Abstract
The precursor of the neurotrophin NGF (proNGF) serves physiological functions distinct from its mature counterpart as it induces neuronal apoptosis through activation of a p75 neurotrophin receptor (p75NTR) and Sortilin death-signalling complex. The neurotrophins BDNF and NT3 provide essential trophic support to auditory neurons. Injury to the neurotrophin secreting cells in the inner ear is followed by irreversible degeneration of spiral ganglion neurons with consequences such as impaired hearing or deafness. Lack of mature neurotrophins may explain the degeneration of spiral ganglion neurons, but another mechanism is possible since unprocessed proNTs released from the injured cells may contribute to the degeneration by induction of apoptosis. Recent studies demonstrate that proBDNF, like proNGF, is a potent inducer of Sortilin:p75NTR mediated apoptosis. In addition, a coincident upregulation of proBDNF and p75NTR has been observed in degenerating spiral ganglion neurons, but the Sortilin expression in the inner ear is unresolved. Here we demonstrate that Sortilin and p75NTR are coexpressed in neurons of the neonatal inner ear. Furthermore, we establish that proNT3 exhibits high affinity binding to Sortilin and has the capacity to enhance cell surface Sortilin:p75NTR complex formation as well as to mediate apoptosis in neurons coexpressing p75NTR and Sortilin. Based on examination of wt and Sortilin deficient mouse embryos, Sortilin does not significantly influence the developmental selection of spiral ganglion neurons. However, our results suggest that proNT3 and proBDNF may play important roles in the response to noise-induced injuries or ototoxic damage via the Sortilin:p75NTR death-signalling complex.
Keywords: Vps10p-domain, neurotrophins, p75NTR, apoptosis
INTRODUCTION
The four neurotrophins (NTs) nerve growth factor-β (NGF), brain-derived nerve growth factor (BDNF), and neurotrophin-3 (NT3) and −4/5 (NT4/5), constitute a family of closely related homodimeric proteins that are essential for the regulation of neuronal growth, differentiation and death (Chao, 2003). Each NT is synthesised as a ~30 kDa precursor molecule (proNT) which is converted to the mature NT by enzymatic cleavage and removal of a ~15 kDa N-terminal prodomain. Both the precursor and the mature forms are secreted (Lee et al., 2001) but at a ratio that may vary with age and environmental circumstances such as conditions that inflict damage to the secreting cell (Harrington et al., 2004). Survival is first and foremost induced via mature NTs and their binding to the three ligand specific tyrosine receptor kinases (TrkA-C) (Chao, 2003; Huang & Reichardt, 2003). In contrast, the precursor forms of NGF and BDNF have been shown to initiate neuronal apoptosis by forming a death-signalling trimeric complex with the multifunctional type-1 receptor Sortilin and the common NT receptor p75NTR (Nykjaer et al., 2004; Teng et al., 2005). Coexpression of Sortilin and p75NTR is essential to this mechanism and neurons deficient in one or both receptors are resistant to proNT mediated induction of death (Jansen et al., 2007). The NTs are strongly expressed in the inner ear during late embryonic life and support afferent innervation and neuronal fiber guidance to target epithelia cells (Pirvola et al., 1992; Ylikoski et al., 1993; Fritzsch et al., 2004). The support is predominantly provided by BDNF and NT3. BDNF is imperative for survival of vestibular sensory neurons whereas the spiral ganglion (SG) neurons depend on NT3 for support (Ernfors et al., 1995). However, BDNF and NT3 are also expressed in the inner ear of adults (Tan & Shepherd, 2006; Sugawara et al., 2007), and auditory neurons of both adults and embryos degenerate in response to loss of their central (cochlear nucleus) or peripheral (organ of Corti) targets (Webster & Webster, 1981). One explanation for this degeneration could be the lack of the life sustaining mature NTs. However, apoptosis resulting from an acute increase in the release of unprocessed proNTs from injured or dying targets cells is another interesting but unexplored possibility. For instance, damage to the inner ear can result in an increased expression of NT-precursors and it is conceivable that release of proNT3 from injured cells of the organ of Corti may contribute to post-traumatic SG-neuronal apoptosis and post-traumatic loss of hearing. The present study was undertaken to determine if the inner ear meets the requirements for such a scenario. We have examined the expression and coexpression of p75NTR and Sortilin, and the ability of proNT3 to bind Sortilin and to promote formation of a death-signalling complex by interacting simultaneously with Sortilin and p75NTR.
MATERIALS AND METHODS
DNA constructs and recombinant proteins
Wildtype Sortilin (Petersen et al., 1997), proSortilin (Munck Petersen et al., 1999) and endocytosis impaired Sortilin (Y14→A, L17→A, L51→A and L52→A, Sortilin-mut) were expressed as described (Nielsen et al., 2001). Rat p75NTR was expressed using the pcDNA3.1/hyg vector (Invitrogen). Bacterial expression of the GST-NT3pro and GST-NT4/5pro have previously been described (Westergaard et al., 2005). In internalisation studies, Surface Plasmon Resonance analysis, and crosslinking experiments were used furin-resistant “uncleavable” recombinant human proNGF and proNT3 expressed in E. coli BL21(DE3) (Novagen). Apoptosis assays were conducted with furin-resistant recombinant proneurotrophins expressed in baculovirus (donated by Kenneth T. Teng). Human recombinant NT3 (G1501, Promega) and neurotensin (N6383, Sigma-Aldrich) were purchased.
Surface Plasmon Resonance (SPR) analysis
Binding of mature NT3, proNT3, and NT3pro to immobilised Sortilin was detected by SPR using a Biacore 3000 instrument (GE Healthcare Europe GmbH, Uppsala, Sweden) as described elsewhere (Munck Petersen et al., 1999). The receptor-ligand interactions were recorded as the difference in response between the immobilised receptor flow cell and a corresponding control flow cell (activated and blocked, but without immobilised receptor).
Kinetic parameters were determined by BIAevaluation 4.1 software using a Langmuir 1:1 binding model and simultaneous fitting of curves in the concentration range.
Transfected cell lines and primary cell cultures
HEK293 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (Bio-Whittaker) supplemented with 10% fetal calf serum (FCS) (Invitrogen). Parental 293 cells and transfectants expressing p75NTR were transfected with Sortilin, proSortilin or Sortilin-mut (endocytosis impaired Sortilin) using FuGENE® transfection reagents (Roche), and stably transfected clones were selected at zeocin 500 μg/ml (Invitrogen).
Internalisation and IF-labelling of internalised ligands
Transfected or wildtype HEK293 cells were incubated with 50nM proNT3 for 2 hours at 4oC before internalisation at 37oC for 40 minutes. The cells were fixed in 4% paraformaldehyde (PFA) and permeabilised with 0.5% saponin in BSS before incubation with anti-NT3 (ab10334, Abcam), anti-NGF (G113, Promega) or anti-GST (27-4577-01, GE Healthcare). Secondary antibodies used were Alexa 488 donkey anti-goat, goat anti-mouse or goat anti-rabbit (Molecular Probes). IF-labelled ligands were visualised by confocal microscopy.
Crosslinking and immunoprecipitation
Crosslinking and immunoprecipitation of Sortilin:p75NTR receptor complexes were done in transfected and wildtype HEK293. Cells were incubated with or without 50nM proNT3 for 1 hour at RT prior to addition of 5mM DSP (Dithiobis[succinimidyl-proprionate], PIERCE). The reaction was stopped by addition of 1M Tris (pH 7.5) and DSP treated cells were then washed and lysed in 1% Triton X-100 supplemented with protease inhibitors (CompleteMini, Roche). For each sample the concentration of protein was determined using a Bicinchoninic Acid Kit (Sigma Aldrich), and equal amounts of total protein were subjected to immunoprecipitation of receptor-ligand complexes using γ-bind G-Sepharose beads (Amersham Pharmacia) coated with p75NTR antibody (ab10494, Abcam). Precipitated proteins and input lysates were analysed by reducing SDS-PAGE and western blotting using monoclonal anti-Sortilin (anti-Neurotensin R3 mAb, Transduction Laboratories), anti-proNT3 (Alomone Labs Ltd.), and anti-p75NTR.
Tissue preparation for immunoblotting
All procedures were approved by the Danish Animal Experiments Inspectorate. Eighteen postnatal day 3–5 (P3–5) Wistar Hannover rats (Harlan Denmark) were sacrificed as previously described (Van de Water & Ruben, 1971; Brors et al., 2003).
The brains were isolated from the bisected crania and stored at −20°C. The temporal bones were harvested and the bony cochlea was gently removed, exposing the membranous labyrinth. The membranous cochlea was isolated and subdivided into 1) the stria vascularis (SV) and lateral ligament, 2) the organ of Corti with Reissner’s membrane (RM) attached, 3) the spiral ganglion (SG), and 4) the modiolus (without the SG). The cochlear tissue fractions from 36 temporal bones were pooled according to their specific origin, centrifuged and the pellets were stored at −20°C.
Immunoblotting
Inner ear tissue fractions were homogenised in 1.2% Triton X-100, 20mM Tris HCl, 10mM EDTA, pH 8.0 for 10 minutes at 4°C. Debris was pelleted and supernatants were subjected to reducing SDS-PAGE (4–16%) and western blotting (~2–5 μg protein/lane). The blots were blocked in 5% defatted milk in TBST buffer (50mM Tris, 500mM NaCl, 0.1% Tween-20, pH 7.4) for 1 hour at RT and subsequently incubated over night at 4°C in blocking buffer with mouse anti-human Sortilin, rabbit anti-human p75NTR (provided by Moses V. Chao), rabbit anti-proNT3, rabbit anti-NT3 (Alomone Labs Ltd.) or mouse anti-beta-actin (Sigma Aldrich). Following wash in 0.5% defatted milk in TBST buffer, blots were incubated with HRP-conjugated anti-rabbit IgG or HRP-conjugated anti-mouse IgG (DAKOR). Detection was performed by enhanced chemiluminescence.
Immunohistochemistry (IHCh) and Immunofluorescence (IF) light microscopy
Five neonatal (P4) Wistar Hannover rats (Harlan Denmark) were anesthetised and in vivo perfused with 2% PFA through the heart. Sections were cut (2μm) from paraffin embedded samples followed by blocking of endogenous peroxidase (0.3% H2O2 in methanol, 30 minutes) and target retrieval using TEG buffer (1.2 g Tris (Sigma), 0.19 g EGTA (Titriplex VI, Merck), distilled water to 1000ml, pH 9.0) and microwave treatment. Sections were incubated with polyclonal anti-Sortilin (1:1400(Munck Petersen et al., 1999)1:700 (BAF2934, R&D), polyclonal anti-p75NTR (1:1500 (donated by Moses Chao), 1:50 (BAF1157, R&D)), polyclonal anti-myosinVIIa (1:500 (ab3481, Abcam), and monoclonal anti-class III β-tubulin (Tuj1) (1:700, MMS-435P, Covance) as indicated. A Sortilin antibody specificity control was performed with Sortilin antibodies preincubated with soluble Sortilin antigen (130μg/ml) (Munck Petersen et al., 1999).
For IHCh, staining was done using HRP-conjugated secondary antibodies (1:200) and Meyers Hematoxylin (Bie & Berntsen) and antibody detection was performed with DiAminoBenzidine (Kem-En-Tec Diagnostics A/S). For IF, Alexa 488 or 568 goat anti-rabbit, 546 donkey anti-rabbit, and 488 goat anti-mouse (1:350, all Molecular Probes) were used as secondary antibodies. All antibodies were diluted in 0.1% BSA and 0.3% Triton X-100 in PBS. Microscopy was performed using a Leica DM R conventional light microscope (IHCh) and a Carl Zeiss LSM 510 Meta confocal microscope (IF).
Dissociated superior cervical ganglion (SCG) tissue cultures
Primary growth medium (PGM)
DMEM containing 10% FCS, 1mM sodium pyruvate, 2mM L-glutamine (all Invitrogen), 100U/ml penicillin, 100μg/ml streptomycin, 20nM floxuridine, 20nM uridine (all Sigma-Aldrich), and 50ng/ml NGF (cat#GF-022-5, Austral Biologicals).
Secondary growth medium(SGM)
PGM without antimetabolites and only 10ng/ml NGF. Twelve to fifteen newborn or postnatal day 1–2 (P0–P2) C57/BL6 mice (Harlan Denmark) were used for each experiment. The animals were cryo-anesthetised, decapitated, and the SCGs were isolated using a Leica MZ125 stereo dissection microscope. The SCGs were pooled, split, and dissociated in HBSS (Bie & Berntsen) containing 10μg/ml DNAse (Sigma-Aldrich). Preheated 0.25% trypsin (Sigma-Aldrich) in Dulbecco’s phosphate-buffered saline (DPBS) without Ca2+ and Mg2+ (Invitrogen) was added and the SCGs were incubated shortly at 37°C. The DNAse/trypsin mixture was inactivated by adding PGM, centrifuged and the pellet was resuspended in PGM. The solution was triturated thoroughly and left intact for a few minutes for non-neural tissue and debris to sediment. The supernatant was transferred to a new tube, further triturated, spun down again, and resuspended in PGM. Cell dissociation was checked with a phase contrast microscope and the cell density was determined using a hemocytometer. A cell suspension volume corresponding to a cell density of 50,000 cells/well was added to each well. Additional PGM was added after adhesion of cells to the coated surface. The growth medium was changed to SGM after 48 hours of incubation.
Dissociated spiral ganglion (SG) tissue cultures
Ten to twelve newborn or postnatal day 3 (P3) C57/BL6 mice (Taconic, Denmark) were used for each experiment. The SGs were cultivated in growth media identical to the SCG media except that no streptomycin (ototoxic) was added to the media. Furthermore, NGF was substituted for 8ng/ml NT3, 8ng/ml BDNF and 100ng/ml leukemia inhibitory factor (LIF). The SGs were isolated from newborn mice as previously described (Van de Water & Ruben, 1971; Brors et al., 2003) and cultivated as described above for the SCGs with the exception that cultures were preplated 2 hours in a non-coated plastic cell suspension dish in an attempt to decrease the proportion of non-neuronal cells and increase the percentage of neurons.
Induction of apoptosis in SCG cultures
Dissociated SCG neurons obtained from fifteen C57/BL6 mice (P0–P1) were plated on coated cover slips and maintained in culture for 5 days before use. Replicate cultures were rinsed 6 × 30 minutes in NGF-free medium to remove all NGF from the SCGs. Subsequently, the cultures were incubated in a modified medium with or without the given additives, as indicated. After 36 hours, SCG cultures were fixed in 4% PFA, washed in DPBS and subsequently in wash buffer (0.1% Triton X-100 in DPBS), blocked in incubation buffer (wash buffer with 10% FCS), and incubated with anti-Tuj1 1:700 (MMS-435p, Covance) in incubation buffer ON at 4°C.
The specimens were then washed again and incubated in secondary antibody (Alexa Fluor 488 goat-anti mouse 1:350 (Molecular Probes). Finally, specimens were washed in DPBS and ddH2O, and mounted in a medium containing DAPI (Vector laboratories Inc). SCG neurons were subjected to apoptosis analysis by morphological evaluation using a Leica DM LB microscope with fluorescence optics. Results represent the mean value of one experiment performed in triplicate. The observer scored apoptotic neurons, defined as Tuj1 positive cells with fragmented nucleus, blindly and at least 800 cells per condition were counted. Statistical significance was determined by student’s t-test.
Induction of apoptosis in SG cultures and counting of apoptotic neurons
SG neurons were rinsed in NT-free medium 6 × 30 minutes and incubated for 36 hours in a growth medium with or without proNT3, as indicated. The neurons were cultivated, fixed, stained, mounted and analysed as described for the SCG cultures, except that results represent a single experiment. The standard derivation bars refer to the differences in apoptosis-ratios between the individual slides (9–12) counted for each condition (~1500 neurons were counted per additive).
Whole mouse heads (E15.5–E16.5) were fixed over-night in phosphate-buffered 4% formaldehyde dehydrated and embedded in paraffin. Five μm thick sections were cut sagittally and every 20th section was sampled. Apoptotic cells were identified with the active caspase-3 technique using rabbit anti-caspase-3 polyclonal antibodies (1:400, Cell Signalling Tech.), affinity purified biotinylated goat anti-rabbit IgG (1:200, DakoCytomation) and DAB (Sigma-Aldrich). All apoptotic cell profiles inside the spiral ganglia were counted.
RESULTS
Sortilin binds proNT3 with high affinity
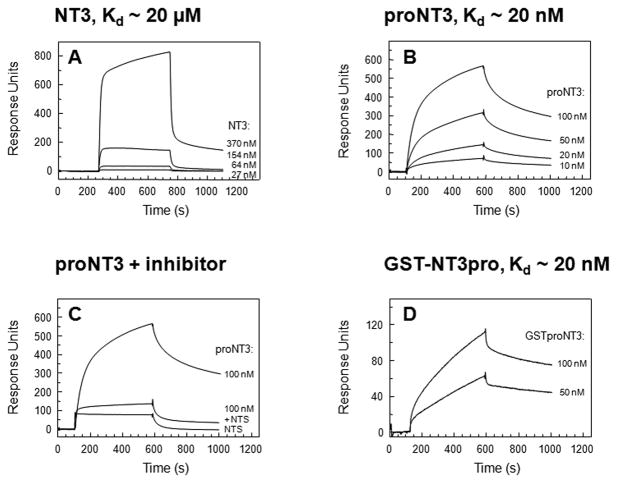
The binding of NT3 and proNT3 to Sortilin was examined by Surface Plasmon Resonance analysis. We found that mature NT3 bound to Sortilin with low affinity (Kd ~ 20μM) (fig. 1A). In contrast, proNT3 exhibited strong binding (Kd ~ 20nM) (fig. 1B) and this interaction could be inhibited by neurotensin, which is a well-known Sortilin ligand (fig. 1C). Furthermore, a similar affinity was found for the interaction between Sortilin and a fusion protein consisting of the prodomain of proNT3 and glutathione-S-transferase (GST-NT3pro) (fig. 1D). By comparison, a fusion protein comprising GST and the prodomain of proNT4/5 (GST-NT4/5pro) bound to Sortilin with an affinity of Kd ~ 1μM (not shown). Thus, Sortilin binds proNT3 through a high affinity interaction with the prodomain sequence of proNT3, as previously demonstrated for the binding of proNGF to Sortilin (Nykjaer et al., 2004).
Figure 1. SPR analysis of ligand binding to immobilised Sortilin.
SPR analysis of receptor-ligand interactions between the NT3 and immobilised Sortilin demonstrated that mature NT3 binds to Sortilin with a Kd ~20 μM (A). This affinity is significantly lower than the affinity found for the interaction between proNT3 and Sortilin (Kd ~20 nM) (B), and for the interaction between the receptor and the prodomain sequence of proNT3 fused to GST (Kd~20 nM) (D). The interaction between Sortilin and proNT3 can be inhibited by the well-known Sortilin ligand neurotensin (C).
NT3 neurotrophin-3, proNT3 proform of NT3, GST-NT3pro prodomain of NT3 coupled to glutathione-s-transferase NTS neurotensin, Kd dissociation constant, SPR Surface plasmon resonance
Receptor mediated internalisation of proNT3 is a Sortilin dependent process
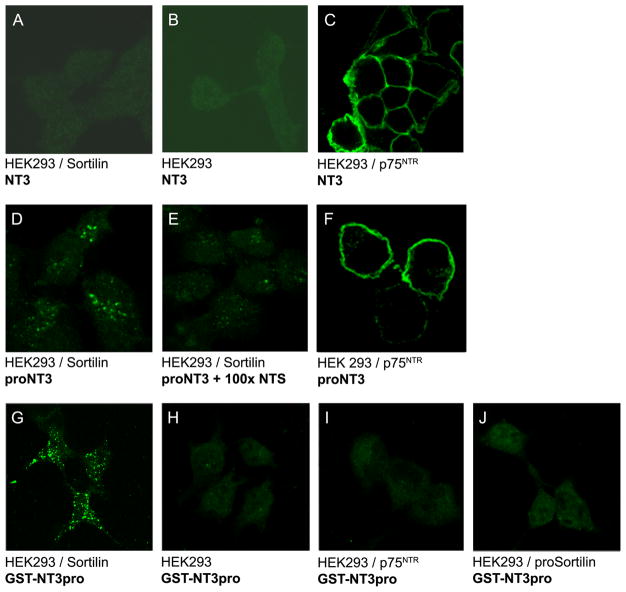
The binding of NT3 and proNT3 to cellular Sortilin and subsequent Sortilin mediated internalisation of these ligands were analysed in HEK293 cells transfected with Sortilin, an uncleavable proform of Sortilin (proSortilin) and/or p75NTR. Cells were incubated with 50nM of ligand at 4°C for 2 hours prior to 40 minutes of internalisation at 37°C. Ligand-binding capacity and uptake was detected by immunofluorescence microscopy of labelled ligands. No binding or internalisation of mature NT3 was detected in cells transfected with Sortilin (fig. 2A) or in untransfected control cells (fig. 2B). Cells expressing p75NTR exhibited a strong NT3 surface labelling, but no endocytosis was detected (fig. 2C). In addition, p75NTR transfectants displayed an intense surface staining after incubation with proNT3 (fig. 2F), but no staining upon incubation with GST-NT3pro fusion protein (fig. 2I), confirming that the interaction between p75NTR and proNT3 depends on the sequence of the mature NT3. In contrast, cells expressing Sortilin demonstrated an effective uptake of both proNT3 (fig. 2D) and GST-NT3pro (fig. 2G) illustrated by accumulating intracellular vesicles and a lack of cell surface staining after 40 minutes of incubation. Cells expressing both Sortilin and p75NTR displayed an augmented uptake of proNT3 combined with a less intense cell surface labelling (not shown). The internalisation of proNT3 was efficiently inhibited by neurotensin (fig. 2E). No binding or uptake of GST-NT3pro was detected in cells transfected with proSortilin (fig. 2J) or in untransfected cells (fig. 2H). Immunostaining of cells incubated with GST-NT3pro at 4°C showed dispersed cell surface labelling compared to the vesicular labelling seen after internalisation at 37°C (supplementary fig. 1). Furthermore, no binding or internalisation of GST-NT4/5pro was seen in Sortilin transfected cells (not shown). Taken together, these observations confirm that receptor mediated endocytosis of proNT3 is a Sortilin dependent process and that proNT3 binding to Sortilin relies on the prodomain sequence of proNT3, thus consolidating the SPR analysis data.
Figure 2. Sortilin-mediated binding and internalisation of NT3 and proNT3.
(A–J) HEK293 cells transfected with Sortilin, p75NTR or proSortilin as indicated were incubated with 50nM of the given ligand (NT3 (A–C), proNT3 (D–F), or GST-NT3pro (G–J)) for 40 minutes at 37° C. Internalised ligand was visualised by immunofluorescence labelling with specific antibodies. Mature NT3 and proNT3 were detected with anti-NT3, whereas GST-NT3pro was detected with an anti-GST antibody. Cells transfected with Sortilin showed no binding and uptake of NT3 (A), but internalisation was detected of both proNT3 (D) and GST-NT3pro (G). The uptake of proNT3 could be inhibited by addition of NTS (E). Cells transfected with p75NTR displayed strong surface labelling after incubation with NT3 (C) and proNT3 (F), but no binding of GST-NT3pro (I).
HEK293 human embryonic kidney 293 cells, NT3 neurotrophin-3, proNT3 proform of NT3, p75NTR p75 neurotrophin receptor, NTS neurotensin, GST-NT3pro prodomain of NT3 coupled to glutathione-s-transferase.
ProNT3 enhance the formation of cell surface Sortilin:p75NTR receptor complexes
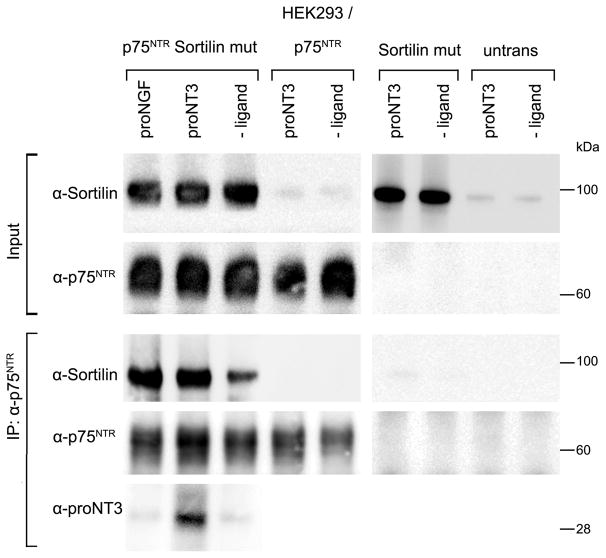
The ability of proNT3 to induce formation of cell surface receptor complexes between Sortilin and p75NTR was examined using crosslinking and precipitation of transfected cells. For these experiments, HEK293 cells were transfected with p75NTR and/or an endocytosis-impaired variant of Sortilin (Sortilin-mut). The Sortilin-mut variant was selected in order to increase the number of Sortilin receptors expressed on the cell surface. Transfectants were incubated 1 hour at RT in the presence or absence of 50nM proNT3 or proNGF (positive control) before crosslinking of receptor:ligand complexes with a reducible membrane-permeable crosslinker, and followed by immunoprecipitation using anti-p75NTR. The precipitates were subjected to reducing SDS-PAGE and co-precipitation of Sortilin and proNT3 were detected by immunoblotting (fig. 3). Sortilin was detected in precipitate from p75NTR/Sortilin-mut cells. A band was detected even in the absence of proNT ligand, indicating some spontaneous interaction between the two receptors. However, the intensity of the Sortilin band is markedly increased in the presence of either proNGF or proNT3, and proNT3 was detected in precipitate from cells incubated with this ligand (fig. 3). No Sortilin precipitation was detected in p75NTR single transfectants, Sortilin-mut single transfectants and untransfected HEK cells (fig. 3). The expression of Sortilin and p75NTR in the input cell lysates was confirmed by immunoblotting (fig. 3). These results demonstrate that both proNT3 and proNGF efficiently upregulates the formation of cell surface Sortilin:p75NTR receptor complexes.
Figure 3. Crosslinking and precipitation of p75NTR:Sortilin:proNT3 complexes.
HEK293 cells transfected with an endocytosis-impaired variant of Sortilin, Sortilin-mut, and p75NTR as indicated were incubated with or without 50 nM ligand (proNT3 or proNGF) before treatment with crosslinker. Receptor:ligand complexes were immunoprecipitated with anti-p75NTR, and components were visualised after reducing SDS-PAGE by immunoblotting with anti-Sortilin, anti-proNT3, and anti-p75NTR. The expression of Sortilin and p75NTR in input cell lysates were confirmed by immunoblotting.
HEK293 human embryonic kidney 293 cells, p75NTR p75 neurotrophin receptor, Sortilin-mut endocytosis-impaired variant of Sortilin, proNGF proform of nerve growth factor, proNT3 proform of neurotrophin-3, NT3 mature form of neurotrophin-3
Sortilin and p75NTR are coexpressed in spiral ganglion neurons
Immunoblotting of subfractions of inner ear tissue
The expression of Sortilin, p75NTR and proNT3 in the inner ear was investigated by immunoblotting using isolated subfractions of dissected inner ear tissue. Figure 4 shows the immunoblots for Sortilin, p75NTR, proNT3 and mature NT3 of pooled tissue homogenates from whole-organ membranous cochleae as well as specific subfractions of membranous cochleae from newborn rats. Sortilin was detected in whole-organ cochleae (Co) in addition to all the subfractions i.e. the stria vascularis (I), the organ of Corti with Reissner’s membrane attached (II), the modiolus (not including the SG) (III), and the SG (IV) (fig. 4A). Even though the expression levels appear fairly uniform in all subfractions, the Sortilin expression level in the organ of Corti with Reissner’s membrane is relatively higher, because of the low total protein concentration in this subfraction, as seen in the corresponding blot for beta-actin (fig. 4A). Expression of p75NTR was detected in the modiolus (III) and the SG (IV) (fig. 4B). Thus, immunoblotting of inner ear tissue revealed that both receptors are expressed in the spiral ganglion. Furthermore proNT3 and mature NT3 were detected in all subfractions (fig. 4C). The specificity of the proNT3 and NT3 antibodies was tested by immunoblotting using recombinant human NT3 and furinresistant proNT3 and proNGF (fig. 4D). The NT3 antibody detects both the mature and the proform of NT3, while anti-proNT3 only recognises the proform.
Figure 4. Immunoblotting of inner ear tissue for Sortilin, p75NTR, proNT3 and mature NT3.
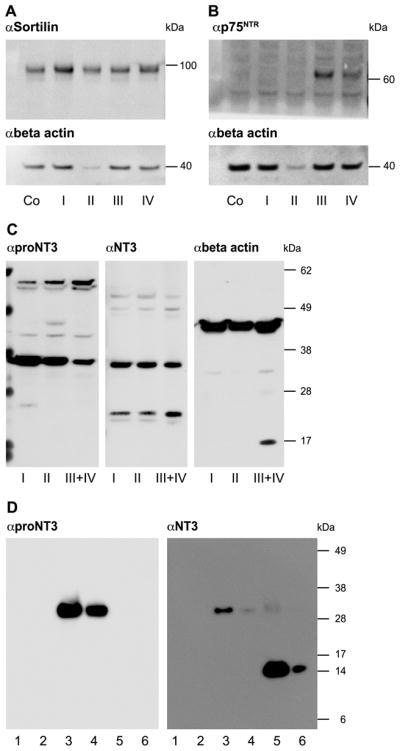
Subfractions of membranous labyrinth from dissected newborn rats (P3–P5) were pooled, homogenised and centrifuged. The supernatants were subjected to SDS-PAGE followed by immunoblotting for Sortilin, p75NTR, proNT3 and mature NT3. Beta-actin (~42 kDa) blots are included for evaluation of total protein concentration. (A) Sortilin (~98 kDa) was detected in whole-organ cochleae (Co) and in all in the subfractions i.e. the stria vascularis (I), the organ of Corti with Reissner’s membrane attached (II), the modiolus (not including the spiral ganglion) (III), and the spiral ganglion (IV). (B) P75NTR (~64 kDa) was detected in the modiolus (III) and the SG (IV). (C) ProNT3 and NT3 were detected in all subfractions. (D) The specificity of proNT3 and NT3 antibodies was tested by immunoblotting of recombinant human proNGF (lane 1 (5 ng) and 2 (1 ng)), proNT3 (lane 3 (5 ng) and 4 (1 ng)), and NT3 (lane 5 (5 ng) and 6 (1 ng)). The proNT3 antibody recognises only proNT3, while the NT3 antibody detects both the proform and the mature form of NT3.
kDa kilodalton, Co whole-organ membranous cochlea, p75NTR p75 neurotrophin receptor, proNT3 proform of neurotrophin-3, NT3 mature form of neurotrophin-3, αSortilin anti-Sortilin, αp75NTR anti- p75NTR, αproNT3 anti-proNT3, αNT3 anti-NT3, αbeta-actin anti-beta actin, P3–P5 postnatal day 3–5
Sortilin and p75NTR detection by IHCh and IF
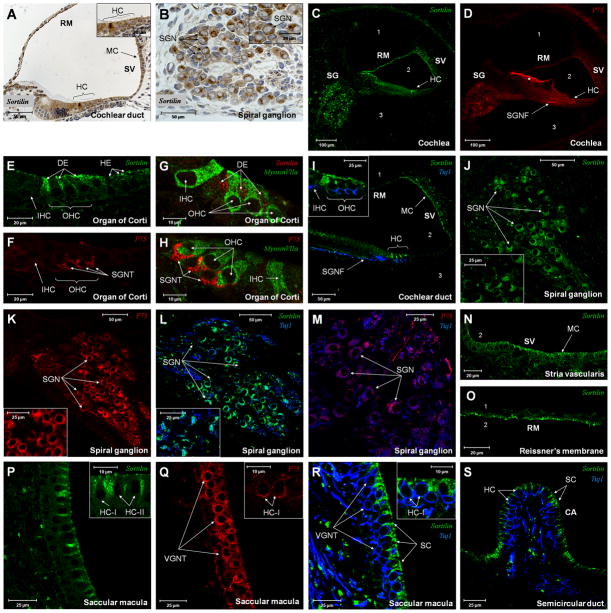
In the cochlea, immunohistochemical labelling for Sortilin showed expression corresponding to various epithelial cells lining the cochlear duct as well as to spiral ganglion neurons (fig. 5A-C). In the organ of Corti, a strong positive response was detected in the apical processes of Deiters cells, whereas a weaker response was seen in Hensen cells (fig. 5E). No labelling was found in sensory hair cells as also demonstrated by double labelling using the hair cell marker myosin VIIa (fig. 5G). In the spiral ganglion, distinct perinuclear labelling was detected in the auditory neurons (fig. 5J). Marginal cells of the stria vascularis were labelled, whereas intermediate cells and basal cells were not (fig. 5N). Finally, a positive response was seen in cells of Reissner’s membrane facing the cochlear duct, whereas cells facing the scala vestibuli were unlabelled (fig. 5O). The specificity of the anti-Sortilin labelling was confirmed by stainings using antibodies preincubated with the Sortilin antigen (supplementary fig. 2G–H).
Figure 5. Immunolabelling of Sortilin and p75NTR in the inner ear of neonatal rats.
(A–D) Expression of Sortilin (A–C) and p75NTR (D) in the cochlea. (E–H) High magnification of the organ of Corti showing Sortilin (E) and p75NTR (F) expression, as well as double labelling of these receptors with the hair cell marker myosin VIIa (G+H). Note the absence of Sortilin in hair cells and the co-localisation of p75NTR with the neuronal marker (Tuj1) in nerve terminals (E–I).
(J–M) Coexpression of Sortilin (J+L) and p75NTR (K+M) in SGN. (N–O) IF detection of Sortilin in marginal cells of the stria vascularis (N) and epithelial cells of Reissner’s membrane (O) facing the cochlear duct. (P–S) Sortilin (P+R+S) and p75NTR (Q) expression in the saccule and semicircular ducts of the vestibular apparatus. Note the differential distribution of Sortilin and p75NTR in the sensory epithelium of the vestibular apparatus in analogy to the hair cell region of the cochlea. RM Reissner’s membrane, SV stria vascularis, MC strial marginal cells, HC hair cells, SGN spiral ganglion neurons, SG spiral ganglion, SGNF spiral ganglion nerve fibers, *) staining artifact, DE Deiters cells, HE Hensen cells, IHC inner hair cells, OHC outer hair cells, SGNT spiral ganglion nerve terminals, Tuj1class III β-tubulin, HC-I type-1 hair cells HC-II type-2 hair cells, VGNT vestibular ganglion nerve terminals, SC supporting cells, CA crista ampullaris.
P75NTR was detected exclusively in auditory neurons (fig. 5D), but in contrast to Sortilin, p75NTR labelling was also detected in nerve fibers and nerve terminals corresponding to the outer spiral bundle (fig. 5D+F+H). The identity of the p75NTR labelled structures was confirmed by double labelling with anti-myosin VIIa and an antibody against the neuronal marker Tuj1 (supplementary fig. 2A–D). The p75NTR labelling pattern found in SGN cell bodies was generally less polarised compared to that of Sortilin (fig. 5J+K). Expression of Sortilin and p75NTR in SGN was confirmed using anti-Tuj1 (fig. 5I+L+M). In the vestibular apparatus, positive labelling for Sortilin was detected in vestibular ganglion neurons (not shown) and in supporting cells corresponding to the maculae of the otolith organs (saccule and utricle) as well as the cristae ampullaris of the semicircular ducts (fig. 5P+R–S). In common with p75NTR expression in the cochlea, p75NTR was only observed in neuronal cell bodies, nerve fibers and nerve terminals, of the vestibular apparatus (fig. 5Q).
ProNT3 induces apoptosis in dissociated SCG neuron cultures
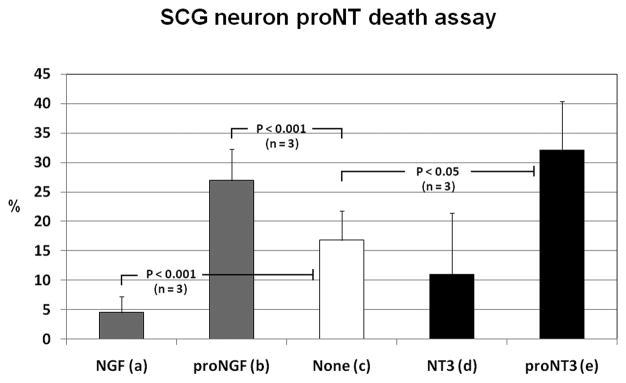
The ability of proNT3 to induce apoptosis in neurons coexpressing p75NTR and Sortilin was tested in SCG cultures, which represent a neuronal model system of Sortilin- and p75NTR-expressing cells. To minimise conversion of proNT into mature NT, which might introduce a bias by facilitating survival in TrkA- and TrkC-positive neurons, furin-resistant mutants of proNGF and proNT3 were used in the death assay. The results (Fig. 6) demonstrate that NGF withdrawal from SCG cultures increases the death rate from 5% to 17% (p < 0.001) (Fig. 6a+c). Conversely, addition of proNGF (p<0.001) or proNT3 (p<0.05) significantly enhances apoptosis in SCG neurons compared to the effect observed for NGF withdrawal alone (Fig. 6b,c,e). In contrast to mature neurotrophins, of which only NGF affected neuronal survival, proNGF and proNT3 exhibited an equal potential to induce apoptosis in SCG neurons.
Figure 6. ProNT3 induced apoptosis in SCG neurons.
The apoptotic potential of proNT3 was tested in SCG neurons. Here is shown the number of apoptotic neurons in percent of the total number counted. SCG neurons were incubated in growth media containing NGF (20ng/ml), NT3 (2ng/ml), proNGF (2ng/ml) or proNT3 (4ng/ml). After 36 hours, SCG cultures were fixed, immunostained with α-Tuj1, α-Sortilin and DAPI and subjected to apoptosis analysis by morphological evaluation. The results demonstrate that NGF (as opposed to NT3) is imperative for SCG neuron survival. In contrast, both proNGF and proNT3 significantly increased the proportion of apoptotic neurons compared to NGF withdrawal alone.
SCG superior cervical ganglion, proNT proneurotrophins, NGF nerve growth factor, proNGF proform of NGF, None no (pro)neurotrophins added, NT3 neurotrophin-3, proNT3 proform of NT3.
ProNT3 enhances apoptosis induced by NT withdrawal in SG neuron cultures
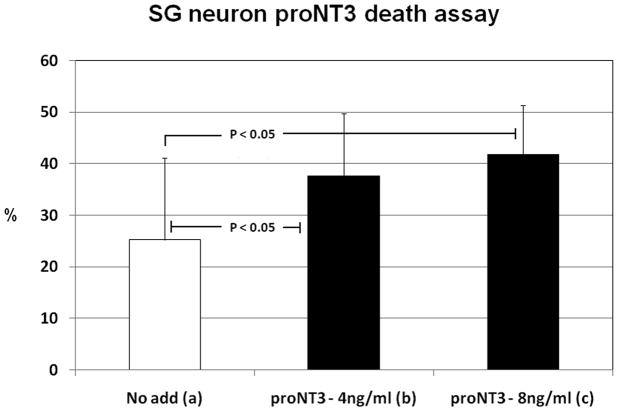
The apoptosis-inducing effect of proNT3 on spiral ganglion neurons was tested in dissociated SG cultures and compared to the effect of NT withdrawal alone (Fig. 7) – which, in a separate experiment, did not differ between wt and Sortilin deficient SGNs (not shown). However, it has been extremely difficult to establish SGN cultures of acceptable purity and with sufficient number of neurons. In one out of several attempts we succeeded and a culture of approximately five thousands neurons was established from 16 animals. The results indicate that proNT3 (p<0.05) significantly enhances apoptosis in SG neurons compared to NT3, BDNF and LIF withdrawal alone (Fig. 7a+b). Furthermore, the death rate seems to increase with increasing concentrations of proNT3 from 4ng/ml (Fig. 7b) to 8ng/ml (Fig. 7c).
Figure 7. ProNT3 induced apoptosis in SG neurons.
ProNT3-induced apoptosis was also investigated in p75NTR- and Sortilin-coexpressing SG neurons. Cultured SGs were rinsed in NT-free medium prior to incubation in modified media containing proNT3 (4ng/ml), proNT3 (8ng/ml) or no NTs. After 36 hours, SG cultures were fixed, stained with DAPI and a neuronal marker, and subjected to apoptosis analysis by morphological evaluation. Here is shown the number of apoptotic cells in percent of total number of neurons counted. The results demonstrate that proNT3 significantly enhanced apoptosis in SG neurons compared to the effect of NT withdrawal alone. The data represents a single experiment and bars indicate standard deviation between apoptosis-ratios on the individual slides (9–12) counted for each condition (~1500 neurons per additive).
SG spiral ganglion, NT neurotrophins, proNT3 proform of neurotrophin-3, No add no neurotrophins added.
Finally, we determined the number of apoptotic SGNs in developing wt and Sortilin−/− mouse embryos (E15.5 – E16.5). As determined by a caspase-3 assay, counting was similar and showed no significant difference between the two species (Suppl. Table 1). Similar data were obtained using a TUNEL-assay (not shown). Thus, Sortilin and proNT induced apoptosis, but does not appear to play any major role in the selection of developing SG neurons.
DISCUSSION
Neurotrophins and their receptors are principal elements of the machinery that sustains the function of mature neuronal cells and governs the survival and death of neurons during development and in response to injury. In the inner ear, neurotrophic support to afferent sensory neurons is provided exclusively by BDNF and NT3 secreted by the sensory epithelial cells and loss of these target cells leads to degeneration of the auditory neurons. One reason for this could simply be deprivation of mature NTs but another possibility is a coincident release of NT-precursors and formation of the previously described Sortilin:p75NTR death-signalling complex. The latter mechanism seems particularly relevant because neuronal death resulting from toxic (aminoglycoside) injury to hair cells was recently demonstrated to be accompanied by an increase in proBDNF as well as an enhanced expression of p75NTR in spiral ganglion neurons (Tan & Shepherd, 2006). However, the expression pattern of Sortilin in the inner ear (notably in SGNs) has up till now been unexamined and it has not been determined whether proNT3, similar to proNGF and proBDNF, can crosslink Sortilin and p75NTR and induce apoptosis.
The NT3 precursor targets the Sortilin β-propeller domain and crosslinks Sortilin and p75NTR
The present data establish that proNT3 binds Sortilin in vitro and in vivo. Binding of mature NT3 is comparatively modest and the high affinity of the precursor is mediated by the prodomain. This is similar to previous observations regarding proNGF (Nykjaer et al., 2004) and proBDNF (Teng et al., 2005). Yet binding to Sortilin cannot be considered a common feature of all NTs since neither NT4/5 nor its prodomain exhibit any significant binding to the receptor. The interactions between Sortilin and proNGF, proBDNF, and proNT3, are all inhibited by the Sortilin-propeptide and by neurotensin. Notably the tripeptide YIL, that constitutes the receptor-binding C-terminus of neurotensin, is a much less effective inhibitor of proNGF binding to Sortilin than the full length peptide (Quistgaard et al., 2009). We recently demonstrated that the ligand-interacting domain of Sortilin is a unique 10-bladed β-propeller with a central tunnel harbouring the binding site for neurotensin (Quistgaard et al., 2009). The present data therefore suggest that proNT3, like proNGF and proBDNF, binds inside or in close proximity to the tunnel but via a separate site than neurotensin. In any case, proNT3 is bound in a manner that mediates its internalisation by Sortilin and allows it to facilitate the previously described interaction between Sortilin and p75NTR in cells that coexpress the two receptors.
The ability of proNT3 to induce apoptosis via the Sortilin:p75NTR complex was assessed in SCGs which express both receptors. We have previously demonstrated that proNGF induces death in SCGs and our present results show that the effect of proNT3 is comparable to that of proNGF. In contrast, death resulting from withdrawal of NGF was not counterbalanced equally well by the mature NTs, which agrees with the observation that SCGs beyond E15.5 express the NGF receptor TrkA but not the NT3 receptor TrkC (Fagan et al., 1996). Similar affect of proNT3 on SCG wt neurons is reported by H. Yano et al., but p75NTR knock out SCG neurons are not susceptible to proNT3 induced cell death (Yano et al., 2009).
Release of proNT3 in the inner ear may result in secondary degeneration of SG neurons
A death-signalling complex consisting of the components proNT3, Sortilin and p75NTR could have a physiological relevance in the inner ear, where NT3 plays a pivotal role in the trophic support of the auditory neurons, as illustrated by the fact that newborn NT3 null mice display a loss of 85% of spiral ganglion neurons (Ernfors et al., 1995). NT3 is expressed in hair cells and supporting cells of the cochlea and hearing impairment and deafness are the results of irreversible loss of these cells followed by a secondary degeneration of the spiral ganglion neurons. In agreement with the functional significance of NT3 and previous expression-analysis of the mature neurotrophin (Sugawara et al., 2007) we detected both mature NT3 and proNT3 the inner ear. Formation of a Sortilin:p75NTR death-signalling complex requires coexpression of the two receptors in the relevant cells of the inner ear. Our data show that Sortilin is expressed corresponding to various epithelial cells lining the cochlear duct as well as to cells supporting the sensory hair cells of the cochlea and the vestibular apparatus. More importantly, we demonstrate that Sortilin coexpresses with p75NTR in spiral ganglion neurons (SGN). In addition to SGN, transient p75NTR expression has previously been described in other cells of the cochlea (Pirvola et al., 1994; Gestwa et al., 1999; Sato et al., 2006). Gestwa et al. studied the expression of p75NTR in the neonatal rat cochlea and detected p75NTR in pillar cells (P0-P4/5), inner sulcus cells (P6–P9) and Schwann cells (P10) (Gestwa et al., 1999), whereas Sato et al. detected p75NTR in pillar cells (P3), cochlear micro vessels (P3) and in inner sulcus cells (P5+) of neonatal mice (Sato et al., 2006). These observations are essentially in agreement with our results, considering the age of the animals used (P5), except that we did not detect p75NTR in pillar cells of neonatal rats. Spiral ganglion neurons coexpress p75NTR and Sortilin and represent an authentic model for examination of the apoptotic-inducing effect of proNT3. Unfortunately, logistic problems only allowed limited experiments with SGN. However our findings in SG neurons support the results seen in SCG cultures and indicate that proNT3 can induce apoptosis. This apoptosis-inducing property has previously been attributed to proNGF (Nykjaer et al., 2004) and to proBDNF (Teng et al., 2005) but until now only hypothesised for proNT3. Thus, the present results establish that proNT3 in common with proNGF and proBDNF has the capacity to activate the death-signalling receptor complex in neurons coexpressing p75NTR and Sortilin.
Recent studies have established that proneurotrophins, including proNGF and proBDNF, are released (Yang et al., 2009), in particular under certain conditions such as trauma (Harrington et al., 2004), and mediate neuronal apoptosis through activation of a Sortilin:p75NTR death-signalling complex. In addition, ototoxic degeneration of spiral ganglion neurons was recently demonstrated to prompt augmented p75NTR expression as well as an upregulation of proBDNF (Tan & Shepherd, 2006). We demonstrate that Sortilin and p75NTR are coexpressed in SGN of the neonatal inner ear. Furthermore, we provide evidence that proNT3 exhibits high affinity binding to Sortilin and has the functional capacity not only to induce cell surface Sortilin:p75NTR receptor complex formation, but also to effectively mediate apoptosis in p75NTR- and Sortilin coexpressing neurons. These results indicate that although Sortilin may have little influence on the selection of SG neurons during development, it may (in concert with proNT3 and proBDNF) play an important role in the response to either noise-induced or ototoxic degeneration of spiral ganglion neurons. In this context, the coexpression of Sortilin with p75NTR in SGN is of fundamental importance because Sortilin acts as a molecular switch governing the p75NTR-mediated apoptotic signals induced by proneurotrophins, and because both receptors are required components for transmission of proneurotrophin-dependent death signals (Jansen et al., 2007). In contrast, the putative physiological functions of Sortilin expressed in epithelial cells lining the cochlear duct as well as in cells supporting cochlear and vestibular sensory hair cells remain undetermined.
Further studies will elucidate whether noise-induced or ototoxic degeneration of spiral ganglion neurons is accompanied by an upregulation of Sortilin expression in parallel to increased p75NTR expression. Studies on the Sortilin knock out mouse have very recently shown that Sortilin-deficiency protects injured corticospinal neurons from apoptosis (Jansen et al., 2007). Future studies will demonstrate whether the Sortilin knock out mouse is also protected against noise induced neuronal degeneration.
Supplementary Material
(A–C) HEK293 cells transfected as indicated were immunostained with an anti-GST antibody after incubation with 50nM of GST-NT3pro for 2 hours at 4° C. Sortilin transfectants display dispersed surface labelling after incubation at 4° C (C) compared to vesicular labelling seen in cells incubated at 4° C followed by incubation at 37° C for 40 min (fig. 2G).
HEK293, human embryonic kidney 293 cells; Sortilin-mut, endocytosis-impaired (surface localised) variant of Sortilin; GST-NT3pro, prodomain of NT3 coupled to glutathione-s-transferase
(A–D) Double immunolabelling of hair cells and nerve terminals in the organ of Corti (A+C) and in the saccular macula (B+D) using a specific hair cell marker (anti-myosin VIIa), a nerve specific marker (anti-Tuj1 class III β-tubulin) and anti-p75NTR. Note the identical staining of anti-p75NTR labelled and nerve specific anti-Tuj1 labelled presynaptic nerve terminals in the organ of Corti as well as the organization of hair cells and their innervating afferent neurons in the saccular macula. (E–H) Anti-Sortilin labelling of spiral ganglion neurons and strial marginal cells in the cochlea. Sortilin antibody specificity was demonstrated by pre-incubation with soluble Sortilin (130 μg/ml) prior to incubation with the primary Sortilin antibody (5.6 μg/ml). Note the lack of immunolabelling in pre-incubated tissue samples (G+H) compared to the positive control (E+F), where labelling was not preceded by pre-incubation with soluble Sortilin.
OHC outer hair cells, IHC inner hair cells, SGNT spiral ganglion nerve terminals, VGNT vestibular ganglion nerve terminals, HC-I type-1 hair cells, SGN spiral ganglion neurons, MC strial marginal cells.
Acknowledgments
We appreciate Anne Marie Bundsgaard and Inger B. Kristoffersen for excellent technical assistance and gratefully thank Morten S. Nielsen for encouraging discussions and advices regarding confocal microscopy and Christian Jacobsen for assistance with SPR Analysis. We also thank Moses V. Chao for donating the p75NTR antibody. The present study was financially supported by the Karen Elise Jensen Foundation, the Skouby Foundation, Emanuel Jensen’s scientific research grant, Oda Pedersen’s research grant, and NIH grant number NS057627 (Kenneth K. Teng). MIND is a Lundbeck Foundation funded research centre.
ABBREVIATIONS
- BDNF
brain-derived nerve growth factor
- GST
gluthathione-S-transferase
- NGF
nerve growth factor
- NT
neurotrophin
- p75NTR
p75 neurotrophin receptor
- SCG
superior cervical ganglion
- SG
spiral ganglion
- SGN
spiral ganglion neurons
References
- Brors D, Bodmer D, Pak K, Aletsee C, Schafers M, Dazert S, Ryan AF. EphA4 provides repulsive signals to developing cochlear ganglion neurites mediated through ephrin-B2 and -B3. J Comp Neurol. 2003;462:90–100. doi: 10.1002/cne.10707. [DOI] [PubMed] [Google Scholar]
- Chao MV. Neurotrophins and their receptors: a convergence point for many signalling pathways. Nat Rev Neurosci. 2003;4:299–309. doi: 10.1038/nrn1078. [DOI] [PubMed] [Google Scholar]
- Ernfors P, Van De Water T, Loring J, Jaenisch R. Complementary roles of BDNF and NT-3 in vestibular and auditory development. Neuron. 1995;14:1153–1164. doi: 10.1016/0896-6273(95)90263-5. [DOI] [PubMed] [Google Scholar]
- Fagan AM, Zhang H, Landis S, Smeyne RJ, Silos-Santiago I, Barbacid M. TrkA, but not TrkC, receptors are essential for survival of sympathetic neurons in vivo. J Neurosci. 1996;16:6208–6218. doi: 10.1523/JNEUROSCI.16-19-06208.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritzsch B, Tessarollo L, Coppola E, Reichardt LF. Neurotrophins in the ear: their roles in sensory neuron survival and fiber guidance. Prog Brain Res. 2004;146:265–278. doi: 10.1016/S0079-6123(03)46017-2. [DOI] [PubMed] [Google Scholar]
- Gestwa G, Wiechers B, Zimmermann U, Praetorius M, Rohbock K, Kopschall I, Zenner HP, Knipper M. Differential expression of trkB.T1 and trkB.T2, truncated trkC, and p75(NGFR) in the cochlea prior to hearing function. J Comp Neurol. 1999;414:33–49. doi: 10.1002/(sici)1096-9861(19991108)414:1<33::aid-cne3>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- Harrington AW, Leiner B, Blechschmitt C, Arevalo JC, Lee R, Morl K, Meyer M, Hempstead BL, Yoon SO, Giehl KM. Secreted proNGF is a pathophysiological death-inducing ligand after adult CNS injury. Proc Natl Acad Sci U S A. 2004;101:6226–6230. doi: 10.1073/pnas.0305755101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang EJ, Reichardt LF. Trk receptors: roles in neuronal signal transduction. Annu Rev Biochem. 2003;72:609–642. doi: 10.1146/annurev.biochem.72.121801.161629. [DOI] [PubMed] [Google Scholar]
- Jansen P, Giehl K, Nyengaard JR, Teng K, Lioubinski O, Sjoegaard SS, Breiderhoff T, Gotthardt M, Lin F, Eilers A, Petersen CM, Lewin GR, Hempstead BL, Willnow TE, Nykjaer A. Roles for the pro-neurotrophin receptor sortilin in neuronal development, aging and brain injury. Nat Neurosci. 2007;10:1449–1457. doi: 10.1038/nn2000. [DOI] [PubMed] [Google Scholar]
- Lee R, Kermani P, Teng KK, Hempstead BL. Regulation of cell survival by secreted proneurotrophins. Science. 2001;294:1945–1948. doi: 10.1126/science.1065057. [DOI] [PubMed] [Google Scholar]
- Munck Petersen C, Nielsen MS, Jacobsen C, Tauris J, Jacobsen L, Gliemann J, Moestrup SK, Madsen P. Propeptide cleavage conditions sortilin/neurotensin receptor-3 for ligand binding. EMBO J. 1999;18:595–604. doi: 10.1093/emboj/18.3.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen MS, Madsen P, Christensen EI, Nykjaer A, Gliemann J, Kasper D, Pohlmann R, Petersen CM. The sortilin cytoplasmic tail conveys Golgi-endosome transport and binds the VHS domain of the GGA2 sorting protein. EMBO J. 2001;20:2180–2190. doi: 10.1093/emboj/20.9.2180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nykjaer A, Lee R, Teng KK, Jansen P, Madsen P, Nielsen MS, Jacobsen C, Kliemannel M, Schwarz E, Willnow TE, Hempstead BL, Petersen CM. Sortilin is essential for proNGF-induced neuronal cell death. Nature. 2004;427:843–848. doi: 10.1038/nature02319. [DOI] [PubMed] [Google Scholar]
- Petersen CM, Nielsen MS, Nykjaer A, Jacobsen L, Tommerup N, Rasmussen HH, Roigaard H, Gliemann J, Madsen P, Moestrup SK. Molecular identification of a novel candidate sorting receptor purified from human brain by receptor-associated protein affinity chromatography. J Biol Chem. 1997;272:3599–3605. doi: 10.1074/jbc.272.6.3599. [DOI] [PubMed] [Google Scholar]
- Pirvola U, Arumae U, Moshnyakov M, Palgi J, Saarma M, Ylikoski J. Coordinated expression and function of neurotrophins and their receptors in the rat inner ear during target innervation. Hear Res. 1994;75:131–144. doi: 10.1016/0378-5955(94)90064-7. [DOI] [PubMed] [Google Scholar]
- Pirvola U, Ylikoski J, Palgi J, Lehtonen E, Arumae U, Saarma M. Brain-derived neurotrophic factor and neurotrophin 3 mRNAs in the peripheral target fields of developing inner ear ganglia. Proc Natl Acad Sci U S A. 1992;89:9915–9919. doi: 10.1073/pnas.89.20.9915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quistgaard EM, Madsen P, Groftehauge MK, Nissen P, Petersen CM, Thirup SS. Ligands bind to Sortilin in the tunnel of a ten-bladed beta-propeller domain. Nat Struct Mol Biol. 2009;16:96–98. doi: 10.1038/nsmb.1543. [DOI] [PubMed] [Google Scholar]
- Sato T, Doi K, Taniguchi M, Yamashita T, Kubo T, Tohyama M. Progressive hearing loss in mice carrying a mutation in the p75 gene. Brain Res. 2006;1091:224–234. doi: 10.1016/j.brainres.2005.12.104. [DOI] [PubMed] [Google Scholar]
- Sugawara M, Murtie JC, Stankovic KM, Liberman MC, Corfas G. Dynamic patterns of neurotrophin 3 expression in the postnatal mouse inner ear. J Comp Neurol. 2007;501:30–37. doi: 10.1002/cne.21227. [DOI] [PubMed] [Google Scholar]
- Tan J, Shepherd RK. Aminoglycoside-induced degeneration of adult spiral ganglion neurons involves differential modulation of tyrosine kinase B and p75 neurotrophin receptor signaling. Am J Pathol. 2006;169:528–543. doi: 10.2353/ajpath.2006.060122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teng HK, Teng KK, Lee R, Wright S, Tevar S, Almeida RD, Kermani P, Torkin R, Chen ZY, Lee FS, Kraemer RT, Nykjaer A, Hempstead BL. ProBDNF induces neuronal apoptosis via activation of a receptor complex of p75NTR and sortilin. J Neurosci. 2005;25:5455–5463. doi: 10.1523/JNEUROSCI.5123-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van de Water TR, Ruben RJ. Organ culture of the mammalian inner ear. Acta Otolaryngol. 1971;71:303–312. doi: 10.3109/00016487109125368. [DOI] [PubMed] [Google Scholar]
- Webster M, Webster DB. Spiral ganglion neuron loss following organ of Corti loss: a quantitative study. Brain Res. 1981;212:17–30. doi: 10.1016/0006-8993(81)90028-7. [DOI] [PubMed] [Google Scholar]
- Westergaard UB, Kirkegaard K, Sorensen ES, Jacobsen C, Nielsen MS, Petersen CM, Madsen P. SorCS3 does not require propeptide cleavage to bind nerve growth factor. FEBS Lett. 2005;579:1172–1176. doi: 10.1016/j.febslet.2004.12.088. [DOI] [PubMed] [Google Scholar]
- Yang J, Siao CJ, Nagappan G, Marinic T, Jing D, McGrath K, Chen ZY, Mark W, Tessarollo L, Lee FS, Lu B, Hempstead BL. Neuronal release of proBDNF. Nat Neurosci. 2009 doi: 10.1038/nn.2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano H, Torkin R, Martin LA, Chao MV, Teng KK. Proneurotrophin-3 Is a Neuronal Apoptotic Ligand: Evidence for Retrograde-Directed Cell Killing. J Neurosci. 2009;29:14790–14802. doi: 10.1523/JNEUROSCI.2059-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ylikoski J, Pirvola U, Moshnyakov M, Palgi J, Arumae U, Saarma M. Expression patterns of neurotrophin and their receptor mRNAs in the rat inner ear. Hear Res. 1993;65:69–78. doi: 10.1016/0378-5955(93)90202-c. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A–C) HEK293 cells transfected as indicated were immunostained with an anti-GST antibody after incubation with 50nM of GST-NT3pro for 2 hours at 4° C. Sortilin transfectants display dispersed surface labelling after incubation at 4° C (C) compared to vesicular labelling seen in cells incubated at 4° C followed by incubation at 37° C for 40 min (fig. 2G).
HEK293, human embryonic kidney 293 cells; Sortilin-mut, endocytosis-impaired (surface localised) variant of Sortilin; GST-NT3pro, prodomain of NT3 coupled to glutathione-s-transferase
(A–D) Double immunolabelling of hair cells and nerve terminals in the organ of Corti (A+C) and in the saccular macula (B+D) using a specific hair cell marker (anti-myosin VIIa), a nerve specific marker (anti-Tuj1 class III β-tubulin) and anti-p75NTR. Note the identical staining of anti-p75NTR labelled and nerve specific anti-Tuj1 labelled presynaptic nerve terminals in the organ of Corti as well as the organization of hair cells and their innervating afferent neurons in the saccular macula. (E–H) Anti-Sortilin labelling of spiral ganglion neurons and strial marginal cells in the cochlea. Sortilin antibody specificity was demonstrated by pre-incubation with soluble Sortilin (130 μg/ml) prior to incubation with the primary Sortilin antibody (5.6 μg/ml). Note the lack of immunolabelling in pre-incubated tissue samples (G+H) compared to the positive control (E+F), where labelling was not preceded by pre-incubation with soluble Sortilin.
OHC outer hair cells, IHC inner hair cells, SGNT spiral ganglion nerve terminals, VGNT vestibular ganglion nerve terminals, HC-I type-1 hair cells, SGN spiral ganglion neurons, MC strial marginal cells.