Abstract
Red blood cell (RBC) storage lesions have been shown to be associated with some adverse reactions; numerous studies have focused on the lesions caused by storage, and few data on the RBC storage lesions caused by prestorage treatments of leucocyte filtration and irradiation. In this study, we examined the changes related with the RBC storage lesions, including 2,3-diphosphatidylglyceric acid (2,3-DPG), pH, free hemoglobin (Hb), supernatant free K+ and Na+ concentration, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH). Along with the increasing storage time, decreases in 2, 3-DPG levels, pH and Na+ concentration, increases in K+ and free Hb concentrations, and significant morphological changes in RBC in all groups were found. The changes in the groups of irradiation, leucocyte filtration and the combined irradiation and leucocyte filtration were more significant than those in the untreated group. Meanwhile, the MCV levels of the three treated groups were significantly lower than those in the untreated group, while the MCH variations were significantly higher. Our results suggest that irradiation and leucocyte filtration before storage may aggravate blood storage lesions.
Introduction
Blood transfusion administration is a necessary and effective method for clinical surgeries, emergency rescue and hematopathy treatment as well. The influence of blood storage lesions (oxygen free radicals damage erythrocyte membranes, increasing their brittleness and decreasing the transformation index) on blood transfusion treatment has been more valuable. Blood will develop different degrees of damage during storage at 4°C, which will significantly affect the health of patients. Meanwhile, procedures such as irradiation and leucocyte filtration before storage will also lead to different degrees of lesion to red blood cell (RBC) [1]–[2].
Filtration and irradiation brought a series of clinical benefit [3]–[4], such as preventing or delaying the non-haemolytic febrile transfusion reaction (NHFTR), reducing the risk of leukocyte-associated virus transmission (CMV, EBV, HTLV), inhabition the cytokine generation and complement activation, preventing transfusion-associated graft-versus-host disease (TA-GVHD), and so on. Leukoreduction of cellular blood components has become the standard and irradiation has widely applied. Due to the acceleration by filtration and irradiation, the US Food and Drug Administration (FDA) limited the maximal storage time of irradiated RBC to 28 days [5], and 35 days by state food and drug administration (SFDA) in China.
To analyze the RBC lesions variations caused by various blood operations and storage time, we investigated the influence of irradiation and/or leukoreduction by filtration on blood quality by measuring 2,3-diphosphatidylglyceric acid (2,3-DPG) levels, pH, free hemoglobin (Hb), supernatant free K+ and Na+ concentration, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC) and RBC morphology.
Results
RBC morphological changes during storage
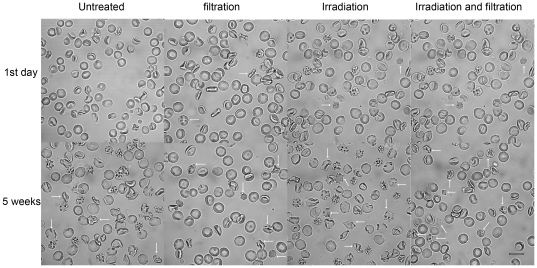
RBC morphology was observed during the storage period using confocal laser scanning microscopy and along with increasing storage time, the normal structure of red blood cells disappeared, and displayed with thin sheet morphology and shrinkage, with a large number of pseudopodia, debris and impurities appearing. This effect was enhanced in all the irradiation and filtration treated groups and treatment was associated with the appearance of a large amount of cell debris (Figure 1).
Figure 1. Changes in RBC morphology during storage.
Untreated group, leucocyte filtration group, Irradiation group, Irradiation and filtration group after storaged 1 day and 35 days were observed (white arrow indicated deformation and lesion RBC). Images were taken under Zeiss 510 META confocal laser scanning microscopy (original magnification ×1000, scale bar = 10 µm).
Changes in Physical and Chemical Indexes in Storage Process
Changes in pH 2,3-DPG levels, free Hb concentrations, supernatant free K+ and Na+ concentration in Storage Process
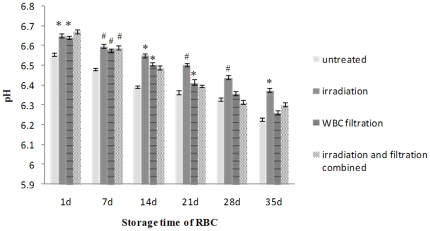
pH was decreased along with the increasing storage time, and was higher in all the treated groups, particularly the irradiation groups, than those of the untreated group (Figure 2).
Figure 2. pH changes in each group during storage.
#: P<0.05 and *: P<0.01 compared with untreated group. WBC, white blood cell.
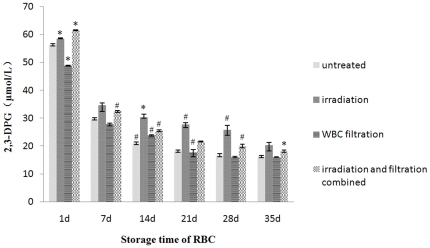
Along with the increasing storage time, the level of 2,3-DPG in each group decreased. The peak period of observed decrease was 1–7 days, and 21 days later this kind of decrease gradually slowed down (Figure 3). Among the 4 experimental groups, the decrease observed in the filtration group was the most significant, and one day after leucocyte filtration, 2,3-DPG levels dropped to 48.786 µmol/L (85.02%), the lowest one. These changes remained significant between days 1 and 21. Interestingly, the smallest decrease in 2,3-DPG levels in the RBC suspension during storage was observed in the irradiation group.
Figure 3. 2,3-DPG changes in each group during storage.
#: P<0.05 and *: P<0.01 compared with untreated group.
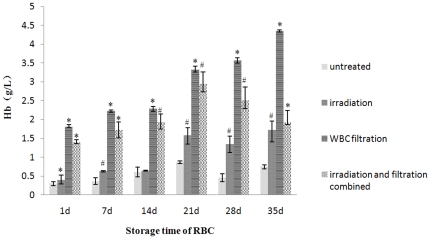
Free Hb concentrations were also increased along with the increasing storage time, and this tendency became more prominent after leucocyte filtration. Compared with the untreated group, the groups after filtration had a rapid and severe increase. Interestingly, after increased to the peak in 21 day, free Hb concentrations in combined irradiation and filtration groups decreased (Figure 4). Changes were seen in all treatment groups at all time points with statistical difference.
Figure 4. Free Hb concentrations changes in each group during storage.
#: P<0.05 and *: P<0.01 compared with untreated group.
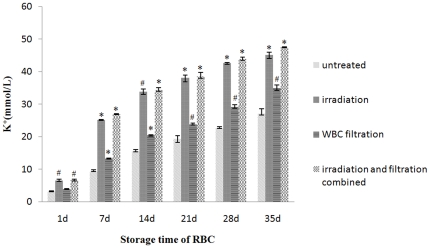
Similar to the free Hb concentrations, the K+ concentration had a dramatic change after filtration. Although affected slightly after irradiation immediately, the K+ concertration of all the samples was increased along with increasing storage time, with the irradiation treatment accelerating the increase in K+ concentration. This had increased to over 40 mmol/L after 28 days storage in the filtration and the combined treatment group (Figure 5).
Figure 5. K+ concentration changes in each group during storage.
#: P<0.05 and *: P<0.01 compared with untreated group.
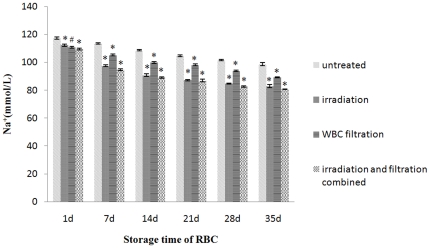
Equally, the Na+ concentration of the untreated group, leucocyte filtration group, irradiation group, and combined treatment group was decreased significantly during days 1–14, and on day 14 dropped to 108.30, 90.63, 99.77 and 89.10 mmol/L respectively. But those of the irradiation group and combined treatment group did not decrease significantly after day 14. The Na+ concentration on day 28 was 101.53, 84.67, 93.80 and 82.63 mmol/L, respectively (Figure 6).
Figure 6. Na+ concentration changes in each group during storage.
#: P<0.05 and *: P<0.01 compared with untreated group.
Changes in MCV and MCH during storage
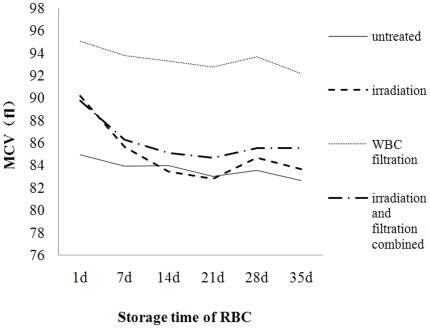
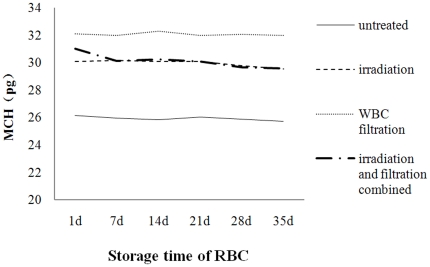
After leucocyte filtration and irradiation, the MCV decreased significantly than that in the untreated group. And the MCH showed a overall downward tendancy during storage, with the MCH of the treated groups higher than than that in the untreated group, but without significance (Figures 7 and 8).
Figure 7. Change in MCV over time during storage at 4°C.
Figure 8. Change in MCH over time during storage at 4°C.
Discussion
Although RBC transfusion is a life-saving procedure in some circumstances, accumulating evidence links transfusion of RBCs with increased morbidity in certain disease [6]–[7] and mortality in certain patients [8]–[9]. The adverse events are related to the structure and function change of RBCs caused by storage. Numerous studies had focused on the changes of RBC's structure and function caused by storage. Substantial evidences revealed that along with the storage time, cell morphology, potassium concentration, pH, free Hb and 2,3-DPG contents had undergone significant changes [10]–[13]. However, these studies followed with relevant blood-banking industry standard operating procedures, which stored after irradiation and/or filtration, with little research about the relationship between prestorage treatment and RBC storage lesions.
Previous studies had shown that irradiation and filtration resulted in a remarkable alteration of the RBC's properties. Gamma irradiation of plasma caused a significant increase in serum Hb, potassium and LDH, a reduction in deformability and viability [13], [14]. Mechanical force (generated by filtration, such as shear force, pressure and so on) will lead to RBC hemolysis [15]–[16]. Consequently, irradiation and filtration will aggravate RBC storage lesions and accelerate RBC hemolysis in vitro. But, there were little light shed on the influence of RBC storage lesions caused by filtration, irradiation, combined filtration and irradiation.
In this study, we found that irradiation, filtration, combined irradiation and filtration in prestorage can cause significant damage in RBC and intensify the RBC storage lesions. Compared with the untreated group, excluding exceptional cases, irradiation and/or filtration resulted in extremely significant difference in all measured factors (pH, 2,3-DPG, free Hb, K+, Na+, MCV, MCH). All these changes occurred in very short time (≤1 day). Free Hb concentration showed most obvious increase after irradiation and/or filtration, especially after filtration. The dramatic change in free Hb concentration suggested that filtration in prestorage would induce great damage to the membrane structure, even led to RBCs lesions (mechanical lesions). This lesions might cause cell debris, RBC's intracellular substances and the contents of leucocyte might secondarily damage RBC and worsen RBCs storage lesions. In the storage process, the free Hb, potassium and sodium concentration of all treated groups became increasingly evident, indicating that irradiation and/or filtration in prestorage would intensify RBCs storage lesions in the same storage conditions.
Interestingly, we did observe that after increased to the peak in 21 day, free Hb concentrations in combined irradiation and filtration groups decreased (Figure 4), which seems not consistent with a closed system. As Kriebardis et al. [17] have shown that during storage RBC-derived vesicles contain hemoglobin and many other proteins. We speculated that these RBC-derived vesicles in our system might explain why the free Hb decreased during the storage.
Reverberi et al. [14] suggested that the blood storage before irradiation was inconsequential. Zimmermann et al. [18] also irradiated the leukoreduced blood on day 14 and they showed that there were no significant differences between RBCs irradiated and the ones non-irradiated with respect to the rate of hemolysis, the potasium levels, ATP per g Hb, and 2,3-DPG per g Hb. However, RBC morphology was found to be adversely influenced by storage time and two treatments, with RBC becoming gradually thinner, changing from a full disk and appearing wrinkled and cracked. Previous studies have indicated that morphological changes of RBC, the degradation of hemoprotein and so on, caused RBC to have a lack of variability, elasticity and shortened life cycle [16], [18]–[22]. The life cycle of leucocytes is shorter than 35 days. So, most leucocytes may have already broken down by the end of the 35-day shelf life. The leucocyte debris may not be efficiently filtered and may still cause some adverse reactions during blood transfusion.
Moreover, some severe changes attracted our attention. While on day 28, the potassium concentration of the irradiation group was over 40 mmol/L and the sodium concentration was lower than 85 mmol/L. High potassium concentration may cause acidosis and potassium poisoning. All treated groups had a high concentration of free Hb. This changes could induce an increase in local ADP concentrations and thereby promote thrombosis [23]. A decrease in the 2,3-DPG levels directly increases the affinity of RBC for oxygen, decreases the viability of RBC after infusion, prolongs the time of RBC movement through capillaries, induces inflammation and directly affects the clinical infusion [24]. After long time storage, the 2,3-DPG concentration levels were very low in four groups, suggesting that the function of RBCs was damaged seriously. These changes were associated with adverse reactions after transfusion, and we also found that: TNF-α and IL-6 level of cancer patients had significant changes after transfusion (data not shown).
Our study was the first comprehensive, systematic analysis on the RBC storage lesion caused by irradiation, filtration, combined irradiation and filtration, which is important both to clinical practitioners and to blood bankers. According to the needs of patients to decide whether irradiation and choose the best time to filter would reduce RBC storage lesions. As Reverberi et al. [14] pointed out, probably it is the best practice to irradiate the filtered blood just before transfusion.
In conclusion, our findings have emphasized the importance of the damage to RBC during storage. In particular, irradiation and leucocytes filtration before storage may lead to greater damage during storage, which will directly influence the effectiveness of RBC transfusion. Reducing the adverse transfusion reactions and also maintaining the activity of red blood cell to guarantee an effective transfusion effects is one of the prominent problems that needs to be further studied and resolved.
Methods
Prestorage treatments and storage
This study was approved by the Medical Ethnic Review Committee of the Second Hospital Affiliated to the Third Military Medical University. Written informed consents were obtained from all the human subjects involved in this study. Two bags of blood with 200 mL in each were obtained from each voluntary blood donor. Totally 4000 mL blood was taken from 10 donors at the Chongqing Blood Center, and was preserved in RBC preservation solution for the irradiation and/or filtration treatment. Each 1000 ml of RBC preservation solution contained (in mM): 27.5 C6H12O6·6H2O, 6.02 NaH2PO4·2H2O, 84.44 NaCl, 80.05 C6H14O6, 1.04 C5H5N5, 5.10 C6H5O7Na3·2H2O and 0.95 C6H8O7·H2O. The irradiation, leucocyte filtration, combined irradiation and filtration were managed on Day 0 and all bags were stored at 4°C. The irradiation, combined irradiation and filtration groups were gamma-irradiated with a dose of 25 Gy by a blood irradiator (BIOBEAM8000, Germany) in Chongqing Blood Center. The irradiation dose was determined using thermoluminescence dosimetry [25]. After the treatments, the blood was randomly divided into 4 groups (untreated, irradiation, leucocyte filtration, irradiation and filtration combined; N = 36 in each). After overnight storage, 24 bags of 25 mL blood sample of Day 1 were shipped to our laboratory at 2 to 8°C. The samples of Day 7, 14, 21, 28 and 35 were treated accordingly to the same procedure for Day 1.
Measurement in vitro
The samples of each time point (Day 1, 7, 14, 21, 28 and 35) were rejuvenated and used for analysis of 2,3-DPG levels, pH, free Hb levels, free K+ and Na+ concentrations, MCV and MCH. The changes in RBC morphology were observed over time. Free K+, Na+ concentrations and pH were analyzed by a blood gas analyzer (sysmex kx-21N hematology analyzer); free Hb and 2,3-DPG levels were determined by corresponding commercial kits (Hb determination kit, purchased from Nanjing Jian Cheng Research Institute, the First Branch of Biological Engineering; 2,3-DPG kit, purchased from USCNLIFE); MCV and MCH were measured by a hematology analyzer (SYSMEX-XE2100, Japan); and cell morphological changes were observed by confocal laser scanning microscopy (Zeiss LSM 510 META).
Statistical analysis
All the data were expressed as mean ± standard deviation (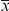 ±s). Student t-test of software SPSS 13.0 was used to analyze the variance among the groups. P<0.05 was considered significant different and P<0.01 was considered extremely significant different.
±s). Student t-test of software SPSS 13.0 was used to analyze the variance among the groups. P<0.05 was considered significant different and P<0.01 was considered extremely significant different.
Acknowledgments
We thank Dr. Jian Liu for the insightful comments and help in manuscript preparation.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: This study was supported by XinQiao Hospital “1520 project” special foundation and Scientific Researching Fund Projects of the Third Military Medical University of Chinese PLA (NO:2007D046). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Tinmouth A, Chin-Yee I. The clinical consequences of the red cell storage lesion. Transfus Med Rev. 2001;15:91–107. doi: 10.1053/tmrv.2001.22613. [DOI] [PubMed] [Google Scholar]
- 2.Tinmouth A, Fergusson D, Yee IC, Hébert PC ABLE Investigators. Clinical consequences of red cell storage in the critically ill. Transfusion. 2006;46:2014–2027. doi: 10.1111/j.1537-2995.2006.01026.x. [DOI] [PubMed] [Google Scholar]
- 3.Nielsen HJ, Hammer JH, Krarup AL, Nielsen LM, Reimert CM, et al. Prestorage leukocyte filtration may reduce leukocyte-derived bioactive substance accumulation in patients operated for burn trauma. Burns. 1999;25:162–170. doi: 10.1016/s0305-4179(98)00157-0. [DOI] [PubMed] [Google Scholar]
- 4.Sugawara A, Nollet KE, Yajima K, Saito S, Ohto H. Preventing platelet-derived microparticle formation–and possible side effects-with prestorage leukofiltration of whole blood. Arch Pathol Lab Med. 2010;134:771–775. doi: 10.5858/134.5.771. [DOI] [PubMed] [Google Scholar]
- 5.Williamson LM, Warwick RM. Transfusion-associated graft-versus-host disease and its prevention. Blood Rev. 1995;9:251–261. doi: 10.1016/s0268-960x(95)90016-0. [DOI] [PubMed] [Google Scholar]
- 6.Zallen G, Offner PJ, Moore EE, Blackwell J, Ciesla DJ, et al. Age of transfused blood is an independent risk factor for postinjury multiple organ failure. Am J Surg. 1999;178:570–572. doi: 10.1016/s0002-9610(99)00239-1. [DOI] [PubMed] [Google Scholar]
- 7.Leal-Noval SR, Jara-López I, García-Garmendia JL, Marín-Niebla A, Herruzo-Avilés A, et al. Influence of erythrocyte concentrate storage time on postsurgical morbidity in cardiac surgery patients. Anesthesiology. 2003;98:815–822. doi: 10.1097/00000542-200304000-00005. [DOI] [PubMed] [Google Scholar]
- 8.Rao SV, Jollis JG, Harrington RA, Granger CB, Newby LK, et al. Relationship of blood transfusion and clinical outcomes in patients with acute coronary syndromes. JAMA. 2004;292:1555–1562. doi: 10.1001/jama.292.13.1555. [DOI] [PubMed] [Google Scholar]
- 9.Kuduvalli M, Oo AY, Newall N, Grayson AD, Jackson M, et al. Effect of peri-operative red blood cell transfusion on 30-day and 1-year mortality following coronary artery bypass surgery. Eur J Cardiothorac Surg. 2005;27:592–598. doi: 10.1016/j.ejcts.2005.01.030. [DOI] [PubMed] [Google Scholar]
- 10.Holme S. Current issues related to the quality of stored RBCs. Transf Apher Sci. 2005;33:55–61. doi: 10.1016/j.transci.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 11.Bennett-Guerrero E, Veldman TH, Doctor A, Telen MJ, Ortel TL, et al. Evolution of adverse changes in stored RBCs. Proc Natl Acad Sci U S A. 2007;104:17063–17068. doi: 10.1073/pnas.0708160104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Niinistö K, Raekallio M, Sankari S. Storage of equine red blood cells as a concentrate. Vet J. 2008;176:227–231. doi: 10.1016/j.tvjl.2007.02.015. [DOI] [PubMed] [Google Scholar]
- 13.Agarwal P, Ray VL, Choudhury N, Chaudhary RK. Effect of pre-storage gamma irradiation on red blood cells. Indian J Med Res. 2005;122:385–387. [PubMed] [Google Scholar]
- 14.Reverberi R, Govoni M, Verenini M. Deformability and viability of irradiated red cells. Ann Ist Super Sanita. 2007;43:176–185. [PubMed] [Google Scholar]
- 15.Yasuda T, Funakubo A, Fukui Y. An investigation of blood damage induced by static pressure during shear-rate conditions. Artif Organs. 2002;26:27–31. doi: 10.1046/j.1525-1594.2002.06786.x. [DOI] [PubMed] [Google Scholar]
- 16.Yasuda T, Funakubo A, Miyawaki F, Kawamura T, Higami T, et al. Influence of static pressure and shear-rate on hemolysis ofred blood cells. ASAIO J. 2001;47:351–353. doi: 10.1097/00002480-200107000-00011. [DOI] [PubMed] [Google Scholar]
- 17.Kriebardis AG, Antonelou MH, Stamoulis KE, Economou-Petersen E, Margaritis LH, et al. RBC-derived vesicles during storage: ultrastructure, protein composition, oxidation, and signaling components. Transfusion. 2008;48:1943–1953. doi: 10.1111/j.1537-2995.2008.01794.x. [DOI] [PubMed] [Google Scholar]
- 18.Zimmermann R, Wintzheimer S, Weisbach V, Strobel J, Zingsem J, et al. Influence of prestorage leukoreduction and subsequent irradiation on in vitro red blood cell (RBC) storage variables of RBCs in additive solution saline-adenine-glucose-mannitol. Transfusion. 2009;49:75–80. doi: 10.1111/j.1537-2995.2008.01920.x. [DOI] [PubMed] [Google Scholar]
- 19.Karon BS, Hoyer JD, Stubbs JR, Thomas DD. Changes in Band 3 oligomeric state precede cell membrane phospholipid loss during blood bank storage of red blood cells. Transfusion. 2009;49:1435–1442. doi: 10.1111/j.1537-2995.2009.02133.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kriebardis AG, Antonelou MH, Stamoulis KE, Economou-Petersen E, Margaritis LH, et al. Progressive oxidation of cytoskeletal proteins and accumulation of denatured hemoglobin in stored red cells. J Cell Mol Med. 2007;11:148–155. doi: 10.1111/j.1582-4934.2007.00008.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sparrow RL, Veale MF, Healey G, Payne KA. Red blood cell (RBC) age at collection and storage influences RBC membrane-associated carbohydrates and lectin binding. Transfusion. 2007;47:966–968. doi: 10.1111/j.1537-2995.2007.01230.x. [DOI] [PubMed] [Google Scholar]
- 22.Greenwalt TJ. The how and why of exocytic vesicles. Transfusion. 2006;46:143–152. doi: 10.1111/j.1537-2995.2006.00692.x. [DOI] [PubMed] [Google Scholar]
- 23.Bosman GJ, Werre JM, Willekens FL, Novotný VM. Erythrocyte ageing in vivo and in vitro: structural aspects and implications for transfusion. Transfus Med. 2008;18:335–347. doi: 10.1111/j.1365-3148.2008.00892.x. [DOI] [PubMed] [Google Scholar]
- 24.Högman CF, Löf H, Meryman HT. Storage of red blood cells with improved maintenance of 2,3-bisphosphoglycerate. Transfusion. 2006;46:1543–1552. doi: 10.1111/j.1537-2995.2006.00893.x. [DOI] [PubMed] [Google Scholar]
- 25.Botter-Jensen L, Christensen P. Transit dose in extracorporeal blood irradiation devices measured by means of thermoluminescence. Acta Radiol Supp. 1972;l313:247–252. [PubMed] [Google Scholar]