Abstract
Herein we report the clinical, histopathological, and molecular features of a cancer syndrome with predisposition to uterine leiomyomas and papillary renal cell carcinoma. The studied kindred included 11 family members with uterine leiomyomas and two with uterine leiomyosarcoma. Seven individuals had a history of cutaneous nodules, two of which were confirmed to be cutaneous leiomyomatosis. The four kidney cancer cases occurred in young (33- to 48-year-old) females and displayed a unique natural history. All these kidney cancers displayed a distinct papillary histology and presented as unilateral solitary lesions that had metastasized at the time of diagnosis. Genetic-marker analysis mapped the predisposition gene to chromosome 1q. Losses of the normal chromosome 1q were observed in tumors that had occurred in the kindred, including a uterine leiomyoma. Moreover, the observed histological features were used as a tool to diagnose a second kindred displaying the phenotype. We have shown that predisposition to uterine leiomyomas and papillary renal cell cancer can be inherited dominantly through the hereditary leiomyomatosis and renal cell cancer (HLRCC) gene. The HLRCC gene maps to chromosome 1q and is likely to be a tumor suppressor. Clinical, histopathological, and molecular tools are now available for accurate detection and diagnosis of this cancer syndrome.
Uterine leiomyomas have a major impact on women's health. Unfortunately little is known about the molecular background of these lesions and inherited leiomyoma susceptibility. Hereditary renal cell cancer can be divided into two syndromes with well defined clinical, histopathological, and molecular features. von Hippel-Lindau (VHL) disease is associated with clear cell cancer as well as extrarenal tumors such as central nervous system hemangioblastomas, retinal angiomas, and pheochromocytomas. In hereditary papillary renal carcinoma (HPRC), multiple bilateral papillary cancers typically occur. VHL is caused by a germline mutation in the VHL tumor-suppressor gene located in chromosome 3p25 (1), whereas HPRC is associated with activating germline changes in MET oncogene in chromosome 7q31 (2). One family segregating a translocation (3;8)(p14;q24) and multifocal clear cell renal cancer has been reported (3). In addition to these syndromes, kidney cancer can occur in context of other tumor-predisposition conditions such as hereditary nonpolyposis colorectal cancer, tuberous sclerosis (4–6), and hereditary thyroid cancer (7). Solitary families segregating clear cell renal cancer with no apparent association to previously described kidney cancer syndromes have been reported also (8).
Although most hereditary cancer syndromes with clinically distinct phenotypes perhaps have been characterized, some disease forms still may be unknown to medical science. Herein we report a clinical, histopathological, and molecular study of a previously unknown syndrome predisposing to uterine leiomyomas as well as renal cell cancer.
Materials and Methods
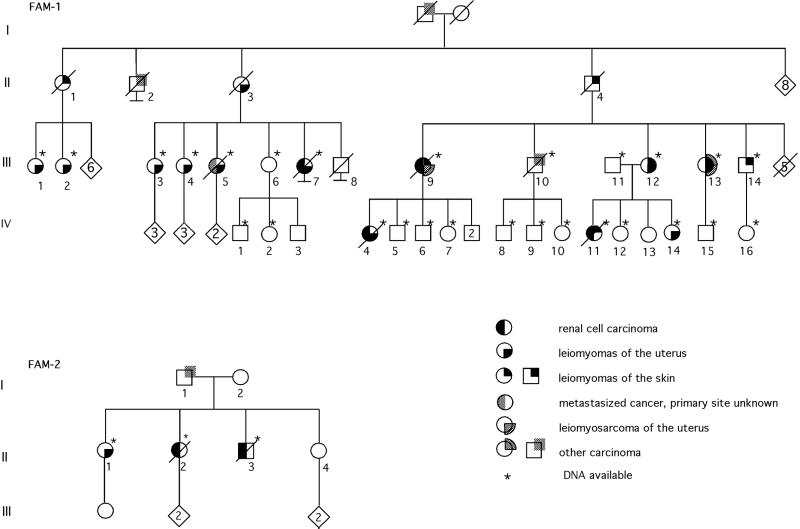
The study focused on two tumor-predisposed families. First, a large kindred (FAM-1; Fig. 1) was scrutinized for clinical, histopathological, and molecular features. In the course of the study, a smaller kindred was identified (Fig. 1; FAM-2).
Figure 1.
Pedigrees of FAM-1 and FAM-2. During the study, members FAM-1/III-1, FAM-1/III-2, and FAM-1/III-14 were ascertained and included in the linkage analyses.
Clinical Documentation.
The proband of FAM-1 (FAM-1/IV-7) was admitted to the Department of Clinical Genetics, Oulu University Hospital for genetic counseling because of renal cell cancer at relatively young age in her mother (FAM-1/III-9) and one of her sisters (FAM-1/IV-4). In addition to data derived from patient records and during patient interviews, a documented family history was obtained by identifying relatives from the official population registries, followed by verifying tumor status in the relatives through patient records and at the Finnish Cancer Registry. Both the population registries and the Cancer Registry have almost complete coverage (9). The family included 11 individuals with uterine leiomyomas and seven with cutaneous nodules. Four individuals had early onset renal cell cancer. Paraffin-embedded tissue blocks were derived from as many cases as possible for histopathological evaluation, including the four renal cell cancers.
Histopathological Evaluation.
Uterine leiomyomas.
Hematoxylin/eosin (H&E) -stained microscope slides were reviewed from two patients, FAM-1/III-12 and FAM-1/III-13, ages 34 and 37 at the time of diagnosis, respectively.
Renal cell cancers.
Four female patients, FAM-1/III-7, FAM-1/III-9, FAM-1/IV-4, and FAM-1/IV-11 (ranging in age from 33 to 48 at the time of diagnosis), had undergone nephrectomy between 1980 and 1998. Pathologists' gross reports, histologic slides, and paraffin-embedded tumors were obtained from the archives of the Department of Pathology at the Oulu University Hospital and other institutes involved. The tumors were classified in terms of the Armed Forces Institute of Pathology classification (10). The histopathology of the tumors was compared with the Delahunt and Eble criteria for papillary renal carcinoma (11). H&E staining, immunohistochemistry for epithelial membrane antigen and vimentin (Dako), cytokeratin 7 (Roche Molecular Biochemicals), and human milk fat globule (HMFG) (NCL-HMFG1 and -2, NovoCastra, Newcastle, UK) were performed on all tumors.
Cutaneous tumors.
H&E slides and paraffin blocks of skin tumors were available from two patients, FAM-1/III-12 and FAM-1/III-13 (ages 40 and 45 at the time of diagnosis, respectively). Immunohistochemistry for desmin, muscle specification activity, and α-SMA (Dako) was performed.
Case with metastases of unknown origin.
From one case (FAM-1/III-5, age 42 at the time of diagnosis) there were slides and paraffin blocks of metastases in the ovaries, omentum, and para-aortic and clavicular lymph nodes. Representative slides were stained with H&E, periodic acid/Schiff reagent, and Giemsa, Grimelius, and Gomori stainings. Immunohistochemistry was used also, as with the renal cell cancers.
In addition, the slides representing breast and bladder carcinomas derived from FAM-1/III-13 and FAM-1/III-10 individuals, respectively, were reevaluated.
Genetic-Marker Analysis.
Before genome-wide linkage analysis in FAM-1, the MET gene region in chromosome 7q was excluded as the predisposing locus [logarithm of odds (lod) score <−2]. Only individuals affected with kidney cancer were included in this analysis. Perkin–Elmer Applied Biosystems (Foster City, CA) PRISM Linkage Mapping Set MD-10 (P/N 450067, 10-cM density) was used in the genome-wide search. PCR products were run on an Applied Biosystems PRISM 377 DNA sequencer (Perkin–Elmer Applied Biosystems). The data were analyzed by the GENESCAN 3.1 software (Perkin–Elmer).
Linkage analyses were carried out by using the GENEHUNTER (genome-wide screening; ref. 12), and FASTLINK (13) programs and a modified MLINK program of the Fastlinkage package LMLINK (14) for inclusion of loss of heterozygosity (LOH) data in linkage analysis. Because the complete pedigree was too large to be analyzed by using GENEHUNTER, the pedigree was split in two for genome-wide screening. Chromosomal regions showing a lod score of 1 or higher were reanalyzed by using the combined pedigree structure. Dominant inheritance for the tumor-predisposition phenotype was assumed, and individuals with cutaneous nodules/leiomyomas, uterine leiomyomas, and renal cell cancer were considered affected. The affection status of individual FAM-1/III-10 with bladder cancer was scored as unknown. Probabilities for phenocopies were set to 0.20 for uterine leiomyomas, 0.00001 for the renal cell cancer phenotype, and 0.002 for cutaneous leiomyomatosis. Penetrance was estimated conservatively at 0.4.
LOH at the susceptibility locus was examined from the available tumor material from both families.
Results
Clinical and Histopathological Features.
The tumor predisposition in FAM-1 seemed dominantly inherited and included susceptibility to uterine leiomyomas, uterine leiomyosarcoma, renal cell cancer, and multiple benign cutaneous tumors (Fig. 1). Table 1 summarizes the features of the affected individuals.
Table 1.
The phenotypes of all affected individuals (individuals with cutaneous nodules/leiomyomas, uterine leiomyomas or leiomyosarcoma, or renal cell cancer)
| Individual | Sex | Skin lesions | Uterine tumors | Renal cell cancer | Other tumors |
|---|---|---|---|---|---|
| FAM-1/II-1 | F | Cutaneous nodules | No | No | No |
| FAM-1/II-3 | F | No | Leiomyomas (40) | No | No |
| FAM-1/II-4 | M | Cutaneous nodules | — | No | No |
| FAM-1/III-1 | F | No | Leiomyomas (44) | No | No |
| FAM-1/III-2 | F | No | Leiomyomas (48) | No | No |
| FAM-1/III-3 | F | No | Leiomyomas (40) | No | No |
| FAM-1/III-4 | F | No | Leiomyomas (34) | No | No |
| FAM-1/III-5 | F | No | Leiomyomas (37) | No | Primary site unknown (42) |
| FAM-1/III-7 | F | No | Leiomyomas (39) | Yes (39) | No |
| FAM-1/III-9 | F | Cutaneous nodules | Leiomyosarcoma (35) | Yes (48) | No |
| FAM-1/III-12 | F | Leiomyomas | Leiomyomas (39) | No | No |
| FAM-1/III-13 | F | Leiomyomas | Leiomyomas, leiomyosarcoma (40) | No | Breast cancer |
| FAM-1/III-14 | M | Cutaneous nodules | — | No | No |
| FAM-1/IV-4 | F | No | Leiomyomas (29) | Yes (42) | No |
| FAM-1/IV-11 | F | Cutaneous nodules | No | Yes (33) | No |
| FAM-1/IV-14 | F | No | Leiomyomas (25) | No | No |
| FAM-2/II-1 | F | No | Leiomyomas (41) | No | No |
| FAM-2/II-2 | F | No | No | Yes (33) | No |
| FAM-2/II-3 | M | No | — | Yes (26) | No |
All listed individuals segregated a disease-associated haplotype. The three individuals representing generation II of FAM-1 are obligate carriers. In addition, one unaffected male displayed a linked haplotype (for reasons of confidentiality, data is not shown). Age at diagnosis, when known, is in parentheses. The cutaneous lesions were, in some cases, present from childhood.
Myomatous uterus was the most common finding; 11 cases were observed in FAM-1. The age at diagnosis ranged from 26 to 48 years. Two patients had been operated for leiomyosarcoma. Leiomyosarcoma was suspected in patient FAM-1/III-7 at the age of 39 on the basis of ultrasound examination, but surgery was not performed because of the general condition of the patient. Histological slides from two patients were reevaluated. Leiomyomas from patient FAM-1/III-12 were benign and composed of groups and bundles of smooth-muscle cells. Patient FAM-1/III-13 had one leiomyoma and one low-grade leiomyosarcoma (1.5-cm diameter).
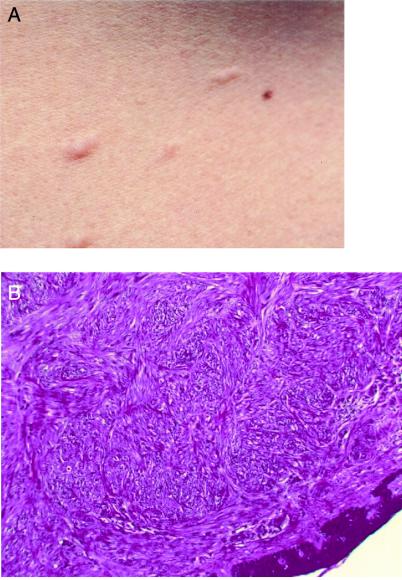
Seven members of FAM-1 had skin lesions. Histological slides from two patients were evaluated. Typical lesions from individuals FAM-1/III-12 and FAM-1/III-13 (Fig. 2) were piloleiomyomas composed of interlacing bundles of smooth-muscle cells. The smooth-muscle origin was confirmed by positive immunohistochemistry for desmin, multiplication-stimulatory activity, and α-SMA.
Figure 2.
(A) Cutaneous leiomyomas from individual FAM-1/III-13. (B) A histological view (FAM-1/III-13, H&E, magnification ×40).
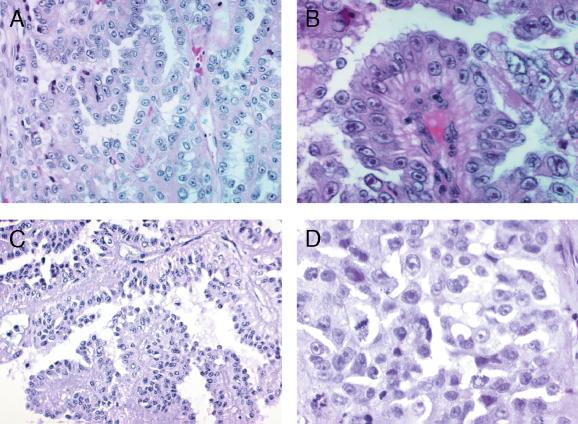
All four renal cell cancers in FAM-1 were solitary. The maximum diameter varied from 5 to 22 cm. The smallest tumor appeared well defined; in three others, the border between the tumor and the kidney parenchyme was irregular. All tumors had metastasized at the time of diagnosis, and none of the patients survived. For histopathological features, see Fig. 3. Two of the four tumors were mainly papillary, whereas one was tubulopapillary and papillary. One showed papillary, solid and cystic areas, and a small sarcomatoid region. Most of the cells had large amphophilic cytoplasm. All tumors had well preserved areas with large nuclei and inclusion-like large eosinophilic nucleoli. The Fuhrman nuclear grade was 3 to 4. Mitoses were moderate, usually less than one in 10 high-power fields. Neither psammoma bodies nor stromal macrophages were seen. All tumors displayed scanty to moderate amounts of round cell infiltrates. Apoptotic cell groups were seen in all lesions. Necroses and hemorrhages occurred in three tumors. None of the tumors displayed rhabdoid features. In immunohistochemical stainings, all tumors were vimentin-positive and cytokeratin 7-negative; two were positive for epithelial membrane antigen (EMA) and HMFG2, and one of these was positive also for HMFG1. Two were negative for EMA and HMFG1 and 2.
Figure 3.
Histopathology of the renal cancers. (A) The papillary structure of the renal cancer of patient FAM-1/IV-11 (H&E, magnification ×200). (B) Cells with inclusion-like nucleoli (patient FAM-1/III-9, H&E, magnification ×400). (C and D) The papillary structure and nuclear features of the lesion from patient FAM-2/II-2 [(C) H&E, magnification ×200; (D) H&E, magnification ×400].
Three other malignancies had occurred in FAM-1. Individual FAM-1/III-5 had been diagnosed with a carcinoma of unknown origin. Histological slides were reevaluated. Small epithelial cell groups without clear organization, but suggestive of a papillary architecture, were seen in lymph nodes, in both ovaries, and omentum. The cells were well defined, and the cytoplasm was large. The nuclei were round and sometimes multiple. Some nuclei with eosinophilic enhanced nucleoli could be seen. Mitoses were scanty. The cells were positive for vimentin, epithelial membrane antigen, and HMFG1 and -2 and negative for cytokeratin 7. The breast cancer from patient FAM-1/III-13 was a lobular infiltrating carcinoma. The urinary bladder tumor from patient FAM-1/III-10 was a grade III transitional cell carcinoma.
The histological picture of the renal cell cancers in FAM-1 was exceptional, and an effort was made to use this feature combined with young age at onset as a tool in diagnosing additional families. Histologic slides of 27 renal cell cancers diagnosed between 15 and 34 years of age (otherwise unselected) were evaluated, and one tumor displayed papillary features similar to tumors in FAM-1 (Fig. 3). The family history of the 33-year-old patient (FAM-2/II-2; Fig. 1) was clarified, and she had had one brother and two sisters. The brother (FAM-2/II-3) had been affected with renal cell cancer at the age of 26 years. Evaluation of this tumor revealed a striking similarity to the lesion of the sister, as well as lesions observed in FAM-1, including metastatic spread at diagnosis. The patient records of the two female siblings were derived and revealed that the other sibling (FAM-2/II-1) had suffered from extensive uterine leiomyomatosis; the weight of the uterus at hysterectomy was 605 g. No evidence of cutaneous lesions was obtained in this family (Table 1). One other malignancy had occurred in the family; the father had been diagnosed with prostate cancer at the age of 65.
Genetic-Marker Analysis.
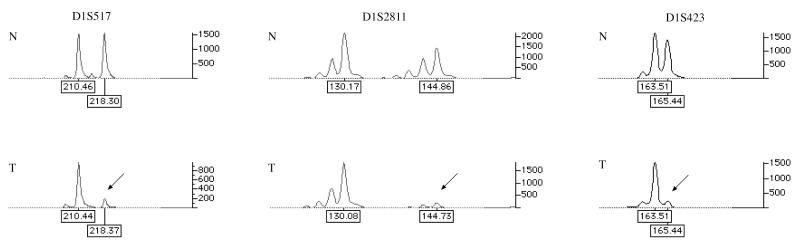
In the genome-wide linkage analysis using 370 microsatellite markers, only one locus, telomeric region of the long arm of chromosome 1, displayed substantial positivity. Altogether, 12 markers located in 1q42-q44 were genotyped to confirm the finding. The highest lod score was at marker D1S2811 (4.11), which was more than 10 times higher than the level of significance (lod score 3) customarily accepted for simple inherited conditions. The four renal cell cancers and one uterine leiomyoma were studied for LOH at this region (Fig. 4). Four of the five lesions, including the leiomyoma, displayed loss of the wild-type chromosome. When these data are incorporated into the lod scores, the score at D1S2811 is 5.31 (Table 2).
Figure 4.
LOH analysis of uterine leiomyoma from the FAM-1/III-1 individual revealing loss of the normal allele. For each marker: (Upper) normal DNA (N); (Lower) tumor DNA (T). Arrows depict LOH.
Table 2.
Two-point lod scores for microsatellite markers flanking the tumor predisposition locus
| θ
|
||||||||
|---|---|---|---|---|---|---|---|---|
| 0.0 | 0.001 | 0.01 | 0.05 | 0.1 | 0.2 | 0.3 | 0.4 | |
| Marker | ||||||||
| Lod scores with LOH information | ||||||||
| 1 D1S2800 | −4.69 | −3.23 | −1.42 | 0.10 | 0.66 | 0.95 | 0.85 | 0.59 |
| 2 D1S517 | −4.44 | −2.55 | −0.70 | 0.49 | 0.83 | 0.90 | 0.71 | 0.44 |
| 3 D1S2785 | 4.07 | 4.07 | 4.01 | 3.74 | 3.40 | 2.68 | 1.91 | 1.12 |
| 4 D1S547 | 2.49 | 2.49 | 2.45 | 2.27 | 2.05 | 1.60 | 1.16 | 0.78 |
| 5 D1S304 | 3.87 | 3.86 | 3.81 | 3.58 | 3.27 | 2.61 | 1.91 | 1.20 |
| 6 D1S2842 | 1.87 | 1.86 | 1.82 | 1.62 | 1.37 | 0.86 | 0.39 | 0.06 |
| 7 D1S404 | 2.39 | 2.38 | 2.33 | 2.11 | 1.83 | 1.26 | 0.69 | 0.21 |
| 8 D1S1609 | 2.36 | 2.36 | 2.32 | 2.14 | 1.91 | 1.42 | 0.92 | 0.48 |
| 9 D1S2811 | 5.31 | 5.30 | 5.24 | 4.95 | 4.56 | 3.75 | 2.86 | 1.91 |
| 10 D1S423 | 0.86 | 0.86 | 0.85 | 0.80 | 0.73 | 0.62 | 0.54 | 0.50 |
| 11 D1S2836 | −1.56 | −0.46 | 0.47 | 1.00 | 1.09 | 0.94 | 0.64 | 0.30 |
| 12 D1S2682 | −1.83 | −0.29 | 0.66 | 1.19 | 1.27 | 1.11 | 0.81 | 0.51 |
| Lod scores without LOH information | ||||||||
| 1 D1S2800 | −4.82 | −3.63 | −1.95 | −0.55 | −0.02 | 0.29 | 0.26 | 0.11 |
| 2 D1S517 | −2.37 | −2.33 | −1.53 | −0.39 | −0.02 | 0.13 | 0.09 | 0.03 |
| 3 D1S2785 | 2.87 | 2.86 | 2.81 | 2.57 | 2.27 | 1.65 | 1.02 | 0.40 |
| 4 D1S547 | 1.59 | 1.59 | 1.55 | 1.37 | 1.16 | 0.75 | 0.41 | 0.15 |
| 5 D1S304 | 2.67 | 2.66 | 2.61 | 2.38 | 2.09 | 1.47 | 0.83 | 0.26 |
| 6 D1S2842 | 1.87 | 1.86 | 1.82 | 1.62 | 1.37 | 0.86 | 0.39 | 0.06 |
| 7 D1S404 | 2.39 | 2.38 | 2.33 | 2.11 | 1.83 | 1.26 | 0.69 | 0.21 |
| 8 D1S1609 | 2.06 | 2.05 | 2.02 | 1.84 | 1.61 | 1.14 | 0.65 | 0.23 |
| 9 D1S2811 | 4.11 | 4.10 | 4.03 | 3.74 | 3.36 | 2.55 | 1.66 | 0.72 |
| 10 D1S423 | 0.26 | 0.26 | 0.25 | 0.22 | 0.17 | 0.1 | 0.04 | 0.01 |
| 11 D1S2836 | −1.79 | −0.76 | 0.17 | 0.70 | 0.79 | 0.66 | 0.40 | 0.13 |
| 12 D1S2682 | 1.18 | 1.18 | 1.16 | 1.06 | 0.92 | 0.61 | 0.31 | 0.08 |
Paraffin-embedded tissue-derived DNA was available from the three affected members of FAM-2 (Fig. 1). All shared the 1q42-q44 region from D1S2800 to D1S2682, and one renal cell cancer (FAM-2/II-3) displayed loss of the wild-type chromosome. In two of the 12 loci (D1S2842 and D1S1609), amplification of the paraffin-embedded tissue DNA was not successful. In three separate loci (D1S2785, D1S304, and D1S2682), the linked allele was of the same size in the two families. In the remaining seven loci, the linked allele was different. Thus, the linked haplotypes in the two families displayed little similarity.
Phenotype in the 20 Individuals Carrying a Disease-Associated Chromosome.
All 19 affected individuals from FAM-1 and FAM-2 with a disease-associated haplotype are listed in Table 1. In addition, one male displaying the linked haplotype in FAM-1 was unaffected. To summarize, 12 of the 16 females (75%) had uterine leiomyomas, and two (13%) had been diagnosed with leiomyosarcoma. Seven individuals (35%) had cutaneous nodules or leiomyomatosis. Six individuals (30%) had renal cell cancer. Because of the small number of males carrying the disease-associated chromosome, comparisons between the sexes were not possible.
Discussion
Multiple lines of evidence demonstrate that the kindreds studied display a previously unreported tumor-predisposition syndrome with distinct clinical, histopathological, and molecular features.
First, the tumor spectrum suggests a novel syndrome. The combination of susceptibility to uterine leiomyomas and renal cell cancer (HLRCC) has not been previously proposed. Of note, one publication of two kindreds from 1973 includes a description of a 20-year-old patient with uterine leiomyosarcoma and metastasized renal cell cancer (15). This patient and two of her four siblings also had cutaneous leiomyomas. The mother had uterine leiomyomas. Considering the rarity of kidney cancer, leiomyomatosis, and uterine leiomyosarcoma at such a young age, this case is of interest and supports the notion that the phenotypes are associated.
Second, the renal cell cancer phenotype alone is unique. The lack of characteristic renal and extrarenal features excludes typical VHL and tuberous sclerosis. The solitary nature of the lesions, as well as very aggressive disease, speak against both VHL and the MET germline mutation phenotype (4, 5, 16). The occurrence of uterine leiomyomas and kidney cancer in Eker rat, an animal model for tuberous sclerosis, is of some interest (17–20), although the human tuberous sclerosis phenotype is different (4, 5). The renal cell carcinomas in the two families displayed a rare and distinct histology with papillary architecture and characteristic cytology as the common features. One tumor had also cystic and solid areas and a sarcomatoid focus; these features occur in papillary carcinomas (12, 21). The epithelium was peculiar and did not correspond to a type 1 papillary renal carcinoma epithelium but had some resemblance to type 2 epithelium. The cytoplasm was abundant and the nuclei were large. In all tumors, very large inclusion-like nucleoli were seen (Fig. 3). The Fuhrman nuclear grade was 3 or 4. Reaction to cytokeratin 7 was null in all four tumors of the large kindred. An abundant presence of apoptotic cells was seen in all four cases. In contrast, papillary renal carcinomas with MET mutations show type 1 histology, small cells with pale cytoplasm, small oval nuclei with inconspicuous nucleoli, and low nuclear grade (22). Oedematous papillae, foamy macrophages, and psammoma bodies are frequent, whereas in type 2 they are uncommon and were not observed in our patients. That the exceptional histology could be used as a tool to identify new cases speaks strongly for a previously unknown hereditary tumor syndrome.
The third and most compelling piece of evidence is the identification of the HLRCC locus. Genome-wide linkage analysis demonstrated that the predisposition locus maps to 1q42–44. Detection of deletions at this locus in both kindreds in five of eight tumors, each deletion targeted at the normal chromosome, strongly supports this finding and suggests that the underlying genetic defect is associated with impaired tumor-suppressor or caretaker function (23). In the three tumors without large deletions, small deletions, inactivating mutations, and gene silencing through promotor hypermethylation are possible causes of loss of function. It is important to determine the full natural history, optimal management, frequency, and molecular background of HLRCC. The data presented here provide clinical, histopathological, and molecular tools for patient identification. Molecular tools include linkage to D1S2811 and the adjacent markers and demonstration of allelic losses targeted at the wild-type chromosome in tumors. Because leiomyomas in the condition are often multiple, many lesions per family are likely to be available.
Although further work is necessary to clarify the natural history of HLRCC, it is clear that the penetrance of the renal cell cancer phenotype is incomplete; in the large kindred, four of the 14 adult individuals with the affected chromosome had kidney cancer. Thus, many kindreds presenting with uterine leiomyomatosis alone,but at risk for renal cell cancer are likely to exist. That evaluation of 27 histological slides of young kidney cancer patients led to identification of a second HLRCC family emphasizes the need to clarify the incidence of the condition urgently.
Understanding the molecular mechanisms involved would facilitate tumor prevention and treatment in hereditary as well as sporadic forms of the disease. It is of particular significance that revealing the molecular background of HLRCC could give insight to formation and neoplastic potential of uterine leiomyomas, one of the most common human tumor types. In addition to pathologists, clinicians treating renal cell cancer and leiomyomas play a key role in patient identification. Additional material will facilitate clinical and molecular studies on the intriguing tumor-predisposition phenotype. Furthermore, accurate diagnosis of HLRCC should result in more-efficient cancer detection and prevention in the affected kindreds.
Acknowledgments
We thank Kristiina Aittomäki and Heikki Järvinen for advice, and Sinikka Lindh, Riitta Mattlar, Siv Lindroos, Annika Lahti, and Kirsi Laukkanen for their technical assistance. Most of the genotyping was done at the Finnish Genome Center (Helsinki, Finland). This study was supported by grants from the Finnish Cancer Society, Helsinki University Central Hospital, Biocentrum Helsinki, the Sigrid Juselius Foundation, and the Academy of Finland (44870, Finnish Center of Excellence Program 2000-2005).
Abbreviations
- VHL
von Hippel-Lindau
- H&E
hematoxylin/eosin
- HMFG
human milk fat globule
- lod
logarithm of odds
- LOH
loss of heterozygosity
References
- 1.Latif F, Tory K, Gnarra J, Yao M, Duh F M, Orcutt M L, Stackhouse T, Kuzmin I, Modi W, Geil L, et al. Science. 1993;260:1317–1320. doi: 10.1126/science.8493574. [DOI] [PubMed] [Google Scholar]
- 2.Schmidt L, Duh F M, Chen F, Kishida T, Glenn G, Choyke P, Scherer S W, Zhuang Z, Lubensky I, Dean M, et al. Nat Genet. 1997;16:68–73. doi: 10.1038/ng0597-68. [DOI] [PubMed] [Google Scholar]
- 3.Cohen A J, Li F P, Berg S, Marchetto D J, Tsai S, Jacobs C S, Brown R S. N Engl J Med. 1979;301:592–595. doi: 10.1056/NEJM197909133011107. [DOI] [PubMed] [Google Scholar]
- 4.Zbar B, Lerman M. Adv Cancer Res. 1998;75:163–201. doi: 10.1016/s0065-230x(08)60742-3. [DOI] [PubMed] [Google Scholar]
- 5.Iliopoulos O, Eng C. Semin Oncol. 2000;27:138–149. [PubMed] [Google Scholar]
- 6.Lynch H T, Lynch J F. Semin Surg Oncol. 2000;18:305–313. doi: 10.1002/(sici)1098-2388(200006)18:4<305::aid-ssu5>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 7.Malchoff C D, Sarfarazi M, Tendler B, Forouhar F, Whalen G, Joshi V, Arnold A, Malchoff D M. J Clin Endocrinol Metab. 2000;85:1758–1764. doi: 10.1210/jcem.85.5.6557. [DOI] [PubMed] [Google Scholar]
- 8.Teh B T, Giraud S, Sari N F, Hii S I, Bergerat J P, Larsson C, Limarcher J M, Nicol D. Lancet. 1997;349:848–849. doi: 10.1016/S0140-6736(05)61751-5. [DOI] [PubMed] [Google Scholar]
- 9.Teppo L, Pukkala E, Lehtonen M. Acta Oncol. 1994;33:365–369. doi: 10.3109/02841869409098430. [DOI] [PubMed] [Google Scholar]
- 10.Murphy W M, Beckwith J B, Farrow G M. Tumors of the Kidney, Bladder and Related Urinary Structures. Washington, DC: Armed Forces Institute of Pathology; 1994. [Google Scholar]
- 11.Delahunt B, Eble J N. Mod Pathol. 1997;10:537–544. [PubMed] [Google Scholar]
- 12.Kruglyak L, Daly M J, Reeve-Daly M P, Lander E S. Am J Hum Genet. 1996;58:1347–1363. [PMC free article] [PubMed] [Google Scholar]
- 13.Cottingham R W, Jr, Idury R M, Schaffer A A. Am J Hum Genet. 1993;53:252–263. [PMC free article] [PubMed] [Google Scholar]
- 14.Rohde K, Teare M D, Scherneck S, Santibanez Koref M. Hum Hered. 1995;45:337–345. doi: 10.1159/000154302. [DOI] [PubMed] [Google Scholar]
- 15.Reed W B, Walker R, Horowitz R. Acta Derm Venereol. 1973;53:409–416. [PubMed] [Google Scholar]
- 16.Walther M M, Choyke P L, Glenn G, Lyne J C, Rayford W, Venzon D, Linehan W M. J Urol. 1999;161:1475–1479. doi: 10.1016/s0022-5347(05)68930-6. [DOI] [PubMed] [Google Scholar]
- 17.Yeung R S, Xiao G-H, Jin F, Lee W-C, Testa J R, Knudson A G. Proc Natl Acad Sci USA. 1994;91:11413–11416. doi: 10.1073/pnas.91.24.11413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kobayashi T, Hirayama Y, Kobayashi E, Kubo Y, Hino O. Nat Genet. 1995;9:70–74. doi: 10.1038/ng0195-70. [DOI] [PubMed] [Google Scholar]
- 19.Everitt J I, Wolf D C, Howe S R, Goldsworthy T L, Walker C. Am J Pathol. 1995;146:1556–1567. [PMC free article] [PubMed] [Google Scholar]
- 20.Hino O, Fukuda T, Satake N, Kobayashi T, Honda S, Orimoto K, Yamashita Y, Kikuchi Y. Prog Exp Tumor Res. 1999;35:95–108. doi: 10.1159/000062006. [DOI] [PubMed] [Google Scholar]
- 21.Reuter V E, Presti J C., Jr Semin Oncol. 2000;27:124–137. [PubMed] [Google Scholar]
- 22.Lubensky I A, Schmidt L, Zhuang Z, Weirich G, Pack S, Zambrano N, Walther M M, Choyke P, Linehan W M, Zbar B. Am J Pathol. 1999;155:517–526. doi: 10.1016/S0002-9440(10)65147-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Knudson A G. Proc Natl Acad Sci USA. 1971;68:820–823. doi: 10.1073/pnas.68.4.820. [DOI] [PMC free article] [PubMed] [Google Scholar]