Abstract
We present the case of a 37 year-old female who had disseminated tuberculosis (TB) with bilateral adrenal involvement resulting in primary adrenal failure (Addison’s disease) with refractory hyponatraemia and possible extrapontine ‘myelinolysis’. Laboratory results were remarkable for adrenal insufficiency. A Mantoux reaction was positive with evidence of pulmonary miliary TB. The magnetic resonance imaging scan showed all the characteristic changes of adrenal TB.
Keywords: TB; Adrenal; Insufficiency; Granuloma; Case report, Oman
Adrenal disseminated tuberculosis causing bilateral adrenal enlargement and Addison’s disease is rarely reported. In many areas of the world, particularly in developing countries, tuberculosis remains a major cause of Addison’s disease. Tuberculosis of the adrenal glands resulting in Addison’s disease must be considered in the right clinical context because early identification and treatment may enable recovery of adrenal function.
Case Report
A 37 year-old woman presented at Sultan Qaboos University Hospital, Oman, with fever, fatigue, vomiting, progressive darkening of the skin, behavioural changes, rigidity and drowsiness over the previous two months. She was born in India, but had lived in Oman for the preceding 15 years. Her last visit to India was 10 years previously. Evidence of a BCG vaccination was present through a scar on the arm. Examination showed a sick looking patient with severe wasting and marked dehydration. She was febrile with low blood pressure (BP 95/64mmHg) and postural drop. The skin and mucosa were hyper-pigmented [Figures 1a and 1b]. She was conscious and oriented, but with markedly diffuse rigidity and bradykinesia. Cranial nerves were normal. Deep tendon reflexes were absent, but plantar reflexes were normal. She had splenomegaly and a large, non-tender, mobile lymph node of 3 × 3 cm in the right axilla. The laboratory findings were as follows: a complete blood count (CBC) showed anaemia (Hb 10.8 g/dl); white blood cells (WBC) 2.97×109/L (monocytes 0.7, eosinophils 1.0) with normal platelet count (240×109/L). The blood film revealed reactive monocytosis and eosinophilia. The sodium was low (120 mmol/L, K 3.7 mmol/L). Serum osmolality was 252mmol/L with a urine sodium of 93mmol/L and urine osmolality of 300mmol/L. Random serum cortisol was very low (<11nmol/L). The adrenocorticotropic hormone (ACTH) level was very high (165.5pmol/L). The Synacthen (ACTH) stimulation test revealed at base line, 30 minutes, and 60 minutes the serum cortisol to be below <11nmol/L. The gonadotrophic hormones, prolactin and thyroid functions tests were within normal limits. The anti-adrenal epithelial cell antibody test was negative. The serology for HIV 1 and 2, cytomegalovirus and toxoplasma gondii were all negative. A tuberculin skin test at 72 hours was positive with a 20mm induration.
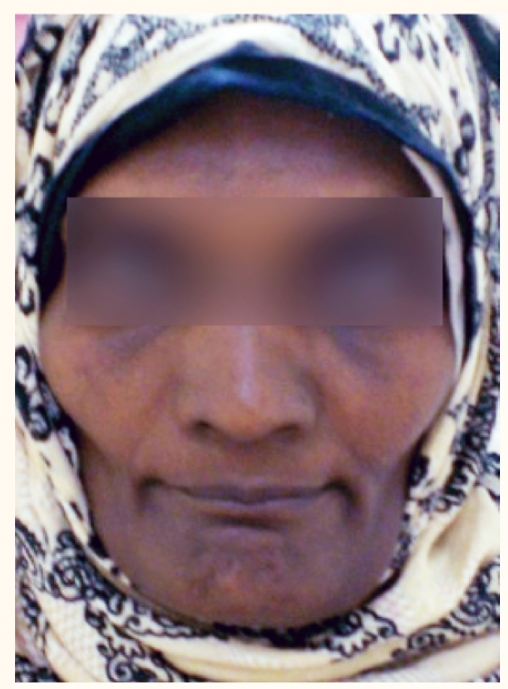
Figure 1a :
Hyperpigmentation of face likely due to hypoadrenalism, in the context of disseminated tuberculosis.
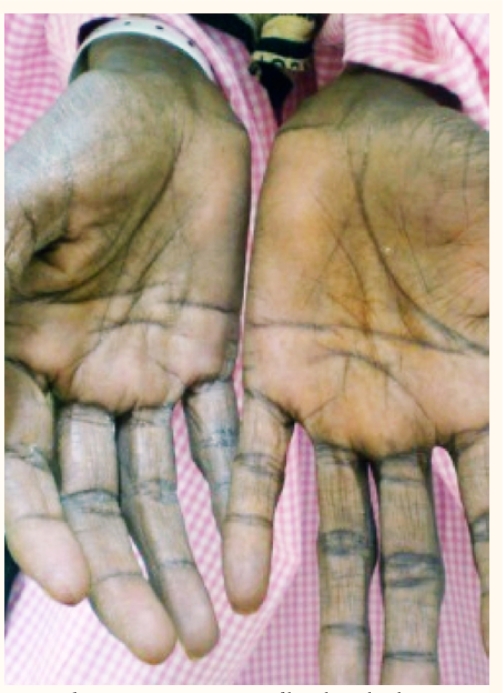
Figure 1b :
Hyperpigmentation of hands and palmar creases.
Imaging, including a chest X-ray, showed a miliary pattern in addition to mild bilateral pleural effusion. A magnetic resonance imaging (MRI) scan of the brain was normal. A repeated MRI brain scan (2 weeks after the initial normal MRI) revealed homogeneous T2 hyper-intensity of bilateral basal ganglia (putamen, caudate, globus pallidus nuclei) with normal signals in the brain stem. There was no meningeal enhancement or basal exudates. An MRI of the abdomen (done because the CT scanner was out of service) revealed moderate enlargement of both adrenal glands with hyper-intensity signals on T2 weighted sequences [Figure 2]. Both adrenal glands showed marginal enhancement with persistent hypo-intensity of the central areas. A lymph node biopsy showed numerous granulomas with central caseous necrosis [Figures 3a and 3b]. No acid fast bacilli were identified; however, a subsequent culture of the lymph node grew mycobacterium tuberculosis sensitive to all first line drugs. The cerebrospinal fluid culture was negative for mycobacterium tuberculosis.
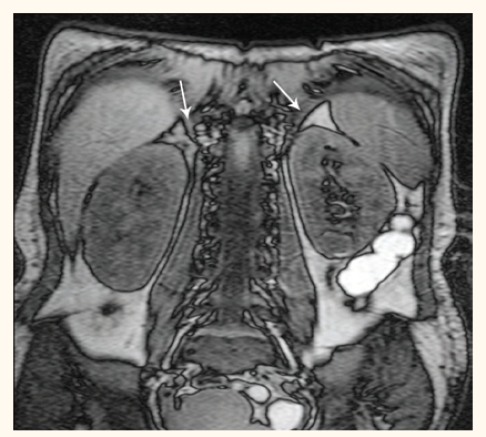
Figure 2:
Abdominal MRI of the patient showing bilaterally enlarged, hyperintense adrenal glands.
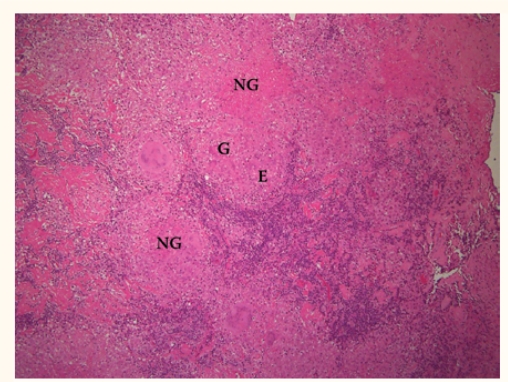
Figure 3a :
Hematoxylin and eosin stain*10 showing a lymph node with multiple necrotising granulomas (NG) composed of multinucleated giant cells (G) and epithelial histiocytes (E).
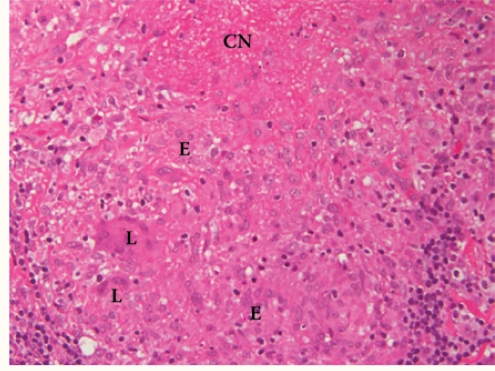
Figure 3b :
Hematoxylin and eosin stain*40 showing granuloma with central necrosis (CN) and composed of epithelioid histiocytes (E) and Langhans giant cells (L).
The patient was started on adrenal hormonal replacement with hydrocortisone 20mg in the morning and 10mg in the evening in addition to fludrocortisone 100μg once daily.
The patient was also empirically commenced on anti-tuberculous medications, on the basis of the clinical suspicion and the radiology findings, with a four-drug regimen consisting of isoniazid, rifampicin, pyrazinamide and ethambutol.
During a month of hospitalisation, the patient made a remarkable recovery with normalisation of the hyponatremia (Na137 mmol/L), resolution of the hypotension, the postural drop and all her other symptoms. At the time of discharge, she was alert and oriented and was able to perform all the functions of daily living. She still had mild bradykinesia and mild diffuse hypertonia, which she seemed to tolerate. Both anti-tuberculous therapy and adrenal hormonal replacement were continued.
Discussion
Disseminated tuberculosis causing bilateral adrenal enlargement and Addison’s disease have been rarely reported.1 Addison’s disease is a primary adrenocortical deficiency that is the result of damage to the adrenal cortex. Overt clinical features of hypoadrenalism occur when 80–90% of both adrenal cortices are destroyed.2
Thomas Addison first described the clinical features and pathogenesis of adrenal insufficiency more than 150 years ago.3 At that time, tuberculosis was the most common cause of this disease. Tuberculosis is still a major cause of adrenal insufficiency in populations with a high prevalence of tuberculosis, but the overall incidence of tuberculous Addison’s disease has decreased.4 Nowadays, tuberculosis accounts for only 7–20% of cases and is often an overlooked cause of Addison’s disease. Recovery of adrenal function is possible after appropriate anti-tuberculosis therapy, though usually it does not occur.2
Tuberculosis may affect many of the endocrine glands including the hypothalamus, pituitary, thyroid, but the most commonly involved endocrine organ is the adrenal gland. In addition to mycobacterial tuberculosis, other mycobacteria, bacteria, viruses and fungi may affect the adrenal glands and lead to the development of adrenal insufficiency. Most cases of adrenal tuberculosis are found 10 to 15 years after the initial infection; hence, tuberculous Addison’s disease has a relatively late onset.2
Bilateral adrenal calcification is the most common radiological feature of Addison’s disease.5 An MRI scan can reveal certain pathological changes of adrenal tuberculosis. In a cohort of 18 patients with proven adrenal tuberculosis, who underwent MRI scans, 97% of the adrenal glands were enlarged with bilateral involvement in 89%, while 83% of the enlarged glands had peripheral rim enhancement and 62% had a central region of T2 hypo/iso-signal intensity.6 Our patient’s MRI study demonstrated all the above characteristic findings.
Conclusion
In many areas of the world, particularly in developing countries, tuberculosis remains a major cause of Addison’s disease. Prompt intervention for Addison’s disease, a high index of suspicion with early identification and treatment of tuberculosis may enable recovery of adrenal function. Tuberculosis of the adrenal glands resulting in Addison’s disease must be considered in the right clinical context.
References
- 1.Bajaj S. Disseminated tuberculosis causing bilateral adrenal enlargement and Addison’s disease. J Assoc Physicians India. 2000;48:919–20. [PubMed] [Google Scholar]
- 2.Alevritis EM, Sarubbi FA, Jordan RM, Peiris AN. Infectious causes of adrenal insufficiency. South Med J. 2003;96:888–90. doi: 10.1097/01.SMJ.0000073269.49575.DF. [DOI] [PubMed] [Google Scholar]
- 3.Addison T. On the constitutional and local effects of disease of the supra-renal capsules. London: Highley Press; 1855. [Google Scholar]
- 4.Nomura K, Demura H, Saruta T. Addison’s disease in Japan: characteristics and changes revealed in a nationwide survey. Intern Med. 1994;33:602–6. doi: 10.2169/internalmedicine.33.602. [DOI] [PubMed] [Google Scholar]
- 5.Orth DN. Adrenal insufficiency. Curr Ther Endocrinol Metab. 1994;5:124–30. [PubMed] [Google Scholar]
- 6.Zhang XC, Yang ZG, Li Y, Min PQ, Guo YK, Deng YP, Dong ZH. Addison’s disease due to adrenal tuberculosis: MRI features. Abdom Imaging. 2008;33:689–94. doi: 10.1007/s00261-007-9352-8. [DOI] [PubMed] [Google Scholar]