Abstract
The inhibitory growth environment of myelin and extracellular matrix proteoglycans in the central nervous system may be overcome by elevating neuronal cAMP or degrading inhibitory proteoglycans with chondroitinase ABC (ChABC). In this study, we asked whether similar mechanisms operate in peripheral nerve regeneration where effective Wallerian degeneration removes myelin and extracellular proteoglycans slowly. We repaired transected common peroneal (CP) nerve in rats and either elevated cAMP in the axotomized neurons by subcutaneous rolipram, a specific inhibitor of phosphodiesterase IV, and/or promoted degradation of proteoglycans in the distal nerve stump by local ChABC administration. Rolipram treatment significantly increased the number of motoneurons that regenerated axons across the repair site at one and two weeks, and increased the number of sensory neurons that regenerated axons across the repair site at two weeks. Local application of ChABC had a similar effect to rolipram treatment in promoting motor axon regeneration, the effect being no greater when rolipram and ChABC were administered simultaneously. We conclude that blocking inhibitors of axon regeneration by elevating cAMP or degrading proteoglycans in the distal nerve stump promotes peripheral axon regeneration after surgical repair of a transected nerve. It is likely that elevated cAMP is sufficient to encourage axon outgrowth despite the the inhibitory growth environment such that simultaneous enzymatic proteoglycan degradation does not promote more axon regeneration than either elevated cAMP or proteoglycan degradation alone.
Keywords: cAMP, chondroitinase ABC, axon regeneration, axon outgrowth, staggered regeneration, motoneurons, DRG neurons
INTRODUCTION
Axons in the peripheral (PNS) but not the central nervous system (CNS) regenerate after injury (Fu and Gordon, 1995, Fu and Gordon, 1995, Fu and Gordon, 1997). Nevertheless there is only a short window of opportunity for effective PNS regeneration to occur, whereby regenerative success of injured nerves progressively decreases after prolonged axotomy and Schwann cell denervation (Fu and Gordon, 1995, Fu and Gordon, 1995, Gordon, et al., 2003, Sulaiman and Gordon, 2000). Axon outgrowth from the proximal stump of transected and surgically repaired nerves is a slow process (Brushart, et al., 2002) and this lengthy period when regenerating axons wander in the suture site of surgically repaired peripheral nerves (Cajal, 1928, Witzel, et al., 2005) accounts, at least in part, for the delays of several weeks for all regenerating axons to cross a repair site (Brushart, et al., 2002). Chondroitin sulfate proteoglycans (CSPGs) of the extracellular matrix and myelin-associated inhibitors, both potent inhibitors of neuronal regeneration in the CNS(Mueller, 1999, Sandvig, et al., 2004, Tang, 2003), are also present in the peripheral nerve. These molecules are up-regulated after nerve injury, show neurite-inhibitory activity (Braunewell, et al., 1995, Shen, et al., 1998, Zuo, et al., 1998), and may play a role in delayed axon outgrowth following peripheral nerve injury. The prolonged time course of weeks for effective removal of myelin debris by macrophages and Schwann cells (Avellino, et al., 1995, Fansa and Keilhoff, 2003, George and Griffin, 1994, Stoll, et al., 1989) and for degradation of glycoproteins of the extracellular matrix (Hughes, et al., 2002) may account for the staggered outgrowth of axons from the proximal stump of an injured nerve (Al-Majed, et al., 2000, Gordon, et al., 2003).
Injured nerves in the CNS may be stimulated to regenerate their axons despite the inhibitory environment by interfering with signaling pathways that are activated by inhibitory myelin associated molecules (Cai, et al., 1999, Dergham, et al., 2002, Lehmann, et al., 1999, Neumann, et al., 2002, Neumann and Woolf, 1999, Qiu, et al., 2002). The inhibition can be overcome by increasing neuronal cAMP levels in vivo (Cai, et al., 1999, Dergham, et al., 2002, Lehmann, et al., 1999, Neumann, et al., 2002, Neumann and Woolf, 1999, Qiu, et al., 2002) and in vitro (Cai, et al., 1999) and by selectively cleaving glycosaminoglycan (GAG) side chains from the protein core of proteoglycans with chondroitinase ABC (ChABC)(Fawcett and Asher, 1999). However, applying these pharmacological approaches to PNS regeneration has generated conflicting data. Some have reported that cAMP does promote axon outgrowth (Gershenbaum and Roisen, 1980, Kilmer and Carlsen, 1987, Pichichero, et al., 1973) while others have failed to demonstrate increased axon outgrowth (Black and Lasek, 1979, Han, et al., 2004, McQuarrie, et al., 1977). The use of ChABC in the PNS has been shown to accelerate axon outgrowth into distal nerve stumps and acellular nerve grafts in rats (Krekoski, et al., 2001, Zuo, et al., 2002). Analyses of the distance of regenerating axons of thy-1 TFP-H transgenic mice demonstrated longer axon profiles in nerve grafts from wild-type littermates with ChABC, which specifically degrades CSPGs but not other proteoglycans such as heparin sulfate proteoglycans (Groves, et al., 2005).
Using retrograde labeling techniques, we aim to further investigate whether elevation of cAMP or removal of GAG side chains from inhibitory proteoglycans in axotomized motor and sensory neurons in the peripheral nerve promotes PNS regeneration. We also question whether a combinational strategy of rolipram and ChABC that has proven to be effective in CNS regeneration (Fouad, et al., 2005, Houle, et al., 2006, Tropea, et al., 2003), is also effective in the PNS.
MATERIAL AND METHODS
All experiments were performed on adult female Sprague Dawley rats (200–220g) and approved by local authorities (Health Sciences Laboratory Animal Services, University of Alberta) according to the Canadian Council for Animal Care guidelines.
Surgery: Nerve repair and delivery of rolipram and/or chondroitinase ABC (ChABC)
All surgeries were performed under Ketamine (Vetalar, Bioniche, Bellville, Ontario) and Xylazine (Rompun, Bayer, Toronto, Ontario) anesthesia at doses of 0.6 and 0.4 mg/kg intraperitoneally respectively. Eye lubricant (Duratears Naturale, Alcon, Ontario) was used to prevent corneal damage during surgery.
Using aseptic technique, the right CP nerve was exposed distal to the sciatic notch and transected 10 mm proximal to its entrance into the flexor muscle group including the tibialis anterior muscle (Fig 1A). Silicone silastic guides (Helix Medical, Inc, Carpinteria, CA) of 0.76 mm interior diameter and 3 mm length were implanted, and the proximal and distal CP nerve stumps were approximated within the nerve guide using 9-0 Ethicon nylon suture (Ethicon, Inc Somerville, NJ, USA) as described in Furey at al (Furey, et al., 2007). An Alzet mini-osmotic pump (Durect Corporation, Cupertino, CA) was then implanted subcutaneously on the back of the rat to release its content of either rolipram or saline systemically. The Alzet pump (model 2ML2) was filled with the appropriate solution for 24 hours and incubated at 37 °C prior to implantation. The pump delivered either rolipram (Nikulina, et al., 2004) diluted in 1:1 Saline/Dimethyl sulfoxide (DMSO) (rolipram group, n=26) or vehicle solution (1:1 Saline/DMSO; control group, n=22), both delivered at a rate of 0.4 μmol/kg/h continuously over 7, 14 or 21 days. The surgical wound was sutured closed and the rats were monitored and kept warm until they recovered from the anesthetic.
Figure 1.
The in vivo model of peripheral nerve regeneration in the common peroneal (CP) nerve in the rat. A. The CP nerve was exposed, transected 10 mm proximal to the nerve entry into the flexor group of muscles that includes the tibialis anterior muscle, and repaired by tubulization. The Alzet osmotic pump was implanted subcutaneously at the back of the rats after the nerve surgery. B. After 7 or 14 days, the nerve was re-exposed, crushed 3 mm distal to the suture site and fluroruby was injected into the nerve via a micropipette that was attached to a picospritzer. C. After 14 or 21 days, the nerve was re-exposed, cut 10 mm distal to the suture site and the proximal stump was dipped for 1h in a solution with fluororuby contained in a vaseline pool. Details are provided in the text.
Chondroitinase ABC application
In 2 groups of rats, the CP nerve was exposed and transected under surgical anesthesia 10 mm proximal to the entrance of the nerve into the flexor muscle group of the hindlimb. In the first group of animals, 5 mm of the cut end of the distal stump was dipped into a Vaseline well containing 20U/ml protease free ChABC (Seikagaku, Japan) in 1% protease free bovine serum albumin for 1h. The nerve ends were then repaired using a silicone silastic guide. An Alzet mini-osmotic pump was then placed on the back of the animal and a vehicle solution of 1:1 Saline/DMSO was delivered systemically over 2 weeks at a delivery rate of 5 μl/ml (ChABC group n=9). In the second group of rats, the ChABC was again applied to the distal nerve stump as described, and an Alzet pump implanted on the back of the rat was filled with rolipram solution for systemic delivery over a 2 week period at the same rate as for the vehicle solution group (Roli+ChABC group, n=8). The skin was closed after surgery and the rats were warmed and monitored during recovery from the anesthetic.
Surgical application of retrograde dyes
A second sterile surgery was carried out under surgical anesthesia at 7 to 21 days following the CP nerve section and repair, and for the systemic delivery of saline/DMSO vehicle or rolipram, and/or local application of ChABC to the CP nerve suture site. Fluororuby (Dextran, tetramethylrodamine, Invitrogen, Molecular Probes, Eugene, Oregon) was either injected 3 mm distal to the site of the nerve repair or applied directly to the proximal stump of the CP nerve 10 mm distal to the repair site (Fig. 1B,C). For the microinjection, the CP nerve was crushed just distal to the cuff (3 mm distal to the suture site) and the dye was injected via a micropipette that was attached to a picospritzer (Intercel Picospritzer III) (Brushart, et al., 2002). For the application of the dye to the proximal stump of the CP nerve cut 10 mm distal to the suture site, the stump was dipped into a 5% Fluororuby solution contained within a vaseline well for 1 hour. Following the dye exposure, excess dye was carefully rinsed off before suturing the skin and allowing the rats to wake. In control experiments, we injected the dye at 3 mm distal to the suture site immediately after nerve repair rather than after 7, 14, and 21 days. This was done to ensure that the injected dye was confined to the injection site within the distal nerve stump and did not penetrate to the axons in the proximal nerve stump.
Tissue removal and analysis of the backlabeled neurons
Rats were deeply anesthetized 5–6 days after back-labeling of the neurons and transcardially perfused with 200 ml saline followed by 500 ml of 4% paraformaldehyde at pH 7.4. After perfusion, the spinal cord was dissected and spinal cord segments T11 to L2 (containing the CP motoneuron pool) and, L4 and L5 dorsal root ganglia (DRGs) (containing most of the cell bodies of the CP sensory nerves) were harvested and post-fixed with 30% sucrose in 4% paraformaldehyde solution overnight. The tissue was then frozen in liquid nitrogen after been embedded in OCT Tissue-Tek Liquid (Sakura, Japan).
Frozen tissues were sectioned in a cryostat (Jung 3000). Longitudinal spinal cord sections were cut at 50 μm thickness and DRG cross-sections were cut at 25 μm thickness. The fluorescent bodies of the labeled motor and sensory neurons of the CP nerve were visualized and counted at 40× magnification under fluorescent microscopy at barrier filters of 580 nm (Fig 2). The number of neurons counted was corrected according to the thickness of the sections and the diameter of the neuron cell bodies (30 μm and 15 μm for motor and DRG neurons respectively) by the method of Abercrombie and Johnson (Abercrombie and Johnson, 1946). The correction factor in our samples was 0.635 for motoneuron counts in the spinal cord sections and 0.574 for the DRG sensory neurons.
Figure 2.
Visualization of motor and sensory neurons backlabeled with fluororuby. A,B. An example of a column of common peroneal motoneurons in 50 μm thick longitudinal sections of the spinal cord at the lumbar level. The neurons are visualized at low and higher magnification to illustrate the laterally located motoneuron column in the ventral horn and the morphology of the motoneurons, respectively B. Sensory neurons in 25 μm thick cross-sections of the L5 dorsal root ganglion. The scale bars are A) 200 μm, B) 50 μm and C) 20 μm.
Morphological analysis of nerve sections
In a third set of rats, surgery was performed to administer rolipram (n=9) or vehicle solution (n=9) immediately after CP nerve transection and repair via an Alzet miniosmotic pump for periods of 4 or 14 days. The surgeries were the same as described for the previous sets of rats. A segment of the CP nerve 3 mm distal to the suture site was removed at 4 day and 14 day time points. The nerve pieces were fixed with gluteraldehyde (3% in 0.1 M phosphate buffer), stained with osmium tetroxide (3% solution in 0.1 M phosphate buffer), dehydrated in ascending alcohols, and embedded in araldite. Sections of 2 μm thickness were stained with methylene blue and observed under the light microscope with a 100X oil-objective lens. In both groups, the number of myelinated fibers with normal appearance and the number of myelinated fibers undergoing Wallerian degeneration were counted to obtain a percentage of degenerative versus intact fibers in the nerve (Ceballos, et al., 2003). The number of phagocytic cells present in the cross-section area was also counted.
Degradation of proteoglycans by ChABC
To guarantee that the ChABC application was successful in degrading the proteoglycans of the extracellular matrix, immunohistochemistry was performed to determine the success of the ChABC in degrading CSPG. CP nerve cross-sections were immunolabeled using a CSPG-neoepitope antibody that binds to new epitopes created on the CSPG core protein after its lysis by chABC (adapted from García-Alías et al 2008). Nerves that were treated with the same concentration of chABC used in the studies of axon regeneration in 4 rats, were fixed with 4% paraformaldhyde and then preserved in 30% PBS-sucrose. Cross-sections of 30 μm were cut in a cryostat. Nerve sections were blocked in PBS with 0.3 % Triton X and 5% FCS, for 1h, then incubated overnight at 4 °C with Proteoglycan Delta Di0S (1B5) anti-mouse (Seikagakubb, 1:5000), incubated for 1h at room temperature with biotinylated goat anti-mouse secondary antibody (1:200), and then anti-goat Cyanine-2 (1:200, Jackson Immunoresearch) for 1h. Samples were washed 3 times with PBS after each step.
Measurement of cAMP levels in DRG neurons and spinal cord homogenates
In the rats with the distal nerve stump removed 4 days after nerve transection and repair for morphological analysis (described above), the right L4 and L5 DRGs were removed and immediately frozen in liquid nitrogen. The segment of the spinal cord where L4 and L5 roots enter was also removed, and the left and right sides were separated and frozen in liquid nitrogen. The tissues were homogenized in 0.1 N hydrochloric acid (HCl) and cAMP levels were measured with a cAMP immunoassay kit (R&D Systems Inc., MN, USA).
Statistics
Statistical comparisons were performed using SPSS 14.0 software. Comparisons between 2 groups were performed using an independent t-test, whereas comparison between 4 groups were performed using one-way analysis of variance (ANOVA), and statistical significance was accepted at p values of <0.05 (*) and <0.01 (**). Values are expressed as means ± SE.
RESULTS
Rolipram delivery accelerates motor and sensory nerve regeneration
In order to increase cellular levels of cAMP pharmacologically in motor and sensory neurons, we infused rolipram systemically at a rate of 0.4 μmol/kg/h (Nikulina, et al., 2004) to inhibit PDE IV, the most common isoform of phosphodiesterase in neural tissue (Jin, et al., 1999). One week after transecting and repairing the nerve, a thin regenerative cord connecting the proximal and the distal stumps was observed through the silicone guide. After 2 to 3 weeks, there was more connective tissue surrounding the guide and the distal stump was obviously thicker and pearly.
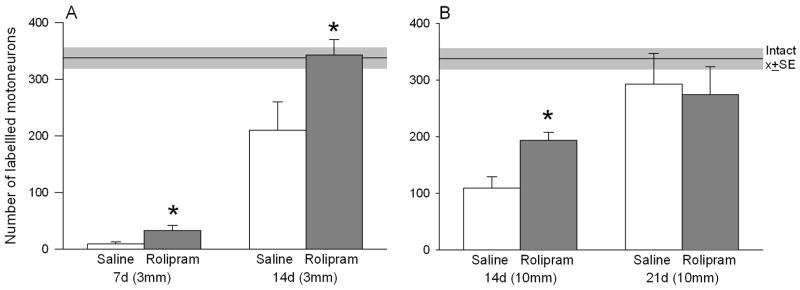
Elevation of cAMP by rolipram has been shown to increase neurite outgrowth of DRG neurons on a non-permissive substrate in vitro (Cai, et al., 1999) but it remains to be determined whether cAMP plays a definitive role in peripheral nerve regeneration in vivo. We asked whether rolipram delivery to regenerating axons promotes axon outgrowth across a surgical repair site and into the distal nerve stump. Rolipram was delivered systemically for 7, 14 and 21 days, after which the motor and sensory neurons that had regenerated their axons across the suture site and into the distal nerve stump were backlabeled with fluororuby either 3 mm or 10 mm distal to the surgical site (Fig. 1). When the fluorescent dye was applied at a distance of 3 mm from the suture site immeditaely after nerve repair, the counts of labeled motoneurons in the spinal cord or sensory neurons in the DRGs were zero. This control experiment demonstrated that the injected retrograde dye was confined within the distal nerve stump. Hence counts of labeled neurons days after nerve transection and repair represented only those neurons whose axons had regenerated across the suture site to the site of dye application. At 7 days after nerve repair, only a few motoneurons were backlabeled with fluororuby 3 mm distal to the repair site. In the vehicle saline treated group, a mean (±SE) of 9.1 ± 3.2 motoneurons were back-labeled in the ventral horn of the right spinal cord (Fig. 3A) whereas with rolipram treatment, the mean (±SE) number of motoneurons was 32.8 ± 9.3, a significant 3.6 fold increase compared to the vehicle group. There were no back-labeled motoneurons in the spinal cord in 2 of 9 animals in the vehicle group and in 1 of 10 animals in the rolipram group. In other words, there were no motoneurons which regenerated their axons across the surgical site at that time point in ~22% and 10% of the vehicle and rolipram-treated rats respectively. One week later, at 14 days after nerve repair and either vehicle or rolipram treatment, all motoneurons had regenerated their axons to 3 mm distal to the suture site in the rolipram treated group with the mean (±SE) number of 343.5 ± 27.1, falling within one SE of the mean number of intact CP motoneurons on the contralateral side of the spinal cord (Fig. 3A). In contrast, the mean (±SE) number of motoneurons in the vehicle treated group (210.9 ± 50.6) was significantly lower. Hence, systemic rolipram administration significantly increased the number of motoneurons that regenerated axons across the surgical repair site.
Figure 3.
Subcutaneous rolipram administration accelerates the onset of motor regeneration after transection and repair of the CP nerve. A. Mean number (±SE) of motoneurons backlabeled 3 mm distal to the suture site at 7 and 14 days in the vehicle (white bars) and the rolipram (grey bars) treated groups. B. Mean number (±SE) of motoneurons backlabeled 10 mm distal to the suture site at 14 and 21 days in the vehicle and the rolipram treated groups. Rolipram increased the number of motoneurons that regenerated axons across the suture site and into the distal nerve stump at 7 and 14 days with all motoneurons regenerating their axons 10 mm from the suture stie by 21 days. The numbers that regenerated axons 21 days after saline and rolipram administration were not statistically different from the numbers of contralateral motoneurons that supply axons to the contralateral intact CP nerve. The dashed lines show the SE of the mean number of intact CP motoneurons. *Statistical significance vs vehicle when p<0.05.
When motor axon regeneration through the distal nerve stump was assessed by back-labeling motoneurons 10 mm distal to the suture site at 14 days following transection and repair of the CP nerve, the mean number (±SE) of motoneurons that regenerated their axons through the distal nerve stump was significantly higher in the rolipram group (193.6 ± 14.3) as compared to the vehicle treated group (109.1 ± 20.3), a ~2-fold increase (Fig 3B). All motoneurons, in both the control and rolipram group, had regenerated their axons by 21d, with mean (±SE) numbers of motoneurons not being significantly different from the number of intact motoneurons in the contralateral intact motoneuron pool (Fig 3B). These data are quite consistent with the view that rolipram accelerates axon outgrowth across the suture site and has little or no effect in accelerating the rate of regeneration through the distal nerve stump.
As expected in a nerve that has ~3 times more sensory than motor fibers (Lozeron, et al., 2004, Swett, et al., 1986), the sensory neuron count in the DRGs were 3X higher than the motoneuron count in the vehicle treated group at each of the different time points after CP nerve transection and repair. There was a positive trend for rolipram treatment to increase the number of DRG neurons that regenerated axons across the suture site at 7 days (66.3±16.8) but the increase was not significant compared to the vehicle treated group (38.3±11.8) (Fig. 4A). At 2 weeks and 10 mm distal to the suture site, the number of DRG neurons that regenerated their axons in the rolipram group was significantly higher than in the vehicle treated group (403.3±49.4 and 210.9±33.6 respectively) (Fig 4B), demonstrating a ~2-fold increase in the number of sensory neurons regenerating axons into the distal nerve stump with rolipram treatment. Thus, rolipram was equally effective in promoting both motor and sensory axon outgrowth across a surgical repair site into the distal nerve stump.
Figure 4.
Rolipram administration significantly increased the number of sensory neurons regenerating axons across the repair site at 14 days following nerve transection and repair (p<0.01). A. Mean number (±SE) of the DRG sensory neurons backlabeled 3 mm distal to the suture site at 7 days in the vehicle (white bars) and the rolipram (grey bars) treated groups. B. Mean (±SE) number of DRG sensory neurons backlabeled 10 mm distal to the suture site at 14 days in the vehicle and the rolipram treated groups.
Clear signs of Wallerian degeneration were evident at 4 days as seen in the micrographs of the distal nerve stump (Fig 5A,C). At this time point, most of the fibers (67±7%) showed signs of degeneration in the saline treated group (Fig. 5A). Some phagocytic cells could be observed in the distal nerve stumps of saline treated rats with mean (±SE) total numbers of 21±7. Rolipram treatment did not alter the proportion of degenerating nerve fibers compared to visually intact fibers (67%±12) nor the number of phagocytic cells (31±9) at 4 days when compared to the vehicle treated group. These findings provide evidence that elevated cAMP levels by rolipram administration are unlikely to affect the rate of Wallerian degeneration. At 14 days after transection and repair of the CP nerve, myelinated axons were observed in 4 of the 5 animals in the vehicle treated group 3 mm distal to the suture site, the mean (±SE) number of axons being 98±116. Axons were thinly myelinated and demonstrated the typical morphology of regenerating fibers (Fig. 5B). In contrast, all the animals in the rolipram treated group showed myelinated regenerating fibers at 14d (Fig. 5D), their numbers being significantly higher at 765±340, an ~8-fold increase. These findings are in agreement with our observations that rolipram treatment increases the number of back-labeled axons that cross the suture site at 7 and 14 days. The relative increase in myelinated fibers seen in the distal nerve stump with rolipram treatment at 14 days was 8-fold as compared to the 2-fold increase in the number of sensory and motor neurons extending axons across the suture site and into the distal nerve stump at 14 days (Fig. 3B, 4B). This demonstrates that rolipram promotes the outgrowth of multiple axon sprouts across the a surgical repair site in addition to accelerating the number of motoneurons that regenerate their axons across the suture site and into the distal nerve stump.
Figure 5.

Cross-sections of distal nerves 3 mm distal to the suture site 4d (A, B) and 14d (C, D) after saline (vehicle) (A, C) and rolipram (B, D) treatment. Quantification of proportions of nerve fibers that had undergone Wallerian degeneration at 4d and of regenerating axons were made, as detailed in the text. Regenerating axons with thinly myelinated sheaths are visible within the distal nerve stumps after CP nerve transection and surgical repair, especially from the rolipram-treated group, at this early time point of axon regeneration. The calibration bar=10 μm. Examples of degenerating axons and the regenerating axons are shown by the symbols ⇒ and →, respectively.
Chondroitinase ABC degrades proteoglycans in vivo promotes peripheral nerve regeneration
ChABC degrades CSPGs present in the endoneurial tissues of the peripheral nerve after nerve injury (Krekoski, et al., 2001, Zuo, et al., 2002). This degradation occurs without compromising the basal lamina scaffold and does not disturb laminin content. The fact that ChABC injected 2 mm distal to the injury site in peripheral nerve accelerates detection of immunoreactive regenerating axons at the site of nerve transection repair, but not after crush injury, strongly indicates that the bacterial enzyme accelerates axon outgrowth rather than rate of regeneration within the nerve stumps (Krekoski, et al., 2001, Zuo, et al., 2002). We treated the distal CP nerve stump with 20U/ml of ChABC, the same dose that was shown to be effective in promoting motor axonal regeneration in the mouse (English, 2005, Groves, et al., 2005) and in degrading CSPGs in the rat sciatic nerve (Krekoski, et al., 2001, Zuo, et al., 2002).
In order to evaluate the success of the in vivo chABC application in degrading the proteoglycans in the CP distal nerve stump, we immunolabeled 4S and 6S chondroitin and dermatan sulphate, new epitopes created on the CSPG core protein after its lysis by chABC. In comparison with there being no immunoreactivity in the intact nerve (Fig. 6A), immunoreactivity that was localized around the nerve fibers in the extracellular space was detected both close to the suture site (Fig. 6B) and 5 mm distal to the site (Fig. 6C). This verified that the in vivo application of ChABC did successfully degraded proteoglycans of the extracellular matrix.
Figure 6.
Cross-sections of an intact CP nerve (A) or nerves treated application of chABC for 1h and visualized 1 day later 3 mm (B) or 8 mm distal (C) to the suture site. Sections were immunolabeled against 4S and 6S chondroitin and dermatan sulfate, degradation products of the proteolgycans. Immunoreactivity is only detectable in the treated nerves (compare A with B and C) both close (B) and 8 mm distal (C) to the suture site, corroborating that chABC is effective in degrading proteoglycans along all the treated nerve. The calibration bar=0.1 mm.
Treatment with ChABC significantly increased the number of motoneurons that regenerated their axons through the distal nerve stump at 14 days post nerve repair as compared with the vehicle saline control (Fig 7A). This effect was similar to the effect of rolipram administration over the same period of time. Hence, ChABC and rolipram, delivered separately, were equally effective in significantly increasing the number of motoneurons that regenerated their axons 10 mm into the distal CP nerve stump (Fig. 7A). When a combination of one time local ChABC application to the CP nerve and continuous systemic delivery of rolipram over 14 days was performed, the increase in the number of motoneurons regenerating their axons was the same as with the application of each agent alone, there being no additive effect when combining rolipram with ChABC (Fig 7A).
Figure 7.
Rolipram and ChABC significantly increased the number of motor and sensory neurons that regenerated their axons across the CP nerve transection and repair site. Combination therapy with both agents did not result in an additive effect above each agent alone. The mean (±SE) number of motoneurons (A) and DRG sensory neurons (B) that were backlabeled 10 mm distal to the suture site 14d after the injury in the vehicle treated group (white bar), the rolipram group (grey bar), the ChABC group (white stripped bar) and the group that received both rolipram treatment and ChABC application (ChABC+Roli, grey stripped bar). Rolipram or ChABC significantly increased the number of motoneurons and sensory neurons that regenerated their axons 10 mm into the distal nerve stump at 14 days, and the combination of the two treatments had similar effects to each of the two treatments applied separately. *Statistical significance vs vehicle when p<0.05 and ** when the p<0.01..
Similar to that observed in motoneurons, ChABC was equally effective in promoting sensory axon regeneration as treatment with rolipram (Fig. 7B). Treatment with ChABC significantly increased the number of DRG sensory neurons that regenerated their axons across the surgical repair site (342.3±68.0) when compared to the vehicle treatment group (210.9±33.6) at 10 mm distal to the repair site. As observed in the motoneurons, combination therapy of local ChABC and systemic rolipram was not additive in effect. Possible reasons for this are considered in the discussion.
cAMP levels in DRG neurons after systemic rolipram administration
In order to verify that systemic rolipram administration was effective in raising neuronal cAMP levels, we prepared another group of rats where the CP nerve was transected and surgically repaired with either saline solution or rolipram delivered systemically via a mini-osmotic pump over 4 days. After 4 days, we harvested the lumbar portion of the spinal cord containing the axotomized CP motoneurons (T12 to L1) and the L4 and L5 DRGs supplying the right hindlimb. The corresponding tissues supplying the left hindlimb with intact CP nerve were harvested. Rolipram significantly increased cAMP levels in the right hemi-sected portion of the spinal cord containing the axotomized CP motoneurons (212.2±30.7 pmol/ml) when compared to the saline group (87.8 ± 30.7 pmol/ml) (Fig. 8A). The same increase with systemic rolipram treatment was observed in the left hemi-sected cord containing the intact motoneurons compared to the saline control. In the right axotomized DRG sensory neurons, there was a similar significant increase in cAMP but the trend to increase cAMP in the left intact DRG sensory neurons did not reach statistical significance (Fig. 8B).
Figure 8.

Rolipram significantly increased intracellular levels of cAMP in axotomized motor (A) and sensory (B) neurons. Intracellular levels of cAMP from L4–L5 DRG homogenates and from hemisected T12-L1 spinal cord segments containing the CP motoneurons from the left intact hindlimb were measured. The neurons were axotomized by cutting and resuturing the right CP nerve (N-N resuture) and the neurons on the contralateral side served as intact controls. Assays were performed 4 days after surgery and treatment with either saline (white bar) or rolipram (grey bar). Rolipram significantly increased cAMP levels in both intact and axotomized motoneurons but only in the axotomized DRG sensory neurons. Mean (±SE) values of cAMP concentrations are plotted in the histograms. Statistical significance p<0.05 (*) or p<0.01 (**).
DISCUSSION
Local inhibitors of regeneration in the distal nerve stump of a transected nerve are a temporal impediment to regeneration in the PNS before Wallerian degeneration rapidly degrades proteoglycans in the extracellular matrix (Hughes, et al., 2002) and clears myelin debris (Fu and Gordon, 1997). However, the onset of Wallerian degeneration is slowed by the delayed entry of macrophages into the distal nerve stump (Bruck, 1997). We provide evidence here that overcoming the inhibitory effects of myelin by rolipram-mediated elevation of cAMP, or the inhibitory effects of proteoglycans through their degradation by ChABC, accelerates the onset of axon regeneration by promoting motor and sensory axon outgrowth across a surgical repair site.
Rolipram delivered at the same concentration reported to be effective in CNS axon regeneration (Nikulina, et al., 2004) accelerated motor and sensory axon outgrowth across a surgical site of peripheral nerve repair. Rolipram administration resulted in a ~3.6-fold increase in the number of motoneurons whose axons had crossed the suture site at 1 week while resulting in a lesser ~2-fold increase in number of motoneurons that regenerated their axons through the distal nerve stump at 2 weeks. The temporal attenuation of effect provides convincing evidence that the primary effect of rolipram is to promote axon outgrowth across a suture site rather than to increase the rate of axon regeneration, similar to that seen with a 1h period of low frequency electrical stimulation of motor and sensory neurons just after surgical repair of a transected peripheral nerve (Brushart, et al., 2002, Brushart, et al., 2005, Geremia, et al., 2007). Accordingly, studies have reported that increased cAMP levels do not enhance the rate of elongation of peripheral motor (Black and Lasek, 1979) and sensory axons (Han, et al., 2004, McQuarrie, et al., 1977).
The effect of the rolipram in rats essentially mimicked the effectiveness of electrical stimulation in accelerating axon outgrowth in both the PNS and CNS whilst not affecting the rate of axonal extension within the distal nerve stumps (Brushart, et al., 2002, Brushart, et al., 2005, Geremia, et al., 2007, Udina, et al., 2008). The accelerated axon outgrowth following electrical stimulation has also been linked to increased neuronal cAMP levels (Ming, et al., 2001, Udina, et al., 2008) and thus rolipram and low frequency electrical stimulation may promote axon outgrowth along similar pathways. It is possible that a more sustained increase in cellular cAMP achieved by continuous rolipram administration accounts for a higher percentage of motoneurons that regenerated their axons across the suture site at 2 weeks in the rolipram treated rats as compared to the application of electrical stimulation for 1h just after nerve repair.
Although effects of systemically delivered rolipram at the site of nerve transection and repair could enhance proliferation of Schwann cells (Levi, et al., 1995, Raff, et al., 1978) and has been reported to accelerate Wallerian degeneration (Gershenbaum and Roisen, 1980), this is unlikely to have made significant contributions to the accelerated outgrowth that was seen at 7 and 14 days after nerve repair and rolipram treatment. Our findings of clear signs of Wallerian degeneration, including the presence of phagocytes and axons undergoing Wallerian degeneration in the distal CP nerve stump, was the same whether or not rolipram was administered.
Rolipram induced elevation of cAMP in axotomized neurons most likely overcomes inhibition from myelin debris and inhibitory CSPGs initially encountered by regenerating axons at the distal nerve stump (Shen, et al., 1998). In the PNS, probably the main myelin associated inhibitor protein is MAG (myelin associated glycoprotein (Shen, et al., 1998)), whilst NogoR1 (the receptor of Nogo) is not present in regenerating peripheral axons. In fact, recent findings suggest that NogoR1 plays an important role in macrophage recruitment during Wallerian degeneration and would not adversely affect axon regeneration (David, et al., 2008). Thus rolipram induced elevation of cAMP in axotomized, neurons most likely overcomes inhibition from MAG contained in myelin debris initially encountered by regenerating axons at the distal nerve stump (Shen, et al., 1998). However, even in the absence of inhibitory molecules, rolipram-induced elevation of cAMP in motoneurons in vitro promotes the outgrowth and elongation of neurites (Aglah, et al., 2008). These recent findings support the view that the effectiveness of rolipram in promoting axon outgrowth in vivo is mediated at the neuronal cell body level rather than at the local site of axonal injury. Whilst cAMP promotes Schwann cell division, the same extent of Wallerian degeneration in the rolipram and saline control groups supports the view that the primary effect of rolipram is at the neuronal cell body.
Our finding that proteoglycan breakdown by localized application of ChABC was equally effective in accelerating motor and sensory axon regeneration as rolipram administration, provides evidence that rolipram and ChABC mediate their growth promoting effects at different locations. In contrast to cAMP, less is known about the mechanism(s) through which proteoglycans inhibit axon regeneration (Sandvig, et al., 2004). CSPGs are abundant and rapidly accumulate after injury in endoneurial tissue (Braunewell, et al., 1995). In the PNS, degradation of CSPGs does not seem to play a major role in nerve crush lesions but accelerates the regeneration of axons across a suture site after repair of a transected nerve (Krekoski, et al., 2001, Zuo, et al., 2002) and improves motor functional recovery (Graham, et al., 2007). In agreement with previous studies (English, 2005, Groves, et al., 2005), we report that local application of ChABC increases the number of motor and sensory neurons that regenerate their axons through the distal nerve stump. Zuo et al (Zuo, et al., 2002) attributed the effects of ChABC to the increased ability of regenerating axons to access the basal laminae of the distal nerve stump, which is left intact. Additionally, ChABC degrades CSPGs without displacing laminin in the extracellular matrix which further promotes axon outgrowth into the distal nerve stump (Bixby and Harris, 1991). CSPG degradation with ChABC treatment was effective in our study consistet with previous reports of immunohistochemical evidence of efficient degradation of CSPGs with ChABC treatment (Zuo, et al., 2002). Additionally, we performed a single treatment with ChABC at the time of nerve transection and repair, which would have degraded the CPSGs present in the distal nerve stump. However, ChABC has a relatively short half-life and thus our treatment method would have had limited impact on de novo CSPG formation in the distal nerve stump. However, the purpose of our experiment was to degrade CSPGs present in the distal nerve stump immediately after nerve transection and repair, thereby limiting the normal delay in degradation which occurs with the progression of Wallerian degeneration.
Although rolipram and ChABC each promote axon outgrowth following nerve injury and repair, the combination of rolipram and ChABC treatments did not result in an additive promotion of axon regeneration. Possible mechanisms to explain this are: (i) Rolipram induced elevation in neuronal cAMP is sufficient to overcome the inhibitory growth environment such that simultaneous enzymatic proteoglycan degradation by ChABC does not impact the outgrowth of regenerating axons; (ii) The individual effects of rolipram and ChABC result in maximum axonal regeneration such that a combination of the two agents would not result in further improvement; and (iii) Both rolipram and ChABC act at different points in the same signaling pathway. The first mechanism seems unlikely because if it where true, the individual effect on axonal regeneration of ChABC would have been less than that of rolipram, which was not observed in our data. The second mechanism is plausible, whereby individual effects on axonal regeneration were similar between the two treatment groups individually and in combination, suggesting that a maximum level of regeneration had been reached. In this explanation, both agents act through different mechanisms, rolipram at the neuronal cell body and ChABC at the local site of nerve injury (Groves, et al., 2005, Zuo, et al., 2002), to produce the same outcome. The third mechanism is also possible, whereby upregulation of cAMP by rolipram and proteoglycan degradation by ChABC both inactivate the inhibitory Rho/ROCK pathway, thereby promoting axon outgrowth via cytoskelatal formation and organization (Fig 9). Thus, the combination of rolipram and ChABC would produce the same end effect on the inhibitory pathway as each of the agents individually.
Figure 9.
Schematic representation of the cross-talk between the inhibitory and regenerative pathways in neurons. Increased cAMP levels by neurotrophic factors (NF), electrical stimulation (ES), a conditioning lesion (CL) or by administration of an inhibitor of phosphodiesterase (PDE) such as rolipram, activate the pro-regenerative pathway and block the pro-inhibitory pathway through phosphokinase A (PKA). Degradation of chondroitin sulfate proteoglycans by local application of chondroitinase ABC (cABC) blocks the inhibitory effects on regeneration induced by proteoglycans (Grados-Munro and Fournier, 2003, Mueller, 1999, Sandvig, et al., 2004, Tang, 2003).
In summary, we have demonstrated that administration of rolipram, a PDEIV specific inhibitor, immediately following peripheral nerve transection and repair was effective in increasing the number of motor and sensory neurons that regenerated axons across the repair site. Our findings are consistent with this effect being mediated through up-regulation of cAMP at the level of the neuronal cell body. Specific breakdown of CSPGs with ChABC was also shown to increase the number of motor and sensory neurons that regenerated axons across the repair site. The findings that simultaneous rolipram and ChABC did not have additive effects demonstrate that either elevation of cAMP or degradation of CSPGs alone is sufficient to overcome inhibitory environmental factors and accelerate axon outgrowth.
Acknowledgments
This work was supported by Canadian Institute for Health Research (CHIR) (T.G.) and by the National Institute of Health (T.M.B.). E.U. was recipient of a postdoctoral fellowship from the Spanish ministry of Sciences. The authors thank Guillermo García-Alías for helpful support with the immunohistochemistry.
References
- 1.Abercrombie M, Johnson ML. Quantitative histology of Wallerian degeneration: I. Nuclear population in rabbit sciatic nerve. J Anat. 1946;80:37–50. [PubMed] [Google Scholar]
- 2.Aglah C, Gordon T, Posse de Chaves EI. cAMP promotes neurite outgrowth and extension through protein kinase A but independently of Erk activation in cultured rat motoneurons. Neuropharmacology. 2008;55:8–17. doi: 10.1016/j.neuropharm.2008.04.005. [DOI] [PubMed] [Google Scholar]
- 3.Al-Majed AA, Neumann CM, Brushart TM, Gordon T. Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J Neurosci. 2000;20:2602–2608. doi: 10.1523/JNEUROSCI.20-07-02602.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Avellino AM, Hart D, Dailey AT, MacKinnon M, Ellegala D, Kliot M. Differential macrophage responses in the peripheral and central nervous system during wallerian degeneration of axons. Exp Neurol. 1995;136:183–198. doi: 10.1006/exnr.1995.1095. [DOI] [PubMed] [Google Scholar]
- 5.Bixby JL, Harris WA. Molecular mechanisms of axon growth and guidance. Annu Rev Cell Biol. 1991;7:117–159. doi: 10.1146/annurev.cb.07.110191.001001. [DOI] [PubMed] [Google Scholar]
- 6.Black MM, Lasek RJ. Slowing of the rate of axonal regeneration during growth and maturation. Exp Neurol. 1979;63:108–119. doi: 10.1016/0014-4886(79)90188-2. [DOI] [PubMed] [Google Scholar]
- 7.Braunewell KH, Pesheva P, McCarthy JB, Furcht LT, Schmitz B, Schachner M. Functional involvement of sciatic nerve-derived versican- and decorin-like molecules and other chondroitin sulphate proteoglycans in ECM-mediated cell adhesion and neurite outgrowth. Eur J Neurosci. 1995;7:805–814. doi: 10.1111/j.1460-9568.1995.tb00683.x. [DOI] [PubMed] [Google Scholar]
- 8.Bruck W. The role of macrophages in Wallerian degeneration. Brain Pathol. 1997;7:741–752. doi: 10.1111/j.1750-3639.1997.tb01060.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brushart TM, Hoffman PN, Royall RM, Murinson BB, Witzel C, Gordon T. Electrical stimulation promotes motoneuron regeneration without increasing its speed or conditioning the neuron. J Neurosci. 2002;22:6631–6638. doi: 10.1523/JNEUROSCI.22-15-06631.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brushart TM, Jari R, Verge V, Rohde C, Gordon T. Electrical stimulation restores the specificity of sensory axon regeneration. Exp Neurol. 2005;194:221–229. doi: 10.1016/j.expneurol.2005.02.007. [DOI] [PubMed] [Google Scholar]
- 11.Cai D, Shen Y, De Bellard M, Tang S, Filbin MT. Prior exposure to neurotrophins blocks inhibition of axonal regeneration by MAG and myelin via a cAMP-dependent mechanism. Neuron. 1999;22:89–101. doi: 10.1016/s0896-6273(00)80681-9. [DOI] [PubMed] [Google Scholar]
- 12.Cajal S. Degeneration and Regeneration of the Nervous System. Oxford University Press; 1928. [Google Scholar]
- 13.Ceballos D, Lago N, Verdu E, Penkowa M, Carrasco J, Navarro X, Palmiter RD, Hidalgo J. Role of metallothioneins in peripheral nerve function and regeneration. Cell Mol Life Sci. 2003;60:1209–1216. doi: 10.1007/s00018-003-3047-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.David S, Fry EJ, Lopez-Vales R. Novel roles for Nogo receptor in inflammation and disease. Trends Neurosci. 2008;31:221–226. doi: 10.1016/j.tins.2008.02.002. [DOI] [PubMed] [Google Scholar]
- 15.Dergham P, Ellezam B, Essagian C, Avedissian H, Lubell WD, McKerracher L. Rho signaling pathway targeted to promote spinal cord repair. J Neurosci. 2002;22:6570–6577. doi: 10.1523/JNEUROSCI.22-15-06570.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.English AW. Enhancing axon regeneration in peripheral nerves also increases functionally inappropriate reinnervation of targets. J Comp Neurol. 2005;490:427–441. doi: 10.1002/cne.20678. [DOI] [PubMed] [Google Scholar]
- 17.Fansa H, Keilhoff G. Factors influencing nerve regeneration. Handchir Mikrochir Plast Chir. 2003;35:72–82. doi: 10.1055/s-2003-40767. [DOI] [PubMed] [Google Scholar]
- 18.Fawcett JW, Asher RA. The glial scar and central nervous system repair. Brain Res Bull. 1999;49:377–391. doi: 10.1016/s0361-9230(99)00072-6. [DOI] [PubMed] [Google Scholar]
- 19.Fouad K, Schnell L, Bunge MB, Schwab ME, Liebscher T, Pearse DD. Combining Schwann cell bridges and olfactory-ensheathing glia grafts with chondroitinase promotes locomotor recovery after complete transection of the spinal cord. J Neurosci. 2005;25:1169–1178. doi: 10.1523/JNEUROSCI.3562-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fu SY, Gordon T. Contributing factors to poor functional recovery after delayed nerve repair: prolonged axotomy. J Neurosci. 1995;15:3876–3885. doi: 10.1523/JNEUROSCI.15-05-03876.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fu SY, Gordon T. Contributing factors to poor functional recovery after delayed nerve repair: prolonged denervation. J Neurosci. 1995;15:3886–3895. doi: 10.1523/JNEUROSCI.15-05-03886.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fu SY, Gordon T. The cellular and molecular basis of peripheral nerve regeneration. Mol Neurobiol. 1997;14:67–116. doi: 10.1007/BF02740621. [DOI] [PubMed] [Google Scholar]
- 23.Furey MJ, Midha R, Xu QG, Belkas J, Gordon T. Prolonged target deprivation reduces the capacity of injured motoneurons to regenerate. Neurosurgery. 2007;60:723–732. doi: 10.1227/01.NEU.0000255412.63184.CC. discussion 732–723. [DOI] [PubMed] [Google Scholar]
- 24.García-Alías G, Lin R, Akrimi SF, Story D, Bradbury EJ, Fawcett J. Therapeutic time window for the application of chondroitinase ABC after spinal cord injury. Exp Neurol. 2008;210:331–338. doi: 10.1016/j.expneurol.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 25.George R, Griffin JW. Delayed macrophage responses and myelin clearance during Wallerian degeneration in the central nervous system: the dorsal radiculotomy model. Exp Neurol. 1994;129:225–236. doi: 10.1006/exnr.1994.1164. [DOI] [PubMed] [Google Scholar]
- 26.Geremia NM, Gordon T, Brushart TM, Al-Majed AA, Verge VM. Electrical stimulation promotes sensory neuron regeneration and growth-associated gene expression. Exp Neurol. 2007;205:347–359. doi: 10.1016/j.expneurol.2007.01.040. [DOI] [PubMed] [Google Scholar]
- 27.Gershenbaum MR, Roisen FJ. The effects of dibutyryl cyclic adenosine monophosphate on the degeneration and regeneration of crush-lesioned rat sciatic nerves. Neuroscience. 1980;5:1565–1580. doi: 10.1016/0306-4522(80)90021-4. [DOI] [PubMed] [Google Scholar]
- 28.Gordon T, Sulaiman O, Boyd JG. Experimental strategies to promote functional recovery after peripheral nerve injuries. J Peripher Nerv Syst. 2003;8:236–250. doi: 10.1111/j.1085-9489.2003.03029.x. [DOI] [PubMed] [Google Scholar]
- 29.Grados-Munro EM, Fournier AE. Myelin-associated inhibitors of axon regeneration. J Neurosci Res. 2003;74:479–485. doi: 10.1002/jnr.10803. [DOI] [PubMed] [Google Scholar]
- 30.Graham JB, Neubauer D, Xue QS, Muir D. Chondroitinase applied to peripheral nerve repair averts retrograde axonal regeneration. Exp Neurol. 2007;203:185–195. doi: 10.1016/j.expneurol.2006.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Groves ML, McKeon R, Werner E, Nagarsheth M, Meador W, English AW. Axon regeneration in peripheral nerves is enhanced by proteoglycan degradation. Exp Neurol. 2005;195:278–292. doi: 10.1016/j.expneurol.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 32.Han PJ, Shukla S, Subramanian PS, Hoffman PN. Cyclic AMP elevates tubulin expression without increasing intrinsic axon growth capacity. Exp Neurol. 2004;189:293–302. doi: 10.1016/j.expneurol.2004.03.010. [DOI] [PubMed] [Google Scholar]
- 33.Houle JD, Tom VJ, Mayes D, Wagoner G, Phillips N, Silver J. Combining an autologous peripheral nervous system “bridge” and matrix modification by chondroitinase allows robust, functional regeneration beyond a hemisection lesion of the adult rat spinal cord. J Neurosci. 2006;26:7405–7415. doi: 10.1523/JNEUROSCI.1166-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hughes PM, Wells GM, Perry VH, Brown MC, Miller KM. Comparison of matrix metalloproteinase expression during Wallerian degeneration in the central and peripheral nervous systems. Neuroscience. 2002;113:273–287. doi: 10.1016/s0306-4522(02)00183-5. [DOI] [PubMed] [Google Scholar]
- 35.Jin SL, Richard FJ, Kuo WP, D’Ercole AJ, Conti M. Impaired growth and fertility of cAMP-specific phosphodiesterase PDE4D-deficient mice. Proc Natl Acad Sci U S A. 1999;96:11998–12003. doi: 10.1073/pnas.96.21.11998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kilmer SL, Carlsen RC. Chronic infusion of agents that increase cyclic AMP concentration enhances the regeneration of mammalian peripheral nerves in vivo. Exp Neurol. 1987;95:357–367. doi: 10.1016/0014-4886(87)90144-0. [DOI] [PubMed] [Google Scholar]
- 37.Krekoski CA, Neubauer D, Zuo J, Muir D. Axonal regeneration into acellular nerve grafts is enhanced by degradation of chondroitin sulfate proteoglycan. J Neurosci. 2001;21:6206–6213. doi: 10.1523/JNEUROSCI.21-16-06206.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lehmann M, Fournier A, Selles-Navarro I, Dergham P, Sebok A, Leclerc N, Tigyi G, McKerracher L. Inactivation of Rho signaling pathway promotes CNS axon regeneration. J Neurosci. 1999;19:7537–7547. doi: 10.1523/JNEUROSCI.19-17-07537.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Levi AD, Bunge RP, Lofgren JA, Meima L, Hefti F, Nikolics K, Sliwkowski MX. The influence of heregulins on human Schwann cell proliferation. J Neurosci. 1995;15:1329–1340. doi: 10.1523/JNEUROSCI.15-02-01329.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lozeron P, Krarup C, Schmalbruch H. Regeneration of unmyelinated and myelinated sensory nerve fibres studied by a retrograde tracer method. J Neurosci Methods. 2004;138:225–232. doi: 10.1016/j.jneumeth.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 41.McQuarrie IG, Grafstein B, Gershon MD. Axonal regeneration in the rat sciatic nerve: effect of a conditioning lesion and of dbcAMP. Brain Res. 1977;132:443–453. doi: 10.1016/0006-8993(77)90193-7. [DOI] [PubMed] [Google Scholar]
- 42.Ming G, Henley J, Tessier-Lavigne M, Song H, Poo M. Electrical activity modulates growth cone guidance by diffusible factors. Neuron. 2001;29:441–452. doi: 10.1016/s0896-6273(01)00217-3. [DOI] [PubMed] [Google Scholar]
- 43.Mueller BK. Growth cone guidance: first steps towards a deeper understanding. Annu Rev Neurosci. 1999;22:351–388. doi: 10.1146/annurev.neuro.22.1.351. [DOI] [PubMed] [Google Scholar]
- 44.Neumann S, Bradke F, Tessier-Lavigne M, Basbaum AI. Regeneration of sensory axons within the injured spinal cord induced by intraganglionic cAMP elevation. Neuron. 2002;34:885–893. doi: 10.1016/s0896-6273(02)00702-x. [DOI] [PubMed] [Google Scholar]
- 45.Neumann S, Woolf CJ. Regeneration of dorsal column fibers into and beyond the lesion site following adult spinal cord injury. Neuron. 1999;23:83–91. doi: 10.1016/s0896-6273(00)80755-2. [DOI] [PubMed] [Google Scholar]
- 46.Nikulina E, Tidwell JL, Dai HN, Bregman BS, Filbin MT. The phosphodiesterase inhibitor rolipram delivered after a spinal cord lesion promotes axonal regeneration and functional recovery. Proc Natl Acad Sci U S A. 2004;101:8786–8790. doi: 10.1073/pnas.0402595101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pichichero M, Beer B, Clody DE. Effects of dibutyryl cyclic AMP on restoration of function of damaged sciatic nerve in rats. Science. 1973;182:724–725. doi: 10.1126/science.182.4113.724. [DOI] [PubMed] [Google Scholar]
- 48.Qiu J, Cai D, Dai H, McAtee M, Hoffman PN, Bregman BS, Filbin MT. Spinal axon regeneration induced by elevation of cyclic AMP. Neuron. 2002;34:895–903. doi: 10.1016/s0896-6273(02)00730-4. [DOI] [PubMed] [Google Scholar]
- 49.Raff MC, Hornby-Smith A, Brockes JP. Cyclic AMP as a mitogenic signal for cultured rat Schwann cells. Nature. 1978;273:672–673. doi: 10.1038/273672a0. [DOI] [PubMed] [Google Scholar]
- 50.Sandvig A, Berry M, Barrett LB, Butt A, Logan A. Myelin-, reactive glia-, and scar-derived CNS axon growth inhibitors: expression, receptor signaling, and correlation with axon regeneration. Glia. 2004;46:225–251. doi: 10.1002/glia.10315. [DOI] [PubMed] [Google Scholar]
- 51.Shen YJ, DeBellard ME, Salzer JL, Roder J, Filbin MT. Myelin-associated glycoprotein in myelin and expressed by Schwann cells inhibits axonal regeneration and branching. Mol Cell Neurosci. 1998;12:79–91. doi: 10.1006/mcne.1998.0700. [DOI] [PubMed] [Google Scholar]
- 52.Stoll G, Griffin JW, Li CY, Trapp BD. Wallerian degeneration in the peripheral nervous system: participation of both Schwann cells and macrophages in myelin degradation. J Neurocytol. 1989;18:671–683. doi: 10.1007/BF01187086. [DOI] [PubMed] [Google Scholar]
- 53.Sulaiman OA, Gordon T. Effects of short- and long-term Schwann cell denervation on peripheral nerve regeneration, myelination, and size. Glia. 2000;32:234–246. doi: 10.1002/1098-1136(200012)32:3<234::aid-glia40>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 54.Swett JE, Wikholm RP, Blanks RH, Swett AL, Conley LC. Motoneurons of the rat sciatic nerve. Exp Neurol. 1986;93:227–252. doi: 10.1016/0014-4886(86)90161-5. [DOI] [PubMed] [Google Scholar]
- 55.Tang BL. Inhibitors of neuronal regeneration: mediators and signaling mechanisms. Neurochem Int. 2003;42:189–203. doi: 10.1016/s0197-0186(02)00094-3. [DOI] [PubMed] [Google Scholar]
- 56.Tropea D, Caleo M, Maffei L. Synergistic effects of brain-derived neurotrophic factor and chondroitinase ABC on retinal fiber sprouting after denervation of the superior colliculus in adult rats. J Neurosci. 2003;23:7034–7044. doi: 10.1523/JNEUROSCI.23-18-07034.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Udina E, Furey M, Busch S, Silver J, Gordon T, Fouad K. Electrical stimulation of intact peripheral sensory axons in rats promotes outgrowth of their central projections. Exp Neurol. 2008;210:238–247. doi: 10.1016/j.expneurol.2007.11.007. [DOI] [PubMed] [Google Scholar]
- 58.Witzel C, Rohde C, Brushart TM. Pathway sampling by regenerating peripheral axons. J Comp Neurol. 2005;485:183–190. doi: 10.1002/cne.20436. [DOI] [PubMed] [Google Scholar]
- 59.Zuo J, Hernandez YJ, Muir D. Chondroitin sulfate proteoglycan with neurite-inhibiting activity is up-regulated following peripheral nerve injury. J Neurobiol. 1998;34:41–54. [PubMed] [Google Scholar]
- 60.Zuo J, Neubauer D, Graham J, Krekoski CA, Ferguson TA, Muir D. Regeneration of axons after nerve transection repair is enhanced by degradation of chondroitin sulfate proteoglycan. Exp Neurol. 2002;176:221–228. doi: 10.1006/exnr.2002.7922. [DOI] [PubMed] [Google Scholar]