Abstract
Study Design
In vitro and in vivo rat tail model to assess effects of torsion on intervertebral disc biomechanics and gene expression.
Objective
Investigate effects of torsion on promoting biosynthesis and producing injury in rat caudal intervertebral discs.
Summary of Background Data
Torsion is an important loading mode in the disc and increased torsional range of motion is associated with clinical symptoms from disc disruption. Altered elastin content is implicated in disc degeneration, but its effects on torsional loading are unknown. Although effects of compression have been studied, the effect of torsion on intervertebral disc gene expression is unknown.
Methods
In vitro biomechanical tests were performed in torsion on rat tail motion segments subjected to 4 treatments: elastase, collagenase, genipin, control. In vivo tests were performed on rats with Ilizarov-type fixators implanted to caudal motion segments with five 90-minute loading groups: 1 Hz cyclic torsion to ±5°, ±15°, and ±30°, static torsion to +30°, and sham. Anulus and nucleus tissues were separately analyzed using qRT-PCR for gene expression of anabolic, catabolic, and proinflammatory cytokine markers.
Results
In vitro tests showed decreased torsional stiffness following elastase treatment and no changes in stiffness with frequency. In vivo tests showed no significant changes in dynamic stiffness with time. Cyclic torsion upregulated elastin expression in the anulus fibrosus. Upregulation of TNF-α and IL-1β was measured at ±30°.
Conclusion
We conclude that strong differences in the disc response to cyclic torsion and compression are apparent with torsion increasing elastin expression and compression resulting in a more substantial increase in disc metabolism in the nucleus pulposus. Results highlight the importance of elastin in torsional loading and suggest that elastin remodels in response to shearing. Torsional loading can cause injury to the disc at excessive amplitudes that are detectable biologically before they are biomechanically.
Keywords: rat tail model, in vivo, animal study, torsion, elastin, proinflammatory cytokines, injury
The causes of intervertebral disc (IVD) degeneration are multifaceted, with contributions from aging, mechanical, genetic, and nutritional factors.1 IVD degeneration is manifested biochemically through a loss of glycosaminoglycans, regional changes in collagen matrix composition2 as well as changes in elastin structure3,4 and content. 5 An increase in expression of proteases and their inhibitors,6,7 including MMP-3, ADAMTS-4, and TIMP-1 as well as cytokines IL-1β8 and TNF-α9 have also been associated with degeneration. Biomechanically, IVD degeneration is characterized by a decrease in intradiscal and osmotic pressure,10,11 an altered range of motion and reduced neutral zone,12-14 and a decrease in creep and creep rate.15,16
Although a combination of loads rather than excess of any one loading mode produces clinical signs of degeneration, 17 it is important to isolate the effects of specific loading modes on IVD responses for a greater understanding of IVD mechanobiology. Many studies of intervertebral compression exist, with fewer studies of torsion in vitro14,18-23, in vivo,24 and in silico.25 Torsion results in interlaminar shears and tension of anulus fibrosus (AF) fibers while axial compression leads to substantially greater nucleus pulposus (NP) pressurization and tensile hoop stresses in the AF. Conflicting results exist on the presence of pressurization during small rotations (<1°),19,20 and it has been postulated26 that an IVD cannot be subjected to pure torsion, since small restoring forces acting through the stretched anulus fibers will slightly compress the disc creating NP pressurization. Studies examining changes in gene expression due to dynamic compression in caudal motion segments have demonstrated an increase in general anabolic and catabolic activity27 with sensitivity to load magnitude, frequency, and duration,28 while static compression has shown a general downregulation of anabolic genes.29,30 Differences between cell responses to compression, which causes mostly nucleus pressurization with some tissue shearing, and torsion, which causes mostly tissue shearing with small amounts of pressurization have not been investigated.
The facet joints of the lumbar spine limits rotation to only ~2° per IVD level in each direction, yet rotations may be as large as 10° per level in the thoracic and cervical regions.31,32 In order to study the effects of larger torsional magnitudes, in vitro human studies remove posterior elements. In vivo rat caudal studies are a useful model, because torsional range of motion is not limited by facet joints, they are easily accessible and they provide a high level of control over mechanical boundary conditions. Torsional failure23 in healthy human lumbar discs in vitro was found at ±10° causing “circumferential splits in the anulus” similar to those in degeneration. This suggests that torsional loading of lumbar IVDs with intact apophyseal joints would not put the IVD at risk of injury.33 However, increased torsional range of motion correlated with clinical symptoms from IVD disruption.34
Torsion subjects AF fibers to tension26 and while the majority of the AF fibrous tissue is collagenous, elastin has been shown to play an important role in tension of tissues such as lungs, skin, and ligaments. Recent studies of elastin in the IVD have found it concentrated along the direction of collagen fibers as well as between lamellae and in cross-bridges across the lamellae.35-37 An increase in elastin has been observed in the inner AF with degeneration, potentially pointing to its function in restoring lamellar structure under radial loads in delamination.5 Biomechanical studies of isolated AF tissues treated with elastase were found to significantly increase deformations within AF lamellae38 and extensibility in radial tension.39 While there is growing knowledge of the biomechanical role of elastin in the IVD, the impact of biomechanics on its gene expression has yet to be explored.
The purpose of this study is to investigate effects of enzymatic digestions on torsional mechanical properties in vitro and the effects of torsion on promoting biosynthesis and producing injury in rat caudal IVDs in vivo. We hypothesize that: (1) torsional mechanical properties will be sensitive to changes in collagen and elastin, while in vivo torsion will upregulate collagen and elastin mRNA expression in the AF; (2) large amplitude torsion will induce injury detectable by increased expression of proinflammatory cytokines and altered biomechanical behaviors with time; and (3) cyclic torsion will promote greater mRNA expression than static torsion.
Materials and Methods
In Vitro Biomechanics
A total of 40 caudal motion segments were harvested from skeletally mature Sprague-Dawley rats obtained from IACUC approved protocols. Three caudal vertebra-disc-vertebra motion segments (c5–6, c7–8, c9–10) were harvested from each rat and the skin and tendons and any remaining soft tissue were removed, wrapped in PBS soaked gauze, immediately following dissection, flash frozen in liquid nitrogen and stored at −80°C. On the day of testing, specimens were thawed at room temperature and potted in 10 mm length 12.5 mm diameter stainless steel tubes (pots), using cyanoacrylate as previously described.40 A custom made clamp insured the motion segments were centered within pots, such that the pots remained parallel, the superior-inferior axis of the disc was aligned with the loading axis and the distance between the IVD and each pot remained constant. The motion segments were randomly assigned to 1 of 4 loading groups (n = 10 per group): control (PBS); collagenase treated, genipin treated, and elastase treated by following a previously described protocol41 where the effects of enzymatic degradation were characterized through protein measurements, and the doses were designed to cause significant biomechanical effects while maintaining structural integrity of the discs. During chemical treatments motion segments were constrained to prevent axial swelling while the samples were soaked for 12 hours. Motion segments were attached with custom grips to AR 2000 Rheometer (TA Instruments, New Castle, DE), and tested in PBS. The testing protocol was angle controlled and consisted of the following stages: (a) tare-compression at 0.15MPa, in vivo resting stress; (b) frequency sweep ranging 0.01 to 10 Hz at 0.15 radians (8.6°); and (c) torque controlled ramp to failure. Custom software in Matlab was used for data analysis.
In Vivo Mechanobiology
Animal Model
As approved by the University IACUC, 50 skeletally mature Sprague-Dawley rats were instrumented with Ilizarov-type devices across caudal disc 8–9 as described previously.30 Each animal was placed under general anesthesia and carbon fiber rings were attached to vertebra c8 and c9, using orthogonal sterile 0.8-mm Kirschner wires (Figure 1). Each surgery was completed in less than 30 minutes and a subcutaneous analgesic (buprenorphine) was administered before surgery as well as 12, 24, and 36 hours after surgery. Mechanical loading protocols were applied to rat tails following a 4-day surgical recovery.
Figure 1.
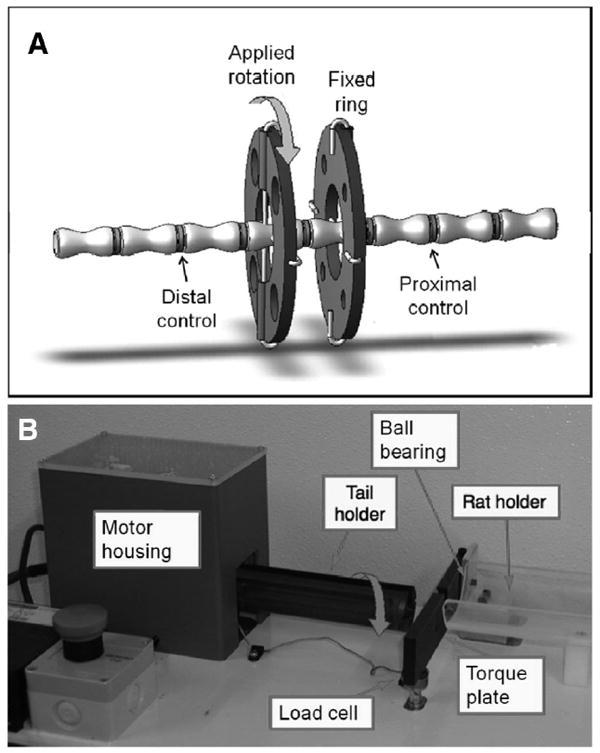
A, In vivo testing involved instrumenting an Ilizarov-type apparatus consisting of carbon fiber rings attached to rat tails, with proximal ring held in torque plate and distal ring rotated in the tail holder. B, Newly designed device to apply torsional loading showing the rat holder, torque plate, and tail holder. The motor housing contains a stepper motor mounted on a linear bearing so that torsional loading could be applied without any axial loading and a motor controller.
Mechanical Loading
Rats were divided into 5 groups (Table 1): based on the in vitro results, cyclic rotation to ±5°, ±15°, and ±30°, static rotation to +30° (static), and sham (carbon fiber rings were implanted but no loading applied). All animals were anesthetized using a gas anesthetic (Isoflurane) for 90 minutes of loading. All animals but shams were loaded using a newly designed torsion device (Figure 1). The proximal ring was attached to a torque plate and the distal ring was attached to the tail holder which was coupled to a stepper motor. Cyclic rotations were applied at 1 Hz sinusoidally, and in the static group a 30° rotation was held for 90 minutes. The rotations of the motor were controlled by custom LabView code, while the resulting torque was recorded by a load cell attached to the torque plate. Torque and angle data were recorded for 10 cycles at a time in 15 minute intervals and these data were further analyzed for stiffness using custom Matlab code.
Table 1.
In Vivo Loading Groups
| Group* | Rotation | Frequency | Duration | n |
|---|---|---|---|---|
| Sham | None | None | 90 min | 10 |
| 1 | ±5° | 1 Hz | 90 min | 10 |
| 2 | ±15° | 1 Hz | 90 min | 10 |
| 3 | ±30° | 1 Hz | 90 min | 10 |
| 4 | +30° | Static | 90 min | 10 |
In vivo loading groups consisted of sham, 3 dynamic loading, and 1 static loading group. All animals were under anesthesia for 90 min, and the tissue was harvested 24 hours post loading.
Gene Expression
Twenty-four hours following the applied loading, animals were killed and the instrumented disc (c8–9) and internal controls (c6–7 and c10–11) were harvested and flash frozen in liquid nitrogen. AF and NP tissue was dissected separately and prepared as described.30 Tissue was pulverized, RNA was extracted by Trizol and purified using an RNA purification kit (Sigma Aldrich), and mRNA was reverse transcribed using Taqman Reverse Transcription Kit (Applied Biosystems, Foster City, CA). Gene expression analysis was performed for 4 anabolic (aggrecan, collagen-I, collagen-II, and elastin), 4 catabolic (MMP3, MMP12, MMP13, and ADAMTS-4) 2 regulatory genes (TIMP-1 and TIMP-3), and 3 proinflammatory cytokines (IL-1β, IL-6, and TNF-α) as well as an endogenous control (18S) using real-time qRT-PCR (GeneAmp 7700 Sequence Detection System, Applied Biosystems). Data were analyzed by the comparative Ct method30 with 18S RNA as endogenous control (ΔCt) and relative levels of genes in instrumented discs normalized to internal controls (ΔΔCt). The 2ˆ-ΔΔCt values were calculated, linearized, and then averaged in each group.
In order to compare the differences in gene expression due to compression and torsion, the genes which were analyzed in torsion but not in a previous dynamic compression study27 were now investigated. Quality of mRNA from that previous study was assessed with nanodrop and its integrity insured by Bioanalyzer Agilent RNA Chip. Gene expression data from previous analyses was used as well as mRNA from dynamic compression was analyzed for elastin, MMP12, IL-1β, IL-6, and TNF-α as well as an endogenous control (18S) as described for torsion.
Statistical Analysis
All statistical analyses were performed with StatView (SAS Institute, Cary, NC), with p < 0.05 considered significant. For mechanical data, an analysis of variance with Fisher PLSD was used to determine differences in each group with time, as well as differences between groups. For qRT-PCR data, a Student t test with a hypothesized mean of 1 was first used to find if in the sham group the loaded level (c8–9) differed from the internal control (c6–7 and c10–11). Next, analysis of variance with Fisher PLSD evaluated if loaded groups differed from shams.
Results
In Vitro
A statistically significant decrease in torsional stiffness was found with elastase, whereas collagenase and genipin treatment led to no significant changes in torsional stiffness (Table 2). Frequency sweep showed no changes in stiffness with frequency for any of the test groups. A neutral zone length was calculated to be 12.9° ± 0.5° for rotation in both directions in control specimens, with no significant differences between treatments (Table 2). Ramp to failure tests (Figure 2) indicated that failure occurred at 40° or above for all samples.
Table 2.
In Vitro Dynamic Biomechanical Testing of Motion Segments
| Group† | NZ Length (°) | K* (Nmm/°) |
|---|---|---|
| Control | 12.9 (0.5) | 2.5 (0.6) |
| Collagenase | 12.6 (0.5) | 1.3 (0.3) |
| Genipin | 12.3 (0.7) | 4.0 (1.1) |
| Elastase | 12.7 (0.3) | 0.9 (0.1) |
All tests were conducted at 8.6° at frequencies ranging from 0.01 to 10 Hz. Since no effect of frequency was found, only data at 1 Hz are reported. Elastin degradation was found to significantly decrease dynamic stiffness at all frequencies (denoted as bold in the table). Neutral zone length was not found to vary with enzyme treatment, and to measure 12.9° in control samples.
Figure 2.
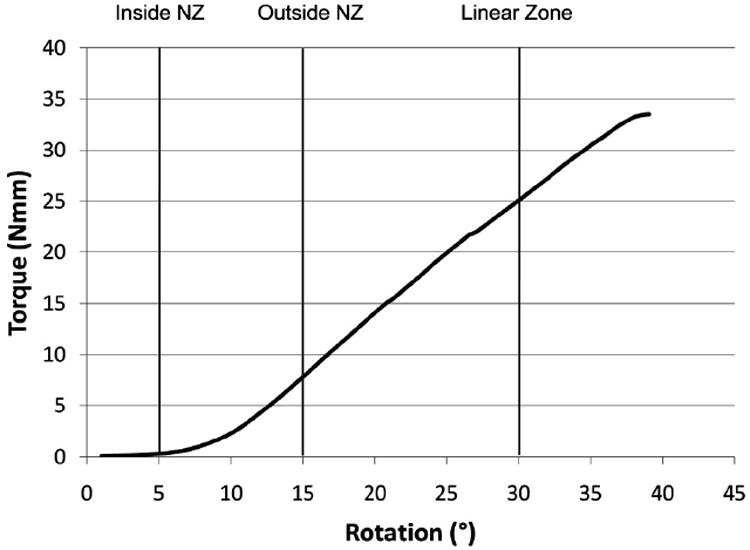
In vitro biomechanical test applied to caudal motion segments consisted of a torque controlled ramp to failure. This typical result demonstrated a transition from the neutral zone to linear region which occurred at 6.5° ± 0.2°. These findings motivated choice of in vivo test conditions to include rotation in the neutral zone (5°), early in the linear zone (15°) and later in the linear zone with possible damage (30°).
In Vivo
Biomechanics
No significant changes in dynamic stiffness were measured with time. A significant increase in stiffness was seen between ±15° and ±30° groups, while the stiffness at ±5° was lower than 1 Nmm and not able to be measured accurately (data not shown) (Figure 3).
Figure 3.
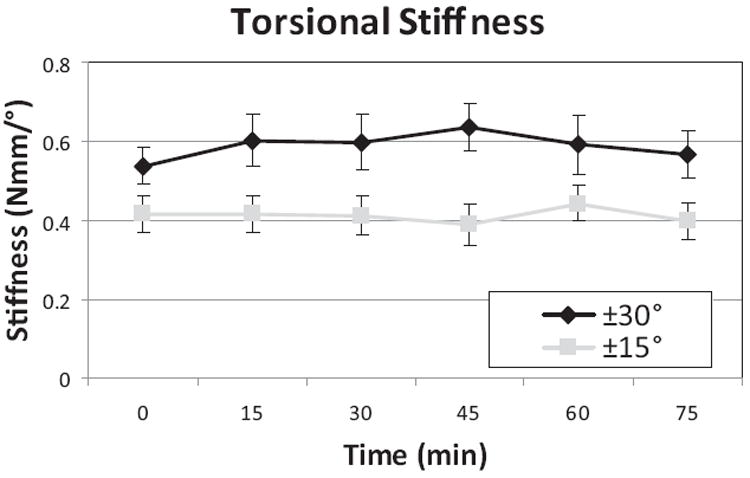
In vivo mechanical measurements were taken every 15 minutes during the 90 minute test. There were no changes in stiffness with time, but stiffness was greater for 30° than 15°, showing nonlinear torsional stiffness behaviors. Stiffness for 5° rotation magnitudes was negligible (not shown) representing loading in the neutral zone.
Gene Expression
Animals in this study tolerated surgery, anesthesia, and the mechanical loading well, maintaining body weight, and showing no outward signs of stress and pin tracks remained very clean with no signs of inflammation. Changes in gene expression with respect to sham as well as sham with respect to the internal control were measured (Figure 4). In the AF, elastin mRNA expression was significantly upregulated in response to cyclic torsion at all amplitudes, while ADAMTS4 was the only catabolic gene significantly upregulated (±30°) with respect to sham. Changes in MMP12 expression in the AF were also detected, but accompanied with changes in sham with respect to the internal control. In the NP, there was a significant upregulation of aggrecan (2-fold) at ±30° as well as a small significant increase (1.3-fold) of TIMP3 also for ±30°. ADAMTS4 was upregulated (3-fold) for the ±15° group but downregulated in the sham compared to internal control. The static condition showed trends of upregulation for similar genes as cyclic ±30° but this was not significant. Proinflammatory cytokines IL-1β and TNF-α showed a significant increase in the AF for ±30°, although for IL-1β this was accompanied by an increase in sham with respect to internal control.
Figure 4.
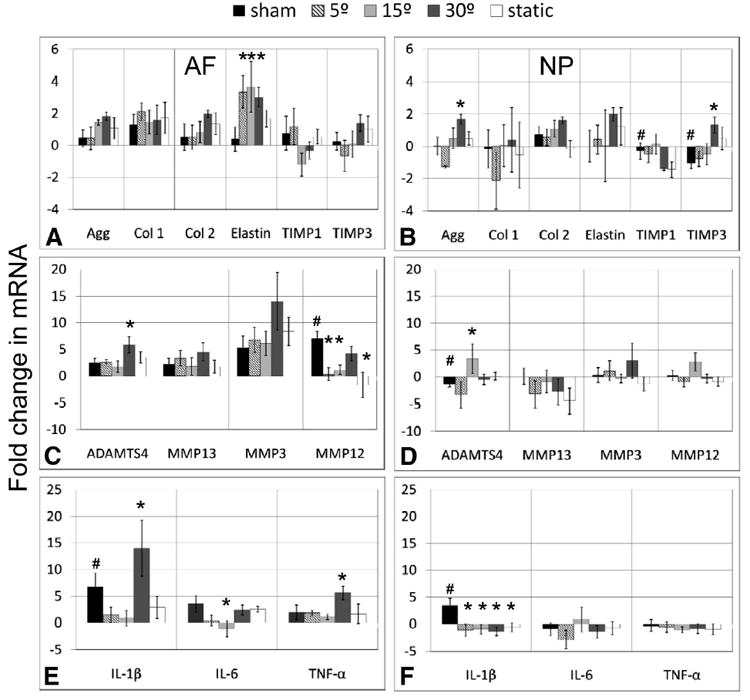
Fold changes in mRNA levels relative to internal control levels (mean ± SEM) for 13 genes where (A, B) are anabolic genes, (C, D) are catabolic genes and (E, F) are proinflammatory cytokines, and (A, C, E) are the measurements from the AF and (B, D, F) from the NP. The mRNA from internal controls was isolated from pooled tissue from proximal and distal internal controls to eliminate potential level effects. *indicates significantly different from sham; #, significantly different from internal controls for sham group only; P < 0.05.
Discussion
In vitro biomechanical testing and in vivo mechanobiology studies were performed on rat caudal IVDs to evaluate proteins most important in determining the biomechanical behaviors and those genes most sensitive to torsional loading. To our knowledge, this is the first study to examine the effect of elastin and collagen on torsional properties of a motion segment or the effects of torsion on gene expression in vivo. Both the in vitro and the in vivo studies point to elastin as the matrix protein most sensitive to torsion. Results showed that 30° of torsion in rat tails did not cause structural changes detectable in our biomechanical testing, yet some increases in proinflammatory cytokines are suggestive of minor injury. Gene expression changes were larger in response to cyclic torsion than static torsion, which is in agreement with previous findings in compression that cyclic loading stimulates gene expression, while static loading or immobilization inhibits it.30
The major finding of this study was the importance of elastin in torsional loading: elastase treatment significantly decreased torsional motion segment stiffness in vitro (Table 2) and cyclic torsion stimulated elastin expression in vivo (Figure 4). Despite the sparseness of elastic fibers in the IVD which make up only 2% of tissue dry weight, previous studies point to elastin as an important matrix protein under AF tissue tension39 and in degeneration. 5 Our findings support current literature on elastin where areas expected to experience greatest tension during torsion are found to be most abundant in elastin: between lamelas and in cross-bridges across the lamellae.36,37,42 No changes were noted in gene expression of MMP12 (elastase) with cyclic torsion, implying that torsion had an impact on anabolic but not catabolic elastin synthesis. This predominantly anabolic response for elastin is consistent with the fact that elastin is one of the most resistant proteins in the body toward proteolysis. 6 However, elastin can also be degraded by other matrix metalloproteases43 including MMP-2, MMP-7, and MMP-9 which were not measured. This study quantified the effects of mechanical loading on gene expression, rather than directly measuring protein level changes since the short duration of the mechanical tests would not be expected to cause protein level changes.
While elastin upregulation in the AF is the major effect of torsion, cyclic compression27 (Table 3) affected a range of anabolic and catabolic genes, most notably aggrecan and ADAMTS4 expression in the NP. Together, these results suggest that torsion and compression elicit distinct cellular responses. Interestingly, some aggrecan and ADAMTS4 upregulation was seen at the higher rotation amplitudes in torsion (although of much smaller amplitudes than in pure compression), which was considered to be associated with increased IVD pressurization at large magnitudes of torsional rotation. It should also be noted that gene expression upregulation that resulted from cyclic compression was usually of a higher magnitude than any of the changes due to torsion, showing compression to have a much greater impact on gene expression in IVDs in general. As the results of this study confirm, torsion affected the AF more than the NP, since at a given rotation the strain experienced by the AF is greater than in the NP due to a greater radial distance. Furthermore, pressurization of the IVD is minimal in torsion, so effects of fluid flow would be expected to be small at most amplitudes and frequencies. In contrast, pressurization is the primary load carriage mechanism of the IVD in compression.
Table 3.
Comparison Between Compression at 1 MPa, 1 Hz, and Torsion at 1 Hz, 30°*
| Genes | AF |
NP |
||
|---|---|---|---|---|
| Torsion | Compression | Torsion | Compression | |
| Agg | 1.8 (±0.2) | 7.1 (±2.8) | 1.7 (±0.3) | 9.8 (±4.0) |
| Col 1 | 1.5 (±0.9) | 3.9 (±2.2) | 0.4 (±1.9) | −0.5 (±7.0) |
| Col 2 | 2.0 (±0.2) | 5.1 (±2.6) | 1.6 (±0.2) | 5.3 (±3.0) |
| Elastin | 3.0 (±0.6) | 5.5 (±1.1)‡ | 2.0 (±0.4) | −0.4 (±1.6)‡ |
| ADAMTS4 | 5.9 (±1.4) | 20.5 (±6.1) | −0.5 (±0.9)§ | 11.7 (±3.6) |
| MMP13 | 4.5 (±1.6) | 8.5 (±3.1)§ | −2.8 (±2.1) | 38.0 (±10.7) |
| MMP3 | 14.0 (±4.8) | 65.5 (±27.0)† | 3.0 (±3.1) | 23.0 (±9.8) |
| MMP12 | 4.2 (±1.3)§ | 7.9 (±4.7)‡ | −0.4 (±0.7) | 6.1 (±0.6)‡ |
| TIMP1 | −0.3 (±0.5) | 14.7 (±2.9) | −1.5 (±0.1)§ | 17.6 (±3.0) |
| TIMP3 | 1.4 (±0.5) | 4.1 (±1.8) | 1.3 (±0.5)§ | 3.1 (±1.2) |
| IL-1β | 14.0 (±3.9)†§ | 15.5 (±9.8)‡ | −1.3 (±0.7)§ | 17.8 (±6.8)‡ |
| IL-6 | 2.4 (±0.9) | 5.0 (±3.4)‡ | −1.3 (±1.0) | 1.8 (±0.4)‡ |
| TNF-α | 5.6 (±1.0) | 8.7 (±3.3)‡ | −0.8 (±0.7) | 3.8 (±1.9)‡ |
All loading lasted 1.5 hours and the tissue was harvested 24-hour post loading.
Different from sham (denoted as bold in the table).
New analysis on prior compression specimens.
Sham significantly different from internal controls.
Contrary to our hypothesis, we found that 30° rotation did not cause structural changes detectable with our biomechanical measurements, although some increases in proinflammatory cytokines were noted in the AF suggesting minor injury may have occurred that was detectable biologically before it was biomechanically. While there were changes in IL-1β expression in both the AF and NP in the cyclic torsion groups, these findings should be considered cautiously with the knowledge they were accompanied by an increase in sham IL-1β expression compared to internal control which may also suggest some effects of surgery. IL-6, IL-1β, and TNF-α were detected in herniated discs,44 whereas other studies found an increase in IL-6 expression only, with no evidence of IL-1β or TNF-α in painful and herniated lumbar discs compared to control.45,46 IL-1β was also found to stimulate matrix degradation in intact human IVD tissue, while TNF-α did not have any measurable effect on matrix degradation.47 Together, we conclude that large amplitudes of torsion are capable of injury to the AF, that the injury is detectable biologically through expression of proinflammatory cytokines before substantial biomechanical changes occur, and that it is possible that cyclic torsion may be capable of regulating proinflammatory cytokine responses in the IVD.
This study was performed on rat caudal IVDs and must be cautiously interpreted in the context with humans. Caudal IVDs have many similarities with lumbar IVDs and are often considered very good models,10,48 yet biomechanical differences exist with larger ranges of motion measured for caudal than for lumbar IVDs.49 Espinoza et al report a higher apparent torsional modulus for lumbar discs compared to caudal, with similarities in the linear zone and differences at the low load neutral zone.50 It is reasonable to expect that the general mechanobiology differences between compression and torsion would be similar across species while magnitudes of biomechanical parameters including range of motion would differ.
Load amplitude and frequency can have effects on biomechanical and biologic responses of the IVD. An in vitro frequency sweep was carried out at 0.01 to 10 Hz, thought to cover a wide range of environmental loading conditions experienced by the spine. No frequency dependence was found on motion segment stiffness in rat caudal discs indicating that torsional amplitude is more important than frequency in determining torsional biomechanical behaviors and perhaps also mechanobiological responses. This is in agreement with the literature, where torsional stiffness was found to increase only slightly with frequency between 0.001 and 1 Hz.51 The choice of rotation angles for the in vivo study of ±5°, ±15°, and ±30° was based on the in vitro ramp to failure (Figure 2) to include rotations that occurred within the neutral zone, early in the linear zone, and late in the linear zone with potential for damage, respectively. While some variations were found in rat tail torsion behaviors particularly for failure, no control specimens failed at rotation amplitudes less than 40°.
We conclude that IVD cells in vivo have distinct responses to torsion and compression. Cyclic torsion stimulates elastin expression with small effects on other genes while cyclic compression results in a more general increase in metabolic response. Results also highlight the importance of elastin in torsional dynamic loading with in vivo findings suggesting that elastin remodels more than collagen in response to torsion and in vitro findings showing that elastin plays an important role in torsional biomechanics. Increased torsional amplitudes caused no structural changes detectable biomechanically, yet some increases in proinflammatory cytokines were present, suggesting minor injury was detectable biologically before mechanically. These mechanobiology findings help elucidate the role of torsional loading in biosynthesis and the relationship of important structural proteins in torsional IVD biomechanics. Results provide baseline information important for tissue engineering approaches as well as improved understanding of healthy and damaging loading on IVDs.
Key Points.
Biomechanically, torsional stiffness of rat caudal intervertebral discs was significantly decreased by elastase treatments.
Cyclic torsional loading in vivo resulted in a significant upregulation of elastin in the AF.
In the context of previous literature, cyclic torsion (which predominantly produces cell stretch and limited pressurization) and compression (which predominantly produces substantial cell pressurization) have distinct effects on intervertebral disc gene expression with torsion upregulating specific target genes including elastin and compression inducing a much greater increase in disc cell metabolic response.
Small increases in aggrecan were found in the NP region of discs loaded under large magnitudes of torsion suggesting increased pressurization at high amplitudes of torsional loading.
Torsional loading on rat caudal discs up to 30° did not affect in vivo and in vitro biomechanical behaviors, suggesting there was no significant injury. However, some upregulation ofmRNAfor TNF-α and IL-1β at ±30° suggest there may be minor injury without major structural disruption.
Acknowledgments
The authors thank for the helpful discussions with Dr. Peter Roughley and the technical assistance of Emir Heco in the design, construction, and evaluation of the loading device.
No funds were received in support of this work. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
The efforts of Dr. Lai were provided through a Graduate Education Attachment Grant from The Hong Kong Polytechnic University.
References
- 1.Hadjipavlou AG, Tzermiadianos MN, Bogduk N, et al. The pathophysiology of disc degeneration: a critical review. J Bone Joint Surg Br. 2008;90:1261–70. doi: 10.1302/0301-620X.90B10.20910. [DOI] [PubMed] [Google Scholar]
- 2.Antoniou J, Steffen T, Nelson F, et al. The human lumbar intervertebral disc: evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J Clin Invest. 1996;98:996–1003. doi: 10.1172/JCI118884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Johnson EF, Berryman H, Mitchell R, et al. Elastic fibres in the anulus fibrosus of the adult human lumbar intervertebral disc: a preliminary report. J Anat. 1985;143:57–63. [PMC free article] [PubMed] [Google Scholar]
- 4.Johnson EF, Chetty K, Moore IM, et al. The distribution and arrangement of elastic fibres in the intervertebral disc of the adult human. J Anat. 1982;135:301–9. [PMC free article] [PubMed] [Google Scholar]
- 5.Cloyd JM, Elliott DM. Elastin content correlates with human disc degeneration in the anulus fibrosus and nucleus pulposus. Spine. 2007;32:1826–31. doi: 10.1097/BRS.0b013e3181132a9d. [DOI] [PubMed] [Google Scholar]
- 6.Roughley PJ. Biology of intervertebral disc aging and degeneration: involvement of the extracellular matrix. Spine. 2004;29:2691–9. doi: 10.1097/01.brs.0000146101.53784.b1. [DOI] [PubMed] [Google Scholar]
- 7.Roberts S, Caterson B, Menage J, et al. Matrix metalloproteinases and aggrecanase: their role in disorders of the human intervertebral disc. Spine. 2000;25:3005–13. doi: 10.1097/00007632-200012010-00007. [DOI] [PubMed] [Google Scholar]
- 8.Le Maitre CL, Freemont AJ, Hoyland JA. The role of interleukin-1 in the pathogenesis of human intervertebral disc degeneration. Arthritis Res Ther. 2005;7:R732–45. doi: 10.1186/ar1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Weiler C, Nerlich AG, Bachmeier BE, et al. Expression and distribution of tumor necrosis factor alpha in human lumbar intervertebral discs: a study in surgical specimen and autopsy controls. Spine. 2005;30:44–53. doi: 10.1097/01.brs.0000149186.63457.20. discussion 54. [DOI] [PubMed] [Google Scholar]
- 10.Urban JP, McMullin JF. Swelling pressure of the lumbar intervertebral discs: influence of age, spinal level, composition, and degeneration. Spine. 1988;13:179–87. doi: 10.1097/00007632-198802000-00009. [DOI] [PubMed] [Google Scholar]
- 11.Nachemson A. Lumbar intradiscal pressure. Experimental studies on postmortem material. Acta Orthop Scand Suppl. 1960;43:1–104. doi: 10.3109/ort.1960.31.suppl-43.01. [DOI] [PubMed] [Google Scholar]
- 12.Mimura M, Panjabi MM, Oxland TR, et al. Disc degeneration affects the multidirectional flexibility of the lumbar spine. Spine. 1994;19:1371–80. doi: 10.1097/00007632-199406000-00011. [DOI] [PubMed] [Google Scholar]
- 13.Tanaka N, An HS, Lim TH, et al. The relationship between disc degeneration and flexibility of the lumbar spine. Spine. 2001;1:47–56. doi: 10.1016/s1529-9430(01)00006-7. [DOI] [PubMed] [Google Scholar]
- 14.Krismer M, Haid C, Behensky H, et al. Motion in lumbar functional spine units during side bending and axial rotation moments depending on the degree of degeneration. Spine. 2000;25:2020–7. doi: 10.1097/00007632-200008150-00004. [DOI] [PubMed] [Google Scholar]
- 15.Kazarian LE. Creep characteristics of the human spinal column. Orthop Clin North Am. 1975;6:3–18. [PubMed] [Google Scholar]
- 16.Koeller W, Muehlhaus S, Meier W, et al. Biomechanical properties of human intervertebral discs subjected to axial dynamic compression—influence of age and degeneration. J Biomech. 1986;19:807–16. doi: 10.1016/0021-9290(86)90131-4. [DOI] [PubMed] [Google Scholar]
- 17.Adams MA, Hutton WC. Prolapsed intervertebral disc. A hyperflexion injury 1981 Volvo Award in Basic Science. Spine (Phila Pa 1976) 1982;7:184–91. [PubMed] [Google Scholar]
- 18.Schultz A, Warwick D, Berkson M, et al. Mechanical properties of human lumbar spine motion segments—part I: responses in flexion, extension, lateral bending and torsion. J Biomech Eng. 1979;101:46–52. [Google Scholar]
- 19.van Deursen DL, Snijders CJ, Kingma I, et al. In vitro torsion-induced stress distribution changes in porcine intervertebral discs. Spine. 2001;26:2582–6. doi: 10.1097/00007632-200112010-00011. [DOI] [PubMed] [Google Scholar]
- 20.Yantzer BK, Freeman TB, Lee WE, III, et al. Torsion-induced pressure distribution changes in human intervertebral discs: an in vitro study. Spine. 2007;32:881–4. doi: 10.1097/01.brs.0000259838.40738.1f. [DOI] [PubMed] [Google Scholar]
- 21.Latorre A, Albareda J, Castiella T, et al. Experimental model of multidirectional disc hernia in rats. Int Orthop. 1998;22:44–8. doi: 10.1007/s002640050206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Costi JJ, Stokes IA, Gardner-Morse M, et al. Direct measurement of intervertebral disc maximum shear strain in six degrees of freedom: motions that place disc tissue at risk of injury. J Biomech. 2007;40:2457–66. doi: 10.1016/j.jbiomech.2006.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Farfan HF, Cossette JW, Robertson GH, et al. The effects of torsion on the lumbar intervertebral joints: the role of torsion in the production of disc degeneration. J Bone Joint Surg Am. 1970;52:468–97. [PubMed] [Google Scholar]
- 24.Hadjipavlou AG, Simmons JW, Yang JP, et al. Torsional injury resulting in disc degeneration: part I. An in vivo rabbit model. J Spinal Disord. 1998;11:312–7. [PubMed] [Google Scholar]
- 25.Shirazi-Adl A, Ahmed AM, Shrivastava SC. Mechanical response of a lumbar motion segment in axial torque alone and combined with compression. Spine. 1986;11:914–27. doi: 10.1097/00007632-198611000-00012. [DOI] [PubMed] [Google Scholar]
- 26.Hukins DW. Disc structure and function. In: Gosh P, editor. The Biology of the Intervertebral Disc. Boca Raton, FL: CRC Press; 1988. pp. 1–37. [Google Scholar]
- 27.MacLean JJ, Roughley PJ, Monsey RD, et al. In vivo intervertebral disc remodeling: kinetics of mRNA expression in response to a single loading event. J Orthop Res. 2008;26:579–88. doi: 10.1002/jor.20560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.MacLean JJ, Lee CR, Alini M, et al. The effects of short-term load duration on anabolic and catabolic gene expression in the rat tail intervertebral disc. J Orthop Res. 2005;23:1120–7. doi: 10.1016/j.orthres.2005.01.020. [DOI] [PubMed] [Google Scholar]
- 29.Lotz JC, Colliou OK, Chin JR, et al. Compression-induced degeneration of the intervertebral disc: an in vivo mouse model and finite-element study. Spine (Phila Pa 1976) 1998;23:2493–506. doi: 10.1097/00007632-199812010-00004. [DOI] [PubMed] [Google Scholar]
- 30.MacLean JJ, Lee CR, Grad S, et al. Effects of immobilization and dynamic compression on intervertebral disc cell gene expression in vivo. Spine. 2003;28:973–81. doi: 10.1097/01.BRS.0000061985.15849.A9. [DOI] [PubMed] [Google Scholar]
- 31.Fujii R, Sakaura H, Mukai Y, et al. Kinematics of the lumbar spine in trunk rotation: in vivo three-dimensional analysis using magnetic resonance imaging. Eur Spine J. 2007;16:1867–74. doi: 10.1007/s00586-007-0373-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.White AA, Panjabi MM. Clinical Biomechanics of the Spine. Philadelphia, PA: Lippincott; 1990. [Google Scholar]
- 33.Adams MA, Hutton WC. The relevance of torsion to the mechanical derangement of the lumbar spine. Spine. 1981;6:241–8. doi: 10.1097/00007632-198105000-00006. [DOI] [PubMed] [Google Scholar]
- 34.Haughton V. Imaging intervertebral disc degeneration. J Bone Joint Surg Am. 2006;88:15–20. doi: 10.2106/JBJS.F.00010. [DOI] [PubMed] [Google Scholar]
- 35.Smith LJ, Fazzalari NL. Regional variations in the density and arrangement of elastic fibres in the anulus fibrosus of the human lumbar disc. J Anat. 2006;209:359–67. doi: 10.1111/j.1469-7580.2006.00610.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yu J, Winlove PC, Roberts S, et al. Elastic fibre organization in the intervertebral discs of the bovine tail. J Anat. 2002;201:465–75. doi: 10.1046/j.1469-7580.2002.00111.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yu J, Fairbank JC, Roberts S, et al. The elastic fiber network of the anulus fibrosus of the normal and scoliotic human intervertebral disc. Spine (Phila Pa 1976) 2005;30:1815–20. doi: 10.1097/01.brs.0000173899.97415.5b. [DOI] [PubMed] [Google Scholar]
- 38.Michalek AJ, Buckley MR, Bonassar LJ, et al. Measurement of local strains in intervertebral disc anulus fibrosus tissue under dynamic shear: contributions of matrix fiber orientation and elastin content. J Biomech. 2009;42:2279–85. doi: 10.1016/j.jbiomech.2009.06.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Smith LJ, Byers S, Costi JJ, et al. Elastic fibers enhance the mechanical integrity of the human lumbar anulus fibrosus in the radial direction. Ann Biomed Eng. 2008;36:214–23. doi: 10.1007/s10439-007-9421-8. [DOI] [PubMed] [Google Scholar]
- 40.Masuoka K, Michalek AJ, MacLean JJ, et al. Different effects of static versus cyclic compressive loading on rat intervertebral disc height and water loss in vitro. Spine. 2007;32:1974–9. doi: 10.1097/BRS.0b013e318133d591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Barbir A, Michalek A, Abbott A, et al. Effects of enzymatic digestion on compressive properties of rat intervertebral discs. J Biomech. 2010;43:1067–73. doi: 10.1016/j.jbiomech.2009.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schollum ML, Robertson PA, Broom ND. A microstructural investigation of intervertebral disc lamellar connectivity: detailed analysis of the translamellar bridges. J Anat. 2009;214:805–16. doi: 10.1111/j.1469-7580.2009.01076.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Smith LJ, Fazzalari NL. The elastic fibre network of the human lumbar anulus fibrosus: architecture, mechanical function and potential role in the progression of intervertebral disc degeneration. Eur Spine J. 2009;18:439–48. doi: 10.1007/s00586-009-0918-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Takahashi H, Suguro T, Okazima Y, et al. Inflammatory cytokines in the herniated disc of the lumbar spine. Spine (Phila Pa 1976) 1996;21:218–24. doi: 10.1097/00007632-199601150-00011. [DOI] [PubMed] [Google Scholar]
- 45.Burke JG, Watson RW, McCormack D, et al. Intervertebral discs which cause low back pain secrete high levels of proinflammatory mediators. J Bone Joint Surg Br. 2002;84:196–201. doi: 10.1302/0301-620x.84b2.12511. [DOI] [PubMed] [Google Scholar]
- 46.Kang JD, Georgescu HI, McIntyre-Larkin L, et al. Herniated lumbar intervertebral discs spontaneously produce matrix metalloproteinases, nitric oxide, interleukin-6, and prostaglandin E2. Spine (Phila Pa 1976) 1996;21:271–7. doi: 10.1097/00007632-199602010-00003. [DOI] [PubMed] [Google Scholar]
- 47.Hoyland JA, Le Maitre C, Freemont AJ. Investigation of the role of IL-1 and TNF in matrix degradation in the intervertebral disc. Rheumatology. 2008;47:809–14. doi: 10.1093/rheumatology/ken056. [DOI] [PubMed] [Google Scholar]
- 48.Demers CN, Antoniou J, Mwale F. Value and limitations of using the bovine tail as a model for the human lumbar spine. Spine (Phila Pa 1976) 2004;29:2793–9. doi: 10.1097/01.brs.0000147744.74215.b0. [DOI] [PubMed] [Google Scholar]
- 49.Elliott DM, Sarver JJ. Young investigator award winner: validation of the mouse and rat disc as mechanical models of the human lumbar disc. Spine (Phila Pa 1976) 2004;29:713–22. doi: 10.1097/01.brs.0000116982.19331.ea. [DOI] [PubMed] [Google Scholar]
- 50.Espinoza Orías AA, Malhotra NR, Elliott DM. Rat disc torsional mechanics: effect of lumbar and caudal levels and axial compression load. Spine J. 2009;9:204–9. doi: 10.1016/j.spinee.2008.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Costi JJ, Stokes IA, Gardner-Morse MG, et al. Frequency-dependent behavior of the intervertebral disc in response to each of six degree of freedom dynamic loading: solid phase and fluid phase contributions. Spine (Phila Pa 1976) 2008;33:1731–8. doi: 10.1097/BRS.0b013e31817bb116. [DOI] [PMC free article] [PubMed] [Google Scholar]


