Abstract
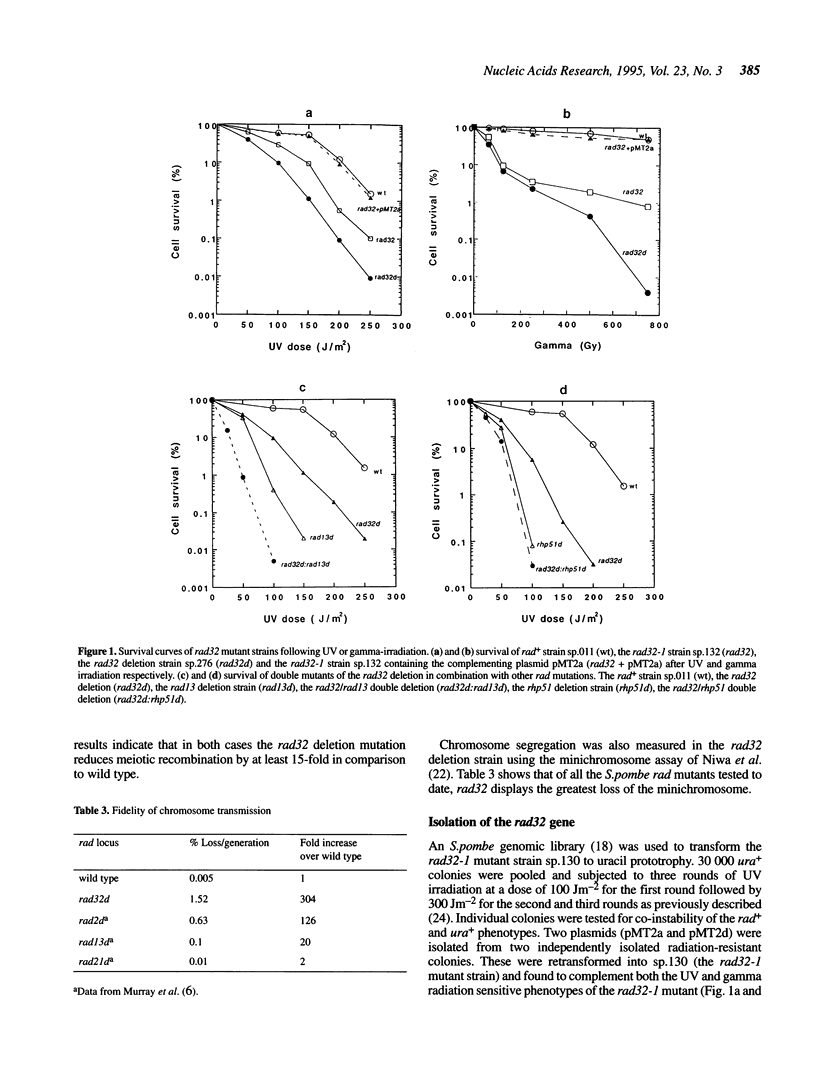
A new Schizosaccharomyces pombe mutant (rad32) which is sensitive to gamma and UV irradiation is described. Pulsed field gel electrophoresis of DNA from irradiated cells indicates that the rad32 mutant, in comparison to wild type cells, has decreased ability to repair DNA double strand breaks. The mutant also undergoes decreased meiotic recombination and displays reduced stability of minichromosomes. The rad32 gene has been cloned by complementation of the UV sensitive phenotype. The gene, which is not essential for cell viability and is expressed at a moderate level in mitotically dividing cells, has significant homology to the meiotic recombination gene MRE11 of Saccharomyces cerevisiae. Epistasis analysis indicates that rad32 functions in a pathway which includes the rhp51 gene (the S.pombe homologue to S.cerevisiae RAD51) and that cells deleted for the rad32 gene in conjunction with either the rad3 deletion (a G2 checkpoint mutation) or the rad2 deletion (a chromosome stability and potential nucleotide excision repair mutation) are not viable.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ajimura M., Leem S. H., Ogawa H. Identification of new genes required for meiotic recombination in Saccharomyces cerevisiae. Genetics. 1993 Jan;133(1):51–66. doi: 10.1093/genetics/133.1.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbet N., Muriel W. J., Carr A. M. Versatile shuttle vectors and genomic libraries for use with Schizosaccharomyces pombe. Gene. 1992 May 1;114(1):59–66. doi: 10.1016/0378-1119(92)90707-v. [DOI] [PubMed] [Google Scholar]
- Benton B. K., Reid M. S., Okayama H. A Schizosaccharomyces pombe gene that promotes sexual differentiation encodes a helix-loop-helix protein with homology to MyoD. EMBO J. 1993 Jan;12(1):135–143. doi: 10.1002/j.1460-2075.1993.tb05639.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezzubova O. Y., Buerstedde J. M. Gene conversion in the chicken immunoglobulin locus: a paradigm of homologous recombination in higher eukaryotes. Experientia. 1994 Mar 15;50(3):270–276. doi: 10.1007/BF01924010. [DOI] [PubMed] [Google Scholar]
- Bezzubova O. Y., Schmidt H., Ostermann K., Heyer W. D., Buerstedde J. M. Identification of a chicken RAD52 homologue suggests conservation of the RAD52 recombination pathway throughout the evolution of higher eukaryotes. Nucleic Acids Res. 1993 Dec 25;21(25):5945–5949. doi: 10.1093/nar/21.25.5945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birkenbihl R. P., Subramani S. Cloning and characterization of rad21 an essential gene of Schizosaccharomyces pombe involved in DNA double-strand-break repair. Nucleic Acids Res. 1992 Dec 25;20(24):6605–6611. doi: 10.1093/nar/20.24.6605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr A. M., Sheldrick K. S., Murray J. M., al-Harithy R., Watts F. Z., Lehmann A. R. Evolutionary conservation of excision repair in Schizosaccharomyces pombe: evidence for a family of sequences related to the Saccharomyces cerevisiae RAD2 gene. Nucleic Acids Res. 1993 Mar 25;21(6):1345–1349. doi: 10.1093/nar/21.6.1345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Veaux L. C., Hoagland N. A., Smith G. R. Seventeen complementation groups of mutations decreasing meiotic recombination in Schizosaccharomyces pombe. Genetics. 1992 Feb;130(2):251–262. doi: 10.1093/genetics/130.2.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feaver W. J., Svejstrup J. Q., Bardwell L., Bardwell A. J., Buratowski S., Gulyas K. D., Donahue T. F., Friedberg E. C., Kornberg R. D. Dual roles of a multiprotein complex from S. cerevisiae in transcription and DNA repair. Cell. 1993 Dec 31;75(7):1379–1387. doi: 10.1016/0092-8674(93)90624-y. [DOI] [PubMed] [Google Scholar]
- Fikes J. D., Becker D. M., Winston F., Guarente L. Striking conservation of TFIID in Schizosaccharomyces pombe and Saccharomyces cerevisiae. Nature. 1990 Jul 19;346(6281):291–294. doi: 10.1038/346291a0. [DOI] [PubMed] [Google Scholar]
- Forsburg S. L., Nurse P. Analysis of the Schizosaccharomyces pombe cyclin puc1: evidence for a role in cell cycle exit. J Cell Sci. 1994 Mar;107(Pt 3):601–613. [PubMed] [Google Scholar]
- Friedberg E. C., Bardwell A. J., Bardwell L., Wang Z., Dianov G. Transcription and nucleotide excision repair--reflections, considerations and recent biochemical insights. Mutat Res. 1994 May 1;307(1):5–14. doi: 10.1016/0027-5107(94)90272-0. [DOI] [PubMed] [Google Scholar]
- Friedberg E. C. Deoxyribonucleic acid repair in the yeast Saccharomyces cerevisiae. Microbiol Rev. 1988 Mar;52(1):70–102. doi: 10.1128/mr.52.1.70-102.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm C., Kohli J., Murray J., Maundrell K. Genetic engineering of Schizosaccharomyces pombe: a system for gene disruption and replacement using the ura4 gene as a selectable marker. Mol Gen Genet. 1988 Dec;215(1):81–86. doi: 10.1007/BF00331307. [DOI] [PubMed] [Google Scholar]
- Gutz H. Site Specific Induction of Gene Conversion in SCHIZOSACCHAROMYCES POMBE. Genetics. 1971 Nov;69(3):317–337. doi: 10.1093/genetics/69.3.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Klapholz S., Waddell C. S., Esposito R. E. The role of the SPO11 gene in meiotic recombination in yeast. Genetics. 1985 Jun;110(2):187–216. doi: 10.1093/genetics/110.2.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieberman H. B., Riley R., Martel M. Isolation and initial characterization of a Schizosaccharomyces pombe mutant exhibiting temperature-dependent radiation sensitivity due to a mutation in a previously unidentified rad locus. Mol Gen Genet. 1989 Sep;218(3):554–558. doi: 10.1007/BF00332423. [DOI] [PubMed] [Google Scholar]
- Maier E., Hoheisel J. D., McCarthy L., Mott R., Grigoriev A. V., Monaco A. P., Larin Z., Lehrach H. Complete coverage of the Schizosaccharomyces pombe genome in yeast artificial chromosomes. Nat Genet. 1992 Jul;1(4):273–277. doi: 10.1038/ng0792-273. [DOI] [PubMed] [Google Scholar]
- Murray J. M., Carr A. M., Lehmann A. R., Watts F. Z. Cloning and characterisation of the rad9 DNA repair gene from Schizosaccharomyces pombe. Nucleic Acids Res. 1991 Jul 11;19(13):3525–3531. doi: 10.1093/nar/19.13.3525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray J. M., Doe C. L., Schenk P., Carr A. M., Lehmann A. R., Watts F. Z. Cloning and characterisation of the S. pombe rad15 gene, a homologue to the S. cerevisiae RAD3 and human ERCC2 genes. Nucleic Acids Res. 1992 Jun 11;20(11):2673–2678. doi: 10.1093/nar/20.11.2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray J. M., Tavassoli M., al-Harithy R., Sheldrick K. S., Lehmann A. R., Carr A. M., Watts F. Z. Structural and functional conservation of the human homolog of the Schizosaccharomyces pombe rad2 gene, which is required for chromosome segregation and recovery from DNA damage. Mol Cell Biol. 1994 Jul;14(7):4878–4888. doi: 10.1128/mcb.14.7.4878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nurse P. Universal control mechanism regulating onset of M-phase. Nature. 1990 Apr 5;344(6266):503–508. doi: 10.1038/344503a0. [DOI] [PubMed] [Google Scholar]
- Ostermann K., Lorentz A., Schmidt H. The fission yeast rad22 gene, having a function in mating-type switching and repair of DNA damages, encodes a protein homolog to Rad52 of Saccharomyces cerevisiae. Nucleic Acids Res. 1993 Dec 25;21(25):5940–5944. doi: 10.1093/nar/21.25.5940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phipps J., Nasim A., Miller D. R. Recovery, repair, and mutagenesis in Schizosaccharomyces pombe. Adv Genet. 1985;23:1–72. doi: 10.1016/s0065-2660(08)60511-8. [DOI] [PubMed] [Google Scholar]
- Schaeffer L., Moncollin V., Roy R., Staub A., Mezzina M., Sarasin A., Weeda G., Hoeijmakers J. H., Egly J. M. The ERCC2/DNA repair protein is associated with the class II BTF2/TFIIH transcription factor. EMBO J. 1994 May 15;13(10):2388–2392. doi: 10.1002/j.1460-2075.1994.tb06522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinohara A., Ogawa H., Matsuda Y., Ushio N., Ikeo K., Ogawa T. Cloning of human, mouse and fission yeast recombination genes homologous to RAD51 and recA. Nat Genet. 1993 Jul;4(3):239–243. doi: 10.1038/ng0793-239. [DOI] [PubMed] [Google Scholar]
- Shinohara A., Ogawa H., Ogawa T. Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell. 1992 May 1;69(3):457–470. doi: 10.1016/0092-8674(92)90447-k. [DOI] [PubMed] [Google Scholar]
- al-Khodairy F., Carr A. M. DNA repair mutants defining G2 checkpoint pathways in Schizosaccharomyces pombe. EMBO J. 1992 Apr;11(4):1343–1350. doi: 10.1002/j.1460-2075.1992.tb05179.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- al-Khodairy F., Fotou E., Sheldrick K. S., Griffiths D. J., Lehmann A. R., Carr A. M. Identification and characterization of new elements involved in checkpoint and feedback controls in fission yeast. Mol Biol Cell. 1994 Feb;5(2):147–160. doi: 10.1091/mbc.5.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]





