Abstract
Background
Fibroblast growth factor 23 (FGF23), uncarboxylated matrix Gla protein (ucMGP), and fetuin-A are regulators of mineral metabolism and inhibitors of vascular calcification. Whether circulating levels of each are associated with cardiovascular disease (CVD) events or mortality in populations without end-stage renal disease is unknown.
Objective
To evaluate the associations of FGF23, ucMGP, and fetuin-A with mortality and CVD events.
Design
Observational study.
Setting
12 outpatient clinics in the San Francisco Bay area.
Patients
833 outpatients with stable coronary artery disease (CAD), recruited from 11 September 2000 to 20 December 2002.
Measurements
Fibroblast growth factor 23, ucMGP, and fetuin-A concentrations were measured at baseline. Participants were followed until 1 December 2008 for mortality and CVD events.
Results
During a median follow-up of 6.0 years, 220 participants died and 182 had CVD events. Compared with participants with FGF-23 levels in the lowest tertile, those in the highest tertile had 2-fold greater risk for mortality (hazard ratio [HR], 2.15 [95% CI, 1.43 to 3.24]) and CVD events (HR, 1.83 [CI, 1.15 to 2.91]) after adjustment for traditional CVD risk factors, C-reactive protein levels, and kidney function. The highest ucMGP tertile was associated with lower mortality risk (HR, 0.48 [CI, 0.31 to 0.75]) and showed a nonsignificant trend toward lower CVD event risk by tertile analysis (HR, 0.65 [CI, 0.40 to 1.05])—an association that was significant when modeled continuously (P = 0.029). No significant association of fetuin-A with mortality (HR, 0.84 [CI, 0.55 to 1.27]) or CVD events (HR, 0.99 [CI, 0.64 to 1.55]) was observed.
Limitation
Participants had prevalent CAD.
Conclusion
In outpatients with stable CAD, higher FGF23 and lower ucMGP levels are independently associated with mortality and CVD events.
Primary Funding Source
American Heart Association.
People with cardiovascular disease (CVD) are at high risk for recurrent CVD events and mortality (1), and many traditional CVD risk factors are not strongly associated with CVD events in this setting (2, 3). New insights into mechanisms for recurrent events might ultimately provide novel therapeutic targets or prevention strategies.
Abnormalities in mineral metabolism are associated with CVD risk in the general population. For example, higher phosphorus levels promote arterial calcification in vitro (4, 5) and are associated with coronary artery calcification and CVD events in the general population, even among persons with normal kidney function (6–9). Similarly, lower serum vitamin D levels and higher serum intact parathyroid hormone levels are associated with CVD events in the general population (10, 11). Recently, several novel regulators of arterial calcification, including fibroblast growth factor 23 (FGF23), uncarboxylated matrix Gla protein (ucMGP), and fetuin-A, have been identified. Yet, the relationship of circulating levels of these proteins with CVD events and mortality is unknown.
Fibroblast growth factor 23 is a bone-derived protein that promotes renal phosphorus wasting and inhibits conversion of 25-hydroxyvitamin D to the active 1,25-dihydroxyvitamin D form (12–16). Studies of mice have suggested that elevated FGF23 levels may reflect a response to dietary phosphorus burden and serve as a marker of arterial calcification (17). Matrix Gla protein (MGP) is produced by chondrocytes and vascular smooth-muscle cells and prevents arterial calcification through local effects in the arterial wall (18–21). Its precursor form is ucMGP, which has affinity for hydroxyapatite and may be depleted from blood in the setting of arterial calcification. Therefore, low serum levels may be a marker of vascular disease (22). Fetuin-A is a liver-secreted protein that complexes with calcium and phosphorus in circulation and prevents the precipitation of these minerals from serum (23, 24). In mice, knockouts of genes encoding FGF23, ucMGP, or fetuin-A result in arterial or soft-tissue calcification or both (24–26). In humans, FGF23 and fetuin-A have been most extensively studied in cohorts with end-stage renal disease (ESRD), where higher FGF23 and lower fetuin-A levels are associated with mortality and CVD events (27–30). Although the temporal association of ucMGP with mortality is uncertain, patients with ESRD or atherosclerosis have lower ucMGP levels than healthy controls (31–34). Because these 3 proteins have complementary biological functions, we measured blood levels of all 3 and evaluated associations with mortality and CVD events in an ambulatory cohort with prevalent coronary artery disease (CAD) and a spectrum of kidney function from normal to moderate chronic kidney disease. On the basis of directions of association observed in cohorts with ESRD, we hypothesized that higher FGF23, lower ucMGP, and lower fetuin-A levels would each be associated with higher CVD events and mortality, independent of traditional CVD risk factors, kidney function, and one another.
Methods
Study Participants
The Heart and Soul Study is an observational study designed to investigate the influence of psychosocial factors on the progression of CVD. Methods are described elsewhere (35). In brief, participants were recruited from out-patient clinics in the San Francisco Bay area if they met at least one of the following inclusion criteria: history of myocardial infarction (MI); angiographic evidence of greater than 50% stenosis in 1 or more coronary vessels; evidence of exercise-induced ischemia by treadmill or nuclear testing; and history of coronary revascularization or documented diagnosis of CAD by an internist or cardiologist. Participants were excluded if they could not walk 1 block, had MI within the past 6 months, or were likely to move out of the area within 3 years. The study protocol was approved by the institutional review boards of participating institutions. All participants provided written informed consent. From September 2000 to December 2002, a total of 1024 participants enrolled and had a baseline study appointment.
Measurements
Predictor Variables
Fasting (12-hour) serum and plasma samples were obtained at the baseline visit, frozen at −70 °C, and later used for measurement of FGF23 (in 2007), ucMGP (in 2007), and fetuin-A (in 2004). Blood levels of each were measured twice in each participant, and results were averaged (the Appendix, available at www.annals.org, includes measurement details).
Outcome Variables
From the baseline examination to 1 December 2008, we conducted annual telephone interviews with participants (or their proxies), inquiring about hospitalizations, cardiac procedures, or death. Two independent, blinded adjudicators reviewed all events, medical records, electrocardiography results, death certificates, and coroner’s reports. In the event of disagreement, the adjudicators conferred, reconsidered their classification, and requested consultation from a third blinded adjudicator, as necessary.
We considered the composite of MI, stroke, transient ischemic attack, or heart failure as a CVD event. Myocardial infarction was defined by cardiac biomarkers, electrocardiography results, and cardiac symptoms or signs according to the American Heart Association criteria (36). We defined “stroke” as a new neurologic deficit not secondary to brain trauma, tumor, infection, or other cause. Transient ischemic attack was defined as a focal neurologic deficit (in the absence of head trauma) lasting more than 30 seconds and no longer than 24 hours, with rapid symptom evolution to the maximal level of deficit in less than 5 minutes and with subsequent complete resolution. We defined “heart failure” as hospitalization for a clinical syndrome involving at least 2 of the following changes from usual status: paroxysmal nocturnal dyspnea, orthopnea, elevated jugular venous pressure, pulmonary rales, third heart sound, and cardiomegaly or pulmonary edema on chest radiography. Mortality was determined by death certificates and coroner’s reports.
Other Measurements
We determined age, sex, race or ethnicity, and smoking status by questionnaire. We instructed participants to bring their medication bottles to the baseline appointment, and study personnel recorded medications. Waist-to-hip ratio was calculated after measuring waist circumference midway between the lower rib margin and iliac crest and hip circumference at the level of the greater trochanters. We determined prevalent hypertension and diabetes by questionnaire. Systolic and diastolic blood pressures were measured after 5 minutes of rest in the supine position. We calculated estimated glomerular filtration rate (GFR) by incorporating creatinine concentrations, age, sex, and race in the abbreviated (4-variable) Modification of Diet in Renal Disease Study equation (37). High-sensitivity C-reactive protein levels were measured with the Roche assay (Indianapolis, Indiana) and the Beckman Extended Range assay (Galway, Ireland) (38). We measured total cholesterol, high-density lipoprotein cholesterol, calcium, and phosphorus levels by using standard clinical chemistry analyzers. Participants had transthoracic echocardiography at baseline, which we used to measure left ventricular ejection fraction. We did a symptom-limited, graded exercise treadmill test according to the standard Bruce protocol and defined peak exercise capacity as total number of metabolic equivalent tasks achieved.
Statistical Analysis
We excluded 5 participants (<1%) who were missing outcome data and 186 participants (18%) who did not have baseline blood specimens for measurement of fetuin-A, ucMGP, or FGF23, resulting in a final sample size of 833 participants. Compared with those of excluded participants, demographic characteristics and event rates were similar among participants who were retained in this analysis (Appendix Table 1, available at www.annals.org). For descriptive statistics, we compared demographic and clinical variables among participants with and without CVD events or mortality during follow-up by using the t test or the Kruskal–Wallis test for continuous variables and the chi-square test or Fisher exact test for categorical variables, as appropriate. We used graphical methods to determine the distribution of FGF23, ucMGP, and fetuin-A in the study sample. Linear regression was used to compare associations of FGF23, ucMGP, and fetuin-A with calcium, phosphorus, and other baseline cardiovascular risks.
We elected to evaluate tertiles of each protein as our primary predictor variables. The lowest tertile served as the reference category. Because the functional form was fairly linear for each, we also evaluated each protein on a continuous scale, to minimize the chance of a type II error. We used Kaplan–Meier curves to evaluate unadjusted time to events and Cox proportional hazards models to evaluate adjusted associations of each novel mineral regulator with CVD events and mortality. Three sequential models were developed: one adjusted for age, sex, and race; one adjusted for age, sex, race and other CVD risk factors (smoking, hypertension, diabetes, waist-to-hip ratio, systolic and diastolic blood pressures, total and high-density lipoprotein cholesterol levels, estimated GFR, C-reactive protein level, ejection fraction, peak exercise capacity, and medications used [Table 1]); and one adjusted for the CVD risk factors plus serum calcium, phosphorus, and the other 2 novel mineral regulators, because we considered it possible that the association of each novel mineral regulator might be mediated through calcium, phosphorus, or the other 2 novel mineral regulators. We assessed proportional hazards assumptions by visually inspecting log-minus-log plots and plots of Schoenfeld residuals versus survival time. No evidence of violation was observed. We used multiplicative interaction terms to evaluate effect modification by diabetes status and for the presence or absence of chronic kidney disease (estimated GFR <60 mL/min per 1.73 m2 vs. ≥60 mL/min per 1.73 m2). Analyses were done using Stata Statistical Software, version 9.2 (StataCorp, College Station, Texas).
Table 1.
Baseline Characteristics of Participants With and Without Events*
| Characteristic | Participants With Events (n = 301) | Participants Without Events (n = 532) | P Value |
|---|---|---|---|
| Mean age (SD), y | 71 (12) | 65 (10) | <0.001 |
| Men, n (%) | 261 (87) | 416 (78) | 0.002 |
| Race, n (%) | 0.080 | ||
| White | 195 (65) | 304 (57) | |
| Black | 44 (15) | 91 (17) | |
| Other | 61 (20) | 137 (26) | |
| Medication use, n (%) | |||
| Aspirin | 231 (77) | 413 (78) | 0.77 |
| β-Blocker | 177 (59) | 301 (57) | 0.53 |
| ACE inhibitor or ARB | 176 (58) | 254 (48) | 0.003 |
| Statin | 187 (62) | 350 (66) | 0.29 |
| Anticoagulant | 30 (10) | 32 (6) | 0.037 |
| Current smoker, n (%) | 66 (22) | 102 (19) | 0.36 |
| Diabetes, n (%) | 100 (33) | 121 (23) | 0.001 |
| Hypertension, n (%) | 225 (75) | 375 (71) | 0.176 |
| Mean systolic BP (SD), mm Hg | 135 (22) | 133 (20) | 0.24 |
| Mean diastolic BP (SD), mm Hg | 74 (12) | 75 (11) | 0.027 |
| Mean waist-to-hip ratio (SD) | 0.97 (0.08) | 0.95 (0.08) | 0.008 |
| Mean peak exercise capacity (SD), METS | 6 (3) | 8 (3) | <0.001 |
| Mean ejection fraction (SD) | 0.59 (0.11) | 0.63 (0.09) | <0.001 |
| Mean estimated GFR (SD), mL/min per 1.73 m2 | 70 (25) | 80 (21) | <0.001 |
| Median total cholesterol level (IQR) | |||
| mmol/L | 4.33 (3.70–5.20) | 4.48 (3.89–5.05) | |
| mg/dL | 167 (143–201) | 173 (150–195) | 0.172 |
| Mean HDL cholesterol level (SD) | |||
| mmol/L | 1.17 (0.36) | 1.22 (0.36) | |
| mg/dL | 45 (14) | 47 (14) | 0.052 |
| Median CRP level (IQR), nmol/L | 25 (12–63) | 19 (8–40) | <0.001 |
| Mean calcium level (SD)† | |||
| mmol/L | 2.4 (0.13) | 2.4 (0.13) | |
| mg/dL | 9.6 (0.5) | 9.6 (0.5) | 0.96 |
| Median phosphorus level (IQR) | |||
| mmol/L | 1.16 (1.07–1.29) | 1.20 (1.07–1.29) | |
| mg/dL | 3.6 (3.3–4.0) | 3.7 (3.3–4.0) | 0.53 |
ACE = angiotensin-converting enzyme; ARB = angiotensin-receptor blocker; BP = blood pressure; CRP = C-reactive protein; GFR = glomerular filtration rate (based on the 4-variable Modification of Diet in Renal Disease Study equation); HDL = high-density lipoprotein; IQR = interquartile range; METS = metabolic equivalent tasks.
Cardiovascular event or death.
Adjusted calcium = total calcium + (4.0 − serum albumin)× 0.8.
Role of the Funding Source
The Heart and Soul Study was supported by the Department of Veterans Epidemiology Merit Review Program; Department of Veterans Affairs Health Services Research and Development service; National Heart, Lung, and Blood Institute; American Federation for Aging Research; Robert Wood Johnson Foundation; and Ischemia Research and Education Foundation. This ancillary study was supported by the American Heart Association and the National Heart, Lung, and Blood Institute. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, approval, or decision to submit the manuscript for review.
Results
Among the 833 participants, the mean age was 67 years (SD, 11), 81% were men, 40% were nonwhite, 22% had moderate chronic kidney disease (estimated GFR <60 mL/min per 1.73 m2), and 27% had type 2 diabetes mellitus. Cardiovascular disease events occurred among 182 individuals during 3913 person-years of follow-up (median, 5.2 years), and 220 deaths occurred during 4360 person-years of follow-up (median, 6.0 years). Participants who had events were older; were more frequently men; were more likely to be receiving an angiotensin-converting enzyme inhibitor or angiotensin-receptor blocker and anticoagulants; and had lower peak exercise capacity, lower ejection fraction, greater waist-to-hip ratio, greater prevalence of diabetes, lower estimated GFR and high-density lipoprotein cholesterol levels, and higher C-reactive protein levels (Table 1).
The distribution of FGF23 was right-skewed, with a median of 43.1 RU/mL (interquartile range, 28.9 to 72.3 RU/mL), so the variable was natural log–transformed to approximate a normal distribution. The distributions of ucMGP and fetuin-A were approximately normal with means of 3290 nM (SD, 1178) and 0.65 g/L (SD, 0.14), respectively (Appendix Figure, available at www.annals.org). After adjustment for age, sex, race, and estimated GFR, associations of mineral regulators with calcium, phosphorus, and one another demonstrated that a higher FGF23 level was strongly associated with phosphorus and weakly associated with calcium and that a higher ucMGP level was associated with a higher fetuin-A level (Appendix Table 2, available at www.annals.org). The association of each novel mineral regulator with major demographic variables and traditional CVD risk factors varied by risk factor (Appendix Table 3, available at www.annals.org), but persons with diabetes had higher levels of each of the 3 mineral regulators.
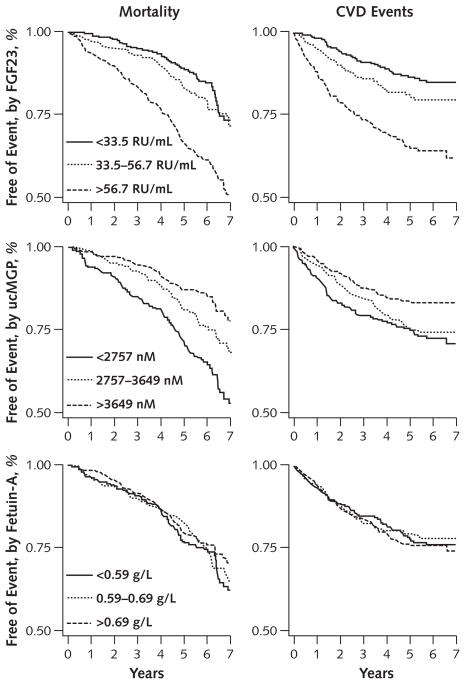
Both higher FGF23 and lower ucMGP levels were associated with greater risk for mortality and CVD events in Kaplan–Meier analysis. In contrast, fetuin-A levels were not associated with either outcome (Figure). Table 2 shows the adjusted associations of each novel mineral regulator with CVD events and mortality. Compared with participants in the lowest FGF23 tertile, those in the highest tertile had approximately 3-fold greater risk for CVD events and mortality in models adjusted for age, sex, and race. Additional adjustment for traditional CVD risk factors, kidney function, C-reactive protein level, ejection fraction, peak exercise capacity, and medication use (multivariate model 1) attenuated this association; however, the highest tertile remained associated with an approximately 2-fold greater risk for either outcome. When FGF23 was evaluated as a continuous predictor, each doubling in FGF23 level was associated with a 41% higher mortality risk and 24% higher CVD event risk. Further adjustment for ucMGP, fetuin-A, calcium, and phosphorus produced essentially unaltered associations. Table 3 shows the associations of FGF23 with the individual components of the composite CVD event outcome, in which associations were qualitatively strongest for heart failure and stroke or transient ischemic attack.
Figure. Unadjusted event-free survival, by tertiles of FGF23, ucMGP, and fetuin-A.
CVD = cardiovascular disease; FGF23 = fibroblast growth factor 23; ucMGP = uncarboxylated matrix Gla protein.
Table 2.
Association of FGF23, ucMGP, and Fetuin-A Levels With All-Cause Mortality or CVD Events
| Variable | HR (95% CI), by Tertile | HR (95% CI), by Continuous Variable in Cox Model | P Value | ||
|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | |||
| FGF23* | Per Doubling of FGF23† | ||||
| All-cause mortality | |||||
| Age-, sex-, and race-adjusted | 1.00 | 1.30 (0.89–1.89) | 2.86 (2.04–4.01) | 1.54 (1.39–1.70) | <0.001 |
| Multivariate model 1‡ | 1.00 | 1.38 (0.92–2.08) | 2.15 (1.43–3.24) | 1.41 (1.23–1.60) | <0.001 |
| Multivariate model 2§ | 1.00 | 1.39 (0.93–2.09) | 2.07 (1.36–3.13) | 1.40 (1.22–1.60) | <0.001 |
| CVD event | |||||
| Age-, sex-, and race-adjusted | 1.00 | 1.50 (0.99–2.27) | 3.09 (2.12–4.52) | 1.49 (1.35–1.66) | <0.001 |
| Multivariate model 1‡ | 1.00 | 1.46 (0.94–2.29) | 1.83 (1.15–2.91) | 1.24 (1.07–1.43) | 0.004 |
| Multivariate model 2§ | 1.00 | 1.47 (0.94–2.30) | 1.83 (1.14–2.94) | 1.24 (1.06–1.44) | 0.006 |
| ucMGP|| | Per 1000-nM Greater ucMGP Level | ||||
| All-cause mortality | |||||
| Age-, sex-, and race-adjusted | 1.00 | 0.71 (0.52–0.96) | 0.51 (0.36–0.74) | 0.75 (0.66–0.85) | <0.001 |
| Multivariate model 1‡ | 1.00 | 0.69 (0.49–0.96) | 0.48 (0.31–0.75) | 0.78 (0.68–0.90) | 0.001 |
| Multivariate model 2§ | 1.00 | 0.70 (0.49–0.99) | 0.49 (0.31–0.78) | 0.79 (0.68–0.92) | 0.002 |
| CVD event | |||||
| Age-, sex-, and race-adjusted | 1.00 | 0.92 (0.66–1.29) | 0.70 (0.48–1.04) | 0.82 (0.71–0.94) | 0.005 |
| Multivariate model 1‡ | 1.00 | 0.96 (0.66–1.41) | 0.65 (0.40–1.05) | 0.84 (0.72–0.98) | 0.029 |
| Multivariate model 2§ | 1.00 | 0.98 (0.66–1.45) | 0.64 (0.39–1.05) | 0.84 (0.72–0.99) | 0.042 |
| Fetuin-A¶ | Per 0.1-g/L Greater Fetuin-A Level | ||||
| All-cause mortality | |||||
| Age-, sex-, and race-adjusted | 1.00 | 0.98 (0.71–1.34) | 1.08 (0.77–1.50) | 0.98 (0.89–1.09) | 0.74 |
| Multivariate model 1‡ | 1.00 | 0.89 (0.62–1.27) | 0.84 (0.55–1.27) | 0.92 (0.81–1.05) | 0.22 |
| Multivariate model 2§ | 1.00 | 0.85 (0.59–1.22) | 0.82 (0.53–1.28) | 0.91 (0.80–1.04) | 0.179 |
| CVD event | |||||
| Age-, sex-, and race-adjusted | 1.00 | 0.99 (0.69–1.43) | 1.29 (0.90–1.84) | 1.10 (0.99–1.23) | 0.071 |
| Multivariate model 1‡ | 1.00 | 0.86 (0.57–1.30) | 0.99 (0.64–1.55) | 1.08 (0.94–1.24) | 0.28 |
| Multivariate model 2§ | 1.00 | 0.92 (0.60–1.39) | 1.12 (0.71–1.78) | 1.14 (0.98–1.31) | 0.081 |
CVD = cardiovascular disease; FGF23 = fibroblast growth factor 23; HR = hazard ratio; ucMGP = uncarboxylated matrix Gla protein.
The values for tertiles 1, 2, and 3 are <33.5, 33.5–56.7, and >56.7 RU/mL, respectively.
HRs for FGF23 as a continuous predictor are expressed as “per doubling of FGF23” because it was natural log–transformed for analysis.
Adjusted for age; sex; race; waist-to-hip ratio; smoking; hypertension; diabetes; systolic and diastolic blood pressures; estimated glomerular filtration rate; total and high-density lipoprotein cholesterol levels; C-reactive protein level; ejection fraction; peak exercise capacity; and use of aspirin, β-blockers, angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers, statins, or anticoagulants.
Adjusted for covariates in multivariable model 1 plus calcium level, phosphorus level, and the 2 other predictor variables (for example, in the FGF23 model, ucMGP and fetuin-A were included as adjustment covariates).
The values for tertiles 1, 2, and 3 are <2757, 2757–3649, and >3649 nM, respectively.
The values for tertiles 1, 2, and 3 are <0.59, 0.59–0.69, and >0.69 g/L, respectively.
Table 3.
HRs for Individual Events per Change in FGF23, ucMGP, or Fetuin-A Level*
| Variable | Per Doubling of FGF23† | Per 1000-nM Greater ucMGP Level | Per 0.1-g/L Greater Fetuin-A Level | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | P Value | HR (95% CI) | P Value | HR (95% CI) | P Value | |
| Death (n =220) | 1.54 (1.39–1.70) | <0.001 | 0.78 (0.68–0.90) | 0.001 | 0.92 (0.81–1.05) | 0.22 |
| Cardiovascular event (n =182) | 1.49 (1.35–1.66) | 0.004 | 0.84 (0.72–0.98) | 0.029 | 1.08 (0.94–1.24) | 0.28 |
| Heart failure (n =119) | 1.31 (1.08–1.59) | 0.005 | 0.88 (0.72–1.08) | 0.22 | 1.04 (0.87–1.25) | 0.67 |
| Myocardial infarction (n =88) | 1.05 (0.85–1.30) | 0.67 | 0.75 (0.59–0.95) | 0.015 | 1.04 (0.85–1.26) | 0.71 |
| Stroke or transient ischemic attack (n =36) | 1.50 (1.11–2.04) | 0.008 | 1.00 (0.72–1.40) | 0.99 | 1.09 (0.82–1.45) | 0.55 |
FGF23 = fibroblast growth factor 23; HR = hazard ratio; ucMGP = uncarboxylated matrix Gla protein.
Adjusted for age; sex; race; waist-to-hip ratio; smoking status; hypertension; diabetes; systolic and diastolic blood pressures; estimated glomerular filtration rate; total and high-density lipoprotein cholesterol levels; C-reactive protein level; ejection fraction; peak exercise capacity; and use of aspirin, β-blockers, angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers, statins, or anticoagulants.
HRs for FGF23 as a continuous predictor are expressed as “per doubling of FGF23” because it was natural log–transformed for analysis.
Serum ucMGP levels were also associated with mortality risk, but in the opposite direction. Compared with participants in the lowest ucMGP tertile, participants in the highest tertile had approximately 50% lower risk for mortality in models adjusted for age, sex, and race (Table 2). This association remained after adjustment for traditional CVD risk factors, kidney function, C-reactive protein level, ejection fraction, peak exercise capacity, and medication use. The association of ucMGP with CVD events was in the same direction but of lesser magnitude. When ucMGP was evaluated as a continuous predictor variable, each 1000-nM higher ucMGP level was associated with a 22% lower risk for mortality and a 16% lower risk for CVD events. Further adjustment for FGF23, fetuin-A, calcium, and phosphorus had little effect on these associations. Among the components of the composite CVD outcome, MI risk was qualitatively more strongly associated with lower ucMGP levels (Table 3).
Compared with participants in the lowest fetuin-A tertile, participants in the highest tertile were not at statistically significantly higher risk for either CVD events or mortality. When fetuin-A was evaluated as a continuous predictor variable, its associations with mortality and CVD events were near unity (Tables 2 and 3).
We evaluated whether these associations were modified by diabetes or chronic kidney disease status. Association of all 3 mineral regulators with mortality or CVD events was similar, regardless of the presence or absence of chronic kidney disease (P ≥ 0.41 for interaction in multivariate model 1 for all). With the exception of the association of ucMGP with mortality, results were also similar in persons with or without diabetes (P ≥ 0.41 for interaction for all). The association of ucMGP with mortality was limited to participants without diabetes (P = 0.035 for interaction). In these persons (n = 610), each 1000-nM higher ucMGP level was associated with 33% lower mortality risk (hazard ratio, 0.67 [95% CI, 0.56 to 0.81]; P < 0.001), whereas there was no significant association of uc-MGP with mortality in 221 participants with diabetes (hazard ratio, 1.10 [CI, 0.85 to 1.41]; P = 0.47).
Discussion
We demonstrate that among persons with stable CAD and a range of kidney function from normal to moderate chronic kidney disease, higher FGF23 levels and lower ucMGP levels are associated with mortality and CVD events, independent of one another, traditional CVD risk factors, kidney function, and C-reactive protein levels. In contrast, fetuin-A levels were not statistically significantly associated with mortality or CVD events.
Higher FGF23 levels are associated with higher mortality in cohorts with ESRD (30). However, to our knowledge, the association of FGF23 with mortality and CVD events in other settings has not previously been evaluated. The mechanisms responsible for this association are uncertain, but several possibilities exist. Fibroblast growth factor 23, an endocrine regulator of phosphorus metabolism, may be a marker of higher time-averaged serum phosphorus levels, and by extension, FGF23 may be a marker of vascular calcification. We and others (30) have demonstrated that higher FGF23 levels are associated with higher serum phosphorus levels, and others have shown that higher serum phosphorus levels are associated with vascular calcification and CVD events in community-living samples with and without severe kidney disease (7–9, 39). However, in our study and in a previous study among patients with ESRD, the associations of FGF23 with mortality were independent of serum phosphorus levels measured at one point in time. It may be that a single serum phosphorus measurement does not accurately represent cumulative phosphorus burden, given the high intraindividual biological variability in serum phosphorus levels when measured repeatedly over time (40). Because active vitamin D regulates the renin–angiotensin axis (41) and proliferation of cardiomyocytes (42), FGF23 may also affect the cardiovascular system through its inhibition of vitamin D activation (43). Fibroblast growth factor 23 is associated with left ventricular hypertrophy (44, 45), a relation that may be explained by the previously mentioned mechanisms, but it is also possible that FGF23 may influence cardiac remodeling through other mechanisms (46– 48). Future studies that elucidate potential direct or indirect effects of FGF23 are required to determine these mechanisms.
To our knowledge, the relationship of ucMGP with mortality has not previously been demonstrated in any setting. Vascular smooth-muscle cells produce MGP, and vitamin K– dependent posttranslational activation produces carboxylated MGP, a locally acting and potent inhibitor of arterial calcification (18, 19, 25). Its precursor, ucMGP, accumulates in arteries near calcium deposits (22)—a process that may lower its blood concentration. Persons with ESRD, calcific uremic arteriolopathy, or cardiac valvular calcification have lower blood ucMGP levels than healthy control participants (31, 32), and ucMGP is inversely correlated with coronary artery calcification (33). The present analysis is novel in demonstrating that a lower ucMGP level is associated with mortality in persons with CAD, and that this association was limited to persons without diabetes. The mechanisms responsible for the effect modification by diabetes are uncertain, but the absence of diabetes may reflect a different underlying biology predisposing to ectopic calcification. We have shown previously that a lower ucMGP level was independently associated with cardiac valve calcification. This association, however, was also limited to persons without diabetes (49).
Synthesized by the liver and secreted into blood, fetuin-A is a circulating inhibitor of arterial calcification that increases the solubility of hydroxyapatite precursors in the bloodstream, reminiscent of mechanisms through which apolipoproteins increase the solubility of lipids (50). In cohorts with ESRD, lower fetuin-A levels have consistently been associated with higher mortality risk (27–29). Whether similar associations extend to nondialysis cohorts is uncertain. In persons with stage 3 or 4 chronic kidney disease, we observed no association of fetuin-A levels with all-cause or CVD mortality (51). Similarly, we observed no association of fetuin-A levels with mortality or CVD events among persons with normal to near-normal kidney function. Studies by other investigators in cohorts without ESRD have provided conflicting results. In 1 study, lower fetuin-A levels were associated with mortality among patients hospitalized with acute MI (52), whereas another study reported that higher fetuin-A levels were associated with MI in a community-living cohort (53). In cohorts with ESRD, fetuin-A has been suggested as a negative acute-phase reactant (27–29), but this association is not consistently observed in other settings (51, 54). In our analysis, we found no statistically significant association between fetuin-A and C-reactive protein levels after correction for other cardiovascular risks and estimated GFR (55). The reasons for conflicting results between ESRD cohorts and other settings remain uncertain. Because persons with ESRD have a high prevalence and severity of vascular calcification and frequently have calcium and phosphorus levels well outside the normal laboratory ranges, we hypothesize that this extreme phenotype may be required to observe the association of low fetuin-A levels with CVD events. Finally, although higher fetuin-A levels have also been associated with insulin resistance and risk for type 2 diabetes mellitus (56), the association of fetuin-A with CVD events and mortality in our study was similar among participants with or without diabetes.
Our study has important limitations. Parathyroid hormone and vitamin D levels were not measured, and whether participants used calcium or vitamin D supplements and bisphosphonates is unknown. All participants had prevalent CAD, and most were older men. Results may differ in younger persons, women, and persons without CAD. Although most deaths in older persons with prevalent CAD are related to cardiovascular events (1), the associations between FGF23 and ucMGP levels with mortality may also reflect noncardiovascular events, and the causes of death were not specifically adjudicated in our study. Heart failure was considered an ischemic event in this cohort with prevalent CAD, yet some heart failure events may have been nonischemic in origin. For the association of fetuin-A and events, our sample size was limited to 833 persons, and it is possible that a true association was missed or that results may differ with other assays; the results should be interpreted within the confines of the 95% CIs.
In conclusion, higher FGF23 and lower ucMGP levels are each associated with mortality and CVD events in outpatients with stable CAD. These findings need to be replicated in independent populations, but they add to the growing body of literature demonstrating that abnormalities in mineral metabolism may identify persons at greater risk for CVD events and death. Because in vitro and animal studies demonstrate that these pathways influence arterial calcification, future studies determining the exact mechanisms may ultimately provide novel therapies for primary or secondary prevention of CVD.
Context
Other investigators recently identified novel regulators of mineral metabolism, which have been associated with cardiovascular disease and mortality in patients with end-stage renal disease.
Contribution
The authors studied 833 persons and found that those with higher levels of fibroblast growth factor 23 and lower levels of uncarboxylated matrix Gla protein had higher cardiovascular disease event rates and mortality after adjustment for traditional risk factors, C-reactive protein levels, and kidney function.
Caution
All study participants had coronary artery disease.
Implication
Better understanding of these relationships might lead to new therapies for cardiovascular disease.
—The Editors
Acknowledgments
Grant Support: By a Hypertension Training Grant (T32 HL007261) through the National Heart, Lung, and Blood Institute (NHLBI) (Dr. Parker), an American Heart Association Fellow-to-Faculty transition grant, and the NHLBI (grant R01HL096851, Dr. Ix). The Heart and Soul Study was supported by the Department of Veterans Epidemiology Merit Review Program; the Department of Veterans Affairs Health Services Research and Development service; the NHLBI (grant R01 HL079235); the American Federation for Aging Research (Paul Beeson Scholars Program); the Robert Wood Johnson Foundation (Generalist Physician Faculty Scholars Program); and the Ischemia Research and Education Foundation. Dr. Ix had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Appendix
Plasma FGF23 concentrations were measured by using a C-terminal human enzyme-linked immunosorbent assay (Immunotopics, San Clemente, California) (57). This assay recognizes 2 epitopes on the C-terminal side cleavage site and thus recognizes both full-length FGF23 and C-terminal cleavage fragments of FGF-23. The intra-assay coefficient of variation was 5.0%; the interassay coefficient of variation was 9.9% at a concentration of 36.4 RU/mL and 12.6% at a level 379 RU/mL. Serum ucMGP (nM) was measured by using a competitive enzyme-linked immunosorbent assay developed at VitaK BV, Maastricht University, Maastricht, the Netherlands (58). The assay detects both phosphorylated and nonphosphorylated ucMGP. The lower limit of detection is 98 nM. The intra-assay and interassay coefficients of variation were 6% and 11%, respectively, Fetuin-A was measured by a nephelometric assay (59). This assay uses the same high-specificity antibody as commercially available enzyme-linked immunosorbent assays and has been used in a previous analysis showing an association between fetuin-A and mortality in hemodialysis patients (60). The assay ranges from 0.05 to 3.5 g/L. The intra-assay coefficient of variation was 7.7%, and the interassay coefficient of variation was 8.1%.
Appendix Figure. Distribution of FGF23*, ucMGP, and fetuin-A levels among 833 participants with coronary artery disease.
FGF23 = fibroblast growth factor 23; ucMGP = uncarboxylated matrix Gla protein.
*16 participants with FGF23 levels >480 RU/mL (range, 529 to 3150 RU/mL) were not included because of space limitations.
Appendix Table 1.
Comparison of Variables in Persons Excluded Because of Missing Measurements
| Variable | Included in Analysis (n = 833) | Excluded From Analysis (n = 191) | P Value |
|---|---|---|---|
| Major baseline characteristics | |||
| Mean age (SD), y | 67 (11) | 67 (10) | 0.89 |
| Women, n (%) | 156 (19) | 28 (14) | 0.137 |
| Race, n (%) | 0.70 | ||
| White | 499 (60) | 120 (62) | – |
| Black | 135 (16) | 34 (17) | – |
| Other | 198 (24) | 41 (21) | – |
| Diabetes, n (%) | 221 (27) | 45 (23) | 0.37 |
| Outcomes, n (%) | |||
| Heart failure | 119 (14) | 24 (13) | 0.62 |
| Myocardial infarction | 88 (11) | 21 (11) | 0.78 |
| Stroke or transient ischemic attack | 36 (4) | 12 (6) | 0.22 |
| Mortality | 221 (27) | 43 (23) | 0.25 |
| Combined outcomes | 301 (36) | 65 (35) | 0.76 |
Appendix Table 2.
Association of Calcium and Phosphorus With ln-FGF23, ucMGP, and Fetuin-A Levels*
| Variable | ln-FGF23 | ucMGP | Fetuin-A | |||
|---|---|---|---|---|---|---|
| Unadjusted β (95% CI) | P Value | Unadjusted β (95% CI) | P Value | Unadjusted β (95% CI) | P Value | |
| ln-FGF23 | – | – | −0.05 (−0.12 to 0.01) | 0.116 | 0.10 (0.03 to 0.17) | 0.004 |
| ucMGP | −0.05 (−0.12 to 0.01) | 0.116 | – | – | 0.27 (0.21 to 0.34) | <0.001 |
| Fetuin-A | 0.10 (0.03 to 0.17) | 0.004 | 0.27 (0.21 to 0.34) | <0.001 | – | – |
| Calcium† | 0.11 (0.04 to 0.18) | 0.001 | 0.06 (0.00 to 0.13) | 0.064 | 0.08 (0.01 to 0.14) | 0.027 |
| Phosphorus | 0.27 (0.21 to 0.34) | <0.001 | 0.10 (0.03 to 0.16) | 0.006 | 0.09 (0.02 to 0.16) | 0.008 |
| Adjusted β (95% CI)‡ | Adjusted β (95% CI)‡ | Adjusted β (95% CI)‡ | ||||
| ln-FGF23 | – | – | −0.03 (−0.1 to 0.04) | 0.39 | 0.06 (−0.01 to 0.13) | 0.111 |
| ucMGP | −0.03 (−0.09 to 0.04) | 0.39 | – | – | 0.25 (0.19 to 0.32) | <0.001 |
| Fetuin-A | 0.05 (−0.01 to 0.12) | 0.111 | 0.25 (0.18 to 0.31) | <0.001 | – | – |
| Calcium† | 0.09 (0.02 to 0.15) | 0.008 | 0.04 (−0.03 to 0.11) | 0.22 | 0.05 (−0.02 to 0.12) | 0.147 |
| Phosphorus | 0.19 (0.12 to 0.25) | <0.001 | 0.08 (0.01 to 0.15) | 0.025 | 0.02 (−0.05 to 0.09) | 0.62 |
ln-FGF23 = natural log–transformed fibroblast growth factor 23; ucMGP = uncarboxylated matrix Gla protein.
Data are coefficients of regression (β) with 95% CIs representing SD change in ln-FGF23, ucMGP, and fetuin-A levels per SD change in predictor variables (FGF23 was log-transformed for regression analysis because of nonparametric distribution).
Adjusted calcium = total calcium + (4.0 − serum albumin) × 0.8.
Adjusted for age, sex, race, and estimated glomerular filtration rate.
Appendix Table 3.
Association Between Baseline Factors and ln-FGF23, ucMGP, and Fetuin-A Levels*
| Variable | ln-FGF23 | ucMGP | Fetuin-A | |||
|---|---|---|---|---|---|---|
| β (95% CI) | P Value | β (95% CI) | P Value | β (95% CI) | P Value | |
| Age (per 10 y) | −0.05 (−0.11 to 0.01) | 0.107 | −0.21 (−0.27 to −0.15) | <0.001 | −0.18 (−0.24 to −0.12) | <0.001 |
| Women | 0.40 (0.23 to 0.57) | <0.001 | 0.15 (−0.02 to 0.32) | 0.093 | 0.26 (0.09 to 0.43) | 0.004 |
| Nonwhite race | −0.04 (−0.18 to 0.10) | 0.56 | 0.06 (−0.08 to 0.20) | 0.37 | 0.07 (−0.07 to 0.21) | 0.35 |
| Smoking status | 0.36 (0.19 to 0.53) | <0.001 | 0.13 (−0.04 to 0.30) | 0.128 | 0.08 (−0.09 to 0.25) | 0.36 |
| Hypertension | 0.14 (−0.01 to 0.29) | 0.077 | −0.05 (−0.20 to 0.11) | 0.54 | −0.02 (−0.17 to 0.14) | 0.84 |
| Diabetes | 0.27 (0.12 to 0.42) | 0.001 | 0.26 (0.11 to 0.41) | 0.001 | 0.27 (0.12 to 0.43) | <0.001 |
| Estimated GFR† | −0.32 (−0.39 to −0.26) | <0.001 | 0.20 (0.13 to 0.27) | <0.001 | −0.01 (−0.08 to 0.06) | 0.83 |
| Waist-to-hip ratio† | −0.03 (−0.10 to 0.04) | 0.36 | 0.03 (−0.04 to 0.10) | 0.40 | 0.04 (−0.03 to 0.11) | 0.21 |
| Total cholesterol level† | 0.01 (−0.06 to 0.08) | 0.82 | 0.16 (0.10 to 0.23) | <0.001 | 0.35 (0.29 to 0.42) | <0.001 |
| HDL cholesterol level† | −0.05 (−0.11 to 0.02) | 0.184 | −0.05 (−0.11 to 0.02) | 0.185 | −0.17 (−0.24 to −0.10) | <0.001 |
| ln-hsCRP level† | 0.20 (0.13 to 0.27) | <0.001 | 0.01 (−0.06 to 0.07) | 0.88 | 0.08 (0.01 to 0.15) | 0.020 |
| Ejection fraction† | −0.11 (−0.18 to −0.05) | 0.001 | 0.05 (−0.02 to 0.12) | 0.149 | −0.07 (−0.14 to 0.00) | 0.050 |
| Max METS† | −0.23 (−0.30 to −0.16) | <0.001 | 0.10 (0.03 to 0.17) | 0.006 | −0.04 (−0.11 to 0.02) | 0.21 |
| Medication use | ||||||
| β-Blocker | 0.04 (−0.10 to 0.18) | 0.55 | 0.02 (−0.11 to 0.16) | 0.73 | 0.06 (−0.08 to 0.20) | 0.40 |
| ACE inhibitor or ARB | 0.12 (−0.02 to 0.25) | 0.092 | 0.04 (−0.10 to 0.18) | 0.56 | 0.02 (−0.12 to 0.16) | 0.79 |
| Aspirin | −0.23 (−0.39 to −0.06) | 0.006 | −0.06 (−0.23 to 0.10) | 0.45 | −0.09 (−0.25 to 0.07) | 0.26 |
| Statin | −0.13 (−0.27 to 0.01) | 0.079 | 0.08 (−0.06 to 0.22) | 0.26 | −0.09 (−0.24 to 0.05) | 0.198 |
| Anticoagulant | 0.63 (0.38 to 0.89) | <0.001 | −0.04 (−0.30 to 0.22) | 0.78 | −0.09 (−0.35 to 0.17) | 0.51 |
ACE = angiotensin-converting enzyme; ARB = angiotensin-receptor blocker; GFR = glomerular filtration rate (based on the 4-variable Modification of Diet in Renal Disease Study equation); HDL = high-density lipoprotein; ln-FGF23 = natural log–transformed fibroblast growth factor 23; ln-hsCRP = natural log–transformed high-sensitivity C-reactive protein; max METS = maximum attainable metabolic equivalent tasks (obtained by using the modified Bruce protocol); ucMGP = uncarboxylated matrix Gla protein.
Data are coefficients of regression (β) with 95% CIs representing SD change in ln-FGF23, ucMGP, and fetuin-A levels per SD change in predictor variables (FGF23 was log-transformed for regression analysis because of nonparametric distribution).
Predictor variable is normalized by its SD for regression analysis.
Footnotes
Potential Conflicts of Interest: Disclosures can be viewed at www.acponline.org/authors/icmje/ConflictOfInterestForms.do?msNum=M09-1886.
Reproducible Research Statement: Study protocol: Available at http://dgim.ucsf.edu/heartandsoulstudy/index.html. Statistical code: Available from Dr. Ix (joeix@ucsd.edu). Data set: Restricted access; see http://dgim.ucsf.edu/heartandsoulstudy/index.html.
Current Author Addresses: Drs. Parker and Ix: Division of Nephrology and Hypertension, University of California, San Diego, and Veterans Affairs San Diego Healthcare System, 3350 La Jolla Village Drive, Mail Code 111-H, San Diego, CA 92161.
Dr. Schurgers: Department of Biochemistry, University of Maastricht, PO Box 616, 6200 MD Maastricht, the Netherlands.
Dr. Brandenburg: Department of Nephrology and Clinical Immunology, University Hospital Aachen, Pauwelsstraße 30, D-52057 Aachen, Germany.
Dr. Christenson: Clinical Pathology, University of Maryland Medical Center, 22 South Greene Street, Baltimore, MD 21201.
Dr. Vermeer: VitaK BV, Maastricht University, BioPartner Center Maastricht, Oxfordlaan 70 6229 EV Maastricht, the Netherlands. Dr. Ketteler: Nephrologische Klinik, Klinikum Coburg, Ketschendorfer Strasse 33, 96450 Coburg, Germany.
Dr. Shlipak and Whooley: Veterans Affairs Medical Center (111A1), 4150 Clement Street, San Francisco, CA 94121.
Author Contributions: Conception and design: J.H. Ix.
Analysis and interpretation of the data: B.D. Parker, L.J. Schurgers, V.M. Brandenburg, R.H. Christenson, C. Vermeer, M. Ketteler, M.G. Shlipak, M.A. Whooley, J.H. Ix.
Drafting of the article: B.D. Parker, J.H. Ix.
Critical revision of the article for important intellectual content: B.D. Parker, L.J. Schurgers, V.M. Brandenburg, R.H. Christenson, C. Vermeer, M. Ketteler, M.G. Shlipak, M.A. Whooley, J.H. Ix.
Final approval of the article: B.D. Parker, L.J. Schurgers, V.M. Brandenburg, R.H. Christenson, C. Vermeer, M. Ketteler, M.G. Shlipak, M.A. Whooley, J.H. Ix.
Provision of study materials or patients: L.J. Schurgers, V.M. Brandenburg, R.H. Christenson, C. Vermeer, M. Ketteler, M.A. Whooley.
Statistical expertise: B.D. Parker, M.G. Shlipak, J.H. Ix.
Obtaining of funding: L.J. Schurgers, V.M. Brandenburg, C. Vermeer, M.A. Whooley, J.H. Ix.
Administrative, technical, or logistic support: B.D. Parker, J.H. Ix.
Collection and assembly of data: L.J. Schurgers, V.M. Brandenburg, R.H. Christenson, C. Vermeer, M. Ketteler, M.A. Whooley, J.H. Ix.
References
- 1.Lloyd-Jones D, Adams R, Carnethon M, De Simone G, Ferguson TB, Flegal K, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2009;119:e21–181. doi: 10.1161/CIRCULATIONAHA.108.191261. [DOI] [PubMed] [Google Scholar]
- 2.Schlant RC, Forman S, Stamler J, Canner PL. The natural history of coronary heart disease: prognostic factors after recovery from myocardial infarction in 2789 men. The 5-year findings of the coronary drug project. Circulation. 1982;66:401–14. doi: 10.1161/01.cir.66.2.401. [DOI] [PubMed] [Google Scholar]
- 3.Vittinghoff E, Shlipak MG, Varosy PD, Furberg CD, Ireland CC, Khan SS, et al. Heart and Estrogen/progestin Replacement Study Research Group. Risk factors and secondary prevention in women with heart disease: the Heart and Estrogen/progestin Replacement Study. Ann Intern Med. 2003;138:81–9. doi: 10.7326/0003-4819-138-2-200301210-00007. [DOI] [PubMed] [Google Scholar]
- 4.Steitz SA, Speer MY, Curinga G, Yang HY, Haynes P, Aebersold R, et al. Smooth muscle cell phenotypic transition associated with calcification: upregulation of Cbfa1 and downregulation of smooth muscle lineage markers. Circ Res. 2001;89:1147–54. doi: 10.1161/hh2401.101070. [DOI] [PubMed] [Google Scholar]
- 5.Jono S, McKee MD, Murry CE, Shioi A, Nishizawa Y, Mori K, et al. Phosphate regulation of vascular smooth muscle cell calcification. Circ Res. 2000;87:E10–7. doi: 10.1161/01.res.87.7.e10. [DOI] [PubMed] [Google Scholar]
- 6.Foley RN, Collins AJ, Ishani A, Kalra PA. Calcium-phosphate levels and cardiovascular disease in community-dwelling adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am Heart J. 2008;156:556–63. doi: 10.1016/j.ahj.2008.05.016. [DOI] [PubMed] [Google Scholar]
- 7.Dhingra R, Sullivan LM, Fox CS, Wang TJ, D’Agostino RB, Sr, Gaziano JM, et al. Relations of serum phosphorus and calcium levels to the incidence of cardiovascular disease in the community. Arch Intern Med. 2007;167:879–85. doi: 10.1001/archinte.167.9.879. [DOI] [PubMed] [Google Scholar]
- 8.Tonelli M, Sacks F, Pfeffer M, Gao Z, Curhan G Cholesterol And Recurrent Events Trial Investigators. Relation between serum phosphate level and cardiovascular event rate in people with coronary disease. Circulation. 2005;112:2627–33. doi: 10.1161/CIRCULATIONAHA.105.553198. [DOI] [PubMed] [Google Scholar]
- 9.Foley RN, Collins AJ, Herzog CA, Ishani A, Kalra PA. Serum phosphorus levels associate with coronary atherosclerosis in young adults. J Am Soc Nephrol. 2009;20:397–404. doi: 10.1681/ASN.2008020141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dobnig H, Pilz S, Scharnagl H, Renner W, Seelhorst U, Wellnitz B, et al. Independent association of low serum 25-hydroxyvitamin d and 1,25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch Intern Med. 2008;168:1340–9. doi: 10.1001/archinte.168.12.1340. [DOI] [PubMed] [Google Scholar]
- 11.Hagström E, Hellman P, Larsson TE, Ingelsson E, Berglund L, Sundström J, et al. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation. 2009;119:2765–71. doi: 10.1161/CIRCULATIONAHA.108.808733. [DOI] [PubMed] [Google Scholar]
- 12.Antoniucci DM, Yamashita T, Portale AA. Dietary phosphorus regulates serum fibroblast growth factor-23 concentrations in healthy men. J Clin Endocrinol Metab. 2006;91:3144–9. doi: 10.1210/jc.2006-0021. [DOI] [PubMed] [Google Scholar]
- 13.Burnett SM, Gunawardene SC, Bringhurst FR, Jüppner H, Lee H, Finkelstein JS. Regulation of C-terminal and intact FGF-23 by dietary phosphate in men and women. J Bone Miner Res. 2006;21:1187–96. doi: 10.1359/jbmr.060507. [DOI] [PubMed] [Google Scholar]
- 14.Gutierrez O, Isakova T, Rhee E, Shah A, Holmes J, Collerone G, et al. Fibroblast growth factor-23 mitigates hyperphosphatemia but accentuates calcitriol deficiency in chronic kidney disease. J Am Soc Nephrol. 2005;16:2205–15. doi: 10.1681/ASN.2005010052. [DOI] [PubMed] [Google Scholar]
- 15.Shimada T, Kakitani M, Yamazaki Y, Hasegawa H, Takeuchi Y, Fujita T, et al. Targeted ablation of Fgf23 demonstrates an essential physiological role of FGF23 in phosphate and vitamin D metabolism. J Clin Invest. 2004;113:561–8. doi: 10.1172/JCI19081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S, et al. Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia. Proc Natl Acad Sci U S A. 2001;98:6500–5. doi: 10.1073/pnas.101545198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.El-Abbadi MM, Pai AS, Leaf EM, Yang HY, Bartley BA, Quan KK, et al. Phosphate feeding induces arterial medial calcification in uremic mice: role of serum phosphorus, fibroblast growth factor-23, and osteopontin. Kidney Int. 2009;75:1297–307. doi: 10.1038/ki.2009.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Boström K, Tsao D, Shen S, Wang Y, Demer LL. Matrix GLA protein modulates differentiation induced by bone morphogenetic protein-2 in C3H10T1/2 cells. J Biol Chem. 2001;276:14044–52. doi: 10.1074/jbc.M008103200. [DOI] [PubMed] [Google Scholar]
- 19.Zebboudj AF, Shin V, Boström K. Matrix GLA protein and BMP-2 regulate osteoinduction in calcifying vascular cells. J Cell Biochem. 2003;90:756–65. doi: 10.1002/jcb.10669. [DOI] [PubMed] [Google Scholar]
- 20.Reynolds JL, Joannides AJ, Skepper JN, McNair R, Schurgers LJ, Proudfoot D, et al. Human vascular smooth muscle cells undergo vesicle-mediated calcification in response to changes in extracellular calcium and phosphate concentrations: a potential mechanism for accelerated vascular calcification in ESRD. J Am Soc Nephrol. 2004;15:2857–67. doi: 10.1097/01.ASN.0000141960.01035.28. [DOI] [PubMed] [Google Scholar]
- 21.Murshed M, Schinke T, McKee MD, Karsenty G. Extracellular matrix mineralization is regulated locally; different roles of two gla-containing proteins. J Cell Biol. 2004;165:625–30. doi: 10.1083/jcb.200402046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schurgers LJ, Teunissen KJ, Knapen MH, Kwaijtaal M, van Diest R, Appels A, et al. Novel conformation-specific antibodies against matrix gamma-carboxyglutamic acid (Gla) protein: undercarboxylated matrix Gla protein as marker for vascular calcification. Arterioscler Thromb Vasc Biol. 2005;25:1629–33. doi: 10.1161/01.ATV.0000173313.46222.43. [DOI] [PubMed] [Google Scholar]
- 23.Heiss A, DuChesne A, Denecke B, Grötzinger J, Yamamoto K, Renné T, et al. Structural basis of calcification inhibition by alpha 2-HS glycoprotein/fetuin-A. Formation of colloidal calciprotein particles. J Biol Chem. 2003;278:13333–41. doi: 10.1074/jbc.M210868200. [DOI] [PubMed] [Google Scholar]
- 24.Schafer C, Heiss A, Schwarz A, Westenfeld R, Ketteler M, Floege J, et al. The serum protein alpha 2-Heremans-Schmid glycoprotein/fetuin-A is a systemically acting inhibitor of ectopic calcification. J Clin Invest. 2003;112:357–66. doi: 10.1172/JCI17202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, Behringer RR, et al. Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature. 1997;386:78–81. doi: 10.1038/386078a0. [DOI] [PubMed] [Google Scholar]
- 26.Sitara D, Razzaque MS, St-Arnaud R, Huang W, Taguchi T, Erben RG, et al. Genetic ablation of vitamin D activation pathway reverses biochemical and skeletal anomalies in Fgf-23-null animals. Am J Pathol. 2006;169:2161–70. doi: 10.2353/ajpath.2006.060329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hermans MM, Brandenburg V, Ketteler M, Kooman JP, van der Sande FM, Boeschoten EW, et al. Netherlands cooperative study on the adequacy of Dialysis (NECOSAD) Association of serum fetuin-A levels with mortality in dialysis patients. Kidney Int. 2007;72:202–7. doi: 10.1038/sj.ki.5002178. [DOI] [PubMed] [Google Scholar]
- 28.Ketteler M, Bongartz P, Westenfeld R, Wildberger JE, Mahnken AH, Böhm R, et al. Association of low fetuin-A (AHSG) concentrations in serum with cardiovascular mortality in patients on dialysis: a cross-sectional study. Lancet. 2003;361:827–33. doi: 10.1016/S0140-6736(03)12710-9. [DOI] [PubMed] [Google Scholar]
- 29.Wang AY, Woo J, Lam CW, Wang M, Chan IH, Gao P, et al. Associations of serum fetuin-A with malnutrition, inflammation, atherosclerosis and valvular calcification syndrome and outcome in peritoneal dialysis patients. Nephrol Dial Transplant. 2005;20:1676–85. doi: 10.1093/ndt/gfh891. [DOI] [PubMed] [Google Scholar]
- 30.Gutiérrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, Shah A, et al. Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med. 2008;359:584–92. doi: 10.1056/NEJMoa0706130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cranenburg EC, Vermeer C, Koos R, Boumans ML, Hackeng TM, Bouwman FG, et al. The circulating inactive form of matrix Gla Protein (ucMGP) as a biomarker for cardiovascular calcification. J Vasc Res. 2008;45:427–36. doi: 10.1159/000124863. [DOI] [PubMed] [Google Scholar]
- 32.Hermans MM, Vermeer C, Kooman JP, Brandenburg V, Ketteler M, Gladziwa U, et al. Undercarboxylated matrix GLA protein levels are decreased in dialysis patients and related to parameters of calcium-phosphate metabolism and aortic augmentation index. Blood Purif. 2007;25:395–401. doi: 10.1159/000108629. [DOI] [PubMed] [Google Scholar]
- 33.Cranenburg EC, Brandenburg VM, Vermeer C, Stenger M, Mühlenbruch G, Mahnken AH, et al. Uncarboxylated matrix Gla protein (ucMGP) is associated with coronary artery calcification in haemodialysis patients. Thromb Haemost. 2009;101:359–66. [PubMed] [Google Scholar]
- 34.Schurgers LJ, Teunissen KJ, Knapen MH, Geusens P, van der Heijde D, Kwaijtaal M, et al. Characteristics and performance of an immunosorbent assay for human matrix Gla-protein. Clin Chim Acta. 2005;351:131–8. doi: 10.1016/j.cccn.2004.08.003. [DOI] [PubMed] [Google Scholar]
- 35.Whooley MA, de Jonge P, Vittinghoff E, Otte C, Moos R, Carney RM, et al. Depressive symptoms, health behaviors, and risk of cardiovascular events in patients with coronary heart disease. JAMA. 2008;300:2379–88. doi: 10.1001/jama.2008.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Luepker RV, Apple FS, Christenson RH, Crow RS, Fortmann SP, Goff D, et al. AHA Council on Epidemiology and Prevention. Case definitions for acute coronary heart disease in epidemiology and clinical research studies: a statement from the AHA Council on Epidemiology and Prevention; AHA Statistics Committee; World Heart Federation Council on Epidemiology and Prevention; the European Society of Cardiology Working Group on Epidemiology and Prevention; Centers for Disease Control and Prevention; and the National Heart, Lung, and Blood Institute. Circulation. 2003;108:2543–9. doi: 10.1161/01.CIR.0000100560.46946.EA. [DOI] [PubMed] [Google Scholar]
- 37.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130:461–70. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 38.Whooley MA, Caska CM, Hendrickson BE, Rourke MA, Ho J, Ali S. Depression and inflammation in patients with coronary heart disease: findings from the Heart and Soul Study. Biol Psychiatry. 2007;62:314–20. doi: 10.1016/j.biopsych.2006.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kestenbaum B, Sampson JN, Rudser KD, Patterson DJ, Seliger SL, Young B, et al. Serum phosphate levels and mortality risk among people with chronic kidney disease. J Am Soc Nephrol. 2005;16:520–8. doi: 10.1681/ASN.2004070602. [DOI] [PubMed] [Google Scholar]
- 40.Chonchol M, Dale R, Schrier RW, Estacio R. Serum phosphorus and cardiovascular mortality in type 2 diabetes. Am J Med. 2009;122:380–6. doi: 10.1016/j.amjmed.2008.09.039. [DOI] [PubMed] [Google Scholar]
- 41.Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest. 2002;110:229–38. doi: 10.1172/JCI15219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.O’Connell TD, Berry JE, Jarvis AK, Somerman MJ, Simpson RU. 1,25-Dihydroxyvitamin D3 regulation of cardiac myocyte proliferation and hypertrophy. Am J Physiol. 1997;272:H1751–8. doi: 10.1152/ajpheart.1997.272.4.H1751. [DOI] [PubMed] [Google Scholar]
- 43.Liu S, Quarles LD. How fibroblast growth factor 23 works. J Am Soc Nephrol. 2007;18:1637–47. doi: 10.1681/ASN.2007010068. [DOI] [PubMed] [Google Scholar]
- 44.Gutiérrez OM, Januzzi JL, Isakova T, Laliberte K, Smith K, Collerone G, et al. Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation. 2009;119:2545–52. doi: 10.1161/CIRCULATIONAHA.108.844506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mirza MA, Larsson A, Melhus H, Lind L, Larsson TE. Serum intact FGF23 associate with left ventricular mass, hypertrophy and geometry in an elderly population. Atherosclerosis. 2009;207:546–51. doi: 10.1016/j.atherosclerosis.2009.05.013. [DOI] [PubMed] [Google Scholar]
- 46.Detillieux KA, Sheikh F, Kardami E, Cattini PA. Biological activities of fibroblast growth factor-2 in the adult myocardium. Cardiovasc Res. 2003;57:8–19. doi: 10.1016/s0008-6363(02)00708-3. [DOI] [PubMed] [Google Scholar]
- 47.Takeshita K, Fujimori T, Kurotaki Y, Honjo H, Tsujikawa H, Yasui K, et al. Sinoatrial node dysfunction and early unexpected death of mice with a defect of klotho gene expression. Circulation. 2004;109:1776–82. doi: 10.1161/01.CIR.0000124224.48962.32. [DOI] [PubMed] [Google Scholar]
- 48.Yu X, Ibrahimi OA, Goetz R, Zhang F, Davis SI, Garringer HJ, et al. Analysis of the biochemical mechanisms for the endocrine actions of fibroblast growth factor-23. Endocrinology. 2005;146:4647–56. doi: 10.1210/en.2005-0670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Parker BD, Schurgers LJ, Vermeer C, Schiller NB, Whooley MA, Ix JH. The association of uncarboxylated matrix Gla protein with mitral annular calcification differs by diabetes status: the Heart and Soul Study. Atherosclerosis. 2010 doi: 10.1016/j.atherosclerosis.2009.11.023. [Forthcoming] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Price PA, Lim JE. The inhibition of calcium phosphate precipitation by fetuin is accompanied by the formation of a fetuin-mineral complex. J Biol Chem. 2003;278:22144–52. doi: 10.1074/jbc.M300744200. [DOI] [PubMed] [Google Scholar]
- 51.Ix JH, Shlipak MG, Sarnak MJ, Beck GJ, Greene T, Wang X, et al. Fetuin-A is not associated with mortality in chronic kidney disease. Kidney Int. 2007;72:1394–9. doi: 10.1038/sj.ki.5002549. [DOI] [PubMed] [Google Scholar]
- 52.Lim P, Collet JP, Moutereau S, Guigui N, Mitchell-Heggs L, Loric S, et al. Fetuin-A is an independent predictor of death after ST-elevation myocardial infarction. Clin Chem. 2007;53:1835–40. doi: 10.1373/clinchem.2006.084947. [DOI] [PubMed] [Google Scholar]
- 53.Weikert C, Stefan N, Schulze MB, Pischon T, Berger K, Joost HG, et al. Plasma fetuin-a levels and the risk of myocardial infarction and ischemic stroke. Circulation. 2008;118:2555–62. doi: 10.1161/CIRCULATIONAHA.108.814418. [DOI] [PubMed] [Google Scholar]
- 54.Hennige AM, Staiger H, Wicke C, Machicao F, Fritsche A, Häring HU, et al. Fetuin-A induces cytokine expression and suppresses adiponectin production. PLoS One. 2008;3:e1765. doi: 10.1371/journal.pone.0001765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ix JH, Chertow GM, Shlipak MG, Brandenburg VM, Ketteler M, Whooley MA. Association of fetuin-A with mitral annular calcification and aortic stenosis among persons with coronary heart disease: data from the Heart and Soul Study. Circulation. 2007;115:2533–9. doi: 10.1161/CIRCULATIONAHA.106.682450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ix JH, Wassel CL, Kanaya AM, Vittinghoff E, Johnson KC, Koster A, et al. Health ABC Study. Fetuin-A and incident diabetes mellitus in older persons. JAMA. 2008;300:182–8. doi: 10.1001/jama.300.2.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jonsson KB, Zahradnik R, Larsson T, White KE, Sugimoto T, Imanishi Y, et al. Fibroblast growth factor 23 in oncogenic osteomalacia and X-linked hypophosphatemia. N Engl J Med. 2003;348:1656–63. doi: 10.1056/NEJMoa020881. [DOI] [PubMed] [Google Scholar]
- 58.Cranenburg EC, Vermeer C, Koos R, Boumans ML, Hackeng TM, Bouwman FG, et al. The circulating inactive form of matrix Gla Protein (ucMGP) as a biomarker for cardiovascular calcification. J Vasc Res. 2008;45:427–36. doi: 10.1159/000124863. [DOI] [PubMed] [Google Scholar]
- 59.Hermans MM, Brandenburg V, Ketteler M, Kooman JP, van der Sande FM, Gladziwa U, et al. Study on the relationship of serum fetuin-A concentration with aortic stiffness in patients on dialysis. Nephrol Dial Transplant. 2006;21:1293–9. doi: 10.1093/ndt/gfk045. [DOI] [PubMed] [Google Scholar]
- 60.Hermans MM, Brandenburg V, Ketteler M, Kooman JP, van der Sande FM, Boeschoten EW, et al. Netherlands cooperative study on the adequacy of Dialysis (NECOSAD) Association of serum fetuin-A levels with mortality in dialysis patients. Kidney Int. 2007;72:202–7. doi: 10.1038/sj.ki.5002178. [DOI] [PubMed] [Google Scholar]