Abstract
Heterotrophic microbial communities in seawater and sediments metabolize much of the organic carbon produced in the ocean. Although carbon cycling and preservation depend critically on the capabilities of these microbial communities, their compositions and capabilities have seldom been examined simultaneously at the same site. To compare the abilities of seawater and sedimentary microbial communities to initiate organic matter degradation, we measured the extracellular enzymatic hydrolysis rates of 10 substrates (polysaccharides and algal extracts) in surface seawater and bottom water as well as in surface and anoxic sediments of an Arctic fjord. Patterns of enzyme activities differed between seawater and sediments, not just quantitatively, in accordance with higher cell numbers in sediments, but also in their more diversified enzyme spectrum. Sedimentary microbial communities hydrolyzed all of the fluorescently labeled polysaccharide and algal extracts, in most cases at higher rates in subsurface than surface sediments. In seawater, in contrast, only 5 of the 7 polysaccharides and 2 of the 3 algal extracts were hydrolyzed, and hydrolysis rates in surface and deepwater were virtually identical. To compare bacterial communities, 16S rRNA gene clone libraries were constructed from the same seawater and sediment samples; they diverged strongly in composition. Thus, the broader enzymatic capabilities of the sedimentary microbial communities may result from the compositional differences between seawater and sedimentary microbial communities, rather than from gene expression differences among compositionally similar communities. The greater number of phylum- and subphylum-level lineages and operational taxonomic units in sediments than in seawater samples may reflect the necessity of a wider range of enzymatic capabilities and strategies to access organic matter that has already been degraded during passage through the water column. When transformations of marine organic matter are considered, differences in community composition and their different abilities to access organic matter should be taken into account.
The degradation of marine organic matter in the ocean is due in large part to the concerted effects of microbial communities in the water column and in the sediments, which collectively reprocess and ultimately remineralize most of the organic matter initially produced in the surface ocean (18, 39). These communities rely on extracellular enzymes to hydrolyze high-molecular-weight organic matter to sizes sufficiently small for cellular uptake. They then respire a fraction of the organic carbon and excrete transformation products as dissolved organic carbon (DOC) that may in turn be used by other members of the microbial community. The composition and capabilities of heterotrophic microbial communities in seawater and sediments thus play a critical role in the global carbon cycle. Although numerous geochemical investigations of carbon cycling have followed the transformations of particulate organic matter as it sinks through the water column into the sediments (49, 74), investigations of microbial activity and composition very rarely cover the same trajectory, focusing instead solely on either sedimentary or seawater environments (19, 20, 27), with very few studies investigating both at the same time and place (41). This fundamental disconnect in carbon cycling studies means that the extent to which seawater and sedimentary microbial communities resemble one another from both compositional and functional perspectives is almost entirely unexplored.
Differences in physical and geochemical parameters (extent of mixing, available electron acceptors, nature and type of organic carbon substrates, nature of surface area) in the water column and in sediments, however, suggest that heterotrophic microbial communities in these environments are confronted by fundamentally different challenges. Differing strategies may be carried out by compositionally different communities, a hypothesis supported by a comparison of data on microbial community composition obtained at different times and different sites. The few studies conducted on sediments and seawater from the same location (in tropical and coral reef environments [41]) have indeed found significant compositional differences. Specific functional comparisons of microbial communities in the water column and sediments are likewise sparse. The observation that organic carbon substrates change in composition and quality with depth in the water column and into the sediment (i.e., through changes in particle C/N ratios and decreases in identifiable biochemical components [49, 74]) suggests that strategies to access organic matter may differ as well.
Tracking microbial transformation of organic matter is extremely challenging, however, due to analytical limitations in efforts to resolve specific changes in substrate structure (40, 49). Investigations of organic matter transformations by heterotrophic microbial communities usually measure changes in bulk parameters or transformations of lower-molecular-weight compounds (72, 77). Initiation of organic matter degradation via extracellular enzymes is typically monitored through use of small-substrate proxies. These proxies consist of an amino acid or monosaccharide linked to a fluorophore that fluoresces upon hydrolysis of the monomer-fluorophore bond (43). Although they are simple to use, these proxies lack the structural features of macromolecules. The relationship between data obtained using low-molecular-weight substrate proxies and the hydrolysis of the polymers that they are intended to represent is uncertain (75). Moreover, these substrate proxies do not reflect the activities of endo-acting extracellular enzymes used to cleave high-molecular-weight substrates midchain (13).
To overcome some of these limitations and to investigate the structural specificities within major classes of extracellular enzymes, alternative approaches using fluorescently labeled high-molecular-weight polysaccharides and peptides were developed (7, 57). Enzyme activity is detected as a change in substrate molecular weight as the substrate is progressively hydrolyzed. These substrates permit comparison of hydrolysis rates as a function of specific composition, and they can detect the activities of endo-acting (midchain-cleaving) enzymes.
Investigations with fluorescently labeled polysaccharides have demonstrated that the rates and structural specificities of extracellular enzymes in seawater and in organic-rich muddy sediments are very different (8, 11). Sedimentary microbial communities produce a broader spectrum of extracellular enzymes than do water column communities from the same locations, differences that do not scale with likely differences in cell counts (8, 11). The sedimentary communities therefore have a broader range of hydrolytic capabilities to initiate carbon cycling, perhaps a necessary strategy in environments where readily accessed substrates may have already been removed during passage through the water column. Whether these observed differences are due to compositionally different microbial communities with a different range of tools or are the outcome of differences in gene regulation between compositionally similar communities is an unanswered question, since comparisons of seawater and sedimentary microbial communities from the same site are almost entirely lacking. Furthermore, the extent to which microbial community composition and capabilities differ may vary by environment. In an investigation of the shallow water column over highly permeable sandy sediments, the spectrum of enzyme activities closely resembled that of the underlying sediments, perhaps due to active flushing of sediments that transported enzymes and/or organisms into the overlying water column (17).
In high-latitude environments, microbial communities in sea ice, the water column, and sediments efficiently cycle organic carbon, despite the permanently cold temperatures (12, 15, 45, 50, 78). Targeted comparisons of pelagic microbial communities with their temperate counterparts have demonstrated that high-latitude communities differ in composition from their more temperate counterparts (21, 22, 32). Bacteria exhibiting growth characteristics well-suited to permanently cold temperatures have also been isolated and characterized from Arctic sediments (46, 73). Direct comparisons of microbial community compositions in seawater and sediments in high-latitude environments, to the best of our knowledge, are entirely lacking. Therefore, we compared the compositional as well as functional characteristics of heterotrophic sedimentary and seawater microbial communities from a high-latitude site.
To investigate microbial community composition as well as microbial function in the water column and sediments in the Arctic, we compared clone libraries and extracellular enzyme activities in surface and near-bottom waters with those in surface (0 to 2 cm) and subsurface (3 to 9 cm) sediments from a fjord of Svalbard, an archipelago in the high Arctic. Our purpose was to determine whether differences previously observed between extracellular enzyme activities in surface waters and surface sediments (11) also extend to deep waters and subsurface sediments and whether these differences are reflected in the composition of the microbial communities in the same samples. For this study, we collected samples simultaneously for measurements of extracellular enzyme activities and for examination of microbial community composition. We focused on the activities of polysaccharide-hydrolyzing enzymes because polysaccharides are major components of phytoplankton biomass, as well as of particulate and dissolved organic carbon in seawater and sediments (14, 24, 30, 56). Previous work with sediments of Svalbard had demonstrated that carbohydrates can potentially meet a significant fraction of benthic microbial carbon requirements (16) and thus can be important substrates for heterotrophic microbial communities.
MATERIALS AND METHODS
Study site and sample collection.
Seawater and sediment samples were collected at Station J, Smeerenburg Fjord (16), 79.42°N, 11.05°W, where the water column depth was 211 m. The surface-water temperature was 5°C, the bottom-water temperature was 2.8°C, and the sediment temperature was 2.3°C. The upper 2 cm of these sediments is soft and brown, grading to darker brown and black at deeper depths. Brittle stars and worm tubes are frequently found at this site (16). Surface water (collected at a depth of ca. 2 m) and bottom water (collected at a depth of 195 m) were transferred to 5-liter carboys from a 10-liter Niskin bottle. Water samples for clone library analysis (750 ml) were immediately filtered with a hand pump through a Millipore Durapore 0.22-μm-pore-size filter. The filters were transferred to sterile plastic petri dishes and immediately frozen on dry ice. Water for enzyme activity measurements was maintained at ca. 4°C until experiments were initiated at the shore lab, after ca. 18 h of transport and setup. Sediment was collected via a Haps core. Sediments for enzyme activity measurements were maintained at ca. 4°C until experiments were initiated at the shore lab. For clone library analysis, surface sediments (25 ml, from the upper 0 to 2 cm; light brown oxidized sediment) were transferred by spatula into a clean 50-ml centrifuge tube. Brown/black sediments (25 ml) with a faint smell of H2S from the sulfate-reducing zone at the 3- to 9-cm depth (13) were also transferred into a 50-ml centrifuge tube, and both tubes were immediately frozen on dry ice. All samples for clone library analysis were kept frozen on dry ice until they were transferred to a −80°C freezer for storage prior to processing.
Measurements of enzyme activities.
Extracellular enzyme activities were measured using fluorescently labeled polysaccharides and algal extracts as substrates (6). Ten substrates that span a range of structural complexity were selected for a focused study of enzyme activities within the polysaccharides, a broad class of biomolecules quantitatively significant for marine carbon metabolism: pullulan [α(1,6)-linked maltotriose (glucose)], laminarin [β(1,3-glucose)], xylan (xylose), fucoidan (sulfated fucose), arabinogalactan (arabinose and galactose), alginic acid (mannuronic and glucuronic acids), and chondroitin sulfate (sulfated N-acetylgalactoseamine and glucuronic acid) were all purchased from Sigma or Fluka. These polysaccharides are constituents of marine algae (56) and/or the activities of enzymes hydrolyzing these substrates have been demonstrated in marine bacteria (3, 5, 25). Genes encoding enzymes that hydrolyze these polysaccharides have also been identified in the genomes of fully sequenced marine bacteria (23, 36, 76). Carbohydrate-rich high-molecular-weight extracts of Isochrysis spp., wakame, and spirulina were extracted and characterized as described previously (6). These algal extracts are chemically more complex than purified polysaccharides; thus, they represent material a step closer to the complex organic matter naturally occurring in marine environments. The Isochrysis extract is primarily (82%) (1,3)-linked glucose, the wakame extract is primarily (75%) fucose and mannuronic acid, and the spirulina extract includes a wide variety of carbohydrates, including 44% rhamnose and 13% glucuronic acid; further detailed chemical compositions are reported elsewhere (11).
The polysaccharides and extracts were labeled with fluoresceinamine (isomer II; Sigma) using the method of Glabe and coworkers (34), as modified by Arnosti (6, 7, 9). In brief, polysaccharides or extracts were dissolved in Milli-Q H2O and activated with CNBr. Fluoresceinamine was added, the reaction mixture was incubated, and the labeled polysaccharides were purified using column chromatography. The labeling density of these substrates is typically quite low; depending on the substrate, between 0.02% and 3% of the monomers in a polymer chain is labeled (9). Physical interference of the fluorescent tag with the polysaccharide is therefore minimal. Previously, it has been demonstrated that labeled and unlabeled polysaccharides do not differ in activity in endothelial monolayer binding assays and in inhibition of lectin-mediated hemagglutination (34).
Enzymatic activities were measured in seawater and sediments in experiments initiated at the shore lab in Ny Ålesund, Svalbard, ca. 18 h after sample collection. Samples analyzed for seawater enzyme activity were collected in glass jars and stored at in situ temperatures. In all experiments, samples (surface and bottom water, as well as surface and subsurface sediments) were subsequently incubated at 4°C in the dark, without shaking. Each 50-ml portion of seawater received a single substrate to achieve a concentration of 3.5 μmol liter−1 monomer-equivalent polysaccharide. This approach ensures the same substrate concentration in every incubation, irrespective of differences in the molecular weights of the polysaccharides. The 50-ml portion was then divided into three 16.7-ml replicates. Killed controls consisted of autoclaved seawater, to which substrate was added after the seawater had cooled. Since the equipment necessary to monitor substrate degradation was not available at the shore lab, sampling time points were selected on the basis of previous work in surface waters at this site (11). Incubations were sampled after 0 days, 2 days, 4 days, 10 days, 15 days, and 21 days. At each time point, ca. 1.5 ml sample was removed from each vial, filtered through a 0.2-μm-pore-size surfactant-free cellulose acetate filter, and then stored frozen until analysis in the home lab.
Measurements of enzyme activities in sediments were made by homogenizing sediments (from 0 to 2 cm of the surface layer and 3 to 9 cm of the subsurface layer) thoroughly and then transferring replicate 20-ml portions of sediments to 50-ml centrifuge tubes for individual incubations. The deep sediments were gassed with N2 during processing and stored in N2-filled gastight bags. For isochrysis extract, wakame extract, spirulina extract, and alginic acid, only 15-ml portions of sediment were used, and substrate addition was reduced proportionately. Since the analytical equipment necessary to measure the substrate signal and monitor substrate transformations was not available at the shore lab in Ny Ålesund, substrate addition levels as well as the time course of incubation were based on previous experience with sediments from this fjord (11, 15). Each substrate was added to triplicate tubes. For surface sediments, two substrate addition levels were used, with triplicate sets receiving 3.5 μmol and 7.0 μmol per tube (equivalent to 175 and 350 μmol liter−1 of sediment, respectively). Substrate addition levels for deep sediments were 7.0 μmol per tube. Controls consisted of a 1:1 slurry of seawater and sediment that was autoclaved and cooled prior to substrate addition. At each time point (0 h, 24 h, 45 h, and 60 h), tubes were centrifuged in a refrigerated centrifuge to obtain a small amount of pore water, which was filtered through a 0.2-μm-pore-size filter and frozen until analysis. Samples were rehomogenized, and deep sediment samples were gassed again with N2 and incubated at 4°C in the dark.
Seawater and pore water samples were shipped frozen to the home lab, where they were thawed and injected via autosampler on a gel permeation chromatography/high-pressure liquid chromatography system with fluorescent detection as described previously (6, 9). Hydrolysis rates were calculated from the molecular weight distribution of the substrates as they were progressively hydrolyzed (6, 9).
The rates reported here are potential rates, since they were measured with an externally added substrate that competes with naturally occurring substrates of unknown concentration for enzyme active sites. The concentration of carbohydrates added to seawater likely doubles the concentration of total dissolved carbohydrates, assuming a DOC concentration of ca. 80 μM and a carbohydrate concentration of 25% of DOC (24); more precise data from Svalbard fjords, to the best of our knowledge, are not available. Sedimentary carbohydrate additions represent approximately a 1- to 2-fold increase in pore water total dissolved carbohydrates (16); particulate carbohydrates, which fuel the dissolved carbohydrate pool, are not included in this calculation. These carbohydrate additions likely represent saturation conditions, which would make the hydrolysis rates zero order (independent of substrate concentration) with respect to enzyme kinetics.
DNA extraction.
To extract and purify DNA from seawater filters in the laboratory, small pieces of frozen filters (∼35-mg filter mass) were put into 2-ml centrifuge tubes (one tube per depth) containing 200 μl of 10 mM Tris-1 mM EDTA buffer (TE) with 0.5% (wt/vol) sodium dodecyl sulfate and proteinase K (50 μg ml−1). Tubes were incubated at room temperature for 30 min; tubes with buffer and no filter material served as blanks. DNA was extracted by adding 200 μl of a pH 8-equilibrated phenol-chloroform-isoamyl alcohol mixture (25:24:1 [vol/vol/vol]) and incubated for 5 min at room temperature. Tubes were then centrifuged at the highest speed for 5 min, and aqueous phases were transferred into new centrifuge tubes. The phenol phases were mixed with 200 μl TE, incubated for 5 min at room temperature, and centrifuged for 5 min. The aqueous extracts were combined with the previous aqueous phases. DNA was precipitated in 10% (vol/vol) 5 M NaCl and 2.5 volumes of 100% ethanol overnight at room temperature. Tubes were then centrifuged at the highest speed for 15 min, and pellets were washed with 70% ethanol, air dried, and resuspended in 200 μl sterile RNA-free water (Qiagen, Valencia, CA). DNA from sediment was extracted and purified from 250 mg sediment using an UltraClean soil DNA kit (MoBio Laboratories, Inc., Solana Beach, CA), following the manufacturer's instructions.
PCR amplification, cloning, and sequencing of 16S rRNA genes.
The bacterial 16S rRNA gene for cloning was amplified by PCR using the bacterial primers 8f and 1492r (70). Each 25-μl PCR mixture contained 1 μl DNA template, 0.3 μl primer solution (100 pmol μl−1), 1 μl bovine serum albumin (10 mg ml−1), 2.5 μl 10× PCR buffer (Fast Buffer 1; Promega, Madison, WI), 4 μl deoxynucleoside triphosphate (2.5 mM each deoxynucleoside triphosphate), 16.075 μl sterile RNA-free water (Qiagen), and 0.125 μl (0.625 units) Speed Star Taq polymerase (Promega). PCR amplification was performed in a Bio-Rad iCycler thermal cycler. The conditions were as follows: an initial denaturation at 94°C for 2 min, followed by 25 cycles, each consisting of 10 s of denaturation at 98°C, 15 s at a primer-annealing temperature of 58°C, and 20 s of elongation at 72°C, plus a final cycle of 72°C for 10 min. The PCR products were visualized on a 1.5% agarose gel using an ethidium bromide stain. Blank extractions, serving as controls for contamination during DNA extraction, yielded no visible products. All PCR products were purified with the MoBio PCR cleanup kit and cloned using the TOPO TA PCR cloning kit before being transformed into Escherichia coli following the manufacturer's protocols (Invitrogen, San Diego, CA). Nearly complete 16S rRNA gene sequences were obtained from Genewiz, Inc. These directly amplified clone colony DNA using a nonspecific amplification procedure, followed by a single-primer sequencing reaction using either 8f/1492r or M13 vector primers.
Sequence analysis.
Sequence reads were checked and assembled into contigs using the Sequencher program (Gene Codes Corp). Assembled contigs were initially screened for chimeras using the Bellerophon 3 program (44) and the default settings. Three percent operational taxonomic unit (OTU) groupings were then defined using the DOTUR program and provided the basis for rarefaction curves (66). Representative sequences for each 3% OTU were then aligned in the ARB program, with final adjustments made by eye, to closest relatives in the SILVA (version 95) reference database (60). After the addition of representative phylotypes, all sequences that appeared in the final tree were checked for chimeras by running them through the Bellerophon 3 program (44), using a window size of 200 bp and a divergence ratio cutoff of 1.08. Selected sequences flagged as chimeras were then double-checked using the Pintail program and excluded from the data set. The alignment used for phylogeny consisted of sequence regions (ca. 1,200 nucleotides) that passed a 40% conservation filter using the filter tool in ARB. The bacterial phylogenies were estimated using the neighbor-joining algorithm (65), a maximum-likelihood-estimated model of evolution, and a gamma-corrected rate distribution in MEGA (version 4.0) software (48). Branch support was estimated with a 2,000-replicate interior branch (IB) test (55), which uses a hypothesis-testing approach to determine the probability that a particular interior branch has a length greater than 0, i.e., that the node in question should not be collapsed into a polytomy. Values greater than 95% should be considered strong support for a particular node (55). In phylogenetic trees of this study, bacterial clades were defined using the most-basal marker sequences from published phylogenies and were named and referenced in accordance with published phylogenies. Bacterial clades showed long-stem branches relative to internal branch lengths and were confirmed by high interior branch support.
Nucleotide sequence accession numbers.
The sequences have GenBank accession numbers GQ259213 to GQ259326 (see Table S1 in the supplemental material).
RESULTS
Extracellular enzyme activities in seawater and sediments.
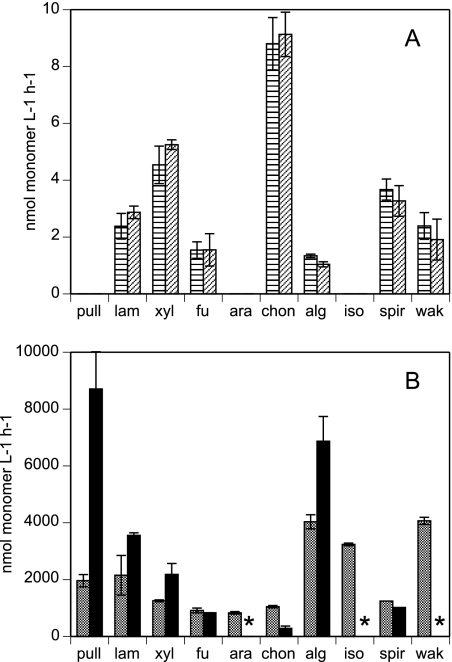
Patterns of enzyme activities in surface waters were identical to those in bottom waters (Fig. 1), despite the 2.2°C temperature difference between surface and bottom waters, which likely indicates a difference in water mass. Laminarin, xylan, fucoidan, chondroitin, alginic acid, wakame extract, and spirulina extract were all hydrolyzed in both surface and bottom waters at virtually identical maximum rates. There was no detectable hydrolysis of pullulan, arabinogalactan, or isochrysis extract over the 21-day time course of incubation in either surface or bottom water. Hydrolysis rates of the substrates in surface and bottom water ranged from nearly 9 nmol monomer liter−1 h−1 (for chondroitin) to undetectable (for pullulan, arabinogalactan, and isochrysis extract). Maximum rates for surface and bottom water (Fig. 1A) were measured after 10 days of incubation for chondroitin, wakame, and spirulina (surface and deep) and xylan (deep), at 15 days for xylan (surface), laminarin (deep), and alginic acid (surface and deep), and at 21 days for laminarin (surface) and fucoidan (surface and deep).
FIG. 1.
Hydrolysis rates of polysaccharides and plankton extracts in surface-water (horizontal-stripe bars) and bottom-water (slanted-stripe bars) samples (A) and in homogenized sediments from depths of 0 to 2 cm (gray bars) and 3 to 9 cm (black bars) (B). Substrates are pull, pullulan; lam, laminarin; xyl, xylan; fu, fucoidan; ara, arabinogalactan; chon, chondroitin sulfate; alg, alginic acid; iso, Isochrysis extract; spir, Spirulina extract; wak, wakame extract. Error bars show standard deviations of triplicate incubations. An asterisk indicates no data.
In sediments, all 10 substrates, purified polysaccharides as well as algal extracts, were hydrolyzed. Hydrolysis rates ranged from ca. 8,700 nmol monomer liter−1 h−1 (pullulan, subsurface) to ca. 280 nmol monomer liter−1 h−1 (chondroitin, subsurface) (Fig. 1B). Note that the hydrolysis of arabinogalactan, isochrysis extract, and wakame extract was not measured in subsurface sediments due to experimental error. Rates measured in surface and subsurface sediments differed somewhat; in particular, the hydrolysis rates of pullulan, laminarin, xylan, and alginic acid were considerably higher (in the case of pullulan, by almost a factor of 5) in subsurface sediments than in surface sediments. Hydrolysis of chondroitin was more rapid in surface than subsurface sediments, and fucoidan hydrolysis was virtually identical at both depths. The order of hydrolysis rates therefore differed between surface and subsurface sediments, with rates being as follows: alginic acid and wakame extract > isochrysis extract > laminarin, pullulan > xylan > chondroitin, arabinogalactan, spirulina extract, fucoidan for surface sediments, and pullulan > alginic acid > laminarin > xylan > fucoidan and spirulina extract > chondroitin for subsurface sediments.
The most striking observation was the contrasting hydrolysis rates and patterns between the seawater and sedimentary communities, irrespective of depth. On a per volume basis, sediment rates were up to 3 orders of magnitude higher than water column rates. Three of the 10 substrates (pullulan, arabinogalactan, and isochrysis) were not hydrolyzed in the water column, but all substrates were hydrolyzed in sediments. Patterns of hydrolysis were also very different, with chondroitin hydrolysis being the most rapid process in seawater and among the least rapid in sediments, while the opposite pattern was observed for alginic acid hydrolysis, being among the most rapid in sediments and among the least rapid (of hydrolyzed substrates) in seawater.
Overview of bacterial community composition.
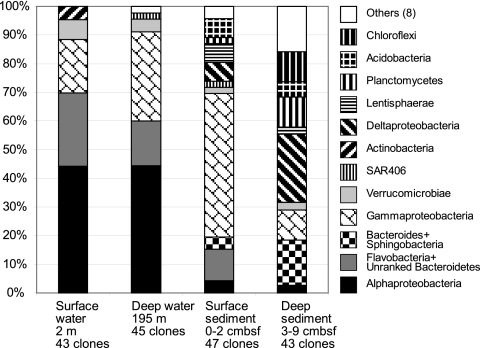
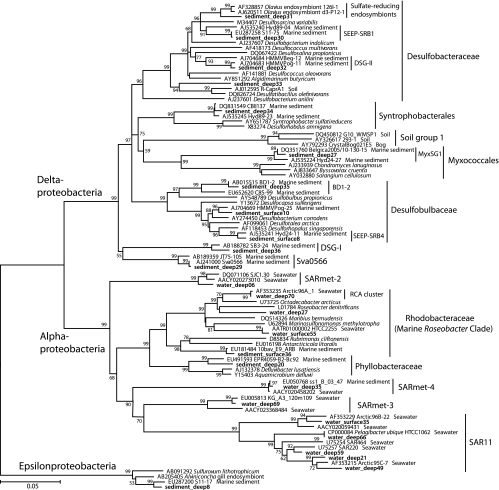
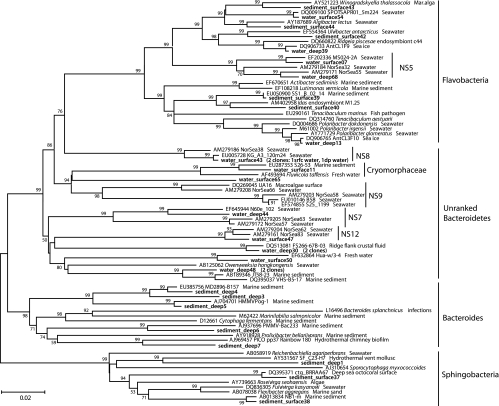
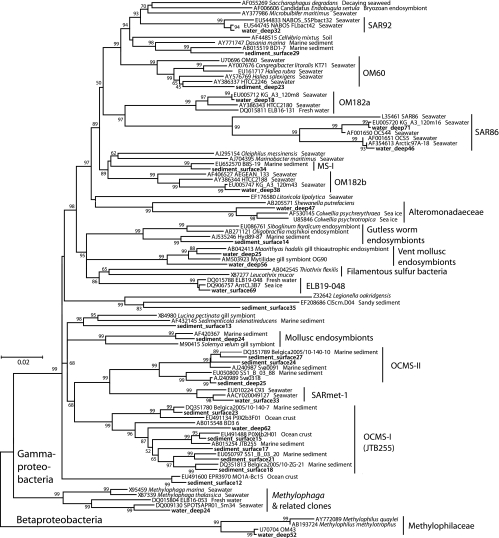
The bacterial communities of the Svalbard water column and sediment samples diverged strongly in composition on phylogenetically broad levels of bacterial phyla and classes (Fig. 2). These changes are primarily manifested in the disappearance of subphylum-level groups (Alphaproteobacteria) in sediments, in a changing taxonomic profile within phyla and subphyla (Bacteroides and Gammaproteobacteria), and in the emergence of bacteria typical of sediment, especially the Deltaproteobacteria (Fig. 2). The 16S rRNA gene clone libraries of the water column samples were dominated by Alphaproteobacteria (44%); their dominant components are the phylogenetically and ecophysiologically distinct pelagic SAR11 and marine Roseobacter lineages (Fig. 3). The SAR11 lineage consists of ultraoligotrophic, globally dominant pelagic bacteria that utilize marine dissolved organic matter (52, 61); their growth depends on exogenous organosulfur compounds (dimethylsulfoniopropionate [DMSP] and methionine) synthesized by other bacteria (71). The marine Roseobacter lineage consists of heterotrophic and photoheterotrophic bacteria that are abundant in coastal waters and often assimilate organosulfur components (28); for example, they respond positively to DMSP-producing algal blooms (37) and they are the most frequently recovered Alphaproteobacteria from metagenomes of DMSP-assimilating laboratory enrichments (53). The next most abundant lineages in the water column are Flavobacteria and unranked Bacteroidetes lineages within the Bacteroidetes phylum (Fig. 4), which accounted for 16 to 26% of all clones. Mostly uncultured marine gammaproteobacterial lineages (Fig. 5), for example, the globally occurring pelagic SAR86 cluster (47), account for 19 to 31%. Detailed phylogenetic analysis shows that the Svalbard clones are most closely related to clones from previous surveys of seawater, polar sea ice, and marine sediment communities (Fig. 3 to 5). Less frequently recovered water column groups include the Verrucomicrobia, Actinobacteria, Betaproteobacteria, and the SAR406 cluster (see Fig. S1 in the supplemental material). The microbial community composition changes drastically in the surficial sediment (0 to 2 cm), where the Alphaproteobacteria decreased to ca. 4% of the bacterial clone library (Fig. 2) and the Gammaproteobacteria dominate (49%). Further shifts between water column and sediment occur within the class-level structure of the Gammaproteobacteria; in the sediment, the oligotrophic, aerobic heterotrophic strains of the OM60 clade (29) and the not-yet-cultured OCMS-II group (Fig. 5) take the place of the pelagic SAR86 cluster (54) (see Fig. S2 and text in the supplemental material). The taxonomic composition within the Bacteroides phylum changes toward a mixed assemblage of Flavobacteria, unranked Bacteroidetes, and Sphingobacteria (Fig. 4) that accounts for 15% of the clone library in surficial sediment. Additional groups such as the Deltaproteobacteria, Acidobacteria, and Lentisphaerae account for ca. 7 to 9% each. In the deeper sediment layer (3 to 9 cm), the Deltaproteobacteria become the largest group in the 16S rRNA gene clone library (20%), while the Planktomycetes and Chloroflexi constitute 9% each. The Gammaproteobacteria are reduced to ca. 9% of the deep sediment clone library, with the Bacteroidetes phylum now being represented by Sphingobacteria and Bacteroides (∼14%). Numerous smaller bacterial groups constitute the remainder of both sediment clone libraries and account for ca. 40% of the clones in the deeper (3 to 9 cm) clone library (Fig. 2); a more detailed discussion of these rarely retrieved phylotypes, as well as additional detail on the major bacterial groups recovered, is included as text in the supplemental material. The greater diversity of the sediment community is reflected not only in the number of phylum-level phylogenetic lineages but also in rarefaction analysis of OTUs. The sediment samples showed a steeper increase of phylotype diversity, as more distinct phylotypes are recovered in the sediment samples than the water column samples (see Fig. S3 in the supplemental material).
FIG. 2.
Percent representation of bacterial phylum- and subphylum-level groups in 16S rRNA gene clone libraries from water column and sediment samples in Smeerenburg Fjord, Svalbard. From left to right, surface water, deep water, surface sediment, and deep sediment community. cmbsf, centimeters below the surface.
FIG. 3.
Neighbor-joining phylogeny of Smeerenburg Fjord alpha- and deltaproteobacterial phylotypes, based on an ∼1,200-bp alignment of bacterial 16S rRNA sequences. The Svalbard phylotypes are labeled with the habitat indicator (water_surface, water_deep, sediment_surface, sediment_deep), followed by the clone number, and are highlighted in boldface.
FIG. 4.
Neighbor-joining phylogeny of Smeerenburg Fjord Bacteroides phylotypes, based on an ∼1,200-bp alignment of bacterial 16S rRNA sequences. The Svalbard phylotypes are labeled with the habitat indicator (water_surface, water_deep, sediment_surface, sediment_deep), followed by the clone number, and are highlighted in boldface.
FIG. 5.
Neighbor-joining phylogeny of Smeerenburg Fjord beta- and gammaproteobacterial phylotypes, based on an ∼1,200-bp alignment of bacterial 16S rRNA sequences. The Svalbard phylotypes are labeled with the habitat indicator (water_surface, water_deep, sediment_surface, sediment_deep), followed by the clone number, and are highlighted in boldface.
DISCUSSION
Microbial communities and activities in water column and sediment.
Understanding the relationships between microbial community composition and function is a major challenge in studying heterotrophic carbon cycling in marine systems. Metagenomic approaches to decipher the metabolic potential of specific organisms as well as entire communities have provided new insights into community potential (31). The expression of the microbial community's enzymatic repertoire in situ (23, 76), however, is most directly assessed by measuring specific enzyme activities in seawater and sediments. Since standard approaches to measure microbial enzyme activities in aquatic systems (i.e., fluorescently labeled low-molecular-weight substrate proxies [43]) provide limited insight into the structural specificities of extracellular enzymes, the extent to which heterotrophic communities vary in their abilities to produce a broad range of extracellular enzymes is, for the most part, unknown. The diverse range of components and structures used as substrates in this study facilitates a detailed investigation of potential enzyme activity and structural specificity that cannot be achieved with commercially available substrate proxies.
The differences in the abilities of water column and sedimentary microbial communities to initiate the degradation of high-molecular-weight organic matter are striking and are reproducible for samples collected in different years (11). These differences cannot be attributed to differences in microbial cell numbers: scaling the sediment and seawater hydrolysis rates by a factor that would account for the 2- to 3-order-of-magnitude difference in cell counts would still not produce similar patterns of hydrolysis (Fig. 1A and B). The extent to which these robust differences may be aligned with differences in community composition was, prior to this study, a completely open question, given the dearth of comparative studies of seawater and sedimentary microbial communities from the same locations, particularly at high latitude.
We hypothesize that the differences in enzyme spectra and activity are due to distinctly different sedimentary and seawater microbial communities, as indicated by the 16S rRNA gene sequencing survey. Our gene sequencing results are congruent with those of other studies of either water column or sediment communities in high latitudes that often yield the most closely related phylotypes in 16S rRNA gene phylogenies (Fig. 3 to 5); joint analyses of polar water column and sediment, as in this study, are not available. The dominant water column communities in high latitudes consisted mostly of Alphaproteobacteria, Gammaproteobacteria, and Bacteroidetes (1, 21, 35, 67, 80), in contrast to Gammaproteobacteria, Deltaproteobacteria, Bacteroidetes, and Planktomycetes, which were the most frequently recovered sediment lineages (8, 26, 27, 59, 62, 63, 64).
Interestingly, one of the key results of this clone library analysis, the complex composition of the sediment community and the high abundance of Deltaproteobacteria in the sediments, is validated by a quantitative microbial community analysis in the upper 20 cm of Smeerenburg Fjord sediment by rRNA blotting (63, 64). The Deltaproteobacteria accounted for ca. 25% of the bacterial 16S rRNA pool in the blotting analysis, compared to 8 to 20% of the 16S rRNA clones in the 0- to 3-cm and 3- to 9-cm depths. The Gammaproteobacteria accounted for ca. 15% of the total 16S rRNA pool in the blotting results, compared to 10 to 50% in the 16S rRNA clone libraries in the 0- to 3-cm and 3- to 9-cm depths. Bacteroidetes constituted ca. 4% of the rRNA blotting signal, compared to ca. 15% of the sediment clone libraries. More than 50% of the bacterial rRNA remained unaccounted for in the blotting analysis, since the 16S rRNA probes for slot blot analysis did not cover the highly diverse bacterial lineages that occur in the sediments (64).
Could the highly diversified benthic microbial lineages (Fig. 2; see Fig. S1 in the supplemental material) represent the principal reason for the expanded spectrum of hydrolytic activities in the sediment compared to the more restricted substrate spectrum measured in the water column? For example, the Chloroflexi are likely to contribute to the diversified repertoire of polysaccharide hydrolases in the Svalbard sediments. The Chloroflexi are a dominant microbial community component of deep marine sediments (69) and include obligate anaerobic dehalogenators (Dehalobium chlorocoercia and Dehalococcoides ethenogenes) and diverse thermophilic, mesophilic, and filamentous sugar and amino acid fermenters within the recently defined class Anaerolineae that are often isolated from anaerobic digesters in wastewater treatment plants or from industrial fermenting bioreactors (81). Some recently isolated strains within the Anaerolineae are able to grow slowly on xylan and starch (81). In molecular assays combining microsphere adhesion to cells with enzyme-labeled fluorescence, several uncultured Chloroflexi populations were shown to express chitinase, esterase, galactosidase, and glucuronidase activity, although this was under aerobic conditions (47). Similar considerations might apply to the Planktomycetes. The Planktomycetes were previously detected to be a significant minority in Svalbard sediments (64) and include cultured isolates that specialize in utilization of sulfated organic compounds (36). In contrast, the Deltaproteobacteria are unlikely candidates for the hydrolysis of complex carbohydrates in the sediment; they are mostly represented by clones that are affiliated with sulfate-reducing bacteria of the families Desulfobacteraceae and Desulfobulbaceae (Fig. 3). The cultured members of these families predominantly oxidize the fermentation products acetate and other low-molecular-weight organic acids, such as propionate to acetate and CO2; their substrate range does not include high-molecular-weight carbohydrates (79).
The problem of linking identity and function.
Although differences in community composition are reflected in or contribute to differences in enzymatic hydrolysis patterns, linking specific organisms and activities for now has the character of a working hypothesis with many caveats: the high number of bacterial phylum- and subphylum-level lineages in the water column and sediment, particularly the high phylum-level diversity of the sediment bacterial populations (Fig. 2); the possibility of overlooking crucial microbial groups due to the methodological limitations of PCR and clone libraries; the nonquantitative character of the clone library composition; and the problems of inferring physiological capabilities from sequence data. Assigning activities of specific enzymes to specific organisms within a complex community is beyond current technical capabilities, due to inadequate coverage of natural microbial communities in databases, as well as the structural/sequence variability of enzymes belonging to the same families and carrying out the same functions. Recognizing these difficulties, we limit this discussion of potential links between hydrolytic and phylogenetic spectra to a single example, the Bacteroidetes phylum, that should be understood to be a working hypothesis for future research.
The Bacteroidetes persist in sediment and the water column while undergoing intraphylum community changes. Within the Bacteroidetes, members of the class Flavobacteria (following the taxonomic outline of Bergey's Manual, 2nd ed. [32a]) account for ca. 15 to 25% of the bacterial clone libraries in the water column; the Flavobacteria are reduced to ca. 10% in the surficial sediment and are replaced in the deep sediment by members of the class Bacteroides and class Sphingobacteria within the same phylum (Fig. 4). Physiologically, this community shift might be best explained in terms of aerobic versus anaerobic metabolism; it represents a change from phylotypes affiliated with aerobic Flavobacteria and taxonomically yet unranked Bacteroidetes in the water column, for example, Bacteroidetes clades NS 7, 8, 9, and 12 from the North Sea water column (4), toward a mixture of aerobic Flavobacteria- and Sphingobacteria-related phylotypes at the sediment surface, followed by mostly anaerobic Bacteroidetes and deeply branching sphingobacterial lineages in the deeper anoxic sediment (Fig. 4; see text in the supplemental material). This habitat specificity appears to be a characteristic feature of the phylum Bacteroidetes. A combined analysis of Bacteroidetes sequences in public databases and from an extensive North Sea water data set demonstrated that most sequence-based clades within the Bacteroidetes were associated with a single habitat type and could be identified to be either marine, freshwater, or sediment derived; only a minority (15%) of Bacteroidetes clades occurred in at least two habitat types (4).
The frequent recovery of flavobacterial sequences from the water column and surficial sediments is most likely related to the presence of phytoplankton and fresh phytodetritus in the water column and on the sediment surface. Numerous studies have shown that the Flavobacteria respond strongly to phytoplankton availability as either natural blooms, experimental blooms, or enrichment experiments on phytodetritus (see references 2 and 58 and references therein). Recently, stable isotope probing experiments with surficial sediments have demonstrated that phytodetritus is quickly assimilated by members of the Bacteroidetes (33). The hydrolysis rates of alginic acid, isochrysis, and wakame extracts at the sediment surface are higher than those of other substrates (Fig. 1); this preference for complex algal extracts might be a consequence of local Bacteroidetes accumulation and enrichment on the sediment surface.
Within the anaerobic sediment, the microbial community shift toward anaerobic Bacteroides reflects a turn to anaerobic, fermentative degradation of carbohydrates, since aerobic degradation is no longer possible. Cultured free-living marine Bacteroides often have psychrotolerant temperature ranges and ferment a wide range of sugars anaerobically (i.e., 42). Bacteroides are also widespread as symbiotic, fermentative degraders of complex plant polymers and marine macroalgal polysaccharides within the digestive tract of metazoans (51). These trends from cultivation studies and biotic surveys are confirmed by more recent whole-genome analyses; for example, the genome of Gramella forsetii contains an abundance of glycosyl hydrolases with close sequence similarities to arabinogalactans (23). Such obligately or facultatively anaerobic Bacteroides species could represent potential contributors to the high pullulan and alginate hydrolysis rates in the anaerobic sediment (Fig. 1); more generally, they could be one of the key bacterial populations that contribute to the generally higher diversity of polysaccharide-hydrolyzing enzymes in the sediment.
Ideally, linkages between enzyme activities and phylogenetic identity should be tested using cultured isolates. Marine Gammaproteobacteria have been cultured from Svalbard Fjord sediments (68) and sea ice (38); these mostly comprise members of the genera Pseudomonas, Pseudoalteromonas, Colwellia, Erythrobacter, Psychrobacter, and Shewanella. Some isolates from these genera hydrolyze starch and Tween 60 (68) and pullulan and alginate (38). The elevated hydrolysis rates of pullulan and alginic acid in sediment compared to the water column (Fig. 1) would be compatible with the presence of these polysaccharide-hydrolyzing Gammaproteobacteria in the surficial sediment. Further, stable carbon isotope probing experiments with sandy sediments of the Gulf of Mexico have demonstrated that the gammaproteobacterial genera Pseudoalteromonas and Vibrio, in addition to members of the Bacteroidetes and Firmicutes, assimilate algal extracts quickly; these gammaproteobacterial genera have a broad range of hydrolases that can be expressed in immediate response to phytoplankton detritus availability (33). However, the overlap of Gammaproteobacteria from our Svalbard clone libraries with these cultured strains is minimal and consists of a single Colwellia-related clone from bottom water (Fig. 5).
Enzyme activities in the context of community composition.
The clone library data clearly indicate differences in community composition, particularly between seawater and sediments, but also between surface and bottom water, as well as between surface and subsurface sediments. These community differences are reflected as well in functional differences between the spectrum of enzyme activities in seawater and sediments (Fig. 1). In surface and bottom water, rates as well as patterns of enzyme activities were very similar. These similarities may be due to functional overlap among the surface- and bottom-water communities, or common members of these communities may be responsible for specific activities. Moreover, since hydrolysis rates are measured with externally added substrates and incubations extended over time courses sufficiently long to allow microbial growth as well as enzyme induction, these measurements provide insight into the potential of a mixed microbial community to respond to substrate input gradually over time, rather than a snapshot view of enzyme activities at the time of sample collection.
Both the surface-water and bottom-water communities can thus respond to a similar extent to substrate addition, but they also share the same fundamental limitations in the spectrum of enzymes produced; some microbial key populations with specific enzymatic capabilities found in the sediments seem to be missing in the water column. Some soluble substrates that are readily hydrolyzed in sediments are microbially recalcitrant in surface and bottom waters, although their compositions on a bulk level are quite similar. For example, laminarin and pullulan are both linear glucose polysaccharides, and isochrysis extract is branched and primarily consists of glucose. Only laminarin, however, was readily hydrolyzed in surface and bottom waters as well as in sediments; pullulan and isochrysis extract were not hydrolyzed in seawater over the time course of incubation.
In sediments, patterns of enzymatic hydrolysis were similar in surface and deep layers, but rates of hydrolysis were typically higher in deep sediments than in surface sediments (Fig. 1). This difference was particularly pronounced for hydrolysis of pullulan (8,700 nmol monomer liter−1 h−1 in deep sediments, 1,960 nmol monomer liter−1 h−1 for surface sediments) but was also evident for alginic acid, laminarin, and xylan. The differences in hydrolysis rates between surface and deep sediments may be due to production of a greater number of enzymes or to enzymes with higher turnover numbers. Only for chondroitin was hydrolysis more rapid in surface than in deep sediments. Chondroitin may also constitute an exception because previous work has provided strong evidence that chondroitin hydrolysis is also induced in Svalbard sediments (10), and the time course of incubation of these sediments (60 h) may have been too short to allow full induction of activity (10).
The striking differences in patterns of hydrolysis between seawater and sediments is consistent with the results of previous investigations of enzyme activities in surface waters and surface sediment from Smeerenburg Fjord (11), with no hydrolysis of pullulan or arabinogalactan occurring in surface water, although these substrates were readily hydrolyzed in surface sediments. In the same study, hydrolysis of isochrysis extract in surface water was detected only after 70 days incubation, an indication that a numerically small and/or slow-growing fraction of the microbial community was responsible for hydrolysis of this substrate.
Outlook.
These data, to the best of our knowledge, are the first from a comparison of microbial community composition in seawater and in sediments from a high latitude and represent data from one of few comparisons of this type from any latitude. The distinct differences in community composition between seawater and sediments are reflected in the differences in community function. Microbial communities in Smeerenburg Fjord sediments may require a broader range of enzymatic capabilities (and/or may have the capability of retaining a genome with a broader range of capabilities) than their water column counterparts. Within the water column, compositional differences in communities were not reflected in differences in function, at least for the substrates/enzymes tested. We note that there is a dearth of data on the physical oceanography of Smeerenburg Fjord, so the extent to which surface and bottom waters remain stratified and/or represent different water masses is presently unknown. The relatively large temperature difference (2.2°C) between surface and bottom waters, however, suggests that fjord water masses are stratified, particularly given the presence of several large glaciers actively releasing icebergs into the fjord waters and thereby providing a source of fresher (less saline) surface water. Given the strong functional differences between the seawater and sedimentary microbial communities, efforts to understand microbial processing of carbon should include both compartments; future work should be aimed at pinning down who does what.
Supplementary Material
Acknowledgments
We thank the captain of R/V Farm and members of the Max Planck Institute's Svalbard 2007 scientific party for excellent seamanship and teamwork during the cruise, Drew Steen for help with seawater sampling and logistics, Sherif Ghobrial for assistance with sample processing, and Enten von Bad Iburg for facilitating the fieldwork.
Partial funding was provided by NSF (grant OCE-0848703 to C.A. and grant NSF-0527167 to A.T.). We are grateful for the generous support of fieldwork and logistics by the Max Planck Institute for Marine Microbiology (Bremen, Germany) that enabled us to carry out this study.
Footnotes
Published ahead of print on 21 January 2011.
Supplemental material for this article may be found at http://aem.asm.org/.
REFERENCES
- 1.Abell, G. C. J., and J. P. Bowman. 2005. Colonization and community dynamics of class Flavobacteria on diatom detritus in experimental mesocosms based on Southern Ocean seawater. FEMS Microbiol. Ecol. 53:379-391. [DOI] [PubMed] [Google Scholar]
- 2.Abell, G. C. J., and J. P. Bowman. 2005. Ecological and biogeographic relationships of class Flavobacteria in the Southern Ocean. FEMS Microbiol. Ecol. 51:265-277. [DOI] [PubMed] [Google Scholar]
- 3.Alderkamp, A.-C., M. van Rijssel, and H. Bolhuis. 2007. Characterization of marine bacteria and the activity of their enzyme systems involved in degradation of the algal storage glucan laminarin. FEMS Microbiol. Ecol. 59:108-117. [DOI] [PubMed] [Google Scholar]
- 4.Alonso, C., F. Warneke, R. Amann, and J. Pernthaler. 2007. High local and global diversity of Flavobacteria in marine plankton. Environ. Microbiol. 9:1253-1266. [DOI] [PubMed] [Google Scholar]
- 5.Araki, T., S. Hashikawa, and T. Morishita. 2000. Cloning, sequencing, and expression in Escherichia coli of the new gene encoding β-1,3-xylanase from a marine bacterium, Vibrio sp. strain XY-214. Appl. Environ. Microbiol. 66:1741-1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arnosti, C. 1995. Measurement of depth- and site-related differences in polysaccharide hydrolysis rates in marine sediments. Geochim. Cosmochim. Acta 59:4247-4257. [Google Scholar]
- 7.Arnosti, C. 1996. A new method for measuring polysaccharide hydrolysis rates in marine environments. Org. Geochem. 25:105-115. [Google Scholar]
- 8.Arnosti, C. 2000. Substrate specificity in polysaccharide hydrolysis: contrasts between bottom water and sediments. Limnol. Oceanogr. 45:1112-1119. [Google Scholar]
- 9.Arnosti, C. 2003. Fluorescent derivatization of polysaccharides and carbohydrate-containing biopolymers for measurement of enzyme activities in complex media. J. Chromatogr. B 793:181-191. [DOI] [PubMed] [Google Scholar]
- 10.Arnosti, C. 2004. Speed bumps and barricades in the carbon cycle: substrate structural effects on carbon cycling. Mar. Chem. 92:263-273. [Google Scholar]
- 11.Arnosti, C. 2008. Functional differences between arctic seawater and sedimentary microbial communities: contrasts in microbial hydrolysis of complex substrates. FEMS Microbiol. Ecol. 66:343-351. [DOI] [PubMed] [Google Scholar]
- 12.Arnosti, C., B. B. Jørgensen, J. Sagemann, and B. Thamdrup. 1998. Temperature dependence of microbial degradation of organic matter in marine sediments: polysaccharide hydrolysis, oxygen consumption, and sulfate reduction. Mar. Ecol. Prog. Ser. 159:59-70. [Google Scholar]
- 13.Arnosti, C. 2011. Microbial extracellular enzymes and the marine carbon cycle. Annu. Rev. Mar. Sci. 3:401-425. [DOI] [PubMed] [Google Scholar]
- 14.Arnosti, C., and M. Holmer. 1999. Carbohydrate dynamics and contributions to the carbon budget of an organic-rich coastal sediment. Geochim. Cosmochim. Acta 63:393-403. [Google Scholar]
- 15.Arnosti, C., N. Finke, O. Larsen, and S. Ghobrial. 2005. Anoxic carbon degradation in Arctic sediments: microbial transformations of complex substrates. Geochim. Cosmochim. Acta 69:2309-2320. [Google Scholar]
- 16.Arnosti, C., and B. B. Jørgensen. 2006. Organic carbon degradation in arctic marine sediments, Svalbard: a comparison of initial and terminal steps. Geomicrobiol. J. 23:551-563. [Google Scholar]
- 17.Arnosti, C., K. Ziervogel, L. Ocampo, and S. Ghobrial. 2009. Enzyme activities in shallow permeable sediments and the water column from the northeastern Gulf of Mexico. Estuar. Coastal Shelf Sci. 84:202-208. [Google Scholar]
- 18.Azam, F. 1998. Microbial control of oceanic carbon flux: the plot thickens. Science 280:694-696. [Google Scholar]
- 19.Baldwin, A. J., et al. 2005. Microbial diversity in a Pacific Ocean transect from the Arctic to Antarctic circles. Aquat. Microb. Ecol. 41:91-102. [Google Scholar]
- 20.Balter, F., J. Aristegui, J. M. Gasol, S. Hernandez-Leon, and G. J. Herndl. 2007. Strong coast-ocean and surface-depth gradients in prokaryotic assemblage structure and activity in a coastal transition zone region. Aquat. Microb. Ecol. 50:63-74. [Google Scholar]
- 21.Bano, N., and J. T. Hollibaugh. 2002. Phylogenetic composition of bacterioplankton assemblages from the Arctic Ocean. Appl. Environ. Microbiol. 68:505-518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bano, N., S. Ruffin, B. Ransom, and J. T. Hollibaugh. 2004. Phylogenetic composition of Arctic Ocean archaeal assemblages and comparison with Antarctic assemblages. Appl. Environ. Microbiol. 70:781-789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bauer, M., et al. 2006. Whole genome analysis of the marine Bacteroidetes ‘Gramella forsetii’ reveals adaptations to degradation of polymeric organic matter. Environ. Microbiol. 8:2201-2213. [DOI] [PubMed] [Google Scholar]
- 24.Benner, R., J. D. Pakulski, M. McCarthy, J. I. Hedges, and P. G. Hatcher. 1992. Bulk chemical characteristics of dissolved organic matter in the ocean. Science 255:1561-1564. [DOI] [PubMed] [Google Scholar]
- 25.Bibel, M., C. Brettl, U. Gosslar, G. Krieghäuser, and W. Libl. 1998. Isolation and analysis of genes for amylolytic enzymes of the hyperthermophilic bacterium Thermotoga maritima. FEMS Microbiol. Lett. 158:9-15. [DOI] [PubMed] [Google Scholar]
- 26.Bowman, J. P., and R. D. McCuaig. 2003. Biodiversity, community structure shifts, and biogeography of prokaryotes within Antarctic continental shelf sediment. Appl. Environ. Microbiol. 69:2463-2483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bowman, J. P., S. A. McCammon, J. A. E. Gibson, L. Robertson, and P. D. Nichols. 2003. Prokaryotic metabolic activity and community structure in Antarctic continental shelf sediments. Appl. Environ. Microbiol. 69:2448-2462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Buchan, A., J. M. González, and A. M. Moran. 2005. Overview of the marine Roseobacter lineage. Appl. Environ. Microbiol. 71:5665-5677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cho, J.-C., and S. J. Giovannoni. 2004. Cultivation and growth characteristics of a diverse group of oligotrophic marine Gammaproteobacteria. Appl. Environ. Microbiol. 70:432-440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cowie, G. L., and J. I. Hedges. 1984. Carbohydrates sources in a coastal marine environment. Geochim. Cosmochim. Acta 48:2075-2087. [Google Scholar]
- 31.DeLong, E. 2009. The microbial ocean from genomes to biomes. Nature 459:200-206. [DOI] [PubMed] [Google Scholar]
- 32.Fuhrman, J. A., et al. 2008. A latitudinal diversity gradient in planktonic marine bacteria. Proc. Natl. Acad. Sci. U. S. A. 105:7774-7778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32a.Garrity, G. M., and J. G. Holt. 2001. The road map to the manual, p. 119-166. In D. R. Boone and R. W. Castenholz (ed.), Bergey's manual of systematic bacteriology, 2nd ed., vol. 1. Springer-Verlag, New York, NY.
- 33.Gihring, T. M., M. Humphrys, H. J. Mills, M. Huettel, and J. E. Kostka. 2009. Identification of phytodetritus-degrading microbial communities in sublitoral Gulf of Mexico sands. Limnol. Oceanogr. 54:1073-1083. [Google Scholar]
- 34.Glabe, C. G., P. K. Harty, and S. D. Rosen. 1983. Preparation and properties of fluorescent polysaccharides. Anal. Biochem. 130:287-294. [DOI] [PubMed] [Google Scholar]
- 35.Glöckner, F. O., B. M. Fuchs, and R. Amann. 1999. Bacterioplankton comparisons of lakes and oceans: a first comparison based on fluorescence in situ hybridization. Appl. Environ. Microbiol. 65:3721-3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Glöckner, F. O., et al. 2003. Complete genome sequence of the marine planctomycete Pirellula sp. strain 1. Proc. Natl. Acad. Sci. U. S. A. 100:8298-8303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.González, J. M., et al. 2005. Bacterial community structure associated with a dimethyl-sulfoniopropionate-producing North Atlantic algal bloom. Appl. Environ. Microbiol. 66:4237-4246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Groudieva, T., et al. 2004. Diversity and cold-active hydrolytic enzymes of culturable bacteria associated with Arctic sea ice, Svalbard. Extremophiles 8:475-488. [DOI] [PubMed] [Google Scholar]
- 39.Hedges, J. I. 1992. Global biogeochemical cycles: progress and problems. Mar. Chem. 39:67-93. [Google Scholar]
- 40.Hedges, J. I., et al. 2000. The molecularly-uncharacterized component (MUC) of nonliving organic matter in natural environments. Org. Geochem. 31:945-958. [Google Scholar]
- 41.Hewson, I., G. A. Vargo, and J. A. Fuhrman. 2003. Bacterial diversity in shallow oligotrophic marine benthos and overlying waters: effects of virus infection, containment, and nutrient enrichment. Microb. Ecol. 46:322-336. [DOI] [PubMed] [Google Scholar]
- 42.Holmes, D. E., K. P. Nevin, T. L. Woodard, A. D. Peacock, and D. R. Lovley. 2007. Prolixibacter bellariivorans gen. nov., sp. nov., a sugar-fermenting, psychrotolerant anaerobe of the phylum Bacteroidetes, isolated from a marine-sediment fuel cell. Int. J. Syst. Evol. Microbiol. 57:701-707. [DOI] [PubMed] [Google Scholar]
- 43.Hoppe, H.-G. 1983. Significance of exoenzymatic activities in the ecology of brackish water: measurements by means of methylumbelliferyl-substrates. Mar. Ecol. Prog. Ser. 11:299-308. [Google Scholar]
- 44.Huber, T., G. Faulkner, and P. Hugenholtz. 2004. Bellerophon: a program to detect chimeric sequences in multiple sequence alignments. Bioinformatics 20:2317-2319. [DOI] [PubMed] [Google Scholar]
- 45.Kirchman, D. L., R. R. Malmstrom, and M. T. Cottrell. 2005. Control of bacterial growth by temperature and organic matter in the western Arctic. Deep Sea Res. II 52:3386-3395. [Google Scholar]
- 46.Knoblauch, C., K. Sahm, and B. B. Jørgensen. 1999. Psychrophilic sulfate-reducing bacteria isolated from permanently cold Arctic marine sediments: description of Desulfofrigus oceanense gen. nov., sp nov., Desulfofrigus fragile sp nov., Desulfofaba gelida gen. nov., sp. nov., Desulfotalea psychrophila gen. nov., sp. nov., and Desulfotalea arctica sp. nov. Int. J. Syst. Bacteriol. 49:1631-1643. [DOI] [PubMed] [Google Scholar]
- 47.Kragelund, C., et al. 2007. Identity, abundance and ecosphysiology of filamentous Chloroflexi species present in activated sludge treatment plants. FEMS Microbiol. Ecol. 59:671-682. [DOI] [PubMed] [Google Scholar]
- 48.Kumar, S., J. Dudley, M. Nei, and K. Tamura. 2008. MEGA: a biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief. Bioinform. 9:299-306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lee, C., S. G. Wakeham, and C. Arnosti. 2004. Particulate organic matter in the sea: the composition conundrum. Ambio 33:559-568. [DOI] [PubMed] [Google Scholar]
- 50.Meiners, K., C. Krembs, and R. Gradinger. 2008. Exopolymer particles: microbial hotspots of enhanced bacterial activity in Arctic fast ice (Chukchi Sea). Aquat. Microb. Ecol. 52:105-207. [Google Scholar]
- 51.Michel, C., and G. T. Macfarlane. 1996. Digestive fates of soluble polysaccharides from marine macroalgae: involvement of the colonic microflora and physiological consequences for the host. J. Appl. Biol. 80:349-369. [DOI] [PubMed] [Google Scholar]
- 52.Morris, R. M., et al. 2002. SAR11 clade dominates ocean surface bacterioplankton communities. Nature 420:806-810. [DOI] [PubMed] [Google Scholar]
- 53.Mou, X., S. Sun, R. A. Edwards, R. E. Hodson, and M. A. Moran. 2008. Bacterial carbon processing by generalist species in the coastal ocean. Nature 451:708-711. [DOI] [PubMed] [Google Scholar]
- 54.Mullins, T. D., T. B. Britschgi, R. L. Krest, and S. J. Giovannoni. 1995. Genetic comparisons reveal the same unknown bacterial lineages in Atlantic and Pacific bacterioplankton communities. Limnol. Oceanogr. 40:148-158. [Google Scholar]
- 55.Nei, M., and S. Kumar. 2000. Molecular evolution and phylogenetics. Oxford University Press, New York, NY.
- 56.Painter, T. J. 1983. Algal polysaccharides, p. 195-285. In G. O. Aspinall (ed.), The polysaccharides, vol. 2. Academic Press, New York, NY. [Google Scholar]
- 57.Pantoja, S., C. Lee, and J. F. Marecek. 1997. Hydrolysis of peptides in seawater and sediments. Mar. Chem. 57:25-40. [Google Scholar]
- 58.Pinhassi, J., et al. 2004. Changes in bacterioplankton composition under different phytoplankton regimens. Appl. Environ. Microbiol. 70:6753-6766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Powell, S., J. P. Bowman, I. Snape, and J. S. Stark. 2003. Microbial community variation in pristine and polluted nearshore Antarctic sediments. FEMS Microbiol. Ecol. 45:135-145. [DOI] [PubMed] [Google Scholar]
- 60.Pruesse, E., et al. 2007. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 35:7188-7196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rappé, M. S., S. A. Connon, K. L. Vergin, and S. J. Giovannoni. 2002. Cultivation of the ubiquitous SAR11 marine bacterioplankton clade. Nature 418:630-633. [DOI] [PubMed] [Google Scholar]
- 62.Ravenschlag, K., K. Sahm, J. Pernthaler, and R. Amann. 1999. High bacterial diversity in permanently cold marine sediments. Appl. Environ. Microbiol. 65:3982-3989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ravenschlag, K., K. Sahm, C. Knoblauch, B. B. Jørgensen, and R. Amann. 2000. Community structure, cellular rRNA content, and activity of sulfate-reducing bacteria in marine Arctic sediments. Appl. Environ. Microbiol. 66:3592-3602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ravenschlag, K., K. Sahm, and R. Amann. 2001. Quantitative molecular analysis of the microbial community in marine arctic sediments. Appl. Environ. Microbiol. 67:387-395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Saitou, N., and M. Nei. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4:406-425. [DOI] [PubMed] [Google Scholar]
- 66.Schloss, P. D., and J. Handelsman. 2005. Introducing DOTUR, a computer program for defining operational taxonomic units and estimating species richness. Appl. Environ. Microbiol. 71:1501-1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Simon, M., F. O. Glöckner, and R. Amann. 1999. Different community structure and temperature optima of heterotrophic picoplankton in various regions of the Southern Ocean. Aquat. Microb. Ecol. 18:275-284. [Google Scholar]
- 68.Srinivas, T. N. R., et al. 2009. Bacterial diversity and bioprospecting for cold-active lipases, amylases and proteases, from culturable bacteria of Kongsfjorden and Ny-Ålesund, Svalbard, Arctic. Curr. Microbiol. 59:537-547. [DOI] [PubMed] [Google Scholar]
- 69.Teske, A. 2006. Microbial communities of deep marine subsurface sediments: molecular and cultivation surveys. Geomicrobiol. J. 23:357-368. [Google Scholar]
- 70.Teske, A., et al. 2002. Microbial diversity of hydrothermal sediments in the Guaymas Basin: evidence for anaerobic methanotrophic communities. Appl. Environ. Microbiol. 68:1994-2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tripp, H. J., et al. 2008. SAR11 marine bacteria require exogenous reduced sulphur for growth. Nature 452:741-744. [DOI] [PubMed] [Google Scholar]
- 72.Valdemarsen, T., and E. Kristensen. 2010. Degradation of dissolved organic monomers and short-chain fatty acids in sandy marine sediments via fermentation and sulfate reduction. Geochim. Cosmochim. Acta 74:1593-1605. [Google Scholar]
- 73.Vandieken, V., N. Finke, and B. B. Jørgensen. 2006. Pathways of carbon oxidation in an Arctic fjord sediment and isolation of psychrophilic and psychrotolerant Fe(III)-reducing bacteria. Mar. Ecol. Prog. Ser. 322:29-41. [Google Scholar]
- 74.Wakeham, S. G., C. Lee, J. I. Hedges, P. J. Pernes, and M. L. Peterson. 1997. Molecular indicators of diagenetic status in marine organic matter. Geochim. Cosmochim. Acta 61:5363-5369. [Google Scholar]
- 75.Warren, R. A. J. 1996. Microbial hydrolysis of polysaccharides. Annu. Rev. Microbiol. 50:183-212. [DOI] [PubMed] [Google Scholar]
- 76.Weiner, R. M., et al. 2008. Complete genome sequence of the complex carbohydrate-degrading marine bacterium, Saccharophagus degradans strain 2-40T. PLoS Genet. 5:e1000087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Westrich, J. T., and R. A. Berner. 1984. The role of sedimentary organic matter in bacterial sulfate reduction: the G model tested. Limnol. Oceanogr. 29:236-249. [Google Scholar]
- 78.Wheeler, P. A., et al. 1996. Active cycling of organic carbon in the central Arctic Ocean. Nature 380:697-699. [Google Scholar]
- 79.Widdel, F., and F. Bak. 1992. Gram-negative mesophilic sulfate-reducing bacteria, p. 3352-3378. In A. Balows, H. G. Trüper, M. Dworkin, W. Harder, and K.-H. Schleifer (ed.), The prokaryotes, 2nd ed. Springer-Verlag, New York, NY.
- 80.Yakimov, M. M., et al. 2004. Crude oil-induced structural shift of coastal bacterial communities of rod bay (Terra Nova Bay, Ross Sea, Antarctica) and characterization of cultured cold-adapted hydrocarbonoclastic bacteria. FEMS Microbiol. Ecol. 49:419-432. [DOI] [PubMed] [Google Scholar]
- 81.Yamada, T., et al. 2006. Anaerolinea thermolimosa sp. nov., Levilinea saccharolytica gen. nov., sp. nov. and Leptolinea tardivitalis gen. nov., sp. nov., novel filamentous anaerobes, and description of the new classes Anaerolineae classis nov. and Caldilineae classis nov. in the bacterial phylum Chloroflexi. Int. J. Syst. Evol. Microbiol. 56:1331-1340. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.