Abstract
Exercise training enhances endothelium-dependent coronary vasodilatation, improving perfusion and contractile function of collateral-dependent myocardium. Paradoxically, studies from our laboratory have revealed increased Ca2+-dependent basal active tone in collateral-dependent arteries of exercise-trained pigs. In this study, we tested the hypothesis that exercise training enhances agonist-mediated contractile responses of collateral-dependent arteries by promoting Ca2+ sensitization. Ameroid constrictors were surgically placed around the proximal left circumflex coronary (LCX) artery of female Yucatan miniature pigs. Eight weeks postoperatively, pigs were randomized into sedentary (pen confined) or exercise-training (treadmill run; 5 days/wk; 14 wk) groups. Arteries (∼150 μm luminal diameter) were isolated from the collateral-dependent and nonoccluded (left anterior descending artery supplied) myocardial regions, and measures of contractile tension or simultaneous tension and intracellular free Ca2+ concentration levels (fura-2) were completed. Exercise training enhanced contractile responses to endothelin-1 in collateral-dependent compared with nonoccluded arteries, an effect that was more pronounced in the presence of nitric oxide synthase inhibition (Nω-nitro-l-arginine methyl ester; 100 μM). Contractile responses to endothelin-1 were not altered by coronary occlusion alone. Exercise training produced increased tension at comparable levels of intracellular free Ca2+ concentration in collateral-dependent compared with nonoccluded arteries, indicative of exercise training-enhanced Ca2+ sensitization. Inhibition of PKC (calphostin C; 1 μM), but not Rho-kinase (Y-27632, 10 μM; or hydroxyfasudil, 30 μM), abolished the training-enhanced endothelin-1-mediated contractile response. Exercise training also increased sensitivity to the PKC activator phorbol 12,13-dibutyrate in collateral-dependent compared with nonoccluded arteries. Taken together, these data reveal that exercise training enhances endothelin-1-mediated contractile responses in collateral-dependent coronary arteries likely via increased PKC-mediated Ca2+ sensitization.
Keywords: coronary artery disease, coronary circulation, endothelin, vascular smooth muscle cell, protein kinase C
the coronary circulation is a highly autoregulated vascular bed. Coronary occlusion/stenosis is a common complication that can lead to alterations in coronary autoregulation in patients with coronary artery disease (5, 7). Patients (22, 32, 42) and animal models (37, 38, 49) with coronary occlusion/stenosis exhibit signs of myocardial ischemia as well as contractile dysfunction during periods of increased cardiac workload. In the compromised myocardial regions distal to occlusion, exercise training produces improvements in vascular function, including alterations in endothelial function (8, 9, 12–16, 52), which contribute to enhanced myocardial perfusion and contractile function (6, 14, 15, 42). However, despite overwhelming evidence for the therapeutic benefits of physical activity, the primary mechanisms by which regular exercise improves vascular function in the setting of coronary artery disease have not been fully established.
Recently, our laboratory has demonstrated the development of apparently opposing exercise-induced adaptations including enhanced Ca2+-dependent basal active tone, nitric oxide production, and K+ channel activity in collateral-dependent coronary arteries (16). In light of these findings, we speculate that these seemingly paradoxical exercise training-induced adaptations may work in parallel to provide a more precise control of arterial tone to aid in the regulation of blood flow to collateral-dependent myocardium, depending on the metabolic needs of the heart. Based on our previous findings, we anticipated that in addition to increased Ca2+-dependent basal active tone (16), small arteries from the collateral-dependent myocardium of exercise-trained animals would also display enhanced contractile responses to vasoactive agonists, further contributing to the autoregulation of coronary blood flow in this region. Therefore, in the present study we tested the hypothesis that exercise training enhances agonist-mediated contractile responses of collateral-dependent arteries by promoting Ca2+ sensitization.
MATERIALS AND METHODS
Experimental animals and surgical instrumentation.
All animal protocols were approved by the Texas A&M University Institutional Animal Care and Use Committee and conformed to the National Institutes of Health's Guide for Care and Use of Laboratory Animals (NIH Publication No. 85-23, Revised 2010). Adult female Yucatan miniature swine (6 to 7 mo of age) were surgically instrumented with ameroid constrictors around the proximal left circumflex coronary (LCX) artery as previously described (16–19). Anesthesia was induced with ketamine (20 mg/kg im), maintained with 2 to 3% isoflurane and supplemental O2 throughout aseptic surgery. Animals recovered from surgery for 8 wk before sedentary or exercise-training experimental protocols were initiated. The duration of this recovery period was chosen in light of previous studies using this animal model of chronic occlusion, which demonstrated that collateral development plateaus approximately 8 wk after ameroid placement (48). Thus we are able to assess collateral development in response to exercise training separately from that in response to occlusion. In our experience, total occlusion ensues approximately 3 wk after ameroid placement.
Sedentary and exercise protocols.
Animals were randomly assigned to either a sedentary or exercise-training group. Exercise-trained (n = 39) pigs underwent a progressive treadmill program (5 days/wk for 14 wk) as previously described (12, 13, 16–19). Sedentary (n = 43) animals were confined to their pens. The effectiveness of the exercise-training program was determined by comparing the heart weight-to-body weight ratio and skeletal muscle citrate synthase activity as previously described (12, 13, 19).
Preparation of coronary arteries.
Following the completion of the 14-wk exercise-training protocol or sedentary confinement, animals were anesthetized with ketamine (35 mg/kg im) and pentothal sodium (30 mg/kg iv). Hearts were removed, placed in Krebs bicarbonate buffer (0–4°C), and weighed. Visual inspection of the ameroid occluder during dissection of the LCX artery indicated 100% occlusion in all animals used for this study. Size-matched arteries (∼150 μm internal luminal diameters) were isolated from both the collateral-dependent LCX (distal to occlusion) and the nonoccluded left anterior descending coronary (LAD) artery regions as previously described (16).
Tension studies.
Arterial rings were studied using specialized isometric microvessel myographs (Danish Myograph Technology) as previously described (16, 30). An endothelin-1 concentration-response curve was done in which arterial rings were incubated at each concentration until the contractile response was at steady state. Steady state was obtained within 3 to 10 min after the addition of each concentration of drug. Developed tension (T) was calculated as the milliNewton of force generated (F) per axial vessel length (g; in mm), where T = F/2 g (30). To assess the contribution of Rho-kinase, PKC, and nitric oxide to endothelin-1-mediated tension development, specific inhibitors [Y-27632, 10 μM; or hydroxyfasudil, 30 μM; calphostin C, 1 μM; and NG-nitro-l-arginine methyl ester (l-NAME), 100 μM, respectively] were added to separate rings 15 min before the initiation of endothelin-1 concentration-response curves. In additional studies, concentration-response curves were generated using the PKC activator phorbol 12,13-dibutyrate (PDBu). Because our preliminary data suggested an incomplete washout of endothelin-1 after the completion of a concentration-response curve, only one curve was completed in each arterial ring.
Simultaneous measures of intracellular free Ca2+ and tension development.
In additional studies, intracellular free Ca2+ (fura-2) and contractile tension were measured simultaneously in denuded arterial rings as previously described (17, 19, 36). Rings were denuded of endothelium by gently rubbing the luminal surface with silk surgical suture.
Solutions and drugs.
Krebs bicarbonate buffer contained (in mM) 131.5 NaCl, 5 KCl, 1.2 NaH2PO4, 1.2 MgCl2, 2.5 CaCl2, 11.2 glucose, 13.5 NaHCO3, and 0.025 EDTA. All drugs were obtained from Sigma Chemical unless otherwise noted. Endothelin-1 was purchased from Peninsula Laboratories. Fura-2 acetoxymethyl ester was purchased from Molecular Probes. Calphostin C was purchased from Biomol International.
Data analysis.
Body weight, heart weight-to-body weight ratio, citrate synthase values, dimensional characteristics of coronary arterial rings, and the basal effects of inhibitors on resting tension were analyzed using Student's t-test. Concentration-response relationships were analyzed using repeated-measures two-way ANOVA, followed by Bonferroni correction for multiple comparisons when a main effect was identified. The concentration of endothelin-1 producing 50% of the maximal contractile response (EC50) was calculated by nonlinear regression analysis. EC50 values were not calculated for curves in the presence of Rho-kinase and PKC inhibitors since these curves did not achieve maximal contractile responses. Maximal contractile responses were those observed independent of endothelin-1 concentration. The individual maximum contraction and EC50 values were averaged for nonoccluded and collateral-dependent arteries in the absence and presence of select inhibitors and compared using Student's t-tests. Ca2+ sensitivity data are presented as milliNewtons of tension as a function of fluorescence ratio. Best fit analysis was accomplished using the Stirling equation: f = y0 + a·[exp(b·x) − 1]/b (SPSS, SigmaPlot 9.0). Coefficients of the equation were compared by linear least squares regression (Microsoft Excel 2002). For all analyses, a P value ≤ 0.05 was considered significant. Data are presented as means ± SE, and n values reflect the number of animals studied.
RESULTS
Efficacy of the exercise-training program.
The effectiveness of the 14-wk exercise-training program was demonstrated by a significant increase in the heart weight-to-body weight ratio and increased skeletal muscle oxidative enzyme capacity in exercise-trained compared with sedentary animals. Although body weight did not differ between sedentary and exercise-trained animals at the time of death (35.6 ± 1.0 vs. 37.0 ± 1.0 kg, respectively), the heart weight-to-body weight ratio was significantly greater from exercise-trained (n = 33) compared with sedentary (n = 40) pigs (5.4 ± 0.1 vs. 4.6 ± 0.1 g/kg, respectively). Citrate synthase activity increased significantly in the deltoid muscle (39.0 ± 2.0 vs. 30.6 ± 1.5 μmol·min−1·g−1) and the medial (42.9 ± 2.2 vs. 33.7 ± 1.5 μmol·min−1·g−1) and long (29.1 ± 1.3 vs. 24.0 ± 0.9 μmol·min−1·g−1) heads of the triceps brachii muscle in exercise-trained (n = 31) compared with sedentary (n = 39) pigs, respectively.
Coronary artery dimensions and characteristics.
No significant differences in dimensional characteristics were observed between arterial rings from the nonoccluded (LAD) or collateral-dependent (LCX) regions of either sedentary or exercise-trained animals (Table 1).
Table 1.
Coronary artery dimensions and characteristics
| n | Outer Diameter, μm | Inner Diameter, μm | Wall Thickness, μm | Axial Length, mm | RT Lmax, mN/mm | |
|---|---|---|---|---|---|---|
| Sedentary | 63 | |||||
| Nonoccluded | 239 ± 5 | 143 ± 4 | 47 ± 1 | 1.49 ± 0.01 | 0.69 ± 0.04 | |
| Collateral dependent | 239 ± 4 | 142 ± 3 | 46 ± 1 | 1.49 ± 0.01 | 0.70 ± 0.06 | |
| Exercise trained | 63 | |||||
| Nonoccluded | 237 ± 4 | 139 ± 4 | 47 ± 1 | 1.50 ± 0.01 | 0.62 ± 0.04 | |
| Collateral dependent | 240 ± 4 | 145 ± 4 | 47 ± 1 | 1.49 ± 0.01 | 0.71 ± 0.04 |
Values are means ± SE; n, number of vessels. RT Lmax, resting tension (RT) where maximal active tension (Lmax) to KCl-induced depolarization is developed. No significant differences exist.
Effects of occlusion and exercise training on the contribution of Rho-kinase, PKC, and nitric oxide to basal tone.
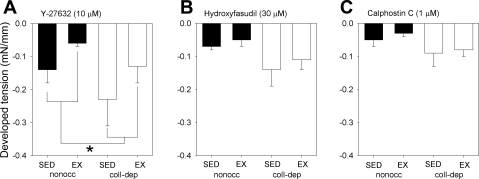
Exposure to the Rho-kinase inhibitors Y-27632 (10 μM) or hydroxyfasudil (30 μM) and the PKC inhibitor calphostin C (1 μM) reduced resting tension in coronary arteries from the nonoccluded and collateral-dependent regions of both sedentary and exercise-trained animals (Fig. 1). Although there is a trend for all inhibitors to reduce tension in collateral-dependent arteries more than nonoccluded vessels, only exposure to Y-27632 had a significantly greater reduction in tension (Fig. 1A). We have previously reported the basal effect of nitric oxide synthase (NOS) inhibition with l-NAME (100 μM) (16).
Fig. 1.
Effects of occlusion and exercise training on the contribution of Rho-kinase and PKC to basal tone. Decrease in baseline tension in nonoccluded (nonocc) and collateral-dependent (coll-dep) arteries in sedentary (SED) and exercise-trained (EX) pigs to 10 μM of Y-27632 (A), 30 μM hydroxyfasudil (B), and 1 μM calphostin C (C). *P < 0.05 nonocc vs. coll-dep.
Endothelin-1 concentration-response relationships.
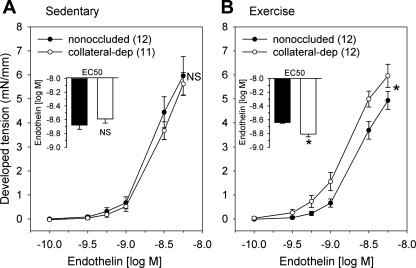
We compared steady-state contractile responses of arteries from the nonoccluded and collateral-dependent myocardial regions to increasing concentrations of endothelin-1 (Fig. 2). In sedentary animals, concentration-dependent contractile responses to endothelin-1 were not different in arteries isolated from the collateral-dependent compared with the nonoccluded region (Fig. 2A). In contrast, collateral-dependent arteries from exercise-trained animals displayed significantly enhanced constriction compared with arteries from the nonoccluded region (Fig. 2B). Furthermore, coronary arteries isolated from the collateral-dependent region displayed significantly increased endothelin-1-mediated contractile responses in exercise-trained compared with sedentary animals. The evaluation of EC50 values revealed that collateral-dependent arteries of exercise-trained animals displayed significantly increased sensitivity to endothelin-1 compared with nonoccluded arteries of exercise-trained and collateral-dependent arteries of sedentary animals (Fig. 2B, inset, and Table 2). EC50 values were not different between collateral-dependent and nonoccluded arteries of sedentary animals (Fig. 2A, inset). Furthermore, maximal endothelin-1-induced contraction was not significantly altered by occlusion or exercise training (Table 2).
Fig. 2.
Endothelin-1 concentration-response relationships. Endothelin-1 concentration-response curves and EC50 values (inset) between nonoccluded and collateral-dependent arteries in sedentary (A) and exercise-trained (B) pigs. Concentration-response curves for endothelin-1 were truncated at the concentration (3 nM) because arteries generally displayed reduced endothelin-1-mediated contraction at subsequent concentrations. NS, no significant difference. *P < 0.05; n, number of animals (shown in parentheses in Figs. 2–7).
Table 2.
Endothelin-1-mediated maximal contraction and EC50 values in the absence and presence of the nitric oxide synthase inhibitor l-NAME
| SED |
EX |
|||
|---|---|---|---|---|
| Nonoccluded | Collateral-dep | Nonoccluded | Collateral-dep | |
| ET-1 | ||||
| Maximal, mN/mm | 6.80 ± 0.75 | 6.55 ± 0.66 | 5.89 ± 0.53 | 6.57 ± 0.59 |
| EC50, log M | −8.65 ± 0.02 | −8.59 ± 0.04 | −8.63 ± 0.04 | −8.79 ± 0.06a,b |
| ET-1 + l-NAME | ||||
| Maximal, mN/mm | 7.69 ± 0.70 | 6.22 ± 0.67 | 6.73 ± 0.84 | 6.65 ± 0.48 |
| EC50, log M | −8.63 ± 0.04 | −8.82 ± 0.05a,d | −8.75 ± 0.05 | −9.04 ± 0.06c,e,f |
Values are means ± SE; n values are same as indicated in respective concentration-response curves from Figs. 2 and 3. Significant difference from
endothelin-1 (ET-1) sedentary (SED) collateral-dependent (dep),
ET-1 exercise-trained (EX) nonoccluded,
ET-1 EX collateral-dependent,
ET-1 + Nω-nitro-l-arginine methyl ester (l-NAME) SED nonoccluded,
ET-1 + l-NAME SED collateral-dependent, and
ET-1 + l-NAME EX nonoccluded pigs is indicated.
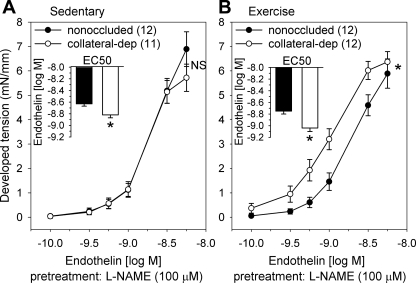
Effect of NOS inhibition on endothelin-1-mediated constriction.
Preincubation with the NOS inhibitor l-NAME (100 μM) resulted in a significant leftward shift of endothelin-1 EC50 values in collateral-dependent arteries of both sedentary and exercise-trained animals compared with EC50 values in the absence of l-NAME (Table 2). In contrast, NOS inhibition did not significantly alter EC50 values in arteries from the nonoccluded region of sedentary or exercise-trained animals. Importantly, the greater response to NOS inhibition in the collateral-dependent arteries of sedentary animals suggests that enhanced nitric oxide production masks an increased sensitivity to endothelin-1 compared with that in nonoccluded arteries. The increased sensitivity to endothelin-1 (EC50) in collateral-dependent arteries of sedentary animals occurred despite no difference in concentration-response curves between nonoccluded and collateral-dependent arteries by analysis of variance and is likely attributable to the tendency for an increased maximal contraction in nonoccluded arteries (Fig. 3A). Furthermore, concentration-response curves to endothelin-1 were significantly enhanced in arteries isolated from the collateral-dependent compared with the nonoccluded region of exercise-trained animals (Fig. 3B), consistent with that observed in the absence of NOS inhibition (Fig. 2B).
Fig. 3.
Effect of NOS inhibition on endothelin-1-mediated constriction. Endothelin-1 concentration-response curves in the presence of 100 μM Nω-nitro-l-arginine methyl ester (l-NAME) between nonoccluded and collateral-dependent arteries in sedentary (A) and exercise-trained (B) pigs. Concentration-response curves for endothelin-1 plus l-NAME were truncated at the concentration 3 nM because arteries generally displayed reduced endothelin-1-mediated contraction at subsequent concentrations. *P < 0.05.
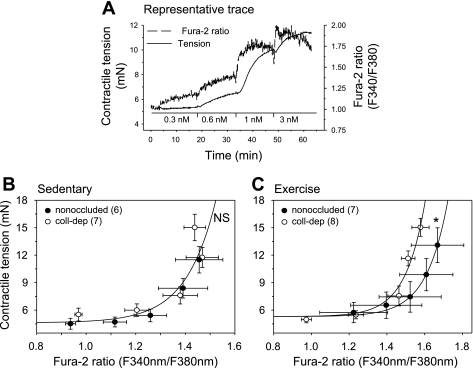
Simultaneous contractile tension and intracellular free Ca2+.
To determine whether enhanced contractile responses to endothelin-1 in collateral-dependent arteries of exercise-trained animals were associated with increased Ca2+ sensitivity, simultaneous measures of endothelin-1-mediated changes in contractile tension and intracellular free Ca2+ levels in endothelium-denuded arteries from the collateral-dependent and nonoccluded regions of both sedentary and exercise-trained pigs were acquired (Fig. 4). Figure 4A represents our experimental protocol in which arterial rings were constricted with increasing concentrations of endothelin-1, and the developed tension, as a function of fura-2 ratio, was obtained. Simultaneous measures of contractile tension and intracellular free Ca2+ levels in response to increasing concentrations of endothelin-1 revealed that occlusion alone did not alter Ca2+ sensitivity in the coronary vasculature, since best fit analysis for each set of data was obtained by the same equation (Fig. 4B). On the other hand, best fit analysis was obtained with different equations for nonoccluded {y = 5.302 + 2.58e-5·[exp(8.874·x) − 1]/8.874} compared with collateral-dependent {y = 5.302 + 2.58e-5·[exp(9.583·x) − 1]/9.583} arteries (Fig. 4C) of exercise-trained pigs. The significantly greater tension at comparable levels of intracellular Ca2+ in the collateral-dependent compared with nonoccluded arteries suggests a synergistic effect of occlusion and exercise training on Ca2+ sensitivity. The comparison of Fig. 4B and 4C demonstrates that exercise training produced an overall reduction in Ca2+ sensitization compared with arteries from sedentary pigs.
Fig. 4.
Simultaneous contractile tension and intracellular free Ca2+ to physiological saline solution, 0.3, 0.6, 1, and 3 nM of endothelin-1. A: experimental protocol depicting developed tension and intracellular free Ca2+ from arterial rings exposed to increasing concentrations of endothelin-1. Developed tension as a function of fura-2 ratio between nonoccluded and collateral-dependent arteries in sedentary (B) and exercise-trained (C) pigs. Best fit analysis was obtained for each data set using the Stirling equation: f = y0 + a·[exp(b·x)−1]/b (SPSS, SigmaPlot 9.0). B: sedentary pigs' best fit analysis was obtained with the same equation: y = 4.621 + 0.0003·[exp(8.383·x) − 1]/8.383. C: exercise-trained pigs' best fit analysis was obtained with different equations for nonoccluded {y = 5.302 + 2.58e-5·[exp(8.874·x) − 1]/8.874} vs. collateral-dependent {y = 5.302 + 2.58e-5·[exp(9.583·x) − 1]/9.583} arteries. *P < 0.05.
Effect of Rho-kinase inhibition on endothelin-1-mediated constriction.
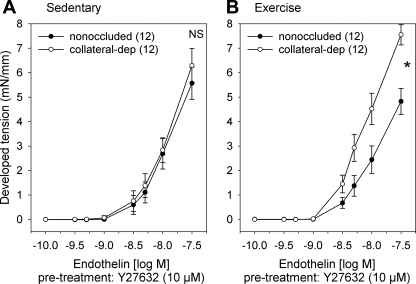
To determine the contribution of the Rho-kinase signaling pathway to endothelin-1-mediated tension development, we compared steady-state contractile responses of arteries from the nonoccluded and collateral-dependent myocardial regions to increasing concentrations of endothelin-1 in the presence of the specific Rho-kinase inhibitor Y-27632 (Fig. 5). Rho-kinase inhibition similarly attenuated endothelin-1-mediated contractile response in arteries from both the nonoccluded and collateral-dependent regions of sedentary (Fig. 5A) and exercise-trained (Fig. 5B) animals. Importantly, enhanced endothelin-1 constriction in collateral-dependent arteries of exercise-trained pigs was not corrected by Rho-kinase inhibition. Additionally, responses in the presence of the Rho-kinase inhibitor hydroxyfasudil (30 μM) were similar to that observed with Y-27632 and also did not correct the exercise training-enhanced endothelin-1-mediated constriction in collateral-dependent arteries (data not shown).
Fig. 5.
Effect of Rho-kinase inhibition on endothelin-1-mediated constriction. Endothelin-1 concentration-response curves in the presence of 10 μM Y-27632 between nonoccluded and collateral-dependent arteries in sedentary (A) and exercise-trained (B) pigs. *P < 0.05.
Effect of PKC inhibition on endothelin-1-mediated constriction.
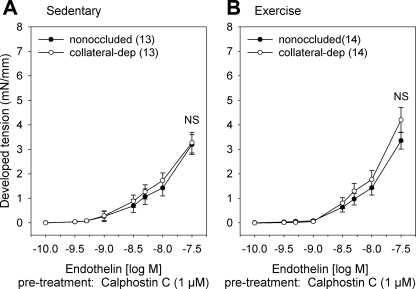
To assess the contribution of PKC to endothelin-1-mediated tension development, we compared steady-state contractile responses of arteries from the nonoccluded and collateral-dependent myocardial regions to increasing concentrations of endothelin-1 in the presence of the specific PKC inhibitor calphostin C (Fig. 6). PKC inhibition significantly attenuated endothelin-1-mediated contractile response in arteries from both the nonoccluded and collateral-dependent regions of sedentary (Fig. 6A) and exercise-trained (Fig. 6B) pigs. In sedentary pigs, PKC inhibition similarly reduced endothelin-1-mediated contractile response in arteries from both the nonoccluded and collateral-dependent regions, suggesting PKC-mediated Ca2+ sensitization was not altered by occlusion alone (Fig. 6A). Importantly, PKC inhibition corrected exercise training-enhanced endothelin-1-mediated constriction in collateral-dependent arteries, resulting in similar contractile responses to endothelin-1 in nonoccluded and collateral-dependent arteries of exercise-trained pigs in the presence of calphostin C (Fig. 6B).
Fig. 6.
Effect of PKC inhibition on endothelin-1-mediated constriction. Endothelin-1 concentration-response curves in the presence of 1 μM calphostin C between nonoccluded and collateral-dependent arteries in sedentary (A) and exercise-trained (B) pigs.
PDBu concentration-response relationships.
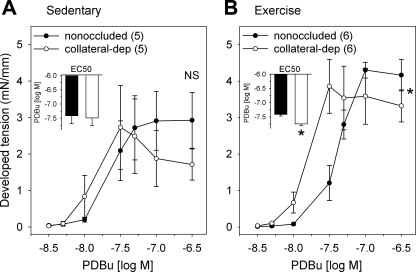
To further investigate the involvement of PKC in the exercise training-enhanced Ca2+ sensitivity of collateral-dependent arteries, we compared steady-state contractile responses of arteries from the nonoccluded and collateral-dependent myocardial regions of both sedentary and exercise-trained animals to increasing concentrations of PDBu (Fig. 7). PDBu is a phorbol ester that activates PKC isozymes by binding to their cysteine-rich zinc-fingerlike sequences (34). Concentration-response curves to PDBu revealed similar responses in nonoccluded and collateral-dependent arteries of sedentary pigs (Fig. 7A). In contrast, collateral-dependent arteries of exercise-trained pigs revealed a significantly enhanced contractile response to PDBu when compared with nonoccluded arteries (Fig. 7B). Further evaluation revealed that sensitivity to PDBu was not different between collateral-dependent and nonoccluded arteries of sedentary animals (Fig. 7A, inset), whereas EC50 values from collateral-dependent arteries from exercise-trained animals displayed a significantly greater sensitivity to PDBu (Fig. 7B, inset) than nonoccluded arteries.
Fig. 7.
Phorbol 12,13-dibutyrate (PDBu) concentration-response curves. PDBu concentration-response curves and EC50 values (inset) between nonoccluded and collateral-dependent arteries in sedentary (A) and exercise-trained (B) pigs. *P < 0.05.
DISCUSSION
In the present study, we document the novel findings that coronary arteries isolated from the collateral-dependent region of exercise-trained pigs display increased endothelin-1-mediated contractile responses compared with arteries from the nonoccluded region of exercise-trained pigs and with both the nonoccluded and collateral-dependent regions of sedentary animals. We also provide evidence that the mechanism for the increased endothelin-1-mediated tension development is enhanced Ca2+ sensitization of the smooth muscle contractile apparatus, as established by the increased tension development at comparable intracellular Ca2+ levels in arteries from the collateral-dependent region of exercise-trained animals. To our knowledge, this is the first report of an effect of exercise training on Ca2+ sensitization of coronary smooth muscle distal to occlusion/stenosis.
Our data also reveal that NOS inhibition produced a leftward shift in the contractile responses to endothelin-1 in collateral-dependent arteries from both sedentary and exercise-trained animals. These findings are supported by our previous observation that phosphorylated endothelial NOS (p-eNOS; p-S1177) protein levels are increased in the collateral-dependent arteries of sedentary and exercise-trained animals in our model of chronic occlusion (16) and suggest that nitric oxide bioavailability is enhanced in collateral-dependent arteries during endothelin-1 stimulation. Interestingly, the effects of NOS inhibition on endothelin-1-mediated contractile responses were most marked in arteries from the collateral-dependent region of exercise-trained animals, as were our previously reported protein levels for both eNOS and p-eNOS (16, 52). However, despite an enhanced contribution of nitric oxide, increased endothelin-1-mediated contraction persisted in the collateral-dependent arteries of exercise-trained animals.
In this study, simultaneous measures of endothelin-1-mediated changes in contractile tension and intracellular free Ca2+ in denuded arteries (Fig. 4) revealed that collateral-dependent vessels of exercise-trained pigs display a greater Ca2+ sensitization compared with their nonoccluded counterparts. Comparing the data in Fig. 4, B and C, reveals that exercise training produced an overall reduction in Ca2+ sensitization when compared with arteries from sedentary pigs, despite the significantly increased endothelin-1 contractile tension in collateral-dependent arteries of exercise-trained animals in Fig. 2. Taken together, these data suggest that the increased contractile responses to endothelin-1 in collateral-dependent arteries of exercise-trained pigs compared with arteries from sedentary animals (Fig. 2) is attributable to a mediator other than Ca2+ sensitization. Exercise training may induce alterations in other mechanisms of Ca2+ regulation, adaptations in signaling pathways that oppose vasoconstriction, or both. Indeed, exercise training has been reported to increase voltage-gated Ca2+ channel current in porcine coronary artery smooth muscle cells (3). Importantly, the comparison of arteries from exercise-trained pigs (Fig. 4C) indicates that Ca2+ sensitization is enhanced in collateral-dependent compared with nonoccluded arteries and likely contributes to the difference in contractile tension between these arteries.
PKC (25, 44) and the Rho/Rho-kinase (21, 43) signaling pathways have been reported to enhance Ca2+ sensitization of the contractile apparatus through inhibition of myosin light chain phosphatase. In this study, pretreatment with the PKC inhibitor calphostin C and not the Rho-kinase inhibitors Y-27632 and hydroxyfasudil reversed the enhanced endothelin-1-mediated contraction and sensitivity to endothelin-1 in collateral-dependent arteries of exercise-trained animals. Importantly, concentrations of calphostin C (1 μM) used in this study have been shown to not alter intracellular Ca2+ levels in porcine coronary arteries (33). Taken together, these data indicate that the enhanced endothelin-1-induced contraction in small collateral-dependent coronary arteries from exercise-trained pigs is likely attributable to enhanced PKC- and not Rho/Rho-kinase-mediated Ca2+ sensitization. However, these data did not discern whether endothelin-1-mediated PKC activation resulted from increases in the PKC signaling pathway and its components and/or enhanced responsiveness of contractile proteins to direct activation of PKC as previously suggested (1). Therefore, we used the diacylglycerol mimetic PDBu to further define the enhanced PKC-mediated Ca2+ sensitization. PDBu concentration-response relationships demonstrated that collateral-dependent arteries isolated from exercise-trained pigs are indeed more responsive to phorbol ester activation compared with the nonoccluded control arteries. Although these findings do not rule out the possibility of endothelin-1-stimulated increases in the PKC signaling pathway and its components, they do provide evidence that the enhanced PKC-mediated Ca2+ sensitization is in part due to an increased responsiveness of contractile proteins to PKC activation.
It is interesting that these unique adaptations were specific to arteries from the collateral-dependent region of exercise-trained animals, suggesting that the combination of occlusion and exercise training stimulated the observed responses. Our observation of enhanced nitric oxide and PKC contributions in small arteries of the collateral-dependent region of exercise-trained animals may reflect a delicate balance of endogenous vasoactive agents that aid in the maintenance of a functional coronary flow reserve that is accessible during periods of increased metabolic demand. Indeed, exercise-induced increases in arterial shear stress likely stimulate nitric oxide production (50, 51) and subsequently enhance vasodilatation of coronary resistance arteries to increase myocardial blood flow into the myocardium at risk. Furthermore, while endothelin-1 contributes to basal vascular tone in resistance arteries of the coronary circulation, the vasoconstrictor effect of endothelin-1 is reduced during exercise (27, 29, 45). Thus the adaptations observed in arteries from the collateral-dependent region of exercise-trained pigs may function to optimize the control of arterial tone at rest, yet maintain the blood flow required to meet the metabolic needs of the heart during exercise. However, it is difficult to reconcile our findings of an increased contribution of PKC in collateral-dependent arteries after exercise training with a growing body of evidence that suggests a role for PKC in the pathogenesis of ischemic heart disease (20, 31). Indeed, studies have reported that PKC modulation provides protection against myocardial ischemic injury in vitro as well as in vivo in various species including pigs (4). On the other hand, it is seemingly paradoxical in our study that both PKC- and nitric oxide-dependent responses are upregulated in collateral-dependent arteries from exercise-trained animals. As we have previously reported, both eNOS and p-eNOS protein levels are increased in these arteries (16), adaptations that may be most exploited during periods of increased metabolic demand (47). The observed increase in PKC-mediated contraction may arise to offset increased nitric oxide production in these arteries, as we have previously reported (16). We speculate that these concurrent adaptations may allow for more precise adjustments in vascular tone to meet the metabolic demands of the heart. It is also noteworthy that PKC may be upregulated for its role in additional cellular functions including migration, proliferation, differentiation, and gene transcription (23), functions that may be highly active in the dynamic environment of collateral growth and development. Our data do not permit us to conclude whether PKC and/or nitric oxide are primary or compensatory mechanisms in coronary collateral growth and function.
We previously reported no change in endothelin-1-mediated Ca2+ sensitization in collateral-dependent epicardial coronary arteries in our porcine model of chronic occlusion (17), which is consistent with current findings in the microcirculation. On the other hand, collateral-dependent vessels from sedentary pigs in our previous study displayed altered Ca2+ sensitivity in response to KCl-induced depolarization (17). It is possible that differences in the Ca2+-sensitizing mechanisms between KCl and endothelin-1 are responsible for the different responses in collateral-dependent arteries of sedentary pigs observed in our previous study. Indeed, it has been shown that depolarization with KCl induces substantial Rho activation and Rho-kinase-dependent contraction (39), whereas this study strongly suggests that PKC, but not Rho-kinase, is responsible for the enhanced endothelin-1-mediated Ca2+ sensitization observed in collateral-dependent arteries from exercise-trained pigs.
Limitations.
Previous studies have revealed that the blockade of endothelin-1 receptors in resting, conscious pigs increases coronary venous blood O2 saturation (27), suggesting that basal endogenous levels of endothelin-1 produce a level of basal active tone (27, 45). Although the endothelin-1 concentrations used in these in vitro studies are higher than plasma concentrations reported in normal or pathological states (24, 26), the reported levels of circulating endothelin-1 are lower than concentrations necessary to elicit contraction in vitro. Furthermore, the administration of physiological plasma levels of endothelin-1 typically produces vasodilatation, likely attributable to endothelin-1-mediated nitric oxide production at lower endothelin-1 concentrations (10, 46). It is widely recognized that at low concentrations of endothelin-1, the stimulation of endothelin receptors subtype B on endothelium produces vasodilatation, whereas higher endothelin-1 concentrations produce vasoconstriction via endothelin receptors subtypes A and B on vascular smooth muscle (41). Thus, since endothelin-1 release is abluminal and based on stoichiometric binding conditions that suggest most endothelin-1 molecules are bound to receptors and are not present in free form (11), circulating endothelin-1 levels likely do not reflect the generally higher concentration at the vascular smooth muscle layer. Therefore, the physiological or pathological relevance of the endothelin-1 concentrations used in our study remains to be determined, especially as relevant to the diseased coronary circulation.
Clinical implications and conclusions.
Despite the dogma that myocardial ischemia associated with coronary artery stenosis causes maximal dilatation of coronary resistance arteries in the ischemic region, emerging clinical evidence indicates that during periods of increased metabolic demand, coronary vasomotor tone persists distal to the site of stenosis and contributes to episodes of ischemia in patients with chronic stable angina (35, 40). Furthermore, in compromised myocardial regions of coronary artery disease patients, paradoxical microvascular constriction occurred as metabolic demand increased (40). Recent evidence highlighting PKC as a potential player in ischemic heart disease (4, 20, 31) has increased the study of PKC modulators as an attractive potential pharmacological treatment of myocardial ischemia. However, the nonpharmacological intervention of exercise training may produce adaptations in multiple vasoactive pathways that function together to optimize blood flow to the compromised region. Indeed, the role of regular exercise has garnered increasing attention from both researchers and clinicians as a mechanism by which myocardial perfusion and function may be substantially improved in patients with coronary artery disease. Whether pharmacological agents, such as selective PKC modulators, would provide benefits in addition to an exercise-training regimen remains to be determined.
In conclusion, our studies not only demonstrate that small coronary arteries (∼150 μm internal luminal diameter) isolated from the collateral-dependent region of exercise-trained animals display increased endothelin-1-induced contraction that is attributable to enhanced Ca2+ sensitization but also implicate the PKC signaling pathway as a strong candidate to mediate this response. In addition, although seemingly paradoxical, we also provide evidence of an increased role for nitric oxide synthase during endothelin-1-mediated constriction in the collateral-dependent arteries following an exercise-training regimen. Because the vasoconstrictor effect of endothelin-1 wanes during exercise (28) and nitric oxide production is increased during exercise (2), these adaptations may function to differentially optimize the control of arterial tone and maintain blood flow appropriate for the metabolic needs of the collateral-dependent myocardial region.
GRANTS
These studies were supported by National Heart, Lung, and Blood Institute Grant R01-HL-064931.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
ACKNOWLEDGMENTS
We greatly appreciate the technical and surgical expertise of Mildred Mattox and excellent technical contributions of Jeff Bray.
REFERENCES
- 1. Abebe W, Mozaffari MS. Vascular reactivity changes in glucose-intolerant rat. J Cardiovasc Pharmacol 50: 590– 597, 2007 [DOI] [PubMed] [Google Scholar]
- 2. Bernstein RD, Ochoa FY, Xu X, Forfia P, Shen W, Thompson CI, Hintze TH. Function and production of nitric oxide in the coronary circulation of the conscious dog during exercise. Circ Res 79: 840– 848, 1996 [DOI] [PubMed] [Google Scholar]
- 3. Bowles DK, Hu Q, Laughlin MH, Sturek M. Exercise training increases L-type calcium current density in coronary smooth muscle. Am J Physiol Heart Circ Physiol 275: H2159– H2169, 1998 [DOI] [PubMed] [Google Scholar]
- 4. Budas GR, Churchill EN, Mochly-Rosen D. Cardioprotective mechanisms of PKC isozyme-selective activators and inhibitors in the treatment of ischemia-reperfusion injury. Pharmacol Res 55: 523– 536, 2007 [DOI] [PubMed] [Google Scholar]
- 5. Christofferson RD, Lehmann KG, Martin GV, Every N, Caldwell JH, Kapadia SR. Effect of chronic total coronary occlusion on treatment strategy. Am J Cardiol 95: 1088– 1091, 2005 [DOI] [PubMed] [Google Scholar]
- 6. Ehsani AA, Heath GW, Hagberg JM, Sobel BE, Holloszy JO. Effects of 12 months of intense exercise training on ischemic ST-segment depression in patients with coronary artery disease. Circulation 64: 1116– 1124, 1981 [DOI] [PubMed] [Google Scholar]
- 7. Fiorilli R, DeFelice F, Violini R. [Chronic total coronary occlusions: an overview]. G Ital Cardiol (Rome) 7: 780– 797, 2006 [PubMed] [Google Scholar]
- 8. Fogarty JA, Delp MD, Muller-Delp JM, Laine GA, Parker JL, Heaps CL. Neuropilin-1 is essential for enhanced VEGF(165)-mediated vasodilatation in collateral-dependent coronary arterioles of exercise-trained pigs. J Vasc Res 46: 152– 161, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Fogarty JA, Muller-Delp JM, Delp MD, Mattox ML, Laughlin MH, Parker JL. Exercise training enhances vasodilation responses to vascular endothelial growth factor in porcine coronary arterioles exposed to chronic coronary occlusion. Circulation 109: 664– 670, 2004 [DOI] [PubMed] [Google Scholar]
- 10. Folta A, Joshua IG, Webb RC. Dilator actions of endothelin in coronary resistance vessels and the abdominal aorta of the guinea pig. Life Sci 45: 2627– 2635, 1989 [DOI] [PubMed] [Google Scholar]
- 11. Frelin C, Guedin D. Why are circulating concentrations of endothelin-1 so low? Cardiovasc Res 28: 1613– 1622, 1994 [DOI] [PubMed] [Google Scholar]
- 12. Griffin KL, Laughlin MH, Parker JL. Exercise training improves endothelium-mediated vasorelaxation after chronic coronary occlusion. J Appl Physiol 87: 1948– 1956, 1999 [DOI] [PubMed] [Google Scholar]
- 13. Griffin KL, Woodman CR, Price EM, Laughlin MH, Parker JL. Endothelium-mediated relaxation of porcine collateral-dependent arterioles is improved by exercise training. Circulation 104: 1393– 1398, 2001 [DOI] [PubMed] [Google Scholar]
- 14. Hambrecht R, Adams V, Erbs S, Linke A, Krankel N, Shu Y, Baither Y, Gielen S, Thiele H, Gummert JF, Mohr FW, Schuler G. Regular physical activity improves endothelial function in patients with coronary artery disease by increasing phosphorylation of endothelial nitric oxide synthase. Circulation 107: 3152– 3158, 2003 [DOI] [PubMed] [Google Scholar]
- 15. Hambrecht R, Wolf A, Gielen S, Linke A, Hofer J, Erbs S, Schoene N, Schuler G. Effect of exercise on coronary endothelial function in patients with coronary artery disease. N Engl J Med 342: 454– 460, 2000 [DOI] [PubMed] [Google Scholar]
- 16. Heaps CL, Mattox ML, Kelly KA, Meininger CJ, Parker JL. Exercise training increases basal tone in arterioles distal to chronic coronary occlusion. Am J Physiol Heart Circ Physiol 290: H1128– H1135, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Heaps CL, Parker JL, Sturek M, Bowles DK. Altered calcium sensitivity contributes to enhanced contractility of collateral-dependent coronary arteries. J Appl Physiol 97: 310– 316, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Heaps CL, Sturek M, Price EM, Laughlin MH, Parker JL. Sarcoplasmic reticulum Ca2+ uptake is impaired in coronary smooth muscle distal to coronary occlusion. Am J Physiol Heart Circ Physiol 281: H223– H231, 2001 [DOI] [PubMed] [Google Scholar]
- 19. Heaps CL, Sturek M, Rapps JA, Laughlin MH, Parker JL. Exercise training restores adenosine-induced relaxation in coronary arteries distal to chronic occlusion. Am J Physiol Heart Circ Physiol 278: H1984– H1992, 2000 [DOI] [PubMed] [Google Scholar]
- 20. Inagaki K, Churchill E, Mochly-Rosen D. Epsilon protein kinase C as a potential therapeutic target for the ischemic heart. Cardiovasc Res 70: 222– 230, 2006 [DOI] [PubMed] [Google Scholar]
- 21. Kitazawa T, Gaylinn BD, Denney GH, Somlyo AP. G-protein-mediated Ca2+ sensitization of smooth muscle contraction through myosin light chain phosphorylation. J Biol Chem 266: 1708– 1715, 1991 [PubMed] [Google Scholar]
- 22. Kolibash AJ, Bush CA, Wepsic RA, Schroeder DP, Tetalman MR, Lewis RP. Coronary collateral vessels: spectrum of physiologic capabilities with respect to providing rest and stress myocardial perfusion, maintenance of left ventricular function and protection against infarction. Am J Cardiol 50: 230– 238, 1982 [DOI] [PubMed] [Google Scholar]
- 23. Larsson C. Protein kinase C and the regulation of the actin cytoskeleton. Cell Signal 18: 276– 284, 2006 [DOI] [PubMed] [Google Scholar]
- 24. Lerman A, Hildebrand FL, Jr, Aarhus LL, Burnett JC., Jr Endothelin has biological actions at pathophysiological concentrations. Circulation 83: 1808– 1814, 1991 [DOI] [PubMed] [Google Scholar]
- 25. Li L, Eto M, Lee MR, Morita F, Yazawa M, Kitazawa T. Possible involvement of the novel CPI-17 protein in protein kinase C signal transduction of rabbit arterial smooth muscle. J Physiol 508: 871– 881, 1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. McMurray JJ, Ray SG, Abdullah I, Dargie HJ, Morton JJ. Plasma endothelin in chronic heart failure. Circulation 85: 1374– 1379, 1992 [DOI] [PubMed] [Google Scholar]
- 27. Merkus D, Haitsma DB, Fung TY, Assen YJ, Verdouw PD, Duncker DJ. Coronary blood flow regulation in exercising swine involves parallel rather than redundant vasodilator pathways. Am J Physiol Heart Circ Physiol 285: H424– H433, 2003 [DOI] [PubMed] [Google Scholar]
- 28. Merkus D, Houweling B, Mirza A, Boomsma F, vandenMeiracker AH, Duncker DJ. Contribution of endothelin and its receptors to the regulation of vascular tone during exercise is different in the systemic, coronary and pulmonary circulation. Cardiovasc Res 59: 745– 754, 2003 [DOI] [PubMed] [Google Scholar]
- 29. Merkus D, Houweling B, vandenMeiracker AH, Boomsma F, Duncker DJ. Contribution of endothelin to coronary vasomotor tone is abolished after myocardial infarction. Am J Physiol Heart Circ Physiol 288: H871– H880, 2005 [DOI] [PubMed] [Google Scholar]
- 30. Mulvany MJ, Halpern W. Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats. Circ Res 41: 19– 26, 1977 [DOI] [PubMed] [Google Scholar]
- 31. Murphy S, Frishman WH. Protein kinase C in cardiac disease and as a potential therapeutic target. Cardiol Rev 13: 3– 12, 2005 [DOI] [PubMed] [Google Scholar]
- 32. Niebauer J, Hambrecht R, Marburger C, Hauer K, Velich T, vonHodenberg E, Schlierf G, Kübler W, Schuler G. Impact of intensive physical exercise and low-fat diet on collateral vessel formation in stable angina pectoris and angiographically confirmed coronary artery disease. Am J Cardiol 76: 771– 775, 1995 [DOI] [PubMed] [Google Scholar]
- 33. Nobe K, Paul RJ. Distinct pathways of Ca2+ sensitization in porcine coronary artery: effects of Rho-related kinase and protein kinase C inhibition on force and intracellular Ca2+. Circ Res 88: 1283– 1290, 2001 [DOI] [PubMed] [Google Scholar]
- 34. Ono Y, Fujii T, Igarashi K, Kuno T, Tanaka C, Kikkawa U, Nishizuka Y. Phorbol ester binding to protein kinase C requires a cysteine-rich zinc-finger-like sequence. Proc Natl Acad Sci USA 86: 4868– 4871, 1989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Pupita G, Maseri A, Kaski JC, Galassi AR, Gavrielides S, Davies G, Crea F. Myocardial ischemia caused by distal coronary-artery constriction in stable angina pectoris. N Engl J Med 323: 514– 520, 1990 [DOI] [PubMed] [Google Scholar]
- 36. Rapps JA, Sturek M, Jones AW, Parker JL. Altered reactivity of coronary arteries located distal to a chronic coronary occlusion. Am J Physiol Heart Circ Physiol 273: H1879– H1887, 1997 [DOI] [PubMed] [Google Scholar]
- 37. Roth DM, Maruoka Y, Rogers J, White FC, Longhurst JC, Bloor CM. Development of coronary collateral circulation in left circumflex Ameroid-occluded swine myocardium. Am J Physiol Heart Circ Physiol 253: H1279– H1288, 1987 [DOI] [PubMed] [Google Scholar]
- 38. Roth DM, White FC, Nichols ML, Dobbs SL, Longhurst JC, Bloor CM. Effect of long-term exercise on regional myocardial function and coronary collateral development after gradual coronary artery occlusion in pigs. Circulation 82: 1778– 1789, 1990 [DOI] [PubMed] [Google Scholar]
- 39. Sakurada S, Takuwa N, Sugimoto N, Wang Y, Seto M, Sasaki Y, Takuwa Y. Ca2+-dependent activation of Rho and Rho kinase in membrane depolarization-induced and receptor stimulation-induced vascular smooth muscle contraction. Circ Res 93: 548– 556, 2003 [DOI] [PubMed] [Google Scholar]
- 40. Sambuceti G, Marzilli M, Marraccini P, Schneider-Eicke J, Gliozheni E, Parodi O, L'Abbate A. Coronary vasoconstriction during myocardial ischemia induced by rises in metabolic demand in patients with coronary artery disease. Circulation 95: 2652– 2659, 1997 [DOI] [PubMed] [Google Scholar]
- 41. Schiffrin EL. Endothelin and endothelin antagonists in hypertension. J Hypertens 16: 1891– 1895, 1998 [DOI] [PubMed] [Google Scholar]
- 42. Schuler G, Hambrecht R, Schlierf G, Niebauer J, Hauer K, Neumann J, Hoberg E, Drinkmann A, Bacher F, Grunze M. Regular physical exercise and low-fat diet. Effects on progression of coronary artery disease. Circulation 86: 1– 11, 1992 [DOI] [PubMed] [Google Scholar]
- 43. Shimokawa H, Seto M, Katsumata N, Amano M, Kozai T, Yamawaki T, Kuwata K, Kandabashi T, Egashira K, Ikegaki I, Asano T, Kaibuchi K, Takeshita A. Rho-kinase-mediated pathway induces enhanced myosin light chain phosphorylations in a swine model of coronary artery spasm. Cardiovasc Res 43: 1029– 1039, 1999 [DOI] [PubMed] [Google Scholar]
- 44. Somlyo AP, Somlyo AV. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev 83: 1325– 1358, 2003 [DOI] [PubMed] [Google Scholar]
- 45. Takamura M, Parent R, Cernacek P, Lavallée M. Influence of dual ETA/ETB-receptor blockade on coronary responses to treadmill exercise in dogs. J Appl Physiol 89: 2041– 2048, 2000 [DOI] [PubMed] [Google Scholar]
- 46. Traverse JH, Judd D, Bache RJ. Dose-dependent effect of endothelin-1 on blood flow to normal and collateral-dependent myocardium. Circulation 93: 558– 566, 1996 [DOI] [PubMed] [Google Scholar]
- 47. Traverse JH, Kinn JW, Klassen C, Duncker DJ, Bache RJ. Nitric oxide inhibition impairs blood flow during exercise in hearts with a collateral-dependent myocardial region. J Am Coll Cardiol 31: 67– 74, 1998 [DOI] [PubMed] [Google Scholar]
- 48. White FC, Carroll SM, Magnet A, Bloor CM. Coronary collateral development in swine after coronary artery occlusion. Circ Res 71: 1490– 1500, 1992 [DOI] [PubMed] [Google Scholar]
- 49. White FC, Roth DM, Bloor CM. The pig as a model for myocardial ischemia and exercise. Lab Anim Sci 36: 351– 356, 1986 [PubMed] [Google Scholar]
- 50. Woodman CR, Muller JM, Rush JW, Laughlin MH, Price EM. Flow regulation of ecNOS and Cu/Zn SOD mRNA expression in porcine coronary arterioles. Am J Physiol Heart Circ Physiol 276: H1058– H1063, 1999 [DOI] [PubMed] [Google Scholar]
- 51. Woodman CR, Price EM, Laughlin MH. Shear stress induces eNOS mRNA expression and improves endothelium-dependent dilation in senescent soleus muscle feed arteries. J Appl Physiol 98: 940– 946, 2005 [DOI] [PubMed] [Google Scholar]
- 52. Zhou M, Widmer RJ, Xie W, Widmer AJ, Miller MW, Schroeder F, Parker JL, Heaps CL. Effects of exercise training on cellular mechanisms of endothelial nitric oxide synthase regulation in coronary arteries after chronic occlusion. Am J Physiol Heart Circ Physiol 298: H1857– H1869, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]