Abstract
Recent advances in nanotechnology offer new hope for disease detection, prevention, and treatment. Nanomedicine is a rapidly evolving field wherein targeted therapeutic approaches using nanotechnology based on the pathophysiology of gastrointestinal diseases are being developed. Nanoparticle vectors capable of delivering drugs specifically and exclusively to regions of the gastrointestinal tract affected by disease for a prolonged period of time are likely to significantly reduce the side effects of existing otherwise effective treatments. This review aims at integrating various applications of the most recently developed nanomaterials that have tremendous potential for the detection and treatment of gastrointestinal diseases.
Keywords: nanoparticles, intestinal tract, diagnosis, therapeutics, nanomaterial, siRNA, gastrointestinal
although nanomaterials (NMs) are widely considered to be an invention of modern science, they actually have a very long history. Nanoparticles (NPs) were empirically used by artisans as far back as the 9th century in Mesopotamia to generate a glittering effect on the surfaces of pots. The first scientific description of nanometer-scale metals was provided by Michael Faraday in his classic paper (25a), but the development of NMs depended mainly on their visualization and the characterization of their physical and chemical properties. Modern instrumental techniques have drastically increased our ability to precisely measure particle size distributions and many other parameters that are correlated with nanoscale objects. For example, techniques such as transmission and scanning electron microscopy and scanning tunneling microscopy have facilitated the direct visualization of individual NPs with atomic accuracy (35, 46). As a result of these advances, scientists have developed exquisite and highly sophisticated methods to generate nanoscale materials with different sizes, compositions, and geometries. These nanoscale materials possess unique and unusual properties and are currently used by multidisciplinary teams of scientists in scientific areas that range from physics and engineering to biochemistry. Consequently, the field of nanomedicine has emerged alongside the development of these materials to harness some of their novel properties. Tremendous progress has been made with respect to the synthesis of a variety of materials that can be used as nanovehicles (carbons, synthetic polymers, polysaccharides, and iron, to name a few) (51, 65) and the techniques used to control the shapes and length scales of NMs. The discovery of quantum effects (size-dependent properties) that produce nanomaterials with specific emissive, absorptive, or scattering spectra has broadened the range of their potential applications in areas such as imaging and diagnostics (10, 11, 33). One of the most attractive applications of NM is as a drug delivery system, in which NMs are used as drug carriers. The potential to target a single cell represents a revolutionary tool for medicine. There are several advantages using NMs: NMs are small in size (1–1,000 nm) with correspondingly large surface area-to-volume ratio; NMs have good stability, robustness, and a long shelf-life compared with molecular carriers; drugs can be loaded into NPs at a high concentration, and the NMs may be made to evade normal digestive processes and thus efficiently deliver drugs to specific sites; the kinetics of drug release can be modulated; chemically tailorable surfaces may be modified with ligands to affect site-specific drug delivery.
Each of these capabilities allows researchers to design and use NM in medicine and gastroenterology to perform diverse functions in bioimaging, diagnostics, and drug delivery that can compete favorably with conventional molecular approaches.
CHARACTERISTICS AND TYPES OF NANOMATERIALS USED IN GASTROENTEROLOGY
The gastrointestinal (GI) tract is an attractive target system for nanotechnology applications (Table 1). It is the site of therapeutic absorption, and the behavior of NMs can be regulated during transit through the digestive tract under conditions of varying pH, transit time, pressure, and bacterial content. The distinct features of each part of the digestive tract introduce many challenges to the application of therapeutics to the gut. NM behavior must be optimized for efficient transit under varying pH, pressure, and enzyme-catalyzed degradation to reach the target site. Engineering a nanocarrier that is robust under the range of conditions presented by the gut represents a challenge in and of itself. The properties of the NMs and the interactions with tissues in the gut depend heavily on size, size distribution, morphology, hydrophilic-hydrophobic balance, and surface functionalization of the NM.
Table 1.
Examples of application of nanotechnology in gastroenterology
| Characteristics and Potentialities | Example of Application in GI (Target, Aim of Study) |
|---|---|
| Polymer micelles/chemically modified drug | |
| √ Loaded molecule control release | √ Liver and jejunum for reduction of Apo B and total cholesterol (chemically modified Apo B siRNA) by intravenous administration (95). |
| √ Gene delivery applications | √ Reduction of HBV DNA in liver by intravenous administration of a chemically engineered hepatitis B virus siRNA (71). |
| √ Low toxicity and antigenicity | √ Micelles (block polyethylene glycol and polyaspartate) loaded with paclitaxel for colon cancer (67). |
| √ Variable stealth according to surface adsorbed molecule. | |
| Liposomes | |
| √ Loaded molecule control release | √ Liver metastasis growth inhibition (loaded with bcl2 siRNA) by intravenous administration (112). |
| √ Gene delivery applications | √ Peritoneal cavity targeting for inhibiting tumor growth and inflammation (loaded IL-12p40 (28) or β-catenin (105). |
| √ Low toxicity and antigenicity | √ Liver cirrhosis remission by encapsulation of hepatocyte growth factor (63) by intravenous administration. |
| √ Variable stealth according to surface adsorbed molecule. | |
| Viral vector | |
| √ No control release | √ Virosomes, presenting hemagglutinin on the surface, to bind and fuse with targeted cells and deliver siRNA (17). |
| √ Gene delivery applications | √ Reduction of DSS induced colitis by intra rectal injection of prohibitin pDNA in inflamed colon (101). |
| √ High toxicity and antigenicity | |
| √ Medium stealth | |
| Polymer NPs | |
| √ Loaded molecule control release | √ Oral delivery of thioketal NPs (sensitive to oxidative agents) loaded with TNF-α siRNA in colonic tissue of mice with DSS-induced colitis (107). |
| √ Gene delivery applications | √ Oral delivery of NPs loaded with TNF-α siRNA encapsulated in a hydrogel (alginate and chitosan) sensitive to colonic bacteria degradation (60). |
| √ Low toxicity and antigenicity | √ Oral delivery of zirconium phosphate NPs loaded with Insulin (21). |
| √ Active cell targeting strategy possible | √ Intestinal immunization using chitosan NPs covered with a high transcytosis peptide (CKS9) for M cells targeting strategy (113). |
| √ Variable stealth according to surface adsorbed molecule. | |
| √ High cell uptake and intracellular targeting | |
GI, gastrointestinal; Apo B, apolipoprotein B; siRNA, small interfering RNA; NPs, nanoparticles; DSS, dextran sodium sulfate.
Characterization of Nanomaterials
NM size.
Size (defined as the diameter of particles over the range from 1 to 1,000 nm) is a key aspect of NMs, since this can influence cellular uptake, physical properties, and interactions with biomolecules. For example, Lamprecht et al. (53) recently characterized the deposition of particles of three sizes, 0.1, 1, and 10 μm, in the gut after oral administration. Interestingly, the authors showed that small particles preferentially deposited in the colon (better bioadhesive properties), with the highest rate of uptake by macrophages in an inflamed colon, relative to the normal intestinal cells. In many cases, the size of a given NM is determined by the procedure used in the synthesis and their chemical composition. For example, 1- to 10-nm particles are typically formed by crystalline iron atoms or micelles of small molecules, whereas larger particles (10–1,000 nm) are typically generated from polymeric materials. In addition to particle size, the composition and surface properties of materials used for micro- or nanoencapsulation can regulate uptake and transport across mucosal barriers and particularly those that pertain to gastroenterology. Importantly, the properties of mucosa and the nature of their interaction with NMs must be carefully considered when developing NMs in gastroenterology. For example, pore size and charge of mucin molecules, which are found along the digestive track (from the stomach to the colon), may vary significantly in each region of the gastric milieu. The mucosa can regulate the interactions between the orally administered NMs and the targeted cells. If the NM is coated with a polymer, the NM size may be modified by other factors, such as the hydrophilic-hydrophobic balance of the polymeric materials. A good solvent for the coated polymer will allow relaxation of the polymer structure to increase the total NM size. These properties define the way in which the NMs may interact with the gastric lining.
The NM size depends on the synthetic process used to prepare the NPs [miniemulsion, sonication, or extrusion (2, 3, 39, 57, 58, 100)]. There are two main classes of NPs: ones that require energy to fragment the bulk materials and others that depend on the nucleation and growth of seeds through chemical processes. During synthesis of liposomes, for example, energy is provided to the system (mechanical, acoustical, or thermal energy)b and this energy determines the size range of the resulting NPs. In general, as more energy is provided to the system, the size distribution narrows and the NPs become smaller. For narrow size distributions to be achieved effectively, the energy introduced into the system (aqueous or organic) must also be homogenously distributed throughout the solution. This condition may be difficult to realize if high-molecular-weight molecules are used to make the particles. For NPs that are formed spontaneously, such as micelles, noble metal, or semiconducting NPs, the size distributions are typically narrower and the size is dictated by nucleation and growth in solution. Thus it is important to consider the intended purpose for the NM when selecting the synthetic process for generating NMs of specific size and size distribution in various applications.
Biomedical applications usually require that the NMs interact with or be taken up by cells; therefore, the particle diameter needs to be much smaller than the size of cells. Lamprecht and colleagues (52) showed that nonphagocytic eukaryotic cells can internalize particles <1 μm in size. Small particles that are 5–110 nm in size are under development as potential carriers of anticancer drugs via intracellular drug delivery. Notably, Lamprecht and colleagues report that particle size does not significantly impact the release kinetics of the drug, but it was beneficial to decrease the “burst effect” of drug release from small NPs. These authors also showed that treatment of a cell culture model for cancer with paclitaxel loaded into NPs produced less cytotoxicity and an efficacy that was 40 times higher than that observed for treatment with the pure drug at the same concentration (52).
NP size is not the only parameter that influences cellular uptake. Particle surface characteristics may be adjusted to optimize biological response. NPs have shown great promise in gastroenterology because their interactions with intestinal epithelial cells, macrophages, immune cells, and M cells are tunable, suggesting their potential as a vehicle for vaccinations.
Surface properties.
In all nanomedicine studies, the major challenge is determining how NMs will interact with the mucosa, tissue, or targeted cells. The first stage of oral drug delivery involves the interactions between the NM surface and the mucosa or GI cells. One such parameter that plays an important role in determining these interactions is the electrostatic surface charge of a NM. The zeta potential (ζ) accurately approximates the charge on a NP and is used to describe cell-NM interactions (22, 24). The charge on a NM depends on the polymer used for the NM matrix or can be modulated by adsorbing specific molecules onto the NM surface. The charge displayed on a NM prevents aggregation. Higher ζ (positive or negative) can produce a stronger electrostatic repulsion between NMs, and thus the NM suspension will become more stable. Digestive diseases, such as ulcerative colitis (UC), Crohn's disease, or cancer, may potentially be treated by an intravenous injection of a NM-encapsulating therapeutic, the surface characteristics of which may strongly affect the therapeutic index. After intravenous administration, NP circulation model studies have shown that the charge and hydrophilic-hydrophobic balance on NMs, but not micelles, regulates the NM interactions with proteins. Proteins adsorb onto the surfaces of NPs to promote opsonization, which leads to aggregation and rapid clearance from the bloodstream (70, 77, 85, 106). Rapid clearance is mediated by splenic filtration and phagocytosis by the mononuclear phagocyte system in the liver. Another important consideration is the role of nonspecific interactions between the biomolecules and the nanomaterials. In many cases, these nonspecific interactions can be minimized when hydrophilic polymers, such as polyethylene glycol, are grafted, conjugated, or absorbed onto the surface of NPs to form a shell. This coating provides steric stabilization and confers “stealth” properties that prevent protein absorption (4, 76). Cell targeting applications require a controlled circulation time for NMs in the bloodstream to allow for efficient interactions with the target tissue. Jung et al. (43) articulated the importance of the ζ on the interactions with the gut mucosa during oral NP delivery. Jani et al. (42) studied the interactions between charged NPs and the intestinal epithelia, demonstrating that the uptake of carboxylated polystyrene NPs was significantly less, especially in M cells, compared with neutral or positively charged polystyrene.
High surface area-to-volume ratio.
Nanomedicine can take advantage of the high surface area-to-volume ratio of NMs and the specificity of the digestive system by engineering diversified interaction sites into the NMs (Fig. 1). Most NMs are spherical (NPs, micelles, or liposomes) and hence have a surface area-to-volume ratio of 3/r, where r is the radius. As r decreases, the surface area-to-volume ratio increases. A particle with a large surface area has more interaction sites available than a particle with a small surface area, and the rate of an interaction at the surface may, therefore, be higher. This has important implications for drug delivery. It should be noted that the inverse relationship between particle size and surface area-to-volume ratio applies to all geometrical shapes and is not restricted to spheres. For example, the main characteristic of nanotubes (cylindrical NMs) is based on the length-to-diameter ratio, which can reach 28,000,000 in some cases. The lightweight and strong carbon nanotubes are used in bone engineering as structural mimics of bone, collagen, or hydroxyapatite.
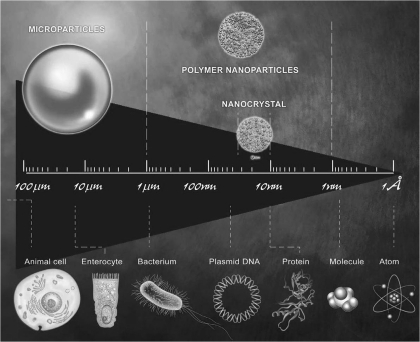
Fig. 1.
Illustration of the difference in surface-to-volume ratio between a microparticle (S1 = 1 mm) and a nanoparticle (S2 = 100 nm).
The dependence of drug delivery rate on particle surface area in nanomedicine applications must be considered. The concern in gastroenterology delivery systems is that the drug may be degraded during transport to the target. The various degradation mechanisms can increase the dosing of the drug and produce unwanted side effects. By their size, physical properties, chemical characteristics, and high surface area-to-volume ratio, NPs may help regulate the pharmacokinetic profile of a drug (36, 94).
Nanomaterial Carriers
The polymers used as particulate vectors may be natural or synthetic, synthesized by standard polymerization chemical methods. In either case, the polymers must be biocompatible, nontoxic, nonimmunogenic, and noncarcinogenic. They must also be (bio)degraded in the body, and their degradation products must be well tolerated and quickly eliminated. Examples of degradable polymers include polyorthoesters, polyanhydrides, polyamides, polyalkylcyanoacrylates, polyesters, lactides/glycolides, polycaprolactones, polyphosphazenes, or pseudo-polyamino acids. Nondegradable polymers are usually composed of silicone, elastomers, polyethylene oxide, polyethylene glycol, or acrylic polymers.
Among the natural polymers, the most commonly used are proteins such as gelatin or albumin, and polysaccharides such as alginate, chitosan, pectin, or amylopectin. Polysaccharides, in particular, have additional advantages in that they are degraded by bacteria and hence can be used as a colonic delivery system (81, 91). The monosaccharide subunit (galactose, glucose, acid derivatives, or N-acetyl) degradation products are easily eliminated or assimilated by the body. A disadvantage of natural polymers is that it is difficult to control their size and, therefore, their average molecular weight (number or mass). Synthetic polymers have been developed to afford this control. The chemical composition of most polymers and copolymers used in particulate vectors is largely modular. Appropriately tuned macromolecular synthetic methods permit regulation of the size and molecular weight distribution. In addition, specific architectures, mechanical properties, viscoelastic properties, or surface energies may be controlled by polymer chemistry methods. Aliphatic polyesters (56, 84, 114) have been widely studied for their biocompatibility and biodegradable properties. Poly(d,l-lactide) (PLA), poly(glycolide) (PGA), and their copolymers degrade in the body to yield lactic acid and glycolic acid, which are natural metabolites. Ultimately, these metabolites are converted into CO2 and H2O. Poly(ϵ-caprolactone), which is more hydrophobic than PLA or PGA, biodegrades slowly in vivo in the presence of lipase. Polyalkylcyanoacrylates can dissolve in biological fluids after hydrolysis of the acrylate function (7).
Nanoparticle Vectors
As shown on Fig. 2, particulate vectors, as they pertain to nanotechnology, refer to the drug carrier system. Particulate vectors are classified according to vector size, targeting mechanism used, and the NM itself: microparticles, micelles vectors, or NPs. Sizes of microparticles and NPs in the range of 1 Å to 1 μm allows for the potential interaction and/or uptake of nano- or microparticles by living unit cells. This ability is first due to the similarity of size between most cells or bacteria and the nano- or microparticles (Fig. 2). The nanocarriers can be particulate dispersions or solid particles in which a drug can be dissolved, entrapped, or attached. There are different types of NP carriers that may be classified on the basis of their constituent elements; these are described below.
Fig. 2.
Illustration of a scale representing the size homology between particles and main units of living identities.
Liposomes (19) are small (nano- or micrometric) spherical vesicles composed of walls that are formed by one or more phospholipids bilayers surrounding a central aqueous phase. They may carry hydrophilic drugs in the central aqueous phase or a lipophilic drug inserted into the phospholipids bilayer. Liposomes can simplify delivery of lipophilic drugs that cannot be stably accommodated for biological release in standard delivery systems. Liposomes have good biocompatibility because the raw materials that compose them are natural phospholipids, sterols, or glycerolipids, and they behave as a modified release system. However, the vesicular structures are chemically unstable (phospholipids are easily oxidized or hydrolyzed) and physically unstable (the micelles may aggregate, fuse, or lose their contents).
Dry emulsions are systems comprising a spherical shell structure with walls composed of surfactant molecules and an oily core in which lipophilic active molecules may be dissolved. Dry emulsions can be prepared by using stabilizers, amphiphilic macromolecules such as proteins, or modified polysaccharides (79). They may be freeze dried to yield a powdered form that is easily reconstituted in water to yield the original oil-in-water emulsion.
Nanocapsules (NCs) or nanospheres (NPhs) are polymeric colloidal structures with diameters between 100 nm and 1 μm, including NPhs and NCs. The polymer networks of the NPhs form spherical matrix structures with polymer shells and oily cores. An active molecule may be incorporated within the polymer network during formation of the NPhs or adsorbed to the surface through hydrophobic, electrostatic, or covalent interactions. The affinity of the NPhs to a cell or tissue type may be enhanced by incorporating tissue- or cell-specific ligands into the NPhs during synthesis. Thus these third-generation vectors may be designed to target cells (9).
NANOPARTICLES AS A DRUG CARRIER: VECTORIZATION AND CHALLENGING CONSIDERATIONS IN THE FIELD OF GASTROENTEROLOGY
The bioavailability of a drug may be optimized by NM encapsulation. The vectorization of millions of molecules of a drug within a single cell or organelle is, without contest, the most powerful treatment possible (1, 110).
Vectorization
The vectorization process consists of formulating an active drug in sufficient quantities to meet the target by using a vector-soluble or water-insoluble inactive therapeutic. Targeted delivery avoids the direct administration of the drug and improves the pharmacokinetics of the active vectorized drug (Fig. 3). The objective of the vectorization is to develop a method of drug administration that improves the bioavailability of an active molecule, for example, extending the half-life of the drug in the body, promoting availability, and, in some cases, facilitating transport of a drug past select biological barriers while protecting the structural integrity of the active functional therapeutic agent (particularly when faced with the many degradation enzymes).
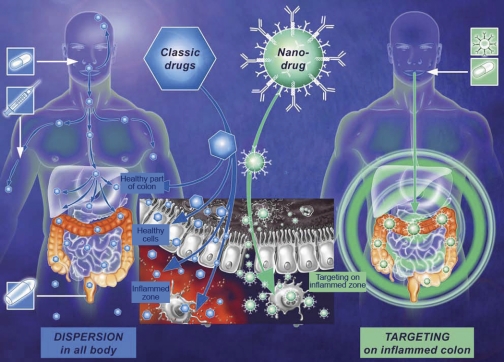
Fig. 3.
Illustration representing the different localization of drug in targeted strategy compared with systemic treatment. For oral intake or intravenous injection of the classical drug, the bioactive component is distributed throughout the body without any distinctions between healthy and inflamed tissue. Enema strategy can only target the distal part of colon. In targeting strategy, nanoparticles (NPs) are covered with an antibody whose ligands are overexpressed in inflamed areas. The NPs accumulate and the drug is released in the specific area.
Indeed, drugs will have a low therapeutic index if they cannot traverse the biological barriers that separate the site of administration from the site of action. Several vectorization methods have been tested, including encapsulation in NPs, which is an important, innovative, and promising technology. As mentioned above, targeted delivery has many advantages. Vectorization of a drug creates a protective coating around the therapeutic molecule to guard against enzymatic and chemical inactivation. Thus the pharmacokinetics of the drug (in terms of dosage and time release) may be optimized. This is particularly useful for drugs that have a short half-life in vivo. The therapeutic efficacy of a bioactive molecule is safeguarded in many treatment regimens by injecting a high drug dose and/or repeating the injection with frequency, both of which can cause undesirable side effects. The gradual release of an active molecule encapsulated within a particle allows reduction of the number of injections and improves the patient's comfort. Vectorization additionally may lead to an optimal pharmacological profile: the variability of concentrations in vivo of an active molecule can be reduced by controlling the drug release profile and maintaining concentrations at an efficient pharmacological level. Controlling the rate of release of active molecules encapsulated within a matrix vector can ensure that the concentration remains constant over a much longer period than is achievable under standard dosing regimens. In nanomedicine, NMs not only decrease the drug concentration, but also permit complete control over the kinetics of drug degradation. Two aspects of drug delivery may, therefore, be optimized: the drug can be introduced in efficient but minimal quantities, and the carrier, usually a polymer, assists in the targeted delivery of the drug.
NM vectors for oral drug administration are designed so that only the matrix, not the drug, is subject to degradation in the digestive tract. The degradation process begins with the mechanical forces imposed by mastication and then proceeds with enzymatic breakdown by the enzymes in the mouth. After oral intake, several different challenges face the NMs.
Barriers to NM Interactions with Cells
Erosive conditions are present throughout the digestive system. In the stomach, the acidic pH (pH = 2–3) can lead to degradation upon contact. Degradation in the small intestine is facilitated by the oxidative and denaturing digestive juices. In the colon, the pH reaches neutrality (pH = 7) and the drug will be exposed to enzymes secreted by bacteria located only in the colon (61). Drugs are degraded not only by bacterial enzymes of the local flora but also by the mechanical pressures applied in the colon where intestinal motility is required. Thus the drug may be specifically released when (time dependence) and where (physical chemistry environment dependence, for example, pH, enzymes, pressure) the matrix is degraded. Because the drug was protected during the degradation process, the positive biological effects of the drug remain once the drug reaches the target site. Delivery is all the more challenging for strategies that target the colon because this is the last part of the digestive system. A number of drug carriers and delivery systems have been investigated for this purpose. Among these, NPs are attractive carriers that have recently shown great promise for promoting drug efficacy. Because water-insoluble drugs pose several challenges with respect to efficient release and bioavailability, NM encapsulation of hydrophobic drugs offers a solution. Bypassing the solubility issues, NM encapsulation shows great promise. For example, strategies designed around the concomitant release of drugs with opposite solubility in water have been studied in the context of lipophilic drug encapsulation (5). In this study (5), the authors synchronized sulpiride (SP) (a low water-soluble drug) and quinidine (Qn) release from hydroxypropylmethylcellulose tablets. They showed that the synchronous release of Qn and SP resulted in the increased bioavailability of SP. Synchronous release efficiency could also be enhanced by targeting NPs to specific cell or tissues. In that case, target-specific ligands were linked to the NP surface.
STRATEGIES USED IN NP DELIVERY IN THE DIGESTIVE SYSTEM
An unmet need in GI disease treatment is the targeted delivery of drugs to the terminal ileum and colon, two sites that are affected by inflammatory diseases and colon cancer. Four strategies for drug delivery systems in nanomedicine may be enumerated on the basis of the relative constant transit time in the digestive tract, the pH, the enzyme environment, and the ambient pressure.
Time-Dependent Strategy
This strategy is based on the transit time in the GI tract. In time-dependent release, the drug is released after the outer layer of the drug delivery system is destroyed. Several properties can be used for this purpose, including the degree of swelling of a polysaccharide matrix or the pH change. Dorkoosh et al. (23) used the change in the enteric coating and the concentration or thickness of the layer to modulate the release time profile. With this technique, one can target any part of the intestine or colon. A similar approach was used to target the colon by Laroui et al. (57) in a study of the behavior of different concentrations of a mixture of two polysaccharides (alginate and chitosan). In this study, a high drug release profile was observed from the NPs in the targeted area. The kinetic of the release was modulated by the osmotic activity of the salt or charged polymers, or the erosion rate of the polymer coating.
pH-Dependent Strategy
The GI tract presents a wide range of pH values: 2–3 in the stomach, 5–6 in the small intestine, and 7 in the colon. NMs that release drugs in a pH-dependent fashion have been commercially developed. Examples include Eudragit poly(meth)acrylate polymers. Those polymers contain acidic or alkaline groups that enable the pH-dependent release of the active ingredient. Polyacrylamide, for example, is a polymer that is highly sensitive to pH. It is stable in acidic pH but degraded in a neutral pH. This characteristic is suitable for drug protection during GI tract transit. Several polysaccharides, such as chitosan, pectin, or alginate, have been developed for delivery purposes by taking advantage of their pH-dependent stability (15, 75, 86). Microspheres can be made to be pH sensitive as well. Lamprecht et al. (55) showed that pH can be used to modify the release of 5-fluorouracil over time using Eudragit P-4581F microspheres. In this study, the authors showed that a relatively weak change in pH (from 6.8 to 7.4) produced a dramatic effect on the kinetics of drug release from the microspheres. Sensitivity to small pH adjustments is relevant to colonic delivery systems. Other strategies call for the encapsulation of a prodrug that is chemically modified to the active drug in a pH-dependent manner, or polysaccharides grafted with a drug for cell targeting. The weak changes in pH convert the prodrug from an inactive form to an active form in the targeted area (44).
Pressure-Dependent Strategy
The strong muscular contractions in the colon serve as a “driving force” to move its contents. Feces must be evacuated, and this organ produces muscular contractions that enable this function. These motions result in intracolonic mechanical pressures that are higher than in other parts of the GI tract. Intestinal pressure-controlled colon delivery capsules (PCDCs) rely on the relatively strong peristaltic waves in the colon for drug release. Such capsules are coated with a water-insoluble polymer, ethyl cellulose (37, 38, 90, 99). After oral administration, PCDCs act as an ethyl cellulose balloon. The suppository base liquefies at body temperature. In the upper GI tract, PCDCs are not directly subjected to the luminal pressures due to the fluid present in the stomach and small intestine. Reabsorption of water in the colon (18), however, increases the viscosity of the luminal contents. As a result, increased intestinal pressures directly affect the system via colonic peristalsis. In response to the increased pressure, PCDCs rupture and release the drug in the colon.
Enzyme-Based Strategy
Enzyme-based strategies depend on the colonic bacteria to degrade a NM and release the drug in the colon. Because bacteria are located mainly in the colon, this strategy is a powerful method for delivering drugs to the colon. Specific enzymes are produced only by colonic bacteria and thus only detectable in that area. That characteristic made enzyme-based strategies very interesting and powerful. In that strategy, Laroui et al. (57) have synthesized polylactic acid NPs loaded with anti-inflammatory tripeptide and succeeded in releasing them in the colon by giving it orally encapsulated in a hydrogel. This hydrogel, made with a specific proportion of alginate and chitosan, is specially degraded by the colonic enzymes. Plenty of polysaccharides can be used since they are specifically degraded by colonic bacteria such as amylase/amylopectin (15) and chitosan (57, 60, 68).
APPLICATION OF NMS IN GASTROENTEROLOGY
Nanomaterials Used as a Theragnostic
Special “theragnostic” NMs are currently being designed with the goal of progressing through preclinical development for cancer imaging and therapy. Theragnostics rely on double encapsulation of both therapeutic and diagnostic molecules, within a single carrier. These NMs are designed to assist in both imaging (diagnosis) and therapeutic applications. Implementation of this concept requires the development of key molecules that can respond to stimuli (biological or chemical) within the area targeted for treatment. Several stimuli may be coupled to the system, for example, inflammatory indicators such as pH, elevation of temperature, hypoxia, or specific binding to an inflammatory ligand. To assist in imaging applications, signaling molecules may be encapsulated or conjugated to the carrier. Polymers may be bonded via noncovalent or covalent interactions directly during the nanocarrier synthesis (104) or after synthesis by surface modification reactions (25). Noncovalent or covalent interactions are selected according to the application and the specificity of the area targeted for treatment. Covalent linkages based on carbonyl (25), amine (66), or silane (96) coupling chemistries allow a wide range of functionalities under various pH or oxidative conditions. Hydrophobic, electrostatic, or hydrogen-bonding interactions present alternatives to covalent linkages, if flexibility under chemical conditions is required (12, 97, 104, 111). Polymers may be selected to include a variety of functional groups and/or hydrophobic sites, leading to a large set of potential linkages between the diagnostic/therapeutic molecule and the nanocarrier. Amide, ester, disulfide, hydrazone, or thioether linkages (103, 108) have been successfully used to enable covalent or hydrophobic interactions (hydrophobic drug loading) or ionic interactions (nucleic acids) (108). As a theragnostic study, Yang et al. (109) demonstrated a promising approach to the treatment of pancreatic cancer. In this study, the author used a urokinase plasminogen activator (uPA) amino-terminal fragment (ATF) peptide as a target ligand with a high binding affinity to uPAR to block the interaction between uPA receptor (uPAR) and its natural ligand, uPA. ATF peptides have been shown to inhibit tumor growth and angiogenesis in several animal tumor models (8, 64). Yang et al. showed that the ATF-coated NPs were efficiently taken up by cancer cells, thereby increasing their tumor-specific accumulation, both for tumor imaging and for the delivery of therapeutic agents.
Tissue Engineering
Nanotechnology may aid in the repair of damaged tissue. Tissue engineering makes use of artificially stimulated cell proliferation by using scaffolds based on suitable materials in conjunction with growth factors. Materials for tissue engineering must contain interconnected pores and require a high surface area for better efficiency. The pores allow for optimal cell culture growth by permitting cell migration, medium diffusion within the biomaterial (diffusion and release of nutriments), and electrical and chemical communication between the cells colonizing the biomaterial (88). Within the GI tract, gastric ulcers can be treated by tissue engineering techniques. Hassani et al. (32) showed that polystyrene microspheres and NPhs adhered to the ulcerated mucosal areas. In this study, the authors showed that this interaction was maximal for small NPs (50 nm), which adhered preferentially to the ulcerated tissue rather than to the healthy tissue. This observation highlights the potential use of NPs as a carrier system for the treatment of gastric ulcers.
Specific Imaging and Therapeutic in Colon Cancer
The small size of NPs endows them with properties that can be very useful in oncology, particularly in imaging. Quantum dots (NPs that display quantum confinement properties, such as size-tunable light emission), when used in conjunction with magnetic resonance imaging, can produce exceptional images of tumor sites. These NPs are much brighter than organic dyes and can be excited by any light source that is blue-shifted relative to the emission spectrum. The fluorescent quantum dots may produce higher-contrast images at a lower cost than organic dyes, which are used as a contrast media. The downside, however, is that quantum dots are usually made of toxic elements. Nanotechnology-based products have already been approved for the treatment of cancer. Approved products include liposomes, such as Doxil, or NPs, such as abraxane. The accumulation of small nanocarriers (10–100 nm) at a tumor site is based on the absence of efficient lymphatic drainage.
Photodynamic therapy involves the illumination, by an external light source, of particles delivered to a specific region of the body. The light is absorbed by the particle, and if the particle is a metal, energy from the light can heat the particle and the surrounding tissue. The light may also be used to produce high-energy reactive oxygen molecules, which may chemically react with and destroy proximal organic molecules. Photodynamic therapy is appealing for several reasons. It does not leave a “toxic trail” of reactive molecules throughout the body (chemotherapy) because it is directed only to the illuminated region where the particles have been delivered. Photodynamic therapy presents a noninvasive therapeutic alternative for treating diseases, growths, and tumors. Kirui et al. (48) recently reported the preparation of multifunctional gold-iron oxide NPs for targeting, imaging, and laser photothermal therapy of cancer cells using laser irradiation at 800 nm. The gold NPs were functionalized with carboxy-terminated phospholipids and conjugated to a single-chain antibody, scFv, which bound to the A33 antigen present in colorectal cancer cells. The A33 antigen is overexpressed on the surface of SW1222 colorectal cancer cells such that the NPs were selectively immobilized on the surfaces of the cancer cells. The NPs were then preferentially taken up by the cancer cells. Upon absorption of 808 nm light, the cancer tissue was selectively destroyed, demonstrating that this strategy is suitable for cancer diagnosis and therapy.
Nanoparticles Used to Deliver Gene Therapy
Currently, a major drawback of gene therapy is the inefficiency of gene transfection. The two main types of vectors used for gene therapy are based on viral or nonviral gene delivery systems. The viral gene delivery system shows a high transfection yield but it has many disadvantages, such as oncogenic effects and immunogenicity. However, cationic polymers, such as chitosan, form complexes with DNA or small interfering RNA (siRNA) and may pose nonviral alternative vectors for gene therapy applications. One major obstacle to DNA and siRNA therapy is the low penetration of naked DNA and siRNA across cell membranes (69). To overcome this problem, several delivery systems have been studied. Recent efforts toward developing tissue-targeted nucleic acid delivery systems based on synthetic reagents have yielded promising results (26, 30, 34, 47, 78, 83, 87, 102). To improve the efficiency of siRNA-loaded polymer NPs, more and more “targeted NPs” are imagined with a surface recovered with a peptide, a chemoattractant agent, or an antibody (Fig. 4) grafted on the polymer matrix to accumulate on a specific area before releasing the active agent. Among drug carriers, NPs, biodegradable or otherwise, have shown an interesting potential for binding and delivering DNA and siRNA (27). Indeed, NPs were found to protect DNA and siRNA against degradation in vitro and in vivo and to significantly enhance their pharmacological activity, not only in cell culture conditions, but also in vivo (27). In the context of inflammatory bowel disease (IBD), a biocompatible system may be most relevant. The biodegradable polymeric envelope may protect and transport the siRNA into the cytosol, thereby allowing the siRNA to be efficiently transfected in vivo (102).
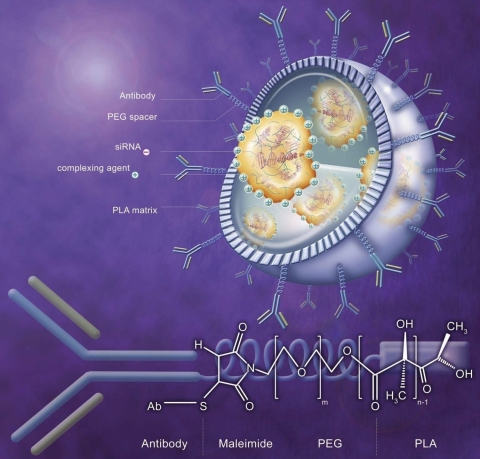
Fig. 4.
Schematic of nanoparticle covered with antibodies grafted on d,l-poly(lactic acid) (PLA) matrix via poly(ethylene glycol) (PEG) spacer. The inside of the NPs is optimized for protecting and loading maximum amount of oligonucleotides by complexation with complexing agent (polyethylenimine, chitosan, poly-l-lysine, or cationic polymer). siRNA, small interfering RNA; Ab, antibody.
Nanoparticles Used in Colitis Therapy
IBD, which includes UC and Crohn's disease, is a chronic debilitating inflammatory condition for which existing effective and targeted treatments are largely limited by significant systemic side effects. Until the previous decade, the treatment options for IBD included anti-inflammatory medications (5-amino salicylic acid, steroids) or immunosuppressants. Despite the efficacy of these medications, their use has been limited by their nonspecific effects on the immune system, resulting in short- and long-term debilitating side effects. Although newer biological therapies such as monoclonal antibodies against tumor necrosis factor-α (TNF-α) have been a therapeutic success, patients develop antibodies against the treatment, increasing the risk of infusion reactions and reducing the response to the treatment.
In an effort to mitigate the side effects of traditional small-molecule drugs for IBD, various sustained-release drug delivery vehicles including pellets, capsules, and tablets have been designed to localize drug release to the colon. These macroscopic drug delivery vehicles limit drug adsorption in the upper intestines by releasing encapsulated agents in response to colon-specific phenomena, such as colonic pH, colon-specific enzymes, or transit time of the carrier to the colon. However, the inflammation resulting from IBD is confined to distinct sites in the intestinal tissue that are distributed throughout the lower intestine and varies greatly from patient to patient. Consequently, current macroscopic drug delivery vehicles deliver drugs to unaffected tissues in the colon and thus lack the specificity needed to target only inflamed intestinal tissues. Furthermore, the efficacy of these single-unit delivery vehicles can be limited by the rapid intestinal clearance resulting from diarrhea, which is a common symptom of IBD experienced by as many as 92% of patients.
To overcome the limitations of current macroscopic drug delivery vehicles for the treatment of IBD, multiple groups have focused on the development of treatment strategies based on drug-loaded NPs. Orally delivered NPs passively target physical changes specific to inflamed tissues, including increases mucous production, accumulation of phagocytic immune cells, and inflammation-induced barrier disruption.
One of the seminal works in the development of nanoparticulate delivery systems for IBD was performed by Nakase et al. (72). In this work, orally administered microparticles composed of poly-dl-lactic acid loaded with dexamethasone were used to treat mice suffering from DSS-induced colitis. Their results showed that orally administered dexamethasone-loaded microparticles were predominantly taken up in inflamed colonic tissues and were more effective than solution administration at the same dose. The accumulation of NPs in inflamed colonic tissues was attributed to particle uptake by phagocytic immune cells, which inundate the submucosa as part of the intestinal inflammatory response.
A more in-depth investigation of micro- and nanoparticle interaction with inflamed intestinal mucosa was performed by Lamprecht et al. (54). In this work, the authors demonstrate that NPs composed of poly(lactic-co-glycolic acid) accumulated preferentially in the thicker mucus layer surrounding colitic ulcers compared with the mucus layers of healthy tissue. This study demonstrated an inverse relationship between particle binding to inflamed intestinal mucosa and particle size, with particles of less than 0.1 μm exhibiting the highest binding affinity. This accumulation of particles at sites of intestinal inflammation significantly increases particle retention near the tissue of interest, allowing for increased drug delivery to the affected tissue.
A central challenge to the targeted delivery of small molecules via NPs is the uncontrolled release of the active molecule in the upper intestinal tract that results from the characteristic initial drug release burst profile that is associated with the high surface area of NPs. To address this issue, the authors (80) covalently tethered the anti-inflammatory molecule 5-amino salicylic acid (5ASA) through a biodegradable linkage to NPs composed of poly(caprolactone). These 5ASA-bound NPs decreased 5ASA delivery to the upper intestinal organs and were as effective as a solution containing 60 times as much 5ASA in the treatment of IBD.
More recently, delivery strategies that incorporate the ability of NPs to passively target inflamed intestinal tissues have been incorporated into systems that actively target encapsulated agents in response to stimuli specific to inflamed intestinal tissues. Laroui et al. (57) demonstrated that a two-component system composed of NPs embedded in a polysaccharide hydrogel has the chemical and physical properties needed to target peptides and other biological macromolecules to inflamed colonic tissues. In this work, NPs loaded with the tripeptide KPV were codelivered with a polysaccharide hydrogel that protects the NPs from the harsh environment of the GI tract, limits KPV release to the upper intestinal tissues, and actively targets the release of the encapsulated NPs in inflamed intestinal tissues. As the hydrogel-nanoparticle hybrid system comes in contact with the distinctive pH of inflamed intestinal tissues, the hydrogel collapses, releasing the NPs, which deliver KPV to inflamed intestinal tissues. The incorporation of KPV-loaded NPs into polysaccharide gels has the ability to produce similar anti-inflammatory results to the free peptide at a 1,000-fold lower dose.
In addition to increasing the efficacy of traditional therapeutics for IBD, nanoparticulate drug carriers have the ability to potentiate the application of next-generation biological therapeutics, such as siRNAs for the treatment of IBD. Given that the aberrant production of proinflammatory cytokines, such as TNF-α, is a central factor in the onset and progression of IBD, siRNAs directed against TNF-α have the potential to transform the treatment of IBD. However, the development of a clinically viable siRNA-based therapeutic has been stymied by two key challenges: 1) degradation in vivo of TNF-α-siRNAs and 2) toxicity involved by systemic reduction of TNF-α. Accordingly, there has been great interest in developing delivery vehicles that can protect TNF-α-siRNA in vivo and target it to sites of intestinal inflammation (60). In this work Laroui et al. (57, 60) showed in vitro efficiency of loaded TNF-α-siRNA NPs on macrophages (RAW 264.7). They targeted TNF-α-siRNA-loaded NPs to the colon using a hydrogel. They showed a targeted decrease of the TNF-α level on mice treated with dextran sodium sulfate, a model of colitis in mice (60). Ideally, we could imagine that NPs could be targeted to a specific part of the colon encapsulated in a specific hydrogel such as in previous work (57). To increase the efficiency of therapeutics, NPs could be improved by recovering it by specific antibodies of macrophages against a receptor potentially overexpressed during inflammation like CD11b (β2-integrin receptor). This potential strategy illustrated on Fig. 5 will have the high interest to knock down colonic TNF-α expression on macrophages mainly involved on TNF-α expression.
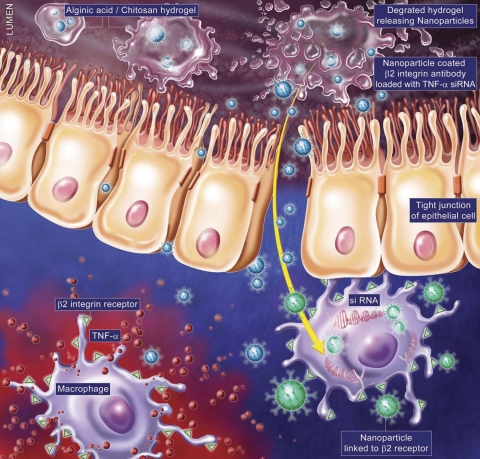
Fig. 5.
Schematic representation of “hierarchical nano-micro particles” with homing capabilities to directly release “molecularly specific” siRNA to target cells. This schematic shows nanoparticle packaged in alginate-chitosan hydrogel designed to home to the colon. Once in the colon the alginate-chitosan degrades releasing the nanoparticle coated with anti-integrin αX/CD11c antibody (to target dendritic cells/macrophages) loaded with TNF-α-siRNA.
Recently, Wilson et al. (107) demonstrated that NPs formulated from a novel polymer that degrades in response to reactive oxygen species (ROS) can be efficiently loaded with TNF-α-siRNA and that, when delivered orally, these siRNA-loaded NPs target TNF-α-siRNA to sites of intestinal inflammation, deplete TNF-α-mRNA in inflamed intestinal tissues, and thus mitigate intestinal inflammation.
The aberrant production of ROS at sites of intestinal inflammation is an important factor in pathogenesis of IBD. For example, biopsies taken from patients suffering from IBD have a ten- to hundredfold increase in mucosal ROS concentrations that are confined to sites of disease development. Kountouras et al. (49) identified the abnormally high levels of ROS produced at sites of intestinal inflammation as a disease specific triggering mechanism for siRNA release. To target orally delivered siRNA to sites of intestinal inflammation, they developed NPs, termed thioketal NPs (TKNs) that release encapsulated agents in response to ROS. TKNs are formulated from poly-(1,4-phenyleneacetone dimethylene thioketal), a new polymer composed of ROS-sensitive thioketal linkages (92) that are stable to acid-, base-, and protease-catalyzed degradation (92, 13). Therefore, TKNs loaded with TNF-α-siRNA remain stable in the harsh environment of the GI tract, thereby protecting siRNA and preventing its release to noninflamed tissues. However, at sites of intestinal inflammation the elevated ROS levels trigger the degradation of the TNF-α-TKNs, thus localizing the release of siRNA to inflamed intestinal tissues. Using a murine model of UC, the authors showed that at a dose of 0.23 mg TNF-α-siRNA per day that orally administered TNF-α-TKNs diminished TNF-α-mRNA levels in the colon and protect mice from experimental UC.
Nanomedicine for the Oral Delivery of Vaccines
One of the most important future applications of NMs is their use as nanocarriers for vaccine delivery to the mucosa (20, 89). NPs can be used as adjuvants, as a delivery system, or both (6, 50, 73). Nano- or microsized particles can target specific sites and mechanisms to enable uptake for delivery of vaccines to the mucosa (74). NPs that are <200 nm in size are taken up by the epithelium mainly in the intestinal epithelial cells on villi tips or by the M cells. Intestinal macrophages can phagocytose particles 1 μm in size, such as the liposomes (29). Tafaghodi et al. (98) successfully developed a liposomal intranasal vaccine against the tetanus toxoid. Following nasal administration in rabbits, they found that the nanocarrier-encapsulated formulation was more effective in inducing a mucosal immune response. Various ligands have been used on the surfaces of NPs and microparticles (74). Such ligands may be proteins, for example, the antibody mAb 5B11, which is used on polystyrene latex particles to target M cells, or the secretory IgA, which targets mouse Peyer's patch M cells (82, 115), or they may be other molecules, for example, the efficient truncated protein Invasin-C192 [a bacterial protein involved in cell attachment and invasion (62)] (40). Ligands may additionally be polysaccharides bonded to lipophilic groups, such as O-palmitoyl mannan (41) or the cholera toxin B subunit (14, 31, 93).
CONCLUSION
This review has summarized the major types of NPs that have potential use in gastroenterology. The application of nanotechnology to medicine is a rapidly developing area of investigation. In the near future, it appears highly likely that nanotechnology will play an important role in assessment and treatment of gastroenterological diseases. Indeed, some of the nanomaterial-based therapies and diagnostics presented here outperform conventional materials in terms of efficacy, reliability, and practicality. Continued optimization of nanomaterial properties will be necessary to determine the applicability of these methods in modern healthcare practice. Importantly, advances in the integration of imaging systems and controllable nanomaterials may ultimately permit the detection of small bowel lesions. Futurist applications are already under investigation. Nanorobots, an active area of innovation, use nanometric robots for surgical and/or endoscopic procedures. Capsule endoscopy currently involves a miniature endoscopic camera that is swallowed by a person and permits detection of small bowel lesions. One can envision camera pills as nanorobots, which will enable diagnostic and therapeutic maneuvers.
GRANTS
This work was supported by grants from the Department of Veterans Affairs and National Institute of Diabetes and Digestive and Kidney Diseases Grants R01-DK071594 (to D. Merlin) and RO1-DK55850 (to S. V. Sitaraman). G. Dalmasso is a recipient of a Research Fellowship Award from the Crohn's and Colitis Foundation of America.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
REFERENCES
- 1. Akita H, Kudo A, Minoura A, Yamaguti M, Khalil IA, Moriguchi R, Masuda T, Danev R, Nagayama K, Kogure K, Harashima H. Multi-layered nanoparticles for penetrating the endosome and nuclear membrane via a step-wise membrane fusion process. Biomaterials 30: 2940–2949, 2009 [DOI] [PubMed] [Google Scholar]
- 2. Altinbas N, Fehmer C, Terheiden A, Shukla A, Rehage H, Mayer C. Alkylcyanoacrylate nanocapsules prepared from mini-emulsions: a comparison with the conventional approach. J Microencapsul 23: 567–581, 2006 [DOI] [PubMed] [Google Scholar]
- 3. An SY, Bui MP, Nam YJ, Han KN, Li CA, Choo J, Lee EK, Katoh S, Kumada Y, Seong GH. Preparation of monodisperse and size-controlled poly(ethylene glycol) hydrogel nanoparticles using liposome templates. J Colloid Interface Sci 331: 98–103, 2009 [DOI] [PubMed] [Google Scholar]
- 4. Avgoustakis K. Pegylated poly(lactide) and poly(lactide-co-glycolide) nanoparticles: preparation, properties and possible applications in drug delivery. Curr Drug Deliv 1: 321–333, 2004 [DOI] [PubMed] [Google Scholar]
- 5. Baluom M, Friedman M, Rubinstein A. Improved intestinal absorption of sulpiride in rats with synchronized oral delivery systems. J Control Release 70: 139–147, 2001 [DOI] [PubMed] [Google Scholar]
- 6. Belyakov IM, Berzofsky JA. Immunobiology of mucosal HIV infection and the basis for development of a new generation of mucosal AIDS vaccines. Immunity 20: 247–253, 2004 [DOI] [PubMed] [Google Scholar]
- 7. Brigger I, Dubernet C, Couvreur P. Nanoparticles in cancer therapy and diagnosis. Adv Drug Deliv Rev 54: 631–651, 2002 [DOI] [PubMed] [Google Scholar]
- 8. Bu X, Khankaldyyan V, Gonzales-Gomez I, Groshen S, Ye W, Zhuo S, Pons J, Stratton JR, Rosenberg S, Laug WE. Species-specific urokinase receptor ligands reduce glioma growth and increase survival primarily by an antiangiogenesis mechanism. Lab Invest 84: 667–678, 2004 [DOI] [PubMed] [Google Scholar]
- 9. Chan LM, Lowes S, Hirst BH. The ABCs of drug transport in intestine and liver: efflux proteins limiting drug absorption and bioavailability. Eur J Pharm Sci 21: 25–51, 2004 [DOI] [PubMed] [Google Scholar]
- 10. Chan WC, Maxwell DJ, Gao X, Bailey RE, Han M, Nie S. Luminescent quantum dots for multiplexed biological detection and imaging. Curr Opin Biotechnol 13: 40–46, 2002 [DOI] [PubMed] [Google Scholar]
- 11. Chen LD, Liu J, Yu XF, He M, Pei XF, Tang ZY, Wang QQ, Pang DW, Li Y. The biocompatibility of quantum dot probes used for the targeted imaging of hepatocellular carcinoma metastasis. Biomaterials 29: 4170–4176, 2008 [DOI] [PubMed] [Google Scholar]
- 12. Choi JS, Park JC, Nah H, Woo S, Oh J, Kim KM, Cheon GJ, Chang Y, Yoo J, Cheon J. A hybrid nanoparticle probe for dual-modality positron emission tomography and magnetic resonance imaging. Angew Chem Int Ed Engl 47: 6259–6262, 2008 [DOI] [PubMed] [Google Scholar]
- 13. Colonna S, Gaggero N, Carrea G, Pasta P. Enantio and diastereoselectivity of cyclohexanone monooxygenase catalyzed oxidation of 1,3-dithioacetals. Tetrahedron 7: 565–570, 1996 [Google Scholar]
- 14. Conacher M, Alexander J, JMB Oral immunization with peptide and protein antigens by formulations in lipid vesicles incorporating bile salts (bilosomes). Vaccine 19: 2965–2974, 2001 [DOI] [PubMed] [Google Scholar]
- 15. Crociani F, Alessandrini A, Mucci MM, Biavati B. Degradation of complex carbohydrates by Bifidobacterium spp. Int J Food Microbiol 24: 199–210, 1994 [DOI] [PubMed] [Google Scholar]
- 16. Das S, Ng KY. Colon-specific delivery of resveratrol: optimization of multi-particulate calcium-pectinate carrier. Int J Pharm 385: 20–28, 2010 [DOI] [PubMed] [Google Scholar]
- 17. De Jonge J, Holtrop M, Wilschut J, Huckriede A. Reconstituted influenza virus envelopes as an efficient carrier system for cellular delivery of small-interfering RNAs. Gene Ther 13: 400–411, 2006 [DOI] [PubMed] [Google Scholar]
- 18. Debongnie JC, Phillips SF. Capacity of the human colon to absorb fluid. Gastroenterology 74: 698–703, 1978 [PubMed] [Google Scholar]
- 19. Delattre J, Couvreur P, Puisieux F, Philippot Schuber F., JR Les liposomes: aspects technologiques, biologiques et pharmacologiques, edited by Doc-Lavoisier T. Paris: INSERM, 1993 [Google Scholar]
- 20. Demento SL, Bonafe N, Cui W, Kaech SM, Caplan MJ, Fikrig E, Ledizet M, Fahmy TM. TLR9-targeted biodegradable nanoparticles as immunization vectors protect against West Nile encephalitis. J Immunol 185: 2989–2997, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Diaz A, David A, Perez R, Gonzalez ML, Baez A, Wark SE, Zhang P, Clearfield A, Colon JL. Nanoencapsulation of insulin into zirconium phosphate for oral delivery applications. Biomacromolecules 11: 2465–2470, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Dittgen MHB. Zeta potential—fundamentals, measurement methods and application to pharmacy. Pharmazie 42: 641–656, 1987 [PubMed] [Google Scholar]
- 23. Dorkoosh FA, Verhoef JC, Borchard G, Rafiee-Tehrani M, Junginger HE. Development and characterization of a novel peroral peptide drug delivery system. J Control Release 71: 307–318, 2001 [DOI] [PubMed] [Google Scholar]
- 24. Douglas SJ, Davis SS, Illum L. Nanoparticles in drug delivery. Crit Rev Ther Drug Carrier Syst 3: 233–261, 1987 [PubMed] [Google Scholar]
- 25. Fang C, Bhattarai N, Sun C, Zhang M. Functionalized nanoparticles with long-term stability in biological media. Small 5: 1637–1641, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25a. Faraday M. The Bakerian lecture: experimental relations of gold (and other metals) to light. Phil Trans R Soc Lond 147: 145–181, 1847 [Google Scholar]
- 26. Farokhzad OC, Jon S, Khademhosseini A, Tran TN, Lavan DA, Langer R. Nanoparticle-aptamer bioconjugates: a new approach for targeting prostate cancer cells. Cancer Res 64: 7668–7672, 2004 [DOI] [PubMed] [Google Scholar]
- 27. Fattal E, Vauthier C, Aynie I, Nakada Y, Lambert G, Malvy C, Couvreur P. Biodegradable polyalkylcyanoacrylate nanoparticles for the delivery of oligonucleotides. J Control Release 53: 137–143, 1998 [DOI] [PubMed] [Google Scholar]
- 28. Flynn MA, Casey DG, Todryk SM, Mahon BP. Efficient delivery of small interfering RNA for inhibition of IL-12p40 expression in vivo. J Inflamm 1: 4, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Godbey WT, Barry MA, Saggau P, Wu KK, Mikos AG. Poly(ethylenimine)-mediated transfection: a new paradigm for gene delivery. J Biomed Mater Res 51: 321–328, 2000 [DOI] [PubMed] [Google Scholar]
- 30. Gunther M, Wagner E, Ogris M. Specific targets in tumor tissue for the delivery of therapeutic genes. Curr Med Chem Anticancer Agents 5: 157–171, 2005 [DOI] [PubMed] [Google Scholar]
- 31. Harokopakis E, Childers NK, Michalek SM, Zhang SS, Tomasi M. Conjugation of cholera toxin or its B subunit to liposomes for targeted delivery of antigens. J Immunol Methods 185: 31–42, 1995 [DOI] [PubMed] [Google Scholar]
- 32. Hassani S, Pellequer Y, Lamprecht A. Selective adhesion of nanoparticles to inflamed tissue in gastric ulcers. Pharm Res 26: 1149–1154, 2009 [DOI] [PubMed] [Google Scholar]
- 33. Hikage M, Gonda K, Takeda M, Kamei T, Kobayashi M, Kumasaka M, Watanabe M, Satomi S, Ohuchi N. Nano-imaging of the lymph network structure with quantum dots. Nanotechnology 21: 185103, 2010 [DOI] [PubMed] [Google Scholar]
- 34. Hood JD, Bednarski M, Frausto R, Guccione S, Reisfeld RA, Xiang R, Cheresh DA. Tumor regression by targeted gene delivery to the neovasculature. Science 296: 2404–2407, 2002 [DOI] [PubMed] [Google Scholar]
- 35. Horber JK, Miles MJ. Scanning probe evolution in biology. Science 302: 1002–1005, 2003 [DOI] [PubMed] [Google Scholar]
- 36. Horter D, Dressman JB. Influence of physicochemical properties on dissolution of drugs in the gastrointestinal tract. Adv Drug Deliv Rev 46: 75–87, 2001 [DOI] [PubMed] [Google Scholar]
- 37. Hu Z, Mawatari S, Shibata N, Takada K, Yoshikawa H, Arakawa A, Yosida Y. Application of a biomagnetic measurement system (BMS) to the evaluation of gastrointestinal transit of intestinal pressure-controlled colon delivery capsules (PCDCs) in human subjects. Pharm Res 17: 160–167, 2000 [DOI] [PubMed] [Google Scholar]
- 38. Hu Z, Mawatari S, Shimokawa T, Kimura G, Yoshikawa Y, Shibata N, Takada K. Colon delivery efficiencies of intestinal pressure-controlled colon delivery capsules prepared by a coating machine in human subjects. J Pharm Pharmacol 52: 1187–1193, 2000 [DOI] [PubMed] [Google Scholar]
- 39. Hussain H, Tan BH, Seah GL, Liu Y, He CB, Davis TP. Micelle formation and gelation of (PEG-P(MA-POSS)) amphiphilic block copolymers via associative hydrophobic effects. Langmuir 26: 11763–11773, 2010 [DOI] [PubMed] [Google Scholar]
- 40. Hussain N, Florence AT. Utilizing bacterial mechanisms of epithelial cell entry: invasin-induced oral uptake of latex nanoparticles. Pharm Res 15: 153–156, 1998 [DOI] [PubMed] [Google Scholar]
- 41. Jain S, Vyas SP. Mannosylated niosomes as adjuvant-carrier system for oral mucosal immunization. J Liposome Res 16: 331–345, 2006 [DOI] [PubMed] [Google Scholar]
- 42. Jani P, Halbert GW, Langridge J, Florence AT. The uptake and translocation of latex nanospheres and microspheres after oral administration to rats. J Pharm Pharmacol 41: 809–812, 1989 [DOI] [PubMed] [Google Scholar]
- 43. Jung T, Kamm W, Breitenbach A, Kaiserling E, Xiao JX, Kissel T. Biodegradable nanoparticles for oral delivery of peptides: is there a role for polymers to affect mucosal uptake? Eur J Pharm Biopharm 50: 147–160, 2000 [DOI] [PubMed] [Google Scholar]
- 44. Jung Y, Kim YM. What should be considered on design of a colon-specific prodrug? Expert Opin Drug Deliv 7: 245–258, 2010 [DOI] [PubMed] [Google Scholar]
- 46. Karoutsos V. Scanning probe microscopy: instrumentation and applications on thin films and magnetic multilayers. J Nanosci Nanotechnol 9: 6783–6798, 2009 [DOI] [PubMed] [Google Scholar]
- 47. Kim B, Tang Q, Biswas PS, Xu J, Schiffelers RM, Xie FY, Ansari AM, Scaria PV, Woodle MC, Lu P, Rouse BT. Inhibition of ocular angiogenesis by siRNA targeting vascular endothelial growth factor pathway genes: therapeutic strategy for herpetic stromal keratitis. Am J Pathol 165: 2177–2185, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Kirui DK, Rey DA, Batt CA. Gold hybrid nanoparticles for targeted phototherapy and cancer imaging. Nanotechnology 21: 105105, 2010 [DOI] [PubMed] [Google Scholar]
- 49. Kountouras J, Chatzopoulos D, Zavos C. Reactive oxygen metabolites and upper gastrointestinal diseases. Hepatogastroenterology 48: 743–751, 2001 [PubMed] [Google Scholar]
- 50. Kozlowski PA, Neutra MR. The role of mucosal immunity in prevention of HIV transmission. Curr Mol Med 3: 217–228, 2003 [DOI] [PubMed] [Google Scholar]
- 51. Kroto HW, Heath JR, O'Brien SC, Curl RF, Smalley RE. C60: Buckminsterfullerene. Nature 318: 162–163, 1985 [Google Scholar]
- 52. Lacoeuille F, Garcion E, Benoit JP, Lamprecht A. Lipid nanocapsules for intracellular drug delivery of anticancer drugs. J Nanosci Nanotechnol 7: 4612–4617, 2007 [PubMed] [Google Scholar]
- 53. Lamprecht A, Scaffer U, Lehr CM. Size dependent targeting of micro- and nano-particulate carriers to the inflamed colonic mucosa. Pharm Res 18: 788–793, 2001 [DOI] [PubMed] [Google Scholar]
- 54. Lamprecht A, Ubrich N, Yamamoto H, Schafer U, Takeuchi H, Maincent P, Kawashima Y, Lehr CM. Biodegradable nanoparticles for targeted drug delivery in treatment of inflammatory bowel disease. J Pharmacol Exp Ther 299: 775–781, 2001 [PubMed] [Google Scholar]
- 55. Lamprecht A, Yamamoto H, Takeuchi H, Kawashima Y. Microsphere design for the colonic delivery of 5-fluorouracil. J Control Release 90: 313–322, 2003 [DOI] [PubMed] [Google Scholar]
- 56. Langer R, Folkman J. Polymers for the sustained release of proteins and other macromolecules. Nature 263: 797–800, 1976 [DOI] [PubMed] [Google Scholar]
- 57. Laroui H, Dalmasso G, Nguyen HT, Yan Y, Sitaraman SV, Merlin D. Drug-loaded nanoparticles targeted to the colon with polysaccharide hydrogel reduce colitis in a mouse model. Gastroenterology 138: 843–853.e41–42, 2010 [DOI] [PubMed] [Google Scholar]
- 58. Laroui H, Grossin L, Leonard M, Stoltz JF, Gillet P, Netter P, Dellacherie E. Hyaluronate-covered nanoparticles for the therapeutic targeting of cartilage. Biomacromolecules 8: 3879–3885, 2007 [DOI] [PubMed] [Google Scholar]
- 60. Laroui H, Theiss AL, Yan Y, Dalmasso G, Nguyen HT, Sitaraman SV, Merlin D. Functional TNFα gene silencing mediated by polyethyleneimine/TNFα siRNA nanocomplexes in inflamed colon. Biomaterials 32: 1218–1228, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Lees CW, Satsangi J. Genetics of inflammatory bowel disease: implications for disease pathogenesis and natural history. Expert Rev Gastroenterol Hepatol 3: 513–534, 2009 [DOI] [PubMed] [Google Scholar]
- 62. Leong JM, Fournier RS, Isberg RR. Identification of the integrin binding domain of the Yersinia pseudotuberculosis invasin protein. EMBO J 9: 1979–1989, 1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Li F, Sun JY, Wang JY, Du SL, Lu WY, Liu M, Xie C, Shi JY. Effect of hepatocyte growth factor encapsulated in targeted liposomes on liver cirrhosis. J Control Release 131: 77–82, 2008 [DOI] [PubMed] [Google Scholar]
- 64. Li H, Soria C, Griscelli F, Opolon P, Soria J, Yeh P, Legrand C, Vannier JP, Belin D, Perricaudet M, Lu H. Amino-terminal fragment of urokinase inhibits tumor cell invasion in vitro and in vivo: respective contribution of the urokinase plasminogen activator receptor-dependent or -independent pathway. Hum Gene Ther 16: 1157–1167, 2005 [DOI] [PubMed] [Google Scholar]
- 65. Li J, Ng HT, Chen H. Carbon nanotubes and nanowires for biological sensing. Methods Mol Biol 300: 191–123, 2005 [DOI] [PubMed] [Google Scholar]
- 66. Liu W, Greytak AB, Lee J, Wong CR, Park J, Marshall LF, Jiang W, Curtin PN, Ting AY, Nocera DG, Fukumura D, Jain RK, Bawendi MG. Compact biocompatible quantum dots via RAFT-mediated synthesis of imidazole-based random copolymer ligand. J Am Chem Soc 132: 472–483, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Matsumura Y. Polymeric micellar delivery systems in oncology. Jpn J Clin Oncol 38: 793–802, 2008 [DOI] [PubMed] [Google Scholar]
- 68. McConnell EL, Murdan S, Basit AW. An investigation into the digestion of chitosan (noncrosslinked and crosslinked) by human colonic bacteria. J Pharm Sci 97: 3820–3829, 2008 [DOI] [PubMed] [Google Scholar]
- 69. Meade BR, Dowdy SF. Exogenous siRNA delivery using peptide transduction domains/cell penetrating peptides. Adv Drug Deliv Rev 59: 134–140, 2007 [DOI] [PubMed] [Google Scholar]
- 70. Moghimi SM, Hunter AC, Murray JC. Long-circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev 53: 283–318, 2001 [PubMed] [Google Scholar]
- 71. Morrissey DV, Lockridge JA, Shaw L, Blanchard K, Jensen K, Breen W, Hartsough K, Machemer L, Radka S, Jadhav V, Vaish N, Zinnen S, Vargeese C, Bowman K, Shaffer CS, Jeffs LB, Judge A, MacLachlan I, Polisky B. Potent and persistent in vivo anti-HBV activity of chemically modified siRNAs. Nat Biotechnol 23: 1002–1007, 2005 [DOI] [PubMed] [Google Scholar]
- 72. Nakase H, Okazaki K, Tabata Y, Chiba T. Biodegradable microspheres targeting mucosal immune-regulating cells: new approach for treatment of inflammatory bowel disease. J Gastroenterol 38, Suppl 15: 59–62, 2003 [PubMed] [Google Scholar]
- 73. Neutra MR, Kozlowski PA. Mucosal vaccines: the promise and the challenge. Nat Rev Immunol 6: 148–158, 2006 [DOI] [PubMed] [Google Scholar]
- 74. O'Hagan DT. The intestinal uptake of particles and the implications for drug and antigen delivery. J Anat 189: 477–482, 1996 [PMC free article] [PubMed] [Google Scholar]
- 75. Onishi H, Koyama K, Sakata O, Machida Y. Preparation of chitosan/alginate/calcium complex microparticles loaded with lactoferrin and their efficacy on carrageenan-induced edema in rats. Drug Dev Ind Pharm 36: 879–884, 2010 [DOI] [PubMed] [Google Scholar]
- 76. Otsuka H, Nagasaki Y, Kataoka K. PEGylated nanoparticles for biological and pharmaceutical applications. Adv Drug Deliv Rev 55: 403–419, 2003 [DOI] [PubMed] [Google Scholar]
- 77. Owens DE, 3rd, Peppas NA. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int J Pharm 307: 93–102, 2006 [DOI] [PubMed] [Google Scholar]
- 78. Pardridge WM. Intravenous, non-viral RNAi gene therapy of brain cancer. Expert Opin Biol Ther 4: 1103–1113, 2004 [DOI] [PubMed] [Google Scholar]
- 79. Pedersen GP, Fäldt P, Bergenstaåhl B, Kristensen HG. Solid state characterisation of a dry emulsion: a potential drug delivery system. Int J Pharm 171: 257–270, 1998 [Google Scholar]
- 80. Pertuit D, Moulari B, Betz T, Nadaradjane A, Neumann D, Ismaili L, Refouvelet B, Pellequer Y, Lamprecht A. 5-Amino salicylic acid bound nanoparticles for the therapy of inflammatory bowel disease. J Control Release 123: 211–218, 2007 [DOI] [PubMed] [Google Scholar]
- 81. Pinhassi RI, Assaraf YG, Farber S, Stark M, Ickowicz D, Drori S, Domb AJ, Livney YD. Arabinogalactan-folic acid-drug conjugate for targeted delivery and target-activated release of anticancer drugs to folate receptor-overexpressing cells. Biomacromolecules 11: 294–303, 2010 [DOI] [PubMed] [Google Scholar]
- 82. Porta C, James PS, Phillips AD, Savidge TC, Smith MW, Cremaschi D. Confocal analysis of fluorescent bead uptake by mouse Peyer's patch follicle-associated M cells. Exp Physiol 77: 929–932, 1992 [DOI] [PubMed] [Google Scholar]
- 83. Raper SE. Gene therapy: the good, the bad, and the ugly. Surgery 137: 487–492, 2005 [DOI] [PubMed] [Google Scholar]
- 84. Ravi S, Peh KK, Darwis Y, Murthy BK, Singh TR, Mallikarjun C. Development and characterization of polymeric microspheres for controlled release protein loaded drug delivery system. Indian J Pharm Sci 70: 303–309, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Romberg B, Hennink WE, Storm G. Sheddable coatings for long-circulating nanoparticles. Pharm Res 25: 55–71, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Sajeesh S, Bouchemal K, Sharma CP, Vauthier C. Surface-functionalized polymethacrylic acid based hydrogel microparticles for oral drug delivery. Eur J Pharm Biopharm 74: 209–218, 2010 [DOI] [PubMed] [Google Scholar]
- 87. Schiffelers RM, Ansari A, Xu J, Zhou Q, Tang Q, Storm G, Molema G, Lu PY, Scaria PV, Woodle MC. Cancer siRNA therapy by tumor selective delivery with ligand-targeted sterically stabilized nanoparticle. Nucleic Acids Res 32: e149, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Serrano F, Lopez GL, Jadraque M, Koper M, Ellis G, Cano P, Martin M, Garrido L. A Nd:YAG laser-microperforated poly(3-hydroxybutyrate-co-3-hydroxyvalerate)-basal membrane matrix composite film as substrate for keratinocytes. Biomaterials 28: 650–660, 2007 [DOI] [PubMed] [Google Scholar]
- 89. Shahiwala A, Vyas TK, Amiji MM. Nanocarriers for systemic and mucosal vaccine delivery. Recent Pat Drug Deliv Formul 1: 1–9, 2007 [DOI] [PubMed] [Google Scholar]
- 90. Shibata N, Ohno T, Shimokawa T, Hu Z, Yoshikawa Y, Koga K, Murakami M, Takada K. Application of pressure-controlled colon delivery capsule to oral administration of glycyrrhizin in dogs. J Pharm Pharmacol 53: 441–447, 2001 [DOI] [PubMed] [Google Scholar]
- 91. Shrivastava PK, Shrivastava SK. Dextran carrier macromolecule for colon specific delivery of celecoxib. Curr Drug Deliv 7: 144–151, 2010 [DOI] [PubMed] [Google Scholar]
- 92. Shukla AK, Verma M, Singh KN. Superoxide induced deprotection of 1,3-dithiolanes: a convenient method of dedithioacetalization. Indian J Chem-Sect B 43: 1748–1752, 2004 [Google Scholar]
- 93. Singh P, Prabakaran D, Jain S, Mishra V, Jaganathan KS, Vyas SP. Cholera toxin B subunit conjugated bile salt stabilized vesicles (bilosomes) for oral immunization. Int J Pharm 278: 379–390, 2004 [DOI] [PubMed] [Google Scholar]
- 94. Solvang S, Finholt P. Effect of tablet processing and formulation factors on dissolution rate of the active ingredient in human gastric juice. J Pharm Sci 59: 49–52, 1970 [DOI] [PubMed] [Google Scholar]
- 95. Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, John M, Kesavan V, Lavine G, Pandey RK, Racie T, Rajeev KG, Rohl I, Toudjarska I, Wang G, Wuschko S, Bumcrot D, Koteliansky V, Limmer S, Manoharan M, Vornlocher HP. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 432: 173–178, 2004 [DOI] [PubMed] [Google Scholar]
- 96. Sun C, Du K, Fang C, Bhattarai N, Veiseh O, Kievit F, Stephen Z, Lee D, Ellenbogen RG, Ratner B, Zhang M. PEG-mediated synthesis of highly dispersive multifunctional superparamagnetic nanoparticles: their physicochemical properties and function in vivo. ACS Nano 4: 2402–2410, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Sun C, Lee JS, Zhang M. Magnetic nanoparticles in MR imaging and drug delivery. Adv Drug Deliv Rev 60: 1252–1265, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Tafaghodi M, Jaafari MR, Tabassi SA. Nasal immunization studies by cationic, fusogenic and cationic-fusogenic liposomes encapsulated with tetanus toxoid. Curr Drug Deliv 5: 108–113, 2008 [DOI] [PubMed] [Google Scholar]
- 99. Takaya T, Ikeda C, Imagawa N, Niwa K, Takada K. Development of a colon delivery capsule and the pharmacological activity of recombinant human granulocyte colony-stimulating factor (rhG-CSF) in beagle dogs. J Pharm Pharmacol 47: 474–478, 1995 [DOI] [PubMed] [Google Scholar]
- 100. Tangirala R, Revanur R, Russell TP, Emrick T. Sizing nanoparticle-covered droplets by extrusion through track-etch membranes. Langmuir 23: 965–969, 2007 [DOI] [PubMed] [Google Scholar]
- 101. Theiss AL, Laroui H, Obertone TS, Chowdhury I, Thompson WE, Merlin D, Sitaraman SV. Nanoparticle-based therapeutic delivery of prohibitin to the colonic epithelial cells ameliorates acute murine colitis. Inflamm Bowel Dis 2010. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Toub N, Bertrand JR, Tamaddon A, Elhamess H, Hillaireau H, Maksimenko A, Maccario J, Malvy C, Fattal E, Couvreur P. Efficacy of siRNA nanocapsules targeted against the EWS-Fli1 oncogene in Ewing sarcoma. Pharm Res 23: 892–900, 2006 [DOI] [PubMed] [Google Scholar]
- 103. Veiseh O, Gunn JW, Zhang M. Design and fabrication of magnetic nanoparticles for targeted drug delivery and imaging. Adv Drug Deliv Rev 62: 284–304, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Veiseh O, Sun C, Fang C, Bhattarai N, Gunn J, Kievit F, Du K, Pullar B, Lee D, Ellenbogen RG, Olson J, Zhang M. Specific targeting of brain tumors with an optical/magnetic resonance imaging nanoprobe across the blood-brain barrier. Cancer Res 69: 6200–6207, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Verma UN, Surabhi RM, Schmaltieg A, Becerra C, Gaynor RB. Small interfering RNAs directed against beta-catenin inhibit the in vitro and in vivo growth of colon cancer cells. Clin Cancer Res 9: 1291–1300, 2003 [PubMed] [Google Scholar]
- 106. Vittaz M, Bazile D, Spenlehauer G, Verrecchia T, Veillard M, Puisieux F, Labarre D. Effect of PEO surface density on long-circulating PLA-PEO nanoparticles which are very low complement activators. Biomaterials 17: 1575–1581, 1996 [DOI] [PubMed] [Google Scholar]
- 107. Wilson DS, Dalmasso G, Wang L, Sitaraman SV, Merlin D, Murthy N. Orally delivered thioketal nanoparticles loaded with TNF-alpha-siRNA target inflammation and inhibit gene expression in the intestines. Nat Mater 9: 923–928, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Xiong XB, Ma Z, Lai R, Lavasanifar A. The therapeutic response to multifunctional polymeric nano-conjugates in the targeted cellular and subcellular delivery of doxorubicin. Biomaterials 31: 757–768, 2010 [DOI] [PubMed] [Google Scholar]
- 109. Yang L, Mao H, Cao Z, Wang YA, Peng X, Wang X, Sajja HK, Wang L, Duan H, Ni C, Staley CA, Wood WC, Gao X, Nie S. Molecular imaging of pancreatic cancer in an animal model using targeted multifunctional nanoparticles. Gastroenterology 136: 1514–1525.e2, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Yang Z, Zhang Y, Yang Y, Sun L, Han D, Li H, Wang C. Pharmacological and toxicological target organelles and safe use of single-walled carbon nanotubes as drug carriers in treating Alzheimer disease. Nanomedicine 6: 427–441, 2010 [DOI] [PubMed] [Google Scholar]
- 111. Yang Z, Zheng S, Harrison WJ, Harder J, Wen X, Gelovani JG, Qiao A, Li C. Long-circulating near-infrared fluorescence core-cross-linked polymeric micelles: synthesis, characterization, and dual nuclear/optical imaging. Biomacromolecules 8: 3422–3428, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Yano J, Hirabayashi K, Nakagawa S, Yamaguchi T, Nogawa M, Kashimori I, Naito H, Kitagawa H, Ishiyama K, Ohgi T, Irimura T. Antitumor activity of small interfering RNA/cationic liposome complex in mouse models of cancer. Clin Cancer Res 10: 7721–7726, 2004 [DOI] [PubMed] [Google Scholar]
- 113. Yoo MK, Kang SK, Choi JH, Park IK, Na HS, Lee HC, Kim EB, Lee NK, Nah JW, Choi YJ, Cho CS. Targeted delivery of chitosan nanoparticles to Peyer's patch using M cell-homing peptide selected by phage display technique. Biomaterials 31: 7738–7747, 2010 [DOI] [PubMed] [Google Scholar]
- 114. Zambaux MF, Bonneaux F, Gref R, Dellacherie E, Vigneron C. Preparation and characterization of protein C-loaded PLA nanoparticles. J Control Release 60: 179–188, 1999 [DOI] [PubMed] [Google Scholar]
- 115. Zhou F, Kraehenbuhl JP, Neutra MR. Mucosal IgA response to rectally administered antigen formulated in IgA-coated liposomes. Vaccine 13: 637–644, 1995 [DOI] [PubMed] [Google Scholar]