Abstract
Panic disorder is a severe anxiety disorder characterized by susceptibility to induction of panic attacks by subthreshold interoceptive stimuli such as 0.5 M sodium lactate infusions. Although studied for four decades, the mechanism of lactate sensitivity in panic disorder has not been understood. The dorsomedial hypothalamus/perifornical region (DMH/PeF) coordinates rapid mobilization of behavioral, autonomic, respiratory and endocrine responses to stress, and rats with disrupted GABA inhibition in the DMH/PeF exhibit panic-like responses to lactate, similar to panic disorder patients. Utilizing a variety of anatomical and pharmacological methods, we provide evidence that lactate, via osmosensitive periventricular pathways, activates neurons in the compromised DMH/PeF, which relays this signal to forebrain limbic structures such as the bed nucleus of the stria terminalis to mediate anxiety responses, and specific brainstem sympathetic and parasympathetic pathways to mediate the respiratory and cardiovascular components of the panic-like response. Acutely restoring local GABAergic tone in the DMH/PeF blocked lactate induced panic-like responses. Autonomic panic-like responses appear to be a result of DMH/PeF-mediated mobilization of sympathetic responses (verified with atenolol) and resetting of the parasympathetically-mediated baroreflex. Based on our findings, DMH/PeF efferent targets such as the C1 adrenergic neurons, paraventricular hypothalamus and the central amygdala are implicated in sympathetic mobilization; the nucleus of the solitary tract is implicated in baroreflex resetting; and the parabrachial nucleus is implicated in respiratory responses. These results elucidate neural circuits underlying lactate induced panic-like responses and the involvement of both sympathetic and parasympathetic systems.
Keywords: panic, c-Fos, hypothalamus, GABA, anxiety, lactate, l-allylglycine
A 'panic' response is a normal physiological survival reflex in humans (Stein and Bouwer, 1997) and can be elicited by either an exteroceptive- or interoceptive-cue perceived as life-threatening (Street et al., 1989; Ehlers and Breuer, 1996). Panic disorder is a severe anxiety disorder characterized by recurrent panic attacks, consisting of pronounced fear, as well as cardiovascular and respiratory responses (DSM-IV, 1994). The initial pathology in these patients appears to be an alteration somewhere in the central neural pathways regulating normal panic responses, thus rendering the patients susceptible to unprovoked panic symptoms when exposed to ordinarily mild interoceptive stressors (Vickers and McNally, 2005). This is supported by findings that panic disorder patients, but not healthy controls, are hyper-responsive to normal interoceptive cues (Hoehn-Saric et al., 2004; Pollock et al., 2006), and are also susceptible to induction of panic attacks by interoceptive stimuli such as intravenous (i.v.) 0.5 M sodium lactate infusions and 7.5% CO2 inhalations (Liebowitz et al., 1986; Gorman et al., 1994). In light of this, these specific interoceptive challenges that fail to induce panic-like responses in healthy controls are often referred to as “subthreshold” interoceptive challenges.
Removal of local tonic γ-amino butyric acid (GABA)-mediated inhibition in the dorsomedial hypothalamic/perifornical region (DMH/PeF) in rats results in rapid mobilization of a response characterized by behavioral, autonomic, respiratory and endocrine components of panic-like responses [see review (DiMicco et al., 2002)]. In light of this, Shekhar and colleagues developed an animal model of panic disorder where chronically inhibiting GABA synthesis in the DMH/PeF of rats leads to heightened anxiety and panic-like responses following exposure to i.v. infusions of 0.5 M sodium lactate (Shekhar et al., 1996; Shekhar and Keim, 1997; Johnson and Shekhar, 2006) or 7.5% CO2 inhalation (Fitz et al., 2003).
Although “osmosensitive” circumventricular organs (CVOs) such as the organum vasculosum of the lamina terminalis (OVLT) appear to be critical for relaying the lactate signal to the DMH/PeF (Johnson and Gross, 1993; Shekhar and Keim, 1997; Thompson and Swanson, 1998), the specific regions of the DMH/PeF involved and the downstream neural pathways through which this signal is propagated to behavioral centers to induce anxiety and elicit increases in heart rate (HR), mean arterial blood pressure (MAP) and respiration rate (RR) are poorly understood. Since evidence from previous studies suggested that disinhibiting the DMH elicits cardioexcitatory responses by mobilizing sympathetic outflow [see review (DiMicco et al., 2002)] and resetting the sensitivity of the baroreflex, which is parasympathetically mediated [see review (McDowall et al., 2006)], we hypothesized that disinhibition of the DMH/PeF alters lactate-induced behavioral, respiratory and cardiovascular responses through efferent targets involved in central sympathetic and parasympathetic autonomic control (Dampney, 1994; Thompson and Swanson, 1998; Fontes et al., 2001).
To identify neural pathways involved in sodium lactate-induced panic-like responses, including specific regions of the DMH/PeF, we studied the effects of sodium lactate or vehicle infusions in rats with prior infusions of the GABA synthesis inhibitor l-allylglycine (l-AG; 2-amino-4-pentenoic acid, an inhibitor of glutamic acid decarboxylase), or the inactive isomer d-allylglycine (d-AG: controls), into the DMH/PeF. We then utilized a variety of immunohistochemical techniques to investigate immediate-early gene (c-Fos) induction in neurochemically and anatomically identified neuronal populations. We also conducted site-specific pharmacological interventions to confirm the involvement of these pathways.
Materials and Methods
Animals
Adult male Sprague-Dawley rats (300–350 g; Harlan Laboratories, Indianapolis, IN), were used in all experiments and were housed individually in plastic cages under standard environmental conditions (22 °C; 12/12 light/dark cycle; lights on at 7:00 A.M.) for 7–10 days prior to the surgical manipulations. Food and water were provided ad libitum. Animal care procedures were conducted in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals (NIH Publication no. 80–23) revised 1996 and the guidelines of the IUPUI Institutional Animal Care and Use Committee.
Surgical procedures
For experiments 1 and 4, rats were anesthetized with an isoflurane system (MGX Research Machine; Vetamic, Rossville, IN). MAP and HR were measured by a femoral arterial line connected to a telemetric probe which contained a pressure transducer [Cat. no. C50-PXT, Data Science International (DSI) St. Paul, MN] and monitored by DSI DATAQUEST software. For experiment 2 and 3, MAP, HR and respiration rate (RR) were monitored by a femoral arterial line attached to a pressure transducer connected to a Beckman R511 Dynograph (Beckman Instruments. Inc., Fullerton CA). In experiments 1 and 4 the HR and MAP data are changes in HR and MAP from the average of the baseline (t -5 to t -1) from each rat over 20 min. In experiments 2 and 3 the data are expressed as peak changes in HR or MAP, which was defined as the highest value sustained for over 1 min. All rats were fitted with venous catheters for i.v. infusions.
After 3 days of recovery, rats were tested for baseline cardiovascular and respiratory responses to lactate, then anesthetized and T-shaped 26 gauge cannulae (Cat. no. 3260PG, Plastics One, Ranoake VA) were directed at cardioexcitatory regions of the DMH (Samuels et al., 2004) based on the following stereotaxic coordinates relative to bregma (using a 10° angle from the vertical plane with the incisor bar set at +5 mm were: Anterior −1.2 mm, Lateral +2.5 mm and Ventral +7.0 mm. Fifty pmol/100 nl of bicuculline methiodide (BMI), a GABAA receptor antagonist, was injected through the vertical arm of the cannula to ascertain that the tip of the cannula was placed in a cardioexcitatory region (i.e., where BMI elicits ≥ 50 beats/min in HR). This microinjection commenced following an ~10 min stable baseline in HR and MAP. The cannulae were then retracted and filled with either l-AG or d-AG solution, then redirected to the previous coordinates. The osmotic minipumps (DURECT Corp. cat. no. 2002) were then attached to the cannula with PE-60 tubing and sutured into place subcutaneously at the nape of the neck. The concentration of the solutions in the minipumps was such that 3.5 nmol/0.5µl per hour of l-AG or d-AG was infused into the DMH region over 5 days prior to i.v. infusions. Previous studies have determined that the dose of l-AG utilized here reduces local GABA concentrations by approximately 60% following unilateral infusions [based on microdialysis study (Abshire et al., 1988), and enzyme assays (Shekhar and DiMicco, 1987; Shekhar et al., 1996; Shekhar and Keim, 1997; Shekhar et al., 2006) and supported by immunohistochemistry (Johnson and Shekhar, 2006) and increases anxiety-like behavior [i.e., as measured by the social interaction (SI) test and elevated plus-maze (EPM)] without increasing cardiorespiratory responses (Shekhar and DiMicco, 1987; Shekhar et al., 1996; Shekhar and Keim, 1997; Shekhar et al., 2006).
Sodium lactate infusion
Five days following stereotaxic surgery, rats randomly received i.v. infusions of 0.5M sodium lactate or 0.9% saline over 15 min, similar to clinical lactate infusions (Liebowitz et al., 1986). Cardiorespiratory responses were recorded continuously for 5 min prior to infusion (baseline) until 15 min following the onset of sodium lactate infusion.
Social interaction test
The SI test was utilized to measure anxiety-like behavior. This test was chosen for the following reasons: 1) the SI test is a fully validated test of experimental anxiety-like behavior in rats (File, 1980) and 2) the dose of l-AG used here increases anxiety-like behavior as measured by the SI test and also other measures of anxiety such as the EPM (Shekhar et al., 1996). Consequently, the SI test may be a sensitive measure of an increase in anxiety-like state following intra-DMH infusions of l-AG. The SI test procedure as used in our laboratory has been described previously (Shekhar and Katner, 1995; Johnson and Shekhar, 2006). See also supplemental methods for detailed description.
Experiment 1: Cellular responses in l-AG- or d-AG-treated rats following lactate or saline challenge
Perfusion
Methods for perfusion and for verification of cannulae placements in experiment 1 have been described previously (Johnson and Shekhar, 2006). Briefly, l-AG and d-AG rats received i.v. infusions of either lactate or saline vehicle over 15 min (n=6/group), were immediately tested in the SI test, then anesthetized and perfused with a 4% paraformaldehyde/1.5% sucrose solution and processed for immunohistochemistry 90 min following the onset of the i.v. infusions as described previously (Johnson and Shekhar, 2006). In the present study, four of the six alternate sets of 30 µm coronal brain sections were stained, one set each for c-Fos (full brain), c-Fos/tyrosine hydroxylase (TH, brainstem only), c-Fos/choline acetyltransferase (ChAT, medulla only), c-Fos/GAD67 (medulla only) and c-Fos/ neuronal marker n (NeuN, midbrain only)]. Immunostaining of TH, ChAT and GAD67 was done to identify catecholaminergic (i.e., norepinephrine- and epinephrine-synthesizing), acetycholinergic and GABAergic neurons, respectively. Noradrenergic (norepinephrine-synthesizing) and adrenergic (epinephrine-synthesizing) cell groups in the brainstem were defined according to a standard stereotaxic atlas of the rat brain [i.e., Paxinos and Watson (1997)] as described previously (Johnson et al., 2005).
Single/double immunohistochemistry
We used immunohistochemical staining for c-Fos, in the presence or absence of other neurochemical markers, to identify specific brain regions involved in responses to l-AG and sodium lactate. Measurement of c-Fos expression is a useful method of identifying functional cellular responses among large populations of neurons in the CNS. Despite some limitations, for example, 1) a lack of cellular c-Fos induction does not necessarily imply a lack of neuronal responses, and 2) measurement of c-Fos expression is not likely to reflect different levels of increased neuronal activity or subtle differences in the patterns of neuronal firing, this technique has been a useful tool in the functional mapping of neural circuitry mediating or modulating many stimuli, including stress-related stimuli [reviewed in (Herman et al., 1996; Herman and Cullinan, 1997; Senba and Ueyama, 1997; Kovacs, 1998)]
Due to the detailed rostrocaudal analyses of c-Fos double immunostaining, chromogens were utilized in place of immunofluorescence. Although fluorescence immunostaining is critical when co-localizing two or more cytoplasmic or nuclear substrates, two chemical substrates can be co-localized using chromogen reactions when one is nuclear and the other cytoplasmic or membrane associated. The use of chromogens, versus fluorescence, also allowed detailed and repeated analyses of archived slides and does not suffer from a reduced signal following routine imaging as is the case with fluorescence. Furthermore, white matter fiber tracts, which serve as anatomical landmarks for identification of the specific rostrocaudal levels for analysis, are easier to locate using sections with chromogen-based immunostaining. Immunostaining for c-Fos protein was accomplished using primary antiserum directed against c-Fos (rabbit anti-c-Fos-polyclonal, affinity-purified antibody, Cat. no. PC38, Ab-5, Calbiochem; diluted 1:10,000) as described previously (Johnson et al., 2005). The chromogen for immunostaining of c-Fos was SG (Cat. no. SK-4700, Vector Laboratories) and the reaction was run for 20 min.
For double immunostaining, immunostaining of c-Fos protein was done first and then, after the first chromogen reaction had been quenched using 1% H2O2, immunostaining of TH, ChAT, GAD67, or NeuN protein immunostaining was done second using primary antibodies/antiserum directed against c-Fos (as above) then TH (rabbit anti-TH-polyclonal, affinity-purified antibody, Cat no. B1562, Chemicon; diluted 1:10,000), ChAT (sheep anti-ChAT polyclonal, antiserum, Cat. no. AB1582, Chemicon, diluted 1:5000), GAD67 (mouse anti-GAD67, monoclonal affinity-purified antibody, Cat. no. AB5406, Chemicon, diluted 1:1500), or NeuN (mouse anti-NeuN monoclonal affinity-purified antibody, Cat. no. MAB377, Chemicon, diluted 1:10,000) using standard immunohistochemical methods as described previously (Johnson et al., 2005) and as described in supplemental methods.
Data generation from immunohistochemistry
Brain regions analyzed for the number of c-Fos-ir nuclei were chosen based on previous studies implicating them in fear circuitry (Singewald and Sharp, 2000; Singewald et al., 2003) and central autonomic and/or osmotic regulation [see reviews (Johnson and Gross, 1993; Dampney, 1994)]. Cell counts for each brain region are based on cell counts from a single brain section from each rat from each treatment group. Each brain section was selected to represent the specific rostrocaudal level, with reference to distance from bregma using a standard stereotaxic atlas of the adult rat brain (Paxinos and Watson, 1997) (see also Fig. S1). In cases where a specific brain region is represented bilaterally, the cell counts for the left and right sides were pooled for a single measurement. Details on data generation from single and double IHC are available in the supplemental methods.
Statistical analyses
Each dependent variable for in vivo analyses (i.e., SI duration, HR, MAP, RR) was analyzed using a two way ANOVA with repeated measures with lactate and isomer as main factors and time as repeated measures. In the presence of significant main effects or main effect × time interactions, post-hoc tests were conducted to define the anatomical location of the effects using Tukey’s HSD tests which corrects for multiple comparisons.
Because we have analyzed a large number of brain regions, we have taken several steps to minimize Type I errors (i.e., detecting a difference when no difference exists). Data for each dependent variable (c-Fos, c-Fos/TH, TH, c-Fos/ChAT, ChAT, c-Fos/NeuN and NeuN) were analyzed using a separate two way ANOVA with repeated measures with lactate and isomer as main factors and brain region as a repeated measure. In the presence of significant main effects or main effect × brain region interactions in the overall model, secondary two way ANOVAs with lactate and isomer as main factors were conducted for each brain region counted. In the presence of appropriate and significant main effects or interactions in both the overall and secondary ANOVAs, post hoc pairwise comparisons were conducted to define the anatomical location of the effects using Tukey’s HSD tests.
Since the full model in the two way ANOVA with repeated measures cannot be estimated if there are missing values, missing values were handled according to the methods of Petersen (Peterson, 1985). Calculated missing values were only used to conduct the overall multifactor ANOVA, and were not used for subsequent secondary ANOVAs, pairwise comparisons or in the report of the data. Details of missing values are described in supplemental methods.
All statistical analyses were carried out using SYSTAT 5.02 (SYSTAT Inc., San Jose, CA) and SPSS 14.0 (SPSS Inc., Chicago IL), and all graphs were generated using SigmaPlot 2001 (SPSS Inc.). Correlations were done for the number of c-Fos-ir cells and the SI duration or autonomic responses in all analyses. Only significant correlations are mentioned in the results.
Experiment 2: Muscimol into the DMH/PeF of l-AG+lactate-treated rats
All rats (n=6) were treated with l-AG as previously described and received unilateral injections of 100 pmol muscimol, a GABAA receptor agonist, in aCSF, or 100nl of aCSF alone (in random order) into the DMH/PeF [using a 33 gauge injector (cat. no. C315I, Plastics One), which extended 1mm past the 24 gauge cannula (cat. no. C315G, Plastics One)] 30 min prior to 0.5 M sodium lactate infusions. 48 hrs was allowed between randomized injections.
Experiment 3: Muscimol into the BNST of l-AG+lactate-treated rats
All rats (n=5) were treated with l-AG into the DMH/PeF as previously described and received unilateral injections of muscimol or saline directed at the BNST 30 min prior to receiving i.v. infusions of 0.5 M sodium lactate. Stereotaxic coordinates relative to bregma for the BNST, using a 10° angle from the vertical plane with the incisor bar set at +5 mm were: Anterior +1.0 mm, Lateral +2.5 mm and Ventral −7.0 mm. On post-pump day 5, l-AG-treated rats were injected, in random order, with either aCSF (100 nl) or muscimol (250 pmol/100 nl) directly into the BSNT [using a 33 gauge injector (cat. no. C315I, Plastics One), which extended 1mm past the 24 gauge cannula (cat. no. C315G, Plastics One)]. 48 h was allowed between randomized injections.
Experiment 4: Intraperitoneal atenolol in l-AG + lactate-treated rats
In order to confirm peripheral sympathetic contributions to panic-like cardiovascular responses, all rats (n=5) were treated with l-AG into the DMH/PeF as previously described and received intraperitoneal (i.p.) injections of 10 mg/kg atenolol 30 min prior to receiving i.v. infusions of 0.5 M sodium lactate. Cardiovascular data were obtained as previously described in methods after rats received 1) l-AG + i.v. lactate, 2) l-AG + i.p. atenolol + i.v. saline or 3) l-AG + i.p. atenolol + i.v. lactate. 48 hrs was allowed between each of the randomized infusions of saline+atenolol or lactate+atenolol or lactate alone.
Results
Experiment 1: Cellular responses in l-AG or d-AG rats following lactate or saline challenge
Behavioral and cardio-respiratory responses in l-AG or d-AG treated rats following i.v. challenges
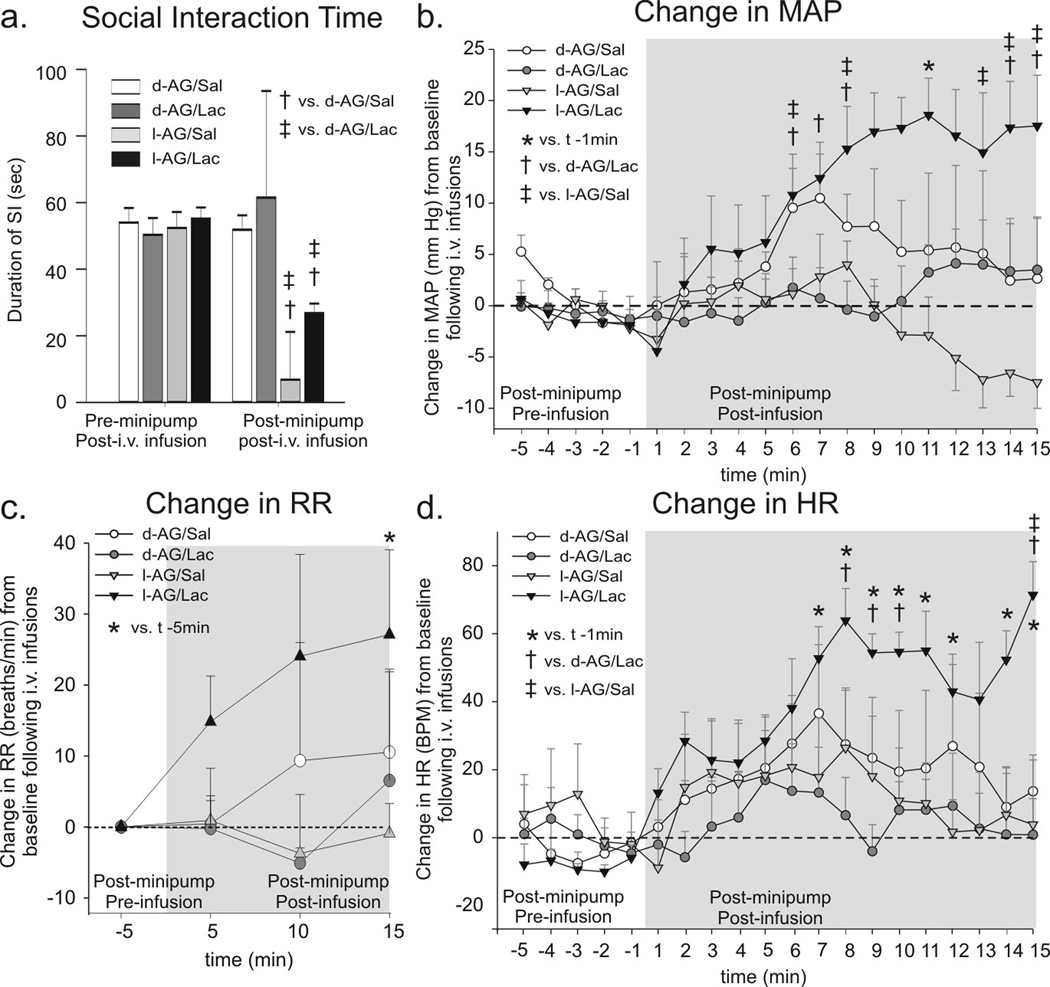
Part of the physiological responses (maximal changes in HR and MAP following i.v. saline or sodium lactate infusions, before and after intra-DMH l-AG or d-AG infusions) from experiment 1 were published previously (Johnson and Shekhar, 2006). In the present article, we wanted to determine the onset and duration of the cardiorespiratory responses following the sodium lactate infusions. We also present for the first time the SI test data and the RR data, which we were able to obtain as an indirect measurement following analysis of normal sinus arrhythmia. Rats with l-AG infusions into the DMH/PeF region for 5 days had less SI (an anxiogenic effect) in the SI test measured immediately after a 20 min i.v. infusion of either vehicle or sodium lactate compared to pre-pump baseline behavior and to d-AG control rats [(Fig. 1a), lactate × isomer × time interaction (F(1,18) = 9.7, P = 0.006)]. There were no significant differences in baseline SI times among treatment groups prior to either d- or l-AG infusions.
Figure 1.
The effect of GABA synthesis inhibition in the DMH region and iv. lactate infusions on anxiety-like behavioral and cardiorespiratory responses from experiment 1. a) Social interaction (SI) time pre and post l-AG (GABA synthesis inhibitor)- or d-AG (control isomer) treatment in rats also receiving i.v. infusions of saline (Sal) or lactate (Lac). Change in b) mean arterial blood pressure (MAP), c) respiration rate (RR), d) and heart rate (HR) from 5 min baseline post-i.v. infusion. The RR data are presented as change from baseline for consistency, but the * symbol indicates a significant difference in actual RR over time. Bars or lines represent the mean + S.E.M. No differences were noted between baseline levels of MAP, RR and HR for each group (data not shown).
Between-subjects analyses revealed that rats with intra-DMH/PeF l-AG infusions, but not d-AG controls, responded to lactate infusions with hypertension [Fig. 1b; lactate × isomer × time interaction (F(192,285) = 4.1, P < 0.001)], tachypnea [Fig. 1c; lactate effect (F(3,16) = 4.0, P < 0.05)] and tachycardia [Fig. 1d; lactate × isomer × time interaction (F(12,285) = 2.6, P < 0.001)]. Within-subjects analyses revealed that that only l-AG+lactate treated rats had an increase in MAP (time effect, F(19,80) = 4.7, P < 0.001), and HR (time effect, F(19,80) = 1.7, P < 0.05) from baseline and an increase in RR (time effect, F(3,16) = 4.0, P < 0.05) over time following i.v. infusions. There were no differences in baseline HR, MAP or RR among treatment groups prior to l-AG or after d-AG treatment (data not shown) and there was no change in the duration of social interaction (Fig. 1a) or cardiorespiratory arousal following lactate infusions in d-AG control rats (Fig. 1b–d).
All minipump cannulae placements resided in regions of the DMH/PeF and posterior hypothalamus (PH) known to be cardioexcitatory and also verified for each infusion site in the present study to be cardioexcitatory (see methods). An illustration of the minipump placements for experiment 1 has been previously published (Johnson and Shekhar, 2006). Some tissue damage at the site of implantation was noted due to the 26 gauge cannula (0.46 mm o.d.), but this damage was equal in the control and experimental groups (data not shown). Previous analyses of the total numbers of N-methyl-d-aspartate (NMDA) and alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate (AMPA) receptor-ir neurons in the DMH did not differ between the side of the DMH which had the cannula track compared to the intact DMH on the contralateral side (Johnson and Shekhar, 2006). Overall this suggests that the tissue damage within the DMH was minimal. Therefore, any treatment effects are unlikely to be due to mechanical damage of the DMH due to the cannula.
Overall analyses of c-Fos cell counts following single immunostaining in experiment 1
Brain regions analyzed for the number of c-Fos-ir nuclei were chosen based on previous studies implicating them in fear circuitry (Singewald and Sharp, 2000; Singewald et al., 2003) and central autonomic and/or osmotic regulation [see reviews (Johnson and Gross, 1993; Dampney, 1994)]. Analysis of the number of c-Fos-ir nuclei in all brain regions analyzed using two-factor ANOVA with repeated measures revealed a lactate (F (1,19)=12.0, P = 0.003) and isomer (F(1,19) = 4.8, P = 0.041) effect, but no lactate × isomer interaction (F(1,19) = 1.2, P = 0.279). Additionally, the effects of both lactate and isomer treatment on the number of c-Fos-ir cells were dependent on brain region [lactate × brain region interaction (F(66,1254) = 6.9, P < 0.001); isomer × brain region interaction (F(66,1254) = 3.3, P < 0.001)]. Furthermore, a lactate × isomer × brain region interaction was observed [(F(66,1254) = 2.7, P < 0.001)]. Overall, l-AG-treated rats, but not d-AG-treated rats, had altered c-Fos responses in some brain regions, but not others, in response to sodium lactate infusions. Detailed descriptions of these effects within each brain region are discussed below. Secondary two-way ANOVAs for each brain region with lactate and isomer as main factors are listed in Table S1a–d.
Circumventricular organs implicated in “sensing” sodium lactate
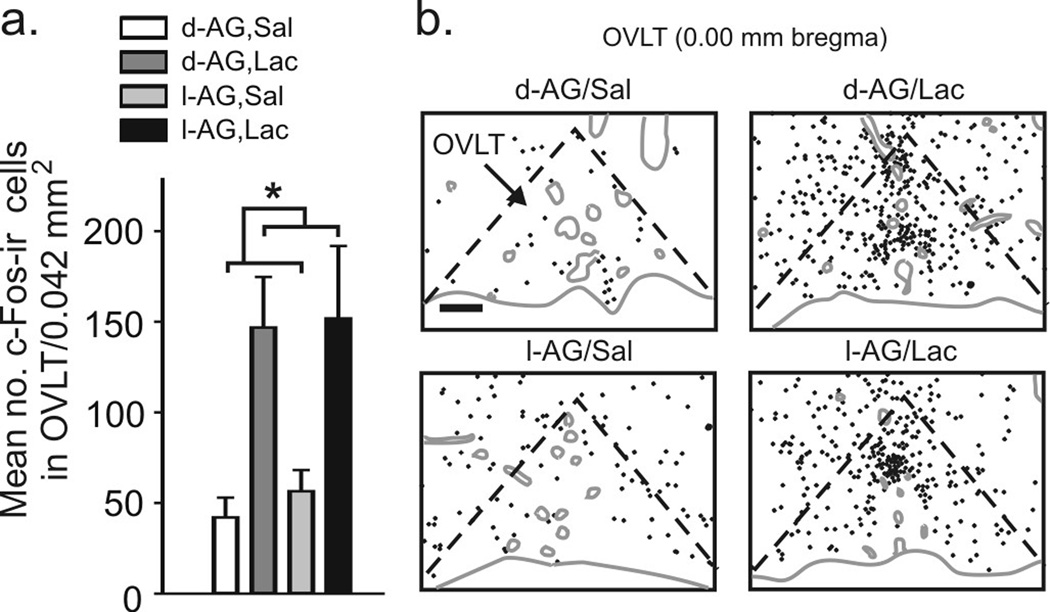
Experiment 1: Lactate infusions increased the number of c-Fos-ir cells in the OVLT (Fig. 2a,b), subfornical organ (SFO) and area postrema (AP) in both l-AG- and d-AG-treated rats (Table 1). However, as predicted, c-Fos responses in the OVLT, SFO and AP did not correlate with SI duration or cardiorespiratory responses (see supplemental results for details).
Figure 2.
Graphs and illustrations of c-Fos-ir cells in the organum vasculosum lamina terminalis (OVLT) from experiment 1. a) Graph illustrates mean (± SEM) numbers of c-Fos-ir cells in the OVLT following i.v. infusions of saline (Sal) or lactate (Lac) in rats previously treated with d- or l-AG infusions into the DMH/PeF region (* P < 0.05). b) Illustrations of c-Fos-ir cells (each black dot = 1 c-Fos-ir cell) in the OVLT of a rat in each treatment group. Gray solid lines indicate the outline of the brain section and blood vessels. Black dashed lines indicate the OVLT. Scale bar: 136 µm.
Table 1.
Mean numbers of single c-Fos-ir cells in anatomically defined subdivisions of the central nervous system following either i.v. infusions of saline (Sal) or sodium lactate (Lac) in rats with prior infusions of d-AG or l-AG into the DMH region from experiment 1 (see table S2 for rostral amygdala data). Column 1 indicates brain regions; no. in parentheses indicate anatomical levels relative to bregma. Columns 2–5 represent a) the no. of c-Fos-ir cells. The titles above columns indicate treatment groups, d-AG/Sal, d-AG/Lac, l-AG/Sal or l-AG/Lac, respectively. Values below columns 2–5 are the mean ± S.E.M. Letters, symbols and boxes adjacent to column 5 are defined in figure legend in table. See methods section for full list of abbreviations.
| single c-Fos-ir cells | d-AG/Sal | d-AG/Lac | I-AG/Sal | I-AG/Lac | |
|---|---|---|---|---|---|
| CVOs | |||||
| OVLT (0.00 mm bregma) | 42 ± 11 | 147 ± 28 | 56 ± 12 | 152 ± 40 | |
| SFO (−0.92 mm bregma) | 11 ± 4 | 66 ± 27 | 43 ± 15 | 94 ± 24 | |
| AP (−13.80 mm bregma) | 4 ± 1 | 29 ± 12 | 3 ± 1 | 46 ± 19 | |
| DMH (−3.14 mm bregma) | |||||
| mDMH ipsi | 38 ± 11 | 32 ± 11 | 60 ± 10 | 50 ± 8 | |
| DA ipsi | 26 ± 8 | 33 ± 10 | 53 ± 11 | 71 ± 12 |
* |
| IDMH ipsi | 48 ± 17 | 50 ± 16 | 57 ± 8 | 105 ± 9c |
# † ‡ |
| mDMH contra | 40 ± 12 | 63 ± 17 | 74 ± 5 | 99 ± 20 | |
| DA contra | 45 ± 13 | 44 ± 12 | 67 ± 10 | 99 ± 13c | |
| IDMH contra | 63 ± 15 | 59 ± 13 | 96 ± 11 | 97 ± 17 | |
| PVN (−1.80 mm bregma) | |||||
| PaDC | 12 ± 3 | 26 ± 6 | 25 ± 4 | 61 ± 19 | |
| PaV | 35 ± 7 | 92 ± 19 | 58 ± 7 | 145 ± 33b | |
| PaMP | 42 ± 8 | 151 ± 40 | 104 ± 12 | 203 ± 42 | |
| PaLM | 51 ± 17 | 188 ± 51 | 69 ± 12 | 318 ± 67b | |
| SON (1.80 mm bregma) | 88 ± 35 | 146 ± 35 | 91 ± 26 | 312 ± 71b |
† ‡ |
| Amygdala (−2.56mm bregma) | |||||
| CeC | 70 ± 31 | 89 ± 18 | 66 ± 19 | 160 ± 28 |
† |
| CeL | 28 ± 6 | 62 ± 18 | 34 ± 13 | 116 ± 17b |
† |
| CeM | 42 ± 14 | 37 ± 8 | 36 ± 6 | 62 ± 11 | |
| BLA | 70 ± 13 | 65 ± 16 | 93 ± 11 | 109 ± 11 |
* |
| BLP | 19 ± 5 | 17 ± 3 | 15 ± 1 | 34 ± 11 | |
| LA | 68 ± 19 | 81 ± 24 | 83 ± 12 | 119 ± 21 | |
| BNST (+0.20 mm bregma) | |||||
| BSTL | 28 ± 10 | 28 ± 8 | 29 ± 3 | 37 ± 6 | |
| BSTM | 41 ± 11 | 60 ± 17 | 89 ± 11 | 90 ± 8 |
* |
| LSV | 103 ± 33 | 144 ± 43 | 196 ± 6 | 245 ± 40 |
* |
| BNST (−0.26 mm bregma) | |||||
| BSTL | 38 ± 13 | 40 ± 8 | 34 ± 12 | 97 ± 14 |
† |
| BSTM | 40 ± 13 | 44 ± 13 | 34 ± 3 | 88 ± 12 | |
| LSV | 41 ± 12 | 68 ± 26 | 68 ± 15 | 139 ± 27 | |
| PBN (−9.30 mm bregma) | 109 ± 28 | 120 ± 29 | 87 ± 10 | 419 ± 80b, c |
# |
| RPa | |||||
| (−11.30 mm bregma) | 26 ± 5 | 64 ± 20 | 27 ± 6 | 46 ± 8 | |
| (−11.60 mm bregma) | 14 ± 4 | 43 ± 11a | 16 ± 4 | 15 ± 5 | |
| Dorsomedial medulla nTS | |||||
| (−13.30 mm bregma) | 23 ± 7 | 77 ± 16 | 32 ± 11 | 153 ± 54 | |
| (−13.68 mm bregma) | 33 ± 7 | 74 ± 14 | 18 ± 4 | 144 ± 29b, c |
† ‡ |
| (−14.20 mm bregma) | 11 ± 2 | 33 ± 13 | 14 ± 7 | 49 ± 20 | |
| DMV (−13.68 mm bregma) | 6 ± 2 | 17 ± 6 | 4 ± 2 | 29 ± 14 |
# |
 non-responsive
non-responsive
 Lac only
Lac only
 I-AG only
I-AG only
 Lac + I-AG and/or correlates with panic like responses
Lac + I-AG and/or correlates with panic like responses
vs d-AG/Sal
vs I-AG/Sal
vs d-AG/Lac
correlates with HR following i.v. infusions
correlates with MAP following i.v. infusions
correlates with RR following i.v. infusions
inversely correlated with change in SI from pre I-AG to post I-AG treatment
c-Fos responses in CNS regions implicated in regulating anxiety-like behavior
Rostral BNST/LSV and BLA
Infusing l-AG in the DMH/PeF, regardless of lactate challenge, increased the number of c-Fos-ir cells in the medial BNST (BSTM) and the ventral part of the lateral septum (LSV), but not in the rostral part of the lateral BNST (BSTL) following exposure to the SI test, compared to d-AG-treated rats (Table 1). Furthermore, analysis of data from all rats in the study revealed that c-Fos responses in the BSTM were inversely correlated with the change in the duration of SI from pre-intra-DMH/PeF to post-intra-DMH/PeF infusions (see supplemental results for details).
Cellular c-Fos responses in the caudal basolateral amygdala (BLA) were similar to the patterns observed in the BSTM. Here l-AG treatment and exposure to the SI test, irrespective of lactate challenge, increased the number of c-Fos-ir cells (Table 1). The number of c-Fos-ir cells in the BLA negatively correlated with the duration of SI from pre-intra-DMH/PeF infusions to post-intra-DMH/PeF infusions (r2 = 0.20, P = 0.040).
c-Fos responses in CNS regions implicated in autonomic/respiratory regulation
Caudal BNST/LSV and CeA
A greater number of c-Fos-ir cells were observed in the caudal BSTL and LSV of l-AG+lactate-treated rats compared to other groups (Table 1). In addition, c-Fos responses in the caudal BSTL were positively correlated with the mean change in HR over the 15 min following i.v. infusions (r2 = 0.33, P = 0.012).
In contrast to the BLA, lactate increased the number of c-Fos-ir cells in the rostral capsular, lateral and medial parts of the central amygdala (CeC, CeL and CeM, respectively; Table 1) and in the caudal CeC and CeL (Table S2, Fig. S2a–b) in l-AG, not d-AG,-treated rats. In addition, c-Fos responses in the rostral CeM (r2=0.27, P = 0.023) and caudal CeC (r2 = 0.30, P = 0.016) and CeL (r2 = 0.32, P = 0.011) were positively correlated with the mean change in HR over the 15 min following i.v. infusions (see Table 1 and S2).
Although c-Fos responses to lactate+l-AG tended to occur throughout the rostro-caudal extent of the amygdala (with the exception being the BLA, where responses were clearly restricted to the caudal part of the nucleus), these responses did not reach statistical significance at all levels nor did they correlate with changes in SI behavior or cardio-respiratory responses.
Dorsomedial medulla (DMM) systems (nTS, DMV)
As observed in the lDMH, rats with lactate infusions had increased c-Fos responses within the DMM [i.e., nTS, dorsal motor nucleus of the vagus (DMV)] of “panic-prone” (l-AG-treated) rats but not d-AG-treated rats (Table 1 & Fig. S2c–d). Furthermore, when data from all rats in the study were considered, c-Fos responses in the nTS at −13.68 mm bregma were positively correlated with the mean change in cardiovascular responses following i.v. infusions [HR (r2 = 022, P = 0.040); MAP (r2 = 0.23, P = 0.044); Table 1]. Responses in the DMV were positively correlated with RR responses following i.v. infusions (r2=0.30, P = 0.033).
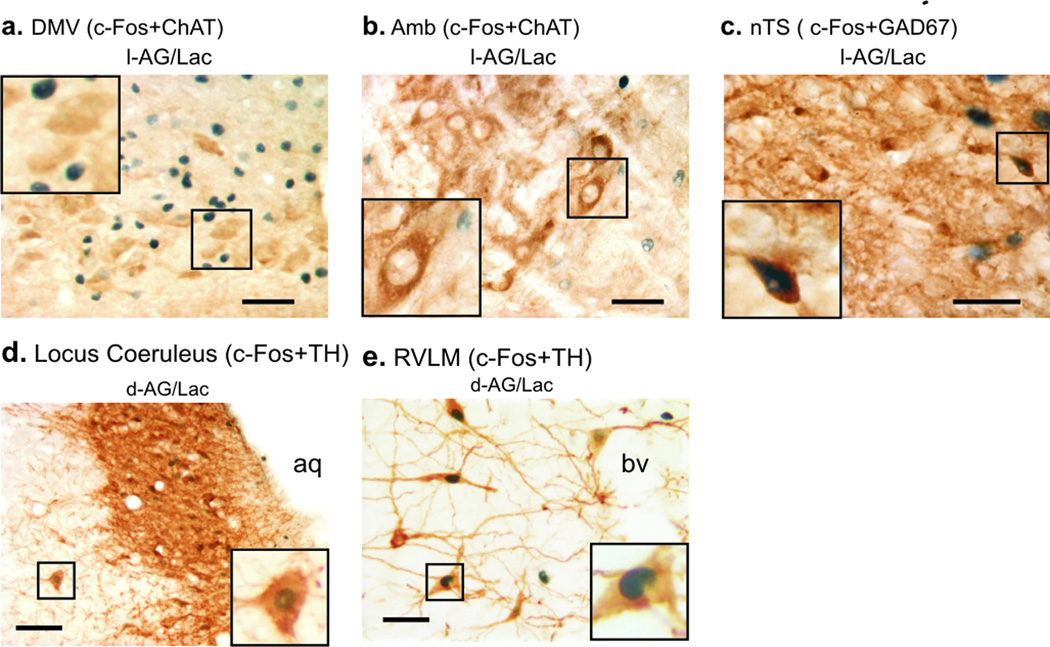
In addition to overall c-Fos analysis, c-Fos expression in the acetylcholinergic parasympathetic preganglionic neurons [c-Fos + choline acetyltransferase (ChAT)] of the DMV and the Amb were investigated. Consistent with the previous immunohistochemical analysis, lactate infusion, compared to vehicle infusion, increased numbers of c-Fos-ir cells in the DMV of l-AG-, but not d-AG-, treated rats (lactate effect; F(1,20) = 11.05, P = 0.003; Table 2b). However, these increases in c-Fos were not occurring in ChAT-ir neurons in either the DMV or Amb (Table 2b & Fig. 3a–b, respectively). No differences in the total numbers of ChAT-ir cells within the DMV or Amb were observed among treatment groups.
Table 2.
Mean numbers of c-Fos-ir cells in anatomically and/or chemically defined subdivisions of the central nervous system following either i.v. infusions of saline (Sal) or sodium lactate (Lac) in rats with prior infusions of d- or l-allylglycine (d- or l-AG) into the DMH region from experiment 1. For table a), b), and c) column 1 indicates brain regions; numbers in parentheses indicate anatomical levels relative to bregma. Columns 2–5 represent a) the no. of c-Fos/tyrosine-hydroxylase (TH)-ir neurons; b) the no. of c-Fos/choline acetyltransferase (ChAT)-ir neurons; and c) the no. of c-Fos/neuronal marker n (NeuN)-ir neurons. No significant differences in the total no. of TH, ChAT or NeuN-ir neurons were observed between groups in any subregion analyzed (see results section for details). The titles above columns 2–5 for a), b) and c) indicate treatment groups, d-AG/Sal, d-AG/Lac, l-AG/Sal or l-AG/Lac, respectively. Values below columns 2–5 are the mean ± S.E.M. Letters, symbols and boxes adjacent to column 5 are defined in figure legend in table. See methods section for full list of abbreviations.
| a.c-Fos/TH-ir neurons | d-AG/Sal | d-AG/Lac | I-AG/Sal | I-AG/Lac | |
|---|---|---|---|---|---|
| Pons | |||||
|
A6 (−10.04 mm bregma) no.c-Fos/TH |
29 ± 9 | 85 ± 18 | 32 ± 12 | 74 ± 25 | |
| Ventrolateral Medulla | |||||
|
C1 (−11.80 mm bregma) no. c-Fos/TH |
3 ± 1 | 16 ± 4 | 5 ± 4 | 17 ± 5 | |
|
C1 (−11.96 mm bregma) no. c-Fos/TH |
3 ± 1 | 17 ± 4 | 7 ± 4 | 20 ± 6 | |
|
C1 (−12.72 mm bregma) no. c-Fos/TH |
6 ± 2 | 17 ± 2a | 6 ± 1 | 23 ± 4b | |
|
A1C1 (−13.68 mm bregma) no. c-Fos/TH |
15 ± 4 | 20 ± 3 | 20 ± 5 | 25 ± 3 | |
| Dorsomedial Medulla | |||||
|
C2 (−13.24 mm bregma) no. c-Fos/TH |
7 ± 2 | 17 ± 3a | 10 ± 3 | 17 ± 2 | |
|
C2 (−13.30 mm bregma) no. c-Fos/TH |
13 ± 3 | 21 ± 4 | 7 ± 1 | 19 ± 2b | |
| b. c-Fos/ChAT-ir neurons | |||||
|
DMV (13.68 mm bregma) no. c-Fos/ChAT |
1 ± 1 | 1 ± 1 | 0 ± 0 | 3 ± 2 | |
|
Amb (11.80 mm bregma) no. c-Fos/ChAT |
0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| c. c-Fos/NeuN-ir neurons | |||||
|
PBN (9.30 mm bregma) no. c-Fos/NeuN |
16 ± 4 | 16 ± 4 | 5 ± 1 | 80 ± 22 | |
![]() non-responsive
non-responsive
 Lac only
Lac only
![]() Lac + I-AG and/or correlates with panic-like responses
Lac + I-AG and/or correlates with panic-like responses
vs d-AG/Sal
vs I-AG/Sal
Figure 3.
Photomicrographs of cellular responses [i.e., c-Fos induction; blue/black immunostaining of nuclei] in neurochemically identified cell groups studied in experiment 1 represented brown cytoplasmic immunostaining. Photographs of c-Fos and choline acetyltransferase (ChAT) immunostaining in the a) dorsal motor nucleus of the vagus (DMV) and b) ambiguus nucleus (Amb) in lactate (Lac) + l-AG-treated rats. Photographs of c) c-Fos and glutamic acid decarboxylase (GAD67) immunostaining in the nucleus of the solitary tract (nTS) of a lactate+l-AG treated rat. Photomicrographs illustrating c-Fos and tyrosine hydroxylase (TH) immunostaining of d) pontine A6 noradrenergic cell group in the locus coeruleus (LC, −10.04 mm bregma) and e) rostroventrolateral medulla (RVLM) C1 adrenergic cell group (−11.96 mm bregma) in saline- (Sal, left) and Lac (right) infused rats with prior infusions of d-allylglycine (d-AG) in the DMH/PeF. Insets at the bottom of low magnification photographs contain higher magnification photographs. Scale bar: 50 µm; lower left insert; a–b) 25 µm, c) 22 µm, d–e) 20 µm.
To determine if the increases in c-Fos expression in these regions were in local GABAergic inhibitory neurons, we performed double immunostaining for GAD67 and c-Fos. Dense synaptic bouton-like staining, as evidenced by small GAD67-ir granules, made visualizing and accurately counting c-Fos/GAD67 double-ir cells difficult and thus a quantitative analysis was not performed. However, there were examples of c-Fos/GAD67-ir cells noted in the nTS of l-AG+lactate-treated rats (Fig. 3c).
Paraventricular hypothalamic nucleus (PVN) and supraoptic nucleus (SON)
Within the PVN, l-AG+lactate-treated rats had increased c-Fos responses in the dorsal cap and ventral PVN (PaDC and PaV, respectively; Table 1 and Fig. S3a,c) compared to other groups. However, c-Fos responses in the PaDC did not correlate with behavioral or cardiorespiratory responses. Lactate infusions also increased c-Fos responses in the lateral magnocellular and medial parvicellular PVN (PaLM and PaMP, respectively) in both l-AG- and d-AG-treated rats. Lactate increased c-Fos responses in the SON, but only in l-AG-treated rats (Table 1). Furthermore, c-Fos responses in the SON were positively correlated with HR (r2 = 0.42, P = 0.003) and MAP (r2 = 0.37, P = 0.007) following i.v. infusions.
Parabrachial nucleus (PBN)
Sodium lactate increased c-Fos responses in the PBN (Table 1, Fig. S3b,d) but only in l-AG-treated rats. In addition, the number of c-Fos-ir cells in the PBN was positively correlated with RR (r2=0.26, P = 0.027), but not HR, MAP or SI duration responses following i.v. infusions (see Table 1).
An estimation of c-Fos induction in neurons in a brain region that showed a robust l-AG+lactate effect (i.e., the PBN) revealed that l-AG+lactate induction of c-Fos in the ventrolateral PBN was exclusively occurring in neurons (see Table S1d and Table 2c). There was no evidence for increased c-Fos expression in non-neuronal cells.
Other CNS regions that were non-responsive or where responses did not correlate with panic-like responses
Surprisingly, no subdivision of the midbrain periaqueductal gray (PAG), or ventrolateral medulla [i.e., Amb, PreBo, LRt, LPGi, CVLM, and RVLM (with the exception of C1 adrenergic neurons)] responded to any independent variable (see Table S3). An increased c-Fos response was observed in the raphe pallidus (RPa) in response to lactate, but only in d-AG-treated control rats (see Table 1). As predicted control sites such as the hypoglossal nucleus (12) expressed low levels of c-Fos in all treatment groups (see Table 1).
Pontine and medullary noradrenergic and adrenergic systems
Lactate-treated rats had altered c-Fos responses in specific noradrenergic and adrenergic (TH-ir) cell groups involved in central autonomic control. Overall, the numbers of TH-ir neurons within neuronal cell groups were largely unaffected by either l-AG or lactate infusion (Table 2a & Table S4).
Lactate infusions increased the number of c-Fos/TH-ir neurons in the A6 cell group (LC) at −10.04 mm bregma in d-AG-, but not l-AG,-treated rats (Table 2a & Table S4, Fig. 3d). Neither lactate nor l-AG infusions altered the number of c-Fos/TH-ir neurons in any other pontine noradrenergic cell group (see Fig. S1).
Lactate infusions increased the number of c-Fos/TH-ir neurons throughout the rostrocaudal extent of both the C1 (located in the rostroventrolateral medulla (RVLM); Table 2a, Fig. 3e & Table S4) and C2 (located in the DMM; Table 2a & Table S4) cell groups in both l-AG- and d-AG-treated rats. Neither l-AG nor lactate altered the number of c-Fos/TH-ir neurons in the C3, A1/C1, or A1 cell groups (see Fig. S1).
Intra- and inter-rater analyses of c-Fos-ir cell counts
Intra-rater and inter-rater analyses of c-Fos-ir cell counts for the OVLT and the CeA revealed that correlations between cell counts were highly significant. The intra-rater analyses of c-Fos counts revealed an r2 = 0.94, P < 0.001 for the OVLT; r2 = 0.97 and P < 0.001 for the CeA. The inter-rater analyses of c-Fos counts revealed an r2 = 0.88, P < 0.001 for the OVLT and an r2=0.98, P < 0.001 for the CeA (data not shown).
Experiment 2: Restoring GABAergic tone in the DMH/PeF blocks l-AG+lactate-induced panic responses
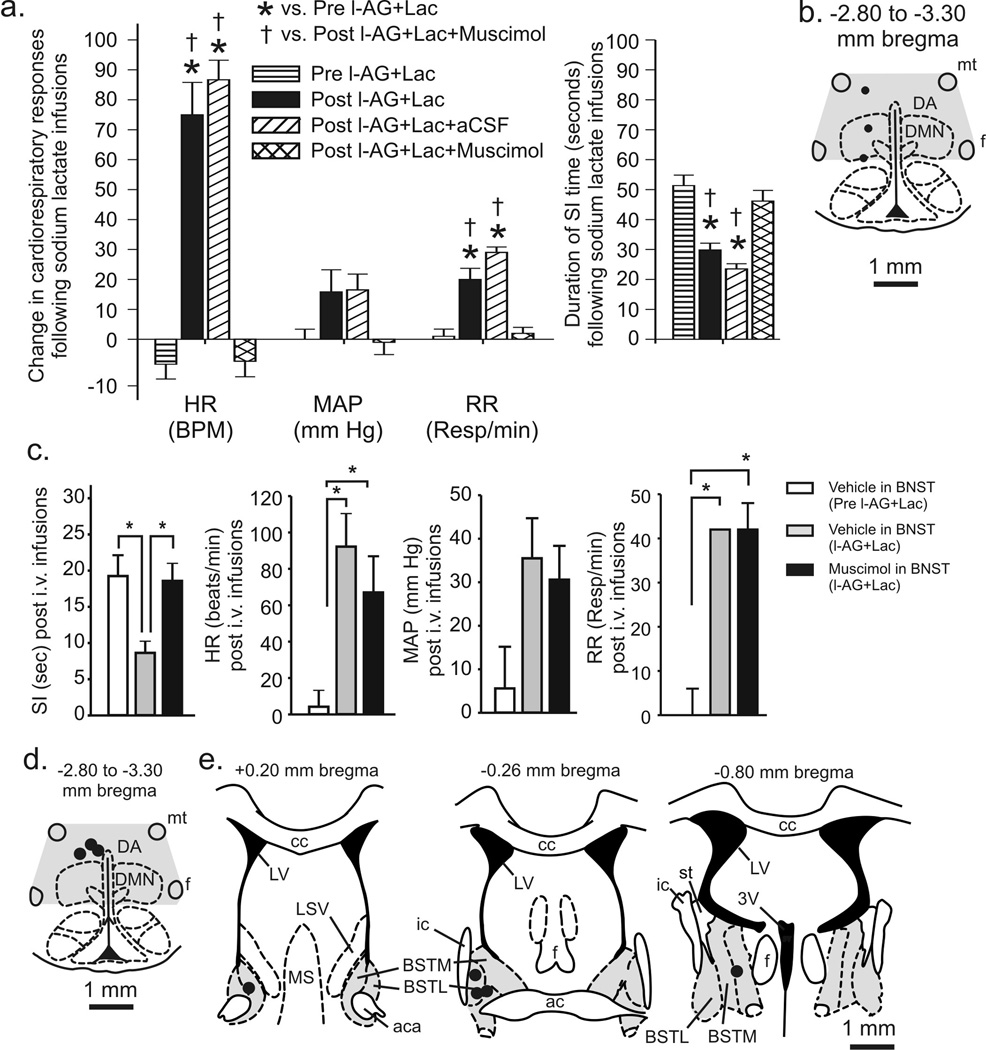
To determine if restoring GABA signaling in the DMH/PeF of l-AG-treated rats was sufficient to block the lactate-induced panic-like effects, injections of muscimol, a GABAA receptor agonist, into the DMH/PeF region 30 min before the onset of lactate infusion blocked l-AG+lactate-induced increases in HR (F(3,20) = 50.2, P < 0.001) and RR (F(3,20) = 28.4, P < 0.001) and blocked l-AG+lactate-induced decreases in SI duration (Fig. 4a; F(3,20) = 21.2, P < 0.001). The l-AG+lactate treatment altered the MAP (F(3,20) = 3.4, P = 0.039), but Tukey’s did not detect a significant change among groups. Pharmacological and histological analyses, as detailed in methods, confirmed successful cannulae implantation in the DMH/PeF region (Fig. 4b).
Figure 4.
Effects of restoring GABAergic tone in the DMH (experiment 2) or bed nucleus of the stria terminalis (BNST, experiment 3) on l-AG+lactate (Lac)-induced behavioral and/or cardiorespiratory responses. a) Graph illustrates the effects of injecting muscimol or artificial cerebrospinal fluid (aCSF) into the DMH region of l-AG+Lac treated rats. Bars represent the mean ± S.E.M. [* (versus Pre l-AG+Lac) P < 0.001, † (versus Post l-AG+Lac+Muscimol) P < 0.01)]. b) Illustration of cannula placements in the DMH region for experiment 2. Black circles represent cannula placements verified histologically (n=3) and gray shading indicates cannula placements from tissue that was micropunched for future neurochemical analyses (n=3). c) First graph, SI duration prior to and after l-AG infusions with aCSF or muscimol injections targeting the BNST followed by i.v. sodium lactate infusions. The following graphs illustrate the changes in the MAP, HR and RR in response to lactate prior to and after l-AG infusions in the DMH and with aCSF or muscimol injections targeting the BNST in experiment 3; (* P<0.05). d) Illustration of cannula placements in the DMH region for experiment 3. Black circle represents one histological placement (n=3) and gray shading represents distribution of cannula placements from tissue micropunched for neurochemical analysis (n=2). e) Illustration of the distribution injection sites in the BNST in experiment 3. Gray shading represents the BNST and black circles indicate aCSF or muscimol injection sites (n=5) verified with histology. See methods section for full list of abbreviations.
Experiment 3: The BNST regulates anxiety-like responses in panic model
To determine if excitation of the BNST is involved with the anxiety-like responses but not cardiorespiratory responses to sodium lactate challenge in l-AG-treated rats, muscimol or vehicle injections were administered to the BNST 30 min prior to a lactate challenge in l-AG treated rats. As reported in Experiments 1–3, l-AG-treated rats had reduced SI duration (F(2,11) = 6.0, P = 0.017, n=5) and increased HR (F(2,11) = 9.3, P = 0.004, n=5) and RR (F(2,3) = 24.5, P = 0.014, n=2) but not MAP (F(2,10) = 3.3, P = 0.078, n=4) in response to lactate, compared to pre-l-AG baseline measurements (Fig. 4c). Post hoc tests revealed that unilateral injections of 250 pmol muscimol (100nl), but not vehicle, into the BNST blocked l-AG-induced decreases in SI duration in the presence of lactate but had no effect on lactate-induced increases in HR or RR in l-AG-treated rats (Fig. 4c). Histological examination confirmed successful implantation of cannulae in the DMH/PeF region (Fig. 4d) and in the BNST region (Fig. 4e).
Experiment 4: Intravenous atenolol infusions block tachycardia responses
In order to verify that a peripheral sympathetic response occured in lactate+l-AG treated rats, systemic application of the selective β1-adrenergic blocker, atenolol was administered i.p. prior to the lactate challenge in “panic-prone” (l-AG-treated) rats. As reported above, challenging l-AG-treated rats with lactate increased cardiovascular responses [MAP, time effect, F(19,100) = 3.3, P < 0.001 (5 min baseline + 15 min post infusion); HR, time effect, F(9,50) = 3.2, P < 0.005 (only when comparing 5 min baseline + 5 min post infusion)](see Fig. S4a,b). Another group of l-AG+lactate treated rats that also received i.v. atenolol had the expected hypertension [time effect, F(19,80) = 1.8, P < 0.05 (5 min baseline + 15 min post infusion)], but failed to have a tachycardia response [time effect, F(11,48) = 0.2, P = 0.997 (5 min baseline + 5 min post infusion)]. Treating l-AG+saline-treated rats with i.v. atenolol did not alter MAP [time effect, F(19,80) = 0.6, P = 0.888 (5 min baseline + 15 min post infusion)] or HR [MAP, time effect, F(11,48) = 0.4, P = 0.950 (5 min baseline + 5 min post infusion)] responses from baseline. The mean baseline values for each treatment group did not differ (see Fig. S4a–b for baselines). Histological examination confirmed successful implantation of cannulae in the DMH/PeF region (Fig. S4c).
Discussion
DMH/PeF as one putative site coordinating panic-like responses
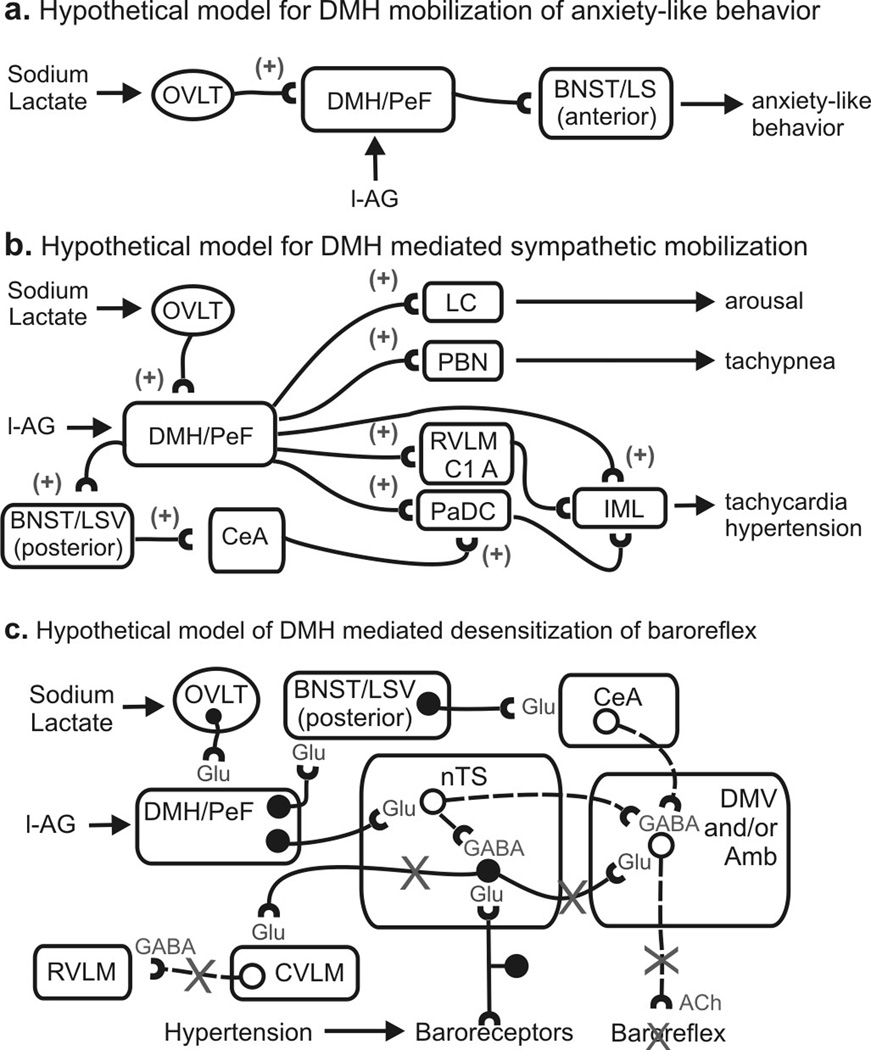
The hypothalamus has long been implicated in generating a coordinated emotional arousal [see review (DiMicco et al., 2002)]. More specifically, blockade of inhibitory GABA neurotransmission in the DMH/PeF elicits increases in HR and RR, BP, plasma catecholamine, adrenocorticotrophic hormone, and corticosterone concentrations, intestinal and colonic motility, and a pattern of peripheral blood flow that is similar to the pattern of peripheral blood flow during the defense reaction [see review (DiMicco et al., 2002)]. This pattern of autonomic responses is similar to that seen during panic attacks in humans (Liebowitz et al., 1986). In addition to the autonomic, respiratory and endocrine responses, activation of the DMH/PeF also elicits escape behaviors (Shekhar and DiMicco, 1987), selective enhancement of fear responses (Shekhar et al., 1987) and anxiety-like behavior as measured by the conflict (Shekhar et al., 1990), elevated plus-maze (Shekhar, 1993) and SI tests (Shekhar and Katner, 1995) in rats. The known neuroanatomical connections of the DMH/PeF are consistent with its proposed key integrative role in anxiety and panic-like responses. Thus, DMH/PeF contains a neuronal system which is essential for behavioral and cardiorespiratory responses associated with emotional arousal. A defect in such a regulatory mechanism could result in susceptibility to panic attacks. In support of these hypotheses, we previously demonstrated in experiment 1 that only panic-prone rats infused with sodium lactate had increased cellular responses in the DMH/PeF which also correlated with cardio-respiratory responses (Johnson and Shekhar, 2006). We have previously demonstrated that the increased cellular responses in the DMH/PeF are almost exclusively occurring in NMDA, but not AMPA, receptor subunit expressing cells, with a morphology (i.e. size and shape) consistent with a neuronal phenotype (Johnson and Shekhar, 2006). Since most glia express AMPA receptor subunits, this suggests that the c-Fos induction in the DMH/PeF is occurring in a unique set of neurons (Seifert et al., 1997). Furthermore, injecting specific NMDA receptor antagonists into the DMH of l-AG treated rats prior to a lactate challenge blocks all anxiety-like behavioral and panic-like cardiorespiratory responses (Johnson and Shekhar, 2006). The neurotransmitter phenotype of these DMH cells remains uncertain, but there are some enticing candidates which may be involved. Neurons that synthesize the neuropeptide orexin express the NMDA receptor [see review (Winsky-Sommerer et al., 2004)] and are only found in the DMH and adjacent hypothalamus (Peyron et al., 1998). Central orexin production is critical for maintaining wakefulness and wakefulness and vigilance [see review (Sakurai, 2007)] and orexin has been demonstrated to mobilize centrally mediated sympathetic responses (Ferguson and Samson, 2003) which is a key component of panic. Overall, this suggests that chronic infusions of l-AG into the DMH/PeF are inducing panic-prone states by making the DMH/PeF susceptible to mobilize panic-like responses to a subthreshold interoceptive cue (i.e., 0.5M sodium lactate). In the present article, this was verified in experiment 2 by injecting muscimol into the DMH/PeF of l-AG treated rats prior to lactate and blocking all aspects of l-AG+lactate-induced panic-like responses. Therefore, the present study clearly demonstrates a critical role for the DMH/PeF region in acutely eliciting panic-like responses following lactate infusions.
Pathways for lactate-induced panic-like responses
A critical component underlying sodium lactate’s ability to induce panic-like cardiorespiratory responses in this panic model involves interactions between the CVOs (OVLT/SFO), areas in the CNS that lack a blood-brain barrier and therefore serve as "sensors" of changes in osmolarity, pH and other plasma parameters (Johnson and Gross, 1993), and the DMH/PeF which is a major target of these CVOs (Thompson and Swanson, 1998). Based on this, neurons in the CVOs are likely to react to sodium lactate infusions (Johnson and Gross, 1993) and propagate this signal to the DMH/PeF in both l-AG- and d-AG-treated rats. As predicted, the lactate challenge increased c-Fos responses in the OVLT of panic-prone and non panic-prone rats equally. This suggests that the sodium lactate signal is being relayed to the DMH/PeF regardless of d-AG or l-AG treatment. This also suggests that in d-AG-treated rats with intact GABAergic tone in the DMH, the subthreshold dose of sodium lactate is not sufficient to activate the DMH. However, in l-AG-treated rats with compromised GABAergic tone in the DMH/PeF, this subthreshold signal is sufficient to activate the DMH where the signal is then further propagated to efferent targets implicated in anxiety and central autonomic control. The effect of l-AG in the hypothalamus may be specific to the DMH/PeF since we previously demonstrated that infusing l-AG into the other surrounding regions (i.e., the paraventricular nucleus of the hypothalamus) does not induce panic-like responses following a sodium lactate challenge (Shekhar et al., 1996). The importance of the OVLT in the DMH/PeF panic model was previously demonstrated in a previous article where injections of a small amount (100 nl) of lactate directly into the OVLT/AV3V region of panic-prone rats (l-AG treated), but not d-AG-treated controls, elicited panic-like responses (Shekhar and Keim, 1997). Furthermore, injecting tetrodotoxin (a voltage gated sodium channel blocker) into the OVLT region of panic-prone rats blocked i.v. lactate induced panic-like responses (Shekhar and Keim, 1997). Overall, the OVLT/AV3V region appears to be an important site for “sensing” plasma sodium lactate and relaying the signal to the DMH/PeF.
A subset of DMH/PeF efferent pathways appears to be activated during the panic-like response induced by lactate infusions. Among the forebrain limbic structures, the BNST, LSV and several subnuclei of the amygdala were selectively activated during panic-like responses. The c-Fos induction in the rostral BNST region correlated strongly with anxiety-like responses, whereas c-Fos induction in the caudal BNST correlated with panic-like cardiovascular responses. Although other studies have demonstrated that the BNST regulates anxiety-like behavior (Sajdyk et al., 2007) and autonomic activity (Crestani et al., 2007), the BNST’s role may depend on the context. In the present article, a subsequent experiment (i.e., experiment 3) demonstrated that inactivation of the rostro-caudal BNST region (see Figure 4) with acute muscimol injections selectively blocked the anxiogenic effects of the lactate+l-AG infusions without affecting the cardiorespiratory activation, providing strong support for the hypothesis that the BNST selectively regulates anxiety behavior, but not panic-like cardiorespiratory responses in the panic model presented here (see Fig. 5a for a hypothetical model).
Figure 5.
Hypothetical models of sodium lactate induced increases in panic-like behavioral and cardiorespiratory responses in panic-prone (i.e., l-AG treated) rats. a) Hypothetical model illustrating the neural pathways of lactate + l-AG-induced anxiety where plus signs indicate an excitatory pathway. b) Illustration of a hypothetical model of sodium lactate + l-AG mobilization of sympathetic nervous system activity. c) Hypothetical model illustrating sodium lactate + l-AG-induced desensitization of the vagally-mediated baroreflex, where solid lines with solid circles indicate excitatory neurons and dashed lines with open circles indicate inhibitory neurons and an “X” over a line indicates paths that are inhibited by the DMH/PeF. Abbreviations: bv, blood vessel; GABA, GABAergic path; Glu, glutamatergic path; IML, intermediolateral cell column; See methods for additional abbreviations.
Although fear conditioning has not been studied in this panic model, the strong CeA responses in rats displaying increased cardiorespiratory responses could potentially be involved in laying down conditioned fear memory [see review (Davis and Shi, 1999)] that may be analogous to secondary phobia following initial panic attacks in panic disorder patients (Starcevic et al., 1993b; Starcevic et al., 1993a). Therefore, we plan in future studies to test for fear conditioning in this model [e.g., using conditioned place-avoidance (Thielen and Shekhar, 2002)] and, in the case that conditioning is occurring, explore the role of the CeA in this conditioning. A final comment is that removing GABAergic tone in the DMH was sufficient to increase anxiety-like behavior and the threshold of anxiety remained high following the lactate challenge. Future studies may try and determine if l-AG+lactate elicit other stress-related behaviors that correlate with panic-like cardiorespiratory responses (e.g., fear associated behavior).
The tachypnea noted after lactate infusions most likely is a result of activating one of several respiratory rhythm generators in the brainstem. In the present study, the PBN was the only major respiratory center that clearly responded during panic-like responses. There is ample anatomical evidence that DMH/PeF neurons project to PBN and therefore could stimulate respiration (Chamberlin and Saper, 1994; Chen et al., 2004).
The finding that systemic injections of atenolol (a β1 adrenergic receptor antagonist) prevented l-AG+lactate-induced tachycardia supports the hypothesis that peripheral sympathetic mechanisms contribute to tachycardia and hypertensive responses during lactate-induced panic. Further analyses of c-Fos responses revealed that discrete brain regions involved in sympathetic and parasympathetic regulation were positively correlated with cardiovascular responses (see Table 1). These regions included the dorsal cap of the PVN (i.e. PaDC), SON, PBN, CeA, caudal BNST and the rostral nTS. Some of the other autonomic regulatory sites responded to lactate infusion (i.e. RPa, LC noradrenergic and C1 and C2 adrenergic neurons), while yet other brainstem regions associated with sympathetic mobilization failed to respond to lactate or l-AG (e.g., subdivisions of the PAG, etc.). In light of the data presented here, there are several ways the DMH could potentially induce cardioexcitatory responses following lactate infusions in l-AG-treated rats (see Fig. 5b–c for a hypothetical model for DMH-mediated cardioexcitatory responses). The DMH/PeF could drive hypertensive responses at efferent targets such as the PaDC, SON, RVLM or sympathetic regions of the spinal cord. The DMH/PeF directly innervates the PaDC, SON (Thompson and Swanson, 1998) and also sympathetic regions of the spinal cord (Hosoya et al., 1987). Neurons in the PaDC also directly innervate sympathetic regions of the spinal cord (Saper et al., 1976). Yet another explanation is that C1 adrenergic neurons were firing at a higher rate in lactate+l-AG-treated rats, relative to lactate+d-AG-treated rats, even though lactate induced a similar level of c-Fos expression in C1 adrenergic neurons in the RVLM similarly in both d-AG and l-AG treated rats challenged with lactate. Consistent with a role for the RVLM/C1 adrenergic cell groups in DMH/PeF-mediated cardiovascular responses, pressor responses, elicited from disinhibition of the DMH, can be severely attenuated by microinjecting muscimol into the RVLM (Fontes et al., 2001). Tachycardia responses following l-AG+lactate infusions are most likely being mobilized by a combination of direct and indirect projections from the DMH/PeF to sympathetic preganglionic neurons.
In order for the sympathetic limb of the autonomic nervous system to simultaneously induce hypertension and tachycardia, the baroreflex mechanism which stimulates vagal parasympathetic output and inhibits the sympathetic output needs to be desensitized [see review (McDowall et al., 2006)]. Within the DMM [a region that is critical for the baroreflex (Catelli and Sved, 1988) and contains parasympathetic motor neurons], infusions of lactate increased c-Fos-ir cells in the nTS but only in l-AG-treated rats. However, increased c-Fos responses in the DMM were not occurring in parasympathetic preganglionic acetylcholinergic neurons (DMV and Amb), or other regions involved in the baroreflex-mediated sympathetic inhibition [e.g., caudal ventrolateral medulla (CVLM)] (Chan and Sawchenko, 1998). The DMH directly innervates the nTS (Fontes et al., 2001), and electrical or chemical [see review (McDowall et al., 2006)] stimulation of the DMH decreases the sensitivity of the baroreflex through a GABAergic mechanism in the nTS/DMV (Jordan et al., 1988). This circuit represents an adaptive means of inhibiting the baroreflex during “fight or flight” responses and exercise (Potts et al., 2003). The nTS contains numerous GABAergic neurons (Fong et al., 2005) which could be dampening parasympathetic activity by inhibiting acetylcholinergic vagal preganglionic neurons. This notion is supported by recent evidence that stimulation of nTS neurons in vitro inhibits DMV neurons (Davis et al., 2003; Sevoz-Couche et al., 2003). Therefore the increased c-Fos-ir cells in the nTS could be occurring in local GABAergic neurons. We demonstrated this with double immunohistochemistry where c-Fos/GAD67-ir neurons were noted in l-AG+lactate-treated rats, even though intense GAD67-ir fibers in the nTS and DMV prevented quantification of double immunostaining.
The finding that panic-related conditions may be a result of parasympathetic and sympathetic imbalance, and baroreflex disruption is not surprising [see review (Friedman and Thayer, 1998)]. Panic disorder patients have higher baseline HR than controls (Gorman et al., 1983; Nesse et al., 1984) and electrocardiograms in panic disorder patients reveal sympathetic dominance and parasympathetic withdrawal (Hamada et al., 1998; McCraty et al., 2001). Panic disorder patients also show disrupted baroreceptor function that persists even when they are in remission (Shioiri et al., 2005). Also, supporting disruption of GABA function in panic disorder are data showing that genetic polymorphisms in the GAD genes are associated with vulnerability to panic disorder (Hettema et al., 2005). Moreover, GABA enhancers such as benzodiazepine agonists attenuate sodium lactate-induced panic-related responses in panic patients (Pohl et al., 1994), and panic patients demonstrate deficits in central GABA concentrations (Goddard et al., 2001).
Technical considerations
It is not certain whether the present anxiety-like responses and cardioexcitatory physiological responses of rats are equivalent to anxiety and panic attacks in humans, respectively. Furthermore, one must use caution when interpreting neural circuitry data based on immediate-early gene induction (Morgan and Curran, 1989). For example, it is possible that supraspinal efferent pathways from the DMH (e.g., PAG) were involved in sympathoexcitatory responses even though no differences in the numbers of c-Fos-ir cells were observed. The phenotype of the cells (i.e., neuronal or glial-like) that expressed c-Fos as a result of l-AG and/or lactate cannot be distinguished from nuclear immunostaining for c-Fos alone. Therefore, attempts were made to identify the extent of c-Fos induction in phenotypically characterized neurons in efferent targets of the DMH/PeF that responded to l-AG and/or lactate. This was done using counterstaining for enzymes restricted to neurons (i.e., TH, ChAT and GAD67) and proteins expressed in the vast majority of neurons of adult rodents [i.e., NeuN in the PBN (Mullen et al., 1992)]. Although identification of c-Fos induction in neurons versus glia could not be conducted in all brain areas in the same group of animals due to limitations of total tissue available, several lines of evidence above demonstrate that at every site we have studied with additional markers, the c-Fos expression has been predominantly seen in the neuronal populations.
Conclusion
The present study demonstrates that selective loss of GABAergic inhibition in critical emotion-regulating regions such as the DMH/PeF region results in lactate-induced panic responses similar to those observed in human panic disorder patients. We provide evidence that lactate infusions activate a compromised DMH/PeF region that in turn activates forebrain limbic structures, including the BNST, that elicit anxiety-related behavioral responses. In addition, data from the present study are consistent with the hypothesis that activation of the DMH/PeF region leads secondarily to activation of specific brainstem nuclei to increase anxiety-related behavior and respiration and to densensitize the baroreflex, thus allowing sympathetically mediated tachycardia responses.
Supplementary Material
Acknowledgements
We gratefully acknowledge Stanley Keim for technical assistance, Pamela E. Kelley for inter-rater analyses and Ryan Wiegand for advice on statistical analyses.
The present work was supported by the National Institute of Mental Health (RO1 MH52619 and RO1 MH065702 to AS); the Wellcome Trust (Research Career Development Fellowship 068558/Z/02/Z to CAL) and the Anxiety Disorders Association of America (Junior Faculty Research Award to PLJ).
Footnotes
Disclosure statement:
The authors’ have no other disclosures to report.
References
- Abshire VM, Hankins KD, Roehr KE, DiMicco JA. Injection of L-allylglycine into the posterior hypothalamus in rats causes decreases in local GABA which correlate with increases in heart rate. Neuropharmacology. 1988;27:1171–1177. doi: 10.1016/0028-3908(88)90013-5. [DOI] [PubMed] [Google Scholar]
- Catelli JM, Sved AF. Enhanced pressor response to GABA in the nucleus tractus solitarii of the spontaneously hypertensive rat. Eur J Pharmacol. 1988;151:243–248. doi: 10.1016/0014-2999(88)90804-7. [DOI] [PubMed] [Google Scholar]
- Chamberlin NL, Saper CB. Topographic organization of respiratory responses to glutamate microstimulation of the parabrachial nucleus in the rat. J Neurosci. 1994;14:6500–6510. doi: 10.1523/JNEUROSCI.14-11-06500.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan RK, Sawchenko PE. Organization and transmitter specificity of medullary neurons activated by sustained hypertension: implications for understanding baroreceptor reflex circuitry. J Neurosci. 1998;18:371–387. doi: 10.1523/JNEUROSCI.18-01-00371.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen T, Hui R, Dong YX, Li YQ, Mizuno N. Endomorphin 1- and endomorphin 2-like immunoreactive neurons in the hypothalamus send axons to the parabrachial nucleus in the rat. Neurosci Lett. 2004;357:139–142. doi: 10.1016/j.neulet.2003.12.079. [DOI] [PubMed] [Google Scholar]
- Crestani CC, Alves FH, Resstel LB, Correa FM. Cardiovascular effects of noradrenaline microinjection in the bed nucleus of the stria terminalis of the rat brain. J Neurosci Res. 2007;85:1592–1599. doi: 10.1002/jnr.21250. [DOI] [PubMed] [Google Scholar]
- Dampney RA. Functional organization of central pathways regulating the cardiovascular system. Physiol Rev. 1994;74:323–364. doi: 10.1152/physrev.1994.74.2.323. [DOI] [PubMed] [Google Scholar]
- Davis M, Shi C. The extended amygdala: are the central nucleus of the amygdala and the bed nucleus of the stria terminalis differentially involved in fear versus anxiety? Ann NY Acad Sci. 1999;877:281–291. doi: 10.1111/j.1749-6632.1999.tb09273.x. [DOI] [PubMed] [Google Scholar]
- Davis SF, Williams KW, Xu W, Glatzer NR, Smith BN. Selective enhancement of synaptic inhibition by hypocretin (orexin) in rat vagal motor neurons: implications for autonomic regulation. J Neurosci. 2003;23:3844–3854. doi: 10.1523/JNEUROSCI.23-09-03844.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiMicco JA, Samuels BC, Zaretskaia MV, Zaretsky DV. The dorsomedial hypothalamus and the response to stress: part renaissance, part revolution. Pharmacol Biochem Behav. 2002;71:469–480. doi: 10.1016/s0091-3057(01)00689-x. [DOI] [PubMed] [Google Scholar]
- DSM-IV. Diagnostic and Statistical Manual - Fourth Edn. (DSM - IV) Washington, DC: American Psychiatric Association; 1994. [Google Scholar]
- Ehlers A, Breuer P. How good are patients with panic disorder at perceiving their heartbeats? Biol Psychol. 1996;42:165–182. doi: 10.1016/0301-0511(95)05153-8. [DOI] [PubMed] [Google Scholar]
- Ferguson AV, Samson WK. The orexin/hypocretin system: a critical regulator of neuroendocrine and autonomic function. Front Neuroendocrinol. 2003;24:141–150. doi: 10.1016/s0091-3022(03)00028-1. [DOI] [PubMed] [Google Scholar]
- Fitz SD, Keim SR, Sajdyk TJ, Shekhar A. Reboxetine, a potent norepinephrine reuptake inhibitor blocks lactate- and CO2-induced panic-like responses in panic-prone rats. Society for Neuroscience. 2003;959:13. [Google Scholar]
- Fong AY, Stornetta RL, Foley CM, Potts JT. Immunohistochemical localization of GAD67-expressing neurons and processes in the rat brainstem: Subregional distribution in the nucleus tractus solitarius. J Comp Neurol. 2005;493:274–290. doi: 10.1002/cne.20758. [DOI] [PubMed] [Google Scholar]
- Fontes MA, Tagawa T, Polson JW, Cavanagh SJ, Dampney RA. Descending pathways mediating cardiovascular response from dorsomedial hypothalamic nucleus. Am J Physiol Heart Circ Physiol. 2001;280:H2891–H2901. doi: 10.1152/ajpheart.2001.280.6.H2891. [DOI] [PubMed] [Google Scholar]
- Friedman BH, Thayer JF. Autonomic balance revisited: panic anxiety and heart rate variability. J Psychosom Res. 1998;44:133–151. doi: 10.1016/s0022-3999(97)00202-x. [DOI] [PubMed] [Google Scholar]
- Goddard AW, Mason GF, Almai A, Rothman DL, Behar KL, Petroff OA, Charney DS, Krystal JH. Reductions in occipital cortex GABA levels in panic disorder detected with 1h-magnetic resonance spectroscopy. Archives of General Psychiatry. 2001;58:556–561. doi: 10.1001/archpsyc.58.6.556. [DOI] [PubMed] [Google Scholar]
- Gorman JM, Levy GF, Liebowitz MR, McGrath P, Appleby IL, Dillon DJ, Davies SO, Klein DF. Effect of acute beta-adrenergic blockade on lactate-induced panic. Arch Gen Psychiatry. 1983;40:1079–1082. doi: 10.1001/archpsyc.1983.01790090041006. [DOI] [PubMed] [Google Scholar]
- Gorman JM, Papp LA, Coplan JD, Martinez JM, Lennon S, Goetz RR, Ross D, Klein DF. Anxiogenic effects of CO2 and hyperventilation in patients with panic disorder. Am J Psychiatry. 1994;151:547–553. doi: 10.1176/ajp.151.4.547. [DOI] [PubMed] [Google Scholar]
- Hamada T, Koshino Y, Misawa T, Isaki K, Gejyo F. Mitral valve prolapse and autonomic function in panic disorder. Acta Psychiatr Scand. 1998;97:139–143. doi: 10.1111/j.1600-0447.1998.tb09976.x. [DOI] [PubMed] [Google Scholar]
- Herman JP, Cullinan WE. Neurocircuitry of stress: central control of the hypothalamo-pituitary-adrenocortical axis. Trends Neurosci. 1997;20:78–84. doi: 10.1016/s0166-2236(96)10069-2. [DOI] [PubMed] [Google Scholar]
- Herman JP, Prewitt CM, Cullinan WE. Neuronal circuit regulation of the hypothalamo-pituitary-adrenocortical stress axis. Crit Rev Neurobiol. 1996;10:371–394. doi: 10.1615/critrevneurobiol.v10.i3-4.50. [DOI] [PubMed] [Google Scholar]
- Hettema JM, Prescott CA, Myers JM, Neale MC, Kendler KS. The structure of genetic and environmental risk factors for anxiety disorders in men and women. Arch Gen Psychiatry. 2005;62:182–189. doi: 10.1001/archpsyc.62.2.182. [DOI] [PubMed] [Google Scholar]
- Hoehn-Saric R, McLeod DR, Funderburk F, Kowalski P. Somatic symptoms and physiologic responses in generalized anxiety disorder and panic disorder: an ambulatory monitor study. Arch Gen Psychiatry. 2004;61:913–921. doi: 10.1001/archpsyc.61.9.913. [DOI] [PubMed] [Google Scholar]
- Hosoya Y, Ito R, Kohno K. The topographical organization of neurons in the dorsal hypothalamic area that project to the spinal cord or to the nucleus raphe pallidus in the rat. Exp Brain Res. 1987;66:500–506. doi: 10.1007/BF00270682. [DOI] [PubMed] [Google Scholar]
- Johnson AK, Gross PM. Sensory circumventricular organs and brain homeostatic pathways. FASEB J. 1993;7:678–686. doi: 10.1096/fasebj.7.8.8500693. [DOI] [PubMed] [Google Scholar]
- Johnson PL, Shekhar A. Panic-prone state induced in rats with GABA dysfunction in the dorsomedial hypothalamus is mediated by NMDA receptors. J Neurosci. 2006;26:7093–7104. doi: 10.1523/JNEUROSCI.0408-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson PL, Hollis JH, Moratalla R, Lightman SL, Lowry CA. Acute hypercarbic gas exposure reveals functionally distinct subpopulations of serotonergic neurons in rats. J Psychopharmacol. 2005;19:327–341. doi: 10.1177/0269881105053281. [DOI] [PubMed] [Google Scholar]
- Jordan D, Mifflin SW, Spyer KM. Hypothalamic inhibition of neurones in the nucleus tractus solitarius of the cat is GABA mediated. J Physiol. 1988;399:389–404. doi: 10.1113/jphysiol.1988.sp017087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovacs KJ. c-Fos as a transcription factor: a stressful (re)view from a functional map. Neurochem Int. 1998;33:287–297. doi: 10.1016/s0197-0186(98)00023-0. [DOI] [PubMed] [Google Scholar]
- Liebowitz MR, Gorman JM, Fyer A, Dillon D, Levitt M, Klein DF. Possible mechanisms for lactate's induction of panic. Am J Psychiatry. 1986;143:495–502. doi: 10.1176/ajp.143.4.495. [DOI] [PubMed] [Google Scholar]
- McCraty R, Atkinson M, Tomasino D, Stuppy WP. Analysis of twenty-four hour heart rate variability in patients with panic disorder. Biol Psychol. 2001;56:131–150. doi: 10.1016/s0301-0511(01)00074-6. [DOI] [PubMed] [Google Scholar]
- McDowall LM, Horiuchi J, Killinger S, Dampney RA. Modulation of the baroreceptor reflex by the dorsomedial hypothalamic nucleus and perifornical area. Am J Physiol Regul Integr Comp Physiol. 2006;290:R1020–R1026. doi: 10.1152/ajpregu.00541.2005. [DOI] [PubMed] [Google Scholar]
- Morgan JI, Curran T. Stimulus-transcription coupling in neurons: role of cellular immediate-early genes. Trends Neurosci. 1989;12:459–462. doi: 10.1016/0166-2236(89)90096-9. [DOI] [PubMed] [Google Scholar]
- Mullen RJ, Buck CR, Smith AM. NeuN, a neuronal specific nuclear protein in vertebrates. Development. 1992;116:201–211. doi: 10.1242/dev.116.1.201. [DOI] [PubMed] [Google Scholar]
- Nesse RM, Cameron OG, Curtis GC, McCann DS, Huber-Smith MJ. Adrenergic function in patients with panic anxiety. Arch Gen Psychiatry. 1984;41:771–776. doi: 10.1001/archpsyc.1984.01790190045005. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain Stereotaxic Coordinates. San Diego: Academic Press; 1997. [Google Scholar]
- Peterson RG. Design and analysis of experiments. New York: Marcel Dekker, Inc.; 1985. [Google Scholar]
- Peyron C, Tighe DK, van den Pol AN, de Lecea L, Heller HC, Sutcliffe JG, Kilduff TS. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J Neurosci. 1998;18:9996–10015. doi: 10.1523/JNEUROSCI.18-23-09996.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pohl R, Balon R, Bechou R, Lycaki H. Lactate-induced anxiety after imapramine and diazepam treatment. Anxiety. 1994;1:54–63. doi: 10.1002/anxi.3070010204. [DOI] [PubMed] [Google Scholar]
- Pollock RA, Carter AS, Amir N, Marks LE. Anxiety sensitivity and auditory perception of heartbeat. Behav Res Ther. 2006;44:1739–1756. doi: 10.1016/j.brat.2005.12.013. [DOI] [PubMed] [Google Scholar]
- Potts JT, Paton JF, Mitchell JH, Garry MG, Kline G, Anguelov PT, Lee SM. Contraction-sensitive skeletal muscle afferents inhibit arterial baroreceptor signalling in the nucleus of the solitary tract: role of intrinsic GABA interneurons. Neuroscience. 2003;119:201–214. doi: 10.1016/s0306-4522(02)00953-3. [DOI] [PubMed] [Google Scholar]
- Sajdyk TJ, Johnson PL, Fitz SD, Shekhar A. Chronic inhibition of GABA synthesis in the Bed Nucleus of the Stria Terminalis elicits anxiety-like behavior. J Psychopharmacol. 2007 doi: 10.1177/0269881107082902. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakurai T. The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nat Rev Neurosci. 2007;8:171–181. doi: 10.1038/nrn2092. [DOI] [PubMed] [Google Scholar]
- Samuels BC, Zaretsky DV, DiMicco JA. Dorsomedial hypothalamic sites where disinhibition evokes tachycardia correlate with location of raphe-projecting neurons. Am J Physiol Regul Integr Comp Physiol. 2004;287:R472–R478. doi: 10.1152/ajpregu.00667.2003. [DOI] [PubMed] [Google Scholar]
- Saper CB, Loewy AD, Swanson LW, Cowan WM. Direct hypothalamo-autonomic connections. Brain Res. 1976;117:305–312. doi: 10.1016/0006-8993(76)90738-1. [DOI] [PubMed] [Google Scholar]
- Seifert G, Rehn L, Weber M, Steinhauser C. AMPA receptor subunits expressed by single astrocytes in the juvenile mouse hippocampus. Brain Res Mol Brain Res. 1997;47:286–294. doi: 10.1016/s0169-328x(97)00059-4. [DOI] [PubMed] [Google Scholar]
- Senba E, Ueyama T. Stress-induced expression of immediate early genes in the brain and peripheral organs of the rat. Neurosci Res. 1997;29:183–207. doi: 10.1016/s0168-0102(97)00095-3. [DOI] [PubMed] [Google Scholar]
- Sevoz-Couche C, Comet MA, Hamon M, Laguzzi R. Role of nucleus tractus solitarius 5-HT3 receptors in the defense reaction-induced inhibition of the aortic baroreflex in rats. J Neurophysiol. 2003;90:2521–2530. doi: 10.1152/jn.00275.2003. [DOI] [PubMed] [Google Scholar]
- Shekhar A. GABA receptors in the region of the dorsomedial hypothalamus of rats regulate anxiety in the elevated plus-maze test. I. Behavioral measures. Brain Res. 1993;627:9–16. doi: 10.1016/0006-8993(93)90742-6. [DOI] [PubMed] [Google Scholar]
- Shekhar A, DiMicco JA. Defense reaction elicited by injection of GABA antagonists and synthesis inhibitors into the posterior hypothalamus in rats. Neuropharmacology. 1987;26:407–417. doi: 10.1016/0028-3908(87)90020-7. [DOI] [PubMed] [Google Scholar]
- Shekhar A, Katner JS. Dorsomedial hypothalamic GABA regulates anxiety in the social interaction test. Pharmacol Biochem Behav. 1995;50:253–258. doi: 10.1016/0091-3057(94)00307-5. [DOI] [PubMed] [Google Scholar]
- Shekhar A, Keim SR. The circumventricular organs form a potential neural pathway for lactate sensitivity: implications for panic disorder. J Neurosci. 1997;17:9726–9735. doi: 10.1523/JNEUROSCI.17-24-09726.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shekhar A, Hingtgen JN, DiMicco JA. Selective enhancement of shock avoidance responding elicited by GABA blockade in the posterior hypothalamus of rats. Brain Res. 1987;420:118–128. doi: 10.1016/0006-8993(87)90246-0. [DOI] [PubMed] [Google Scholar]
- Shekhar A, Hingtgen JN, DiMicco JA. GABA receptors in the posterior hypothalamus regulate experimental anxiety in rats. Brain Res. 1990;512:81–88. doi: 10.1016/0006-8993(90)91173-e. [DOI] [PubMed] [Google Scholar]
- Shekhar A, Keim SR, Simon JR, McBride WJ. Dorsomedial hypothalamic GABA dysfunction produces physiological arousal following sodium lactate infusions. Pharmacol Biochem Behav. 1996;55:249–256. doi: 10.1016/s0091-3057(96)00077-9. [DOI] [PubMed] [Google Scholar]
- Shekhar A, Johnson PL, Sajdyk TJ, Fitz SD, Keim SR, Kelley PE, Gehlert DR, DiMicco JA. Angiotensin-II is a putative neurotransmitter in lactate-induced panic-like responses in rats with disruption of GABAergic inhibition in the dorsomedial hypothalamus. J Neurosci. 2006;26:9205–9215. doi: 10.1523/JNEUROSCI.2491-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shioiri T, Kojima-Maruyama M, Hosoki T, Kitamura H, Tanaka A, Yoshizawa M, Bando T, Someya T. Dysfunctional baroreflex regulation of sympathetic nerve activity in remitted patients with panic disorder. A new methodological approach. Eur Arch Psychiatry Clin Neurosci. 2005;255:293–298. doi: 10.1007/s00406-005-0561-2. [DOI] [PubMed] [Google Scholar]
- Singewald N, Sharp T. Neuroanatomical targets of anxiogenic drugs in the hindbrain as revealed by Fos immunocytochemistry. Neuroscience. 2000;98:759–770. doi: 10.1016/s0306-4522(00)00177-9. [DOI] [PubMed] [Google Scholar]
- Singewald N, Salchner P, Sharp T. Induction of c-Fos expression in specific areas of the fear circuitry in rat forebrain by anxiogenic drugs. Biol Psychiatry. 2003;53:275–283. doi: 10.1016/s0006-3223(02)01574-3. [DOI] [PubMed] [Google Scholar]
- Starcevic V, Uhlenhuth EH, Kellner R, Pathak D. Comparison of primary and secondary panic disorder: a preliminary report. J Affect Disord. 1993a;27:81–86. doi: 10.1016/0165-0327(93)90080-4. [DOI] [PubMed] [Google Scholar]
- Starcevic V, Kellner R, Uhlenhuth EH, Pathak D. The phenomenology of panic attacks in panic disorder with and without agoraphobia. Compr Psychiatry. 1993b;34:36–41. doi: 10.1016/0010-440x(93)90033-z. [DOI] [PubMed] [Google Scholar]
- Stein DJ, Bouwer C. A neuro-evolutionary approach to the anxiety disorders. J Anxiety Disord. 1997;11:409–429. doi: 10.1016/s0887-6185(97)00019-4. [DOI] [PubMed] [Google Scholar]
- Street LL, Craske MG, Barlow DH. Sensations, cognitions and the perception of cues associated with expected and unexpected panic attacks. Behav Res Ther. 1989;27:189–198. doi: 10.1016/0005-7967(89)90078-8. [DOI] [PubMed] [Google Scholar]
- Thielen SK, Shekhar A. Amygdala priming results in conditioned place avoidance. Pharmacol Biochem Behav. 2002;71:401–406. doi: 10.1016/s0091-3057(01)00696-7. [DOI] [PubMed] [Google Scholar]
- Thompson RH, Swanson LW. Organization of inputs to the dorsomedial nucleus of the hypothalamus: a reexamination with Fluorogold and PHAL in the rat. Brain Res Brain Res Rev. 1998;27:89–118. doi: 10.1016/s0165-0173(98)00010-1. [DOI] [PubMed] [Google Scholar]
- Vickers K, McNally RJ. Respiratory symptoms and panic in the National Comorbidity Survey: a test of Klein's suffocation false alarm theory. Behav Res Ther. 2005;43:1011–1018. doi: 10.1016/j.brat.2004.06.019. [DOI] [PubMed] [Google Scholar]
- Winsky-Sommerer R, Yamanaka A, Diano S, Borok E, Roberts AJ, Sakurai T, Kilduff TS, Horvath TL, de Lecea L. Interaction between the corticotropin-releasing factor system and hypocretins (orexins): a novel circuit mediating stress response. J Neurosci. 2004;24:11439–11448. doi: 10.1523/JNEUROSCI.3459-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.