6 Previous genetic association studies mainly focused on the relationship between IBD and the genetic variants in HLA genes instead of on genes involved in immune regulation and inflammatory response. Recent identification of polymorphisms in the tumor necrosis factor-α (TNF-α) gene gave suggestions on understanding the genetic predisposition of these two diseases.7, 8
The human TNF-α gene was first cloned in 1985. It maps to chromosome 6p21.3 and encodes a multifunctional proinflammatory cytokine that belongs to the TNF superfamily. TNF-α has several actions, such as stimulating the acute phase response, leading to an increase in the level of C-reactive protein, and producing IL-1 oxidant and inflammatory lipid prostaglandin E2. Large amounts of TNF-α are released in response to lipopolysaccharides, other bacterial products, and interleukin-1 (IL-1). A high concentration of TNF-α could induce shock-like symptoms; a low concentration of TNF-α with long-term exposure could result in cachexia and wasting syndrome (http://en.wikipedia.org/wiki/Tumor_necrosis_factor-alpha).
Studies have highlighted that small differences at cytokine levels as a result of genetic variants may have an important effect on inflammatory response and may influence the pathophysiology. Two SNPs located at nucleotides −238 and −308 have been extensively researched on the susceptibility to a range of autoimmune disorders, including rheumatoid arthritis,9 exfoliation glaucoma,10 and many kinds of cancers.11, 12, 13 The hypothesis that TNF-α-308 G>A may have important roles in the development of UC, CD, and CRC has been debated. The results of previous association studies were inconsistent. Moreover, whether the increased risk of CRC in IBD patients could be partly ascribed to the polymorphism of TNF-α remains unclear. Therefore, to further explore the relationship between the promoter −308 G>A polymorphism in TNF-α and the risk of UC, CD, and CRC, we conducted a systematical review and a meta-analysis.
Materials and methods
Search strategy
PubMed, Cochrane and Embase were retrieved for the association studies focused on the relationship between the polymorphism of TNF-α-308 G>A and the risk of IBD and CRC primarily. The key words and subject terms used were as follows: ‘colorectal' and ‘cancer and/or carcinoma', ‘Crohn's disease' or ‘CD', ‘ulcerative colitis' or ‘UC', ‘TNF-α-308 G>A', ‘polymorphism'. The relevant articles and publications on the same topic in reference lists of the reviewed articles were also retrieved according to the inclusion criteria. Only the full-text papers published in English were included.
Criteria of inclusion and exclusion
Any human association study, regardless of sample size, was eligible (1) if it pertained to1 the relationship between the polymorphism of TNF-α-308 G>A and the risk of IBD or CRC; (2) the genetic information of included studies was from unrelated populations; (3) controls of included studies were from a healthy population or were subjects without diseases related to IBD or CRC; (4) the genotype distribution of the control population was in Hardy–Weinberg equilibrium; and (5) for the articles with overlap data of the same population resource, only the latest and largest report was included.
Data extraction and quality assessment
The information extracted from each study includes the following: the name of the authors, the year of publication, the ethnicity of subjects, the study design, demography characteristics of cases and controls, genotype frequency, matching condition, OR, and the adjustment of confounders. Baseline information and data were extracted by two reviewers independently using the same standard. In addition, the quality of studies was assessed by these two reviewers adopting the quality assessment score method, which was based on traditional epidemiological considerations and genetic issues.14 Another reviewer adjudicated the differences between them.
Statistical analysis
Variant allele frequencies were compared between cases and controls in different ethnic populations. Compared with wild-type homozygote GG, ORs, and 95% CIs for the heterozygote GA and the variant homozygote AA were calculated, respectively. The relationship between the polymorphism of TNF-α-308 G>A and the risk of UC, CD, and CRC was further analyzed by categorizing into different ethnic populations. The associations with p<0.05 were considered as being statistically significant. Homogeneity among the included studies was assessed using the Cochrane Q value and I2. When obvious heterogeneity was detected, a random-effects model was used; otherwise, a fixed-effects model was adopted. Publication bias was investigated using graphical evaluation of funnel plots. In addition, the possible presence of publication bias was assessed by rank correlation and linear regression. If significant publication bias was detected, ORs and 95% CIs would be adjusted by trim and fill.
Analyses were performed with the software SPSS (13.015, Permanent licence, Stats Data Mining Co., Ltd, Beijing, China). ORs and 95% CIs were generated by CMA (Comprehensive Meta Analysis V2(2005) Biostat, Inc., Englewood, NJ, USA).
Results
Main characteristics of included studies
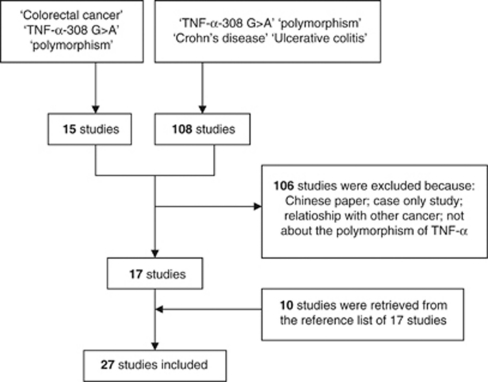
Details for literature search were shown in Figure 1. Finally, 27 studies were included in this meta-analysis. Six of them were on Asians, and the others were conducted on a European population. In all, 25 of the 27 studies were population-based case–control studies, except for two hospital-based case–control studies. Data were obtained from 14, 18, and 7 studies for the association between the polymorphism of TNF-α-308 G>A and UC, CD, and CRC, respectively. Genotype and variant allele frequencies were presented in Table 1 . In these included studies, IBD patients were diagnosed with conventional pathological examination or selected from definite cases in the hospital. CRC patients were confirmed by histological diagnosis. All cases were matched with unrelated healthy individuals or volunteers recruited from the same ethnic population. In these 27 studies, PCR-RFLP or PCR-SSCP was chosen for genotyping in 18 studies, and real-time PCR assays with (TaqMan, Life Technologies Corporation, Carlsbad, CA, USA) primers and probes were used in the others.
Figure 1.
Flow diagram of the literature search and study selection protocol.
Table 1. Information of the included studies that investigated the relationship between TNF-α-308G>A and the risk of inflammatory bowel disease and colorectal cancer.
| First Author (Year)a | Country (Racial descent)b | Study Design | Case (−308 G>A)c | Control (−308 G>A)c | Variant allele frequency |
|---|---|---|---|---|---|
| Bouma et al14 | Netherlands (European) | Pop c/c | CD (25/10/0) | (65/29/4) | 0.189 |
| Sashio et al15 | Japan (Asian) | Pop c/c | UC (89/16/1) | (107/4/0) | 0.018 |
| CD (119/4/1) | |||||
| Martin et al16 | Germany (European) | Pop c/c | UC (74/19/0) | (87/32/0) | 0.134 |
| CD (78/17/0) | |||||
| Gonzalez et al17 | Spain (European) | Pop c/c | CD (41/9/0) | (76/21/3) | 0.135 |
| Vatay et al18 | Hungary (European) | Pop c/c | UC (41/9/0) | (93/41/4) | 0.178 |
| CD (63/11/0) | |||||
| Balding et al19 | Ireland (European) | Pop c/c | UC (78/23/7) | (233/140/16) | 0.221 |
| CD (37/26/1) | |||||
| Yamamoto-Furusho K et al20 | Mexico (European) | Pop c/c | UC (61/19/0) | (96/3/0) | 0.015 |
| Fowler et al21 | Australia (European) | Pop c/c | CD (198/91/15) | (150/61/6) | 0.168 |
| Cantor et al22 | Canada (European) | Pop c/c | UC (35/18/2) | (63/27/2) | 0.168 |
| CD (103/30/5) | |||||
| Song et al23 | China (Asian) | Pop c/c | UC (86/16/1) | (211/9/0) | 0.020 |
| CD (26/2/0) | |||||
| Ferreira et al24 | Portugal (European) | Pop c/c | CD (171/55/9) | (684/229/12) | 0.136 |
| Zipperlen et al25 | Canada (European) | Pop c/c | CD (85/41/2) | (67/33/3) | 0.189 |
| Castro-Santos et al26 | Spain (European) | Pop c/c | CD (108/31/7) | (265/71/7) | 0.124 |
| UC (70/26/3) | |||||
| Yang et al27 | Korea (Asian) | Pop c/c | CD (255/29/4) | (342/54/3) | 0.075 |
| Sykora et al28 | Zech (European) | Pop c/c | CD (38/8/0) | (71/10/1) | 0.073 |
| UC (22/12/2) | |||||
| Qian et al29 | Chinese (Asian) | Pop c/c | UC (79/30/1) | (242/48/2) | 0.089 |
| Celik et al30 | Turkey (Asian) | Pop c/c | CD (56/14/0) | (86/18/1) | 0.095 |
| UC (96/23/1) | |||||
| Cucchiara et al31 | Italy (European) | Pop c/c | UC (140/28/5) | (308/37/2) | 0.059 |
| CD (137/46/5) | |||||
| Mittal et al32 | India (European)d | Pop c/c | UC (9/79/4) | (10/147/7) | 0.490 |
| CD (3/17/2) | |||||
| Ferguson et al33 | NewZealand (European) | Pop c/c | UC (258/122/23) | (282/123/10) | 0.172 |
| CD (260/112/16) | |||||
| Jang et al34 | Korea (Asian) | Pop c/c | CRC (24/3/0) | (85/7/0) | 0.038 |
| Landi et al35 | Spain (European) | Hosp c/c | CRC (278/80/5) | (234/76/10) | 0.150 |
| Macarthur et al36 | Scotland (European) | Pop c/c | CRC (157/74/15) | (224/145/20) | 0.238 |
| Gunter et al37 | USA (European) | Pop c/c | CRC (146/59/12) | (139/57/6) | 0.170 |
| Theodoropoulos et al38 | Greece (European) | Pop c/c | CRC (152/56/14) | (146/44/10) | 0.160 |
| Toth et al39 | Hungary (European) | Pop c/c | CRC (132/48/3) | (111/30/0) | 0.106 |
| Garrity-Park et al40 | Ireland (European) | Hosp c/c | CRC (52/49/13) | (92/20/2) | 0.105 |
Abbreviations: CD, Crohn's disease; UC, Ulcerative colitis; CRC, Colorectal cancer.
Reference number.
The world population falls into three basic racial groups: Mongoloid (such as Asian), Negroid (African), and European (Caucasian or White).
Wild-type homozygote/heterozygote/variant homozygote.
It was reported that most inhabitants' ancestor of Indians are White. Although, U.S. population is highly heterogeneous, more than 70% residents are White. Thus, the population of these two countries were categorized as European.
Quantitative synthesis
Ulcerative colitis
Pooled ORs and 95% CIs were generated on the basis of data from 10 studies on a European population and 4 from an Asian population. No statistically significant differences in variant allele frequencies between cases and controls were found in either Europeans (P=0.689) or Asians (P=0.097) (Table 2). However, significant association was found for GA vs GG in Asians (OR, 2.360; 95% CI, 1.269–4.390). The pooled OR and 95% CI for AA vs GG in Europeans were 1.836 (1.160–2.904) with slight heterogeneity (I2=7.151%) and nonsignificant publication bias (P=1.000 for Rank correlation; P=0.715 for linear regression). In addition, according to the results of a dominant genetic model, the genotype GA+AA vs GG was still significant in Asians (OR, 2.427; 95% CI, 1.260–4.677; I2=67.373%) and in the total analysis (OR, 1.474; 95% CI, 1.028–2.112; I2=78.143%); however, heterogeneity was obvious.
Table 2. Comparisons of variant allele frequencies between cases and controls in two ethnic populations.
| Allele frequency (mean±SD) | |||||
|---|---|---|---|---|---|
| Population | Disease | NO. Data (case/control) | Case | Control | P-valuea |
| Europeans | Ulcerative colitis | 10 (1983/3532) | 0.186±0.111 | 0.164±0.131 | 0.689 |
| Crohn's disease | 14 (1119/2188) | 0.167±0.100 | 0.174±0.101 | 0.859b | |
| Colorectal cancer | 6 (1345/1366) | 0.199±0.071 | 0.155±0.049 | 0.245c | |
| Total | 30 | 0.179±0.097 | 0.169±0.102 | 0.616 | |
| Asians | Ulcerative colitis | 4 (440/835) | 0.105±0.028 | 0.056±0.042 | 0.097 |
| Crohn's disease | 4 (509/728) | 0.056±0.338 | 0.052±0.390 | 0.887 | |
| Colorectal cancer | 1 (27/92) | 0.056 | 0.038 | ||
| Total | 9 | 0.078±0.037 | 0.052±0.036 | 0.153 | |
Test of difference between cases and controls.
Compared with the controls of Asian Crohn's disease, P-value=0.047.
Compared with the controls of Asian Colorectal cancer, γ-value=0.002.
Crohn's disease
There were no statistically significant differences of variant allele frequencies between cases and controls in both Europeans and Asians. Compared with Asian controls (mean±SD, 0.052±0.390), European controls carried a higher frequency (mean±SD, 0.174±0.101; P=0.047) of the −308A allele (Table 2). As shown in Table 3, a statistically significant association between genotype AA and the risk of CD was observed, especially in Europeans (OR, 1.693; 95% CI, 1.151–2.492, adjusted by the trim and fill method because of significant publication bias: P=0.024 for rank correlation; P=0.005 for linear regression) with slight heterogeneity (I2=12.467%). The increased risk (OR, 1.683; 95% CI, 1.163–2.434) in the total analysis was mainly generated by the contribution of the European population. There were no significant associations for the comparisons of GA vs GG and GA+AA vs GG in these two ethnic populations.
Table 3. Pooled analysis for the associations between the polymorphism of TNF-α-308G>A and the risk of inflammatory bowel disease and colorectal cancer.
| Data | Test of association | Test of heterogeneity | Test of publication (P-value) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Disease | Comparison | Ethnicity | NO. | OR (95% CI) | P-value (Z test) | χ2 | I2 (%) | P-value | Rank correlation | Linear regression |
| Ulcerative colitis | GA vs GG | Europeans | 10 | 1.157 (0.769–1.742)a | 0.485 | 38.150 | 76.409 | 0.000 | 0.211 | 0.487 |
| Asians | 4 | 2.360 (1.269–4.390)a | 0.007 | 8.035 | 62.663 | 0.045 | 0.734 | 0.338 | ||
| Total | 14 | 1.420 (0.986–2.045)a | 0.060 | 57.437 | 77.366 | 0.000 | 0.063 | 0.158 | ||
| AA vs GG | Europeans | 8 | 1.836 (1.160–2.904) | 0.017 | 7.539 | 7.151 | 0.375 | 1.000 | 0.715 | |
| Asians | 4 | 2.142 (0.517–8.884) | 0.294 | 1.115 | 0.000 | 0.773 | 0.734 | 0.281 | ||
| Total | 14 | 1.862 (1.204–2.882) | 0.005 | 8.696 | 0.000 | 0.650 | 0.945 | 0.957 | ||
| GA+AA vs GG | Europeans | 10 | 1.213 (0.808–1.821)a | 0.351 | 40.559 | 77.810 | 0.000 | 0.371 | 0.597 | |
| Asians | 4 | 2.427 (1.260–4.677)a | 0.008 | 9.195 | 67.373 | 0.027 | 0.734 | 0.318 | ||
| Total | 14 | 1.474 (1.028–2.112)a | 0.035 | 59.478 | 78.143 | 0.000 | 0.080 | 0.205 | ||
| Crohn's disease | GA vs GG | Europeans | 14 | 0.975 (0.772–1.231)a | 0.832 | 31.324 | 58.498 | 0.003 | 0.155 | 0.295 |
| Asians | 4 | 0.872 (0.596–1.275) | 0.479 | 2.052 | 0.000 | 0.562 | 0.308 | 0.244 | ||
| Total | 18 | 1.003 (0.881–1.142) | 0.967 | 33.967 | 49.952 | 0.008 | 0.705 | 0.485 | ||
| AA vs GG | Europeans | 14 | 1.693 (1.151–2.492)b | 0.008 | 13.709 | 12.467 | 0.320 | 0.024 | 0.005 | |
| Asians | 4 | 1.574 (0.448–5.521) | 0.479 | 0.606 | 0.000 | 0.738 | 1.000 | 0.745 | ||
| Total | 18 | 1.683 (1.163–2.434)b | 0.006 | 14.328 | 0.000 | 0.501 | 0.043 | 0.005 | ||
| GA+AA vs GG | Europeans | 14 | 0.985 (0.773–1.254)a | 0.901 | 35.759 | 63.646 | 0.001 | 0.063 | 0.158 | |
| Asians | 4 | 0.910 (0.631–1.315) | 0.617 | 1.580 | 0.000 | 0.664 | 0.308 | 0.093 | ||
| Total | 18 | 0.992 (0.806–1.221)a | 0.942 | 37.942 | 55.195 | 0.003 | 0.495 | 0.308 | ||
| Colorectal cancer | GA vs GG | Europeans | 6 | 0.920 (0.576–1.467)a | 0.328 | 26.486 | 81.122 | 0.000 | 0.009 | 0.007 |
| Asians | 1 | 1.518 (0.365–6.320) | 0.566 | — | — | — | — | — | ||
| Total | 7 | 1.241 (0.838–1.838)a | 0.281 | 26.714 | 77.540 | 0.000 | 0.036 | 0.157 | ||
| AA vs GG | Europeans | 6 | 1.064 (0.458–2.472)a,b | 0.231 | 13.786 | 63.731 | 0.017 | 0.260 | 0.286 | |
| Asians | 1 | — | — | — | — | — | — | — | ||
| Total | 6 | 1.064 (0.458–2.472)a,b | 0.231 | 13.786 | 63.731 | 0.017 | 0.260 | 0.286 | ||
| GA+AA vs GG | Europeans | 6 | 1.298 (0.834–2.020)a,b | 0.247 | 33.274 | 84.973 | 0.000 | 0.009 | 0.007 | |
| Asians | 1 | 1.518 (0.365–6.320) | 0.566 | — | — | — | — | — | ||
| Total | 7 | 0.940 (0.592–1.492)a,b | 0.210 | 33.461 | 82.069 | 0.000 | 0.036 | 0.171 | ||
Abbreviations: OR, Odds Ratio; CI, Confidence Interval.
Random effect model.
Trim and fill.
CRC
No significant differences were detected for the contrasts of variant allele frequencies between cases and controls in Europeans and Asians. As only one study carried out in Asians was included, one-sample T-test was used to compare the differences of allele frequencies between European and Asian controls (P=0.002). No significant association was observed between the polymorphism of TNF-α-308 G>A and the risk of CRC. As the limited included studies had different genotyping methods, study design, and various ethnic populations, obvious heterogeneity (63.731–84.973%) and publication bias were observed and could hardly be avoided. Most ORs and 95% CIs adjusted by the trim and fill method were listed in Table 3.
Test of heterogeneity and publication bias
There was significant heterogeneity for the comparison of GA with GG in the total analysis of UC (χ2=57.437, I2=77.366%, df=13, P=0.000). According to the results of stratification analysis, we explored the source of heterogeneity from the subgroup analyses of both Europeans (χ2=38.150, I2=76.409%, df=9, P=0.000) and Asians (χ2=8.035, I2=62.663%, df=3, P=0.045). For the meta-analysis of CD, heterogeneity was found for GA vs GG in the European population (χ2=31.324, I2=58.498%, df=13, P=0.003). Extensive heterogeneity among the included studies for CRC was observed (Table 3).
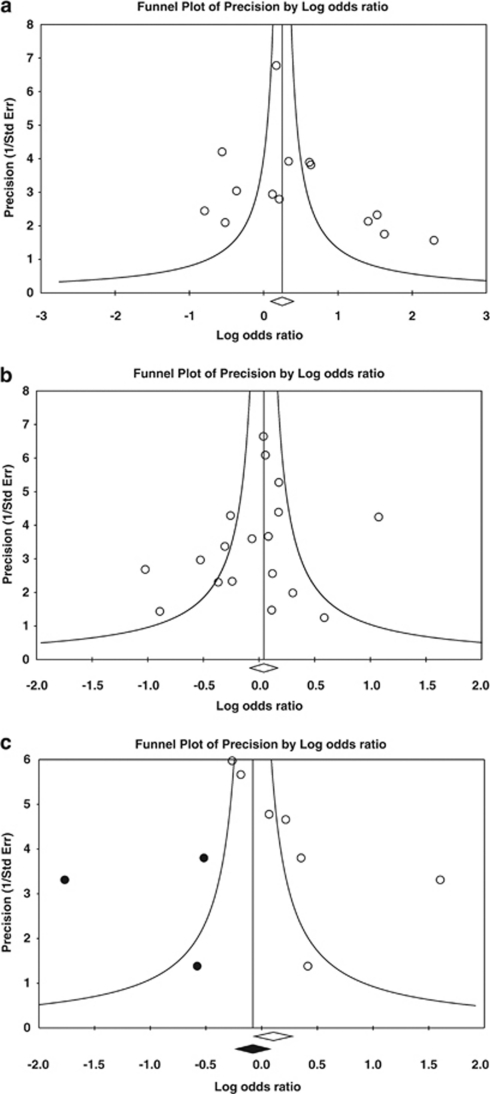
As shown in Figures 2a and b, the graphics of funnel plot revealed that no publication bias in the dominant genetic model was observed in the total analysis of UC and CD. Only the funnel plot of CRC was asymmetrical and adjusted by the trim and fill method (Figure 2c). Rank correlation and the linear regression approach have also been used to evaluate potential publication bias. Detailed results are listed in Table 3.
Figure 2.
Funnel plots for the total analysis of ulcerative colitis, Crohn's disease and colorectal cancer in a dominant genetic model. (a) GA+AA vs GG for ulcerative colitis. (b) GA+AA vs GG for Crohn's disease. (c) GA+AA vs GG for colorectal cancer.
Discussion
IBD comprises two chronic inflammatory disorders of the gastrointestinal tract: UC and CD. The IBD incidence rate has increased gradually over the past decade. It has been the most common health problem worldwide, particularly in developed countries. Moreover, studies have revealed that atypical hyperplasia and canceration are more likely to occur in the mucous membrane of UC patients.15 The susceptibility of CD patients to CRC would be 440-fold that of the common population and the onset age would be 10 years earlier.41
IBD was considered as resulting from the imbalance between luminal bacterial flora and the immune system.16 Regular use of non-steroidal anti-inflammatory drugs (NSAID) could reduce the incidence of IBD and CRC. Thus, inflammation may be important in the development of IBD and CRC. Recent evidence has also suggested the genetic susceptibility of IBD. Polymorphisms in inflammatory genes have been researched for the association with IBD and CRC; however, the results are inconsistent. Whether the polymorphism of TNF-α-308 G>A is directly involved in modifying cytokine production and the pathophysiology of IBD and CRC, or serves merely as a marker that is in linkage with genetic predisposition, remains unclear. Meanwhile, we conducted this systematic review and a meta-analysis to further explore the relationship between the polymorphism of TNF-α-308 G>A and the risk of IBD and CRC, to give an explanation for the genetic correlation between IBD and CRC.
Balding et al17 found that the TNF-α-308 A allele was less frequent in both UC and CD patients compared with healthy controls. Ferguson et al18 found that the control group contained a lower frequency of the TNF-α-308 A allele compared with UC and CD patients. In this meta-analysis, it appeared that there were no significant differences between cases and controls in IBD and CRC (Table 2).
According to human ethnicity, populations of the included studies were categorized into three subgroups: European (Caucasian or White), Mongoloid (Asian), and Negroid (African). No study conducted in Africans was included in this meta-analysis. By calculating the variant allele frequencies and comparing the differences between Europeans and Asians, significant differences were detected for the comparisons of controls in CD and CRC. However, as there were relatively few studies for the pooled analysis of CRC in Europeans and Asians, the detection of significant difference was not stable. In addition, no significant associations were found between the polymorphisms of TNF-α-308 and the risk of CRC in these two ethnic populations. We could not assure that the different susceptibility to CRC between Europeans and Asians was caused by the different carriage of the −308 A allele.
Susceptibility to IBD and CRC may be provoked by a complex signaling network of cytokines. It has been pointed out that the increased TNF-α secretion could lead to the initiation of stimulating IL-1A and IL-6. TNF-α and IL-1A stimulate each other to induce the increased epithelial antigen uptake in the ileum of CD patients and T-helper A cytokines, which is important for the onset and progression of IBD.19 Most studies included in this meta-analysis have also assessed the effect of polymorphisms in IL-6, IL-1, IL-10 on the risk of IBD and CRC. However, not all these studies explored the same genetic variations; we could not carry out a meta-analysis by combining the interaction of polymorphisms in TNF-α and other cytokines.
Rutter et al20 analyzed a colonoscopy surveillance database and indicated that inflammation has an important role in the development of CRC. However, as the patients have different genetic predisposition, not all patients exposed to prolonged inflammation would develop CRC. Several studies have suggested a positive association between the polymorphisms in cytokines and the risk of CRC.38, 39 In this meta-analysis, we analyzed the relationship between the polymorphism of TNF-α-308 G>A and the susceptibility to CRC. However, no significant association was observed. Thus, prospective studies with a big sample size and combination analysis of other cytokines and environmental factors are requested.
Although, by combining the data of individual studies, we have increased the statistical power to detect the slight association, there are still some limitations. Bouma et al42 pointed out that UC is a disease with multiple divergent clinical courses; failure to note the heterogeneity in selection of patients may limit the power to identify genetic associations. Not all the included studies described the details about the selection criteria of UC cases; we could not assess the heterogeneity among the UC cases of different studies. Second, although the random effects model, trim and fill method have been used in this meta-analysis, significant heterogeneity and publication bias within some subgroup analyses lead the failure to confirm marginal associations.
With these limitations in mind, our results are still statistically credible. The results of this meta-analysis suggest that the polymorphism of TNF-α-308 G>A participates in modifying the susceptibility to IBD in both Europeans and Asians, although not to a highly significant degree. No significant association was observed for CRC. In view of IBD and CRC being multifactorial diseases, the polymorphisms of cytokines cannot be seen as the unique crucial factor of pathopoiesis. Future studies are expected to give further etiological explanation for IBD and CRC by considering all relevant interactions.
Acknowledgments
This study was supported by grants from the National Natural foundation (30972539) and the Graduate Foundation, supported by the Scientific Research of Heilongjiang Province (YJSCX2009-224HLJ).
The authors declare no conflict of interest.
References
- Edwards CN, Griffith SG, Hennis AJ, Hambleton IR. Inflammatory bowel disease: incidence, prevalence, and disease characteristics in Barbados, West Indies. Inflamm Bowel Dis. 2008;14:1419–1424. doi: 10.1002/ibd.20495. [DOI] [PubMed] [Google Scholar]
- Kraus S, Arber N. Inflammation and colorectal cancer. Curr Opin Pharmacol. 2009;9:405–410. doi: 10.1016/j.coph.2009.06.006. [DOI] [PubMed] [Google Scholar]
- Triantafillidis JK, Nasioulas G, Kosmidis PA. Colorectal cancer and inflammatory bowel disease: epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 2009;29:2727–2737. [PubMed] [Google Scholar]
- Sun X-w, Wu S-l, Lin Y-j, Bo W, Han H-l, Dai X-d. Trend of morbidity and mortality of colorectal carcinom a in Nangang District of Harbin from 1992 to 2001. World Chin J Digestol. 2004;12:2302–2306. [Google Scholar]
- Bernstein CN, Rawsthorne P, Cheang M, Blanchard JF. A population-based case control study of potential risk factors for IBD. Am J Gastroenterol. 2006;101:993–1002. doi: 10.1111/j.1572-0241.2006.00381.x. [DOI] [PubMed] [Google Scholar]
- El-Tawil AM. A population-based case-control study of potential risk factors for IBD. Am J Gastroenterol. 2009;104:1064. doi: 10.1038/ajg.2008.165. [DOI] [PubMed] [Google Scholar]
- Kim TH, Kim BG, Shin HD, et al. [Tumor necrosis factor-alpha and interleukin-10 gene polymorphisms in Korean patients with inflammatory bowel disease] Korean J Gastroenterol. 2003;42:377–386. [PubMed] [Google Scholar]
- Fidder HH, Heijmans R, Chowers Y, et al. TNF-857 polymorphism in Israeli Jewish patients with inflammatory bowel disease. Int J Immunogenet. 2006;33:81–85. doi: 10.1111/j.1744-313X.2006.00572.x. [DOI] [PubMed] [Google Scholar]
- Llanos C, Soto L, Sabugo F, et al. [The influence of −238 and −308 TNF alpha polymorphisms on the pathogenesis and response to treatment in rheumatoid arthritis] Rev Med Chil. 2005;133:1089–1095. doi: 10.4067/s0034-98872005000900014. [DOI] [PubMed] [Google Scholar]
- Mossbock G, Renner W, El-Shabrawi Y, et al. TNF-alpha −308 G>A and −238 G>A polymorphisms are not major risk factors in Caucasian patients with exfoliation glaucoma. Mol Vis. 2009;15:518–522. [PMC free article] [PubMed] [Google Scholar]
- Flego V, Radojcic Badovinac A, Bulat-Kardum L, et al. Primary lung cancer and TNF-alpha gene polymorphisms: a case-control study in a Croatian population. Med Sci Monit. 2009;15:CR361–CR365. [PubMed] [Google Scholar]
- Yang JJ, Ko KP, Cho LY, et al. The role of TNF genetic variants and the interaction with cigarette smoking for gastric cancer risk: a nested case-control study. BMC Cancer. 2009;9:238. doi: 10.1186/1471-2407-9-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohaar I, Tiwari P, Kumar R, et al. Association of single nucleotide polymorphisms (SNPs) in TNF-LTA locus with breast cancer risk in Indian population. Breast Cancer Res Treat. 2009;114:347–355. doi: 10.1007/s10549-008-0006-5. [DOI] [PubMed] [Google Scholar]
- Bouma G, Xia B, Crusius JB, et al. Distribution of four polymorphisms in the tumour necrosis factor (TNF) genes in patients with inflammatory bowel disease (IBD) Clin Exp Immunol. 1996;103:391–396. doi: 10.1111/j.1365-2249.1996.tb08292.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sashio H, Tamura K, Ito R, et al. Polymorphisms of the TNF gene and the TNF receptor superfamily member 1B gene are associated with susceptibility to ulcerative colitis and Crohn's disease, respectively. Immunogenetics. 2002;53:1020–1027. doi: 10.1007/s00251-001-0423-7. [DOI] [PubMed] [Google Scholar]
- Martin K, Radlmayr M, Borchers R, Heinzlmann M, Folwaczny C. Candidate genes colocalized to linkage regions in inflammatory bowel disease. Digestion. 2002;66:121–126. doi: 10.1159/000065592. [DOI] [PubMed] [Google Scholar]
- Gonzalez S, Rodrigo L, Martinez-Borra J, et al. TNF-alpha -308A promoter polymorphism is associated with enhanced TNF-alpha production and inflammatory activity in Crohn's patients with fistulizing disease. Am J Gastroenterol. 2003;98:1101–1106. doi: 10.1111/j.1572-0241.2003.07416.x. [DOI] [PubMed] [Google Scholar]
- Vatay A, Bene L, Kovacs A, et al. Relationship between the tumor necrosis factor alpha polymorphism and the serum C-reactive protein levels in inflammatory bowel disease. Immunogenetics. 2003;55:247–252. doi: 10.1007/s00251-003-0575-8. [DOI] [PubMed] [Google Scholar]
- Balding J, Livingstone WJ, Conroy J, et al. Inflammatory bowel disease: the role of inflammatory cytokine gene polymorphisms. Mediators Inflamm. 2004;13:181–187. doi: 10.1080/09511920410001713529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto-Furusho JK, Uscanga LF, Vargas-Alarcon G, Rodriguez-Perez JM, Zuniga J, Granados J. Polymorphisms in the promoter region of tumor necrosis factor alpha (TNF-alpha) and the HLA-DRB1 locus in Mexican mestizo patients with ulcerative colitis. Immunol Lett. 2004;95:31–35. doi: 10.1016/j.imlet.2004.05.015. [DOI] [PubMed] [Google Scholar]
- Fowler EV, Eri R, Hume G, et al. TNFalpha and IL10 SNPs act together to predict disease behaviour in Crohn's disease. J Med Genet. 2005;42:523–528. doi: 10.1136/jmg.2004.027425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantor MJ, Nickerson P, Bernstein CN. The role of cytokine gene polymorphisms in determining disease susceptibility and phenotype in inflammatory bowel disease. Am J Gastroenterol. 2005;100:1134–1142. doi: 10.1111/j.1572-0241.2005.40979.x. [DOI] [PubMed] [Google Scholar]
- Song Y, Wu KC, Zhang L, et al. Correlation between a gene polymorphism of tumor necrosis factor and inflammatory bowel disease. Chin J Dig Dis. 2005;6:170–174. doi: 10.1111/j.1443-9573.2005.00226.x. [DOI] [PubMed] [Google Scholar]
- Ferreira AC, Almeida S, Tavares M, et al. NOD2/CARD15 and TNFA, but not IL1B and IL1RN, are associated with Crohn's disease. Inflamm Bowel Dis. 2005;11:331–339. doi: 10.1097/01.mib.0000158153.71579.b4. [DOI] [PubMed] [Google Scholar]
- Zipperlen K, Peddle L, Melay B, Hefferton D, Rahman P. Association of TNF-alpha polymorphisms in Crohn disease. Hum Immunol. 2005;66:56–59. doi: 10.1016/j.humimm.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Castro-Santos P, Suarez A, Lopez-Rivas L, Mozo L, Gutierrez C. TNFalpha and IL-10 gene polymorphisms in inflammatory bowel disease. Association of -1082 AA low producer IL-10 genotype with steroid dependency. Am J Gastroenterol. 2006;101:1039–1047. doi: 10.1111/j.1572-0241.2006.00501.x. [DOI] [PubMed] [Google Scholar]
- Yang SK, Lee SG, Cho YK, Lim J, Lee I, Song K. Association of TNF-alpha/LTA polymorphisms with Crohn's disease in Koreans. Cytokine. 2006;35:13–20. doi: 10.1016/j.cyto.2006.07.002. [DOI] [PubMed] [Google Scholar]
- Sykora J, Subrt I, Didek P, et al. Cytokine tumor necrosis factor-alpha A promoter gene polymorphism at position −308 G—>A and pediatric inflammatory bowel disease: implications in ulcerative colitis and Crohn's disease. J Pediatr Gastroenterol Nutr. 2006;42:479–487. doi: 10.1097/01.mpg.0000221917.80887.9e. [DOI] [PubMed] [Google Scholar]
- Qian C, Qin Z, Minliang W, Weiling H, Min G, Jianmin S. Genetic susceptibility to ulcerative colitis in the Chinese Han ethnic population: association with TNF polymorphisms. Chin Med J. 2006;14:1198–1203. [PubMed] [Google Scholar]
- Celik Y, Dagli U, Kilic MY, et al. Cytokine gene polymorphisms in Turkish patients with inflammatory bowel disease. Scand J Gastroenterol. 2006;41:559–565. doi: 10.1080/00365520500349523. [DOI] [PubMed] [Google Scholar]
- Cucchiara S, Latiano A, Palmieri O, et al. Polymorphisms of tumor necrosis factor-alpha but not MDR1 influence response to medical therapy in pediatric-onset inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2007;44:171–179. doi: 10.1097/MPG.0b013e31802c41f3. [DOI] [PubMed] [Google Scholar]
- Mittal RD, Manchanda PK, Bid HK, Ghoshal UC. Analysis of polymorphisms of tumor necrosis factor-alpha and polymorphic xenobiotic metabolizing enzymes in inflammatory bowel disease: study from northern India. J Gastroenterol Hepatol. 2007;22:920–924. doi: 10.1111/j.1440-1746.2006.04538.x. [DOI] [PubMed] [Google Scholar]
- Ferguson LR, Huebner C, Petermann I, et al. Single nucleotide polymorphism in the tumor necrosis factor-alpha gene affects inflammatory bowel diseases risk. World J Gastroenterol. 2008;14:4652–4661. doi: 10.3748/wjg.14.4652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang WH, Yang YI, Yea SS, et al. The −238 tumor necrosis factor-alpha promoter polymorphism is associated with decreased susceptibility to cancers. Cancer Lett. 2001;166:41–46. doi: 10.1016/s0304-3835(01)00438-4. [DOI] [PubMed] [Google Scholar]
- Landi S, Moreno V, Gioia-Patricola L, et al. Association of common polymorphisms in inflammatory genes interleukin (IL)6, IL8, tumor necrosis factor alpha, NFKB1, and peroxisome proliferator-activated receptor gamma with colorectal cancer. Cancer Res. 2003;63:3560–3566. [PubMed] [Google Scholar]
- Macarthur M, Sharp L, Hold GL, Little J, El-Omar EM. The role of cytokine gene polymorphisms in colorectal cancer and their interaction with aspirin use in the northeast of Scotland. Cancer Epidemiol Biomarkers Prev. 2005;14:1613–1618. doi: 10.1158/1055-9965.EPI-04-0878. [DOI] [PubMed] [Google Scholar]
- Gunter MJ, Canzian F, Landi S, Chanock SJ, Sinha R, Rothman N. Inflammation-related gene polymorphisms and colorectal adenoma. Cancer Epidemiol Biomarkers Prev. 2006;15:1126–1131. doi: 10.1158/1055-9965.EPI-06-0042. [DOI] [PubMed] [Google Scholar]
- Theodoropoulos G, Papaconstantinou I, Felekouras E, et al. Relation between common polymorphisms in genes related to inflammatory response and colorectal cancer. World J Gastroenterol. 2006;12:5037–5043. doi: 10.3748/wjg.v12.i31.5037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toth EK, Kocsis J, Madaras B, et al. The 8.1 ancestral MHC haplotype is strongly associated with colorectal cancer risk. Int J Cancer. 2007;121:1744–1748. doi: 10.1002/ijc.22922. [DOI] [PubMed] [Google Scholar]
- Garrity-Park MM, Loftus EV, Jr, Bryant SC, Sandborn WJ, Smyrk TC. Tumor necrosis factor-alpha polymorphisms in ulcerative colitis-associated colorectal cancer. Am J Gastroenterol. 2008;103:407–415. doi: 10.1111/j.1572-0241.2007.01572.x. [DOI] [PubMed] [Google Scholar]
- Heuschen UA, Hinz U, Allemeyer EH, et al. Backwash ileitis is strongly associated with colorectal carcinoma in ulcerative colitis. Gastroenterology. 2001;120:841–847. doi: 10.1053/gast.2001.22434. [DOI] [PubMed] [Google Scholar]
- Bouma G, Crusius JB, Garcia-Gonzalez MA, et al. Genetic markers in clinically well defined patients with ulcerative colitis (UC) Clin Exp Immunol. 1999;115:294–300. doi: 10.1046/j.1365-2249.1999.00797.x. [DOI] [PMC free article] [PubMed] [Google Scholar]