Abstract
Beckwith–Wiedemann syndrome (BWS) is a phenotypically and genotypically heterogeneous overgrowth syndrome characterized by somatic overgrowth, macroglossia and abdominal wall defects. Other usual findings are hemihyperplasia, embryonal tumours, adrenocortical cytomegaly, ear anomalies, visceromegaly, renal abnormalities, neonatal hypoglycaemia, cleft palate, polydactyly and a positive family history. BWS is a complex, multigenic disorder associated, in up to 90% of patients, with alteration in the expression or function of one or more genes in the 11p15.5 imprinted gene cluster. There are several molecular anomalies associated with BWS and the large proportion of cases, about 85%, is sporadic and karyotypically normal. One of the major categories of BWS molecular alteration (10–20% of cases) is represented by mosaic paternal uniparental disomy (pUPD), namely patients with two paternally derived copies of chromosome 11p15 and no maternal contribution for that. In these patients, in addition to the effects of IGF2 overexpression, a decreased level of the maternally expressed gene CDKN1C may contribute to the BWS phenotype. In this paper, we reviewed a series of nine patients with BWS because of pUPD using several methods with the aim to evaluate the percentage of mosaicism, the methylation status at both loci, the extension of the pUPD at the short arm and the breakpoints of recombination. Fine mapping of mitotic recombination breakpoints by single-nucleotide polymorphism-array in individuals with UPD and fine estimation of epigenetic defects will provide a basis for understanding the aetiology of BWS, allowing more accurate prognostic predictions and facilitating management and surveillance of individuals with this disorder.
Keywords: Beckwith–Wiedemann syndrome, uniparental disomy, breakpoint, SNP-array
Introduction
Beckwith–Wiedemann syndrome (BWS (MIM 130650)) is a phenotypically and genotypically heterogeneous overgrowth syndrome characterized by somatic overgrowth, macroglossia and abdominal wall defects. Other usual findings are hemihyperplasia, embryonal tumours, adrenocortical cytomegaly, ear anomalies, visceromegaly, renal abnormalities, neonatal hypoglycaemia, cleft palate, polydactyly and a positive family history.1, 2, 3, 4
BWS is a complex, multigenic disorder associated, in up to 90% of patients, with alteration in the expression or function of one or more genes in the 11p15.5 imprinted gene cluster5 (Figure 1a). There are several molecular anomalies associated with BWS and the large proportion of cases, about 85%, is sporadic and karyotypically normal.
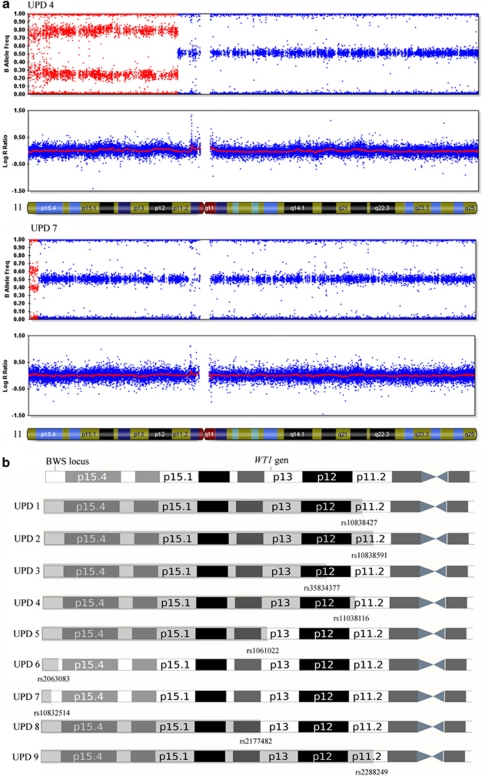
Figure 1.
(a) Schematic view of chromosome 11. Zoom of a part of 11p showing the location of genes and ICs of 11p15.5 BWS locus and WT1 gene. (b) Location of the six STR markers evaluated in patients and their parents.
Chromosomal rearrangements are relatively rare (∼2–3% of cases) and comprise translocations or inversions (typically maternally inherited), microdeletions of KvDMR6 or H19DMR7 (maternally inherited) and paternal duplications. Point mutations in CDKN1C, a cyclin-dependent kinase inhibitor acting as negative regulator of cell proliferation, have been found in 5–7% of sporadic BWS cases8, 9, 10, 11 and in approximately 40% of cases with a positive family history.12
The largest molecular subgroup, about 60% of cases, is represented by BWS patients that carry an epigenetic error on one or more members of 11p15.5 imprinted gene cluster. Two main epigenetic alterations in BWS patients have been described, one is represented by gain of methylation at the paternal H19DMR (observed in 5–10% of cases), and it is associated with loss of H19 expression and IGF2 biallelic expression.13, 14 The other alteration is loss of maternal methylation of KvDMR (observed in ∼50% of BWS patients), usually accompanied by biallelic expression of the KCNQ1OT1 transcript and downregulation of CDKN1C.15, 16, 17 Finally, another large category of BWS molecular alteration, 10–20% of cases, is represented by paternal uniparental disomy (pUPD), namely patients with two paternally derived copies of chromosome 11p15 and no maternal contribution for that region.18, 19 In these patients, in addition to the effects of IGF2 overexpression, a decreased level of the maternally expressed gene CDKN1C may contribute to the BWS phenotype.9, 12, 20
It has been demonstrated that BWS patients with paternal UPD always show mosaicisms, suggesting that all cases had arisen as a postzygotic event19, 21, 22; a possible explanation is that lack of one or more chromosome 11 maternally expressed genes may lead to embryonic lethality.23 It was also suggested that the degree of mosaicism and the location of the genetic abnormality in different tissue is strongly associated with the pathological phenotype.24 The extent of pUPD at chromosome 11p has been studied by means of STR markers; the critical region for pUPD is telomeric to chromosome 11p13 and always includes the region where map some BWS genes (IGF2, H19 and CDKN1C).19, 21, 23 Some cases with mosaic pUPD for the whole chromosome 11 have been also described and the clinical findings did not differ from patients with pUPD restricted to a small part of 11p.23, 25 Although the extent of segmental disomy and proportion of cells with pUPD is variable, in all BWS cases the paternal UPD is isodisomic. This suggests that cells with paternal UPD of chromosome 11 may have a selective growth advantage and that maternal UPD cells die or there is a partition to different parts of the conceptus.26
Clinically, a clear association between mosaic UPD and hemihyperplasia exists.15, 19, 22, 27 In addition, it was noted that neoplasias and Wilms' tumours are more frequent in BWS patients with pUPD or H19DMR hypermethylation than in BWS patients with other molecular defect.27, 28, 29 Moreover, it has been hypothesized that extremely high levels of UPD might drive severe phenotypic expression of BWS;30 but it is often difficult to determine if levels of UPD correlate with severity of the phenotype especially when the tissues/organs involved are not usually directly tested. Finally, in view of the evidence of imprinted transcripts at the Wilms' tumour-suppressor gene (WT1 at 11p13)31, 32, 33 it is interesting to evaluate whether disomy extended to WT1 influenced the risk of neoplasia (Figure 1a).
In this paper, we evaluated a series of nine patients with BWS because of pUPD of 11p using several methods with the aim to evaluate the percentage of mosaicism, the methylation status at both loci, the extent of the pUPD at the short arm and their breakpoints. Fine mapping of mitotic recombination breakpoints by single-nucleotide polymorphism (SNP)-array in UPD and fine estimation of epigenetic defects will provide a basis for understanding the aetiology of BWS. In addition, it would allow more accurate prognostic predictions providing a better management and surveillance of BWS children.
Patients and methods
Patients
From a total of 132 patients with presumptive diagnosis of BWS included in the Spanish Overgrowth Syndrome Registry, 92 had confirmed molecular diagnosis of the disorder. Among these patients, we detected 71 with hypomethylation at KvDMR, 8 with point mutation on CDKN1C gene, 2 with hypermethylation of H19DMR and 2 with paternal duplication of 11p15.5 region. Finally, 9 out of these 92 patients had laboratory results indicative of pUPD11. Clinical data of BWS individuals including personal and family history, clinical, laboratory and X-rays, pedigree, and follow-up evolution were included in a database. Parental data were also recorded. The Institutional IRB at Hospital Universitario La Paz approved this study as part of the research study at the Spanish Overgrowth Syndrome Registry (# HULP-PI446).
Cytogenetics and DNA extraction
Karyotypes were performed by standard methods in all patients. A minimum of 20 cells were counted with a resolution of at least 550 bands. FISH analysis using the D11S2071 probe was applied to all patients. DNA was extracted from blood using Qiagen kits Puregene Blood Core Kit (Gaithersburg, MD, USA) in all patients and available parents.
STRs segregation analyses
A panel of six microsatellites mapping to the short arm of chromosome 11 was evaluated in patients and their parents (when available) (Figure 1b). We used the following STRs: TH01, D11S1318, D11S4088, D11S1338, D11S1346 and D11S4046. Sequences of the primers were obtained from the NCBI public database (http://www.ncbi.nlm.nih.gov/sites/entrez). We calculated the percentage of mosaicism as recommended by Sasaki et al.34 The calculation was as follows: (K−1)/(K+1) × 100; where K is the ratio of the intensity of the parental alleles (paternal/maternal ratio) of the test sample.
Methylation-specific MLPA (MS–MLPA)
MS–MLPA with 100 ng of genomic DNA, the SALSA ME030-B1 (11p15 region, BWS/SRS; MRC-Holland, Amsterdam, Holland) and the methylation-sensitive restriction enzyme HhaI (Promega Corp., Madison, WI, USA) was performed according to the manufacturer's recommendations. MS–MLPA PCR products were analyzed on an ABI 3130 automated sequencer (Applied Biosystems, Foster City, CA, USA). To calculate the gene dosage, the analysis of results was based on the comparison between the signals of undigested samples and undigested controls. Calculation of the methylation level of probes containing the HhaI restriction site was performed comparing the signals of restriction digested samples with undigested samples. The analysis of raw data was carried out using an Excel-based in-house program (Meth-HULP v1.1; available on request). We normalized the raw value of peak areas in both controls and patient samples for undigested and digested samples and quantified gene dosage and methylation level for each sample.
Sodium bisulphite conversion and MS–high-resolution melting analysis (MS–HRMA)
Before MS–HRM assay, genomic DNA of controls and patients were treated with sodium bisulphite using the EZ DNA methylation Kit (Zymo Research, Orange, CA, USA) according to the manufacturer's instructions. Bisulphite converts unmethylated cytosines to uracil, whereas methylated cytosines remain unreactive.
Specific primers (available on request) to amplify H19 and KvDMR from bisulphite-converted DNA were designed by means of Methylation Primer Express Software v1.0 (Applied Biosystems). The primers amplify both methylated and unmethylated template, according to the principles set out to compensate for PCR bias.35 Specificity of primers was confirmed by sequencing of the PCR products. PCR amplification and MS–HRM analysis were done in a LyghtCycler 480 Real-Time PCR System (Roche Diagnostics, Penzberg, Germany). Normalization of the melting curves was carried out using the software provided with the LyghtCycler 480 (Gene Scanning Software v1.5, Roche Diagnostics). To visually estimate the curves, we included two synthetic DNA with standard methylation of 0 and 100% as controls (EpiTect PCR Control DNA Set, Qiagen). We calculated the level of methylation by the MS–HRM assay, evaluating the area under the curve in the plateau phase. The position of the plateau at the vertical scale represents the ratio between the methylated and unmethylated allele.
Pyrosequencing of imprinted centres at 11p15
Quantitative analysis of methylation at the KvDMR and H19 imprinting centres (ICs) was also performed by pyrosequencing. Specific primers (available on request) to amplify and to pyrosequence the two amplicons were designed by means of Pyrosequencing Assay Design Software (Biotage, Charlottesville, VA, USA). The forward primer for H19 and reverse primers for KvDMR amplification were 5′-biotinylated to facilitate single-strand DNA template isolation for the pyrosequencing reaction. Preparation of the single-strand DNA template for pyrosequencing was performed using the PSQ Vacuum Prep tool (Biotage) according to the manufacturer's instructions. The biotinylated PCR product was immobilized on streptavidin-coated Sepharose high-performance beads (Amersham Biosciences, Piscataway, NJ, USA) and processed to obtain single-strand DNA using the PSQ 96 Sample Preparation Kit (Biotage) according to the manufacturer's instructions. The sequencing-by-synthesis reaction of the complementary strand was automatically performed on a PSW 96MA instrument (Biotage) at room temperature using PyroGold reagents (Biotage). As nucleotides were dispensed, a light signal was generated proportional to the amount of each incorporated nucleotide. These light signals were detected by a charge-coupled device camera and converted to peaks in a sequencing program that was automatically generated in real time for each sample. The Pyro Q-CpG Software (Biotage) automatically determines individual methylation frequencies for all CpG sites in the amplicon; the degree of methylation is calculated from the ratio of the peak heights of C and T.
High-density SNP-arrays
DNAs were extracted by routine methods and were quantified using PicoGreen (Invitrogen Corp., Carlsbad, CA, USA). A genome-wide scan of 620 901 tag SNPs was conducted on the patients, using the Illumina Human610-Quad BeadChip according to the manufacturer's specifications (Illumina, San Diego, CA, USA). GenCall scores <0.15 at any locus were considered ‘no calls'. Image data were analyzed using the Chromosome Viewer tool contained in Beadstudio 3.2 (Illumina). The metric used was the log R ratio, which is the log (base 2) ratio of the observed normalized R value for a SNP divided by the expected normalized R value.36 In addition, an allele frequency analysis was applied for all SNPs. All genomic positions were based on NCBI Build 36 (dbSNP version 126, http://www.ncbi.nlm.nih.gov/ncbisearch).
Results
Paternal UPD and establishment of mosaicism percentage
All nine cases of UPD have been diagnosed by analysis of STRs markers mapping at the short arm of chromosome 11, and have shown paternal isodisomy (Supplementary Figure 1). We calculated the percentage of mosaicism by means of STRs and SNP-arrays (Figure 2a). The calculation of mosaicism using STRs has been done as recommended by Sasaki et al34 (Table 1a). Taking into account that isodisomy is paternal, we could estimate the percentage of mosaicism in the SNP-array as the higher allele B frequency (Table 2).
Figure 2.
(a) SNP-array results for two UPD patients (UPD 4 and UPD 7). The B allele frequency is shown in the upper panel of each patient. The red zone represents the chromosome fragment with UPD and the level of mosaicism for each SNP. The graph log R ratio is the measure of genomic dosage (normal in both patients). Note the differences presented by these two cases on the extent of UPD fragment and their level of mosaicism, which is higher for patient UPD 4 than UPD 7. (b) Graphic representation of the UPD extension for each patients and the latest SNP involved in the breakpoint. The color reproduction of this figure is available on the html full text version of the manuscript.
Table 1. Estimated percentage of cells with UPD calculated for each STR markers (a), methylation index calculated by MS–MLPA (b), MS–HRM (c) and pyrosequencing (d) for each patient.
| a | ||||||
|---|---|---|---|---|---|---|
| STR markers | ||||||
| Patient | D11S4046 | D11S1318 | D11S4088 | D11S1338 | D11S1346 | TH01 |
| UPD 1 | 85.77% | 82.01% | 77.77% | 53.49% | — | 89.38% |
| UPD 2 | 16.82% | 47.91% | 31.82% | 48.47% | 50.52% | 37.52% |
| UPD 3 | — | 57.44% | 21.49% | — | 51.23% | 41.39% |
| UPD 4 | 74.14% | 73.08% | 73.62% | 51.26% | 67.87% | 64.08% |
| UPD 5 | 33.35% | 45.58% | 40.59% | — | 74.83% | 63.51% |
| UPD 6 | 81.70% | 66.48% | 31.26% | — | 9.07% | 36.31% |
| UPD 7 | 12.93% | 28.35% | 32.39% | 22.79% | — | — |
| UPD 8 | 50.19% | — | 63.11% | 50.46% | 53.73% | 49.17% |
| UPD 9 | 58.78% | 38.82% | 73.84% | 37.68% | 45.54% | — |
| b | c | d | ||||
| MS–MLPA | MS–HRM | CpG Q-Pyro | ||||
|
Patient |
H19DMR 1 |
KvDMR |
H19DMR |
KvDMR |
H19DMR |
KvDMR |
| UPD 1 | 114.77% | 8.89% | 60.98% | 24.19% | 90.61% | 10.96% |
| UPD 2 | 81.25% | 31.70% | 46.01% | 48.91% | 73.15% | 27.94% |
| UPD 3 | 59.81% | 42.76% | 46.17% | 53.24% | 64.80% | 28.31% |
| UPD 4 | 79.99% | 21.38% | 50.41% | 40.36% | 86.78% | 20.50% |
| UPD 5 | 73.47% | 23.22% | 50.84% | 36.46% | 79.22% | 18.59% |
| UPD 6 | 69.27% | 33.98% | 51.99% | 50.68% | 72.07% | 26.05% |
| UPD 7 | 70.32% | 42.05% | 44.37% | 48.52% | 67.75% | 29.13% |
| UPD 8 | 69.09% | 34.47% | 52.41% | 45.96% | 74.26% | 28.34% |
| UPD 9 | 86.18% | 35.85% | 51.89% | 50.48% | 73.26% | 22.99% |
| Controls' mean | 50.26% | 54.69% | 39.64% | 59.67% | 52.11% | 40.68% |
| SD | 7.18% | 4.63% | 2.07% | 3.74% | 2.02% | 5.17% |
For the methylation analysis it has been calculated the normal methylation index as controls' mean value and SD, for each technique.
Table 2. SNP-array analyses results.
| Patient | Latest UPD SNP | Position | First Het SNP | Position | Min UPD size (bp) | Gap (bp) | Max UPD size (bp) | Inferior B allele frequency (%) | Superior B allele frequency (%) | SD (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| UPD 1 | rs10838427 | 45 138 258 | rs11038507 | 45 495 455 | 45 138 | 357 197 | 45 495 | 10.5 | 89.5 | 4.8 |
| UPD 2 | rs10838591 | 46 230 719 | rs8914 | 46 655 700 | 46 231 | 424 981 | 46 656 | 31.6 | 68.4 | 4.2 |
| UPD 3 | rs35834377 | 41 774 946 | rs11036703 | 42 105 122 | 41 775 | 330 176 | 42 105 | 35.5 | 64.5 | 4.2 |
| UPD 4 | rs11038116 | 44 680 447 | rs704664 | 44 743 777 | 44 681 | 63 330 | 44 744 | 23.1 | 76.9 | 4.6 |
| UPD 5 | rs1061022 | 32 083 259 | rs7105777 | 32 997 914 | 32 083 | 914 655 | 32 998 | 23.1 | 76.9 | 4.3 |
| UPD 6 | rs2063082 | 6 624 904 | rs11040978 | 6 690 629 | 6625 | 65 725 | 6691 | 35.7 | 64.3 | 6.5 |
| UPD 7 | rs10832514 | 2 645 102 | rs151288 | 2 742 878 | 2645 | 97 776 | 2743 | 39.1 | 60.9 | 3.3 |
| UPD 8 | rs2177482 | 31 758 933 | rs11031505 | 31 855 115 | 31 759 | 96 182 | 31 855 | 30.0 | 70.0 | 3.5 |
| UPD 9 | rs2288249 | 46 290 815 | rs876701 | 46 327 343 | 46 291 | 36 528 | 46 327 | 29.4 | 70.6 | 3.7 |
The minimum and maximum size estimated for the UPD extent and the gap between these two values are shown. The breakpoints were estimated by means of the identification of the last SNP affected by UPD and the first SNP in heterozygosis. The lower and higher B allele frequencies for all SNPs were calculated. Taking into account that isodisomy is paternal, the percentage of mosaicism of UPD cells is considered to be represented by the frequency of superior B allele.
Correlation between the percentage of the UPD cells and the methylation index
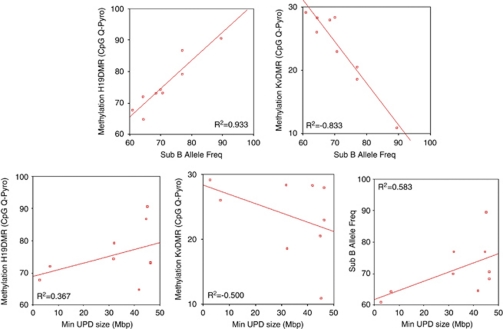
As expected, the degree of mosaicism, calculated by SNP-array, and the level of methylation defect, estimated by pyrosequencing (Table 1d), showed very high and statistically significant correlation: R2=+0.933 for H19DMR and R2=−0.833 for KvDMR (Figure 3a; Supplementary Table 1).
Figure 3.
Scatter graph of Spearman's correlations and relatives R2 values. (a) These two graphs represent the direct and inverse correlation between the percentage of mosaicism and the methylation level at H19DMR and KvDMR loci, respectively. (b) Graphic representation showing that there is no correlation between the size of the UPD fragment and the methylation level of both ICs and the percentage of mosaicism.
Extent of the pUPD and correlation with methylation index and mosaicism
Fine mapping of mitotic recombination breakpoint in pUPD of chromosome 11 has been performed by SNP-array (Figures 2a and b). To estimate the extent of pUPD, we mapped the latest SNP in pUPD and the first SNP in heterozygosis and we evaluated the minimum and the maximum size of pUPD region for each patient (Table 2). We calculated the methylation index in both ICs of 11p15.5 region, H19 and KvDMR, through MS–MLPA, MS–HRM and pyrosequencing analyses (Supplementary Figures 1b, 2a and b; Tables 1b–d). We also performed a correlation analysis between the extent of the pUPD and the level of methylation (obtained by CpG Q-Pyro) and mosaicism (calculated by STR analysis). As we expected, there has been no correlation among these parameters (Figure 3b; Supplementary Table 1).
Fine mapping of the breakpoints
Fine mapping of mitotic recombination breakpoints in pUPD of chromosome 11 by SNP-arrays did not provide strong evidence for recombination hot-spots. The locations of mitotic breakpoints were different in all patients (Figure 2b; Table 2). However, despite of variability of the extent of isodisomy, the region of paternal UPD invariably includes the whole imprinted gene cluster at 11p15.5 (Figure 2b).
Discussion
Paternal UPD of chromosome 11p is a relatively common and cancer-prone mechanism in BWS patients;27 thus patients with this molecular subtype should be closely evaluated and prospectively followed up. Regional or partial UPD are either caused by abnormal recombination between non-sister chromatids during mitosis or by deletion of a region and further duplication of the homologous region of the other chromosome.
From a total of 92 patients with confirmed molecular diagnosis of BWS only 9 patients (10%) had patUPD11p. This proportion is slightly lower than previous reports where close to 15–20% of the patients with BWS showed patUPD11p. This lower proportion may be due to the specific characteristics of our Registry with a high number of pregnancies obtained after assisted reproductive technologies, a procedure that it is hypothesized that predisposes to aberrant methylation of the centromeric IC2.37
Percentage of 11p mosaicism and BWS phenotype
It has been demonstrated that phenotypic expression of BWS correlates with elevated levels of UPD in the organs and tissues that were tested.24 However, it must be noted that the level of UPD has not been found to correlate with phenotypic expression. Our patients had clinical heterogeneity and lack of correlation with the percentage of mosaicism as well as the extension onto the short arm of chromosome 11. The only constant finding in all patients with patUPD11p in our series is hemihypertrophy. As an example of such lack of correlation, patient UPD 8 showed macrosomia at birth, hemihypertrophy and mild macroglossia and had 31Mb of extension and 70% of mosaicism and patient UPD 6 who has a smaller extension (6.6Mb) and 64% of mosaicism showed a more expanded phenotype including macrosomia, polyhydramnios, umbilical hernia, hemihypertrophy, macroglossia and hypoglycaemia (Supplementary Table 2). Thus, larger size of mosaic UPD and/or higher percentage of mosaicism do not directly imply a more florid manifestation of the disorder. This could be due to the fact that routine testing of a single tissue, such as blood, may not actually reflect the level of UPD in the tissues more susceptible to overgrowth in BWS.30
Comparison of mitotic and meiotic recombination breakpoints at 11p
Postfertilization errors such as nondisjunction with reduplication, mitotic recombination or gene conversion might lead to complete or partial isodisomy. Mosaicism might be frequent with postfertilization errors, because there would be no selection against the original disomic line. The site of breakpoints were different in all patients suggesting that in contrast to some common breakpoints observed in meiosis there seem to be no common mitotic recombination regions23, 38 (Table 2).
Correlation of the tests
We found that a combination of two tests (MS–MLPA + MS–HRMA or MS–MLPA + pyrosequencing) is the most useful approach for clinical diagnosis. SNP-arrays alone has been useful for diagnosing UPD11p, however, it cannot discriminate between paternal and maternal contribution. The costs of the studies are also important because the SNP-arrays are still expensive and should be set up in experienced laboratories. The limitations of our study are that only nine patients were evaluated and only one tissue (blood) has been tested in these patients. In contrast, we applied five different techniques to approach patUPD11p, including high-density SNP-arrays, which in fact it would have been useful itself to solve most of the issues because it can interrogate not only the dosage but also can demonstrate the percentage of mosaicism and its extent through the short arm of chromosome 11.
Summing up, SNP-arrays have been useful and very informative for clinical diagnosis in patients with BWS with mosaic UPD11p and might be used as the unique tool in clinical diagnostic laboratories, not only to evaluate mosaicism but also to map the mitotic site and consequently the extent of UPD at the short arm of chromosome 11.
Acknowledgments
Grant sponsor: Fondo de Investigación Sanitaria (FIS); Grant number: PI08/1360.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on European Journal of Human Genetics website (http://www.nature.com/ejhg)
Supplementary Material
References
- Beckwith JB. Extreme cytomegaly of the adrenal fetal cortex, omphalocele, hyperplasia of kidneys and pancreas, and Leydig-cell hyperplasia: another syndrome. Western Soc Pediatr Res (Abstr) (Los Angeles) 1963. p. 20.
- Elliott M, Maher ER. Beckwith-Wiedemann syndrome. J Med Genet. 1994;31:560–564. doi: 10.1136/jmg.31.7.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez RMY. Clinical features in the Wiedemann-Beckwith syndrome. Clin Genet. 1996;50:272–274. [PubMed] [Google Scholar]
- Wiedemann H-R. Familal malformation complex with umbilical hernia and macroglossia: a ‘new syndrome'. J Genet Hum. 1964;13:223–232. [PubMed] [Google Scholar]
- Maher ER, Reik W. Beckwith-Wiedemann syndrome: imprinting in clusters revisited. J Clin Invest. 2000;105:247–252. doi: 10.1172/JCI9340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niemitz EL, DeBaun MR, Fallon J, et al. Microdeletion of LIT1 in familial Beckwith-Wiedemann syndrome. Am J Hum Genet. 2004;75:844–849. doi: 10.1086/425343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riccio A, Sparago A, Verde G, et al. Inherited and sporadic epimutations at the IGF2-H19 locus in Beckwith-Wiedemann syndrome and Wilms' tumor. Endocr Dev. 2009;14:1–9. doi: 10.1159/000207461. [DOI] [PubMed] [Google Scholar]
- Lam WW, Hatada I, Ohishi S, et al. Analysis of germline CDKN1C (p57KIP2) mutations in familial and sporadic Beckwith-Wiedemann syndrome (BWS) provides a novel genotype-phenotype correlation. J Med Genet. 1999;36:518–523. [PMC free article] [PubMed] [Google Scholar]
- Lee MP, DeBaun M, Randhawa G, Reichard BA, Elledge SJ, Feinberg AP. Low frequency of p57KIP2 mutation in Beckwith-Wiedemann syndrome. Am J Hum Genet. 1997;61:304–309. doi: 10.1086/514858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romanelli V, Belinchon A, Campos-Barros A, et al. CDKN1C mutations in HELLP/preeclamptic mothers of Beckwith-Wiedemann syndrome (BWS) patients. Placenta. 2009;30:551–554. doi: 10.1016/j.placenta.2009.03.013. [DOI] [PubMed] [Google Scholar]
- Romanelli V, Belinchon A, Benito-Sanz S, et al. CDKN1C (p57(Kip2)) analysis in Beckwith-Wiedemann syndrome (BWS) patients: genotype-phenotype correlations, novel mutations, and polymorphisms. Am J Med Genet A. 2010;152A:1390–1397. doi: 10.1002/ajmg.a.33453. [DOI] [PubMed] [Google Scholar]
- O'Keefe D, Dao D, Zhao L, et al. Coding mutations in p57KIP2 are present in some cases of Beckwith-Wiedemann syndrome but are rare or absent in Wilms tumors. Am J Hum Genet. 1997;61:295–303. doi: 10.1086/514854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reik W, Brown KW, Slatter RE, Sartori P, Elliott M, Maher ER. Allelic methylation of H19 and IGF2 in the Beckwith-Wiedemann syndrome. Hum Mol Genet. 1994;3:1297–1301. doi: 10.1093/hmg/3.8.1297. [DOI] [PubMed] [Google Scholar]
- Schneid H, Seurin D, Vazquez MP, Gourmelen M, Cabrol S, Le Bouc Y. Parental allele specific methylation of the human insulin-like growth factor II gene and Beckwith-Wiedemann syndrome. J Med Genet. 1993;30:353–362. doi: 10.1136/jmg.30.5.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel JR, Smallwood A, Harper A, et al. Epigenotype-phenotype correlations in Beckwith-Wiedemann syndrome. J Med Genet. 2000;37:921–926. doi: 10.1136/jmg.37.12.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee MP, DeBaun MR, Mitsuya K, et al. Loss of imprinting of a paternally expressed transcript, with antisense orientation to KVLQT1, occurs frequently in Beckwith-Wiedemann syndrome and is independent of insulin-like growth factor II imprinting. Proc Natl Acad Sci USA. 1999;96:5203–5208. doi: 10.1073/pnas.96.9.5203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smilinich NJ, Day CD, Fitzpatrick GV, et al. A maternally methylated CpG island in KvLQT1 is associated with an antisense paternal transcript and loss of imprinting in Beckwith-Wiedemann syndrome. Proc Natl Acad Sci USA. 1999;96:8064–8069. doi: 10.1073/pnas.96.14.8064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry I, Bonaiti-Pellie C, Chehensse V, et al. Uniparental paternal disomy in a genetic cancer-predisposing syndrome. Nature. 1991;351:665–667. doi: 10.1038/351665a0. [DOI] [PubMed] [Google Scholar]
- Slatter RE, Elliott M, Welham K, et al. Mosaic uniparental disomy in Beckwith-Wiedemann syndrome. J Med Genet. 1994;31:749–753. doi: 10.1136/jmg.31.10.749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatada I, Ohashi H, Fukushima Y, et al. An imprinted gene p57KIP2 is mutated in Beckwith-Wiedemann syndrome. Nat Genet. 1996;14:171–173. doi: 10.1038/ng1096-171. [DOI] [PubMed] [Google Scholar]
- Catchpoole D, Lam WW, Valler D, et al. Epigenetic modification and uniparental inheritance of H19 in Beckwith-Wiedemann syndrome. J Med Genet. 1997;34:353–359. doi: 10.1136/jmg.34.5.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry I, Puech A, Riesewijk A, et al. Somatic mosaicism for partial paternal isodisomy in Wiedemann-Beckwith syndrome: a post-fertilization event. Eur J Hum Genet. 1993;1:19–29. doi: 10.1159/000472384. [DOI] [PubMed] [Google Scholar]
- Cooper WN, Curley R, Macdonald F, Maher ER. Mitotic recombination and uniparental disomy in Beckwith-Wiedemann syndrome. Genomics. 2007;89:613–617. doi: 10.1016/j.ygeno.2007.01.005. [DOI] [PubMed] [Google Scholar]
- Itoh N, Becroft DM, Reeve AE, Morison IM. Proportion of cells with paternal 11p15 uniparental disomy correlates with organ enlargement in Wiedemann-Beckwith syndrome. Am J Med Genet. 2000;92:111–116. [PubMed] [Google Scholar]
- Dutly F, Baumer A, Kayserili H, et al. Seven cases of Wiedmann-Beckwith syndrome, including the first reported case of mosaic paternal isodisomy along the whole chromosome 11. Am J Med Genet. 1998;79:347–353. doi: 10.1002/(sici)1096-8628(19981012)79:5<347::aid-ajmg4>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- Bischoff FZ, Feldman GL, McCaskill C, Subramanian S, Hughes MR, Shaffer LG. Single cell analysis demonstrating somatic mosaicism involving 11p in a patient with paternal isodisomy and Beckwith-Wiedemann syndrome. Hum Mol Genet. 1995;4:395–399. doi: 10.1093/hmg/4.3.395. [DOI] [PubMed] [Google Scholar]
- Cooper WN, Luharia A, Evans GA, et al. Molecular subtypes and phenotypic expression of Beckwith-Wiedemann syndrome. Eur J Hum Genet. 2005;13:1025–1032. doi: 10.1038/sj.ejhg.5201463. [DOI] [PubMed] [Google Scholar]
- Bliek J, Maas SM, Ruijter JM, et al. Increased tumour risk for BWS patients correlates with aberrant H19 and not KCNQ1OT1 methylation: occurrence of KCNQ1OT1 hypomethylation in familial cases of BWS. Hum Mol Genet. 2001;10:467–476. doi: 10.1093/hmg/10.5.467. [DOI] [PubMed] [Google Scholar]
- DeBaun MR, Niemitz EL, McNeil DE, Brandenburg SA, Lee MP, Feinberg AP. Epigenetic alterations of H19 and LIT1 distinguish patients with Beckwith-Wiedemann syndrome with cancer and birth defects. Am J Hum Genet. 2002;70:604–611. doi: 10.1086/338934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith AC, Shuman C, Chitayat D, et al. Severe presentation of Beckwith-Wiedemann syndrome associated with high levels of constitutional paternal uniparental disomy for chromosome 11p15. Am J Med Genet A. 2007;143A:3010–3015. doi: 10.1002/ajmg.a.32030. [DOI] [PubMed] [Google Scholar]
- Malik K, Salpekar A, Hancock A, et al. Identification of differential methylation of the WT1 antisense regulatory region and relaxation of imprinting in Wilms' tumor. Cancer Res. 2000;60:2356–2360. [PubMed] [Google Scholar]
- Malik KT, Wallace JI, Ivins SM, Brown KW. Identification of an antisense WT1 promoter in intron 1: implications for WT1 gene regulation. Oncogene. 1995;11:1589–1595. [PubMed] [Google Scholar]
- Ward A, Dutton JR. Regulation of the Wilms' tumour suppressor (WT1) gene by an antisense RNA: a link with genomic imprinting. J Pathol. 1998;185:342–344. doi: 10.1002/(SICI)1096-9896(199808)185:4<342::AID-PATH136>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- Sasaki K, Soejima H, Higashimoto K, et al. Japanese and North American/European patients with Beckwith-Wiedemann syndrome have different frequencies of some epigenetic and genetic alterations. Eur J Hum Genet. 2007;15:1205–1210. doi: 10.1038/sj.ejhg.5201912. [DOI] [PubMed] [Google Scholar]
- Wojdacz TK, Hansen LL. Reversal of PCR bias for improved sensitivity of the DNA methylation melting curve assay. Biotechniques. 2006;41:274–278. doi: 10.2144/000112240. [DOI] [PubMed] [Google Scholar]
- Simon-Sanchez J, Scholz S, Fung HC, et al. Genome-wide SNP assay reveals structural genomic variation, extended homozygosity and cell-line induced alterations in normal individuals. Hum Mol Genet. 2007;16:1–14. doi: 10.1093/hmg/ddl436. [DOI] [PubMed] [Google Scholar]
- Amor DJ, Halliday J. A review of known imprinting syndromes and their association with assisted reproduction technologies. Hum Reprod. 2008;23:2826–2834. doi: 10.1093/humrep/den310. [DOI] [PubMed] [Google Scholar]
- Russo S, Finelli P, Recalcati MP, et al. Molecular and genomic characterisation of cryptic chromosomal alterations leading to paternal duplication of the 11p15.5 Beckwith-Wiedemann region. J Med Genet. 2006;43:e39. doi: 10.1136/jmg.2005.038398. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.