Abstract
Background
Amongst the Plasmodium species in humans, only P. vivax and P. ovale produce latent hepatic stages called hypnozoites, which are responsible for malaria episodes long after a mosquito bite. Relapses contribute to increased morbidity, and complicate malaria elimination programs. A single drug effective against hypnozoites, primaquine, is available, but its deployment is curtailed by its haemolytic potential in glucose-6-phosphate dehydrogenase deficient persons. Novel compounds are thus urgently needed to replace primaquine. Discovery of compounds active against hypnozoites is restricted to the in vivo P. cynomolgi-rhesus monkey model. Slow growing hepatic parasites reminiscent of hypnozoites had been noted in cultured P. vivax-infected hepatoma cells, but similar forms are also observed in vitro by other species including P. falciparum that do not produce hypnozoites.
Methodology
P. falciparum or P. cynomolgi sporozoites were used to infect human or Macaca fascicularis primary hepatocytes, respectively. The susceptibility of the slow and normally growing hepatic forms obtained in vitro to three antimalarial drugs, one active against hepatic forms including hypnozoites and two only against the growing forms, was measured.
Results
The non-dividing slow growing P. cynomolgi hepatic forms, observed in vitro in primary hepatocytes from the natural host Macaca fascicularis, can be distinguished from similar forms seen in P. falciparum-infected human primary hepatocytes by the differential action of selected anti-malarial drugs. Whereas atovaquone and pyrimethamine are active on all the dividing hepatic forms observed, the P. cynomolgi slow growing forms are highly resistant to treatment by these drugs, but remain susceptible to primaquine.
Conclusion
Resistance of the non-dividing P. cynomolgi forms to atovaquone and pyrimethamine, which do not prevent relapses, strongly suggests that these slow growing forms are hypnozoites. This represents a first step towards the development of a practical medium-throughput in vitro screening assay for novel hypnozoiticidal drugs.
Introduction
In mammalian hosts, malaria infection is initiated by the inoculation of Plasmodium sporozoites by an infected anopheline mosquito. These parasite forms then make their way to the liver where they specifically invade hepatocytes, the host cells permissive to full maturation leading to the production of merozoites that initiate the erythrocytic cycle. In most Plasmodium species, invasion of the hepatocyte by the sporozoite proceeds seamlessly to the development of the hepatic parasite culminating in the release of merozoites into the blood stream. The minimum time between the infective bite and the blood infection varies with the parasite species (2 to 15 days). However, in five parasite species, three in southeast Asian monkeys (P. cynomolgi, P. fieldi and P. simiovale) and two in humans (P. vivax and P. ovale), a subset of the sporozoites that have invaded the hepatocyte do not develop beyond the early hepatic trophozoite stage, but remain in a state of latency in which they can persist for many years [1], [2]. These forms, called hypnozoites, resume their development to generate infective merozoites many weeks, months or years later, though the timing and trigger for this “re-activation” remains mysterious. The hypnozoites probably provide a significant evolutionary advantage, particularly in temperate climates, as they effectively extend the duration of infection and, thus the opportunities to transmit successfully via mosquitoes to new hosts.
The demonstration of hypnozoites in P. cynomolgi-infected rhesus macaques in the early 1980's provided an explanation for the relapses observed during infections with these five parasite species [3]. A relapse is a blood stage episode that can occur after full clearance of parasites from the erythrocytic parasites (naturally or via treatment) from a primary or a recrudescent episode. In P. vivax, relapses are often observed regularly over many months and even years after the infectious mosquito bite. A similar pattern is also observed for P. ovale. Moreover, some temperate P. vivax strains produce sporozoites that nearly all transform into hypnozoites such that the primary blood infection is only observed many months after the infective mosquito bite, when some of the hypnozoite reactivate [1], [2]. Given that P. vivax is the most widely distributed of the four species of malaria parasites specific to humans and the most prevalent, except for sub-Saharan Africa where P. falciparum dominates and P. ovale infections are common [4], the occurrence of hypnozoites/relapses adds to their morbidity burden and makes these species especially difficult to eradicate.
Only one antimalarial drug capable of eliminating all liver stages including hypnozoites, the 8-aminoquinoline primaquine, is currently available [5], [6]. Its short half-life in the serum necessitates a lengthy treatment regimen that is not conducive to good adherence. Another recently developed 8-aminoquinoline with a longer half-life, tafenoquine, is equally effective against hypnozoites. However, the major shortcoming of primaquine and tafenoquine is that administration to persons with glucose-6-phosphate dehydrogenase deficiency (G6PD) is associated with a high risk of severe haemolytic anaemia. This precludes their widespread use because carriage of this genetic trait is common in the majority of the populations in P. vivax and P. ovale endemic areas.
The revival of eradication as the ultimate goal of malaria control programmes and the rediscovery that the clinical and socio-economic burdens of vivax malaria rival those of falciparum malaria, makes it imperative to develop novel compounds able to eliminate hypnozoites. Such a drug discovery programme cannot be currently envisaged simply because the only model available to screen for activity against hypnozoites is the P. cynomolgi sporozoite infection in rhesus monkeys. This host-parasite combination, whose development was initiated in 1944, remains the acknowledged biological and chemotherapeutic model for P. vivax infections, including erythrocytic and hepatic stages [7], [8], [9], [10]. However, an in vitro model is necessary in order to screen large numbers of new compounds for hypnozoiticidal activity. Hopes to achieve this were raised with the establishment of in vitro cultured hepatic stages of P. vivax [11], [12] and then P. cynomolgi [13], [14] in their respective primary host hepatocytes. In parallel, P. vivax hepatic stage maturation was obtained in the hepatoma cell line HepG2-A16 [15]. Observation of non-dividing P. vivax forms in HepG2-A16 cells when most hepatic forms have developed to form mature schizonts (5–8 days after sporozoite inoculation), prompted the suggestion that these corresponded to hypnozoites [15]. This was re-enforced when the proportion of these non-dividing forms correlated with the expected proportion of hypnozoites to dividing hepatic parasites for different P. vivax strains, namely in equal numbers from tropical strains and with a predominance of hypnozoites from temperate strains [16], [17]. However, these morphological observations alone are of insufficient weight to establish the actual nature of the non-dividing forms observed as hypnozoites. Failure of hepatic parasites to reach to first mitotic division might be due to other factors, such as defective sporozoites, or failure to thrive in a metabolically unsuitable host cell, or in a sub-optimal culture environment. Indeed, cultured hepatic parasites, as well as those found in vivo under natural conditions of infection, are often asynchronous, with large variations in size and developmental stage as cultivation progresses. Furthermore, in our experience small uninuclear hepatic forms are equally regularly observed along with mature hepatic schizonts in primary hepatocyte or hepatoma cell cultures infected with sporozoites from Plasmodium species that do not form hypnozoites, such as P. falciparum or P. berghei.
We have recently proposed that the differential susceptibility of normal hepatic forms and of hypnozoites to defined anti-malarial drugs might serve to indicate the presence of hypnozoites in infected hepatocyte cultures [18]. This was tested in parallel cultures of P. falciparum and P. cynomolgi infections in their host's respective primary hepatocytes, because this provides a means to establish if the non-dividing parasites obtained from the relapsing species (P. cynomolgi) and the non-relapsing species (P. falciparum) are biologically distinct.
Results and Discussion
Slow growing and normally developing hepatic forms occur in hepatocyte cultures
Primary hepatocytes were isolated from liver segments obtained from humans or from Macaca fascicularis, the natural host of P. cynomolgi [1], [2]. The primary hepatocyte cultures were infected with P. falciparum or P. cynomolgi sporozoites respectively, and then maintained for 11 days. Each day some wells from the cultures were fixed and the numbers and sizes of the hepatic forms present were measured. The hepatic forms were visualized using two stains: DAPI that reveals the nuclei and an antibody raised against a P. falciparum heat shock protein 70.1 (HSP70.1) [19], which recognizes both P. falciparum and P. cynomolgi parasites. HSP70.1 expression in hepatic stages is initiated about three hours after hepatocyte invasion and is maintained until full maturation of the hepatic schizont.
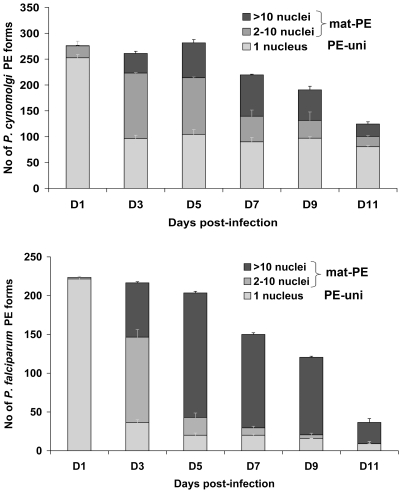
As expected, from Day 3 post-inoculation onwards, most P. falciparum and about half of P. cynomolgi hepatic forms obtained developed normally (parasite nuclear division and size increase), however in a proportion of the infected hepatocytes the parasites remained uninucleate (Fig. 1) and did not grow beyond their size on Day 1. The smaller uninucleate pre-erythrocytic (PE) forms were termed PE-uni, while the maturing forms were termed mat-PE. As the infection progressed, some differences were observed between the P. falciparum and P. cynomolgi cultures (Fig. 2). Nearly 40% of the hepatic forms in the P. cynomolgi were of the PE-uni type on Day 3 and these forms remained unchanged by Day 11, at which time they represented nearly two thirds of the hepatic forms as by that time nearly all developing forms had fully matured and burst. In contrast, the P. falciparum PE-uni forms were fewer in numbers from the outset and although their absolute numbers remained constant between Day 5 and Day 9, they had significantly declined by Day 11 (p<0.05, Mann-Whitney test). When both cultures were stopped on Day 11, most of the remaining mat-PE forms were large mature hepatic schizonts each with a large number of visible parasite nuclei. Similar proportions of long-lived PE-uni forms were obtained when P. cynomolgi sporozoites were used to infect cultured rhesus monkey (M. mulatta) primary hepatocytes (data not shown).
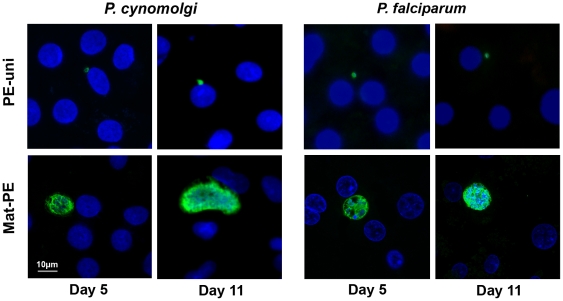
Figure 1. Slow growing and normally developing pre-erythrocytic (PE) forms.
Two types of hepatic parasites can be clearly distinguished from days 5 post-infection onwards, in in vitro cultured human or M. fascicularis primary hepatocytes infected with P. falciparum or P. cynomolgi sporozoites, respectively. The representative photomicrographs were made on cultures fixed on Day 5 and Day 11 post-infection. The parasite and host nuclei are stained with DAPI (blue), while the parasites are labelled by an antibody specific to the HSP70 of the two parasite species (green). Mature PE forms (Mat-PE) and uninucleate small PE forms (PE-uni) are clearly distinguishable.
Figure 2. Maturation dynamics of cultured P. falciparum and P. cynomolgi hepatic forms.
The proportion of PE forms at different stages of maturation (as assessed by the extent of nuclear division) is presented for cultures of increasing age. Uninucleate parasite forms correspond to the small forms (PE-uni) shown in Figure 1, while all others correspond to the mat-PE forms. The data was representative of three independent experiments.
Differential activity of selected antimalarial drugs distinguished the two hepatic forms
The PE-uni observed in the P. falciparum (a non-hypnozoite producing species) and in the P. cynomolgi (a hypnozoite producing species) hepatic cultures are indistinguishable from the non-dividing forms postulated to be hypnozoites that were observed in cultured HepG2 cells infected with P. vivax strains of different geographical origins [15], [16], [17], [20].
In order to provide unequivocal proof that the PE-uni forms observed in the P. cynomolgi (or P. vivax) hepatic cultures are indeed hypnozoites, it would be necessary to observe after a suitable period of latency (at least 3–4 weeks), a reactivation of the maturation process that would lead to the release of merozoites. This would be difficult for HepG2 cultures because beyond a week of incubation, the multiplication of the hepatoma cells will irrevocably lead to an unacceptable dilution of the few non-dividing forms. This can be overcome by prior irradiation of the HepG2 cells prior to inoculation with sporozoites, though it is not clear that hypnozoites are capable of forming and/or being reactivated in hepatoma cells. Primary hepatocytes do not divide in culture, but maintaining their viability beyond a few weeks has proven difficult.
Given these limitations, we postulated that data consistent with a hypnozoite identity for the P. cynomolgi PE-uni forms could be derived by exploiting the contrasting activities of three current anti-malarial drugs against hypnozoites [18]. Pyrimethamine (an inhibitor of dihydrofolate reductase), atovaquone (an inhibitor of the mitochondrial cytochrome c) and primaquine (putatively a mitochondrial inhibitor), have all been demonstrated to fully inhibit normal hepatic stage parasites in vivo and in vitro [10], [21], [22], [23], [24], [25]. Only primaquine however, is able additionally to eliminate hypnozoites [5], [6].
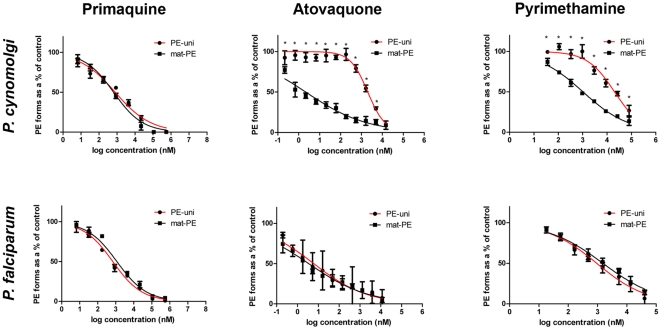
Having established above that the PE-uni forms are most evident between Day 5 and Day 9 of cultivation, during which time their numbers remain stable for both P. falciparum and P. cynomolgi, we obtained the IC50 values for these drugs against the PE-uni and the mat-PE forms following exposure to the drugs for three days from Day 5 to Day 8 (Fig. 3 and Table 1). As expected, the two types of hepatic forms (mat-PE and PE-uni) from both parasite species were eliminated with primaquine, with similar IC50 values (650 nM to 1166 nM, equivalent to 0.3 µg/ml–0.53 µg/ml of the diphosphate salt). In all cases, full inhibition was observed as the concentration reached 110 µM (50 µg/ml), though it should be noted that M. fascicularis hepatocytes viability decreased with exposure to primaquine concentrations higher than 22 µM (10 µg/ml) as assessed by MTT (TC50SH = 56 µM). By contrast, while both pyrimethamine and atovaquone inhibited the development of the mat-PE forms of both parasite species as well as the non-dividing PE-uni of P. falciparum, the P. cynomolgi PE-uni forms showed resistance to these drugs, displaying a 900-fold and a 20-fold increase in the IC50 values for atovaquone and pyrimethamine, respectively.
Figure 3. Dose-response curves for primaquine, atovaquone and pyrimethamine against the distinct hepatic forms observed for P. falciparum and P. cynomolgi.
The infected primary hepatocyte cultures were exposed to the varying drug concentrations from Day 5 to Day 8 post-inoculation and fixed on D8. The curves obtained for the PE-uni forms are presented in red, while those for the Mat-PE forms are presented in black. The data was representative of two independent assays for pyrimethamine (for both parasite species) and for primaquine (P. falciparum); and of three independent experiments for atovaquone (both parasite species) and primaquine (P. cynomolgi). *, p<0.05.
Table 1. IC50 values for selected drugs against the different hepatic parasites.
| Primaquine IC50 in µM (95% CI) | Atovaquone IC50 in nM (95% CI) | Pyrimethamine IC50 in µM (95% CI) | ||||
| PE-uni | mat-PE | PE-uni | mat-PE | PE-uni | mat-PE | |
| P. cynomolgi | 0.80 (0.55–1.17) | 0.65 (0.50–0.85) | 1997 (1638–2435) | 2.13 (1.38–3.30) | 20.31 (14.95–27.60) | 1.03 (0.84–1.25) |
| P. falciparum | 0.69 (0.51–0.94) | 1.17 (0.80–1.70) | 6.09 (2.67–13.94) | 3.48 (2.21–.47) | 0.94 (0.65–.37) | 1.53 (1.18–1.99) |
The inhibition of the PE-uni forms by primaquine indicates that they are metabolically active. However, the resistance of the P. cynomolgi PE-uni forms to atovaquone and pyrimethamine, as compared to the sensitivity of the P. falciparum PE-uni forms to these drugs, suggests that the metabolism of the cultured non-dividing hepatic parasites differs between the two species. The concordance of these observations in vitro with the known inability of pyrimethamine and atovaquone to prevent relapses and thus, eliminate hypnozoites in vivo, strongly suggests that the majority of the P. cynomolgi PE-uni forms we observed in culture represent hypnozoites.
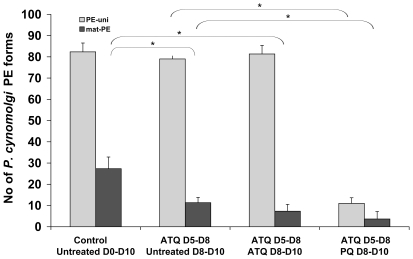
Since the ultimate aim is to develop a viable screening assay to identify novel hypnozoitocidal drugs, we modified the assay to include a PE-uni selection step before testing for primaquine activity against these forms. This was achieved by treating P. cynomolgi hepatic cultures with atovaquone between Day 5 and Day 8. Subsequent to this, most of the hepatic parasites present in the cultures were the PE-uni forms, and these forms persisted unaffected whether atovaquone treatment was withdrawn or maintained another three days, up to Day 10. By contrast, their numbers were significantly diminished by exposure to primaquine on Day 8 to Day 10 (Fig. 4).
Figure 4. Selection of P. cynomolgi PE-uni forms prior to screening for hypnozoiticidal activity.
Cultures were treated with atovaquone (ATQ) at 67 ng/ml (182 nM) for three days starting on Day 5 post-inoculation in order to eliminate the mat-PE parasites. Subsequently the cultures were either left untreated, treated for a further two days with ATQ (as above), or primaquine (PQ) at 10 µg/ml (22 nM). The cultures were then stopped by methanol fixation on Day 10 and the hepatic forms present enumerated. The data was representative of three independent experiments. *, p<0.05.
Conclusions
Here we demonstrate that the non-dividing P. cynomolgi and P. falciparum hepatic forms that are observed in cultured primary hepatocytes isolated from their respective natural hosts are distinct. The higher PE-uni numbers observed for P. cynomolgi, their maintenance in undiminished numbers after 10 days of cultivation, and their resistance to inhibition by atovaquone and pyrimethamine distinguishes them from the P. falciparum PE-uni forms. These observations, when associated to susceptibility to elimination by primaquine and to the contrasting relapse patterns of sporozoite-induced P. cynomolgi infections in rhesus monkeys after primaquine or pyrimethamine treatment, provide compelling support for the hypothesis that these in vitro PE-uni forms actually represent hypnozoites. In any case, the PE-uni forms seem to provide a hypnozoite surrogate suitable for drug discovery programs. Given the long-established biological and chemotherapeutic equivalence of P. vivax and P. cynomolgi infections [8], [9], [10], we consider it likely that similar PE-uni forms will be observed for P. vivax. It would also be reasonable to suggest that the non-dividing P. vivax forms initially observed in HepG2-A16 cells in mid 1980 [15], [16] and very recently [20] could also represent hypnozoites. Nonetheless, this would need a validation similar to the one presented here because it is not known whether hepatoma cells lines present a suitable environment for hypnozoite formation and eventual reactivation.
The work described here represents a first significant step towards the development of a screening assay aimed at the discovery of novel hypnozoiticidal compounds. The use of P. cynomolgi for this endeavour is justified by the fact that the requisite provision of a constant supply of sporozoites from the same strain, or eventually clone, can be achieved without the necessity to infect chimpanzees as is the case for P. vivax [20], or rely on patients infected by different P. vivax strains. On the other hand, if medium-throughput screening programs are envisaged it would be necessary to have access to easy-to-culture cell lines that are at least as susceptible to sporozoite infection as the primary hepatocytes isolated from monkey liver segments. Finally, the availability of a potential in vitro hypnozoite model opens the way to conduct detailed investigations into the nature and mechanisms of latency in Plasmodium parasites, a hitherto closed avenue of fundamental biological interest.
Methods
Ethics Statement
Human hepatocytes were isolated from liver segments taken after oral informed consent from adult patients undergoing partial hepatectomy as part of their medical treatment (Service de Chirurgie Digestive, Hépato-Bilio-Pancréatique et Transplantation Hépatique, Hôpital Pitié-Salpêtrière, Paris, France). The collection and use of this material for the purposes of the study presented here were undertaken in accordance with French national ethical guidelines under Article L. 1121-1 of the “Code de la Santé Publique”. Given that the tissue samples are classed as surgical waste, that they were used anonymously (the patient's identity is inaccessible to the researchers), and that they were not in any way genetically manipulated, article L. 1211-2 stipulates that their use for research purposes is allowed provided that the patient does not express any opposition to this to the surgeon prior to surgery after being informed of the nature of the research in which they might be potentially employed. Within this framework, the collection and use of this material was furthermore approved by the Institutional Review Board (Comité de Protection des Personnes) of the Centre Hospitalo-Universitaire Pitié-Salpêtrière, Assistance Publique-Hôpitaux de Paris, Paris, France.
All Macaca mulatta in this study were captive bred for research purposes. BPRC housing and animal care procedures are in compliance with Dutch law on animal experiments, European directive 86/609/EEC, and with the “Standard for Humane Care and Use of Laboratory Animals by Foreign Institutions”, identification number A5539-01, provided by the Department of Health and Human Services of the US National Institutes of Health (NIH). The local independent ethical committee, constituted according to Dutch law on animal experiments, approved the study protocols prior to start of the experiments. Adult cynomolgus macaques (Macaca fascicualris) were imported from Mauritius and housed in the facilities of the Centre d'Energie Atomique (CEA) (Fontenay-aux-Roses, France). Non-human primates (NHP, that includes M. fascicularis) are used at the CEA in accordance with French national regulation and under the inspections of national veterinary inspectors. The CEA is in compliance with Standards for Human Care and Use of Laboratory Animals (Animal Welfare Assurance, OLAW number #A5826-01). The use of NHP at CEA is in accordance with recommendation of the Weatherall report as follows: NHP are used at CEA only when no other models (in vitro or in vivo) are suitable for the aims of the project (recommendation n°1); the study is in the field of infectious diseases (recommendation n°2), Plasmodium physio-pathology; NHP are breed following the recommendation of the ETS123 and in accordance with the newly published Eureopean Directive (2010/63) (recommendation n°9). It should be stressed that none of the animal were specifically used for this work, since the liver fragments were collected at necropsy from animals that were euthanized in the course of unrelated studies, thus no suffering was specifically associated with the surgical procedure to obtain the liver fragments. This approach is fully in accordance with the 3R and reduces the number of animal used. Animal suffering avoidance and refinement of procedures are included in CEA aims. The animals were used under the supervision of the veterinarians in charge of the animal facility and the protocols employed were reviewed by the Ethical Animal Committee of the CEA. During ethical review, the end points and analgesia were reviewed and optimised. Experimental procedures were conducted in strict accordance with the recommendations of the European guidelines for the care and use of laboratory animals (European directive 86/609/EEC, OJ, L358, December 18, 1986). In addition, the CEA facilities was accredited (assurance identification number #A5826-01) by the National Institute of Health (USA) Office for Laboratory Animal Welfare (OLAW). The protocols and the use of hepatocytes for the purpose of the work described herein were approved by the Ethical Animal Committee of the CEA (Permit Number A 92-032-02).
Drugs
Primaquine, 8-(4-amino-1-methylbutylamino)-6-methoxyquinoline diphosphate salt, and pyrimethamine, 5-(4-chlorophenyl)-6-ethyl-2,4-pyrimidinediamine, were purchased from Sigma-Aldrich St Louis, MO, USA (P9504 and P771, respectively); atovaquone, trans-2-[4-(4-chlorophenyl)cyclohexyl]-3-hydroxy-1,4-naphthalenedione was purchased from Laboratoire GlaxoSmithKline, France.
Sporozoites
P. falciparum (strain NF54) sporozoites were obtained from infected Anopheles stephensi salivary glands collected on days 14–21 after an infective blood meal on a membrane-based feeder system (Department of Medical Microbiology, University Medical Centre St. Radboud, Nijmegen, The Netherlands). Plasmodium cynomolgi (M strain) sporozoites were obtained from infected Anopheles stephensi salivary glands collected on days 14–35 after an infective blood meal on blood from a Macaca mulatta, infected with blood stage P. cynomolgi parasites using membrane-based glass feeders (Biomedical Primate Research Centre, Rijswijk, The Netherlands).
Primary hepatocytes
Human hepatocytes were isolated from liver segments taken from adult patients in the course of partial hepatectomy (Service de Chirurgie Digestive, Hépato-Bilio-Pancréatique et Transplantation Hépatique, Hôpital Pitié-Salpêtrière, Paris, France). Simian hepatocytes were isolated from Macaca fascicularis or M. mulatta liver segments (provided by Roger Le Grand, commissariat à l'Energie Atomique CEA, Fontenay-aux-Roses, France, or by Clemens H M Kocken, Biomedical Primate Research Centre, Rijswijk, The Netherlands, respectively) taken from healthy animals. The primary hepatocytes were isolated using two-step enzymatic perfusion as previously described [26]. Briefly, the hepatic segments were successively perfused with HBSS-HEPES buffer and with collagenase (C5138, Sigma-Aldrich St Louis, MO, USA), and then dissociated. Viable cells were recovered after centrifugation over a 37% Percoll layer. The isolated simian primary hepatocytes that were obtained were immediately cryopreserved. On the days when human liver segments became available, and when the viability of the primary hepatocytes isolated from these was deemed satisfactory for infection with parasites, an aliquot of the M. fascicularis primary hepatocytes was thawed and the two host cell types were separately seeded and incubated for 24 h before infection.
The primary hepatocytes were seeded at a density of 8.3×104 cells per well allowing cell confluence in µclear plate Black 96 wells (Greiner Bio-one, France), or 2.1×105 cells per well in Lab-Tek 8-chambers slides, in both cases coated with rat tail collagen I (Becton Dickinson, Le Pont de Claix, France) in complete medium: William's medium E (Gibco, Cergy-Pontoise, France) supplemented with 10% foetal calf serum (Perbio), 5×10−5 M hydrocortisone hemisuccinate (Upjohn SERB Laboratoire), 5 µg/ml Insulin (Sigma), 2 mM L-Glutamine (25030-024, Gibco), 0.02 U/ml–0.02 µg/ml Penicillin-streptomycin (15140-122, Life Technologies) and incubated at 37°C, 5% CO2 for 24 h before infection.
Pre-erythrocytic cultures and drug assays
After isolation, the P. falciparum or P. cynomolgi sporozoites were suspended in complete medium and added to their respective host's primary hepatocyte cultures. For drug assays, 3×104 sporozoites were added to each well, or 1×105 P. cynomolgi sporozoites for cultures initiated in Lab-Tek 8-chambers slides. Infected cultures were fixed with cold 100% methanol on Days 1, 3, 5, 7, 9 and 11 post-infection in order to count and measure the size of the P. cynomolgi or P. falciparum pre-erythrocytic (PE) forms obtained. For the drug assays, the antimalarial drugs were added at different concentrations to complete medium. Infected cells were exposed to the drug for 3 days from Day 5 until fixation at Day 8 post-infection. In a second set of experiments, PE-uni selection was included by treatment of P. cynomolgi hepatic cultures with atovaquone at 67 ng/ml (182 nM) between Day 5 and Day 8. This was followed by a 2 days treatment with either the same concentration of ATO or with PQ at 10 µg/ml (22 nM), until fixation at Day 10 post-infection. In all cases, the medium containing the drug was renewed daily during treatment. Assays for each drug were generally conducted in two or three independent experiments. In an individual experiment, each concentration was tested in duplicate or triplicate wells.
Hepatic parasites were enumerated by immunofluorescence analysis. Briefly, following fixation, the parasites were specifically labelled with a mouse polyclonal serum raised against the P. falciparum heat shock protein 70.1 (PfHSP70.1) obtained after immunization with the recombinant protein. This serum also cross-reacts with P. cynomolgi HSP70. The labelled parasites were visualized with Alexa 488-conjugated goat anti-mouse immunoglobulin (Invitrogen). Parasite and host cell nuclei were stained with 1 µg/ml of diamidino-phenylindole (DAPI; Sigma). Parasites were enumerated by examination of the cultures under a fluorescence microscope at 200× magnification (Leica DMI 4000 B). Photomicrographs were obtained using a confocal Olympus BX61 microscope at 600× magnification on cells cultures in Lab-Tek 8-chambers slides.
The cytotoxicity of the anti-malarial drugs was evaluated on M. fascicularis hepatocytes treated as for the drug activity assays, through the colorimetric methylthiazolyldiphenyl-tetrazolium bromide test (MTT) [27]. Briefly, the primary hepatocytes were exposed to various concentrations of drug and three days later, a 100 µl MTT solution (500 µg/mL) was added in each well. After 4 h of incubation at 37°C, the plates were read in a spectrophotometer (540 nm absorbance wavelength) and the results were expressed as percentage of cellular viability compared to the non-treated control.
Data analysis
Dose-response curves and inhibitory concentrations 50% (IC50) were calculated by non-linear regression analysis using GraphPad Prism software with the data previously normalized to the untreated controls. Mean parasite numbers were compared using the Mann-Whitney non-parametric test. A p-value of less than 0.05 was considered to be statistically significant in the tests.
Acknowledgments
We wish to thank Thierry Diagana (Novartis Institute for Tropical Diseases, Singapore) for his invaluable support as well as for his important input in data analysis and manuscript preparation. We wish to thank Anne Charlotte Grüner for excellent critical comments, S van Amsterdam and A. vd Berg for expert technical assistance in developing the P. cynomolgi transmission platform, Benoit Delache and the TIPIV staff of the CEA for excellent technical assistance, and Christophe Joubert and the CEA animal facility staff.
Footnotes
Competing Interests: Georges Snounou is a PLoS Academic Editor.
Funding: This work was supported by a grant from the Medicines for Malaria Venture and a translational research grant (WT078285) from the Wellcome Trust to the Novartis Institute for Tropical Diseases, the Biomedical Primates Research Center and the Université Pierre et Marie Curie. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Garnham PCC. Oxford: Blackwell Scientific Publications; 1966. Malaria parasites and other haemosporidia. pp. 1–1114. [Google Scholar]
- 2.Coatney GR, Collins WE, Warren M, Contacos PG. Washington DC: U.S. Government Printing Office; 1971. The primate malarias. pp. 1–366. [Google Scholar]
- 3.Krotoski WA. Discovery of the hypnozoite and a new theory of malarial relapse. Trans R Soc Trop Med Hyg. 1985;79:1–11. [PubMed] [Google Scholar]
- 4.Sutherland CJ, Tanomsing N, Nolder D, Oguike M, Jennison C, et al. Two nonrecombining sympatric forms of the human malaria parasite Plasmodium ovale occur globally. J Infect Dis. 2010;201:1544–1550. doi: 10.1086/652240. [DOI] [PubMed] [Google Scholar]
- 5.Baird JK, Hoffman SL. Primaquine therapy for malaria. Clin Infect Dis. 2004;39:1336–1345. doi: 10.1086/424663. [DOI] [PubMed] [Google Scholar]
- 6.Hill DR, Baird JK, Parise ME, Lewis LS, Ryan ET, et al. Primaquine: report from CDC expert meeting on malaria chemoprophylaxis I. Am J Trop Med Hyg. 2006;75:402–415. [PubMed] [Google Scholar]
- 7.Kocken CHM, Remarque EJ, Dubbeld MA, Wein S, Van Der Wel AM, et al. Statistical model to evaluate in vivo activities of antimalarial drugs in a Plasmodium cynomolgi-macaque model for Plasmodium vivax malaria. Antimicrob Agents Chemother. 2009;53:421–427. doi: 10.1128/AAC.00576-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schmidt LH. Plasmodium cynomolgi infections in the rhesus monkey. Background studies. Am J Trop Med Hyg. 1982;31:609–611. doi: 10.4269/ajtmh.1982.31.609. [DOI] [PubMed] [Google Scholar]
- 9.Schmidt LH, Fradkin R, Genther CS, Rossan RN, Squires WL. Plasmodium cynomolgi infections in the rhesus monkey. I. The characteristics of untreated sporozoite-induced and trophozoite-induced infections. Am J Trop Med Hyg. 1982;31:612–645. [PubMed] [Google Scholar]
- 10.Schmidt LH, Fradkin R, Genther CS, Rossan RN, Squires WL. Plasmodium cynomolgi infections in the rhesus monkey. II. Responses of sporozoite-induced and trophozoite-induced infections to standard anitmalarial drugs. Am J Trop Med Hyg. 1982;31:646–665. [PubMed] [Google Scholar]
- 11.Mazier D, Landau I, Miltgen F, Druilhe P, Guguen-Guillouzo C, et al. Infestation in vitro d'hépatocytes humains par des sporozoïtes dee Plasmodium vivax: schizogonie et libération de mérozoïtes infestants pour les hématies humaines. Ann Parasitol Hum Comp. 1983;58:405–406. [PubMed] [Google Scholar]
- 12.Mazier D, Landau I, Druilhe P, Miltgen F, Guguen-Guillouzo C, et al. Cultivation of the liver forms of Plasmodium vivax in human hepatocytes. Nature. 1984;307:367–369. doi: 10.1038/307367a0. [DOI] [PubMed] [Google Scholar]
- 13.Millet PG, Jiang J-B, Lun Z-R, Bray RS, Canning EU, et al. Obtention in vitro de schizontes de Plasmodium cynomolgi bastianelli dans des hépatocytes de Macaca rhesus. Ann Parasitol Hum Comp. 1987;62:5–7. doi: 10.1051/parasite/19876215. [DOI] [PubMed] [Google Scholar]
- 14.Millet PG, Fisk TL, Collins WE, Broderson JR, Nguyen-Dinh P. Cultivation of exoerythrocytic stages of Plasmodium cynomolgi, P. knowlesi, P. coatneyi, and P. inui in Macaca mulatta hepatocytes. Am J Trop Med Hyg. 1988;39:529–534. doi: 10.4269/ajtmh.1988.39.529. [DOI] [PubMed] [Google Scholar]
- 15.Hollingdale MR, Collins WE, Campbell CC, Schwartz AL. In vitro culture of two populations (dividing and nondividing) of exoerythrocytic parasites of Plasmodium vivax. Am J Trop Med Hyg. 1985;34:216–222. doi: 10.4269/ajtmh.1985.34.216. [DOI] [PubMed] [Google Scholar]
- 16.Hollingdale MR, Collins WE, Campbell CC. In vitro culture of exoerythrocytic parasites of the North Korean strain of Plasmodium vivax in hepatoma cells. Am J Trop Med Hyg. 1986;35:275–276. doi: 10.4269/ajtmh.1986.35.275. [DOI] [PubMed] [Google Scholar]
- 17.Shu H, Lou S, Liu D, Fu R. [Observation on hypnozoite of different isolates of Plasmodium vivax in cultured materials]. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 1995;13:185–188. [PubMed] [Google Scholar]
- 18.Mazier D, Rénia L, Snounou G. A pre-emptive strike against malaria's stealthy hepatic forms. Nat Rev Drug Discov. 2009;8:854–864. doi: 10.1038/nrd2960. [DOI] [PubMed] [Google Scholar]
- 19.Mattei DM, Scherf A, Bensaude O, Pereira Da Silva LH. A heat shock-like protein from the human malaria parasite Plasmodium falciparum induces autoantibodies. Eur J Immunol. 1989;19:1823–1828. doi: 10.1002/eji.1830191010. [DOI] [PubMed] [Google Scholar]
- 20.Chattopadhyay R, Velmurugan S, Chakiath C, Andrews Donkor L, Milhous W, et al. Establishment of an in vitro assay for assessing the effects of drugs on the liver stages of Plasmodium vivax malaria. PLos One. 2010;5:e14275. doi: 10.1371/journal.pone.0014275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fisk TL, Millet PG, Collins WE, Nguyen-Dinh P. In vitro activity of antimalarial compounds on the exoerythrocytic stages of Plasmodium cynomolgi and P. knowlesi. Am J Trop Med Hyg. 1989;40:235–239. doi: 10.4269/ajtmh.1989.40.235. [DOI] [PubMed] [Google Scholar]
- 22.Jiang J-B, Bray RS, Krotoski WA, Canning EU, Liang D-S, et al. Observations on early and late post-sporozoite tissue stages in primate malaria. V. The effect of pyrimethamine and proguanil upon tissue hypnozoites and schizonts of Plasmodium cynomolgi bastianellii. Trans R Soc Trop Med Hyg. 1988;82:56–58. [PubMed] [Google Scholar]
- 23.Povinelli L, Fox BC, Parise ME, Monson TA, Morrisey JM, et al. Plasmodium vivax malaria in spite of atovaquone/proguanil (malarone) prophylaxis. J Travel Med. 2003;10:353–355. doi: 10.2310/7060.2003.9318. [DOI] [PubMed] [Google Scholar]
- 24.Jiménez BC, Navarro M, Huerga H, López-Román E, Mendoza A, et al. Tertian malaria (Plasmodium vivax and Plasmodium ovale) in two travelers despite atovaquone-proguanil prophylaxis. J Travel Med. 2006;13:373–375. doi: 10.1111/j.1708-8305.2006.00073.x. [DOI] [PubMed] [Google Scholar]
- 25.Maguire JD, Llewellyn DM. Relapsing vivax malaria after 6 months of daily atovaquone/proguanil in Afghanistan: the case for expanded use of primaquine as a causal prophylactic. J Travel Med. 2007;14:411–414. doi: 10.1111/j.1708-8305.2007.00153.x. [DOI] [PubMed] [Google Scholar]
- 26.Guguen-Guillouzo C, Campion JP, Brissot P, Glaise D, Launois B, et al. High yield preparation of isolated human adult hepatocytes by enzymatic perfusion of the liver. Cell Biol Int Rep. 1982;6:625–628. doi: 10.1016/0309-1651(82)90187-4. [DOI] [PubMed] [Google Scholar]
- 27.Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]