Abstract
Radiation exposure accompanying medical imaging associates with cancer risk. Patients with recurrent or chronic diseases may be especially at risk, because they may undergo more of these procedures. The aim of this study was to assess the individual cumulative effective doses (CEDs), which quantify radiation from medical imaging procedures, in a cohort of 106 hemodialysis patients during a median follow-up of 3 years. We retrospectively calculated individual radiation exposures by collecting the number and type of radiologic procedures from hospital records. We also estimated organ doses for computed tomography procedures. The mean and median annual CEDs were 21.9 and 11.7 mSv per patient-year, respectively. The mean and median total CEDs per patient during the study period were 57.7 and 27.3 mSv, respectively. By radiation dose group, we classified 22 patients as low (<3 mSv/yr), 51 as moderate (3 to <20 mSv/yr), 22 as high (20 to <50 mSv/yr), and 11 as very high (≥50 mSv/yr). Seventeen patients had a total CED >100 mSv, a value associated with a substantial increase in risk for cancer- mortality. Of the total CED,s 76% was a result of CT scanning. The annual CED significantly associated with age and transplant waitlist status. In summary, this study shows that a significant fraction of surviving hemodialysis patients during a 3-year period receives estimated radiation doses that may put them at an increased risk for cancer.
Medical uses of radiation have risen rapidly over the past decade, and as of 2006, medical uses represent the largest source of exposure to the U.S. population, accounting for 3.0 mSv against an estimated 2.4 mSv from a natural background.1 Advances in medical imaging have been associated with increased radiation exposure, especially for patients with chronic and recurrent conditions.2 The association of ionizing radiation and cancer risk is assumed to be continuous and graded over the entire range of exposure,3 and approximately 29,000 future cancers have been related to computed tomography (CT) examinations performed in the United States in 2007.4 Patients on hemodialysis require ongoing care that often results in repeated imaging and repeated exposure to ionizing radiation for both diagnostic and therapeutic purposes.5 Maintenance hemodialysis, besides being such a chronic condition, is associated with an increased incidence of cancer of unclear etiology. Patients with ESRD have a fourfold higher risk of cancer compared with the general population, but the cancer risk is different according to the renal replacement therapy: there is an increase of 1 to 1.5 times during dialysis and 2.5 to 5 times after kidney transplantation for both uremia and drug-related immunosuppression.6,7 The excess risk of cancer associated with radiation exposure8 adds in these patients to the increased incidence of cancer because of the inherent pathology and must be taken into careful consideration particularly in younger patients and in those eligible for kidney transplantation. Cumulative effective dose (CED) allows for comparisons/summation of radiation exposure generated from different origins and has been previously used to quantify the radiation exposure from medical imaging procedures.9
The aim of this retrospective study is to quantify the CED of ionizing radiation in hemodialysis patients, to identify the subgroups that are at an increased risk, and to consider the potential health consequences of this radiation exposure.
RESULTS
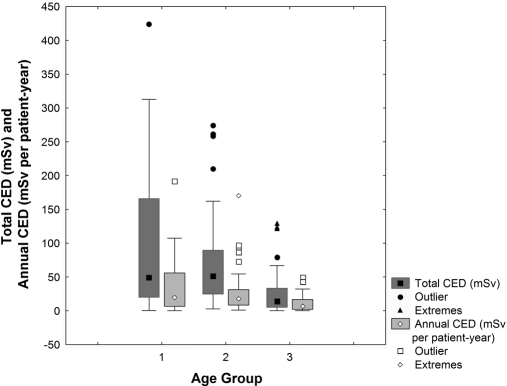
The 106 study patients (63 men) were followed for a median of 3.0 years. During the study period, 23 patients (21.6%) died, whereas 6 (5.6%) underwent kidney transplantation. In these cases, the data were censored at the date of death or of transplantation. Thus, a total of 281 patient-years were available for follow-up. The mean ± SD age at study entry was 65.3 ± 14.6 years. Among the subjects, 14 were in the 18- to 50-year age group, 41 were in the 50- to 70-year age group, and 51 were >70 years. In all, 77% of the subjects were prevalent, with a median (intraquartile range [IQR]) dialysis period of 4.0 (1.6 to 8.3) years, and the remaining 23% initiated dialysis during the study period. The average radiation exposure was significantly associated to the younger-aged patients who were exposed to higher total CEDs (P = 0.0001) and annual CEDs (P = 0.0002) than the older patients (Fig. 1). Also the transplant waiting list status was associated with a significantly higher (P = 0.04) annual CED (Table 1). Overall, 35 patients started the screening for renal transplantation. There were five patients denied renal transplantation on the basis of an abdominal/pelvic CT study showing severe atherosclerosis of the iliac arteries. Of the 30 patients in the active transplant waiting list, they underwent, in total, 283 conventional radiology, 85 CT, 51 nuclear medicine, and 27 interventional procedures with 10 cardiac catheterizations. Average annual CED was not significantly different for men and women, for comorbid conditions, or for death during the study period.
Figure 1.
Total and annual CED are higher in younger patients. Box plot showing median and intraquartile range of average cumulative effective dose of radiation from radiologic procedures by age group at study entry.
Table 1.
Patient characteristics for the study population and comparison of average annual CED for gender, transplant waiting list status, and comorbid conditions including death
| Patient Characteristics | Total [N (%)] | Annual CED (mSv per Patient-Year) (Mean ± SD) |
P | |
|---|---|---|---|---|
| Yes | No | |||
| Male | 63 (59.4%) | 22.0 ± 31.0 | 21.6 ± 32.0 | 0.44 |
| Diabetes mellitus | 26 (24.5%) | 20.0 ± 18.6 | 22.5 ± 34.4 | 0.33 |
| Ischemic heart disease | 44 (41.5%) | 21.1 ± 23.5 | 22.4 ± 35.9 | 0.30 |
| Tumor | 20 (18.9%) | 25.8 ± 23.3 | 20.9 ± 328 | 0.11 |
| Renal transplant eligible | 30 (28.3%) | 30.5 ± 40.0 | 18.4 ± 26.5 | 0.04 |
| Death | 23 (21.7%) | 32.4 ± 45.2 | 18.9 ± 25.7 | 0.40 |
Among the subjects, 22 were in the low (<3 mSv/yr), 51 in the moderate (3 to <20 mSv/yr), 22 in the high (20 to <50 mSv/yr), and 11 in the very high (≥50 mSv/yr) radiation dose groups. Seventeen patients had a total CED >100 mSv.
The total number of radiologic procedures for all patients was 1303, and the annual median and mean CEDs are shown in Table 2. The median (IQR) total CED per subject over the study period was 27.3 mSv (9.8 to 60.0). The mean total CED was 55.7 ± 73.6 mSv. The mean levels are much higher than the median annual and total CED, which reflects the dramatic right-skew in this distribution of patients with increasing CED.
Table 2.
Number of radiologic procedures and annual and total CED by procedure type
| Procedure | Number of Examinations [N (%)] | Annual CED (mSv per Patient-Year) [Median (IQR)] | Annual CED (mSv per Patient-Year) (mean ± SD) | Total CED mSv (%) |
|---|---|---|---|---|
| Overall total | 1303 (100%) | 11.7 (4.3 to 24.7) | 21.9 ± 31.2 | 5901.4 (100%) |
| Conventional diagnostic radiology | 848 (65.0%) | 1.4 (0.7 to 2.7) | 1.9 ± 1.5 | 488.7 (8.3%) |
| CT | 248 (19.0%) | 6.5 (0 to 18.6) | 16.6 ± 29.2 | 4484.0 (76.0%) |
| Nuclear medicine | 108 (8.2%) | 0.0 (0 to 2.8) | 1.6 ± 1.3 | 451.1 (7.6%) |
| Interventional | 99 (7.6%) | 0.0 (0 to 2.3) | 1.8 ± 1.5 | 477.5 (8.1%) |
IQR, intraquartile range.
The median (IQR) number of radiologic procedures was 4.3 (2.3 to 6.3) per patient-year. All patients underwent at least one procedure involving exposure to ionizing radiation: 65% had at least one CT examination during the study period; this percentage decreases to 50% for nuclear medicine and to 52% for interventional procedures. The proportion of total radiation exposure attributable to different types of investigations is also shown in Table 2. CT examinations accounted for 76% of the total CED, while accounting for only 19% of the total number of radiological procedures.
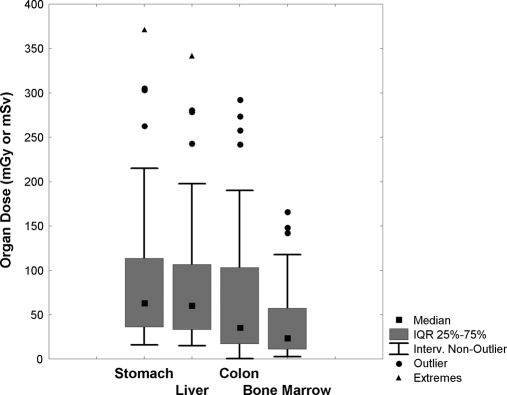
Among the 248 CT procedures, 101 (40.7%) were performed to diagnose a specific disease (i.e., when patients had symptoms such as neurologic signs, hemorrhages, respiratory distress, trauma, acute pain, suspected cancer, abscess, appendicitis or diverticulitis, suspected aneurysm or dissection), 101 (40.7%) were performed to monitor a previously diagnosed disease, and 46 (18.6%) were for transplant screening. In 96 of 248 (38.7%) cases, the CT examinations resulted in findings that were judged by the referring physicians as relevant for the patient's clinical management and/or therapy. On the other hand, in 118 (47.6%) cases, the CT findings, although showing some abnormalities, did not changed the patient's management, and in 34 (13.7%), produced negative results. Conventional diagnostic radiology, nuclear medicine, and interventional procedures accounted for 65, 8.2, and 7.6% of the frequency in procedures and for 8.3, 7.6, and 8.1% of total CED, respectively. The proportion of total CED to different types of CT examinations is shown in Table 3. Although comprising only 43.1% of the CT procedures, abdominal/pelvic examinations resulted in 73.2% of the CT radiation exposure and 55.6% of the total CED. For 50 of 106 (47%) patients who were submitted to at least one abdominal/pelvic CT examination during the study period, cumulated doses to relevant organs (stomach, liver, colon, and bone marrow) were estimated. Effective dose and organ doses for abdominal and abdominal/pelvic CT examinations as performed in our institution are shown in Table 4. Frequency distributions of cumulated organ doses are represented in Figure 2 by means of box and whiskers plots. The median cumulative organ doses in this subgroup were 64, 60, 35, and 24 mSv for stomach, liver, colon, and bone marrow, respectively.
Table 3.
Number of CT examinations, CT scans, and related total CED
| Procedure | Number of Examinations, [N (%)] | Number of Scans (Average N of Scans per Examination) | Total CED mSv(%) |
|---|---|---|---|
| Overall total | 248 (100%) | 507 (2.0) | 4484 (100%) |
| Head/neck | 65 (26.2%) | 79 (1.2) | 181 (4.0%) |
| Chest | 74 (29.8%) | 141 (1.9) | 987 (22.1%) |
| Abdomen/pelvis | 107 (43.1%) | 285 (2.4) | 3286 (73.2%) |
| Other | 2 (0.008%) | 2 (1.0) | 30 (0.007%) |
Table 4.
Effective dose and organ radiation doses for abdominal/pelvic CT examinations estimated at our institution
| Routine Abdomen-Pelvis | Suspected Aneurysm or Dissection | Routine Upper Abdomen | |
|---|---|---|---|
| Effective dose (mSv)a | 14.8 | 17.2 | 6.2 |
| Organs or tissues | Organ Dose (mGy or mSv)b | ||
| gonads | 31.3 | 35.9 | 0.8 |
| bone marrow (red) | 12.4 | 14.3 | 3.8 |
| colon | 23.2 | 26.9 | 0.7 |
| lung | 7.4 | 8.7 | 7.5 |
| stomach | 24.8 | 29.4 | 21.6 |
| bladder | 32.4 | 38.9 | 0.2 |
| breast | 1.4 | 1.6 | 1.3 |
| liver | 22.7 | 27.3 | 20.5 |
| esophagus (thymus) | 1.1 | 1.3 | 1.1 |
| thyroid | 0.1 | 0.1 | 0.1 |
| skin | 8.8 | 10.2 | 3.3 |
| bone surface | 16.4 | 19.1 | 6.9 |
| remainder | 12.1 | 14.0 | 3.8 |
aEffective doses and organ doses are reported for a single scan.
bThe radiation dose, a measure of ionizing energy absorbed per unit of mass, is expressed in grays (Gy) or milligrays (mGy); 1 Gy = 1 joule per kilogram. The radiation dose is often expressed as an equivalent dose in Sieverts (Sv) or millisieverts (mSv). For x-ray radiation, which is the type used in CT scanners, 1 mSv = 1 mGy.
Figure 2.
Abdominal/pelvic CT exposes patients to high organ doses. Box plot showing median and intraquartile range of cumulative organ dose of radiation in a subgroup of 50 patients who underwent at least one abdominal/pelvic CT.
The main analyses were repeated in the subset of 86 noncancer patients. Both the total CED and annual CED were significantly higher in younger patients than in older patients (P = 0.0009 and P = 0.0029, respectively). In this subset, the transplant waiting list status was associated with a significantly higher (P = 0.02) annual CED, whereas average radiation exposure was not significantly different for men and women, for comorbid conditions, or for death during the study period (Table 5).
Table 5.
Patient characteristics for the subset of 86 noncancer patients and comparison of average annual CED for gender, transplant waiting list status, and comorbid conditions including death
| Patient Characteristics | Total [N (%)] | Annual CED (mSv per Patient-Year) (Mean ± SD) |
P | |
|---|---|---|---|---|
| Yes | No | |||
| Male | 50 (58.1%) | 20.3 ± 32.0 | 21.8 ± 34.4 | 0.23 |
| Diabetes mellitus | 22 (25.6%) | 17.6 ± 17.6 | 22.1 ± 36.7 | 0.42 |
| Ischemic heart disease | 38 (44.1%) | 18.4 ± 23.3 | 23.0 ± 38.9 | 0.51 |
| Renal transplant eligible | 25 (29.1%) | 32.6 ± 43.4 | 16.1 ± 26.3 | 0.03 |
| Death | 18 (20.9%) | 28.6.±48.4 | 18.9 ± 27.5 | 0.97 |
DISCUSSION
This study showed that within 3 years, a significant fraction of surviving hemodialysis patients received estimated radiation doses that may put them at an increased risk of cancer. The cumulative radiation exposure was significantly higher in relatively younger patients and in those who are transplant eligible. This is of particular concern given the anticipated life expectancy of these subjects and the ongoing use of immunosuppressive agents in the latter.
Although there is some disagreement about the level of risk from low-level radiation exposure over time, the current consensus favors the linear no-threshold model.10 Under this model, the cumulative exposure to radiation over a lifetime is linearly associated with an increased risk of cancer. Some contend that the model fails to account for the rate of radiation exposure or for cell capacity to repair radiation damage.11 The assumed risk estimates are ultimately derived from analyses of mortality data based on Japanese atomic bomb survivors7 exposed to intermediate radiation doses (approximately 40 mSv), like two or three typical CT scans in adults. The atomic bomb data provide strong evidence of an increased cancer mortality risk at equivalent doses >100 mSv, good evidence of an increased risk for doses between 50 and 100 mSv, and reasonable evidence for an increased risk for doses between 10 and 50 mSv.8 However, while recognizing these controversies about the validity of the linear no-threshold model, it must also be recognized that the organ doses for a typical CT study of the body involving two or three scans are in the range in which there is direct evidence of a statistically significant increase in the risk of cancer, and the corresponding CT-related risks can thus be directly assessed from epidemiologic data, without the need to extrapolate measured risk at lower doses. For instance, Brenner et al.12 reported an estimated lifetime cancer mortality risks attributable to the radiation exposure from a single typical CT examination of abdomen in an adult of 0.02%, based on typical organ doses very similar to our estimates, reported in Table 4 (the higher values for gonads and colon are mainly caused by the involvement of the pelvic region). The number of CT scans in a given study is an important factor in determining the dose. For example, Mettler et al.13 reported that in virtually all patients undergoing CT of the abdomen or pelvis, more than one scan was obtained during the examination; among all patients undergoing CT, the authors reported that at least three scans were obtained in 30% of patients, more than five scans in 7%, and nine or more scans in 4%. These findings are paralleled by our data showing that the average number of scans per single CT examination was 2.4 for abdominal/pelvic, 1.9 for chest, and 1.3 for head/neck examinations. The published estimates of typical effective doses in CT procedures are usually reported for a single CT scan. The simple recording of a CT examination without the recording of the exact number of scans per single examination may lead to a severe underestimation of the contribution of CT to both effective and organ doses. For example, with an average of 2.4 scans for abdominal/pelvic CT examinations, the estimated lifetime cancer mortality risks attributable to the radiation exposure from a typical abdominal/pelvic CT examination rises to 0.05% in an adult. Although this figure still represents a small increase in cancer mortality over the specific background rate of hemodialysis patient, this finding may be viewed in the light of the elevated number of dialysis patients, which are >300,000 in the United States.14
As for nuclear medicine procedures, the administered radiopharmaceutical activity is the single most relevant predictor of absorbed dose (although the distribution and elimination kinetics of the radiopharmaceutical play a role). The recording of individual administered activities allows us to obtain more accurate estimates of nuclear medicine contribution to the patient effective dose than the simple attribution of a typical effective dose from published studies.
For nephrologists, the association between cancer and chronic kidney disease has been recognized for a long time.15,16 Chronic kidney disease is a risk factor for cancer, because of several not definitely understood mechanisms. The first is the carcinogenesis related to medications administered for treating the renal disease: glomerulonephritides and vasculitides require citotoxic therapy such as cyclophosphamide and azathioprine, which are known to induce bladder, kidney,17 skin cancer, or lymphomas.18 The second is the carcinogenesis related to underlying renal disease: acquired polycystic kidney disease is associated with increased risk of renal cell carcinoma.19 The third is the carcinogenesis caused by impaired immunity such as lymphocytes disregulation20 and DNA damage.21 Therefore, we can hypothesize that the ionizing radiations, which are recognized to induce DNA and RNA alterations and to reduce the capacity of repair of the molecules, would not be only a risk factor but also a concomitant cause of cancer in our population. The excess mortality cancer risk began at a GFR of 55 ml/min per 1.73 m2 and increases of about 30% for every 10 ml/min decrease of GFR, with the greatest risk for GFR <40 ml/min.22,23 Patients with ESRD have fourfold higher risk of cancer compared with the general population, but the risk is different according to the renal replacement therapy: it is increased of 1 to 1.5 times during dialysis and 2.5 to 5 times after kidney transplantation.6,7 Finally, it was shown that more than 5 years of dialysis before transplantation is also associated with increased cancer risk.6 Moreover, by atomic bomb survivor data, we can argue a 14% per Sv increased risk of noncancer disease mortality, such as heart, brain, digestive, and respiratory diseases,24 which are much more difficult to study in our population but which are also at increased prevalence for causes not completely understood.
Dialysis patients receive a higher radiation dose than other chronically ill patients. Stein et al.2 reported a mean total CED after 3 years of 12.3 mSv for 1711 patients with hydrocephalus, 21.7 mSv for 3220 patients with pulmonary thromboembolic disease, 18.7 mSv for 5855 patients with renal colic, and 14 mSv for 11,072 patients with cardiac disease. Kroecker et al.25 reported a mean total CED of 14.3 mSv after 5 years for 371 patients with a diagnosis of Crohn's disease. Chen et al.26 reported a mean total CED over 3 years of 23.1 mSv for 90,121 patients who underwent more than one cardiac imaging procedure. These values must be compared with our estimate of mean total CED over 3 years of 55.7 mSv in 106 dialysis patient or to the corresponding estimate of 34.2 mSv reported by Kinsella et al.5 in 100 dialysis patient for a median of 3.4 years of follow-up.
Altogether, these findings emphasize the need to begin tracking at least the CT-related exposure, as recently suggested by the American College of Radiology,27 to develop and increment alternative strategies to reduce patient-specific radiation burden. The most effective way to reduce the population dose from CT is simply to decrease the number of CT studies that are prescribed. Although the retrospective nature of this study does not allow us to draw conclusive inferences about the percentage of CT studies that could have been avoided, the significant number of examinations that resulted in non-notable findings or in negative results points toward the need of a more stringent process of justification of CT referral. As institutions begin to implement radiation reduction and exposure tracking programs, special attention should be paid not only to individuals but also to cohorts, such as the hemodialysis patients in this study. This will also aid in incrementing the awareness of the medical community (including radiologists and emergency room physicians) of the much higher radiation burden associate with CT examinations in comparison with other radiologic procedures.28
To our knowledge, there is only one published study on the CEDs that maintenance hemodialysis patients accrue over time.5 Notwithstanding a shorter period of follow-up (281 patient-years) in our study compared with the one reported by Kinsella et al. (345 patient-years) and a lower number of total radiologic procedures registered (1303 in our study versus 2575 reported by Kinsella et al.), the median estimated total CED was higher (27.3 versus 21.7 mSv), and even more relevant, the median annual CED was almost doubled in our study (11.7 versus 6.9 mSv). This is mainly because of the severe underestimation in the study of Kinsella et al. of the contribution of CT exposure. On average, in the study of Kinsella et al., a CT study contributed to an average 7.4 mSv to the CED compared with a corresponding figure of 18 mSv in our study. This underestimation is most likely attributable to the nonconsideration of the number of scans in a single CT study, which, as previously mentioned, is a crucial factor in determining the total examination dose. To a lesser extent, the underestimation may also be attributable to the absence of the contribution of nuclear medicine procedures. It should be underlined that the effective doses calculated for abdominal/pelvic CT examinations using organ doses and International Commission on Radiologic Protection 103 tissue-weighting factors were less than International Commission on Radiologic Protection 60 values.29
The results of this study should be interpreted in the context of several limitations. First and most important, it was conducted in a single center; the pattern of use of radiation-related procedures and the resulting patient exposure are highly variable depending on both available technologies and clinical practices. Moreover, we only registered total CEDs during the study period on dialysis and as performed at our medical center for dialysis; this by definition underestimates the radiation exposure of the subjects. Second, the inclusion in the sample population of patients already diagnosed with cancer could be criticized because these subjects may inflate the CED because of therapeutic imaging/monitoring. The main analyses were thus repeated in the subset of 86 noncancer patients with similar conclusions. This finding can be partly explained by the consideration that dialyzed patients are already exposed to a considerable amount of imaging procedures because of their multiple comorbid conditions, and their likelihood of being imaged is not further increased when diagnosis of cancer is made. Third, we did not use measures of radiation dose that are specific to the subject we studied, but instead, we relied on estimates of effective doses or organ doses (for CT procedures), which are neither precisely measured nor subject specific. This limitation was partly compensated in our study by careful recording of the number and location of scans in individual CT examinations, by the estimation of organ dose for CT examinations derived from the institutional acquisition protocols, by the use of an anthropomorphic phantom and Monte Carlo–based dosimetry calculations, and by the estimation of effective dose from nuclear medicine procedures obtained from individual administered activities. Finally, our characterization of the effective dose in the subgroup of subjects represents an application of this quantity beyond its formal definition.
In conclusion, the dialyzed patients received high ionizing radiation doses, because of several comorbidities requiring several diagnostic and therapeutic radiologic procedures. This may be translated to an excess of cancer risk, mainly among young subjects that will receive renal transplantation.
CONCISE METHODS
Data Sources and Study Population
We conducted a retrospective study of period prevalent maintenance hemodialysis patients attending a single university-based dialysis center between June 30, 2007 and June 30, 2010. The center's medical imaging equipment included two magnetic resonance units, three CT scanners, and two pieces of angiography equipment in the radiology department, one positron emission tomography and three single-photon emission tomography scanners in the Nuclear Medicine Department, and two pieces of dedicated cardiac angiography equipment in the cardiology department. Since the first kidney transplant was performed at Novara University Hospital in October 1998, >500 transplants have been carried out. Patients with <6 months of follow-up were excluded. Comorbidities such as mellitus diabetes, neoplasia, and ischemic heart disease (myocardial infarction or angina) were obtained by reviewing medical notes, clinical summaries, and patient interviews. Details of all radiologic procedures performed on patients in the cohort during the study period were obtained from the Radiology Information System present in our institution. For CT procedures, the numbers of scans in each patient and in each anatomic region were obtained by examining individual examinations in the Picture Archiving and communication system of the Hospital Radiology Department.
Estimates of Radiation Doses
To evaluate the radiation exposure for each imaging procedure, we obtained estimates of typical effective doses (assessed in millisieverts). The effective dose is calculated by weighting the concentrations of energy deposited in each organ from a radiation exposure with parameters that reflect the type of radiation and the potential for radiation-related mutagenic changes in each organ in a reference subject.30 It is used for dose distributions that are not homogeneous (which is always the case with CT and nuclear medicine) and is designed to be proportional to a generic estimate of the overall harm to a patient caused by the radiation exposure. Thus, it allows for useful population-level comparisons across different types of radiation exposures31 but provides only an approximate estimate of the true risk. For risk estimation, the organ dose is the preferred quantity.32 Indeed, it is possible to estimate the cancer risks associated with the radiation exposure from any given CT examination by estimating the organ doses involved and applying organ-specific cancer incidence or mortality data that were derived from studies of atomic bomb survivors.12
For common radiology procedures, we relied primarily on data summarized in a recent review.33 For instances in which this source was insufficient, we obtained estimates from other published sources or extrapolated from data reported in similar procedures.34 For all examinations, CTs with and without contrast were counted as two scans, and the radiation dose was doubled. CT with contrast could be counted as having multiple series within the scan to account for multiphasic imaging.32,35
We estimated effective doses and organ doses for abdominal/pelvic CT using the most complete computational method based on Monte Carlo simulations36,37 that explicitly uses tissue weighing coefficients as specified by International Commission on Radiologic Protection 103. CT scans were performed with a 64-row multidetector CT (Lightspeed VCT; GE, Milwaukee, WI) using both z-axis and angular tube current modulation. The institutional examination protocol parameters used to scan the phantom are summarized in Table 6. An average adult was simulated with an anthropomorphic phantom (Rando, The Phantom Laboratory, Salem, NY). Automatic exposure-activated scans follow a lateral topogram acquisition. The tube current values for all of the acquired scans were recorded after extracting the information from the DICOM header of the stored images by means of an automatic routine.38 These values were multiplied by the rotation time used to obtain the milliampere time second values. The volumetric CT dose index provided by the equipment user interface was recorded for every exposure condition. The accuracy of these provided values was verified by using values measured during the routine quality controls, finding a maximum difference of 5%. The patient dose was evaluated in terms of effective dose and organ doses. The software ImPACT CT PATIENT DOSE CALCULATOR v1.02 (ImPACT, London, UK) was used after a modification of the calculation procedure to consider the actual distribution of organ doses consequent to the tube current modulation.38 Briefly a Microsoft Excel macro was developed that considers the milliampere current values for every slab of thickness 1 cm of the scanned volume; it calculates the relative effective dose contribution and adds all of the contributions to determine the total effective dose.
Table 6.
Protocol parameters used to scan the phantom in abdominal/pelvic CT examinations
| Examination | Scan Range | kV | Noise Index | Slice Thickness (mm) | Rotation Time (seconds) | Pitch | Beam Collimation (mm) | FOV (cm) |
|---|---|---|---|---|---|---|---|---|
| 1. Routine abdomen-pelvis | Diaphragmatic cupola to pubic symphysis | 120 | 12 | 2.5 | 0.6 | 0.984 | 40 | 36 |
| 2. Suspected aneurysm or dissection | Diaphragmatic cupola to pubic symphysis | 120 | 16 | 1.25 | 0.5 | 0.984 | 40 | 36 |
| 3. Routine abdomen | Diaphragmatic cupola to iliac wings | 120 | 12 | 2.5 | 0.6 | 0.984 | 40 | 36 |
To err on the side of underestimating exposure, a value of 14.0 mSv was assigned to each complete abdomen-pelvis scan irrespective of the acquisition protocol adopted (routine or vascular). This is equivalent to the average value reported by Mettler et al.33 in a review of >20 publications on dosimetry in abdominal CT. On the other hand, a value of 6.2 mSv was assigned to each upper abdomen scan, which is lower than the corresponding estimate of 8 mSv by Mettler et al.33 It is worth noting that Please spell out units of measure when not used with a numeral. International Commission on Radiologic Protection 60 estimation of effective dose in the routine abdominal/pelvic examination provided a value of 18.7 mSv, which differed from corresponding International Commission on Radiologic Protection 103 estimate by +20%, mainly because of the higher weighting factor for gonads. This is in agreement with the existing literature comparing International Commission on Radiologic Protection 60 and International Commission on Radiologic Protection 103 estimates of effective doses.29
The effective dose from nuclear medicine was estimated as follows39: for the different types of procedures, the individual administered activity of a specific radiopharmaceutical was recorded. Coefficients relating effective doses to administered activity were obtained from the addenda to International Commission on Radiologic Protection Publication 53 (International Commission on Radiologic Protection 80, 1998).40
Procedural frequencies and CEDs of radiation were calculated for the entire study population over the 3-year study period. CED is expressed for each patient as a summation over the study period (total CED [mSv]) and as annual CED (mSv per patient year). Organ doses for patients who underwent abdominal/pelvic CT were estimated for the entire study period. Subjects were categorized according to gender and age at the beginning of the study period (18 to 50, 50 to 70 and >70 years). Procedures were subdivided in conventional diagnostic radiology, CT, nuclear medicine, and interventional radiology, as performed in Mettler et al.33
We calculated population-based rates of effective doses for the overall study population according to the following annual CED categories9: low (≤3 mSv/yr, the background level of radiation from natural sources in the world), moderate (>3 to 20 mSv/yr, the upper annual limit for occupational exposure for at-risk workers in European Union), high (>20 to 50 mSv/yr, the upper annual limit for occupational exposure for at-risk workers in any given year in United States), and very high (>50 mSv/yr).
Statistical Analysis
Data were described using mean and SD or using median and IQR for non-normal distributions. Comparison between groups was performed using Fisher's exact test for categorical variables and Mann-Whitney U test or Kruskal-Wallis for non-normally distributed continuous variables with two groups or more than two groups, respectively. All statistical analyses were carried out with Statistica software, Version 6.0 (Statsoft) using a two-sided type I error rate of 0.05.
DISCLOSURES
None.
Supplementary Material
Acknowledgments
Portions of the data contained within this manuscript have been submitted in abstract form to the 52 Annual Meeting of the Italian Society of Nephrology (SIN), October 6–9ç 2010, Rimini, Italy.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “Radiation Exposure in Dialysis Patients,” on pages 406–407.
REFERENCES
- 1. Mettler FA, Bhargavan M, Faulkner K, Gilley DB, Gray JE, Ibbott GS, Lipoti JA, Mahesh M, McCrohan JL, Stabin MG, Thomadsen BR, Yoshizumi TT: Radiologic and nuclear medicine studies in the United States and worldwide: Frequency, radiation dose, and comparison with other radiation sources-1950–2007. Radiology 253: 520–531, 2009 [DOI] [PubMed] [Google Scholar]
- 2. Stein EG, Haramati LB, Bellin E, Ashton L, Mitsopoulos G, Schoenfeld A, Amis ES: Radiation exposure form medical imaging in patients with chronic and recurrent conditions. J Am Coll Radiol 7: 351–359, 2010 [DOI] [PubMed] [Google Scholar]
- 3. Committee to assess health risks form exposure to low levels of ionizing radiation, National Research Council: Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII-Phase 2, Washington, DC, National Academies Press, 2005 [PubMed] [Google Scholar]
- 4. Berrington de González A, Mahesh M, Kim KP, Bhargavan M, Lewis R, Mettler F, Land C: Projected cancer risks from computed tomographic scans performed in the United States in 2007. J Arch Intern Med 169: 2071–2077, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kinsella SM, Coyle JP, Long EB, McWilliams SR, Maher MM, Clarkson MR, Eustace JA: Maintenance hemodialysis patients have high cumulative radiation exposure. Kidney Int 78: 724–725, 2010 [DOI] [PubMed] [Google Scholar]
- 6. Vajdic CM, McDonald SP, McCredie MR, van Leeuwen MT, Stewart JH, Law M, Chapman JR, Webster AC, Kaldor JM, Grulich AE: Cancer incidence before and after kidney transplantation. JAMA 296: 2823–2831, 2006 [DOI] [PubMed] [Google Scholar]
- 7. Stewart JH, Vajdic CM, van Leeuwen MT, Amin J, Webster AC, Chapman JR, McDonald SP, Grulich AE, McCredie MR: The pattern of excess cancer in dialysis and transplantation. Nephrol Dial Transplant 24: 3225–3231, 2009 [DOI] [PubMed] [Google Scholar]
- 8. Pierce DA, Shimizu Y, Preston DL, Vaeth M, Mabuchi K: Studies of the mortality of atomic bomb survivors. Report 12, part I. Cancer: 1950– 1990. Radiat Res 146: 1–27, 1996 [PubMed] [Google Scholar]
- 9. Fazel R, Krumholtz HM, Wang Y, Ross JS, Chen J, Ting HH, Shah ND, Nasir K, Eistein AJ, Nallamothu BK: Exposure to low-dose ionizing radiation from medical imaging procedures. N Engl J Med 361: 849–857, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Little MP, Wakeford R, Tawn EJ, Bouffler SD, Berrington de Gonzalez A: Risks associated with low doses and low dose rates of ionizing radiation: Why linearity may be (almost) the best we can do. Radiology 251: 6–12, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tubiana M, Feinendegen LE, Yang C, Kaminski JM: The linear no threshold relationship is inconsistent with radiation biologic and experimental data. Radiology 251: 13–22, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Brenner DJ, Elliston CD, Hall EJ, Berdon WE: Estimated risks of radiation-induced fatal cancer from pediatric CT. AJR 176: 289–296, 2001 [DOI] [PubMed] [Google Scholar]
- 13. Mettler FA, Wiest PW, Locken JA, Kelsey CA: CT scanning: Patterns of use and dose. J Radiol Prot 20: 353–359, 2000 [DOI] [PubMed] [Google Scholar]
- 14. Van Dijk C, Ledesma SG, Tetelbaum I: Patient characteristics associated with defects of the peritoneal cavity boundary. Perit Dial Int 25: 367–373, 2005 [PubMed] [Google Scholar]
- 15. Stengel B: Chronic kidney disease and cancer: A troubling connection. J Nephrol 23: 253–262, 2010 [PMC free article] [PubMed] [Google Scholar]
- 16. Mandayam S, Shahinian VB: Are chronic dialysis patients at increased risk for cancer? J Nephrol 21: 166–174, 2008 [PubMed] [Google Scholar]
- 17. Travis LB, Curtis RE, Glimelius B, Holowaty EJ, Van Leeuwen FE, Lynch CF, Hagenbeek A, Stovall M, Banks PM, Adami J, Gospodarowicz MK, Wacholder S, Inskip PD, Tucker MA, Boice JD, Jr: Bladder and kidney cancer following cyclophosphamide therapy for non-Hodgkin's lymphoma. J Natl Cancer Inst 87: 524–530, 1995 [DOI] [PubMed] [Google Scholar]
- 18. Silman AJ, Petrie J, Hazleman, Evans SJ: Lymphoproliferative cancer and other malignancy in patients with rheumatoid artritis treated with azathioprine: A 20 year follow up study. Ann Rheum Dis 47: 988–992, 1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Marple JT, MacDougall M, Chonko AM: Renal cancer complicating acquired cystic kidney disease. J Am Soc Nephrol 4: 1951–1956, 1994 [DOI] [PubMed] [Google Scholar]
- 20. Goldblum SE, Reed WP: Host defence and immunologic alterations associated with chronic hemodialysis. Ann Intern Med 93: 597–613, 1980 [DOI] [PubMed] [Google Scholar]
- 21. Malachi T, Zevin D, Gafter U, et al. : DNA repair and recovery of RNA synthesis in uremic patients. Kidney Int 44: 385–389, 1993 [DOI] [PubMed] [Google Scholar]
- 22. Fried LF, Katz R, Sarnak MJ, Shlipak MG, Chaves PH, Jenny NS, Stehman-Breen C, Gillen D, Bleyer AJ, Hirsch C, Siscovick D, Newman AB: Kidney function as a predictor of noncardiovascualar mortality J Am Soc Nephrol 16: 3728–3735, 2005 [DOI] [PubMed] [Google Scholar]
- 23. Wong G, Hayen A, Chapman JR, Webster AC, Wang JJ, Mitchell P, Craig JC: Association of CKD and cancer risk in older people. J Am Soc Nephrol 20: 1341–1350, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Preston DL, Shimizu Y, Pierce DA, Suyama A, Mabuchi K: Studies of mortality of atomic bomb survivors. Report 13: Solid cancer and noncancer disease mortality 1950–1997. Radiat Res 160: 381–407, 2003 [DOI] [PubMed] [Google Scholar]
- 25. Kroeker KI, Lam S, Birchall I, Fedorak RN: Patients with IBD are exposed to high levels of ionizing radiation through CT scan diagnostic imaging: A five-year study. J Clin Gastroenterol 2010, epub ahead of print [DOI] [PubMed] [Google Scholar]
- 26. Chen J, Einstein AJ, Fazel R, Krumholz HM, Wang Y, Ross JS, Ting HH, Shah ND, Nasir K, Nallamothu BK: Cumulative exposure to ionizing radiation from diagnostic and therapeutic cardiac imaging procedures: A population-based analysis. J Am Coll Cardiol 56: 702–711, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Amis ES, Jr, Butler PF, Applegate KE, Birnbaum SB, Brateman LF, Hevezi JM, Mettler FA, Morin RL, Pentecost MJ, Smith GG, Strauss KJ, Zeman RK: American College of Radiology white paper on radiation dose in medicine. J Am Coll Radiol 4: 272–284, 2007 [DOI] [PubMed] [Google Scholar]
- 28. Lee CI, Haims AH, Monico EP, Brin JS, Forman HP: Diagnostic CT scans: Assessment of patient, physician, and radiologist awareness of radiation doses and possible risks Radiology 231: 393–398, 2004 [DOI] [PubMed] [Google Scholar]
- 29. Christner JA, Kofler JM, McCollough CH: Estimating effective dose for CT using dose-length product compared with using organ doses: Consequences of adopting International Commission on Radiological Protection publication 103 or dual-energy scanning. AJR 194: 881–889, 2010 [DOI] [PubMed] [Google Scholar]
- 30. Martin CJ: The application of effective dose to medical exposures. Radiat Prot Dosimetry 128: 1–4, 2008 [DOI] [PubMed] [Google Scholar]
- 31. The 2007 recommendations of the International Commission on Radiological Protection: ICRP publication 103. Ann ICRP 37: 1–332, 2007 [DOI] [PubMed] [Google Scholar]
- 32. Brenner DJ, Hall EJ, Phil D: Computed tomography: An increasing source of radiation exposure. N Engl J Med 357: 2277–2284, 2007 [DOI] [PubMed] [Google Scholar]
- 33. Mettler FA, Huda W, Yoshizumi TT, Mahesh M: Effective doses in radiology and diagnostic nuclear medicine: A catalog. Radiology 248: 254–263, 2008 [DOI] [PubMed] [Google Scholar]
- 34. Hart D, Wall BF: Effective Radiation Exposure of the UK Population from Medical and Dental X-Ray Examinations, Oxon, UK, NRPN-W4, 2002 [Google Scholar]
- 35. Smith-Bindman R, Lipson J, Marcus R, Kim KP, Mahesh M, Gould R, Berrington de González A, Miglioretti DL: Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med 169: 2078–2086, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Jones DG, Shrimpton PC: Survey of CT Practice in the UK. Part 3. Normalized Organ Doses Calculated Using Monte Carlo Techniques, Chilton, UK, National Radiological Protection Board, 1991 [Google Scholar]
- 37. Zankl M, Panzer W, Drexler G: The Calculation of Dose from External Photon Exposures Using Reference Human Phantoms and Monte Carlo Methods. Part VI. Organ Doses from Computed Tomographic Examinations, Neuherberg, Germany, Forschungszentrum fur Umwelt und Gesundtheit, Institut fur Strahlenschutz, 1991 [Google Scholar]
- 38. Rampado O, Marchisio F, Izzo A, Garelli E, Bianchi CC, Gandini G, Ropolo R: Effective dose and image quality evaluations of an automatic CT tube current modulation system with an anthropomorphic phantom. Eur Radiol 72: 181–187, 2009 [DOI] [PubMed] [Google Scholar]
- 39. Hart D, Wall BF: A Survey of Nuclear Medicine in the UK in 2003/04, Chilton, UK, Health Protection Agency, 2005 [Google Scholar]
- 40. Radiation dose to patients from radiopharmaceuticals: ICRP publication 80. Ann ICRP 28: 1–130, 1998 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.