Abstract
Adolescence is a developmental period often characterized as a time of impulsive and risky choices leading to increased incidence of unintentional injuries and violence, alcohol and drug abuse, unintended pregnancy and sexually transmitted diseases. Traditional neurobiological and cognitive explanations for such suboptimal choices and actions have failed to account for nonlinear changes in behavior observed during adolescence, relative to childhood and adulthood. This review provides a biologically plausible conceptualization of the mechanisms underlying these nonlinear changes in behavior, as an imbalance between a heightened sensitivity to motivational cues and immature cognitive control. Recent human imaging and animal studies provide a biological basis for this view, suggesting differential development of subcortical limbic systems relative to top-down control systems during adolescence relative to childhood and adulthood. This work emphasizes the importance of examining transitions into and out of adolescence and highlights emerging avenues of future research on adolescent brain development.
Keywords: Adolescence, brain, development, fMRI, risk, incentive, cognitive control, connectivity
Introduction
Adolescence is characterized as a time when we act more impulsively, fail to consider long-term consequences, and engage in riskier behavior than we do as adults (Gardner & Steinberg, 2005; Scott, 1992; Steinberg, et al., 2008). This propensity to take risks is reflected in higher incidences of accidents, suicides, unsafe sexual practices, and criminal activity (Scott, 1992). Juveniles fifteen years of age and younger act more impulsively than do older adolescents, but even sixteen- and seventeen year old youth fail to exhibit adult levels of self-control (Feld, 2008).
In the past decade, a number of cognitive and neurobiological hypotheses have been postulated for why adolescents engage in impulsive and risky acts. Traditional accounts of adolescence suggest that it is a period of development associated with progressively greater efficiency of cognitive control capacities. This efficiency in cognitive control is described as dependent on maturation of the prefrontal cortex as evidenced by imaging (Galvan, et al., 2006; Gogtay, et al., 2004; Hare, et al., 2008; Sowell, et al., 2003) and post mortem studies (Bourgeois, Goldman-Rakic, & Rakic, 1994; Huttenlocher, 1979; Rakic, 1994) showing continued structural and functional development of this region well into young adulthood.
The general pattern of improved cognitive control with maturation of the prefrontal cortex (Crone & van der Molen, 2007) suggests a linear increase in development from childhood to adulthood. If cognitive control and an immature prefrontal cortex were the basis for suboptimal choice behavior alone, then children should look remarkably similar or presumably worse than adolescents, given their less developed prefrontal cortex and cognitive abilities (Casey, Getz, & Galvan, 2008). Yet suboptimal choices and actions observed during adolescence represent an inflection in development (Windle, et al., 2008) that is unique from either childhood or adulthood, as evidenced by the National Center for Health Statistics on adolescent behavior and mortality (Eaton, et al., 2008).
This review addresses the primary question of how the brain is changing during adolescence in ways that may explain inflections in risky behavior. We outline a testable neurobiological model that emphasizes the dynamic interplay between subcortical and cortical brain regions and speculate on the emergence of these systems from an evolutionary perspective. We provide evidence from behavioral and human brain imaging studies to support this model in the framework of actions in motivational contexts (Cauffman, et al., 2010; Figner, Mackinlay, Wilkening, & Weber, 2009; Galvan, Hare, Voss, Glover, & Casey, 2007; Galvan, et al., 2006) and address why some teenagers may be at greater risk than others for making suboptimal decisions leading to poorer long-term outcomes (Galvan, et al., 2007; Hare, et al., 2008).
Neurobiological Model of Adolescence
An accurate conceptualization of cognitive and neurobiological changes during adolescence must treat adolescence as a transitional developmental period (Spear, 2000), rather than a single snapshot in time. In other words, to understand this developmental period, characterizing transitions into and out of adolescence are necessary for distinguishing distinct attributes of this period of development (Casey, Galvan, & Hare, 2005; Casey, Tottenham, Liston, & Durston, 2005). Establishing developmental trajectories for cognitive processes is essential in characterizing these transitions and constraining interpretations about changes in behavior during this period.
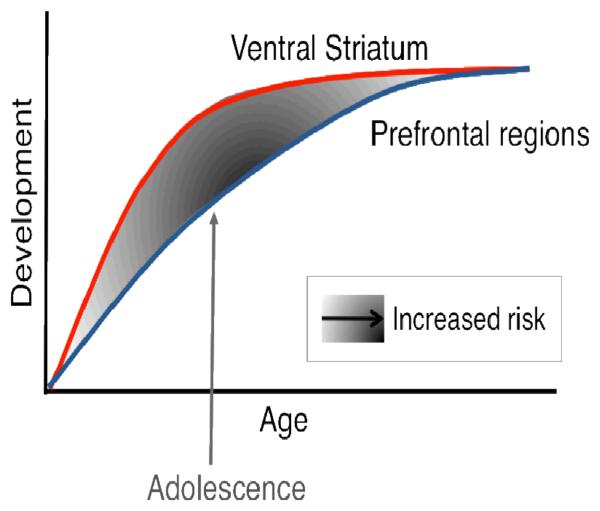
We have developed a testable neurobiological model of adolescent development within this framework that builds on rodent models (Brenhouse, Sonntag, & Andersen, 2008; Laviola, Adriani, Terranova, & Gerra, 1999; Spear, 2000) and recent imaging studies of adolescence (Ernst, et al., 2005; Galvan, et al., 2007; Galvan, et al., 2006; Hare, et al., 2008; Somerville, Hare, & Casey, in press; Van Leijenhorst, Moor, et al., 2010; Van Leijenhorst, Zanolie, et al., 2010). Figure 1 depicts this model. This characterization of adolescence goes beyond exclusive association of risky behavior to the immaturity of the prefrontal cortex. Rather, the proposed neurobiological model illustrates how subcortical and cortical top-down control regions must be considered together. The cartoon illustrates different developmental trajectories for these systems, with subcortical systems such as the ventral striatum developing earlier than prefrontal control regions. According to this model, the individual is biased more by functionally mature subcortical regions during adolescence (i.e., imbalance of subcortical relative to prefrontal cortical control), compared to children, for whom these systems (i.e., subcortical and prefrontal) are both still developing, and compared to adults, for whom these systems are fully mature.
Figure 1.
Neurobiological model depicting later development of top down prefrontal regions relative to subcortical regions involved in desire and fear. This imbalance in development of these systems is proposed to be at the core of risky choice behavior in adolescents in contrast to the popular view of adolescent behavior being due to the protracted development of the prefrontal cortex alone (From (Somerville, Jones, & Casey, 2010).
This perspective provides a basis for nonlinear shifts in risky behavior across development, due to earlier maturation of subcortical systems relative to less mature top down prefrontal control systems. With development and experience, the functional connectivity between these regions provides a mechanism for top down control of this circuitry (Hare, et al., 2008). Further, the model reconciles the contradiction of health statistics of risky behavior during adolescence, with the astute observation by Reyna and Farley (2006) that adolescents are quite capable of rational decisions and understand risks of behaviors in which they engage. However, in emotionally salient situations, subcortical systems will win out (accelerator) over control systems (brakes) given their maturity relative to the prefrontal control system.
This model is consistent with models of adolescent development (Ernst, Pine, & Hardin, 2006; Ernst, Romeo, & Andersen, 2009; Geier & Luna, 2009; Nelson, Leibenluft, McClure, & Pine, 2005; Steinberg, 2008; Steinberg, et al., 2009) that suggest differential development of subcortical and cortical regions. For example, the triadic model proposed by Ernst and colleagues (Ernst, et al., 2006) describes motivated behavior as having three distinct neural circuits (approach, avoidance and regulatory). The approach system relates to reward behaviors and is largely controlled by the ventral striatum. The avoidance system relates to avoidance behaviors and is mostly controlled by the amygdala. Lastly, the regulatory system balances the approach and avoidance systems and is largely controlled by the prefrontal cortex. Accordingly, increased risk taking behavior during adolescence is due to greater influence of the approach system and a weaker influence of the regulatory system.
Our model differs from others in that it is based on empirical evidence for brain changes not only in the transition from adolescence to adulthood, but also the transition into adolescence from childhood. Further, we do not suggest that the striatum and amygdala are specific to approach and avoidant behavior given recent studies showing valence independence of these structures (Levita, et al., 2009), but rather that they are systems important in detecting motivationally and emotionally relevant cues in the environment that can bias behavior. This sensitivity to appetitive and emotive cues during adolescence has been described across species (see Spear, 2009) and is reviewed here.
Comparative and Evolutionary Perspectives on Adolescence
A question that emerges from the imbalance model of adolescent brain development is why the brain might be programmed to develop in this way. This question may be addressed by taking a step backward and considering the definition of adolescence as the transitional period between childhood and adulthood. Puberty marks the onset of adolescence with the beginnings of sexual maturation (Graber & Brooks-Gunn, 1998) and can be defined by biological markers. Adolescence can be described as a progressive transition into adulthood with a nebulous ontogenetic time course (Spear, 2000, p.419). A complete discussion of the effect of pubertal hormones on brain and behavior is beyond the scope of this paper; see (Forbes & Dahl, 2010; Romeo, 2003) for detailed reviews on the subject.
Evolutionarily speaking, adolescence is a period of gaining independence from the protection of the family, which simultaneously may put the individual in harms way (Kelley, Schochet, & Landry, 2004). Independence-seeking behaviors are observed across mammalian species, with increases in peer-directed social interactions and intensification in novelty seeking which impact adolescents' propensity for risky behavior (Brown, 2004; Chassin, et al., 2004; Collins & Laursen, 2004; Laviola, et al., 1999). This risky behavior may be defined as the product of a biologically driven imbalance between increased novelty and sensation seeking in conjunction with immature “self-regulatory competence” (Steinberg, 2004). Speculation would suggest that this developmental pattern is an evolutionary feature in that an individual needs to engage in high-risk behavior to leave a safe and familiar niche in order to find a mate and procreate (Spear, 2000). Thus, risk taking appears to coincide with the time in which hormones drive adolescents to seek out sexual partners. In today's society -when adolescence may extend indefinitely- with children living with parents and having financial dependence and choosing mates later in life, this behavior may be less adaptive. Our neurobiological model suggests this occurs through differential development of subcortical and cortical systems. Empirical behavioral and imaging data are reviewed in support of this view.
Adolescent Behavioral Development
A core component of behavioral development is the ability to suppress inappropriate actions in favor of goal-directed ones, especially in the presence of compelling incentives. This ability is typically referred to as cognitive control (Casey, Galvan, et al., 2005; Casey, Giedd, & Thomas, 2000; Casey, Thomas, et al., 2000). We review classic cognitive developmental literature in the context of changes in cortically driven cognitive processes with age and provide behavioral and neuroanatomical evidence for its distinction from risky behaviors.
A number of classic developmental studies have shown that cognitive control develops throughout childhood and adolescence (Case, 1972; Flavell, Beach, & Chinksy, 1966; Keating & Bobbitt, 1978; Pascual-Leone, 1970). Several theorists have argued that this development is due to increases in processing speed and efficiency (e.g., (Bjorklund, 1985, 1987; Case, 1972)), but others have suggested “inhibitory” processes are the key factor (Harnishfeger & Bjorklund, 1993). According to this account, suboptimal choices in childhood are due to greater susceptibility to interference from competing sources that must be suppressed (e.g., (Brainerd & Reyna, 1993; Casey, Thomas, Davidson, Kunz, & Franzen, 2002; Dempster, 1993; Diamond, 1985; Munakata & Yerys, 2001). Thus optimal decision-making requires the control of impulses (Mischel, Shoda, & Rodriguez, 1989) and this ability matures in a linear fashion across childhood and adolescence (Eigsti, et al., 2006).
In contrast, risk taking or reward seeking behaviors, seem to peak during adolescence and then decline in adulthood (Eaton, et al., 2008; Windle, et al., 2008) and are associated with puberty maturation (Dahl, 2004; Martin, et al., 2001). A recent study by Steinberg et al. (2008) delineated the construct of impulse/cognitive control from sensation seeking behaviors, defined as the desire to seek out novel experiences and taking risks in order to achieve them. They tested individuals between the ages of 10 and 30 and showed that differences in sensation seeking with age followed a curvilinear pattern, with peaks in sensation seeking increasing between 10 and 15 years and declining or remaining stable thereafter. In contrast, age differences in impulsivity followed a linear pattern, with decreasing impulsivity with age.
These findings suggest distinct developmental trajectories for the two constructs. Specifically, impulsivity diminishes with age across childhood and adolescence (Casey, Galvan, et al., 2005; Casey, Thomas, et al., 2002; Galvan, et al., 2007), although there are differences in the degree to which a given individual is impulsive or not, regardless of age (Eigsti, et al., 2006). In contrast to impulse/cognitive control, sensation seeking/risk taking appears to show a curvilinear pattern, with an increase during adolescence relative to childhood and adulthood (Cauffman, et al., 2010; Figner, et al., 2009; Galvan, et al., 2007). As will be reviewed in the following sections, these findings suggest a distinct neural system for the construct of risky behavior, separate from the neural system for impulse control, with earlier development of risk taking behavior relative to protracted development of impulse control (Galvan, et al., 2007; Steinberg, et al., 2008).
Adolescent Brain Development
Recent investigations of adolescent brain development have been based on advances in neuroimaging methodologies that can be easily used with developing human populations. These methods rely on magnetic resonance imaging (MRI) methods and include: structural MRI, which is used to measure the size and shape of structures; functional MRI (fMRI) which is used to measure patterns of brain activity; and diffusion tensor imaging (DTI) which is used to index connectivity of white matter fiber tracts. Evidence for our developmental model of competition between cortical and subcortical regions is supported by immature structural and functional connectivity as measured by DTI and fMRI, respectively.
MRI Studies of Human Brain Development
Several studies have used structural MRI to map the anatomical course of normal brain development (see review (Casey, Tottenham, et al., 2005)). Although total brain size is approximately 90% of its adult size by age six, the gray and white matter subcomponents of the brain continue to undergo dynamic changes throughout adolescence. Data from recent longitudinal MRI studies indicate that gray matter volume has an inverted U-shape pattern, with greater regional variation than white matter (Giedd, 2004; Gogtay, et al., 2004; Sowell, et al., 2003; Sowell, Thompson, & Toga, 2004). In general, regions subserving primary functions, such as motor and sensory systems, mature earliest; higher-order association areas, which integrate these primary functions, mature later (Gogtay, et al., 2004; Sowell, et al., 2004). For example, studies using MRI-based measures show that cortical gray matter loss occurs earliest in the primary sensorimotor areas and latest in the dorsolateral prefrontal and lateral temporal cortices (Gogtay, et al., 2004). This pattern is consistent with nonhuman primate and human postmortem studies showing that the prefrontal cortex is one of the last brain regions to mature (Bourgeois, et al., 1994; Huttenlocher, 1979) while subcortical and sensorimotor regions develop sooner. In contrast to gray matter, white matter volume increases in a roughly linear pattern, increasing throughout development well into adulthood (Gogtay, et al., 2004). These changes presumably reflect ongoing myelination of axons by oligodendrocytes enhancing neuronal conduction and communication of relevant connections.
Although less attention has been given to subcortical regions when examining structural changes, some of the largest changes in the brain across development are seen in portions of the basal ganglia such as the striatum (Sowell, Thompson, Holmes, Jernigan, & Toga, 1999), especially in males (Giedd, et al., 1996). These developmental changes in structural volume within basal ganglia and prefrontal regions suggest that cortical connections are becoming more refined consistent with neural developmental processes (e.g. dendritic arborization, cell death, synaptic pruning, myelination) that are occurring during childhood and adolescence (Huttenlocher, 1979). These processes allow for fine-tuning and strengthening of connections between prefrontal and subcortical regions with learning that may coincide with greater cognitive control (e.g., signaling of prefrontal control regions to adjust behavior) (Casey, Amso, & Davidson, 2006; Casey & Durston, 2006).
It is unclear exactly how structural changes relate to behavior changes. A few studies have shown indirect associations between MRI-based volumetric change and cognitive function using neuropsychological measures (e.g., (Casey, Castellanos, et al., 1997; Sowell, et al., 2003)). Specifically, associations have been reported between MRI-based prefrontal cortical and basal ganglia regional volumes and measures of cognitive control (i.e., ability to override an inappropriate choice/action in favor of another (Casey, Castellanos, et al., 1997) (Casey, Trainor, et al., 1997)). These findings suggest that cognitive changes are reflected in structural changes in the brain and underscore the importance of subcortical (striatum) as well as cortical (e.g., prefrontal cortex) development.
DTI Studies of Human Brain Development
The MRI-based morphometry studies reviewed suggest that cortical connections are being fine-tuned with the elimination of an overabundance of synapses and strengthening of relevant connections with development and experience. Recent advances in MRI technology like DTI provide a tool for examining the developmental modulation of specific white matter tracts and their relation to behavior. In one study, development of cognitive control was positively correlated with prefrontal-parietal fiber tracts (Nagy, Westerberg, & Klingberg, 2004) consistent with functional neuroimaging studies showing differential recruitment of these regions in children relative to (Klingberg, Forssberg, & Westerberg, 2002).
Using a similar approach, Liston and colleagues (2006) examined the strength of white matter tracts in frontostriatal circuits, which continue to develop across childhood into adulthood. The frontostriatal fiber tracts were defined by connecting two regions of interest in the striatum and ventral prefrontal cortex identified in an fMRI study using the same task (Durston, Thomas, Worden, Yang, & Casey, 2002; Epstein, et al., 2007). Across these developmental DTI studies, fiber tract measures across the entire brain were correlated with development. However, there was specificity in which particular fiber tracts were associated with cognitive control (Casey, et al., 2007; Liston, et al., 2006) or cognitive ability (Nagy, et al., 2004). Specifically, frontostriatal connection strength positively predicted impulse control capacity, as measured by performance on a go/nogo task (Casey, et al., 2007; Liston, et al., 2006). These findings underscore the importance of examining not only regional structural changes, but also circuitry related changes when making claims about age-dependent maturation of neural substrates of cognitive development.
Functional MRI Studies of Behavioral and Brain Development
Although structural changes as measured by MRI and DTI have been associated with behavioral changes during development, a more direct approach for examining structure-function associations is to measure changes in the brain and behavior simultaneously, as with fMRI. The ability to measure functional changes in the developing brain with MRI has significant potential for the field of developmental science. In the context of the current article, fMRI provides a means for constraining interpretations of adolescent decision-making. As stated previously, the development of the prefrontal cortex is believed to play an important role in the maturation of higher cognitive abilities such as decision-making and goal oriented choice behavior (Casey, Tottenham, & Fossella, 2002; Casey, Trainor, et al., 1997). Many paradigms have been used, together with fMRI, to assess the neurobiological basis of these abilities. These paradigms include go/nogo, (participants must respond to one stimulus but suppress responses to a second stimulus) flanker (participants choose the directionality of a target surrounded by symbols that are either compatible or incompatible with the target), stop signal (participants respond as fast as possible to a stimulus but must suppress this response when they receive a stop signal such as an auditory tone), and antisaccade tasks (participants must inhibit reflexive eye movements to gaze in the opposite direction of a target) (Bunge, Dudukovic, Thomason, Vaidya, & Gabrieli, 2002; Casey, Giedd, et al., 2000; Casey, Trainor, et al., 1997; Durston, et al., 2003; Luna, et al., 2001). Collectively, these studies show that children recruit distinct but often larger, more diffuse prefrontal regions when performing these tasks than do adults. The pattern of activity within brain regions central to task performance (i.e., that correlate with cognitive performance) become more focal or fine-tuned with age; while regions not correlated with task performance diminish in activity with age. This pattern has been observed across both cross-sectional (Brown, et al., 2005) and longitudinal studies (Durston, et al., 2006) and across a variety of paradigms.
Although neuroimaging studies cannot definitively characterize the mechanism of such developmental changes (e.g. dendritic arborization, synaptic pruning) the findings reflect development within, and refinement of, projections to and from activated brain regions with maturation. Further, the findings suggest that these neuroanatomical changes occur over a protracted period of time (Brown, et al., 2005; Bunge, et al., 2002; Casey, Thomas, et al., 2002; Casey, Trainor, et al., 1997; Crone, Donohue, Honomichl, Wendelken, & Bunge, 2006; Luna, et al., 2001; Moses, et al., 2002; Schlaggar, et al., 2002; Tamm, Menon, & Reiss, 2002; Thomas, et al., 2004; Turkeltaub, Gareau, Flowers, Zeffiro, & Eden, 2003).
How can this methodology inform us about whether adolescent decisions are indeed impulsive or are risky? Impulse control as measured by tasks such as the go/nogo task show a linear pattern of development across childhood and adolescence as described above. However, recent neuroimaging studies have begun to examine reward-related processing relevant to risk-taking in adolescents (Bjork, et al., 2004; Ernst, et al., 2005; Galvan, et al., 2005; May, et al., 2004; Van Leijenhorst, Moor, et al., 2010). These studies have focused primarily on the region of the ventral striatum, a region implicated in learning and predicting reward outcomes.
Sensitivity to Appetitive cues in Adolescence
Our neurobiological model suggests that the combination of heightened responsiveness to motivational cues and immaturity in behavioral control may bias adolescents to seek immediate, rather than long-term gains. Tracking subcortical (e.g., ventral striatum) and cortical (e.g., prefrontal) development across childhood through adulthood provides constraints on whether changes reported in adolescence are specific to this period of development, or reflect maturation that is steadily occurring in a somewhat linear pattern from childhood to adulthood.
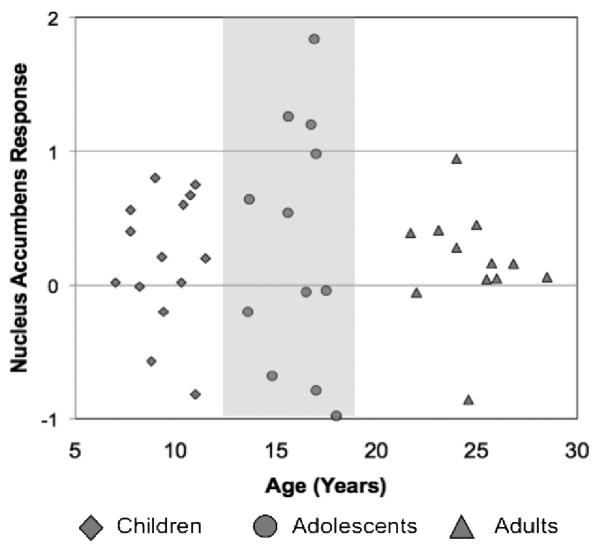
Several groups have shown that adolescents show heightened activation of the ventral striatum in anticipation and/or receipt of rewards compared to adults (Ernst, et al., 2005; Galvan, et al., 2006; Geier, Terwilliger, Teslovich, Velanova, & Luna, 2009; Van Leijenhorst, Zanolie, et al., 2010), coupled with less activation in the prefrontal cortex relative to adults. In one of the first studies to examine this response across the full range of childhood to adulthood Galvan and her colleagues examined behavioral and neural responses to reward manipulations in 6 to 29 year olds. They focused on brain circuitry implicated in reward related learning and behavior in animal studies (Hikosaka & Watanabe, 2000; Pecina, Cagniard, Berridge, Aldridge, & Zhuang, 2003; Schultz, 2006), adult human imaging studies (e.g. (Knutson, Adams, Fong, & Hommer, 2001; O'Doherty, Kringelbach, Rolls, Hornak, & Andrews, 2001; Zald, et al., 2004)) and in studies of addiction (Hyman & Malenka, 2001; Volkow & Li, 2004). Based on rodent models (Laviola, et al., 1999; Spear, 2000) and previous imaging work (Ernst, et al., 2005), they hypothesized that relative to children and adults, adolescents would show exaggerated activation of the ventral striatum in concert with less mature recruitment of top down prefrontal control regions. Their results supported this hypothesis showing that the spatial extent of brain activity in adolescents in the ventral striatum to reward, was similar to that observed in adults, whereas the extent of activity in prefrontal regions was more similar to children. The extent of activity between these two regions was associated with elevated magnitude of activity in the ventral striatum in adolescents relative to children and adults assumed to result from the imbalance in corticosubcortial development (see Figure 2). Recent work showing delayed functional connectivity between prefrontal and subcortical regions in adolescence relative to adults provides a mechanism for the lack of top down control of regions related to processing motivational cues (Hare, et al., 2008).
Figure 2.
Activity in the ventral striatum to anticipated reward as a function of age, for each individual subject, showing enhanced activity between roughly 13 to 18 years (Adapted from Galvan et al., 2006; 2007).
These findings are consistent in part with rodent models (Laviola, Macri, Morley-Fletcher, & Adriani, 2003) and previous imaging studies (Ernst, et al., 2005; Van Leijenhorst, Moor, et al., 2010) showing enhanced ventral striatal activity to rewards and anticipation of rewards during adolescence. Relative to children and adults, adolescents showed an exaggerated ventral striatal response to reward. However, both children and adolescents showed a less mature response in prefrontal control regions than adults. These findings suggest different developmental trajectories for these regions may underlie the enhancement in ventral striatal activity, relative to children or adults, which may in turn relate to the increased risky decisions observed during this period of development (Figner, et al., 2009). It is relevant to note that while several laboratories (Ernst, et al., 2005; Galvan, et al., 2006; Geier, et al., 2009; Somerville, et al., in press; Van Leijenhorst, Moor, et al., 2010) have shown this heightened response in the ventral striatum in adolescents, one laboratory has failed to observe this response (Bjork, et al., 2004; Bjork, Smith, Chen, & Hommer, 2010) Future studies will be needed to clarify the specific conditions under which this pattern of brain activity is or is not observed.
Differential recruitment of prefrontal and subcortical regions have been reported across a number of developmental fMRI studies (Casey, Thomas, et al., 2002; Geier, et al., 2009; Luna, et al., 2001; Monk, et al., 2003; Thomas, et al., 2004; Van Leijenhorst, Zanolie, et al., 2010). Typically these findings have been interpreted in terms of immature prefrontal regions rather than an imbalance between prefrontal and subcortical regional development. Given evidence of prefrontal regions in guiding appropriate actions in different contexts (Miller & Cohen, 2001), immature prefrontal activity might hinder appropriate estimation of future outcomes and appraisal of risky choices, and might thus be less influential on reward valuation than the ventral striatum. This pattern is consistent with previous research showing elevated subcortical, relative to cortical activity when decisions are biased by immediate over long-term gains (McClure, Laibson, Loewenstein, & Cohen, 2004). During adolescence, relative to childhood or adulthood, immature prefrontal cortex engagement may not provide sufficient top-down control of robustly activated reward processing regions (e.g., ventral striatum), resulting in less influence of prefrontal systems relative to the ventral striatum in reward valuation.
While differential recruitment of cortical and subcortical regions has been robustly reported across development, only a few studies have addressed how cognitive control and reward systems interact. A recent study by (Geier, et al., 2009) examined this interaction using a version of an antisaccade task during fMRI in adolescents and adults. Their findings showed that on trials for which money was at stake, performance was enhanced, with the greatest enhancement (faster and more accurate responses) observed in adolescents. This performance was paralleled by exaggerated activation in the ventral striatum in adolescents following a cue that the next trial would be rewarded while they were preparing for and subsequently executing the antisaccade. Adolescents also showed elevated prefrontal activity in regions important for controlling eye movements. These findings suggest a reward-related upregulation in these control regions.
The Geier study provides an example of how appetitive cues can facilitate cognitive performance in adolescents, but high risk behavior in adolescence in every day life suggest that appetitive cues may impair cognitive decisions. To test this hypothesis, Somerville and colleagues (Somerville, et al., in press) tested children, adolescents and adults while performing a go/nogo task on which they had to suppress a response to an appetitive social cue. She showed that adolescents had greater difficulty resisting appetitive social cues compared to children and adults, as evidenced by more false alarms to these cue than neutral ones. This behavioral performance was paralleled by enhanced activity in the ventral striatum. In contrast, activation in the prefrontal cortex was associated with overall accuracy and showed a linear decrease in activity with improvement in performance and age. A functional connectivity analyses identified the dorsal striatum as a key convergence point for cortical and subcortical signals. Collectively, these studies suggest that differences in adolescent behavior from adults depend on the context of the behavior. In appetitively charged situations, subcortical systems involved in detection of appetitive cues will win out (accelerator) over cortical control systems (brakes), given differential regional development. However, in situations in which appetitive or emotive cues are not present, cortical control systems are not compromised, leading to more optimal performance in adolescents.
Adolescence and Individual Differences
Individuals vary in their ability to control impulses and in risk taking, a phenomenon that has been recognized in psychology for some time (Benthin, Slovic, & Severson, 1993). Therefore, some adolescents will be more likely to engage in risky behaviors, and be more prone to poorer outcomes. Thus, examining individual variability may help to identify potential bio-behavioral markers to identify individuals who might be at greater risk for poor outcomes during adolescence.
A classic example of individual differences reported in these abilities in the social, cognitive and developmental psychology literature is that of delay of gratification (Mischel, et al., 1989). Delay of gratification is typically assessed in 3 to 4 year old children. The child is asked whether they would prefer a small reward (one marshmallow) now or a large reward (two marshmallows) later. The child is then told that the experimenter will leave the room in order to prepare for upcoming activities and explains to the child that if she remains in her seat and does not eat a marshmallow during that time, then she will receive the large reward of both marshmallows. If the child does not or cannot wait, she should ring a bell to summons the experimenter and thereby receive the smaller reward. Once it is clear the child understands the task, she is seated at the table with the two rewards and the bell. Distractions in the room are minimized, with no toys, books or pictures. The experimenter returns after 15 minutes or after the child has rung the bell, eaten the rewards, or shown any signs of distress. Using this paradigm, Mischel showed that children typically behave in one of two ways on this task: 1) they ring the bell almost immediately in order to have the marshmallow, which means they only get one; 2) they wait and optimize their gains, and receive both marshmallows. This observation suggests that some individuals are better than others in their ability to control impulses in the face of highly salient incentives and this bias can be detected in early childhood (Mischel, et al., 1989) and appear to remain throughout adolescence and young adulthood (Eigsti, et al., 2006).
What might explain individual differences in optimal choice behavior? Some theorists have postulated that dopaminergic mesolimbic circuitry, implicated in reward processing, underlies risky behavior (Blum, et al., 2000). Developmental studies provide neurochemical evidence indicates that the balance in the adolescent brain between cortical and subcortical dopamine systems begins to shift toward greater cortical dopamine levels during adolescence (Brenhouse, et al., 2008; Spear, 2000). Similarly, there is a delayed time course of dopaminergic enervation of the nonhuman primate prefrontal cortex through adolescence into adulthood suggesting that functional maturity is not reached until adulthood (Rosenberg & Lewis, 1995). Individual differences in this circuitry, such as allelic variants in dopamine-related genes, resulting in too little or too much dopamine in subcortical regions, might relate to the propensity of some to engage in risky behavior more than others (O'Doherty, 2004).
The ventral striatum has been shown to increase in activity immediately prior to making risky choices on monetary-risk paradigms (Kuhnen & Knutson, 2005; Matthews, Simmons, Lane, & Paulus, 2004; Montague & Berns, 2002) and as described previously, adolescents show exaggerated striatal activity to rewarding outcomes relative to children or adults (Ernst, et al., 2005; Galvan, et al., 2006). Collectively, these data suggest that adolescents may be more prone to risky choices as a group (Figner, et al., 2009; Gardner & Steinberg, 2005), but some adolescents will be more prone than others to engage in risky behaviors, putting them at potentially greater risk for negative outcomes.
To explore individual differences in risk taking behavior, Galvan and colleagues (2007) examined the association between activity in reward-related neural circuitry in response to a large monetary reward with personality trait measures of risk taking and impulsivity in adolescence. Functional magnetic resonance imaging and anonymous self-report rating scales of risky behavior, risk perception and impulsivity were acquired in individuals between the ages of 7 and 29 years. There was a positive association between ventral striatal activity and the likelihood of engaging in risky behavior across development. This activity varied as a function of individuals' ratings of anticipated positive or negative consequences of such behavior. The individuals who perceived risky behaviors as leading to dire consequences, activated the ventral striatum less to reward. This negative association was driven by the child participants, whereas a positive association was seen in the adults who rated the consequences of such behavior as positive.
In addition to linking risk taking to reward circuitry, Galvan showed no association between activity of this circuitry and ratings of impulsivity (Galvan, et al., 2007). Instead she showed that impulsivity was negatively correlated with age. This finding is consistent with a recent report Steinberg (2008) showing differential development of sensation seeking and impulsivity, with sensation seeking increasing during adolescence relative to childhood and adulthood, but impulsivity followed a linear pattern of decreasing with age. These findings suggest that during adolescence, some individuals may be more prone to engage in risky behaviors due to developmental changes in concert with variability in a given individual's predisposition to engage in risky behavior, rather than to simple changes in impulsivity. Further, these individual and developmental differences may help explain vulnerability in some individuals to risk-taking associated with substance use, and ultimately, addiction.
Conclusion
Human imaging studies show structural and functional changes in corticosubcortical circuitry (for review, (Casey, Tottenham, et al., 2005; Giedd, et al., 1999; Giedd, et al., 1996; Jernigan, et al., 1991; Sowell, et al., 1999)) that parallel increases in cognitive control and self-regulation (Casey, Trainor, et al., 1997; Luna & Sweeney, 2004; Luna, et al., 2001; Rubia, et al., 2000; Steinberg, 2004; Steinberg, et al., 2008). These changes show a shift in activation of prefrontal regions from diffuse to more focal recruitment over time (Brown, et al., 2005; Bunge, et al., 2002; Casey, Trainor, et al., 1997; Durston & Casey, 2006; Moses, et al., 2002) and elevated recruitment of subcortical regions during adolescence (Casey, Thomas, et al., 2002; Durston & Casey, 2006; Luna, et al., 2001). Although neuroimaging studies cannot definitively characterize the mechanism of such developmental changes, these changes in volume and structure may reflect development within, and refinement of, projections to and from these brain regions during maturation suggestive of fine-tuning of the system with development (Hare, et al., 2008; Liston, et al., 2006).
Taken together, the findings synthesized here indicate that increased risk taking behavior in adolescence is associated with different developmental trajectories of subcortical motivational and cortical control regions. However, this is not to say that adolescents are incapable of making rational decisions. Rather, in emotionally charged situations, the more mature limbic system may win over the prefrontal control system in guiding actions.
Although adolescence has been distinguished as a period characterized by reward seeking and risk taking behaviors (Gardner & Steinberg, 2005; Spear, 2000) individual differences in neural responses to reward, predispose some adolescents to take more risks than others, putting them at greater risk for poor outcomes such as addiction, substance abuse and mortality. These findings provide crucial groundwork by synthesizing the various findings related to impulsivity and risk taking in adolescence and in understanding individual differences and developmental markers for propensities for suboptimal choices leading to negative consequences.
Acknowledgments
This work was supported in part by NIDA R01 DA018879, NIMH P50 MH62196, NSF 06-509, and NSF 0720932 to BJC, the Mortimer D. Sackler family, the Dewitt-Wallace fund, and by the Weill Cornell Medical College Citigroup Biomedical Imaging Center and Imaging Core.
References
- Benthin A, Slovic P, Severson H. A psychometric study of adolescent risk perception. Journal of Adolescence. 1993;16:153–168. doi: 10.1006/jado.1993.1014. [DOI] [PubMed] [Google Scholar]
- Bjork JM, Knutson B, Fong GW, Caggiano DM, Bennett SM, Hommer DW. Incentive-elicited brain activation in adolescents: similarities and differences from young adults. Journal of Neuroscience. 2004;24(8):1793–1802. doi: 10.1523/JNEUROSCI.4862-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjork JM, Smith AR, Chen G, Hommer DW. Adolescents, adults and rewards: comparing motivational neurocircuitry recruitment using fMRI. PLoS One. 2010;5(7):e11440. doi: 10.1371/journal.pone.0011440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjorklund DF. The role of conceptual knowledge in the development of organization in children's memory. In: Brainerd CJ, Pressley M, editors. Basic processes in memory development: Progress in cognitive development research. Springer-Verlag; New York: 1985. pp. 103–142. [Google Scholar]
- Bjorklund DF. How age changes in knowledge base contribute to the development of children's memory: An interpretive review. Developmental Review. 1987;7:993–130. [Google Scholar]
- Blum K, Braverman ER, Holder JM, Lubar JF, Monastra VJ, Miller D, et al. Reward deficiency syndrome: a biogenetic model for the diagnosis and treatment of impulsive, addictive, and compulsive behaviors. J Psychoactive Drugs. 2000;32(Suppl, i-iv):1–112. doi: 10.1080/02791072.2000.10736099. [DOI] [PubMed] [Google Scholar]
- Bourgeois JP, Goldman-Rakic PS, Rakic P. Synaptogenesis in the prefrontal cortex of rhesus monkeys. Cerebral Cortex. 1994;4:78–96. doi: 10.1093/cercor/4.1.78. [DOI] [PubMed] [Google Scholar]
- Brainerd CJ, Reyna VF. Memory independence and memory interference in cognitive development. Psychological Review. 1993;100:42–67. doi: 10.1037/0033-295x.100.1.42. [DOI] [PubMed] [Google Scholar]
- Brenhouse HC, Sonntag KC, Andersen SL. Transient D1 dopamine receptor expression on prefrontal cortex projection neurons: relationship to enhanced motivational salience of drug cues in adolescence. J Neurosci. 2008;28(10):2375–2382. doi: 10.1523/JNEUROSCI.5064-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown BB. Adolescents' Relationships with Peers. In: Lerner RM, Steinberg LD, editors. Handbook of adolescent psychology. John Wiley & Sons, Inc; Hoboken: 2004. pp. 363–394. [Google Scholar]
- Brown TT, Lugar HM, Coalson RS, Miezin FM, Petersen SE, Schlaggar BL. Developmental changes in human cerebral functional organization for word generation. Cerebral Cortex. 2005;15:275–290. doi: 10.1093/cercor/bhh129. [DOI] [PubMed] [Google Scholar]
- Bunge SA, Dudukovic NM, Thomason ME, Vaidya CJ, Gabrieli JD. Immature frontal lobe contributions to cognitive control in children: evidence from fMRI. Neuron. 2002;33(2):301–311. doi: 10.1016/s0896-6273(01)00583-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Case R. Validation of a neo-Piagetian mental capacity construct. Journal of Experimental Child Psychology. 1972;14:287–302. [Google Scholar]
- Casey BJ, Amso D, Davidson MC. Learning about learning and development with neuroimaging. In: Johnsons M, Munakata Y, editors. Attention and Performance XXI: Processes of Change in Brain and Cognitive Development. MIT; Cambridge, MA: 2006. [Google Scholar]
- Casey BJ, Castellanos FX, Giedd JN, Marsh WL, Hamburger SD, Schubert AB, et al. Implication of right frontostriatal circuitry in response inhibition and attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1997;36(3):374–383. doi: 10.1097/00004583-199703000-00016. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Durston S. From behavior to cognition to the brain and back: what have we learned from functional imaging studies of attention deficit hyperactivity disorder? Am J Psychiatry. 2006;163(6):957–960. doi: 10.1176/ajp.2006.163.6.957. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Epstein JN, Buhle J, Liston C, Davidson MC, Tonev ST, et al. Frontostriatal connectivity and its role in cognitive control in parent-child dyads with ADHD. Am J Psychiatry. 2007;164(11):1729–1736. doi: 10.1176/appi.ajp.2007.06101754. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Galvan A, Hare TA. Changes in cerebral functional organization during cognitive development. Curr Opin Neurobiol. 2005;15(2):239–244. doi: 10.1016/j.conb.2005.03.012. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Getz S, Galvan A. The adolescent brain. Dev Rev. 2008;28(1):62–77. doi: 10.1016/j.dr.2007.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Giedd JN, Thomas KM. Structural and functional brain development and its relation to cognitive development. Biol Psychol. 2000;54(1-3):241–257. doi: 10.1016/s0301-0511(00)00058-2. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Thomas KM, Davidson MC, Kunz K, Franzen PL. Dissociating striatal and hippocampal function developmentally with a stimulus-response compatibility task. Journal of Neuroscience. 2002;22(19):8647–8652. doi: 10.1523/JNEUROSCI.22-19-08647.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Thomas KM, Welsh TF, Badgaiyan RD, Eccard CH, Jennings JR, et al. Dissociation of response conflict, attentional selection, and expectancy with functional magnetic resonance imaging. Proceedings of the National Academy of Sciences of the United States of America. 2000;97(15):8728–8733. doi: 10.1073/pnas.97.15.8728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Tottenham N, Fossella J. Clinical, imaging, lesion, and genetic approaches toward a model of cognitive control. Dev Psychobiol. 2002;40(3):237–254. doi: 10.1002/dev.10030. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Tottenham N, Liston C, Durston S. Imaging the developing brain: what have we learned about cognitive development? Trends in Cognitive Science. 2005;9(3):104–110. doi: 10.1016/j.tics.2005.01.011. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Trainor RJ, Orendi JL, Schubert AB, Nystrom LE, Giedd JN, et al. A developmental functional MRI study of prefrontal activation during performance of a go-no-go task. Journal of Cognitive Neuroscience. 1997;9:835–847. doi: 10.1162/jocn.1997.9.6.835. [DOI] [PubMed] [Google Scholar]
- Cauffman E, Shulman EP, Steinberg L, Claus E, Banich MT, Graham S, et al. Age differences in affective decision making as indexed by performance on the Iowa Gambling Task. Dev Psychol. 2010;46(1):193–207. doi: 10.1037/a0016128. [DOI] [PubMed] [Google Scholar]
- Chassin L, Hussong A, Barrera M, Jr., Molina BSG, Trim R, Ritter J. Adolescent Substance Use. In: Lerner RM, Steinberg LD, editors. Handbook of adolescent psychology. John Wiley & Sons, Inc; Hoboken: 2004. pp. 665–696. [Google Scholar]
- Collins AW, Laursen B. Parent-Adolescent Relationships and Influences. In: Lerner RM, Steinberg LD, editors. Handbook of adolescent psychology. John Wiley & Sons, Inc; Hoboken: 2004. pp. 331–362. [Google Scholar]
- Crone EA, Donohue SE, Honomichl R, Wendelken C, Bunge SA. Brain regions mediating flexible rule use during development. J Neurosci. 2006;26(43):11239–11247. doi: 10.1523/JNEUROSCI.2165-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crone EA, van der Molen MW. Development of decision making in school-aged children and adolescents: evidence from heart rate and skin conductance analysis. Child Dev. 2007;78(4):1288–1301. doi: 10.1111/j.1467-8624.2007.01066.x. [DOI] [PubMed] [Google Scholar]
- Dahl R. Adolescent brain development: A period of vulnerabilities and opportunities. Annals of the New York Academy of Sciences. 2004;1021:1–22. doi: 10.1196/annals.1308.001. [DOI] [PubMed] [Google Scholar]
- Dempster FN. Resistance to interference: Developmental changes in a basic processing mechanism. In: Howe ML, Pasnak R, editors. Emerging themes in cognitive development. Vol. 1. Springer; New York: 1993. pp. 3–27. [Google Scholar]
- Diamond A. Development of the ability to use recall to guide action, as indicated by infants' performance on AB. Child Development. 1985;56:868–883. [PubMed] [Google Scholar]
- Durston S, Casey BJ. A shift from diffuse to focal cortical activity with development: the authors' reply. Dev Sci. 2006;9(1):18–20. doi: 10.1111/j.1467-7687.2005.00458.x. [DOI] [PubMed] [Google Scholar]
- Durston S, Davidson MC, Thomas KM, Worden MS, Tottenham N, Martinez A, et al. Parametric manipulation of conflict and response competition using rapid mixed-trial event-related fMRI. Neuroimage. 2003;20(4):2135–2141. doi: 10.1016/j.neuroimage.2003.08.004. [DOI] [PubMed] [Google Scholar]
- Durston S, Davidson MC, Tottenham N, Galvan A, Spicer J, Fossella JA, et al. A shift from diffuse to focal cortical activity with development. Dev Sci. 2006;9(1):1–8. doi: 10.1111/j.1467-7687.2005.00454.x. [DOI] [PubMed] [Google Scholar]
- Durston S, Thomas KM, Worden MS, Yang Y, Casey BJ. The effect of preceding context on inhibition: an event-related fMRI study. Neuroimage. 2002;16(2):449–453. doi: 10.1006/nimg.2002.1074. [DOI] [PubMed] [Google Scholar]
- Eaton LK, Kann L, Kinchen S, Shanklin S, Ross J, Hawkins J, et al. Youth Risk Behavior Surveillance - United States, 2007, surveillance summaries. Morbidity and Mortality Weekly Report. 2008;57(SS04):1–131. [PubMed] [Google Scholar]
- Eigsti IM, Zayas V, Mischel W, Shoda Y, Ayduk O, Dadlani MB, et al. Predicting cognitive control from preschool to late adolescence and young adulthood. Psychol Sci. 2006;17(6):478–484. doi: 10.1111/j.1467-9280.2006.01732.x. [DOI] [PubMed] [Google Scholar]
- Epstein JN, Casey BJ, Tonev ST, Davidson MC, Reiss AL, Garrett A, et al. ADHD- and medication-related brain activation effects in concordantly affected parent-child dyads with ADHD. J Child Psychol Psychiatry. 2007;48(9):899–913. doi: 10.1111/j.1469-7610.2007.01761.x. [DOI] [PubMed] [Google Scholar]
- Ernst M, Nelson EE, Jazbec S, McClure EB, Monk CS, Leibenluft E, et al. Amygdala and nucleus accumbens in responses to receipt and omission of gains in adults and adolescents. Neuroimage. 2005;25(4):1279–1291. doi: 10.1016/j.neuroimage.2004.12.038. [DOI] [PubMed] [Google Scholar]
- Ernst M, Pine DS, Hardin M. Triadic model of the neurobiology of motivated behavior in adolescence. Psychol Med. 2006;36(3):299–312. doi: 10.1017/S0033291705005891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ernst M, Romeo RD, Andersen SL. Neurobiology of the development of motivated behaviors in adolescence: a window into a neural systems model. Pharmacol Biochem Behav. 2009;93(3):199–211. doi: 10.1016/j.pbb.2008.12.013. [DOI] [PubMed] [Google Scholar]
- Feld BC. A slower form of death: Implications of Roper v. Simmons for juveniles sentenced to life without parol. Notre Dame Journal of Law, Ethics, & Public Policy. 2008;22:9–65. [Google Scholar]
- Figner B, Mackinlay RJ, Wilkening F, Weber EU. Affective and deliberative processes in risky choice: age differences in risk taking in the Columbia Card Task. J Exp Psychol Learn Mem Cogn. 2009;35(3):709–730. doi: 10.1037/a0014983. [DOI] [PubMed] [Google Scholar]
- Flavell JH, Beach DR, Chinksy JM. Spontaneous verbal rehearsal in a memory task as a function of age. Child Development. 1966;37:283–299. [PubMed] [Google Scholar]
- Forbes EE, Dahl RE. Pubertal development and behavior: hormonal activation of social and motivational tendencies. Brain Cogn. 2010;72(1):66–72. doi: 10.1016/j.bandc.2009.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galvan A, Hare T, Voss H, Glover G, Casey BJ. Risk-taking and the adolescent brain: who is at risk? Dev Sci. 2007;10(2):F8–F14. doi: 10.1111/j.1467-7687.2006.00579.x. [DOI] [PubMed] [Google Scholar]
- Galvan A, Hare TA, Davidson M, Spicer J, Glover G, Casey BJ. The role of ventral frontostriatal circuitry in reward-based learning in humans. J Neurosci. 2005;25(38):8650–8656. doi: 10.1523/JNEUROSCI.2431-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galvan A, Hare TA, Parra CE, Penn J, Voss H, Glover G, et al. Earlier development of the accumbens relative to orbitofrontal cortex might underlie risk-taking behavior in adolescents. Journal of Neuroscience. 2006;26(25):6885–6892. doi: 10.1523/JNEUROSCI.1062-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner M, Steinberg L. Peer influence on risk taking, risk preference, and risky decision making in adolescence and adulthood: an experimental study. Dev Psychol. 2005;41(4):625–635. doi: 10.1037/0012-1649.41.4.625. [DOI] [PubMed] [Google Scholar]
- Geier CF, Luna B. The maturation of incentive processing and cognitive control. Pharmacol Biochem Behav. 2009;93(3):212–221. doi: 10.1016/j.pbb.2009.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geier CF, Terwilliger R, Teslovich T, Velanova K, Luna B. Immaturities in Reward Processing and Its Influence on Inhibitory Control in Adolescence. Cereb Cortex. 2009 doi: 10.1093/cercor/bhp225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giedd JN. Structural magnetic resonance imaging of the adolescent brain. Ann N Y Acad Sci. 2004;1021:77–85. doi: 10.1196/annals.1308.009. [DOI] [PubMed] [Google Scholar]
- Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A, et al. Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci. 1999;2(10):861–863. doi: 10.1038/13158. [DOI] [PubMed] [Google Scholar]
- Giedd JN, Snell JW, Lange N, Rajapakse JC, Casey BJ, Kaysen D, et al. Quantitative magnetic resonance imaging of human brain development: ages 4-18. Cerebral Cortex. 1996;6:551–560. doi: 10.1093/cercor/6.4.551. [DOI] [PubMed] [Google Scholar]
- Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC, et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proceedings of the National Academy of Sciences, USA. 2004;101(21):8174–8179. doi: 10.1073/pnas.0402680101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graber JA, Brooks-Gunn J. Puberty. In: Blechman EA, Brownell KD, editors. Behavioral medicine and women: A comprehensive handbook. Guilford Press; New York, NY: 1998. pp. 51–58. [Google Scholar]
- Hare TA, Tottenham N, Galvan A, Voss HU, Glover GH, Casey BJ. Biological substrates of emotional reactivity and regulation in adolescence during an emotional go-nogo task. Biol Psychiatry. 2008;63(10):927–934. doi: 10.1016/j.biopsych.2008.03.015015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harnishfeger KK, Bjorklund DF. The ontogeny of inhibition mechanisms: A renewed approach to cognitive development. In: Howe ML, Pasnek R, editors. Emerging themes in cognitive development. Vol. 1. Springer-Verlag; New York: 1993. [Google Scholar]
- Hikosaka K, Watanabe M. Delay activity of orbital and lateral prefrontal neurons of the monkey varying with different rewards. Cereb Cortex. 2000;10(3):263–271. doi: 10.1093/cercor/10.3.263. [DOI] [PubMed] [Google Scholar]
- Huttenlocher PR. Synaptic density in human frontal cortex - developmental changes and effects of aging. Brain Research. 1979;163:195–205. doi: 10.1016/0006-8993(79)90349-4. [DOI] [PubMed] [Google Scholar]
- Hyman SE, Malenka RC. Addiction and the brain: the neurobiology of compulsion and its persistence. Nat Rev Neurosci. 2001;2(10):695–703. doi: 10.1038/35094560. [DOI] [PubMed] [Google Scholar]
- Jernigan TL, Zisook S, Heaton RK, Moranville JT, Hesselink JR, Braff DL. Magnetic resonance imaging abnormalities in lenticular nuclei and cerebral cortex in schizophrenia. Arch Gen Psychiatry. 1991;48(10):881–890. doi: 10.1001/archpsyc.1991.01810340013002. [DOI] [PubMed] [Google Scholar]
- Keating DP, Bobbitt BL. Individual and developmental differences in cognitive processing components of mental ability. Child Development. 1978;49:155–167. [Google Scholar]
- Kelley AE, Schochet T, Landry CF. Risk taking and novelty seeking in adolescence: introduction to part I. Ann N Y Acad Sci. 2004;1021:27–32. doi: 10.1196/annals.1308.003. [DOI] [PubMed] [Google Scholar]
- Klingberg T, Forssberg H, Westerberg H. Increased brain activity in frontal and parietal cortex underlies the development of visuospatial working memory capacity during childhood. J Cogn Neurosci. 2002;14(1):1–10. doi: 10.1162/089892902317205276. [DOI] [PubMed] [Google Scholar]
- Knutson B, Adams CM, Fong GW, Hommer D. Anticipation of increasing monetary reward selectively recruits nucleus accumbens. J Neurosci. 2001;21(16):RC159. doi: 10.1523/JNEUROSCI.21-16-j0002.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhnen CM, Knutson B. The neural basis of financial risk taking. Neuron. 2005;47(5):763–770. doi: 10.1016/j.neuron.2005.08.008. [DOI] [PubMed] [Google Scholar]
- Laviola G, Adriani W, Terranova ML, Gerra G. Psychobiological risk factors for vulnerability to psychostimulants in human adolescents and animal models. Neurosci Biobehav Rev. 1999;23(7):993–1010. doi: 10.1016/s0149-7634(99)00032-9. [DOI] [PubMed] [Google Scholar]
- Laviola G, Macri S, Morley-Fletcher S, Adriani W. Risk-taking behavior in adolescent mice: psychobiological determinants and early epigenetic influence. Neurosci Biobehav Rev. 2003;27(1-2):19–31. doi: 10.1016/s0149-7634(03)00006-x. [DOI] [PubMed] [Google Scholar]
- Levita L, Hare TA, Voss HU, Glover G, Ballon DJ, Casey BJ. The bivalent side of the nucleus accumbens. Neuroimage. 2009;44(3):1178–1187. doi: 10.1016/j.neuroimage.2008.09.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liston C, Watts R, Tottenham N, Davidson MC, Niogi S, Ulug AM, et al. Frontostriatal microstructure modulates efficient recruitment of cognitive control. Cerebral Cortex. 2006;16(4):553–560. doi: 10.1093/cercor/bhj003. [DOI] [PubMed] [Google Scholar]
- Luna B, Sweeney JA. The emergence of collaborative brain function: FMRI studies of the development of response inhibition. Ann N Y Acad Sci. 2004;1021:296–309. doi: 10.1196/annals.1308.035. [DOI] [PubMed] [Google Scholar]
- Luna B, Thulborn KR, Munoz DP, Merriam EP, Garver KE, Minshew NJ, et al. Maturation of widely distributed brain function subserves cognitive development. Neuroimage. 2001;13(5):786–793. doi: 10.1006/nimg.2000.0743. [DOI] [PubMed] [Google Scholar]
- Martin CA, Logan TK, Portis C, Leukefeld CG, Lynam D, Staton M, et al. The association of testosterone with nicotine use in young adult females. Addict Behav. 2001;26(2):279–283. doi: 10.1016/s0306-4603(00)00094-0. [DOI] [PubMed] [Google Scholar]
- Matthews SC, Simmons AN, Lane SD, Paulus MP. Selective activation of the nucleus accumbens during risk-taking decision making. Neuroreport. 2004;15(13):2123–2127. doi: 10.1097/00001756-200409150-00025. [DOI] [PubMed] [Google Scholar]
- May JC, Delgado MR, Dahl RE, Stenger VA, Ryan ND, Fiez JA, et al. Event-related functional magnetic resonance imaging of reward-related brain circuitry in children and adolescents. Biological Psychiatry. 2004;55(4):359–366. doi: 10.1016/j.biopsych.2003.11.008. [DOI] [PubMed] [Google Scholar]
- McClure SM, Laibson DI, Loewenstein G, Cohen JD. Separate neural systems value immediate and delayed monetary rewards. Science. 2004;306(5695):503–507. doi: 10.1126/science.1100907. [DOI] [PubMed] [Google Scholar]
- Miller EK, Cohen JD. An integrative theory of prefrontal cortex function. Annu Rev Neurosci. 2001;24:167–202. doi: 10.1146/annurev.neuro.24.1.167. [DOI] [PubMed] [Google Scholar]
- Mischel W, Shoda Y, Rodriguez MI. Delay of gratification in children. Science. 1989;244(4907):933–938. doi: 10.1126/science.2658056. [DOI] [PubMed] [Google Scholar]
- Monk CS, McClure EB, Nelson EE, Zarahn E, Bilder RM, Leibenluff E, et al. Adolescent immaturity in attention-related brain engagement to emotional facial expressions. Neuroimage. 2003;20:420–428. doi: 10.1016/s1053-8119(03)00355-0. [DOI] [PubMed] [Google Scholar]
- Montague PR, Berns GS. Neural economics and the biological substrates of valuation. Neuron. 2002;36(2):265–284. doi: 10.1016/s0896-6273(02)00974-1. [DOI] [PubMed] [Google Scholar]
- Moses P, Roe K, Buxton RB, Wong EC, Frank LR, Stiles J. Functional MRI of global and local processing in children. Neuroimage. 2002;16(2):415–424. doi: 10.1006/nimg.2002.1064. [DOI] [PubMed] [Google Scholar]
- Munakata Y, Yerys BE. All together now: when dissociations between knowledge and action disappear. Psychol Sci. 2001;12(4):335–337. doi: 10.1111/1467-9280.00361. [DOI] [PubMed] [Google Scholar]
- Nagy Z, Westerberg H, Klingberg T. Maturation of white matter is associated with the development of cognitive functions during childhood. J Cogn Neurosci. 2004;16(7):1227–1233. doi: 10.1162/0898929041920441. [DOI] [PubMed] [Google Scholar]
- Nelson EE, Leibenluft E, McClure EB, Pine DS. The social re-orientation of adolescence: a neuroscience perspective on the process and its relation to psychopathology. Psychol Med. 2005;35(2):163–174. doi: 10.1017/s0033291704003915. [DOI] [PubMed] [Google Scholar]
- O'Doherty JP. Reward representations and reward-related learning in the human brain: insights from neuroimaging. Curr Opin Neurobiol. 2004;14(6):769–776. doi: 10.1016/j.conb.2004.10.016. [DOI] [PubMed] [Google Scholar]
- O'Doherty JP, Kringelbach ML, Rolls ET, Hornak J, Andrews C. Abstract reward and punishment representations in the human orbitofrontal cortex. Nat Neurosci. 2001;4(1):95–102. doi: 10.1038/82959. [DOI] [PubMed] [Google Scholar]
- Pascual-Leone JA. A mathematical model for transition in Piaget's developmental stages. Acta Psychologica. 1970;32:301–345. [Google Scholar]
- Pecina S, Cagniard B, Berridge KC, Aldridge JW, Zhuang X. Hyperdopaminergic mutant mice have higher “wanting” but not “liking” for sweet rewards. J Neurosci. 2003;23(28):9395–9402. doi: 10.1523/JNEUROSCI.23-28-09395.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakic P. e. a. Synaptic development of the cerebral cortex: implications for learning, memory and mental illness. Prog. Brain Res. 1994;102:227–243. doi: 10.1016/S0079-6123(08)60543-9. [DOI] [PubMed] [Google Scholar]
- Romeo RD. Puberty: a period of both organizational and activational effects of steroid hormones on neurobehavioural development. J Neuroendocrinol. 2003;15(12):1185–1192. doi: 10.1111/j.1365-2826.2003.01106.x. [DOI] [PubMed] [Google Scholar]
- Rosenberg DR, Lewis DA. Postnatal maturation of the dopaminergic innervation of monkey prefrontal and motor cortices: a tyrosine hydroxylase immunohistochemical analysis. J Comp Neurol. 1995;358(3):383–400. doi: 10.1002/cne.903580306. [DOI] [PubMed] [Google Scholar]
- Rubia K, Overmeyer S, Taylor E, Brammer M, Williams SC, Simmons A, et al. Functional frontalisation with age: mapping neurodevelopmental trajectories with fMRI. Neurosci Biobehav Rev. 2000;24(1):13–19. doi: 10.1016/s0149-7634(99)00055-x. [DOI] [PubMed] [Google Scholar]
- Schlaggar BL, Brown TT, Lugar HM, Visscher KM, Miezin FM, Petersen SE. Functional neuroanatomical differences between adults and school-age children in the processing of single words. Science. 2002;296(5572):1476–1479. doi: 10.1126/science.1069464. [DOI] [PubMed] [Google Scholar]
- Schultz W. Behavioral theories and the neurophysiology of reward. Annu Rev Psychol. 2006;57:87–115. doi: 10.1146/annurev.psych.56.091103.070229. [DOI] [PubMed] [Google Scholar]
- Scott ES. Judgment and Reasoning in Adolescent Decisionmaking. Villanova Law Review. 1992;37:1607–1669. [PubMed] [Google Scholar]
- Somerville LH, Hare TA, Casey BJ. Frontostriatal maturation predicts cognitive control failure to appetitive cues in adolescents. Journal of Cognitive Neuroscience. doi: 10.1162/jocn.2010.21572. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somerville LH, Jones RM, Casey BJ. A time of change: behavioral and neural correlates of adolescent sensitivity to appetitive and aversive environmental cues. Brain Cogn. 2010;72(1):124–133. doi: 10.1016/j.bandc.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sowell ER, Peterson BS, Thompson PM, Welcome SE, Henkenius AL, Toga AW. Mapping cortical change across the human life span. Nat Neurosci. 2003;6(3):309–315. doi: 10.1038/nn1008. [DOI] [PubMed] [Google Scholar]
- Sowell ER, Thompson PM, Holmes CJ, Jernigan TL, Toga AW. In vivo evidence for post-adolescent brain maturation in frontal and striatal regions. Nat Neurosci. 1999;2(10):859–861. doi: 10.1038/13154. [DOI] [PubMed] [Google Scholar]
- Sowell ER, Thompson PM, Toga AW. Mapping changes in the human cortex throughout the span of life. Neuroscientist. 2004;10(4):372–392. doi: 10.1177/1073858404263960. [DOI] [PubMed] [Google Scholar]
- Spear LP. The adolescent brain and age-related behavioral manifestations. Neuroscience and Biobehavioral Reviews. 2000;24(4):417–463. doi: 10.1016/s0149-7634(00)00014-2. [DOI] [PubMed] [Google Scholar]
- Spear LP. The Behavioral Neuroscience of Adolescence. W.W. Norton & Company; New York: 2009. [Google Scholar]
- Steinberg L. Risk taking in adolescence: what changes, and why? Ann N Y Acad Sci. 2004;1021:51–58. doi: 10.1196/annals.1308.005. [DOI] [PubMed] [Google Scholar]
- Steinberg L. A social neuroscience perspective on adolescent risk-taking. Developmental Review. 2008;28:78–106. doi: 10.1016/j.dr.2007.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg L, Albert D, Cauffman E, Banich M, Graham S, Woolard J. Age differences in sensation seeking and impulsivity as indexed by behavior and self-report: evidence for a dual systems model. Dev Psychol. 2008;44(6):1764–1778. doi: 10.1037/a0012955. [DOI] [PubMed] [Google Scholar]
- Steinberg L, Graham S, O'Brien L, Woolard J, Cauffman E, Banich M. Age differences in future orientation and delay discounting. Child Dev. 2009;80(1):28–44. doi: 10.1111/j.1467-8624.2008.01244.x. [DOI] [PubMed] [Google Scholar]
- Tamm L, Menon V, Reiss AL. Maturation of brain function associated with response inhibition. J Am Acad Child Adolesc Psychiatry. 2002;41(10):1231–1238. doi: 10.1097/00004583-200210000-00013. [DOI] [PubMed] [Google Scholar]
- Thomas KM, Hunt RH, Vizueta N, Sommer T, Durston S, Yang Y, et al. Evidence of developmental differences in implicit sequence learning: an fMRI study of children and adults. J Cogn Neurosci. 2004;16(8):1339–1351. doi: 10.1162/0898929042304688. [DOI] [PubMed] [Google Scholar]
- Turkeltaub PE, Gareau L, Flowers DL, Zeffiro TA, Eden GF. Development of neural mechanisms for reading. Nat Neurosci. 2003;6(7):767–773. doi: 10.1038/nn1065. [DOI] [PubMed] [Google Scholar]
- Van Leijenhorst L, Moor BG, Op de Macks ZA, Rombouts SA, Westenberg PM, Crone EA. Adolescent risky decision-making: Neurocognitive development of reward and control regions. Neuroimage. 2010 doi: 10.1016/j.neuroimage.2010.02.038. [DOI] [PubMed] [Google Scholar]
- Van Leijenhorst L, Zanolie K, Van Meel CS, Westenberg PM, Rombouts SA, Crone EA. What motivates the adolescent? Brain regions mediating reward sensitivity across adolescence. Cereb Cortex. 2010;20(1):61–69. doi: 10.1093/cercor/bhp078. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Li TK. Drug addiction: the neurobiology of behaviour gone awry. Nat Rev Neurosci. 2004;5(12):963–970. doi: 10.1038/nrn1539. [DOI] [PubMed] [Google Scholar]
- Windle M, Spear LP, Fuligni AJ, Angold A, Brown JD, Pine D, et al. Transitions into underage and problem drinking: developmental processes and mechanisms between 10 and 15 years of age. Pediatrics. 2008;121:S273–S289. doi: 10.1542/peds.2007-2243C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zald DH, Boileau I, El-Dearedy W, Gunn R, McGlone F, Dichter GS, et al. Dopamine transmission in the human striatum during monetary reward tasks. J Neurosci. 2004;24(17):4105–4112. doi: 10.1523/JNEUROSCI.4643-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]