Abstract
BACKGROUND: Caveolin-1 is a protein that displays promotive versus preventive roles in cancer progression according to circumstances. Temozolomide (TMZ) is the standard chemotherapeutic to treat glioma patients. The present work aims to characterizeTMZ-induced effects on caveolin-1 expression in glioma cells. METHODS: Human astroglioma (U373 and T98G) and oligodendroglioma (Hs683) cell lines were used in vitro as well as in vivo orthotopic xenografts (Hs683 and U373) into the brains of immunocompromisedmice. In vitro TMZ-induced effects on protein expression and cellular localization were determined by Western blot analysis and on the actin cytoskeleton organization by means of immunofluorescence approaches. In vivo TMZ-induced effects in caveolin-1 expression in human glioma xenografts were monitored by means of immunohistochemistry. RESULTS: TMZ modified caveolin-1 expression and localization in vitro and in vivo after an administration schedule that slightly, if at all, impaired cell growth characteristics in vitro. Caveolin-1 by itself (at a 100-ng/ml concentration) was able to significantly reduce invasiveness (Boyden chambers) of the three human glioma cell lines. The TMZ-inducedmodification in caveolin-1 expression in flotation/raft compartments was paralleled by altered Cyr61 and β1 integrin expression, two elements that have already been reported to collaborate with caveolin-1 in regulating glioma cell biology, and all these features led to profound reorganization of the actin cytoskeleton. An experimental Src kinase inhibitor, AZD0530, almost completely antagonized the TMZ-induced modulation in caveolin-1 expression. CONCLUSION: TMZ modifies caveolin-1 expression in vitro and in vivo in glioma cells, a feature that directly affects glioma cell migration properties.
Introduction
The combination of temozolomide (TMZ) with radiotherapy has been shown to improve survival in glioblastoma (GBM) patients after surgical resection [1,2]. However, the prognosis associated with GBM patients remains dismal because i) GBMs cannot be completely removed surgically because of the highly infiltrative nature of these tumors into the brain parenchyma [1,3] and ii) GBM cells defend themselves against TMZ during chronic treatments, with the emergence of TMZ-resistant tumors observed at both the experimental [4] and the clinical [1,2] levels.
The therapeutic benefits contributed by TMZ relates to the fact that it induces double-strand DNA breaks through generation of methylguanosine [5] concomitantly with sustained autophagy-related processes [6,7], all these features ending with apoptosis in GBM cells [8]. TMZ also displays antiangiogenic effects [9]. In contrast, as mentioned above, TMZ treatment of GBMs can lead to the emergence of TMZ-resistant tumors. Fisher et al. [10] showed that treatment of human malignant glioma cells with TMZ activate stress mechanisms that include up-regulation of angiogenesis-inducing proteins, notably hypoxia-inducible factor-1α and vascular endothelial growth factor. We recently demonstrated that TMZ activates galectin-1 (a hypoxia-regulated protein [11]) expression in malignant glioma cells, a process that leads to marked proangiogenic effects [12] and the activation of chemoresistance [13]. The fact of decreasing galectin-1 expression in experimental malignant gliomas [12,13] and melanomas [14] leads in turn to increased therapeutic benefits contributed by TMZ.
Numerous hypoxia-regulated proteins are implicated in glioma biology [15,16] among which caveolin-1 [17], and we were therefore wondering whether TMZ could modify caveolin-1 expression and/or cellular localization in GBM cells.
Caveolin-1 is a 21- to 24-kDa scaffold protein and an essential constituent of caveolae, which are flask-shaped invaginations of the plasma membrane [18]. In caveolae, caveolin-1 directly interacts with several signaling molecules, including growth factor receptors (e.g., the epidermal growth factor receptors [EGFRs]), kinases (including Src), G proteins, and adhesion molecules [19,20]. Caveolin-1 contributes to the regulation of multiple cancer-associated processes, including cellular transformation, tumor growth, cell migration and metastasis, cell death and survival, multiple drug resistance, and angiogenesis [21–23]. However, both cancer growth-enhancing and growth-inhibiting effects have been reported for caveolin-1 under different conditions [21–23]. Caveolin-1 is expressed in normal and malignant glial cells [24–27] as well as in brain blood vessels [28].
The present work thus aims to i) investigate as whether TMZ is able to modify caveolin-1 expression and/or cellular localization in glioma cells, ii) characterize caveolin-1-mediated effects on glioma cell invasion, and iii) identify some signaling elements by which TMZ modifies caveolin-1 expression in detergent-insoluble floating complexes of glioma cells.
We used three GBM models. The U373 and T98G models are of astrocytic origin [29,30]. U373 GBM cells display resistance to proapoptotic stimuli [30,31] but actual sensitivity to sustained proautophagic stimuli, leading to massive cell death [32]. The Hs683 GBM model is of oligodendroglial origin [29,33] and displays sensitivity to proapoptotic drugs [30,31]. Hs683 GBM cells contain only one Notch2 gene copy per diploid genome as do oligodendrogliomas, whereas U373 GBM cells contain two copies as do astrogliomas [34]. The HS683 model might correspond to the few GBMs displaying an oligodendroglial origin [35] and/or component [36]. These three models markedly and diffusely invade the brain parenchyma when orthotopically grafted into the brains of immunocompromised mice [4,9,12,30,37].
Materials and Methods
Cell Cultures and Compounds
Hs683 (ATCC code HTB-138), U373 (ATCC code HTB-17), and T98G (ATCC code CRL-1690) human GBM cell lines were obtained from the American Type Culture Collection (ATCC, Manassas, VA) and maintained in our laboratory, as detailed previously [9,12,13,32,33,38,39].
TMZ was obtained from Schering Plough (Brussels, Belgium). Caveolin-1 recombinant protein purified on glutathione sepharose 4 Fast Flow was obtained from Abnova (Taipei, Taiwan), Nycodenz (5-(N-2,3-dihydroxypropylacetamido)-2,4,6-tri-iodo-N-N′-bis(2,3 dihydroxypropyl) isophthalamide), a nonionic iodinated gradient medium was purchased from Gentaur (Brussels, Belgium), and the EGFR kinase inhibitor (erlotinib) and Src tyrosine kinase inhibitor (AZD0530) were obtained from Selleck Chemicals Co (Shanghai, China).
Protein Extraction for Raft Resident Caveolin-1
Caveolin-1 is a part of caveolae that are Triton-insoluble complexes and are difficult to solubilize using conventional detergents found in typical lysis buffers. Therefore, flotation of detergent-insoluble proteins was performed as described by Naslavsky et al. [40].
Briefly, in all experiments, confluent cells growing in three 75-mm dishes were pelleted and lysed with 750 µl of lysis buffer (1% of TX-100 in TNE (150 mM NaCl, 25 mM Tris-HCl, and 5 mM EDTA, pH 7.5)) on ice for 30 minutes. Lysates were spun at low speed (3000 rpm, 4°C, 5 minutes) to generate a postnuclear supernatant. All subsequent steps were performed on ice. Lysates were adjusted to 35% Nycodenz by adding an equal volume of ice-cold 70% Nycodenz dissolved in TNE. One-and-a-half milliliters of the samples was loaded at the bottom of 4-ml tubes (Beckman Coulter, Analis, Suarlee, Belgium). An 8% to 25% Nycodenz linear step gradient in TNE was then overlaid above the lysate (350 µl each of 25%, 22.5%, 20%, 18%, 15%, 12%, and 8% Nycodenz). The tubes were spun at 55,000 rpm for 4 hours at 4°C in a Beckman Optima LE-80K with a SW 60 Ti rotor (Beckman Coulter). Twelve 330-µl fractions were collected from the bottom of the tube.
Protein Expression Measurements
Western blot analyses were performed to measure protein expression, as detailed previously [9,12,13,38,39]. The primary antibody incubation step was omitted as a negative control experiment. Integrity and quantity of the extracts were assessed by tubulin immunoblot analysis. Proteins were detected by Western blot analysis using the following primary antibodies: anti-caveolin-1 (1:250 dilution; BD Transduction Laboratories, Erembodegem, Belgium), antitubulin (1:3000 dilution; Abcam, Cambridge, UK), anti-Cyr61 (1:1000 dilution; Abcam, Cambridge, UK), and anti-β1 integrin (1:500 dilution; BD Transduction Laboratories). Secondary antibodies were purchased from Pierce (PerbioScience, Erembodegem, Belgium). Western blots were developed using the Pierce SuperSignal Chemiluminescence System.
Cytology
The effect of TMZ (100 µM) on actin cytoskeleton organization of GBM cells was investigated using fluorescent probes, as detailed elsewhere [39,41]. The green fluorochrome Alexa Fluor 488-conjugated phallacidin (Molecular Probes, Inc, Invitrogen, Merelbeke, Belgium) was used to label fibrillary actin, whereas the red fluorochrome Fluor 594-conjugated DNAseI (Molecular Probes, Inc) was used to label globular actin.
Invasion Assay
Invasive features of T98G, U373, and Hs683 GBM cells were assessed in vitro using the Boyden transwell invasion system (BD BioCoat Matrigel invasion chambers; BD Biosciences Discovery Labware, Bedford, MA), as detailed elsewhere [33]. We studied the effects of caveolin-1 on malignant glioma cell migration when applied to the top as opposed to the bottom of the Boyden chambers.
Immunohistochemical Procedures for Human Glioma Xenografts
We used histologic slides for caveolin-1 immunohistochemical staining in Hs683 and U373 GBM xenografts from a previous in vivo experiment, in which we demonstrated TMZ-induced antiangiogenic effects and improvements in survival [9]. In this previous experiment, immunocompromised mice bearing orthotopic Hs683 or U373 xenografts were treated (or left untreated as control) three times a week (Monday, Wednesday, and Friday) for three and five consecutive weeks, respectively, with TMZ (40 mg/kg, intravenously) [9].
The mouse brains were fixed in 4% buffered formaldehyde, routinely processed, and paraffin-embedded. Three-micrometer sections were prepared and routinely stained with hematoxylin and eosin. Two additional sections, collected on SuperFrost Plus Slides, were used for immunohistochemical analysis. Caveolin-1 immunoreactivity was studied in all cases, and the immunohistochemical reaction with the anti-caveolin-1 antibody (rabbit polyclonal antibody, diluted 1:350; Santa Cruz Biotechnology, Santa Cruz, CA) was performed in an automated immunostainer (Ventana BenchMark Auto-Stainer; Ventana Medical Systems, Tucson, AZ).
Results
Temozolomide Modulates Raft Resident Caveolin-1 Expression in Human GBM Cell Lines In Vitro
We previously determined the 50% reduction in in vitro growth (IC50) concentrations for TMZ on after 3 days of glioma cell culture with the drug, and we obtained 118 ± 3 µM for U373, 634 ± 23 µM for T98G, and 719 ± 12 µM for Hs683 GBM cells [9]. In the current experiment, U373, T98G, and Hs683 GBM cells were chronically treated in vitro with 100 µM TMZ, which was added to the culture medium each day for 7 hours (after which fresh medium replaced the medium with drugs for 17 hours) for five consecutive days. Protein analyses were performed at 3, 5, and 7 days after the fifth TMZ addition to the GBM culture medium (Figures 1 and 2). We previously showed that 5 x 100 µM TMZ treatment (with observation 7 days after the fifth TMZ administration) did not impair T98G cell population growth, slightly impaired Hs683 cell population growth, and moderately impaired U373 cell population growth [9]. Thus, the in vitro 5 x 100 µM treatment adopted here for three distinct human GBM cell lines slightly, if at all, impaired cell growth characteristics.
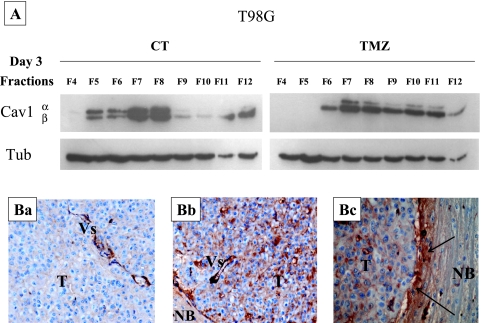
Figure 1.
Temozolomide modulates raft resident caveolin-1 expression in vitro and increases caveolin-1 expression in vivo in experimental GBMs. (A) Western blot analysis of the expression of raft resident caveolin-1 in the different flotation fractions (F4–F12) obtained from T98G GBM cells left untreated (CT) or treated with 100 µM TMZ for 7 h/d for five consecutive days. Raft resident caveolin-1 expression was measured on the third day (day 3) after the end of the TMZ treatment. (B) Caveolin-1 immunoreactivity in Hs683 GBM tumors implanted into brains of immunodeficient mice and treated (Bb and Bc) or not treated (Ba) with TMZ (40 mg/kg per os, three times per week for three consecutive weeks, with immunohistochemical analyses performed 1 week after the end of the treatment). Caveolin-1 expression was absent from the Hs683 tumor (T) left untreated (Ba), in which only the vessels (Vs) were immunoreactive for caveolin-1. Caveolin-1 expression was marked in Hs683 tumors (T) after TMZ treatment (Bb). Caveolin-1 expression in the tumor bulk (T) was associated with accumulation of caveolin-1 in the extracellular matrix around the invasive part of the tumor (black arrows in Bc). The normal brain (NB) expressed weak levels of caveolin-1 (Bc).
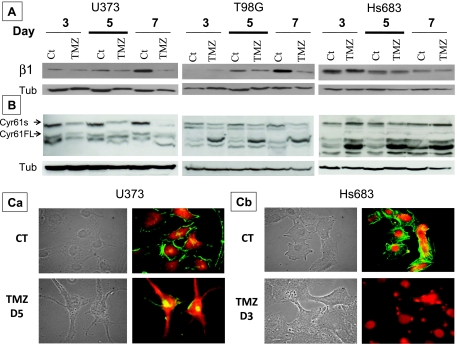
Figure 2.
Temozolomide decreases β1 integrin and modulates Cyr61 and actin cytoskeleton organization in GBM cell lines. (A) Western blot analysis of the expression of β1 integrin in U373, T98G, and Hs683 GBM cells left untreated (CT) or treated with 100 µM TMZ for 7 h/d for five consecutive days. The β1 integrin expression was measured on the third (day 3), fifth (day 5), and seventh (day 7) days after the end of the TMZ treatment. (B) Western blot analyses of the expression of Cyr61 in U373, T98G, and Hs683 GBM cell lines left untreated (CT) or treated with 100 µM TMZ using a treatment schedule identical to that described above for β1 integrin. (C) Fluorescence microscopic visualization of the actin cytoskeleton (red fluorescence shows globular (nonpolymerized) actin and green fluorescence shows fibrillary (polymerized) actin) under control conditions (CT) and 3 (TMZ D3) and 5 (TMZ D5) days after termination of TMZ (100 µM) treatment in U373 (Ca) and Hs683 (Cb) glioma cells according to a treatment schedule identical to that described above for β1 integrin.
Raft resident caveolin-1 was expressed in flotation fractions 5 to 12 of the T98G GBM cell line with the largest expression in fractions 5 to 8 (Figure 1A). TMZ treatment, 3 days after the fifth in vitro administration into the culture medium, changed the expression and therefore the localization of raft resident caveolin-1 into the different flotation compartments: caveolin-1 disappeared from fraction 5, decreased in fractions 6 to 8, and increased in fractions 9 to 11 with TMZ treatment (Figure 1A). We obtained similar results with the two other GBM cell lines (data not shown). Raft resident caveolin-1 was not expressed in fractions 1 to 4 of the three GBM cell lines (data not shown).
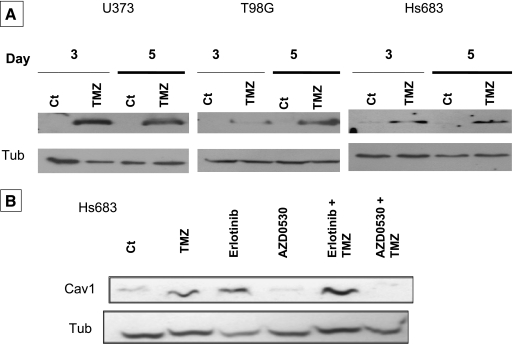
By contrast, using a classic extraction method, the three GBM cell lines did not express soluble caveolin-1 under control conditions, whereas they displayed increased intracellular soluble caveolin-1 expression 3 to 5 days after the fifth in vitro administration of TMZ into the culture medium (Figure 4A).
Figure 4.
The TMZ-induced caveolin-1 modulation is Src-dependent in Hs683 GBM cells. (A) Western blot analysis of the expression of soluble caveolin-1 in U373, T98G, and Hs683 GBM cells left untreated (CT) or treated with 100 µM TMZ for 7 h/d for five consecutive days. Soluble caveolin-1 expression was measured on the third (day 3) and fifth (day 5) days after the end of the TMZ treatment. (B) Western blot analyses of soluble caveolin-1 expression in Hs683 glioma cells treated with TMZ (100 µM) four times per week (day 1–4) for 7 h/d, the EGFR inhibitor (10 µM) (erlotinib; day 1), the Src inhibitor AZD0530 (10 µM) (day 1), and combination of the inhibitors and TMZ (+TMZ) compared with control untreated cells (Ct). Soluble caveolin-1 expression was measured on day 5.
Temozolomide Increases Caveolin-1 Expression in Human GBM Xenografts In Vivo
Chronic TMZ treatment modified raft resident caveolin-1 expression in vitro in T98G GBM cells (Figure 1A) and in Hs683 and U373 (data not shown) and in vivo in Hs683 (Figure 1B) and in U373 (data not shown) GBM xenografts. Immunohistochemical analyses further revealed that the highest TMZ-induced increases in caveolin-1 expression was observed in the extracellular matrix, particularly around the invasive areas (the black arrows in Figure 1Bc) of the tumor (T in Figure 1Bc) compared with normal brain parenchyma (NB in Figure 1Bc).
Temozolomide Decreases β1 Integrin Expression and Modulates Cyr61 Signaling and Actin Cytoskeleton Polymerization in Human GBM Cell Lines In Vitro
Caveolin-1 is part of the signaling pathways controlling integrin function and expression, as previously demonstrated for α5β1 integrins [42]. Caveolin-1 has also been identified as a component in cysteinerich 61 (Cyr61) integrin signaling [43]. Cyr61 is a matricellular protein, and expression of this protein is known to correlate with poor prognosis in GBM patients [44]. TMZ-induced effects on α5β1 integrin and Cyr61 expression were investigated in Hs683, U373, and T98G GBM cells in vitro (Figure 2). We again adopted a regimen of daily in vitro TMZ (100 µM) treatment for 7 h/d over five consecutive days.
TMZ did not modify α5 integrin expression patterns in these three GBM cell lines (data not shown). In contrast, we found that TMZ significantly decreased expression levels of β1 integrin in the GBM cell lines of astroglial origin (i.e., U373 and T98G) 5 or 7 days after the fifth in vitro administration of TMZ (Figure 2A). In addition, β1 integrin expression levels progressively increased over time in these two GBM models of astroglial origin (Figure 2A). In contrast, β1 integrin expression levels progressively decreased over time in the Hs683 GBM model of oligodendroglial origin, and TMZ failed to further decrease levels over control (Figure 2A). We previously demonstrated marked differences in β integrin expression levels between astrogliomas and oligodendrogliomas, and between U373 and T98G versus Hs683 GBM cells [29]. Whereas U373 and T98G GBM cells display marked β1 expression, as do astrogliomas [29,45], Hs683 GBM cells display high levels of β4 integrin, as do oligodendrogliomas [29,45]. The differences in β integrin expression between U373 and T98G GBM cells with an astroglioma origin versus Hs683 GBM cells with an oligodendroglioma origin [29] could explain, at least partly, why TMZ did not induce similar patterns of β1 integrin expression in U373, T98G, and Hs683 GBM cells (Figure 2A).
The same features have been observed with respect to Cyr61. Indeed, chronic in vitro TMZ (100 µM) treatment for 7 h/d for five consecutive days significantly decreased expression levels of the secreted form of Cyr61 in U373 and T98G GBM cell lines of astrocytic origin, whereas this treatment had no effect or even slightly increased expression of the Cyr61 secreted form in the Hs683 GBM cell line of oligodendroglial origin (Figure 2B). The reasons for the differences in the full-length Cyr61 observed with TMZ treatment are not yet understood for the moment (Figure 2B).
Malignant glioma adhesion depends on various integrins [29,45–47], and modifications in integrin signaling and/or expression can modify actin cytoskeleton organization [48]. Figure 2C reveals that TMZ treatment significantly depolymerized actin stress fibers not only in U373 and T98G cells (data not shown) but also in Hs683 GBM cells. This effect was sustained even 5 days after termination of TMZ treatment (Figure 2Ca). The finding that TMZ depolymerized the actin cytoskeleton in Hs683 oligodendroglioma cells but did not change β1 integrin expression may indicate the involvement of other β integrins [29,45–47], expression of which may be modified by TMZ treatment.
Extracellular Caveolin-1 Significantly Decreases U373, T98G, and Hs683 GBM Cell Invasiveness In Vitro
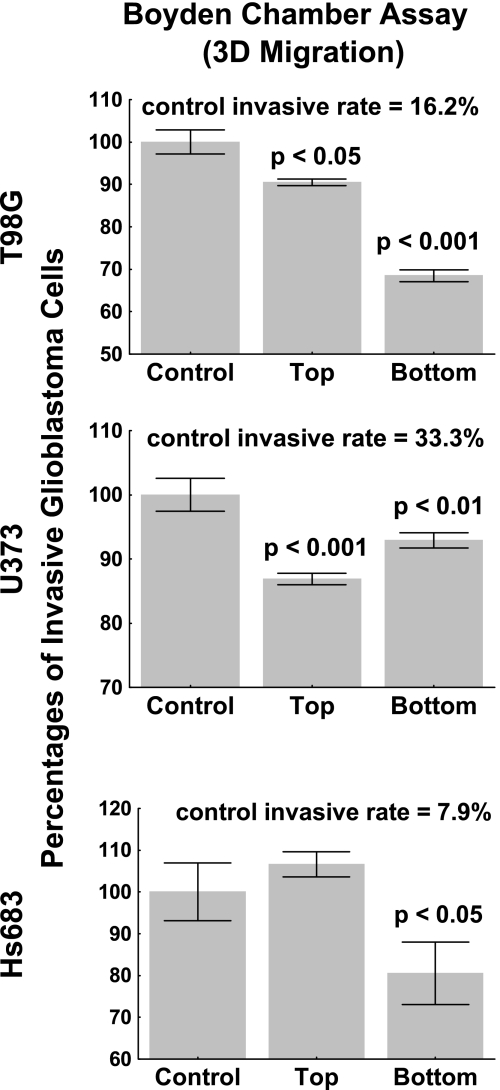
Caveolin-1 is secreted by androgen-insensitive prostate cancer cells and is detected in the serum of patients with prostate cancer [49,50]. The highest reported serum level of caveolin-1 was approximately 100 ng/ml [49]. We therefore used 10 and 100 ng/ml caveolin-1 to investigate whether caveolin-1 can modify invasiveness of glioma cells cultured in Boyden chambers in vitro. As shown in Figure 3, 100 ng/ml caveolin-1 applied to the top of the Boyden chambers significantly decreased the invasiveness levels in the two GBM models of astrocytic origin (i.e., U373 and T98G) but not in the Hs683 GBM model of oligodendroglial origin. In contrast, caveolin-1 added to the bottom of the Boyden chamber reduced invasiveness in the three models, suggesting that caveolin-1 exerted repulsive, or nonpermissive, effects on glioma cell migration and invasion. Lower concentrations (10 ng/ml) of caveolin-1 did not modify glioma cell invasiveness (data not shown). Altogether, these data suggest that part of the therapeutic benefits contributed by TMZ treatment in GBM patients could relate to TMZ-induced modification in caveolin-1 expression, a feature that in turn seems to lower GBM cell invasiveness.
Figure 3.
Extracellular caveolin-1 decreases GBM cell line invasion. Invasion was evaluated using Matrigel Boyden chambers. The number of invasive cells was counted in 10 fields per chamber (the chamber surface includes 30 fields), and the experiment was conducted in triplicate. The mean of the 10 values was used to calculate the percentage of invasive cells in the population seeded, and the control invasion rate shown on each graph was reported to be 100%. The effects of 100 ng/ml recombinant caveolin-1 were examined both when the protein was added in the upper (“Top”; promigratory or antimigratory factor) or the lower (“Bottom”; chemoattractant vs chemorepulsive factor) compartments of the chamber. Results are expressed as mean of the triplicate ± SE.
Temozolomide-Induced Caveolin-1 Modulation in GBM Cells Is at Least Partly Mediated through Src Signaling
Caveolin-1 actively interacts with Src and EGFR [19,20], and several Src and EGFR inhibitors are currently assayed in clinical trials as potential treatments for recurrent GBM patients (clinicaltrial.gov) [51]. We therefore investigated the effects of inhibitors of EGFR kinase (erlotinib) and Src (AZD0530) on TMZ-induced intracellular soluble caveolin-1 expression in Hs683 GBM cells. Figure 4 shows that AZD0530 prevented TMZ-induced soluble caveolin-1 expression but did not change basal levels of soluble caveolin-1 in Hs683 GBM cells. In contrast, erlotinib increased soluble caveolin-1 expression to a similar degree as TMZ in these Hs683 GBM cells (Figure 4). These data suggest that Src could be a signaling element for TMZ when increasing intracellular soluble caveolin-1 expression in GBM cells. In contrast, EGFR seems to negatively regulate soluble caveolin-1 expression in GBM cells and its inhibition leads to increased soluble caveolin-1 expression, a feature that becomes additive to the TMZ-induced effects on soluble caveolin-1 expression in these Hs683 GBM cells (Figure 4). Reconciling the data from Figure 3 with those from Figure 4 suggests that a treatment combining an anti-EGFR compound with TMZ would reinforce an increase in soluble caveolin-1 expression (Figure 4), which in turn would decrease GBM cell invasiveness (Figure 3).
Discussion
GBM cells develop TMZ resistance during long-term treatment, and this resistance is partly mediated by O(6)-methylguanine-DNA methyltransferase (MGMT) [5,52]. The MGMT promoter methylation status [53], but not anti-MGMT immunohistochemistry [54], can be used to predict responses to TMZ treatment in GBM patients. Identification of additional biomarkers that would predict distinct clinical responses at the single patient level within MGMT-positive and -negative groups of GBM patients will help in tailoring a drug regimen to the specific biochemistry and particular vulnerabilities of each patient's tumor, thereby increasing effectiveness and avoiding unnecessary toxicities [32,51]. We already suggested such a potential role for galectin-1 [55], and the current study investigated such a potential role for caveolin-1. Data from the present study show that TMZ-treated GBM cells differently express raft resident and soluble caveolin-1 compared with untreated GBM cells.
Evidence for a role of caveolin-1 in cancer biology remains controversial [56–61]. Even within the specific group of gliomas, immunohistochemical caveolin-1 expression varies among glioma subgroups [25,26].
Caveolin-1 is upregulated in cancer cells facing adverse events. Specifically, caveolin-1 induction in tumor cells and tumor endothelial cells after radiation was previously shown to be responsible for the acquisition of a radio-resistant phenotype [62,63]. Conversely, HDAC inhibitors, proteasome inhibitors, and vascular endothelial growth factor receptor inhibitors, which are in clinical trials for GBM [51], were shown to reduce caveolin-1 levels. Moreover, Palozza et al. [64] recently demonstrated that β-carotene acts as a growth-inhibitory agent in caveolin-1-positive human colon and prostate cancer cells but not in caveolin-1-negative cells. This finding suggests that modifications in caveolin-1 expression in glioma cells treated with TMZ could also modify the response of cells to future treatments.
The fact remains that as long as cancer cell biology is taken into account, caveolin-1-related roles seem to be tightly dependent on the cancer cell type may be because various cancer cell types express various activated signaling pathways. Data from the present study clearly evidence opposite roles for EGFR and Src on TMZ-induced modulation in caveolin-1 expression (Figure 4).
Accumulating experimental evidences indicate that caveolin-1 regulates signal transduction-associated proteins in caveolae [19,20,22]. For example, caveolin-1 modifies expression levels of Src, EGFR, endothelial nitric oxide synthase, G protein α and subunits, and H-Ras [65]. Several groups have provided rationale for interaction between caveolin-1 and β1 integrin [62,66,67]. Caveolin-1 promotes Fyn-dependent shc phosphorylation and MAPK activation in response to integrin ligation through association with β1 integrins and the Src-related kinase Fyn [66]. This would strengthen the rationale to explore these pathways in gliomas. Conversely, Src and other kinases phosphorylate caveolin-1 on Tyr14 in response to various stimuli, and this phosphorylation is crucial for caveolin-1 functions [48]. The interaction at Tyr14 contributes to modifications in the actin cytoskeleton organization [48,68].
By manipulating the levels of caveolin-1 in a GBM cell line (U87MG), Martin et al. [42] showed that reduced levels of caveolin-1 shifted cells toward a more aggressive phenotype, whereas forced expression of caveolin-1 slowed proliferation, clonogenicity and invasion. The current data perfectly fit in with those data reported by Martin et al. [42] because we show here that caveolin-1 reduces GBM cell invasion (Figure 3).
In addition, it is already well documented that GBM microenvironment directly affects GBM cell biology [15,16,69]. Our current data suggest that GBM microenvironment may also be a major determinant of caveolin-1-mediated roles in cancer cell biology according to the fact that TMZ-mediated increased caveolin-1 expression GBM xenografts mainly occur in the glioma microenvironment (Figure 1Bc).
In conclusion, TMZ modified caveolin-1 expression in vitro and in vivo in human GBM models of astrocytic versus oligodendroglial origin, and this effect seemed to be mediated partly through activation of Src signaling but not of EGFR signaling. In contrast, blocking EGFR also leads to increases in soluble caveolin-1 expression in GBM cells. TMZ-induced modification in caveolin-1 expression in GBM cells was paralleled by the decrease in β1 integrin expression and disorganization of the actin cytoskeleton. Caveolin-1 decreases GBM cell invasion.
Footnotes
The present work was funded by grants awarded by the Fonds National de la Recherche Scientifique (Brussels, Belgium).
References
- 1.Lefranc F, Sadeghi N, Camby I, Metens T, Dewitte O, Kiss R. Present and potential future issues in glioblastoma treatment. Expert Rev Anticancer Ther. 2006;6:719–732. doi: 10.1586/14737140.6.5.719. [DOI] [PubMed] [Google Scholar]
- 2.Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. [DOI] [PubMed] [Google Scholar]
- 3.Lefranc F, Brotchi J, Kiss R. Possible future issues in the treatment of glioblastomas: special emphasis on cell migration and the resistance of migrating glioblastoma cells to apoptosis. J Clin Oncol. 2005;23:2411–2422. doi: 10.1200/JCO.2005.03.089. [DOI] [PubMed] [Google Scholar]
- 4.Le Calvé B, Rynkowski M, Le Mercier M, Bruyère C, Lonez C, Gras T, Haibe-Kains B, Bontempi G, Decaestecker C, Ruysschaert JM, et al. Long term in vitro treatment of human glioblastoma cells with temozolomide confers resistance in vivo through up-regulation of GLUT transporter and aldo-keto reductase enzyme AKR1C expression. Neoplasia. 2010;12:727–739. doi: 10.1593/neo.10526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hegi ME, Liu L, Herman JG, Stupp R, Wick W, Weller M, Mehta MP, Gilbert MR. Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMTactivity. J Clin Oncol. 2008;26:4189–4199. doi: 10.1200/JCO.2007.11.5964. [DOI] [PubMed] [Google Scholar]
- 6.Kanzawa T, Germano IM, Komata T, Ito H, Kondo Y, Kondo S. Role of autophagy in temozolomide-induced cytotoxicity for malignant glioma cells. Cell Death Differ. 2004;11:448–457. doi: 10.1038/sj.cdd.4401359. [DOI] [PubMed] [Google Scholar]
- 7.Katayama M, Kawaguchi T, Berger MS, Pieper RO. DNA damaging agent-induced autophagy produces a cytoprotective adenosine triphosphate surge in malignant glioma cells. Cell Death Differ. 2007;14:548–558. doi: 10.1038/sj.cdd.4402030. [DOI] [PubMed] [Google Scholar]
- 8.Roos WP, Batista LF, Naumann SC, Wick W, Weller M, Menck CF, Kaina B. Apoptosis in malignant glioma cells triggered by the temozolomide-induced DNA lesion O6-methylguanine. Oncogene. 2007;26:186–197. doi: 10.1038/sj.onc.1209785. [DOI] [PubMed] [Google Scholar]
- 9.Mathieu V, De Neve N, LeMercier M, Dewelle J, Gaussin JF, Dehoux M, Kiss R, Lefranc F. Combining bevacizumab with temozolomide increases the antitumor efficacy of temozolomide in a human glioblastoma orthotopic xenograft model. Neoplasia. 2008;10:1383–1392. doi: 10.1593/neo.08928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fisher T, Galanti G, Lavie G, Jacob-Hirsch J, Kventsel I, Zeligson S, Winkler R, Simon AJ, Amariglio N, Rechavi G, et al. Mechanisms operative in the antitumor activity of temozolomide in glioblastoma multiforme. Cancer J. 2007;13:335–344. doi: 10.1097/PPO.0b013e318157053f. [DOI] [PubMed] [Google Scholar]
- 11.Le QT, Shi G, Cao H, Nelson DW, Wang Y, Chen EY, Zhao S, Kong C, Richardson D, O'Byrne KJ, et al. Galectin-1: a link between tumor hypoxia and tumor immune privilege. J Clin Oncol. 2005;23:8932–8941. doi: 10.1200/JCO.2005.02.0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Le Mercier M, Mathieu V, Haibe-Kains B, Bontempi G, Mijatovic T, Decaestecker C, Kiss R, Lefranc F. Knocking down galectin 1 in human hs683 glioblastoma cells impairs both angiogenesis and endoplasmic reticulum stress responses. J Neuropathol Exp Neurol. 2008;67:456–469. doi: 10.1097/NEN.0b013e318170f892. [DOI] [PubMed] [Google Scholar]
- 13.Le Mercier M, Lefranc F, Mijatovic T, Debeir O, Haibe-Kains B, Bontempi G, Decaestecker C, Kiss R, Mathieu V. Evidence of galectin-1 involvement in glioma chemoresistance. Toxicol Appl Pharmacol. 2008;229:172–183. doi: 10.1016/j.taap.2008.01.009. [DOI] [PubMed] [Google Scholar]
- 14.Mathieu V, Le Mercier M, De Neve N, Sauvage S, Gras T, Roland I, Lefranc F, Kiss R. Galectin-1 knockdown increases sensitivity to temozolomide in a B16F10 mouse metastatic melanoma model. J Invest Dermatol. 2007;127:2399–2410. doi: 10.1038/sj.jid.5700869. [DOI] [PubMed] [Google Scholar]
- 15.Amberger-Murphy V. Hypoxia helps glioma to fight therapy. Curr Cancer Drug Targets. 2009;9:381–390. doi: 10.2174/156800909788166637. [DOI] [PubMed] [Google Scholar]
- 16.Oliver L, Olivier C, Marhuenda FB, Campone M, Vallette FM. Hypoxia and the malignant glioma microenvironment: regulation and implications for therapy. Curr Mol Pharmacol. 2009;2:263–284. doi: 10.2174/1874467210902030263. [DOI] [PubMed] [Google Scholar]
- 17.Mann GE, Yudilevich DL, Sobrevia L. Regulation of amino acid and glucose transporters in endothelial and smooth muscle cells. Physiol Rev. 2003;83:183–252. doi: 10.1152/physrev.00022.2002. [DOI] [PubMed] [Google Scholar]
- 18.Stan RV. Structure of caveolae. Biochim Biophys Acta. 2005;1746:334–348. doi: 10.1016/j.bbamcr.2005.08.008. [DOI] [PubMed] [Google Scholar]
- 19.Carver LA, Schnitzer JE. Caveolae: mining little caves for new cancer targets. Nat Rev Cancer. 2003;3:571–581. doi: 10.1038/nrc1146. [DOI] [PubMed] [Google Scholar]
- 20.van Deurs B, Roepstorff K, Hommelgaard AM, Sandvig K. Caveolae: anchored, multifunctional platforms in the lipid ocean. Trends Cell Biol. 2003;13:92–100. doi: 10.1016/s0962-8924(02)00039-9. [DOI] [PubMed] [Google Scholar]
- 21.Burgermeister E, Liscovitch M, Rocken C, Schmid RM, Ebert MP. Caveats of caveolin-1 in cancer progression. Cancer Lett. 2008;268:187–201. doi: 10.1016/j.canlet.2008.03.055. [DOI] [PubMed] [Google Scholar]
- 22.Goetz JG, Lajoie P, Wiseman SM, Nabi IR. Caveolin-1 in tumor progression: the good, the bad and the ugly. Cancer Metastasis Rev. 2008;27:715–735. doi: 10.1007/s10555-008-9160-9. [DOI] [PubMed] [Google Scholar]
- 23.Quest AF, Gutierrez-Pajares JL, Torres VA. Caveolin-1: an ambiguous partner in cell signalling and cancer. J Cell Mol Med. 2008;12:1130–1150. doi: 10.1111/j.1582-4934.2008.00331.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cameron P, Liu C, Smart D, Hantus ST, Fick JR, Cameron RS. Caveolin-1 expression is maintained in rat and human astroglioma cell lines. Glia. 2002;37:275–290. doi: 10.1002/glia.10036. [DOI] [PubMed] [Google Scholar]
- 25.Cassoni P, Senetta R, Castellano I, Ortolan E, Bosco M, Magnani I, Ducati A. Caveolin-1 expression is variably displayed in astroglial-derived tumors and absent in oligodendrogliomas: concrete premises for a new reliable diagnostic marker in gliomas. Am J Surg Pathol. 2007;31:760–769. doi: 10.1097/01.pas.0000213433.14740.5d. [DOI] [PubMed] [Google Scholar]
- 26.Senetta R, Trevisan E, Ruda R, Maldi E, Molinaro L, Lefranc F, Chiusa L, Lanotte M, Soffietti R, Cassoni P. Caveolin 1 expression independently predicts shorter survival in oligodendrogliomas. J Neuropathol Exp Neurol. 2009;68:425–431. doi: 10.1097/NEN.0b013e31819ed0b7. [DOI] [PubMed] [Google Scholar]
- 27.Silva WI, Maldonado HM, Velazquez G, Rubio-Dávila M, Miranda JD, Aquino E, Mayol N, Cruz-Torres A, Jardón J, Salgado-Villanueva IK. Caveolin isoform expression during differentiation of C6 glioma cells. Int J Dev Neurosci. 2005;23:599–612. doi: 10.1016/j.ijdevneu.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 28.Virgintino D, Robertson D, Errede M, Benagiano V, Tauer U, Roncali L, Bertossi M. Expression of caveolin-1 in human brain microvessels. Neuroscience. 2002;115:145–152. doi: 10.1016/s0306-4522(02)00374-3. [DOI] [PubMed] [Google Scholar]
- 29.Belot N, Rorive S, Doyen I, Lefranc F, Bruyneel E, Dedecker R, Micik S, Brotchi J, Decaestecker C, Salmon I, et al. Molecular characterization of cell substratum attachments in human glial tumors relates to prognostic features. Glia. 2001;36:375–390. doi: 10.1002/glia.1124. [DOI] [PubMed] [Google Scholar]
- 30.Branle F, Lefranc F, Camby I, Jeuken J, Geurts-Moespot A, Sprenger S, Sweep F, Kiss R, Salmon I. Evaluation of the efficiency of chemotherapy in in vivo orthotopic models of human glioma cells with and without 1p19q deletions and in C6 rat orthotopic allografts serving for the evaluation of surgery combined with chemotherapy. Cancer. 2002;95:641–655. doi: 10.1002/cncr.10710. [DOI] [PubMed] [Google Scholar]
- 31.Ingrassia L, Lefranc F, Dewelle J, Pottier L, Mathieu V, Spiegl-Kreinecker S, Sauvage S, El Yazidi M, Dehoux M, Berger W, et al. Structure-activity relationship analysis of novel derivatives of narciclasine (an Amaryllidaceae isocarbostyril derivative) as potential anticancer agents. J Med Chem. 2009;52:1100–1114. doi: 10.1021/jm8013585. [DOI] [PubMed] [Google Scholar]
- 32.Lefranc F, Mijatovic T, Kondo Y, Sauvage S, Roland I, Debeir O, Krstic D, Vasic V, Gailly P, Kondo S, et al. Targeting the α1 subunit of the sodium pump to combat glioblastoma cells. Neurosurgery. 2008;62:211–221. doi: 10.1227/01.NEU.0000311080.43024.0E. [DOI] [PubMed] [Google Scholar]
- 33.Le Mercier M, Fortin S, Mathieu V, Roland etI, Spiegl-Kreinecker S, Haibe-Kains B, Bontempi G, Decaestecker C, Berger W, Lefranc F, et al. Galectin 1 proangiogenic and promigratory effects in the Hs683 oligodendroglioma model are partly mediated through the control of BEX2 expression. Neoplasia. 2009;11:485–496. doi: 10.1593/neo.81526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sivasankaran B, Degen M, Ghaffari A, Hegi ME, Hamou MF, Ionescu MC, Zweifel C, Tolnay M, Wasner M, Mergenthaler S, et al. Tenascin-C is a novel RBPJκ-induced target gene for Notch signaling in gliomas. Cancer Res. 2009;69:458–465. doi: 10.1158/0008-5472.CAN-08-2610. [DOI] [PubMed] [Google Scholar]
- 35.Decaestecker C, Camby I, Gordower L, Dewitte O, Cras P, Martin JJ, Pasteels JL, Van Ham P, Brotchi J, Kiss R, et al. Characterization of astroglial versus oligodendroglial phenotypes in glioblastomas by means of quantitative morphonuclear variables generated by computer-assisted microscopy. J Neuropathol Exp Neurol. 1998;57:791–802. doi: 10.1097/00005072-199808000-00008. [DOI] [PubMed] [Google Scholar]
- 36.He J, Mokhtari K, Sanson M, Marie Y, Kujas M, Huguet S, Leuraud P, Capelle L, Delattre JY, Poirier J, et al. Glioblastomas with an oligodendroglial component: a pathological and molecular study. J Neuropathol Exp Neurol. 2001;60:863–871. doi: 10.1093/jnen/60.9.863. [DOI] [PubMed] [Google Scholar]
- 37.Lefranc F, James S, Camby I, Gaussin JF, Darro F, Brotchi J, Gabius J, Kiss R. Combined cimetidine and temozolomide, compared with temozolomide alone: significant increases in survival in nude mice bearing U373 human glioblastoma multiforme orthotopic xenografts. J Neurosurg. 2005;102:706–714. doi: 10.3171/jns.2005.102.4.0706. [DOI] [PubMed] [Google Scholar]
- 38.Lefranc F, Sauvage S, Van Goietsenoven G, Mégalizzi V, Lamoral-Theys D, Debeir O, Spiegl-Kreinecker S, Berger W, Mathieu V, Decaestecker C, et al. Narciclasine, a plant growth modulator, activates Rho and stress fibers in glioblastoma cells. Mol Cancer Ther. 2009;8:1739–1750. doi: 10.1158/1535-7163.MCT-08-0932. [DOI] [PubMed] [Google Scholar]
- 39.Megalizzi V, Mathieu V, Mijatovic T, Gailly P, Debeir O, De Neve N, Van Damme M, Bontempi G, Haibe-Kains B, Decaestecker C, et al. 4-IBP, a σ1 receptor agonist, decreases the migration of human cancer cells, including glioblastoma cells, in vitro and sensitizes them in vitro and in vivo to cytotoxic insults of proapoptotic and proautophagic drugs. Neoplasia. 2007;9:358–369. doi: 10.1593/neo.07130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Naslavsky N, Stein R, Yanai A, Friedlander G, Taraboulos A. Characterization of detergent-insoluble complexes containing the cellular prion protein and its scrapie isoform. J Biol Chem. 1997;272:6324–6331. doi: 10.1074/jbc.272.10.6324. [DOI] [PubMed] [Google Scholar]
- 41.Camby I, Decaestecker C, Lefranc F, Kaltner H, Gabius HJ, Kiss R. Galectin-1 knocking down in human U87 glioblastoma cells alters their gene expression pattern. Biochem Biophys Res Commun. 2005;335:27–35. doi: 10.1016/j.bbrc.2005.07.037. [DOI] [PubMed] [Google Scholar]
- 42.Martin S, Cosset EC, Terrand J, Maglott A, Takeda K, Dontenwill M. Caveolin-1 regulates glioblastoma aggressiveness through the control of α(5)β(1) integrin expression and modulates glioblastoma responsiveness to SJ749, an α(5)β(1) integrin antagonist. Biochim Biophys Acta. 2009;1793:354–367. doi: 10.1016/j.bbamcr.2008.09.019. [DOI] [PubMed] [Google Scholar]
- 43.Jin Y, Kim HP, Cao J, Zhang M, Ifedigbo E, Choi AM. Caveolin-1 regulates the secretion and cytoprotection of Cyr61 in hyperoxic cell death. FASEB J. 2009;23:341–350. doi: 10.1096/fj.08-108423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xie D, Yin D, Tong X, O'Kelly J, Mori A, Miller C, Black K, Gui D, Said JW, Koeffler HP. Cyr61 is overexpressed in gliomas and involved in integrin-linked kinase-mediated Akt and β-catenin-TCF/Lef signaling pathways. Cancer Res. 2004;64:1987–1996. doi: 10.1158/0008-5472.can-03-0666. [DOI] [PubMed] [Google Scholar]
- 45.Ikota H, Kinjo S, Yokoo H, Nakazato Y. Systematic immunohistochemical profiling of 378 brain tumors with 37 antibodies using tissue microarray technology. Acta Neuropathol. 2006;111:475–482. doi: 10.1007/s00401-006-0060-1. [DOI] [PubMed] [Google Scholar]
- 46.Gladson CL. The extracellular matrix of gliomas: modulation of cell function. J Neuropathol Exp Neurol. 1999;58:1029–1040. doi: 10.1097/00005072-199910000-00001. [DOI] [PubMed] [Google Scholar]
- 47.Rutka JT. The K.G. McKenzie award lecture—1986. Effects of extracellular matrix proteins on the growth and differentiation of an anaplastic glioma cell line. Can J Neurol Sci. 1986;13:301–306. doi: 10.1017/s0317167100036611. [DOI] [PubMed] [Google Scholar]
- 48.Lee H, Volonte D, Galbiati F, Iyengar P, Lublin DM, Bregman DB, Wilson MT, Campos-Gonzalez R, Bouzahzah B, Pestell RG, et al. Constitutive and growth factor-regulated phosphorylation of caveolin-1 occurs at the same site (Tyr-14) in vivo: identification of a c-Src/Cav-1/Grb7 signaling cassette. Mol Endocrinol. 2000;14:1750–1775. doi: 10.1210/mend.14.11.0553. [DOI] [PubMed] [Google Scholar]
- 49.Tahir SA, Frolov A, Hayes TG, Mims MP, Miles BJ, Lerner SP, Wheeler TM, Ayala G, Thompson TC, Kadmon D. Preoperative serum caveolin-1 as a prognostic marker for recurrence in a radical prostatectomy cohort. Clin Cancer Res. 2006;12:4872–4875. doi: 10.1158/1078-0432.CCR-06-0417. [DOI] [PubMed] [Google Scholar]
- 50.Tahir SA, Ren C, Timme TL, Gdor Y, Hoogeveen R, Morrisett JD, Frolov A, Ayala G, Wheeler TM, Thompson TC. Development of an immunoassay for serum caveolin-1: a novel biomarker for prostate cancer. Clin Cancer Res. 2003;9:3653–3659. [PubMed] [Google Scholar]
- 51.Djedid R, Kiss R, Lefranc F. Targeted therapy of glioblastomas: a 5-year view. Future Medicine. 2009;6:351–370. [Google Scholar]
- 52.Kesari S, Schiff D, Drappatz J, LaFrankie D, Doherty L, Macklin EA, Muzikansky A, Santagata S, Ligon KL, Norden AD, et al. Phase II study of protracted daily temozolomide for low-grade gliomas in adults. Clin Cancer Res. 2009;15:330–337. doi: 10.1158/1078-0432.CCR-08-0888. [DOI] [PubMed] [Google Scholar]
- 53.Murat A, Migliavacca E, Gorlia T, Lambiv WL, Shay T, Hamou MF, de Tribolet N, Regli L, Wick W, Kouwenhoven MC, et al. Stem cell-related “self-renewal” signature and high epidermal growth factor receptor expression associated with resistance to concomitant chemoradiotherapy in glioblastoma. J Clin Oncol. 2008;26:3015–3024. doi: 10.1200/JCO.2007.15.7164. [DOI] [PubMed] [Google Scholar]
- 54.Preusser M, Charles Janzer R, Felsberg J, Felsberg J, Reifenberger G, Hamou MF, Diserens AC, Stupp R, Gorlia T, Marosi C, et al. Anti-O6-methylguanine-methyltransferase (MGMT) immunohistochemistry in glioblastoma multiforme: observer variability and lack of association with patient survival impede its use as clinical biomarker. Brain Pathol. 2008;18:520–532. doi: 10.1111/j.1750-3639.2008.00153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Le Mercier M, Fortin S, Mathieu V, Kiss R, Lefranc F. Galectins and gliomas. Brain Pathol. 2010;20:17–27. doi: 10.1111/j.1750-3639.2009.00270.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cantiani L, Manara MC, Zucchini C, De Sanctis P, Zuntini M, Valvassori L, Serra M, Olivero M, Di Renzo MF, Colombo MP, et al. Caveolin-1 reduces osteosarcoma metastases by inhibiting c-Src activity and met signaling. Cancer Res. 2007;67:7675–7685. doi: 10.1158/0008-5472.CAN-06-4697. [DOI] [PubMed] [Google Scholar]
- 57.Cokakli M, Erdal E, Nart D, Yilmaz F, Sagol O, Kilic M, Karademir S, Atabey N. Differential expression of caveolin-1 in hepatocellular carcinoma: correlation with differentiation state, motility and invasion. BMC Cancer. 2009;9:65. doi: 10.1186/1471-2407-9-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Di Vizio D, Morello M, Sotgia F, Pestell RG, Freeman MR, Lisanti MP. An absence of stromal caveolin-1 is associated with advanced prostate cancer, metastatic disease and epithelial Akt activation. Cell Cycle. 2009;8:2420–2424. doi: 10.4161/cc.8.15.9116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Logozzi M, DeMilito A, Lugini L, Borghi M, Calabrò L, Spada M, Perdicchio M, Marino ML, Federici C, Iessi E, et al. High levels of exosomes expressing CD63 and caveolin-1 in plasma of melanoma patients. PLoS One. 2009;4:e5219. doi: 10.1371/journal.pone.0005219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tanase CP, Dima S, Mihai M, Raducan E, Nicolescu MI, Albulescu L, Voiculescu B, Dumitrascu T, Cruceru LM, Leabu M, et al. Caveolin-1 overexpression correlates with tumour progression markers in pancreatic ductal adenocarcinoma. J Mol Histol. 2009;40:23–29. doi: 10.1007/s10735-008-9209-7. [DOI] [PubMed] [Google Scholar]
- 61.Witkiewicz AK, Dasgupta A, Nguyen KH, Liu C, Kovatich AJ, Schwartz GF, Pestell RG, Sotgia F, Rui H, Lisanti MP. Stromal caveolin-1 levels predict early DCIS progression to invasive breast cancer. Cancer Biol Ther. 2009;8:1071–1079. doi: 10.4161/cbt.8.11.8874. [DOI] [PubMed] [Google Scholar]
- 62.Cordes N, Frick S, Brunner TB, Pilarsky C, Grützmann R, Sipos B, Klöppel G, McKenna WG, Bernhard EJ. Human pancreatic tumor cells are sensitized to ionizing radiation by knockdown of caveolin-1. Oncogene. 2007;26:6851–6862. doi: 10.1038/sj.onc.1210498. [DOI] [PubMed] [Google Scholar]
- 63.Regina A, Jodoin J, Khoueir P, Rolland Y, Berthelet F, Moumdjian R, Fenart L, Cecchelli R, Demeule M, Béliveau R. Down-regulation of caveolin-1 in glioma vasculature: modulation by radiotherapy. J Neurosci Res. 2004;75:291–299. doi: 10.1002/jnr.10865. [DOI] [PubMed] [Google Scholar]
- 64.Palozza P, Sestito R, Picci N, Lanza P, Monego G, Ranelletti FO. The sensitivity to β-carotene growth-inhibitory and proapoptotic effects is regulated by caveolin-1 expression in human colon and prostate cancer cells. Carcinogenesis. 2008;29:2153–2161. doi: 10.1093/carcin/bgn018. [DOI] [PubMed] [Google Scholar]
- 65.Head BP, Patel HH, Tsutsumi YM, Hu Y, Mejia T, Mora RC, Insel PA, Roth DM, Drummond JC, Patel PM. Caveolin-1 expression is essential for N-methyl-d-aspartate receptor-mediated Src and extracellular signal-regulated kinase 1/2 activation and protection of primary neurons from ischemic cell death. FASEB J. 2008;22:828–840. doi: 10.1096/fj.07-9299com. [DOI] [PubMed] [Google Scholar]
- 66.Wary KK, Mariotti A, Zurzolo C, Giancotti FG. A requirement for caveolin-1 and associated kinase Fyn in integrin signaling and anchorage-dependent cell growth. Cell. 1998;94:625–634. doi: 10.1016/s0092-8674(00)81604-9. [DOI] [PubMed] [Google Scholar]
- 67.Razani B, Woodman SE, Lisanti MP. Caveolae: from cell biology to animal physiology. Pharmacol Rev. 2002;54:431–467. doi: 10.1124/pr.54.3.431. [DOI] [PubMed] [Google Scholar]
- 68.Swaney JS, Patel HH, Yokoyama U, Head BP, Roth DM, Insel PA. Focal adhesions in (myo)fibroblasts scaffold adenylyl cyclase with phosphorylated caveolin. J Biol Chem. 2006;281:17173–17179. doi: 10.1074/jbc.M513097200. [DOI] [PubMed] [Google Scholar]
- 69.Barcellos-Hoff MH, Newcomb EW, Zagzag D, Narayana A. Therapeutic targets in malignant glioblastoma microenvironment. Semin Radiat Oncol. 2009;19:163–170. doi: 10.1016/j.semradonc.2009.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]