Abstract
Background
Plasma exchange may be effective adjunctive treatment for renal vasculitis. We performed a systematic review and meta-analysis of randomized control trials of plasma exchange for renal vasculitis.
Study Design
Systematic review and meta-analysis of manuscripts identified from electronic databases, bibliographies, and studies identified by experts. Data was abstracted in parallel by two reviewers.
Setting & Population
Adults with idiopathic renal vasculitis or rapidly progressive glomerulonephritis
Selection Criteria for Studies
Randomized controlled trials that compared standard care with standard care plus adjuvant plasma exchange in adult patients with either renal vasculitis or idiopathic rapidly progressive glomerulonephritis.
Intervention
Adjuvant plasma exchange
Outcome
Composite of end-stage renal disease or death.
Results
We identified 9 trials including 387 patients. In a fixed-effects model the pooled relative risk of end-stage renal disease or death was 0.80 for patients treated with adjunctive plasma exchange compared to standard care alone (95% confidence interval 0.65 to 0.99; p=0.04). No significant heterogeneity was detected (p=0.5; I2=0%). The effect of plasma exchange did not differ significantly across the range of baseline serum creatinine values (p=0.7) or number of plasma exchange treatments (p=0.8). The relative risk for end-stage renal disease was 0.64 (95% confidence interval 0.47 to 0.88; p=0.006) while the relative risk for death alone was 1.01 (95% confidence interval 0.71 to 1.4; p=0.9).
Limitations
Although the primary result was statistically significant, there is insufficient statistical information to reliable determine if plasma exchange reduces the composite of end-stage renal disease or death.
Conclusions
Plasma exchange may reduce the composite endpoint of end-stage renal disease or death in renal vasculitis. Further trials are required given the limited data available.
Keywords: vasculitis, glomerulonephritis, plasma exchange, meta-analysis
Introduction
Wegener’s granulomatosis and microscopic polyangiitis are autoimmune diseases often associated with the presence of circulating anti-neutrophil cytoplasm antibodies (ANCA) and are a common cause of rapidly progressive glomerulonephritis (RPGN) 1. The majority of cases of RPGN have a pauci-immune pattern on immunoflourescence and have circulating ANCA, representing a form of ANCA associated vasculitis (AAV). The introduction of immunosuppression and glucocorticoids has improved outcomes 2, 3 but AAV still has high rates of end-stage renal disease (ESRD) and mortality, particularly in those with glomerulonephritis and low kidney function at presentation 4, 5.
Plasma exchange (PLEX) is a method of removing circulating plasma constituents (e.g. immunoglobulins) and may be an effective treatment for inflammatory autoimmune diseases mediated by auto-antibodies 6. PLEX was proposed as a treatment for severe forms of idiopathic RPGN, the majority of which were due to AAV, based on early anecdotal reports 7, 8. The biologic plausibility of PLEX in AAV has been advanced by discovery of circulating ANCA and their putative pathogenic role 9, 10. Given AAV remains complicated by unacceptably high ESRD and death rates, the addition of PLEX to the standard of care has been studied in several clinical trials.
We systematically reviewed available literature for randomized control trials (RCTs) examining the effect of PLEX in idiopathic RPGN and AAV with renal involvement to understand the effect of adding PLEX to standard therapies on the outcomes of ESRD and death.
Methods
Search Strategy
We searched Medline, EMBASE and the Cochrane Controlled Clinical Trials Register (all to August 2009) using the OVID search engine and two comprehensive search themes which we combined with the Boolean operator “and”. The first theme used terms for vasculitis or glomerulonephritis while the second used terms for plasma exchange. The results were filtered for randomized controlled trials 11.
We identified additional citations from reference lists of review articles, conference proceedings of the American Society of Nephrology (2003 to 2008) and through experts.
Study Selection and Data Abstraction
Two individuals (MW and FC) independently evaluated articles for eligibility in a two stages. First, all identified abstracts were reviewed. Those that met the inclusion criteria, or for which there was uncertainty, were selected for full text review in the second stage. In the second stage full text articles were evaluated for inclusion criteria. Inclusion criteria consisted of: 1) study design (RCT); 2) study population (patients with AAV with renal involvement or RPGN of which >75% were reported pauci-immune or idiopathic); and 3) the outcome (trials that report mortality or renal function parameters). Studies were eligible for inclusion whether published in full or abstract form and irrespective of language.
The same reviewers extracted data in duplicate from all eligible studies to a standardized form. Disagreements were resolved by consensus with a third author (DJ). Extracted data included study design, details of the treatment protocol, and baseline demographic, clinical and laboratory data. The methodological quality of the included studies was assessed using the criteria suggested by Jadad et al (i.e. method of randomization, blinding, use of placebo, reporting of losses to follow-up) 12. Additionally, we abstracted data on allocation concealment, intention to treat analysis and comment on important baseline differences between groups.
Study Outcomes
The primary outcome was the composite of ESRD (defined as dialysis dependency or renal transplantation) or death. A composite outcome was chosen to account for the potential of death to act as a competing endpoint for ESRD (i.e. patients that die cannot develop ESRD and a therapy that results in excess death may therefore appear superior in terms of preventing ESRD). Conversely, follow-up censored at the occurrence of ESRD or other renal parameters may result in differential follow-up. A therapy that results in reduced ESRD may therefore underestimate the effects of the therapy on death because of more complete follow-up. We also assessed the number of adverse events.
Quantitative Data Synthesis
We used the Maentel-Haenszel fixed-effects model to estimate the pooled relative risk with 95% confidence intervals for study outcomes using data from all eligible RCTs 11. We performed sensitivity analyses using the DerSimonian and Laird random effect model 13. The presence of heterogeneity across studies was evaluated using the I2 and Q statistics 14 and we performed meta-regression to test potential explanations of heterogeneity. We tested for the presence of publication bias using funnel plots, Begg’s and Egger’s tests. All statistical analyses were performed using Stata ver 10 (Statcorp, College Station, TX).
To assess the reliability of the statistical inferences, from the pooled analysis on our primary outcome (ESRD or death), we calculated the optimal information size (i.e. the required meta-analysis sample size) and applied methods adapted from formal interim monitoring boundaries for cumulative meta-analysis 15. For the optimal information size, we assumed a moderate relative risk reduction of 25% as clinically important and a 38% event rate in the control arm (a conservative estimate from recent trials). We allowed for 30% heterogeneity and required 90% power and a maximum 5% false positive rate. If our meta-analysis did not surpass the optimal information size, we adjusted the thresholds for statistical significance using Lan-DeMets monitoring boundaries for the cumulative meta-analysis Z-statistic 15, 16. If the Z-statistic did not cross the monitoring boundaries, we added a hypothetical trial that demonstrated a 25% relative risk reduction to the current meta-analysis to determine how much further evidence is required before subsequent meta-analysis results are unlikely to be falsely positive. The hypothetical trial’s size was increased until the cumulative Z-statistic crossed the monitoring boundaries.
Results
Identification of Studies
Progress through stages of the systematic review is summarized in Figure 1. A total of 613 unique citations were identified by our search strategy, of which, 579 were excluded on initial screening. Of the 34 studies that underwent further review, 7 met our eligibility criteria 17-23. We found two additional eligible RCTs from abstract screening and contacting experts; one was available in full text 24 and one as abstract only with updated details from the author 25 but was subsequently published in full 26.
Figure 1.
Literature search and selection.
Study Characteristics
A total of 387 patients were included in 9 RCTS, of whom 201were treated with PLEX and 186 were treated without PLEX (Table 1). One study is a patient level meta-analysis of only the patients with glomerulonephritis from two trials conducted at the same centres 19, 27, 28. This study presents data as a single large study and is treated as one study in our analysis. The mean age of patients ranged from 41 to 67 years old of whom the majority were male (64 to 79%). Follow-up ranged from 6 to 127 months. Mean baseline serum creatinine measurements ranged from 3.6 to 13.5 mg/dL in the PLEX group and 3.2 to 13.1 mg/dL in the no PLEX group. The vast majority of patients had either AAV or idiopathic RPGN (≥87.5% in every trial; >95% of patients overall). Three studies included patients with pulmonary hemorrhage 17, 22, 25.
Table 1.
Characteristics of patients included in trials for meta-analysis.
| Study | Follow-Up (months) | N | Age (mean years) | Female (%) | Baseline Creatinine (mg/dL) | Dialysis at Entry (%) | Lung Hem | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLEX | Ctr | PLEX | Ctr | PLEX | Ctr | PLEX | Ctr | PLEX | Ctr | |||
| Rifle (1980) | 22 | 6 | 8 | 41 | 52 | 50 | 25 | 10.1 | 12.9 | 67 | 88 | No |
| Mauri (1985) | NR | 12 | 10 | NR | NR | NR | NR | 13.5 | 13.1 | 50 | 50 | NR |
| Glockner (1988) | 6 | 16 | 15 | 56 | 47 | 38 | 20 | 6.6 | 6.8 | 50 | 27 | NR |
| Pusey (1991) | 58 | 25 | 23 | 52 | 51 | 36 | 39 | 9.0 | 7.2 | 44 | 34 | Yes |
| Cole (1992) | 12 | 16 | 16 | NR | NR | NR | NR | 7.2 | 9.0 | 25 | 43 | NR |
| Guillevin (1997) | 12 | 19 | 13 | 47 | 62 | 47 | 38 | 5.0 | 3.2 | 32 | 15 | NR |
| Szpirt (1999) | 60 | 16 | 16 | 58 | 56 | 25 | 19 | 3.6 | 3.8 | 13 | 25 | Yes |
| Zauner (2002) | 127 | 21 | 18 | 55 | 56 | 29 | 22 | NR | NR | NR | NR | Yes |
| Jayne (2007) | 12 | 70 | 67 | 67 | 66 | 41 | 36 | 8.5 | 8.1 | 67 | 71 | No |
PLEX=plasma exchange group; Ctr=control group; Lung Hem=patients with lung hemorrhage included; NR = not reported NOTE: multiple serum creatinine by 88.4 to obtain values in μmol/L
Seven RCTs used a cyclophosphamide based treatment regimen in all patients while one trial included no cyclophosphamide 23 and one study treated only some patients with cyclophosphamide 19. Four trials 19, 21–23 used intravenous methylprednisolone in both PLEX and no PLEX groups while one study 20 used intravenous methylprednisolone only in the no PLEX group. One study utilized adjunctive anticoagulation in both study groups 21. One study utilized azathioprine in addition to cyclophosphamide 17. PLEX prescriptions varied between trials (Table 2). Two trials specified PLEX by centrifugation, two by filter separation, two allowed either according to local practice, and three did not report modality. PLEX treatments ranged between 4 and 28 over 1 to 8 weeks. Albumin was the most common replacement fluid (6 trials) while fresh frozen plasma was the primary replacement fluid in one trial 22 and two trials did not specify replacement fluid. The prescribed volume of replacement was variably calculated as absolute volume, percent of plasma volume or volume per unit body weight (Table 2).
Table 2.
Characteristics of treatments in trials for meta-analysis.
| Study | PLEX Method | Number of PLEX | Volume/ PLEX | Weeks of PLEX | CYC | IV MeP |
|---|---|---|---|---|---|---|
| Rifle (1980) | NR | Up to 28 | 1.5 plasma volumes | Up to 8 | Yes | Yes |
| Mauri (1985) | Either | 6 | 3.5 L | 2 | Yes | No |
| Glockner (1988) | Filter | ≥9 | 50 ml/kg | 4 | Yes | No |
| Pusey (1991) | Centrifuge | 9 | 4 L | 1 to 5 | Yes | No |
| Cole (1992) | Centrifuge | ≥10 | 1 plasma volume | 2.25 | No | Yes |
| Guillevin (1997) | Either | 9 to 12 | 60 ml/kg | 3 | Some | Yes |
| Szpirt (1999) | Filter | 6 to 12 | 4 L | 2 | Yes | No |
| Zauner (2002) | NR | 3 to 12 | 40 ml/kg | NR | Yes | Yes |
| Jayne (2007) | Either | 7 | 60 ml/kg | 2 | Yes | Non-PLEX Only |
PLEX=plasma exchange; CYC=cyclophosphamide; IV MeP=intravenous methylprednisolone; NR = not reported
Risk of Bias
Only one RCT included greater than 100 patients 20. Trial quality was limited (see Supplementary Table S1 [provided as online supplementary material]). Unsurprisingly given the nature of the intervention, no trials were blinded or utilized placebo/sham procedures. Allocation concealment was not described in any of the trials. There were small but potentially meaningful differences between groups in studies. For example, the proportion of patients on dialysis at randomization in each group differed in the trials by Glockner et al (PLEX group 50% vs control group 27%), Cole et al (PLEX group 25% vs control group 44%), and Guillevin et al (PLEX group 35% vs control group 15%) although these differences were less pronounced over the trials taken as a whole (PLEX groups 44% vs control groups 46%). Baseline serum creatinine levels were similar between PLEX and control groups in all trials except Guillevin et al (PLEX group 5.0 vs control group 3.2 mg/dL) although the stability or rate of change of these values is not clear in any trial.
Quantitative Data Synthesis
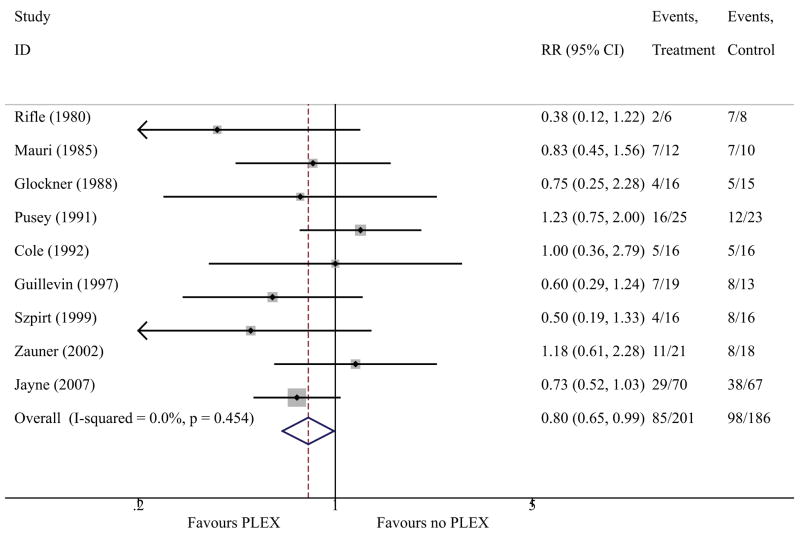
All nine trials were included in quantitative analyses. Overall there were 183 events. No individual study showed a significant difference between the PLEX and no PLEX groups with respect to the primary outcome (the composite of ESRD or death). The pooled relative risk of ESRD or death for PLEX treated compared to no PLEX patients was 0.80 (95% CI 0.65 to 0.99; p=0.04) (Figure 2). There was minimal between trial heterogeneity (I2 0.0%; Q statistic p-value 0.45).
Figure 2.
Forest plot of the effects of adjunctive plasma exchange (PLEX) on the composite endpoint of end-stage renal disease or death in patients with ANCA associated vasculitis.
Figure 3 presents the analysis of death as an endpoint and demonstrates a RR of 1.0 for PLEX compared to control (95% CI 0.71 to 1.42; p=0.9) with minimal heterogeneity between trials (p=0.4; I2=3.9%). Figure 4 presents the analysis of ESRD as an endpoint and demonstrates a RR of 0.64 for PLEX compared to control (95% CI 0.47 to 0.88; p=0.007) with minimal heterogeneity (p=0.5; I2=0%).
Figure 3.
Forest plot of the effects of adjunctive plasma exchange (PLEX) on the endpoint of death in patients with ANCA associated vasculitis.
Figure 4.
Forest plot of the effects of adjunctive plasma exchange (PLEX) on the endpoint of end-stage renal disease in patients with ANCA associated vasculitis.
PLEX had the largest effect in the studies with the lowest baseline serum creatinine (Guillevin and Szpirt). However, meta-regression using mean baseline serum creatinine as the independent variable had no significant association with the primary composite outcome (p=0.7), death (p=0.3) or ESRD (p=0.8). Similarly, the minimum number of PLEX treatments was not associated with the effect of PLEX (p=0.8). A sensitivity analysis omitting the study by Jayne and colleagues due to differences in intravenous glucocorticoid treatment did not significantly alter the point estimate of the effect (RR 0.84 compared to 0.80) although the result was no longer significant (p=0.2). Random effects models made little difference in the effect estimates but marginally widened the confidence intervals for each outcome. Using a random effects model, the pooled estimate for the composite endpoint of ESRD or death was 0.82 (95% CI 0.67 to 1.01), for death it was 0.97 (95% CI 0.67 to 1.42) and for ESRD it was 0.67 (95% CI 0.49 to 0.91). Similarly, sensitivity analyses in which cases that were unlikely AAV or studies that contained cases that were definitely not AAV, made no material difference to the estimated effect of PLEX on the composite endpoint of ESRD or death (RR 0.81; 95% CI 0.66 to 1.00). Little data was available on adverse event rates between treatment arms. There were no gross differences in adverse event rates or severe infection rates in the studies that presented data (Table 3).
Table 3.
Summary of adverse events reported.
| Study | Adverse Events n/N (%) | Infections n/N (%) | ||
|---|---|---|---|---|
| PLEX | Ctr | PLEX | Ctr | |
| Rifle (1980) | 1/6 (17) | 0/8 (0) | 1/6 (17) | 0/8 (0) |
| Mauri (1985) | NR | NR | NR | NR |
| Glockner (1988) | 5/16 (31) | 5/15 (33) | 4/16 (25) | 4/15 (27) |
| Pusey (1991) | NR | NR | 5/25 | 3/23 |
| Cole (1992) | NR | NR | 4/16 (25) | 2/16 (13) |
| Guillevin (1997) | NR | NR | NR | NR |
| Szpirt (1999) | 2/16 (13) | 2/16 (13) | 2/16 (13) | 1/16 (6) |
| Zauner (2002) | NR | NR | NR | NR |
| Jayne (2007) | 35/70 (50) | 32/67 (48) | 31/70 (44) | 30/67 (45) |
NR = not reported
There was no evidence of publication bias using either Egger’s test (p=0.7) or Begg’s test for asymmetry (p=0.1) although there were few trials from which to draw conclusions.
Reliability and Conclusiveness of Results
Although the primary result of this meta-analysis was statistically significant result, the calculated optimal information size suggests at least 1478 patients are required to be confident that PLEX provides at least a 25% relative risk reduction. The monitoring boundaries have not been crossed indicating that the cumulative evidence is unreliable and inconclusive (i.e., there is inadequate information to conclude that PLEX effectively reduces ESRD or death despite the p-value below the conventional limits of significance). We calculated that the addition of a hypothetical trial that randomizes only 500 patients and demonstrates at least a 25% relative risk reduction to the current meta-analysis, would provide sufficient information according to the stopping rules to have confidence that PLEX is of at least moderate benefit in AAV (Figure 5).
Figure 5.

Results of a cumulative meta-analysis after the addition of a hypothetical trial, including 500 patients and demonstrating a 25% relative risk reduction, to the current meta-analysis results. The intersection of the Moderate Evidence and Cumulative Z-statistic lines demonstrates the number of patients required to achieve moderate evidence for the effect of PLEX.
Discussion
In this meta-analysis of 9 trials including 387 patients we could not demonstrate that there is conclusive evidence that adjunctive PLEX reduces ESRD and death in patients with AAV. Despite statistically significant results suggesting a 20% relative risk reduction, there are too few patients randomized and sensitivity analyses were not sufficiently robust to reliably conclude that PLEX has at least a moderate reduction of the composite endpoint of ESRD or death. Further, whether PLEX affects survival or only the development of ESRD is unclear.
PLEX has a strong rationale to support its use in AAV. There is evidence that suggests ANCA are pathogenic 9, 10. The rapid removal of ANCA may therefore result in rapid control of disease activity and prevent the accumulation of organ damage. PLEX also removes proinflammatory mediators which would augment the clinical effects of autoantibody clearance 6. Although previous studies suggested PLEX may only be efficacious in patients with substantially decreased kidney function 17, we observed no significant decrement in the effect of PLEX across a range of serum creatinine at study entry. Furthermore, although there was no significant difference in the effect of PLEX on the composite endpoint across the spectrum of kidney function, there were trends towards benefit in preventing death in trials enrolling patients with more preserved kidney function and benefit in preventing ESRD in trials enrolling patients with lower kidney function. Differences both within and between trials with respect to the timing of PLEX in relation to the onset of disease may also have resulted in differing results. One would expect PLEX to be most useful when instituted early in patients with severe underlying disease (i.e. before the onset of significant renal scarring). However, discriminating between patients with and without significant scarring is difficult and not documented well in the trials in this meta-analysis.
We noted a large apparent effect of PLEX on ESRD but no apparent effect on mortality. One would assume that a reduction in ESRD would result in a decrease in overall mortality. Patients with ESRD due to AAV may, however, have reasonable survival on dialysis or with transplantation and therefore extend their lives beyond the follow-up duration of most studies. Additionally, some studies likely censored follow-up time at the point of ESRD and therefore lost information on differential survival rates. Alternatively, death due to severe infection rather than ESRD or progressive AAV is common in patients with very low kidney function and PLEX would not be expected to have a major impact on this causal pathway, thus the effects of PLEX on death would therefore be diluted or the effects of PLEX on death may be negligible. However, ESRD is associated with poor health related quality of life and high health care costs and the prevention of ESRD alone is an important advance for patients with AAV.
Few patients with pulmonary hemorrhage were included in the trials. This is likely due to a prevalent belief that PLEX is medically indicated for patients with pulmonary hemorrhage on the basis of observational data and the systematic exclusion of these patients from most trials. Although data on patients with lung hemorrhage was insufficient to perform meaningful subgroup analyses, it is notable that none of the three trials that included these patients made special mention of particularly poor outcomes in patients with lung hemorrhage not treated with PLEX. Further, infection is the most commonly cited cause of death in these patients irrespective of whether PLEX was given 29, 30. Together, these data call to question the utility of PLEX in patients with pulmonary hemorrhage.
Our meta-analysis has several notable strengths. We utilized objective and clinically important endpoints. This is particularly notable given the difficultly in performing RCTs in rare diseases like AAV. Establishing effective therapies is, however, important given the poor outcomes of patients with AAV. Although several RCTs in AAV have demonstrated the non-inferiority of reduced toxicity regimens compared to standard regimens 31–35, there has been little progress in establishing therapies that are more effective than cyclophosphamide and glucocorticoids. It is therefore a major advancement in the field of AAV to demonstrate that PLEX may reduce the occurrence of important clinical endpoints. Furthermore, no other adjuvant treatments or alternate treatment regimens have ever shown trends to improved mortality or ESRD rates in AAV. PLEX therefore represents the best candidate to improve patient-important outcomes in AAV.
Although our meta-analysis is encouraging, it should be interpreted in light of its limitations. The included studies were heterogeneous with respect to the treatment regimens, the patients included and the duration of follow-up. It is reassuring that despite these clinical disparities there was no statistical heterogeneity and results from the random-effect models are similar to the fixed-effect models. Further, sensitivity analyses excluding all patients without confirmed AAV did not demonstrate significant differences from the primary analysis. Also, although our meta-analysis is comprehensive it still does not reach optimal information size. Despite this, PLEX has accumulated more evidence any other therapy in this rare disease and the addition of a trial of 500 patients demonstrating at least a 25% relative risk reduction would reliably confirm a treatment effect of at least moderate size. A trial of this size failing to demonstrate a 25% relative risk reduction would require further trials be undertaken.
We demonstrated that PLEX is a promising therapy and may improve clinically meaningful outcomes in a broad spectrum of patients with AAV. Despite statistically significant results, we also found that according to monitoring boundaries for meta-analyses and the calculated optimal information size, enthusiasm for PLEX in AAV must be tempered. Until further RCT data is available, widespread use of PLEX for patients with renal vasculitis cannot be endorsed due to its expensive, resource intensive and invasive nature. PEXIVAS (ISRCTN number 07757494/Clincaltrials.gov number NCT00987389), a trial underway that is powered to detect a moderate to large treatment effect of PLEX may provide sufficient additional information to make confident statements about the efficacy of PLEX for AAV.
Supplementary Material
Acknowledgments
MW is supported by fellowship grants from the KRESCENT program, Canadian Institutes of Health Research RCT Mentoring Program, and Alberta Heritage Foundation for Medical Research. FC was supported by a fellowship grant from the European Renal Association–European Dialysis and Transplantation Association. KT is supported by a fellowship grant from the Canadian Network and Centre for Trials Internationally. CP is supported by the Imperial College NIHR Comprehensive Biomedical Research Centre.
Role of the Funding Source
No external funding was obtained. All authors had full access to study data and all take full responsibility for the decision to submit for publication.
Footnotes
Disclosures
Michael Walsh has no conflicts of interest to disclose.
Fausta Catapano has no conflicts of interest to disclose.
Wladimir Szpirt has no conflicts of interest to disclose.
Kristian Thorlund has no conflicts of interest to disclose.
Annette Bruchfeld has no conflicts of interest to disclose.
Loic Guillevin has no conflicts of interest to disclose.
Chen Au Peh has no conflicts of interest to disclose.
Peter Merkel has no conflicts of interest to disclose.
Charles Pusey has no conflicts of interest to disclose.
Marion Haubitz has no conflicts of interest to disclose.
David Jayne has no conflicts of interest to disclose.
Author’s Contributions
Michael Walsh contributed to the concept, design, data collection, analysis, interpretation, and manuscript preparation of this study.
Fausta Catapano contributed to the data collection, interpretation and manuscript preparation of this study.
Wladimir Szpirt contributed to the concept, data collection, interpretation, and manuscript preparation of this study.
Kristian Thorlund contributed to the design, analysis, interpretation, and manuscript preparation of this study.
Annette Bruchfeld contributed to the concept, interpretation, and manuscript preparation of this study.
Loic Guillevin contributed to the concept, interpretation, and manuscript preparation of this study.
Chen Au Peh contributed to the concept, interpretation, and manuscript preparation of this study.
Peter Merkel contributed to the concept, interpretation, and manuscript preparation of this study.
Charles Pusey contributed to the concept, interpretation, and manuscript preparation of this study.
Marion Haubitz contributed to the concept, interpretation, and manuscript preparation of this study.
David Jayne contributed to the concept, design, data collection, interpretation, and manuscript preparation of this study.
Ethics Committee Approval
This study dealt with only published aggregate data and was exempt from requiring approval at the local ethics committee.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Reference List
- 1.Jennette JC, Falk RJ, Andrassy K, et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum. 1994;37(2):187–192. doi: 10.1002/art.1780370206. [DOI] [PubMed] [Google Scholar]
- 2.Bosch X, Guilabert A, Espinosa G, Mirapeix E. Treatment of antineutrophil cytoplasmic antibody associated vasculitis: a systematic review. JAMA. 2007;298(6):655–669. doi: 10.1001/jama.298.6.655. [DOI] [PubMed] [Google Scholar]
- 3.Menahem S, Hiremagalur B, Mudge D, Toussaint N, Walters G. The CARI guidelines. Induction and maintenance therapy in ANCA-associated systemic vasculitis. Nephrology (Carlton ) 2008;13 (Suppl 2):S24–S36. doi: 10.1111/j.1440-1797.2008.00995.x. [DOI] [PubMed] [Google Scholar]
- 4.Booth AD, Almond MK, Burns A, et al. Outcome of ANCA-associated renal vasculitis: a 5-year retrospective study. Am J Kidney Dis. 2003;41(4):776–784. doi: 10.1016/s0272-6386(03)00025-8. [DOI] [PubMed] [Google Scholar]
- 5.Slot MC, Tervaert JW, Franssen CF, Stegeman CA. Renal survival and prognostic factors in patients with PR3-ANCA associated vasculitis with renal involvement. Kidney Int. 2003;63(2):670–677. doi: 10.1046/j.1523-1755.2003.00769.x. [DOI] [PubMed] [Google Scholar]
- 6.Wood L, Jacobs P. The effect of serial therapeutic plasmapheresis on platelet count, coagulation factors, plasma immunoglobulin, and complement levels. J Clin Apher. 1986;3(2):124–128. doi: 10.1002/jca.2920030209. [DOI] [PubMed] [Google Scholar]
- 7.D'Amico G, Sinico R, Fornasieri A, et al. Effect of intensive plasma exchange (PE) in rapidly progressive crescentic glomerulonephritis (RPCGN) International Journal of Artificial Organs. 1983;6 (Suppl 1):3–9. [PubMed] [Google Scholar]
- 8.Stevens ME, McConnell M, Bone JM. Aggressive treatment with pulse methylprednisolone or plasma exchange is justified in rapidly progressive glomerulonephritis. Proceedings of the European Dialysis & Transplant Association. 1983;19:724–731. [PubMed] [Google Scholar]
- 9.Falk RJ, Jennette JC. ANCA are pathogenic--oh yes they are! J Am Soc Nephrol. 2002;13(7):1977–1979. doi: 10.1681/ASN.V1371977. [DOI] [PubMed] [Google Scholar]
- 10.Xiao H, Heeringa P, Hu P, et al. Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulonephritis and vasculitis in mice. J Clin Invest. 2002;110(7):955–963. doi: 10.1172/JCI15918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Egger M, Smith GD, Altman D. Systematic Reviews in Health Care: Meta-analysis in context. London: BMJ Publishing Group; 2001. [Google Scholar]
- 12.Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12. doi: 10.1016/0197-2456(95)00134-4. [DOI] [PubMed] [Google Scholar]
- 13.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 14.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–1558. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 15.Pogue JM, Yusuf S. Cumulating evidence from randomized trials: utilizing sequential monitoring boundaries for cumulative meta-analysis. Control Clin Trials. 1997;18(6):580–593. doi: 10.1016/s0197-2456(97)00051-2. [DOI] [PubMed] [Google Scholar]
- 16.Thorlund K, Devereaux PJ, Wetterslev J, et al. Can trial sequential monitoring boundaries reduce spurious inferences from meta-analyses? Int J Epidemiol. 2009;38(1):276–286. doi: 10.1093/ije/dyn179. [DOI] [PubMed] [Google Scholar]
- 17.Pusey CD, Rees AJ, Evans DJ, Peters DK, Lockwood CM. Plasma exchange in focal necrotizing glomerulonephritis without anti-GBM antibodies. Kidney International. 1991;40(4):757–763. doi: 10.1038/ki.1991.272. [DOI] [PubMed] [Google Scholar]
- 18.Glockner WM, Sieberth HG, Wichmann HE, et al. Plasma exchange and immunosuppression in rapidly progressive glomerulonephritis: a controlled, multi-center study. Clinical Nephrology. 1988;29(1):1–8. [PubMed] [Google Scholar]
- 19.Guillevin L, Cevallos R, Durand-Gasselin B, Lhote F, Jarrousse B, Callard P. Treatment of glomerulonephritis in microscopic polyangiitis and Churg-Strauss syndrome. Indications of plasma exchanges, Meta-analysis of 2 randomized studies on 140 patients, 32 with glomerulonephritis. [Review] [17 refs] Annales de Medecine Interne. 1997;148(3):198–204. [PubMed] [Google Scholar]
- 20.Jayne DR, Gaskin G, Rasmussen N, et al. Randomized trial of plasma exchange or high-dosage methylprednisolone as adjunctive therapy for severe renal vasculitis. [see comment] Journal of the American Society of Nephrology. 2007;18(7):2180–2188. doi: 10.1681/ASN.2007010090. [DOI] [PubMed] [Google Scholar]
- 21.Rifle G, Chalopin JM, Zech P, et al. Treatment of idiopathic acute crescentic glomerulonephritis by immunodepression and plasma-exchanges. A prospective randomised study. Proc Eur Dial Transplant Assoc. 1981;18:493–502. [PubMed] [Google Scholar]
- 22.Zauner I, Bach D, Braun N, et al. Predictive value of initial histology and effect of plasmapheresis on long-term prognosis of rapidly progressive glomerulonephritis. American Journal of Kidney Diseases. 2002;39(1):28–35. doi: 10.1053/ajkd.2002.29874. [DOI] [PubMed] [Google Scholar]
- 23.Cole E, Cattran D, Magil A, et al. A prospective randomized trial of plasma exchange as additive therapy in idiopathic crescentic glomerulonephritis. The Canadian Apheresis Study Group. [see comment] American Journal of Kidney Diseases. 1992;20(3):261–269. doi: 10.1016/s0272-6386(12)80699-8. [DOI] [PubMed] [Google Scholar]
- 24.Mauri JM, Gonzales MT, Poveda R, et al. Therapeutic plasma exchange in the treatment of rapidly progressive glomerulonephritis. Plasma Ther Transfus Technol. 1985;6:587–591. [Google Scholar]
- 25.Szpirt W, Rasmussen N, Petersen J. Long term outcome and prognostic factors in randomized study of plasma exchange and cyclosporine A in Wegeners granulomatosis. J Am Soc Nephrol. 1999;10(11):A1. Ref Type: Abstract. [Google Scholar]
- 26.Szpirt WM, Heaf JG, Petersen J. Plasma exchange for induction and cyclosporine A for maintenance of remission in Wegener's granulomatosis--a clinical randomized controlled trial. Nephrol Dial Transplant. 2010 doi: 10.1093/ndt/gfq360. [DOI] [PubMed] [Google Scholar]
- 27.Guillevin L, Jarrousse B, Lok C, et al. Longterm followup after treatment of polyarteritis nodosa and Churg-Strauss angiitis with comparison of steroids, plasma exchange and cyclophosphamide to steroids and plasma exchange. A prospective randomized trial of 71 patients. The Cooperative Study Group for Polyarteritis Nodosa. [see comment] Journal of Rheumatology. 1991;18(4):567–574. [PubMed] [Google Scholar]
- 28.Guillevin L, Lhote F, Cohen P, et al. Corticosteroids plus pulse cyclophosphamide and plasma exchanges versus corticosteroids plus pulse cyclophosphamide alone in the treatment of polyarteritis nodosa and Churg-Strauss syndrome patients with factors predicting poor prognosis. A prospective, randomized trial in sixty-two patients. Arthritis & Rheumatism. 1995;38(11):1638–1645. doi: 10.1002/art.1780381116. [DOI] [PubMed] [Google Scholar]
- 29.Gallagher H, Kwan JT, Jayne DR. Pulmonary renal syndrome: a 4-year, single-center experience. Am J Kidney Dis. 2002;39(1):42–47. doi: 10.1053/ajkd.2002.29876. [DOI] [PubMed] [Google Scholar]
- 30.ter Maaten JC, Franssen CF, Gans RO, van Schijndel RJ, Hoorntje SJ. Respiratory failure in ANCA-associated vasculitis. Chest. 1996;110(2):357–362. doi: 10.1378/chest.110.2.357. [DOI] [PubMed] [Google Scholar]
- 31.DeGroot K, Rasmussen N, Bacon PA, et al. Randomized trial of cyclophosphamide versus methotrexate for induction of remission in early systemic antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis & Rheumatism. 2005;52(8):2461–2469. doi: 10.1002/art.21142. [DOI] [PubMed] [Google Scholar]
- 32.Jayne D, Rasmussen N, Andrassy K, et al. A randomized trial of maintenance therapy for vasculitis associated with antineutrophil cytoplasmic autoantibodies. New England Journal of Medicine. 2003;349(1):36–44. doi: 10.1056/NEJMoa020286. [DOI] [PubMed] [Google Scholar]
- 33.DeGroot K, Harper L, Jayne DR, et al. Pulse versus daily oral cyclophosphamide for induction of remission in antineutrophil cytoplasmic antibody-associated vasculitis: a randomized trial. Ann Intern Med. 2009;150(10):670–680. doi: 10.7326/0003-4819-150-10-200905190-00004. [DOI] [PubMed] [Google Scholar]
- 34.Jones RB, Tervaert JW, Hauser T, et al. Rituximab versus cyclophosphamide in ANCA-associated renal vasculitis. N Engl J Med. 2010;363(3):211–220. doi: 10.1056/NEJMoa0909169. [DOI] [PubMed] [Google Scholar]
- 35.Stone JH, Merkel PA, Spiera R, et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N Engl J Med. 2010;363(3):221–232. doi: 10.1056/NEJMoa0909905. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.