Abstract
Purpose
To investigate the association between dry eye syndrome (DE) and serum levels of interleukin (IL)-17 in patients with systemic immune-mediated diseases.
Methods
IL-17 and IL-23 levels were measured in the sera of patients whose tear production was <5 mm on the Schirmer test. Subjects included patients with chronic graft-versus-host disease (GVHD), rheumatoid arthritis (RA), Sjogren's syndrome (SS), systemic lupus erythematosus (SLE), and no systemic disease. Corneal/conjunctival fluorescein staining was scored and the correlation between the score and the IL-17 level was evaluated.
Results
A strong correlation existed between IL-17 level and the type of systemic disease. IL-17 was significantly elevated in patients with chronic GVHD compared to those with RA and SS. IL-17 was not detectable in patients with SLE or in those without systemic disease. IL-23 was not detected in any of the subjects. IL-17 was significantly increased in patients with high fluorescein staining scores.
Conclusions
Our data suggest that IL-17 is involved in the pathogenesis of DE in patients with systemic immune-mediated diseases.
Keywords: Chronic graft-versus-host disease, Dry eye syndromes, Interleukin-17, Rheumatoid arthritis, Sjogren's syndrome
Dry eye syndrome (DE) is one of the most important complications of systemic immune-mediated diseases such as chronic graft-versus-host disease (GVHD), rheumatoid arthritis (RA), Sjogren's syndrome (SS), and systemic lupus erythematosus (SLE). All of these disorders are characterized by destruction of acinar tissue within the lacrimal glands and ocular surface inflammation, which both lead to DE.
Interleukin (IL)-17 is a pro-inflammatory cytokine produced by a number of human immune cells, including Th17 cells, neutrophils, and peripheral blood mononuclear cells. Recent studies have revealed that IL-17 is upregulated in various autoimmune inflammatory diseases and is involved in the pathogenesis of RA, SS, SLE, and GVHD [1-4]. Regarding ocular diseases, an upregulation of IL-17 or IL-23 has been observed in patients with uveitis and scleritis [5-7]. IL-23 is an essential cytokine for Th17 cell differentiation and function [8,9]. Also, IL-23 has been shown to be necessary for the development of systemic autoimmune diseases such as inflammatory bowel disease and collagen-induced arthritis [10-12]. These findings suggest that the IL-23/IL-17 pathway is involved in the development of DE associated with systemic immune-mediated diseases. However, the association between the IL-23/IL-17 pathway and DE has not been established. Given that drugs designed to neutralize IL-23 and IL-17 are in the pipeline, efforts to elucidate the role of those cytokines in DE are worthwhile. Therefore, we performed this study in order to investigate the association between DE and IL-23/IL-17 in DE patients with associated systemic immune-mediated diseases.
Materials and Methods
The patients enrolled in this study had tear production <5 mm by the non-anesthetic Schirmer test. Subjects included patients with chronic GVHD (n = 8), RA (n = 8), SS (n = 12), SLE (n = 6), and no systemic disease (n = 10). The systemic diseases of the patients were under control with/out immunosuppressants. Sera were obtained from peripheral blood samples collected from patients without anticoagulant agents and stored at ≤-20℃ until assay time. The serum levels of IL-17 and IL-23 were measured by the ELISA method using R&D Systems kits (Minneapolis, MN, USA) according to the instructions provided by the manufacturer. ELISA experiments were performed in triplicate to ensure the reproducibility of the data. The lower limits of sensitivity for IL-17 and IL-23 ELISA were 15 pg/mL and 4 pg/mL, respectively.
DE severity and ocular surface inflammation were evaluated with corneal/conjunctival fluorescein staining. The ocular surface was examined with a slit lamp biomicroscope in cobalt blue light, three minutes after fluorescein instillation. Punctate staining was recorded with a standardized scoring system; the corneal and conjunctival surfaces were divided into nine areas and the numbers of stained divisions were summed (0 to 9) [13]. Fluorescein staining was graded as mild (scores 0 to 3), moderate (4 to 6), or severe (6 to 9). We evaluated the correlation between the fluorescein staining score and IL-17 level.
Data were expressed as means ± SD and compared using the student's t-test and simple correlation analysis. A p-value of <0.05 was considered statistically significant.
Results
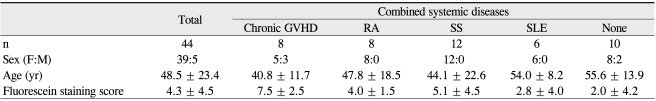
The clinical features and demographic data of the patients in each group are shown in Table 1. The differences in age and sex were not significant among the groups. The IL-17 levels did not show a significant correlation with patient age (p = 0.246).
Table 1.
Clinical and demographic data of dry eye patients enrolled in the study
GVHD = graft-versus-host disease; RA = rheumatoid arthritis; SS = Sjogren's syndrome; SLE = systemic lupus erythematosus.
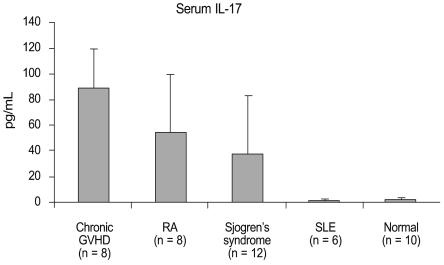
Notably, a strong correlation existed between the IL-17 level and the type of systemic disease (Fig. 1). The IL-17 level in patients with chronic GVHD was 89.0 ± 30.2 pg/mL, which was significantly higher than the level in patients with RA (54.0 ± 46.0 pg/mL; p = 0.02) or SS (37.0 ± 46.7 pg/mL; p = 0.043). In contrast, IL-17 was not detected in the sera of DE patients with SLE or in patients without systemic disease. The IL-17 elevation in patients with RA and SS was statistically significant compared to the levels in patients with SLE and those without systemic disease (p = 0.021 and 0.036, respectively). IL-23 was not detected in any of the subjects.
Fig. 1.
Levels of interleukin (IL)-17 in sera from dry eye patients with systemic diseases and dry eye subjects without systemic disease (control). GVHD = graft-versus-host disease; RA = rheumatoid arthritis; SLE = systemic lupus erythematosus.
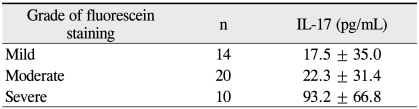
In addition, IL-17 level was significantly correlated with the fluorescein staining score (Table 2). Patients with severe fluorescein staining on the ocular surface (defined as staining scores 6 to 9) had higher IL-17 serum levels than those with moderate or mild fluorescein staining (p = 0.01 and 0.024, respectively).
Table 2.
Levels of interleukin (IL)-17 in dry eye patients with regard to the corneal/conjunctival fluorescein staining score
Discussion
The present study demonstrated that IL-17 production was significantly increased in the sera of DE patients with systemic autoimmune diseases, whereas IL-17 was not elevated in DE patients without systemic inflammatory co-morbidities. Interestingly, our study revealed significant IL-17 elevation in patients with chronic GVHD. Although there has been a study evaluating IL-17 in an acute GVHD animal model [14], the present study is, to our knowledge, the first to report an association between IL-17 and GVHD in humans.
In our study, IL-17 level was also associated with the severity of DE, as represented by fluorescein staining scores on the ocular surface. Given the fact that patients with chronic GVHD had more severe fluorescein staining of their eyes, it is possible that severe ocular surface inflammation induced IL-17 production and led to the IL-17 elevation seen in the sera of the DE patients with chronic GVHD. However, previous reports have demonstrated that IL-17 and Th17 cells are linked to systemic autoimmune diseases such as RA and SS in humans [1-5]. Moreover, a recent report revealed that IL-17 significantly disrupts the corneal epithelial barrier by inducing metalloproteinase production on the ocular surface [15]. Taken together, it is more likely that increased IL-17 in patients with chronic GVHD, RA, or SS induced corneal epithelial barrier disruption and led to severe DE in our study.
In our study, IL-23 was not detectable in any of the patients. Recent reports have suggested that IL-23 promotes IL-17 production by CD4+ T cells and increases susceptibility to autoimmune inflammation [16]. As such, the fact that no IL-23 was detected in any of the subjects in this study might be related to the fact that our patients' systemic inflammatory diseases were under good control.
DE is an inflammatory process localized to the ocular surface-lacrimal gland loop [17]. In addition, according to a recent report, desiccating stress has been shown to induce a Th17 response localized in the cornea and conjunctiva [15]. Therefore, evaluation of IL-17 in the lacrimal glands, ocular surface, or tears would be valuable in confirming the role of IL-17 in DE. We did not perform such studies because lacrimal gland biopsy is invasive and the volume of tears that can be acquired from DE patients with very low tear production is insufficient to perform an ELISA assay. Thus, our results do not exclude the possibility that the IL-17 pathway is involved in DE in a manner unrelated to systemic immune-mediated disease. In addition, to evaluate the role of IL-17 in the pathogenesis of DE, animal studies using an IL-17 knockout animal or siRNA would be valuable, as demonstrated by several reports [18].
Our study is limited in that the number of subjects was not sufficient to draw firm conclusions, especially in disorders as heterogeneous as GVHD, RA, SS, and SLE. Furthermore, the patients in our study were on various immunosuppressant medications that could potentially affect serum IL-17 levels. Further studies involving a large number of subjects are needed. Also, as previously mentioned, the serum level of IL-17 may not directly correlate with the level of IL-17 in tears. However, we believe our study sheds light on the possible association between IL-17 and DE.
In conclusion, our study showed that IL-17 was elevated in DE patients with chronic GVHD, RA, and SS. It also showed that IL-17 elevation was more significant in DE patients with severe fluorescein staining on the ocular surface. All these results suggest that the IL-17 pathway is involved in the pathogenesis of DE associated with systemic inflammatory diseases. Drugs designed to neutralize IL-17 are in the pipeline, and they have proven to be effective in treating RA. Hence, further studies on the role of IL-17 in DE will provide new strategies for the treatment of DE.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Nguyen CQ, Hu MH, Li Y, et al. Salivary gland tissue expression of interleukin-23 and interleukin-17 in Sjogren's syndrome: findings in humans and mice. Arthritis Rheum. 2008;58:734–743. doi: 10.1002/art.23214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sakai A, Sugawara Y, Kuroishi T, et al. Identification of IL-18 and Th17 cells in salivary glands of patients with Sjogren's syndrome, and amplification of IL-17-mediated secretion of inflammatory cytokines from salivary gland cells by IL-18. J Immunol. 2008;181:2898–2906. doi: 10.4049/jimmunol.181.4.2898. [DOI] [PubMed] [Google Scholar]
- 3.Garrett-Sinha LA, John S, Gaffen SL. IL-17 and the Th17 lineage in systemic lupus erythematosus. Curr Opin Rheumatol. 2008;20:519–525. doi: 10.1097/BOR.0b013e328304b6b5. [DOI] [PubMed] [Google Scholar]
- 4.Tesmer LA, Lundy SK, Sarkar S, Fox DA. Th17 cells in human disease. Immunol Rev. 2008;223:87–113. doi: 10.1111/j.1600-065X.2008.00628.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chi W, Zhu X, Yang P, et al. Upregulated IL-23 and IL-17 in Behçet patients with active uveitis. Invest Ophthalmol Vis Sci. 2008;49:3058–3064. doi: 10.1167/iovs.07-1390. [DOI] [PubMed] [Google Scholar]
- 6.Amadi-Obi A, Yu CR, Liu X, et al. TH17 cells contribute to uveitis and scleritis and are expanded by IL-2 and inhibited by IL-27/STAT1. Nat Med. 2007;13:711–718. doi: 10.1038/nm1585. [DOI] [PubMed] [Google Scholar]
- 7.Abraham C, Cho J. Interleukin-23/Th17 pathways and inflammatory bowel disease. Inflamm Bowel Dis. 2009;15:1090–1100. doi: 10.1002/ibd.20894. [DOI] [PubMed] [Google Scholar]
- 8.Diveu C, McGeachy MJ, Cua DJ. Cytokines that regulate autoimmunity. Curr Opin Immunol. 2008;20:663–668. doi: 10.1016/j.coi.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 9.McGeachy MJ, Chen Y, Tato CM, et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314–324. doi: 10.1038/ni.1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Langrish CL, Chen Y, Blumenschein WM, et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med. 2005;201:233–240. doi: 10.1084/jem.20041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yen D, Cheung J, Scheerens H, et al. IL-23 is essential for T cell-mediated colitis and promotes inflammation via IL-17 and IL-6. J Clin Invest. 2006;116:1310–1316. doi: 10.1172/JCI21404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Murphy CA, Langrish CL, Chen Y, et al. Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J Exp Med. 2003;198:1951–1957. doi: 10.1084/jem.20030896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Norn MS. Lissamine green. Vital staining of cornea and conjunctiva. Acta Ophthalmol (Copenh) 1973;51:483–491. doi: 10.1111/j.1755-3768.1973.tb06027.x. [DOI] [PubMed] [Google Scholar]
- 14.Kappel LW, Goldberg GL, King CG, et al. IL-17 contributes to CD4-mediated graft-versus-host disease. Blood. 2009;113:945–952. doi: 10.1182/blood-2008-08-172155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.De Paiva CS, Chotikavanich S, Pangelinan SB, et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009;2:243–253. doi: 10.1038/mi.2009.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sutton CE, Lalor SJ, Sweeney CM, et al. Interleukin-1 and IL-23 induce innate IL-17 production from gammadelta T cells, amplifying Th17 responses and autoimmunity. Immunity. 2009;31:331–341. doi: 10.1016/j.immuni.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 17.Mathers WD. Why the eye becomes dry: a cornea and lacrimal gland feedback model. CLAO J. 2000;26:159–165. [PubMed] [Google Scholar]
- 18.Chauhan SK, El Annan J, Ecoiffier T, et al. Autoimmunity in dry eye is due to resistance of Th17 to Treg suppression. J Immunol. 2009;182:1247–1252. doi: 10.4049/jimmunol.182.3.1247. [DOI] [PMC free article] [PubMed] [Google Scholar]