Abstract
In this report, we describe an effect of photodynamic therapy (PDT) on membrane trafficking in murine 1c1c7 hepatoma cells. A brief exposure of 1c1c7 cells to a 20 nM concentration of the phosphatidylinositol kinase class-3 antagonist wortmannin led to the rapid appearance of cytoplasmic vacuoles. Fluorescence monitoring of plasma membrane-associated 1-[4-(trimethylamino)-phenyl]-6-phenylhexa-1,3,5-triene (TDPH) over time demonstrated that the wortmannin-induced vacuoles were derived from endocytosed plasma membrane. Low-dose photodamage catalyzed by the lysosomal photosensitizer NPe6, prior to the addition of wortmannin, prevented formation of these vacuoles. NPe6 was found to suppress for several hours the normal trafficking of TDPH-labeled plasma membrane to the cytosol, and the formation of punctate TDPH-labeled cytoplasmic vesicles. The ability of NPe6-induced photodamage to suppress wortmannin-induced vacuolization occurred under conditions that did not disrupt lysosomes and were at or below the threshold of cytostatic/cytotoxic effects. Furthermore, the suppressive effects of NPe6-PDT were not prevented by inclusion of an agent that stabilized lysosomal membranes, or by E64d, an inhibitor of lysosomal cathepsin proteases. Mitochondrial photodamage was less effective at preventing wortmannin-induced vacuole formation and PDT directed against the ER had no effect. The role of photodamage to the endocytic pathway may be a hitherto unexplored effect on cells that selectively accumulate photosensitizing agents. These results indicate that photodamage directed against endosomes/lysosomes has effects independent of the release of lysosomal proteases.
Introduction
Photodynamic therapy (PDT) involves the use of photosensitizing agents that localize in neoplastic tissues and/or their vasculature.1 While the term PDT implies a therapeutic intent in a clinical context, it is used here to indicate the effects of irradiation on photosensitized cells in culture. Irradiation at an appropriate wavelength leads to an interaction between the photosensitizer and molecular oxygen resulting in formation of reactive oxygen species that can initiate cytotoxic effects. Although PDT can lead to both autophagy2 and apoptosis,3 effects on membrane trafficking have seldom been explored.
The process of endocytosis is responsible for such critical processes as nutrient uptake, receptor signaling, growth, differentiation, adhesion and migration. The endocytic pathway entails the internalization of the plasma membrane and its trafficking to early endosomes, where membrane material can be subsequently re-utilized to form new plasma membrane or eventually trafficked to lysosomes for degradation. Wortmannin is a potent and irreversible antagonist of a PI-3 kinase involved in regulation of membrane traffic.4–6 Several authors have reported the formation of cytoplasmic vacuoles upon exposure of cultured mammalian cells to wortmannin.5–7 Such vacuoles are believed to derive from endocytosed plasma membrane whose normal processing is blocked by wortmannin. Reaves et al.5 concluded that these represent swollen late endosomes reflecting an inhibition of membrane traffic from endosomes to lysosomes.
Initiation of the canonical pathway for autophagosome formation is dependent on a class III PI-3 kinase.8 Exposure of cells to wortmannin, a PI-3 K antagonist, is therefore expected to prevent autophagy. We previously used wortmannin in our studies as a tool to determine the role of PDT-induced autophagy as a pro-survival and/or death pathway.9
Santus’ group had earlier demonstrated that lysosomal photodamage mediated by fluoroquinolones could lead to impaired endocytosis.10,11 It was proposed that this result derived from release of lysosomal proteases into the cytoplasm, since the irreversible cysteine-protease inhibitor E64d prevented the impairment. The current study was prompted by a finding that wortmannin induces formation of cytoplasmic vacuoles, an effect blocked after PDT involving NPe6, an agent that targets endosomes and lysosomes.
In this report, we demonstrate that the wortmannin-induced vacuoles are derived from endocytosis of plasma membrane, that PDT involving NPe6 suppresses aspects of plasma membrane endocytosis, and that lysosomal proteases have no role in the process. We also explored the effect of PDT involving lysosomal, mitochondrial and ER targets on membrane trafficking in 1c1c7 murine hepatoma cells.
Results and discussion
Wortmannin-induced vacuole formation
Cultures of control (untreated) murine hepatoma 1c1c7 cells exhibited <3 cytoplasmic vacuoles per cell (Fig. 1A). Multiple vacuoles began to appear within 15 min of treatment with 20 nM wortmannin (Fig. 1B). This effect became much more pronounced after 30 min (Fig. 1C). Similar effects were produced by the PI-3 K inhibitor LY294002 but not by its inactive analog P30351 (not shown). The effects of wortmannin were reversible. Vacuoles began to disappear within 1 h after replacing the medium (Fig. 1D) with <3 vacuoles per cell seen after a 2 h incubation in wortmannin-free medium (Fig. 1E). The vacuoles persisted for several hours if the subsequent incubation in drug-free medium was carried out at 15°C (not shown).
Fig. 1.
Time-course of vacuole formation induced by wortmannin. (A) Control cells. (B) Cultures after a 15 min exposure to 20 nM wortmannin. (C) Cultures after a 30 min exposure to wortmannin. (D,E) Cells incubated with 20 nM wortmannin for 30 min before being washed, placed in fresh medium and incubated with fresh medium for 1 h (D) or 2 h (E). White bar in panel A = 20 μm. These images represent typical fields. Some 1c1c7 cells exhibited a slightly compressed cell morphology (e.g., panel A) but this does not appear to affect the response to wortmannin or PDT. In replicate studies, we found 100% of cells were uniformly highly vacuolated after a 30 min exposure to wortmannin, with <10% of cells showing vacuoles after 2 h in fresh medium.
Examination of the vacuoles by electron microscopy indicated the presence of material within the lumens, although the nature of the encapsulated material was not obvious (Fig. 2A). The micrographs shown in Fig. 2A are quite similar to those observed in rat kidney cells following treatment with wortmannin.7 Electron microscopy also revealed that wortmannin-induced vacuoles were contained within a contiguous single membrane, as opposed to the characteristic double-membrane of autophagosomes. To further corroborate that the observed vacuoles were not autophagosomes, we monitored the effects of wortmannin on LC3 distribution in a 1c1c7 sub-line that stably expressed a GFP-LC3 fusion protein (Fig. 2, B–E). Autophagy induces the clustering of GFP-LC3 into intense fluorescent punctate loci that correspond to autophagosomes.2 GFP-LC3 was diffusely distributed in control cultures (panel C), and remained diffusively distributed following exposure to wortmannin (panel E). Loss of fluorescence in cytoplasmic regions between the nucleus and the plasma membrane was often observed and may represent a thinning of the cytoplasm in this region as a result of interference with membrane recycling processes. No punctate fluorescence was, however, observed.
Fig. 2.
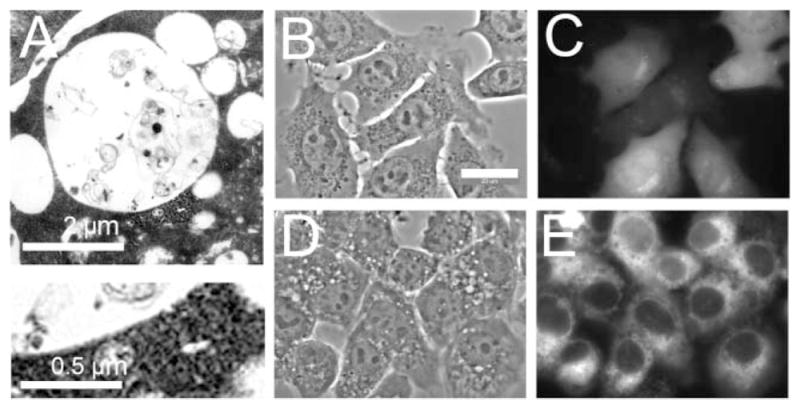
Wortmannin vacuoles are not autophagosomes: (A) 1c1c7 cultures were treated with 20 nM wortmannin for 30 min at 37 °C prior to being processed for electron microscopy. White bar = 2 μm. An enlarged portion of the vacuole membrane is shown in the lower image. The wortmannin-induced vacuoles lack the double-membrane structure characteristic of autophagosomes. (B–E) 1c1c7 cells that stably expressed a GFP-LC3 fusion protein were treated with nothing (B,C) or 20 nM wortmannin for 30 min (D,E) before phase (B,D) or fluorescence (C,E) microscopy. White bar in panel B = 20 μm.
Endocytic internalization studies with TDPH
Because of the affinity of TDPH for hydrophobic loci in the plasma membrane, this probe can be used to monitor endocytic internalization of the plasma membrane.12,13 A 30 s incubation of 1c1c7 cells with TDPH led to a fluorescent labeling pattern that involved only the plasma membrane (Fig. 3A). A subsequent 30 min ‘chase’ in fresh medium at 37 °C led to migration of the TDPH label into the cytoplasm and the appearance of some small punctate cytoplasmic fluorescent structures (Fig. 3B). A similar effect was seen when the ‘chase’ was carried out in the presence of 20 nM wortmannin: plasma membrane fluorescence decreased while cytoplasmic fluorescence increased (Fig. 3C). However, in the wortmannin-treated cultures there were far fewer small punctate fluorescent structures, and a dramatic increase in numbers of large vacuoles whose membranes were highly fluorescent (Fig. 3C,D). This result is consistent with the proposal that the wortmannin-induced vacuoles are derived from plasma membrane material that has undergone endocytosis.
Fig. 3.
Plasma membrane origin of wortmannin-induced vacuoles. 1c1c7 cells were treated with TDPH for 30 s, the medium replaced, and the cells imaged immediately (A) or 30 min later (B) by phase-contrast (left column) and fluorescence (right column) microscopy. For panel C, cells were treated with TDPH for 30 s, then placed in fresh medium containing 20 nM wortmannin for 30 min prior to imaging. Panel D represents an enlargement of a portion of the cell culture shown in panel C. To examine the effect of PDT on TDPH distribution, cells were given an LD30 PDT dose with NPe6, then exposed to TDPH for 30 s followed by a 30 min chase in fresh medium (Panel E). In other studies, the interval between irradiation and the TDPH treatment was expanded to 4 h (panel F) and 16 h (panel G). A significant partition of TDPH to the cytoplasm only occurred with the 16 h interval. To demonstrate effects of the 4–16 h time frame on wortmannin responsiveness, we treated cells with an LD30 PDT dose followed by exposure to 20 nM wortmannin 4 h (panel H) or 16 h (panel I) later. In only the latter culture were vacuoles detected. White bars in panels A and D = 20 μm.
Reduced labeling of the cytoplasm by TDPH after a 30 min chase was observed if cultures were first incubated with NPe6 and then irradiated with an LD30 light dose before a 30 s incubation in TDPH (Fig. 3E). This result was not significantly different if the interval between PDT and the TDPH exposure + chase occurred 4 h after irradiation (Fig. 3F). If this interval was increased to 16 h, there was a redistribution of TDPH from plasma membrane to the cytoplasm (Fig. 3G).
The ability of wortmannin to induce vacuole formation after an LD30 PDT dose was also examined. When cells were exposed to 20 nM wortmannin for 30 min, 4 h after PDT, <10% of the cells showed any evidence of vacuole formation, and only 2–3 vacuoles per cell were observed (Fig. 3H). This is far below the level of vacuolization produced by wortmannin in control cells (e.g., Fig. 3 panel C or Fig. 1 panel C). Prolonging the interval between PDT and treatment with wortmannin to 16 h removed the impairment to wortmannin-induced vacuolization (Fig. 3I). In this case, >90% of the cells were vacuolated, with >10 vacuoles per cell.
Other studies on PDT and endocytosis
Berg’s group reported on a possibly related process termed ‘photochemical internalization’ (PCI) that uses PDT to target endosomes in order to facilitate therapeutic delivery.14–16 PCI employs photosensitizers that localize in endosomes. Irradiation then results in an intracellular release of chemotherapeutic agents that had been sequestered in endosomes following their entry into cells by endocytosis. The protocol adopted involved irradiation after endocytosis of the photosensitizer + chemotherapeutic agents.15,16 Any interference with the endocytic process derived from subsequent photodamage would therefore not interfere with accumulation of either the photosensitizer or the chemotherapeutic agent.
Viability data
Using a 5 day MTT17 assay as described below, we determined LD30 and LD90 PDT doses for three photosensitizing agents. Light bandwidths and doses employed were: NPe6 (660 ± 10 nm) LD10 = 15 mJ cm−2; LD30 = 30 mJ cm−2; LD90 = 145 mJ cm−2. BPD (690 ± 10 nm) LD30 = 70 mJ cm−2, LD90 = 120 mJ cm−2. mTHPC (650 ± 10 nm) LD30 = 30 mJ cm−2, LD90 = 55 mJ cm−2. These results did not significantly differ from data obtained from clonogenic assays.
Relationship between vacuole formation and lysosomal photodamage
The lysosomal photosensitizer NPe6 is phototoxic because of the induction of lysosomal membrane permeabilization (LMP) followed by the release of proteases that can activate the mitochondrial apoptotic pathway.18,19 Lysosomal photodamage was monitored by following patterns of Lysosensor Green (LSG) fluorescence. Fig. 4 compares phase-contrast and fluorescence images of control vs. irradiated cultures. Panels A and B illustrate the punctate lysosomal localization of LSG in control cells. Panels C and D show a similar punctate pattern for NPe6 in control cells. Although the studies shown in Fig. 4A–D do not directly demonstrate labeling of the same structures by LSG and NPe6, we have shown such a co-localization in a prior study.18
Fig. 4.
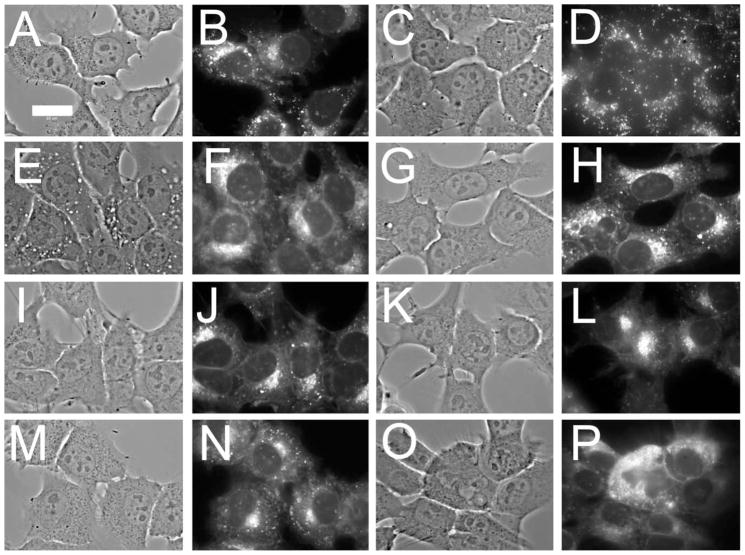
PDT-induced lysosomal photodamage and suppression of wortmannin-induced vacuolization in cells photosensitized with NPe6. Each pair of images represents a phase-contrast and a LysoSensor Green fluorescence image, except for panel D that demonstrates the NPe6 labeling pattern. Treatments are as follows: A,B = control cultures; C,D = cultures loaded with 40 μM NPe6; E,F = Cultures treated with 20 nM wortmannin for 30 min. The remaining panels involved cultures that were photosensitized with NPe6, irradiated with different light doses, then incubated with 20 nM wortmannin for 30 min prior to microscopy. The light doses were: G,H = 15 mJ cm−2; I,J = 30 mJ cm−2; K,L = 45 mJ cm−2; M,N = 90 mJ cm−2; O,P = 180 mJ cm−2. In all studies LSG was added for the final 10 min prior to imaging. All exposures for image acquisition were 40 ms except for panel D = 2000 ms. White bar in panel A = 20 μm.
Treatment of 1c1c7 cultures with 20 nM wortmannin induced vacuole formation (Fig. 4E) along with some loss of the punctate LSG labeling pattern observed in controls (Fig. 4F). Irradiation of NPe6-sensitized cultures with a non-cytotoxic 7.5 mJ cm−2 light dose affected neither the patterns nor the intensity of LSG fluorescence, nor was the ability of wortmannin to induce vacuolization altered (not shown). Wortmannin-induced vacuolization was, however, strongly suppressed if NPe6-photosensitized cultures were irradiated with light doses ≥15 mJ cm−2 (Fig. 4G). Indeed, a light dose of 15 mJ cm−2 was as effective (Fig. 4G) as 30 mJ cm−2 (Fig. 4I), 45 mJ cm−2 (Fig. 4K), 90 mJ cm−2 (Fig. 4M) or 180 mJ cm−2 (Fig. 4O) in suppressing wortmannin-induced vacuole formation. Moreover, the ability of NPe6 PDT to suppress the effect of wortmannin was independent of effects on LSG fluorescence. Suppression of vacuolization occurred at light doses that had no effect (Fig. 4H and J), or that clearly disrupted LSG labeling intensity and patterns (Fig. 4P). Since light doses of 15, 30 and 145 mJ cm−2 represent ~LD10, LD30 and LD90 conditions, respectively, the ability of NPe6-PDT to suppress wortmannin-induced formation of cytoplasmic vacuoles did not quantitatively correlate with either disruption of lysosomes or induction of phototoxicity.
Lysosomal proteases and vacuolization
We employed two additional approaches to demonstrate that the suppression of wortmannin-induced vacuolization by NPe6 PDT was not a consequence of released lysosomal proteases. First, we took advantage of our previous findings that pretreatment of 1c1c7 cultures with 3-O-methyl sphingomyelin (3-O-MeSM) stabilized late endosomes/lysosomes and prevented LMP-induced by NPe6-catalyzed photodamage.19 A control culture and cells treated with an LD30 PDT dose with NPe6 are shown in Fig. 5, panels A and B. Panel C illustrates the result of exposure to wortmannin and panel D shows the ability of this PDT dose to prevent vacuole formation. Exposure of cultures to 50 μM 3-O-MeSM alone, or in combination with NPe6 PDT, did not affect 1c1c7 morphology (Fig. 5E and F). Pretreatment with 3-O-MeSM did not suppress wortmannin-induced vacuolization (Fig. 5G), nor the ability of NPe6 PDT to inhibit wortmannin-induced vacuole formation (see Fig. 5H).
Fig. 5.
Released proteases are not responsible for NPe6 PDT suppression of wortmannin-induced vacuolization. 1c1c7 cells were either untreated (left column) or given an LD30 PDT dose using NPe6 (right column). A = control culture; (B) cells imaged 60 min after an LD30 PDT dose using NPe6; (C) cells imaged 30 min after treatment with 20 nM wortmannin; (D) cells given an LD30 PDT dose with NPe6 30 min prior to the addition of wortmannin, then incubated for 30 min. (E) Cells imaged after a 90 min exposure to 50 μM 3-O-MeSM; (F) cells treated with NPe6 + 50 μM 3-O-MeSM for 60 min, irradiated and imaged 30 min later. (G) Cells treated with 50 μM 3-O-MeSM for 1 h prior to addition of 20 nM wortmannin and imaged 30 min later. (H) Cells treated with 50 μM 3-O-MeSM for 60 min prior to irradiation and then treated with wortmannin for 30 min. (I,J) Treatments were similar to those described in panels G and H except that 10 μM E64d was substituted for 3-O-MeSM. White bar in panel A = 20 μm.
Second, the mechanism-based cysteine protease inhibitor E64d has been widely used to implicate the role of released lysosomal cysteine proteases in a variety of physiological processes. Pretreatment with E64d had no effect on the ability of wortmannin to induce vacuolization (Fig. 5I), nor did it inhibit the ability of NPe6 PDT to suppress wortmannin-induced vacuolization (Fig. 5J). These results complement the LSG studies, and indicate that the effects of NPe6-PDT do not derive from overt lysosomal photodamage. Rather, they represent an unrecognized consequence of very subtle lysosomal photodamage, or the effects on a hitherto unidentified additional target.
Effect of other PDT targets on wortmannin-induced vacuole formation
Fig. 6 and Table 1 provide a comparison of photosensitizers that target different organelles with regard to PDT-induced suppression of wortmannin-induced vacuole formation. These comparisons were carried out using LD30 and LD90 PDT doses. With the lysosomal photosensitizer NPe6, either PDT condition effectively suppressed wortmannin-induced vacuole formation (compare Fig. 6B with C and D). With BPD, an agent that preferentially targets mitochondria,20 an LD30 PDT dose appeared to have a slight inhibitory effect on vacuole formation (Fig. 6E), but this was not statistically significant, whereas, an LD90 PDT dose abolished vacuole formation (Fig. 6F). In the case of mTHPC, an agent that targets the ER,21 no suppression of wortmannin-induced vacuolization occurred at either an LD30 (Fig. 6G) or an LD90 PDT dose (Fig. 6H). An analysis of these data is shown in Table 1. As a control, we determined the effects of photodamage from BPD or mTHPC alone. We had previously observed extensive vacuolization of 1c1c7 cells within 2–3 h after photodamage induced by mTHPC.2 No vacuoles were, however, observed in cultures within 30 min of treatment with LD90 PDT doses of either BPD or mTHPC (Fig. 6I and J, respectively).
Fig. 6.
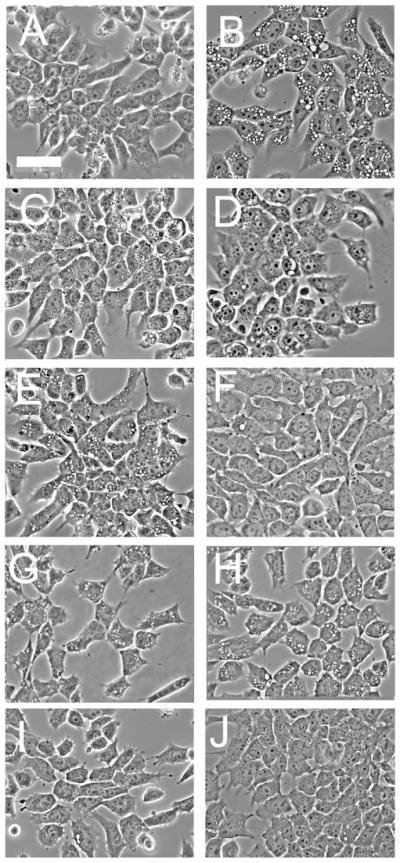
Inhibition of wortmannin-induced vacuole formation as a function of the PDT target. (A) A control culture; (B) effect of a 30 min exposure to 20 nM wortmannin; (C,D) cells given an LD30 PDT dose (C) or an LD90 (D) dose using NPe6 prior to a 30 min exposure to wortmannin. (E,F) Cells treated with an LD30 (E) or an LD90 (F) PDT dose using BPD prior to exposure to 20 nM wortmannin for 30 min. (G,H) Cells were given an LD30 (G) or an LD90 (H) PDT dose with mTHPC before a 30 min exposure to 20 nM wortmannin. (I,J) Cells were exposed to an LD90 PDT dose with BPD (I) or mTHPC (J) and then incubated for 30 min. White bar in panel A = 50 μm.
Table 1.
Vacuole formation in control vs. photodamaged cells 30 min after incubation with 20 nM wortmannin
| Conditions | Vacuoles per cella |
|---|---|
| Controls | 0 |
| Wortmannin alone | 19 ± 8.3 |
| NPe6 (LD30), wortmannin | 0 |
| BPD (LD30), wortmannin | 12.3 ± 2.3 |
| BPD (LD90), wortmannin | 0 |
| mTHPC (LD30), wortmannin | 12.2 ± 6.4 |
| mTHPC (LD90), wortmannin | 13.6 ± 8.2 |
Data represent mean ± SD of 10–15 cells per treatment group. A one way Anova analysis of variance indicated that PDT doses of LD30 (BPD) or LD30/90 (mTHPC) did not statistically (P < 0.05) reduce the number of vacuoles per cell compared to wortmannin treatment alone.
The studies reported in Fig. 6 raise three questions. Do PDT effects on endocytosis uniformly vary with the target of the photosensitizer (e.g., lysosomes vs. mitochondria vs. ER)? What is the nature of the lysosomal photodamage that initiates those processes that suppress endocytosis? What processes are involved in mediating the suppression? These issues are currently being investigated. In studies reported in preliminary form,22 we reported that wortmannin-induced vacuole formation in the murine P388 leukemia was antagonized by photodamage to any of the three sites mentioned above: lysosomes, mitochondria or the ER.
There are numerous types of plasma membrane-associated endocytic processes, e.g., phagocytosis, pinocytosis, receptor-mediated endocytosis. The endocytic process observed in our studies most likely reflects events associated with the normal turnover of the plasma membrane, which is a constitutive and fairly rapid, dynamic process. At issue is whether NPe6 PDT affects other endocytic processes such as receptor-mediated endocytosis. We also do not know whether photodamage, e.g., with NPe6, also affects exocytosis (e.g., the fusion of cytoplasmic vacuoles/vesicles with the plasma membrane). Answers to such questions are clearly relevant to growth factor and death receptor signaling, cancer cell invasion, and mechanisms of infectious diseases. It is also important to consider that some examples of receptor-mediated endocytosis, e.g., internalization of the epidermal growth factor receptor, are not dependant on PI-3 K activity.23
Experimental
Drugs and chemicals
Benzoporphyrin derivative (Verteporfin, BPD) was purchased from VWR (West Chester, PA, USA). Frontier Scientific (Logan, UT, USA) provided m-tetrahydroxyphenyl chlorin (mTHPC). Stock solutions of these agents were prepared in DMSO. N-Aspartyl chlorine e6 (NPe6) was provided by Prof. Kevin M. Smith, Louisiana State University, Baton Rouge, LA, USA. Solutions were prepared in 10 mM NaHCO3. Amino acids and tissue culture media were purchased from Sigma–Aldrich, St. Louis, MO, USA. Fluorescent probes were obtained from Invitrogen/Molecular Probes, Eugene, OR, USA. Other chemicals were provided by Fisher Scientific Co. (Chicago, IL, USA) and Sigma–Aldrich (St. Louis, MO, USA).
Cells and cell culture
The conditions and media used for the culturing of the murine hepatoma Hepa 1c1c7 cell line have been reported,19 as has the preparation of a sub-line transfected with GFP-LC3 to aid in the detection of autophagy.2 Short-term experiments were conducted using growth medium with 20 mM HEPES (pH 7.4) replacing NaHCO3 so that a neutral pH can be maintained in the absence of CO2.
PDT protocols
1c1c7 cells cultured on 1 cm glass coverslips were incubated with 0.5 μM mTHPC for 16 h, or with 0.5 μM BPD or 40 μM NPe6 for 60 min, all at 37 °C. The medium was then replaced and the cells were irradiated. The length of time of irradiation was adjusted to yield the desired loss of viability as determined by MTT assay. The light source was a 600 W quartz-halogen lamp with IR radiation attenuated by a 10 cm layer of water. Irradiation was carried out at 690 ± 10 nm (BPD) or 660 ± 10 nm (NPe6, mTHPC) as defined by the use of interference filters (Oriel, Stratford, CT, USA).
Viability
1c1c7 cells were sparsely plated on 35 mm dishes. After 24 h, cultures were incubated with photosensitizing agents for the times specified above in PDT protocol. At the end of the loading period the medium was replaced and the cells were irradiated using specified light doses. The dishes were then returned to a CO2 incubator and allowed to grow until the controls were nearly confluent (5 days). Cell survival was assessed by an MTT assay17 or, alternatively, by colony counting using an Oxford Optronix GelCount device. All such experiments were carried out in triplicate. The results obtained with the MTT assay (reported here) did not significantly differ from those obtained using clonogenic assays.
Wortmannin-induced vacuole formation
Vacuolization of 1c1c7 cells was achieved by treatment of cultures with 20 nM wortmannin for 30 min prior to visual analyses. In PDT studies cultures were treated with wortmannin after irradiation (unless stated differently in the text), and viewed 30 min later.
Phase-contrast, fluorescence and electron microscopy
Images were acquired with a Nikon E-600 microscope using a Photometrics Coolsnap HQ CCD camera. NPe6 fluorescence was determined using 400–450 nm excitation and 590–700 nm emission. Late endosomes/lysosomes were identified by incubating cultures with 2 μM LysoSensor Green (LSG) for 10 min at 37 °C. LSG fluorescence at 500–550 nm was measured using 470 ± 20 nm excitation.
Incubations with 1 μM TDPH, a label for hydrophobic membrane loci,12,13 were carried out at 37 °C for 30 s. Thereafter, the cells were washed with PBS and immediately examined by microscopy. Alternatively, the medium was replaced and the cells incubated for 30 min at 37 °C to determine whether TDPH could be chased from the plasma membrane to intracellular loci. In the case of PDT studies, cultures were first photosensitized and irradiated, labeled with TDPH and then placed in fresh media for 30 min. TDPH fluorescence at 430 ± 20 nm was monitored using 360 ± 20 nm excitation.
For electron microscopy, cells were trypsinized, fixed with glutaraldehyde and osmium tetroxide, treated with uranyl acetate + lead citrate for enhanced protein and lipid staining, and then dehydrated in ethanol. The cell pellets were embedded in epon resin and cut with an ultramicrotome to a 70 nm thickness before viewing.
Conclusions
The current study demonstrates that a minimal degree of photodamage with an agent that primarily targets lysosomes/endosomes can suppress plasma membrane endocytosis in the murine 1c1c7 hepatoma cell line. Mitochondrial photodamage was less effective in this regard while targeting the ER had no detectable effect. The role of impairment of membrane trafficking in the overall response to photodynamic therapy remains to be determined.
Acknowledgments
These studies were supported by grant CA 23378 from the NIH. Mr Price is supported by NIH grant GM-058905-11. Ann Marie Santiago provided excellent technical assistance during the course of this work. We thank Dr James Hatfield, Department of Pathology at the John Dingell VA Hospital, for electron microscopy.
Abbreviations
- BPD
Benzoporphyrin derivative
- E64d
2S,3S-trans-(ethoxycarbonyloxirane-2-carbonyl)-L-leucine-(3-methylbutyl) amide (an inhibitor of calpain and cysteine protease activity)
- ER
Endoplasmic reticulum
- LDxx
PDT dose that reduces viability to xx% of controls
- LMP
Lysosomal membrane permeabilization
- LSG
LysoSensor Green
- mTHPC
meso-Tetrahydroxyphenylporphine
- NPe6
N-Aspartyl chlorine e6
- 3-O-MeSM
3-O-Methyl sphingomyelin
- PCI
Photochemical internalization
- PDT
Photodynamic therapy
- PI
Phosphatidyl inositol
- TDPH
1-[4-(Trimethylamino)phenyl]-6-phenylhexa-1,3,5-triene
References
- 1.Dougherty TJ, Gomer CJ, Henderson BW, Jori G, Kessel D, Korbelik M, Moan J, Peng Q. Photodynamic therapy. J Natl Cancer Inst. 1998;90:889–905. doi: 10.1093/jnci/90.12.889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Reiners JJ, Jr, Agostinis P, Berg K, Oleinick NL, Kessel D. Assessing autophagy in the context of photodynamic therapy. Autophagy. 2010;6:7–18. doi: 10.4161/auto.6.1.10220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Oleinick NL, Morris RL, Belichenko I. The role of apoptosis in response to photodynamic therapy: what, where, why, and how. Photochem Photobiol Sci. 2002;1:1–21. doi: 10.1039/b108586g. [DOI] [PubMed] [Google Scholar]
- 4.Kjeken R, Mousavi SA, Brech A, Griffiths G, Berg T. Wortmannin-sensitive trafficking steps in the endocytic pathway in rat liver endothelial cells. Biochem J. 2001;357(2):497–503. doi: 10.1042/0264-6021:3570497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Reaves BJ, Bright NA, Mullock BM, Luzio JP. The effect of wortmannin on the localisation of lysosomal type I integral membrane glycoproteins suggests a role for phosphoinositide 3-kinase activity in regulating membrane traffic late in the endocytic pathway. J Cell Sci. 1996;109:749–762. doi: 10.1242/jcs.109.4.749. [DOI] [PubMed] [Google Scholar]
- 6.Shpetner H, Joly M, Hartley D, Corvera S. Potential sites of PI-3 kinase function in the endocytic pathway revealed by the PI-3 kinase inhibitor, wortmannin. J Cell Biol. 1996;132:595–605. doi: 10.1083/jcb.132.4.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bright NA, Lindsay MR, Stewart A, Luzio JP. The relationship between lumenal and limiting membranes in swollen late endocytic compartments formed after wortmannin treatment or sucrose accumulation. Traffic. 2001;2:631–642. doi: 10.1034/j.1600-0854.2001.20906.x. [DOI] [PubMed] [Google Scholar]
- 8.Scarlatti F, Maffei R, Beau I, Ghidoni R, Cagodno P. Non-canonical autophagy: an exception or an underestimated form of autophagy? Autophagy. 2008;4:1083–1085. doi: 10.4161/auto.7068. [DOI] [PubMed] [Google Scholar]
- 9.Kessel D, Vicente MG, Reiners JJ., Jr Initiation of apoptosis and autophagy by photodynamic therapy. Lasers Surg Med. 2006;38:482–488. doi: 10.1002/lsm.20334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ouédraogo G, Morlière P, Mazière C, Mazière JC, Santus R. Alteration of the endocytotic pathway by photosensitization with fluoroquinolones. Photochem Photobiol. 2000;72:458–463. doi: 10.1562/0031-8655(2000)072<0458:aotepb>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 11.Ouédraogo G, Morlière P, Bazin M, Santus R, Kratzer B, Miranda MA, Castell JV. Lysosomes are sites of fluoroquinolone photosensitization in human skin fibroblasts: a microspectro-fluorometric approach. Photochem Photobiol. 1999;70:123–129. [PubMed] [Google Scholar]
- 12.Prendergast FG, Haugland RP, Callahan PJ. 1-[4-(Trimethylamino)phenyl]-6-phenylhexa-1,3,5-triene: synthesis, fluorescence properties and use as a fluorescent probe of lipid bilayers. Biochemistry. 1981;20:7333–7338. doi: 10.1021/bi00529a002. [DOI] [PubMed] [Google Scholar]
- 13.Illinger D, Kuhry JG. The kinetic aspects of intracellular fluorescence labeling with TMA-DPH support the maturation model for endocytosis in L929 cells. J Cell Biol. 1994;125:783–794. doi: 10.1083/jcb.125.4.783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lou PJ, Lai PS, Shieh MJ, Macrobert AJ, Berg K, Bown SG. Reversal of doxorubicin resistance in breast cancer cells by photochemical internalization. Int J Cancer. 2006;119:2692–2698. doi: 10.1002/ijc.22098. [DOI] [PubMed] [Google Scholar]
- 15.Norum OJ, Selbo PK, Weyergang A, Giercksky KE, Berg K. Photochemical internalization (PCI) in cancer therapy: from bench towards bedside medicine. J Photochem Photobiol, B. 2009;96:83–92. doi: 10.1016/j.jphotobiol.2009.04.012. [DOI] [PubMed] [Google Scholar]
- 16.Prasmickaite L, Høgset A, Selbo PK, Engesaeter BØ, Hellum M, Berg K. Photochemical disruption of endocytic vesicles before delivery of drugs: a new strategy for cancer therapy. Br J Cancer. 2002;86:652–657. doi: 10.1038/sj.bjc.6600138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Twentyman PR, Luscombe M. A study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br J Cancer. 1987;56:279–285. doi: 10.1038/bjc.1987.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reiners JJ, Jr, Caruso JA, Mathieu PA, Chelladurai B, Yin XH, Kessel D. Release of cytochrome c and activation of pro-caspase-9 following lysosomal photodamage involves Bid cleavage. Cell Death Differ. 2002;9:934–944. doi: 10.1038/sj.cdd.4401048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Caruso JA, Mathieu PA, Reiners JJ., Jr Sphingomyelins suppress the targeted disruption of lysosomes/endosomes by the photosensitizer NPe6 during photodynamic therapy. Biochem J. 2005;392:325–334. doi: 10.1042/BJ20050313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Peng TI, Chang CJ, Guo MJ, Wang YH, Yu JS, Wu HY, Jou MJ. Mitochondrion-targeted photosensitizer enhances the photodynamic effect-induced mitochondrial dysfunction and apoptosis. Ann N Y Acad Sci. 2005;1042:419–428. doi: 10.1196/annals.1338.035. [DOI] [PubMed] [Google Scholar]
- 21.Teiten MH, Bezdetnaya L, Morlière P, Santus R, Guillemin F. Endoplasmic reticulum and Golgi apparatus are the preferential sites of Foscan localisation in cultured tumour cells. Br J Cancer. 2003;88:146–152. doi: 10.1038/sj.bjc.6600664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kessel D. Effects of PDT on the endocytic pathway. Proc SPIE. 2010;7551:1–7. [Google Scholar]
- 23.Chen X, Wang Z. Regulation of intracellular trafficking of the EGF receptor by Rab5 in the absence of phosphatidylinositol 3-kinase activity. EMBO Rep. 2001;2:68–74. doi: 10.1093/embo-reports/kve005. [DOI] [PMC free article] [PubMed] [Google Scholar]