Abstract
Background
The goals of this prospective longitudinal study were to monitor levels of psychosocial stress in children and adolescents with Tourette syndrome (TS) and/or obsessive-compulsive disorder (OCD) compared to healthy control subjects and to examine the relationship between measures of psychosocial stress and fluctuations in tic, obsessive-compulsive (OC), and depressive symptom severity.
Methods
Consecutive ratings of tic, OC and depressive symptom severity were obtained for 45 cases and 41 matched healthy control subjects over a two-year period. Measures of psychosocial stress included youth self-report, parental report, and clinician ratings of long-term contextual threat. Structural equation modeling for unbalanced repeated measures was used to assess the temporal sequence of psychosocial stress with the severity of tic, OC and depressive symptoms.
Results
Subjects with TS and OCD experienced significantly more psychosocial stress than did the controls. Estimates of psychosocial stress were predictive of future depressive symptoms. Current levels of psychosocial stress were also a significant predictor of future OC symptom severity, but not vice versa. Current OC symptom severity was a predictor of future depressive symptom severity, but not vice versa. Current levels of psychosocial stress and depression were independent predictors of future tic severity, even after controlling for the effect of advancing chronological age.
Conclusions
The impact of antecedent psychosocial adversity is greater on future depressive symptoms than for tic and/or OC symptoms. Worsening OC symptoms are also a predictor of future depressive symptoms. Advancing chronological age is robustly associated with reductions in tic severity.
Keywords: Tourette syndrome, obsessive-compulsive disorder, depression, psychosocial stress, latent variables, longitudinal study
Tourette syndrome (TS) and early-onset obsessive-compulsive disorder (OCD) are etiologically related, chronic, familial neuropsychiatric disorders. These disorders are characterized by intrusive sensory urges, motor and vocal tics, as well as recurrent thoughts and impulses and repetitive behaviors or mental acts that wax and wane in severity (Leckman, 2002). This fluctuation complicates clinical management as well as the interpretation of efficacy and effectiveness of treatment studies. Many studies have emphasized the importance of psychosocial stress in mediating the progression of a number of disease states including headache (Fernandez & Sheffield, 1996), multiple sclerosis (Mohr et al., 2000), human immunodeficiency virus (Evans et al., 1997; Moss, Bose, Wolters, & Brouwers, 1998; Murphy, Moscicki, Vermund, & Muenz, 2000), diabetes (Goldston, Kovacs, Obrosky, & Iyengar, 1995; Thernlund et al., 1995) and major depression (Brown & Harris, 1978; Kendler, Karkowski, & Prescott, 1999; Kendler, Gardner, & Prescott, 2002; Goodyer, Kolvin, & Gatzanis, 1985). Earlier cross-sectional and longitudinal studies of TS and early-onset OCD have consistently suggested that these disorders are sensitive to psychosocial stress (Bornstein, 1990; Chappell et al., 1994; Findley et al., 2003; Hoekstra, Steenhuis, Kallenberg, & Minderaa, 2004; Silva, Munoz, Barickman, & Friedhoff, 1995; Surwillo, Shafii, & Barrett, 1978; Thomsen, 1995).
The goals of this prospective longitudinal study were to monitor levels of psychosocial stress in children and adolescents with TS and/or OCD compared to healthy control subjects and to examine the relationship between measures of psychosocial stress and fluctuations in tic, obsessive-compulsive (OC), and depressive symptom severity. A structural equation modeling framework with latent-time varying constructs was used to maximize the available data obtained at different time points.
Method
Subjects
Subjects in this study were 86 children and adolescents, aged 7 to 17 years, who had been diagnosed with TS alone (N = 17), OCD alone (N = 8), both TS and OCD (N = 20), as well as 41 healthy control subjects (Table 1). As previously described, all patients were being followed at the Yale Child Study Center Tic Disorder/Obsessive-Compulsive Disorder Specialty Clinic (Findley et al., 2003; Lin et al., 2002; Luo et al., 2004). Expert clinicians using DSM-IV criteria made all psychiatric diagnoses based on all available information (see below). Exclusion criteria for both groups included an IQ < 75; serious medical illness; major sensory handicaps (blindness, deafness); major neurological disease (including a seizure disorder); head trauma resulting in loss of consciousness; current (past 6 months) psychiatric disorder that could interfere with participation, such as major depression; psychosis; autism or another pervasive developmental disorder. All parents provided written informed consent after the study was described to them in detail. A separate assent form was used to ensure the informed participation of the child and adolescent subjects.
Table 1.
Demographic and clinical characteristics at baseline
| Variable | n | Tic/OC patients (N = 45) | n | Unaffected controls (N = 41) |
|---|---|---|---|---|
| Age in years (SD), range | 45 | 12.0 (2.7), 7.5–16.9 | 41 | 12.7 (2.7), 7.5–17 |
| Sex (% female)* | 11 | 24% | 20 | 49% |
| Ethnicity (%Caucasian) | 42 | 93% | 34 | 83% |
| Parent education in years (SD) | 44 | 15.2 (2.0) | 41 | 15.6 (2.1) |
| Age of onset (SD), range | ||||
| TS (including chronic tics) | 37a | 5.9 (2.3), 2–12 | NA | NA |
| OCD | 28b | 7.4 (3.0), 3–13 | ||
| Baseline symptom severity by diagnosis | ||||
| TS (including chronic tics)1 | 37a | 15.4 (8.1), 0–34 | NA | NA |
| OCD2 | 28b | 12.8 (7.9), 0–29 | ||
| Stress characteristics at baseline | ||||
| Perceived Stress Scale Parent (PSSP)** | 44 | 6.5 (3.1), 2–13 | 38 | 2.8 (2.0), 0–8 |
| Daily Life Stressors Scale (DLSS)** | 45 | 15.3 (12.6), 0–58 | 40 | 4.7 (5.5), 0–24 |
| Yale Children’s Global Stress Index (YCGSI) | 43 | 1.8 (1.1), 0–4 | 38 | 1.7 (1.1). 0–3.75 |
| ADHD at baseline (%) | 26 | 58% | NA | NA |
| Anxietyc (SD), range** | 43 | 10.2 (6.5), 0–25 | 40 | 3.8 (4.2), 0–19 |
| Depressiond (SD), range** | 45 | 26.4 (7.9), 17–48 | 41 | 18.1 (1.5), 17–24 |
TS = Tourette syndrome, OCD = Obsessive-compulsive disorder, NA = Not applicable.
Total tic severity score on the YGTSS (Yale Global Tic Severity Scale), range 0–50.
Total score on the CY-BOCS (Children’s Yale-Brown Obsessive-Compulsive Scale), range 0–40.
Combined scores on the YGTSS and CY-BOCS, range 0–90.
Number of subjects out of 45 patients who had TS (including chronic tics).
Number of subjects out of 45 patients who had OCD.
Raw score on the Revised Children’s Manifest Anxiety Scale (RCMAS), range 0–28. T scores were used for data analysis purposes.
Total score on the Children’s Depression Rating Scale – Revised (CDRS-R), range 17–113
Significant differences between Tic/OCD patients and unaffected controls:
p < .05;
p < .0001.
Baseline assessment
When a family entered the study, information concerning the patient was collected in a two-stage process (Findley et al., 2003; Lin et al., 2002; Luo et al., 2004). The first stage consisted of the collection of information concerning symptoms associated with TS and OCD using a self- and family-report (Tourette Syndrome Association International Consortium for Genetics, 1999) based on the tic inventory and ordinal severity scales of the Yale Global Tic Severity Scale (YGTSS) (Leckman et al., 1989), and the symptom checklist and ordinal scales of the Children’s Yale-Brown Obsessive Compulsive Scale (CY-BOCS) (Scahill et al., 1997). In a second stage of assessment, an experienced clinician reviewed these symptom ratings with the child and the parent to ensure their accuracy and validity. Comorbid psychiatric diagnoses, including attention deficit hyperactivity disorder (ADHD), were made using all available information, including data collected using the Schedule for Affective Disorders and Schizophrenia for School-Age Children (Kaufman et al., 1997). In addition, depression was assessed with the Children’s Depression Rating Scale-Revised (CDRS-R) (Poznanski, Freeman, & Mokros, 1985), and anxiety was measured with the Revised Children’s Manifest Anxiety Scale (RCMAS) (Paget & Reynolds, 1984).
Measurements of psychosocial stress at baseline were made using parent report (Parent Perceived Stress Scale, PSS-P) (Cohen, Kamarck, & Mermelstein, 1983; youth self-report (Daily Life Stressors Scale, DLSS) (Kearney, Drabman, & Beasley, 1993), and a clinician-rated measure of long-term contextual threat (Yale Children’s Global Stress Index, YCGSI) (Findley et al., 2003). Normal controls were also assessed for their stress level for comparison with the subjects who had been diagnosed with TS and/or OCD.
Longitudinal ratings
Monthly ratings included an assessment of tic severity by expert raters using the tic portion of the YGTSS. Other monthly ratings included OC symptom severity using the CY-BOCS and the CDRS-R both completed by expert raters. The PSS-P (parental report) was also administered on a monthly basis. The DLSS (self-report) and the clinician-rated YCGSI and CDRS-R were collected every four months. Most subjects completed the monthly visitation schedule during the two-year period, although the number of visits varied among the subjects (min. = 4, max. = 26, mean = 21, median = 24, Scahill et al., 1997).
Medication status
This was a naturalistic prospective longitudinal study of two years’ duration; consequently each subject’s medication history was monitored each month according to the class of psychotropic agents (adrenergic agonists [clonidine, guanfacine], classical neuroleptics [haloperidol, pimozide, fluphenazpine], atypical neuroleptics [risperidone, ziprasidone, olanzapine, aripiprazole], serotonin reuptake inhibitors and other antidepressants [citalopram, clomipramine, fluoxetine, fluvoxamine, paroxetine, sertaline, venlafaxine], mood stabilizers [lithium, divalproex sodium, topiramate], psychostimulants [methylphenidate, dextroamphetamine], benzodiazpines [clonazepam], other miscellaneous agents). Baseline medication status and changes in medication status were included in the structural equation modeling.
Pathway analyses of the relationship between longitudinal measures of psychosocial stress and tic, OC and depressive symptom severity
The longitudinal design makes it feasible to investigate possible temporal relationships between stress, severity of tic and OC symptoms, and depression. A structural equation modeling framework was employed to explore possible relationships. All the coefficients in a structural equation model are simultaneously estimated via maximum likelihood method with STATA add-on module GLLAMM (Rabe-Hesketh, Skrondal, & Pickles, 2004) since it accommodates different measurement schedules among multiple longitudinal measures. A latent time-varying stress construct (SC) was modeled to represent the underlying true stress level manifested by the three available longitudinal stress measures: the YCGSI, the PSS-P and the DLSS. Time-lag effect was studied with mixed effect submodels in a structural equation framework by using the nearest recorded past reading of a longitudinal measure as a covariate for a current reading. The effects of previous stress level on current symptom severity and vice versa; the effects of previous stress level on current depression; and the effects of previous depression level on current symptom severity and vice versa were all explored.
Statistical models
Relationships between psychosocial stress and tic, OC and depressive symptom severity were studied via the submodels within the above structural equation modeling framework. Specifically, we developed a latent measure (Stress Construct, SC) of psychosocial stress that was related to each of the three underlying stress measures as specified in the following submodel (Bollen, 1989):
| (1) |
where Sijk is the value of type k stress measure recorded at time point j for subject i, γ0k is the corresponding intercept for the type k stress measure that accommodates scale difference in multiple stress measures, γ1k is the factor loading that represents relative contribution of the type k stress measure to the time-varying stress construct ηij at time point j for subject i, and ζijk is independently, normally distributed residual with mean zero and variance For the sake of model identifiability, the first factor loading γ11 is set to one.
The correlations among different stress measures and among different time points of a same measure in a same subject were accommodated by the stress construct ηij that could be affected by other covariates in the following submodel (Bollen, 1989):
| (2) |
where α is a vector of regression coefficients for the corresponding effects of predictors X in subject i, for example, X could include time-lagged measure of depression, and εij is the residual that is assumed to follow a multivariate normal distribution with mean zero and covariance of compound symmetry to accommodate within-subject correlations across different time points j.
Finally, the dependence of the longitudinal measures of symptom severity on the SC is described in the following submodel (Bollen, 1989):
| (3) |
where Yijl is the value of type l symptom severity measure (for example, l = 1 for YGTSS, l = 2 for CY-BOCS, l = 3 for CDRS-R) recorded at time point j for subject i; β0l is the corresponding fixed intercept; b0il is the corresponding random intercept for subject i that accommodates within subject correlation and is assumed to be normally distributed with mean zero and variance ; β1l is the regression coefficient for the effect of the time-lagged stress construct ηi,j−1 at time point j–1 on type l severity measure for subject i; is a vector of regression coefficients for the corresponding effects of predictors Zil, for example, it can include a time-lagged depression measure for Yijl of symptom severity or vice versa, and eijl is the independently, normally distributed residual with mean zero and variance The γ’s, α’s and β’s are simultaneously estimated by the GLLAMM module in STATA (Rabe-Hesketh et al., 2004). The effects represented by γ1k,α and β1l are represented by arrow lines. An effect is deemed to be statistically significant if the p-value for Wald test for each of the γ’s, α’s and β’s is smaller than .05.
We also incorporated baseline medication status, a change in medication, and a diagnosis of ADHD as class variables into the longitudinal path models.
Results
Description of study cohort
Distributions of the demographic and clinical variables including age, gender, race, age of symptom onset, and symptom severity scores, stress, anxiety, and depression are presented in Table 1. The sample is a relatively typical group of clinically referred pediatric patients with TS and early onset OCD and age matched healthy control subjects.
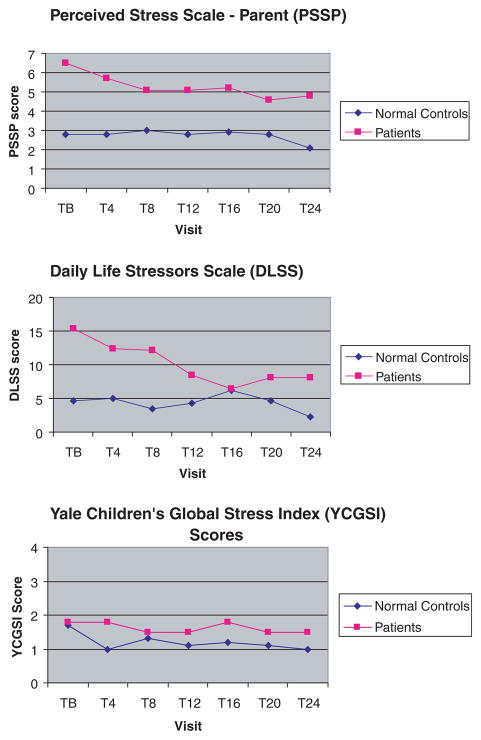
Overall stress levels, regardless of the informant, were higher among the TS and/or OCD patients compared to the normal controls (Figure 1). For example, on the blindly rated YCGSI, 47% (21 of the 45) of the patients were judged to be under either a high-moderate or severe level of contextual threat stress on 38 occasions compared to just 20% (8 of 41) of the healthy controls on 17 occasions. Each of the independently determined measures of psychosocial stress was correlated with one another. This was true in both the patient and healthy controls, when the data was analyzed separately by group (data not shown). The parental ratings of perceived stress (PSS-P) were correlated in the range of .65–.67 (p < .0001) with each of the other two measures. However, the clinician ratings of long-term contextual threat were only correlated at the level of .43 (p < .002) with the subject’s self-report of daily life stress (DLSS).
Figure 1.
Group-specific stress measures. Stress scores at each time point (Baseline = TB, visit 4, 8, 12, 16, 20, 24 months after baseline = T4, T8, T12, T16, T20, T24, respectively) over the two-year period were averaged separately across patients and controls. Stress scores are plotted for Perceived Stress Scale – Parent (PSS-P), Daily Life Stressors Scale Parent (DLSS), and Yale Children’s Global Stress Index (YCGSI). Stress levels for all the three stress ratings PSS-P, DLSS, and YCGSI over the two-year period are significantly higher among the patients with TS and/or OCD than those among the normal controls (Z = 6.97, p < .0001 for PSS-P; Z = 3.95, p < .0001 for DLSS; and Z = 2.84, p = .0045 for YCGSI)
Overall tic symptom severity and obsessive-compulsive symptom severity improved over the 2-year period (Figure 2). Accounting for the longitudinal nature of the tic and OC symptom severity measures, the correlation between YGTSS and CY-BOCS was .34 (p = .02). The correlation coefficients between symptom severities of tic (YGTSS) and OC (CY-BOCS) and depression (CDRS-R) were .25 (p < .001) and .40 (p < .001), respectively. The correlation coefficients between YGTSS and each of the stress measure including PSS-P, DLSS, and clinician ratings in patients were .20 (p < .0001), .13 (p = .023), and .12 (p = .037), respectively. The correlation coefficients between CY-BOCS and each of the stress measures including PSS-P, DLSS, and clinician ratings in patients were .21 (p < .0001), .18 (p = .003), and .082 (p = .051), respectively. The correlation coefficients between depression (CDRS-R) and each of the stress measures including PSS-P, DLSS, and clinician ratings in patients were .60 (p < .0001), .37 (p < .0001), and .27 (p < .0001), respectively.
Figure 2.
Trajectories of YGTSS and CY-BOCS. Yale Global Tic Severity Scale (YGTSS) and Children’s Yale-Brown Obsessive Compulsive Scale (CY-BOCS) symptom severity scores are plotted among the patients over the two-year period (TB to T24)
A minority of subjects were rated has having clinically significant levels of depression (CDRS-R ≥40). Specifically, 27% (12 of the 45) of the patients were judged to have a clinically significant level of depression on 20 occasions compared to just 2% (1 of 41) of the healthy controls on a single occasion.
Thirty-nine out of the 45 subjects were taking psychotropic medications at baseline. The treatment of 18 subjects remained unchanged over the entire course of the study (stable medication regimen = 12; and medication free for the duration of the study = 6). At baseline, 13 (29%) subjects were taking neuroleptics, 21 (47%) subjects were on selective serotonin reuptake inhibitors (SSRIs), and 18 (40%) subjects were on alpha agonists. Subjects taking SSRIs had significantly higher baseline ratings of both OC and depressive symptom severity (CY-BOCS (F [1,41] = 12.03, p = .001), CDRS-R (F [1,42] = 10.44, p = .002). Subjects on alpha agonists had significantly higher baseline ratings of both tic and depressive symptom severity (YGTSS (F [1,41] = 5.41, p = .025), CDRS (F [1,42] = 8.14, p = .007). At baseline, no significant differences in tic, OCD, stress or depression scores were observed among the subjects on neuroleptics vs. the remaining subjects.
Exploring relationship between age, stress, depression, tic and OC symptom severity
Increasing chronological age is associated with tic improvement (Figure 3A), e.g., the standardized coefficient of .38 means that following an increase of 2.7 years of age (1 SD of all measures of age in the patient population), we observed a reduction of 38% of 1.0 SD of all the YGTSS scores (.38 × 9.0 = 3.4). Even after controlling for this age effect, current levels of psychosocial stress and depression were still independent and significant (although modest) predictors of future tic symptom severity (Figure 3A). In contrast, current tic severity was not a statistically significant predictor of psychosocial stress or of depressive symptoms (data not shown).
Figure 3.
Pathway analyses of the relationships between longitudinally collected measures of psychosocial stress and tic, obsessive-compulsive (OC) and depressive symptom severity. Squares depict measured variables and circles represent latent variables. The standardized coefficients represent the size of the effects and the p-values in parentheses represent the significance of these effects as a result of the Wald test. An effect is deemed to be statistically significant if the p-value for the Wald test for each of the γ’s,α’s and β’s is smaller than .05 (see text). The standardized coefficients are the proportion of 1.0 standard deviation (SD) of the predicted variable that can be explained by 1.0 SD change in the predicting variable. A solid line indicates a statistically significant positive relationship; a dashed line indicates a significant inverse relationship. The thickness of the lines reflects the relative significance of the interactions. An arrow indicates a predictive relationship, while a line without an arrow head indicates the relative contributions to a latent variable. (A) Tic symptoms. In this model, future tic improvement was associated with advancing chronological age. The standardized coefficient of .38 means that following an increase of 2.7 years of age (1 SD of all measures of age in the patient population), we observed a reduction of 38% of 1.0 SD of all the YGTSS scores (.38 × 9.0 = 3.4). In addition, increases in past depressive symptoms (Children’s Depression Rating Scale-Revised, CDRS-RT−1) predict higher levels of current psychosocial stress (Stress Construct, SC), which in turn modestly, but significantly, predict increases in future tic severity (Yale Global Tic Severity Scale, YGTSST+1). Increases in current depressive symptoms (Children’s Depression Rating Scale-Revised, CDRS-RT) are also modestly, and independently, predictive of increases in future tic symptom severity (YGTSST+1). Prior tic symptom severity (YGTSST−1) does not affect current depressive symptom ratings; and past YGTSS scores also do not significantly affect current stress levels (data not shown). The SC is a latent variable derived from the Yale Children’s Global Stress Index (YCGSI), the Perceived Stress Scale–Parent (PSS-P) and the Daily Life Stress Scale (DLSS). (B) Obsessive-compulsive (OC) symptoms. Higher levels of psychosocial stress in the past (SCT−1), predict higher levels of current obsessive-compulsive symptom severity (Children’s Yale-Brown Obsessive Compulsive Scale [CY-BOCST]), which subsequently predicts future depressive symptom severity (CDRS-RT+1). The standardized coefficient of .47 means that following an increase of 8.2 units on the CY-BOCS (1.0 SD of all measures of OC symptom severity in the patient population), we estimated an increase of 47% of 1.0 SD of all the CDRS-R scores (.47 × 8.7 = 4.1 units). (C) Depressive symptoms. The previous (time-lagged) depressive symptom severity (CDRS-RT−1) strongly predicts current levels of psychosocial stress (SC), which subsequently predicts future levels of depressive symptoms (CDRS-RT+1). The standardized coefficient of .73 means that following an increase of 3.3 units on the PSS-P (1.0 SD of all measures on the PSS–P in the patient population), we estimated an increase of 73% of 1.0 SD of all the CDRS-R scores (.73 × 8.7 = 6.4 units)
Current stress levels were a robust predictor of future OC symptom severity (Figure 3B). We also observed that current OC symptom severity was a potent predictor of future depressive symptoms (Figure 3B), e.g., the standardized coefficient of .47 means that following an increase of 8.2 units on the CY-BOCS (1.0 SD of all measures of OC symptom severity in the patient population), we estimated an increase of 47% of 1.0 SD of all the CDRS-R scores (.47 ×8.7 = 4.1 units). However, current depressive severity was not a statistically significant predictor of future OC symptoms (data not shown). When the antecedent tic and OC symptom severity ratings were included in the model the effects of psychosocial stress on tic and OC outcomes remained significant, but with reduced effect sizes (data not shown).
Current estimates of psychosocial stress were strongly predictive of future depressive symptoms while current ratings of depressive symptoms were only modestly predictive of future ratings of psychosocial stress (Figure 3C), e.g., the standardized coefficient of .73 means that following an increase of 3.3 units on the PSS-P (1.0 SD on of all measures on the PSS-P in the patient population), we estimated an increase of 73% of 1.0 SD of all the CDRS-R scores (.73 ×8.7 = 6.4 units).
In each of these models the parental report of current stress (PSS-P) was the most robust predictor of stress-related outcomes followed by youth self-report and clinician-rated long-term object threat ratings, in that order.
The severity of anxiety (as measured using the RCMAS) at entry was not statistically related to the severity of tics or OC symptoms recorded over the course of the study (data not shown). Likewise, a diagnosis of ADHD at the entry of the study was not statistically related to the severity of tics or OC symptoms recorded over the course of the study (data not shown). When data concerning changes in a subject’s medication status were included as a predictor in Z in submodel (3), there was no influence on the relative significance of the directional findings depicted in Figure 3A, 3B, or 3C (data not shown).
Discussion
As we have reported before, the patient group overall experienced higher levels of stress using all the stress measures than did healthy controls (Bornstein, 1990; Chappell et al., 1994; Findley et al., 2003; Hoekstra et al., 2004; Silva et al., 1995; Surwillo et al., 1978; Thomsen, 1995).
The results from our structural equation modeling framework with latent-time varying constructs support the conclusion that antecedent psychosocial stress, particularly as perceived by the parent, is a significant predictor of future depressive and OC symptom severity and is a less robust indicator of future tic severity. Our data also indicates that depressive symptoms do predict modest increases in tic severity consistent with earlier predictions (Robertson, Williamson, & Eapen, 2006). Other findings of note include that chronological age has a strong inverse relationship to tic severity.
Still, understanding the interrelationships between tics, OCD, depression, and ADHD is being complicated by the use of cross-sectional designs in most studies of clinically referred subjects. Although the etiological interrelationships between depression and ADHD with TS and OCD have not been fully elucidated, the co-occurrence of these disorders in children and adults with TS or OCD is commonplace and associated with distinctive cognitive profiles, worse social adjustment, increased psychosocial dysfunction, and diminished quality of life (Leckman et al., 1998; Peterson, Pine, Cohen, & Brook, 2001; Bloch et al., 2006; Carter et al., 2000; Dykens et al., 1990; Elstner, Selai, Trimble, & Robertson, 2001; Robertson, Banerjee, Eapen, & Fox-Hiley, 2002; Robertson et al., 2006).
Limitations
These results are from a demographically homogeneous sample of clinically referred TS and OCD patients and might not extrapolate to other populations. On the other hand, our findings are largely consistent with earlier population-based longitudinal studies suggesting the patterns of interactive comorbidity are true over the course of months as well as over the course of years (Peterson et al., 2001).
Given the naturalistic design of this prospective study, we did not formally assess the role of medication on the course of tic, OC and depressive symptomatology. At baseline, however, it appears that medication use may be more a reflection of the treating clinician’s perception of which symptoms were the most problematic nature of the presenting symptoms rather than a reflection of the effectiveness of these agents.
Clinical implications
Ratings of psychosocial stress, particularly parent-ratings of perceived stress, appear to be the best predictor of subsequent depressive and OC symptom severity and a modest predictor of tic severity. This suggests that parental perceptions of child stress are a worthwhile variable to monitor. It is surely a cost-effective variable given the considerable resources needed to determine the long-term contextual threat associated with stressful life events compared with simply asking a parent the four questions on the PSS-P. The magnitude of these effects can be estimated by the standardized coefficients such that an increase of 1.0 standard deviation (SD) on the PSS-P is estimated to increase the future ratings of depressive symptoms by 6.4 units on the CDRS-R (four months later, Figure 3). Similarly, an increase of 1.0 SD on the CY-BOCS is estimated to increase the future ratings of depressive symptoms by 4.1 units on the CDRS-R (four months later). To place these changes in context, depressed adolescents treated with a combination of cognitive behavioral therapy and fluoxetine on average improved by 27.0 units on the CDRS-R over the course of a three-month clinical trial (March et al., 2004). Alternatively, an increase of 1.0 standard deviation (SD) on the PSS-P is estimated to increase the future ratings of OC symptoms by 6.4 units on the CY-BOCS (one month later).
We found a clear relationship between future depression ratings and antecedent ratings of psychosocial stress. In contrast to earlier adult studies (where long-term contextual threat ratings have consistently been found to be the most powerful predictor of depression) (Brown & Harris, 1978; Kendler et al, 1999; Kendler et al., 2002; Kendler, Kuhn, & Prescott, 2004), we found that the level of psychosocial stress as perceived by the patient (usually the mother) was the most predictive. Although this conclusion is constrained by the number of cases who were seriously depressed during the course of the study, it is important for clinicians working with children and families to be sensitive to the importance of parental perception of psychosocial stress and the future course of tic and OC symptoms as well as depression.
Clinically it is also important to note that these findings replicate and extend earlier studies with regard to the natural history of tic disorders where age 9–12 is an inflection point beyond which tic severity gradually declines in a majority of cases (Leckman et al., 1998; Bloch et al., 2006; Coffey et al., 2004). To further contextualize these findings, we note that in a group of 33 children and adolescents with TS and ADHD randomized to treatment with a combination of clonidine and methylphenidate, the total tic score on the YGTSS on average improved by 12.0 units over the course of a four-month clinical trial (The Tourette’s Syndrome Study Group, 2002). Overall, these findings provide an optimistic long-term outlook for parents and children beset with severe and worsening tics in preadolescence.
However, the presence of comorbid ADHD is another important consideration if clinicians are to provide the best possible care for these complex cases given its potential adverse impact on the developmental course of children and adolescents (Peterson et al., 2001; Bloch et al., 2006; Carter et al., 2000; Dykens et al., 1990; Stokes, Bawden, Camfield, Backman, & Dooley, 1991; Sukhodolsky et al., 2003, 2005). This is especially true when multiple comorbidities occur in the same child. The presence of multiple coexisting conditions is likely to magnify the level of stress the family is experiencing and create negative multidirectional interactions within the family and peer group as well as further jeopardize academic performance.
Future directions
The pathogenesis of TS and early-onset OCD remains in doubt. In addition to chronological age and psychosocial stress, recent attention has focused on genetic factors (Abelson et al., 2005), the possible role of post-infectious autoimmune mechanisms (Leckman et al., 2005), and the development of basal ganglia structures (Kalanithi et al., 2005; Bloch, Leckman, Zhu, & Peterson, 2005). Future prospective longitudinal studies are needed to deepen our understanding of the role of psychosocial stress and its interaction with genetic vulnerability factors and immune function to influence the course of brain structure and function to produce the broad range of outcomes typical of these of these complex disorders.
Acknowledgments
This research was funded by NIH Grants MH066187, MH493515, MH61940, MH014235, DA017713, DA076750, RR00044, and RR00125. Additional support was provided by the Echlin Foundation and the Kaiser Family. The authors also wish to thank Virginia Eicher, Susan Quatrano, Nancy Thompson, and Barbara Peterson-Cremer for their invaluable assistance in completing this study.
Abbreviations
- TS
Tourette syndrome
- OCD
obsessive-compulsive disorder
Footnotes
Conflict of interest statement: No conflicts declared.
References
- Abelson JF, Kwan KY, O’Roak BJ, Baek DY, Stillman AA, Morgan TM, Mathews CA, Pauls DL, Rašin MR, Gunel M, Davis NR, Ercan-Sencicek AG, Guez DH, Spertus JA, Leckman JF, Dure LS, Kurlan R, Singer HS, Gilbert DL, Farhi A, Louvi A, Lifton RP, Šestan N, State MW. Mutations in SLITRK1 are associated with Tourette syndrome. Science. 2005;310:317–320. doi: 10.1126/science.1116502. [DOI] [PubMed] [Google Scholar]
- Bloch MH, Leckman JF, Zhu HT, Peterson BS. Basal ganglia volumes and future tic severity in children with Tourette syndrome. Neurology. 2005;65:1253–1258. doi: 10.1212/01.wnl.0000180957.98702.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloch MH, Peterson BS, Scahill L, Otka J, Katsovich L, Zhang H, Leckman JF. Clinical predictors of future tic and OCD severity in children with Tourette syndrome. Archives of Pediatrics and Adolescent Medicine. 2006;160:63–69. doi: 10.1001/archpedi.160.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bollen KA. Structural equations with latent variables. New York: John Wiley & Sons; 1989. [Google Scholar]
- Bornstein RA. Neuropsychological performance in children with Tourette’s syndrome. Psychiatry Research. 1990;33:73–81. doi: 10.1016/0165-1781(90)90150-4. [DOI] [PubMed] [Google Scholar]
- Brown GW, Harris TO. Social origins of depression: A study of psychiatric disorder in women. London: Tavistock; 1978. [Google Scholar]
- Carter AS, O’Donnell DA, Schultz RT, Scahill L, Leckman JF, Pauls DL. Social and emotional adjustment in children affected with Gilles de la Tourette syndrome: Associations with ADHD and family functioning. Journal of Child Psychology and Psychiatry. 2000;41:215–223. [PubMed] [Google Scholar]
- Chappell P, Riddle M, Anderson G, Scahill L, Hardin M, Walker D, Cohen D, Leckman JF. Enhanced stress responsivity of Tourette syndrome patients undergoing lumbar puncture. Biological Psychiatry. 1994;36:35–43. doi: 10.1016/0006-3223(94)90060-4. [DOI] [PubMed] [Google Scholar]
- Coffey BJ, Biederman J, Geller D, Frazier J, Spencer T, Doyle R, Gianini L, Small A, Frisone DF, Magovcevic M, Stein N, Faraone SV. reexamining tic persistence and tic-associated impairment in Tourette’s disorder: Findings from a naturalistic follow-up study. Journal of Nervous and Mental Disease. 2004;192:776–780. doi: 10.1097/01.nmd.0000144696.14555.c4. [DOI] [PubMed] [Google Scholar]
- Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. Journal of Health and Social Behaviour. 1983;24:385–396. [PubMed] [Google Scholar]
- Dykens E, Leckman DL, Riddle MA, Hardin MT, Schwartz S, Cohen D. Intellectual, academic and adaptive functioning of Tourette syndrome children with and without attention deficit disorder. Journal of Abnormal Child Psychology. 1990;18:607–615. doi: 10.1007/BF01342750. [DOI] [PubMed] [Google Scholar]
- Elstner K, Selai CE, Trimble MR, Robertson MM. Quality of Life (QOL) of patients with Gilles de la Tourette’s syndrome. Acta Psychiatrica Scandinavica. 2001;103:52–59. doi: 10.1034/j.1600-0447.2001.00147.x. [DOI] [PubMed] [Google Scholar]
- Evans DL, Leserman J, Perkins DO, Stern RA, Murphy C, Zheng BY, Gettes D, Longmate JA, Silva SG, van der Horst CM, Hall CD, Folds JD, Golden RN, Petitto JM. Severe life stress as a predictor of early disease progression in HIV infection. American Journal of Psychiatry. 1997;154:630–634. doi: 10.1176/ajp.154.5.630. [DOI] [PubMed] [Google Scholar]
- Fernandez E, Sheffield J. Relative contributions of life events versus daily hassles to the frequency and intensity of headaches. Headache. 1996;36:595–602. doi: 10.1046/j.1526-4610.1996.3610595.x. [DOI] [PubMed] [Google Scholar]
- Findley DB, Leckman JF, Katsovich L, Lin H, Zhang H, Grantz H, Otka J, Lombroso PJ, King RA. Development of the Yale Children’s Global Stress Index (YCGSI) and its application in children and adolescents with Tourette syndrome and obsessive-compulsive disorder. American Journal of Child and Adolescent Psychiatry. 2003;4:450–457. doi: 10.1097/01.CHI.0000046816.95464.EF. [DOI] [PubMed] [Google Scholar]
- Goldston DB, Kovacs M, Obrosky DS, Lyengar S. A longitudinal-study of life events and metabolic control among youths with insulin-dependent diabetes-mellitus. Health Psychology. 1995;14:409–414. doi: 10.1037//0278-6133.14.5.409. [DOI] [PubMed] [Google Scholar]
- Goodyer IM, Kolvin I, Gatzanis S. Recent undesirable life events and psychiatric disorder in childhood and adolescence. British Journal of Psychiatry. 1985;147:517–523. doi: 10.1192/bjp.147.5.517. [DOI] [PubMed] [Google Scholar]
- Hoekstra PJ, Steenhuis MP, Kallenberg CGM, Minderaa RB. Association of small life events with self reports of tic severity in pediatric and adult tic disorder patients: A prospective longitudinal study. Journal of Clinical Psychiatry. 2004;65:426–431. doi: 10.4088/jcp.v65n0320. [DOI] [PubMed] [Google Scholar]
- Kalanithi PAS, Zheng W, DiFiglia M, Grantz H, Saper CB, Schwartz ML, Leckman JF, Vaccarino FM. Altered parvalbumin-positive neuron distribution in basal ganglia of individuals with Tourette syndrome. Proceedings of National Academy of Science USA. 2005;102:13307–13312. doi: 10.1073/pnas.0502624102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P, Williamson D, Ryan N. Schedule for Affective Disorders and schizophrenia for school-age children present and lifetime version: Initial reliability and validity data. American Journal of Child and Adolescent Psychiatry. 1997;36:980–988. doi: 10.1097/00004583-199707000-00021. [DOI] [PubMed] [Google Scholar]
- Kearney CA, Drabman RS, Beasley JF. The trials of childhood: The development, reliability, and validity of the daily life stressors scale. Journal of Child and Family Study. 1993;2:371–388. [Google Scholar]
- Kendler KS, Gardner CO, Prescott CA. Toward a comprehensive developmental model for major depression in women. American Journal of Psychiatry. 2002;159:1133–1145. doi: 10.1176/appi.ajp.159.7.1133. [DOI] [PubMed] [Google Scholar]
- Kendler KS, Karkowski LM, Prescott CA. Causal relationship between stressful life events and the onset of major depression. American Journal of Psychiatry. 1999;156:837–841. doi: 10.1176/ajp.156.6.837. [DOI] [PubMed] [Google Scholar]
- Kendler KS, Kuhn J, Prescott CA. The interrelationship of neuroticism, sex, and stressful life events in the prediction of episodes of major depression. American Journal of Psychiatry. 2004;161:631–6. doi: 10.1176/appi.ajp.161.4.631. [DOI] [PubMed] [Google Scholar]
- Leckman JF. Tourette syndrome. Lancet. 2002;360:1577–1586. doi: 10.1016/S0140-6736(02)11526-1. [DOI] [PubMed] [Google Scholar]
- Leckman JF, Katsovich L, Kawikova I, Lin H, Zhang H, Krönig H, Morshed S, Parveen S, Grantz H, Findley DB, Lombroso PJ, King RA. Increased serum levels of Tumor Necrosis Factor-alpha and IL-12 in Tourette syndrome. Biological Psychiatry. 2005;57:667–673. doi: 10.1016/j.biopsych.2004.12.004. [DOI] [PubMed] [Google Scholar]
- Leckman JF, Riddle MA, Hardin MT, Ort SI, Swartz KL, Stevenson J, Cohen DJ. The Yale Global Tic Severity Scale: Initial testing of a clinician-rated scale of tic severity. Journal of American Academy of Child and Adolescent Psychiatry. 1989;28:566–573. doi: 10.1097/00004583-198907000-00015. [DOI] [PubMed] [Google Scholar]
- Leckman JF, Zhang H, Vitale A, Lahnin F, Lynch K, Bondi C, Kim YS, Peterson BS. Course of tic severity in Tourette syndrome: the first two decades. Pediatrics. 1998;102:14–19. doi: 10.1542/peds.102.1.14. [DOI] [PubMed] [Google Scholar]
- Lin HQ, Yeh CB, Peterson BS, Scahill L, Grantz H, Findley D, Katsovich L, Otka J, Lombroso PJ, King RA, Leckman JF. Assessment of symptom exacerbations in a longitudinal study of children with Tourette syndrome or obsessive-compulsive disorder. American Journal of Child and Adolescent Psychiatry. 2002;41:1070–1077. doi: 10.1097/00004583-200209000-00007. [DOI] [PubMed] [Google Scholar]
- Luo F, Leckman JF, Katsovich L, Findley D, Grantz H, Tucker DM, Lombroso PJ, King RA, Bessen DE. Prospective longitudinal study of children with tic disorders and/or obsessive-compulsive disorder: Relationship of symptom exacerbations to newly acquired streptococcal infections. Pediatrics. 2004;113:e578–e585. doi: 10.1542/peds.113.6.e578. [DOI] [PubMed] [Google Scholar]
- March J, Silva S, Petrycki S, Curry J, Wells K, Fairbank J, Burns B, Domino M, McNulty S, Vitiello B, Severe J. Treatment for Adolescents with Depression Study (TADS) Team. Fluoxetine, cognitive-behavioral therapy, and their combination for adolescents with depression: Treatment for Adolescents with Depression Study (TADS) randomized controlled trial. Journal of American Medical Association. 2004;292:807–20. doi: 10.1001/jama.292.7.807. [DOI] [PubMed] [Google Scholar]
- Mohr DC, Goodkin DE, Bacchetti P, Boudewyn AC, Huang L, Marrietta P, Cheuk W, Dee B. Psychological stress and the subsequent appearance of new brain MRI lesions in MS. Neurology. 2000;55:55–61. doi: 10.1212/wnl.55.1.55. [DOI] [PubMed] [Google Scholar]
- Moss H, Bose S, Wolters P, Brouwers P. A preliminary study of factors associated with psychological adjustment and disease course in school-age children infected with the human immunodeficiency virus. Journal of Development and Behavioral Pediatrics. 1998;19:18–25. doi: 10.1097/00004703-199802000-00003. [DOI] [PubMed] [Google Scholar]
- Murphy DA, Moscicki AB, Vermund SH, Muenz LR Adolescent Med HIV AIDS Research Network. Psychological distress among HIV+ adolescents in the REACH study: Effects of life stress, social support, and coping. Journal of Adolescent Health. 2000;27:391–398. doi: 10.1016/s1054-139x(00)00158-0. [DOI] [PubMed] [Google Scholar]
- Paget KD, Reynolds CR. Dimensions, levels and reliabilities on the revised children’s manifest anxiety scale with learning-disabled children. Journal of Learning Disabilities. 1984;17:137–141. doi: 10.1177/002221948401700302. [DOI] [PubMed] [Google Scholar]
- Peterson BS, Pine DS, Cohen P, Brook JS. Prospective, longitudinal study of tic, obsessive- compulsive, and attention-deficit/hyperactivity disorders in an epidemiological sample. Journal of American Academy of Child and Adolescent Psychiatry. 2001;40:685–695. doi: 10.1097/00004583-200106000-00014. [DOI] [PubMed] [Google Scholar]
- Poznanski EO, Freeman LN, Mokros HB. Children’s Depression Rating Scale–Revised. Psychopharmacological Bulletin. 1985;21:979–989. [Google Scholar]
- Rabe-Hesketh S, Skrondal A, Pickles A. GLLAMM Manual. U.C. Berkeley Division of Biostatistics Working Paper Series. Working Paper 160. 2004 Available from: http://www.bepress.com/ucbbiostat/paper160.
- Robertson MM, Banerjee S, Eapen V, Fox-Hiley P. Obsessive-compulsive behaviour and depressive symptoms in young people with Tourette syndrome. A controlled study. European Journal Child and Adolescent Psychiatry. 2002;11:261–265. doi: 10.1007/s00787-002-0301-3. [DOI] [PubMed] [Google Scholar]
- Robertson MM, Williamson F, Eapen V. Depressive symptomatology in young people with Gilles de la Tourette Syndrome – a comparison of self-report scales. Journal of Affective Disorders. 2006 Feb 4; doi: 10.1016/j.jad.2005.12.046. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- Scahill L, Riddle MA, McSwiggan-Hardin MT, Ort SI, King RA, Goodman WK, Leckman JF. Children’s Yale-Brown Obsessive Compulsive Scale: Reliability and validity. American Journal of Child and Adolescent Psychiatry. 1997;36:844–852. doi: 10.1097/00004583-199706000-00023. [DOI] [PubMed] [Google Scholar]
- Silva RR, Munoz DM, Barickman J, Friedhoff AJ. Environmental factors and related fluctuation of symptoms in children and adolescents with Tourette disorder. Journal of Child Psychology and Psychiatry. 1995;36:305–12. doi: 10.1111/j.1469-7610.1995.tb01826.x. [DOI] [PubMed] [Google Scholar]
- Stokes A, Bawden H, Camfield P, Backman J, Dooley J. Peer problems in Tourette disorder. Pediatrics. 1991;87:936–42. [PubMed] [Google Scholar]
- Sukhodolsky DG, Rosario-Campos MC, Scahill L, Katsovich L, Pauls DL, Peterson BS, King RA, Lombroso PJ, Findley D, Leckman JF. Adaptive, emotional, and family functioning of children with obsessive-compulsive disorder and comorbid attention deficit hyperactivity disorder. American Journal of Psychiatry. 2005;162:1125–1132. doi: 10.1176/appi.ajp.162.6.1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sukhodolsky DG, Scahill L, Zhang H, Peterson B, King R, Lombroso PJ, Katsovich L, Findley D, Leckman JF. Disruptive behavior in children with Tourette syndrome: Association with ADHD comorbidity, tic severity, and functional impairment. Journal of American Academy of Child and Adolescent Psychiatry. 2003;42:98–105. doi: 10.1097/00004583-200301000-00016. [DOI] [PubMed] [Google Scholar]
- Surwillo WW, Shafii M, Barrett CL. Gilles-de-la-Tourette syndrome – 20-month study of effects of stressful life events and haloperidol on symptom frequency. Journal of Nervous and Mental Disease. 1978;166:812–816. [PubMed] [Google Scholar]
- The Tourette’s Syndrome Study Group. Treatment of ADHD in children with tics: A randomized controlled trial. Neurology. 2002;58:527–536. doi: 10.1212/wnl.58.4.527. [DOI] [PubMed] [Google Scholar]
- Thernlund GM, Dahlquist G, Hansson K, Ivarsson SA, Ludvigsson J, Sjoblad S, Hagglof B. Psychological stress and the onset of IDDM in children – a case–control study. Diabetes Care. 1995;18:1323–1329. doi: 10.2337/diacare.18.10.1323. [DOI] [PubMed] [Google Scholar]
- Thomsen PH. Obsessive-compulsive disorder in children and adolescents: A study of parental psychopathology and precipitating events in 20 consecutive Danish cases. Psychopathology. 1995;28:161– 167. doi: 10.1159/000284916. [DOI] [PubMed] [Google Scholar]
- Tourette Syndrome Association International Consortium for Genetics. A complete genome screen in sib pairs affected by Gilles de la Tourette syndrome. American Journal of Human Genetics. 1999;65:1428–1436. doi: 10.1086/302613. [DOI] [PMC free article] [PubMed] [Google Scholar]