Abstract
Breast Tumor Kinase (Brk/PTK6) has a relatively limited expression profile in normal tissue. Its expression is restricted to epithelial cells that are differentiating such as those in the epidermis, and Brk expression appears to be absent from proliferating cells in normal tissue. Also, there is now some evidence to suggest that Brk plays a functional role in the differentiation of the keratinocytes in the epidermis. We have, therefore, investigated the role that Brk/PTK6 plays in normal human primary keratinocytes by suppressing protein levels using RNA interference. We show that as primary human keratinocytes are induced to differentiate in vitro, Brk levels decrease. Decreasing Brk protein levels lead to an increase in the number of cells with a permeable plasma membrane, a decrease in epidermal growth factor receptor (EGFR) and a parallel increase in keratin 10 levels, but classical markers of apoptosis or terminal differentiation are not affected. We propose Brk, Keratin 10 and EGFR are co-regulated during differentiation and that manipulating Brk expression can influence the differentiation of normal primary human keratinocytes.
Keywords: Keratinocyte, Cell differentiation, Breast tumor kinase (Brk/PTK6)
Introduction
Keratinocyte differentiation is tightly controlled and involves a range of signals and events, including tyrosine kinase activation, that are important in regulation. Signaling downstream of the epidermal growth factor receptor (EGFR) in the presence of constitutively active AKT initiates the growth arrest and differentiation process in response to elevated calcium [1], while decreased EGFR levels and signaling are associated with terminal differentiation [14].
The breast tumor kinase (Brk/PTK6) is a known potentiator of EGF signaling [8, 9] and an inhibitor of Akt [5, 25] and, its suppression decreases proliferation of breast cancer cells [6]. Although originally identified in breast carcinomas [13], Brk’s expression profile in normal tissue is relatively restricted. Brk and its mouse orthologue, Sik, are found in differentiating epithelia. Brk expression has been identified in epithelial cells of the GI tract [12], epidermis [19, 21], prostate [3], and oral mucosa [15].
In addition to the expression profile in differentiating epithelia, there is an evidence to suggest that Brk plays a functional role in keratinocyte differentiation. Brk over-expression in the keratinocyte cell line HaCaT results in moderately elevated expression of a known keratinocyte differentiation marker, keratin 10 [21]. Increased Brk-associated kinase activity was observed in HaCaT cells, induced to differentiate with calcium [21]. Similar results were seen in primary murine keratinocytes where activity of Sik increased rapidly following the calcium treatment [20].
We investigated the role that Brk plays in normal human primary keratinocytes by suppressing protein levels using RNA interference. We hypothesized that since differentiating agents can increase Brk activity, the suppression of Brk expression would alter primary keratinocyte differentiation in vitro.
Materials and methods
Cell culture
Normal human adult keratinocytes were purchased from TCS Cell Works, maintained in Epilife media supplemented with full supplement and antibiotics (Cascade Biologics Inc., USA) and used below passage ten.
For calcium differentiation studies, cells were plated at 37,500 cm−2. 24 h later medium was changed for medium plus 1.2 mM Ca2+ for 5 days, at which point cells displayed an altered morphology and decreased growth compared with controls.
Methylcellulose (MC) studies were performed according to Watt [23]. Briefly, 2 × 105 cells were suspended for 48 h in 2 ml methylcellulose per well of a polyHEMA-coated 6-well plate (Sigma).
RNA interference
Cells were transfected with Brk-targeting (5′-GGUGAUUUCUCGAGACAACTT-3′) or Control siRNA (5′-GGACACCAUCAAGUGUUCGTT-3′) (Dharmacon). Two transfections were performed 24 h apart on 105 cells with the siRNA duplex (120 nM) and Oligofectamine (Invitrogen) in OptiMEM medium using the manufacturer’s guidelines.
Trypan blue staining
Cell viability was determined using trypan blue exclusion. Cells were mixed with the stain to a final trypan blue concentration of 0.1% and counted using an improved neubauer hemocytometer. Cells that appeared blue when visualized by light microscopy were classed as nonviable.
Cell death ELISA
Cell death levels were measured using a cell death detection ELISA (Roche).
Western blotting
Cells were lysed in 2× SDS-PAGE loading buffer and separated by SDS-PAGE. Equal loading was determined by coomassie-blue staining and the presence of housekeeping proteins. Separated proteins were transferred to nitrocellulose membranes by electro-blotting in Towbin buffer. Membranes were incubated with antibodies to GAPDH, β-actin (Abcam), EGFR, K10, involucrin (Sigma) and Brk (ICR-100) [9] and proteins visualized with an appropriate HRP-conjugated secondary antibody and chemiluminescence.
Results and discussion
Keratinocytes are one of the few sites of physiological Brk expression [20, 21], however, Brk’s role in these cells has yet to be fully characterized. In this study normal human keratinocytes were used as model for studying the role of Brk in human primary cells.
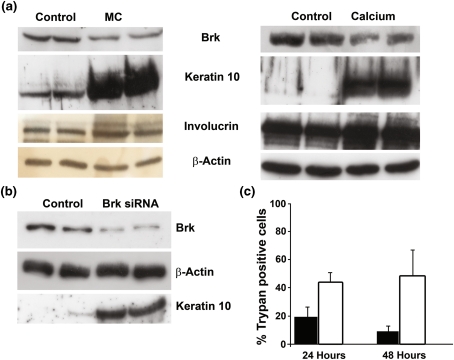
Primary non-immortalized keratinocytes were induced to differentiate by either increasing the calcium concentration of the medium or by resuspending the cells in methylcellulose. When cells were suspended in methylcellulose for 48 h or cultured in calcium for up to 5 days the levels of both Keratin 10 and involucrin increased. Brk protein levels decreased under these conditions suggesting that, as cells become more terminally differentiated Brk protein decreases (Fig. 1a).
Fig. 1.
Brk levels are reduced in differentiation. a Normal human primary keratinocytes were induced to differentiate by either culturing in methylcellulose for 48 h (MC) (left hand panel) or in medium containing 1.2 mM Calcium ions for 5 days (Calcium) (right hand panel). Control cells were cultured under normal adherent culture conditions (Control). Cells were lysed in hot SDS-PAGE lysis buffer and relative protein levels determined by western blotting. Brk levels were then suppressed in normal human primary keratinocytes by RNAi. b 48 h following the second transfection with control (Control) or Brk-targeting siRNA (Brk siRNA), cells were lysed in hot SDS-PAGE lysis buffer and relative protein levels determined by western blotting. c 24 and 48 h following the second transfection with control (filled bars) or Brk-targeting siRNA (open bars) cells were harvested, resuspended in 0.1% trypan blue and the percentage of nonviable cells determined
To determine whether experimentally decreasing Brk expression resulted in differentiation, RNA interference was used to decrease Brk protein expression. Brk suppression (Fig. 1b) resulted in an increase in the percentage of cells incorporating trypan blue both 24 and 48 h following the second transfection compared with control transfectants (Fig. 1c). This increase was seen with two different Brk-targeting siRNA sequences (data not shown).
Membrane permeabilization to trypan blue could be regarded as a sign of cell death, however, it is also an early sign of keratinocyte differentiation. Western blot analysis revealed that on Brk suppression, Keratin 10 levels increased (Fig. 1b), although there were no observable changes in levels of the differentiation markers Keratin 1 and involucrin, or activation markers Keratin 6 and 16 (data not shown).
The effects of Brk-suppression on cell membrane integrity coupled with an increase in Keratin 10 suggest that Brk suppression may contribute to primary keratinocyte differentiation. However, as levels of other differentiation markers were unaffected, it is likely that downregulation of Brk may be an early/intermediate event in differentiation and is not a sufficient signal alone to fully mediate the differentiation process. Activation status could be important, although Brk expression is differential in epidermal layers [20, 21].
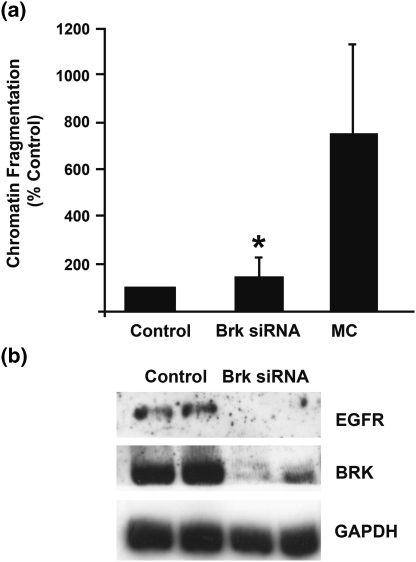
The level of cytosolic, histone-bound DNA was determined by DNA ELISA as a measure of chromatin fragmentation, and a significant increase in DNA fragmentation in cells with suppressed Brk, compared with controls, was observed (Fig. 2a).
Fig. 2.
Brk suppression induces chromatin fragmentation and suppression of EGFR. Brk levels were suppressed in normal human primary keratinocytes by RNAi. a 48 h following the second transfection with control (Control) or Brk-targeting siRNA (Brk siRNA), cells were harvested and assayed for chromatin fragmentation. As a positive control, primary keratinocytes were also cultured in methyl cellulose (MC) for 48 h prior to harvesting and assaying with the cell death ELISA. *P = 0.036. b Parallel wells were also lysed in hot SDS-PAGE lysis buffer and relative protein levels determined by western blotting
Keratinocyte terminal differentiation is associated with some cell death features such as DNA fragmentation although these findings are inconsistent [17, 24].
Therefore to determine whether the observed increase in cell permeability and chromatin fragmentation, in Brk-suppressed cells, might be a result of differentiation, keratinocytes were induced to differentiate by increasing culture calcium concentrations or by suspension culture in methylcellulose. The well-documented method of differentiating keratinocytes in methylcellulose [18] resulted in DNA fragmentation (Fig. 2a). However, differentiation with increased calcium did not result in any observable increase in chromatin fragmentation using the cell death ELISA (unpublished observation) suggesting that DNA fragmentation can accompany keratinocyte differentiation, but this phenomenon is observed only in certain types of differentiation which may account for previously inconsistent findings. It is also striking that the level of chromatin fragmentation observed when cells were cultured on methylcellulose is much greater than that observed on Brk suppression. It is probable that methylcellulose culture affects a number of cellular pathways and processes involved in differentiation and that Brk contributes to this. However, it is unlikely that Brk is the only contributor to differentiation, which would account for the lesser effects in Brk-suppressed cells.
We have previously reported that Brk expression protected breast cancer cells from suspension-induced cell death [7]. Given that in vivo, Brk expression is increased in differentiating cells that are in transit away from the epidermal basement membrane, and that untransformed cells are dependent on contact with basement membrane/substratum for survival [4], we hypothesize that a function of Brk in early differentiating cells may be to promote their survival. The subsequent decrease in Brk levels that occurred when keratinocytes continued to differentiate might occur as a requirement of the differentiation process in primary cells. The expression of cell surface receptors associated with Brk signaling, such as EGFR, has been shown to decrease in vivo as cells commit to terminal differentiation [11, 14]. Inhibition of EGFR signaling induces growth arrest and keratinocyte differentiation [16]. As Brk enhances the effects of signaling via Erb receptors [8, 9] suppression of Brk would be predicted to reduce the effects of Erb signaling, in which this cellular context results in a more differentiated phenotype. Conversely EGF treatment of a serum-free epidermal culture model resulted in decreased differentiation [2] and an elevated expression of EGFR in differentiating cells, and presumably therefore activation of EGFR signaling, resulted in hyperproliferation [22]. Downregulation of EGFR signaling appears to be a requirement of keratinocyte differentiation and the reduction in signaling could be enhanced by reduced Brk levels. We propose that this may be a supplementary mechanism that contributes to the subsequent differentiation. In addition, we found that EGFR protein levels were decreased on Brk suppression in primary keratinocytes (Fig. 2b), which supports recent findings that Brk inhibits EGFR downregulation [10]. Brk suppression in keratinocytes may, therefore, be a mechanistic prerequisite for reduced EGFR levels in differentiation.
Conclusions
This is the first study in normal primary human cells demonstrating co-regulation of Brk and keratin 10 in differentiation. The data presented link Brk mechanistically to keratinocyte differentiation (via Keratin 10 and EGFR expression) and demonstrate that manipulating Brk affected phenotypic outcome. The results of this and other studies [20, 21] suggest that Brk plays an important, complex role in normal human keratinocyte differentiation and that this role could be keratinocyte-specific.
Acknowledgments
This work was supported by a project grant from the Biotechnology and Biological Sciences Research Council, UK. The funder has played no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Calautti E, Li J, Saoncella S, Brissette JL, Goetinck PF. Phosphoinositide 3-kinase signaling to Akt promotes keratinocyte differentiation versus death. J Biol Chem. 2000;280(38):32856–32865. doi: 10.1074/jbc.M506119200. [DOI] [PubMed] [Google Scholar]
- 2.Chen CS, Lavker RM, Rodeck U, Risse B, Jensen PJ. Use of a serum-free epidermal culture model to show deleterious effects of epidermal growth factor on morphogenesis and differentiation. J Invest Dermatol. 1995;104(1):107–112. doi: 10.1111/1523-1747.ep12613595. [DOI] [PubMed] [Google Scholar]
- 3.Derry JJ, Richard S, Valderrama Carvajal H, Ye X, Vasioukhin V, Cochrane AW, Chen T, Tyner AL. Sik (BRK) phosphorylates Sam68 in the nucleus and negatively regulates its RNA binding ability. Mol Cell Biol. 2000;20(16):6114–6126. doi: 10.1128/MCB.20.16.6114-6126.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Frisch SM, Francis H. Disruption of epithelial cell-matrix interactions induces apoptosis. Cell Biol. 1994;24(4):619–626. doi: 10.1083/jcb.124.4.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haegebarth A, Perekatt AO, Bie W, Gierut JJ, Tyner AL. Induction of protein tyrosine kinase 6 in mouse intestinal crypt epithelial cells promotes DNA damage-induced apoptosis. Gastroenterology. 2009;137(3):945–954. doi: 10.1053/j.gastro.2009.05.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harvey AJ, Crompton MR. Use of RNA interference to validate Brk as a novel therapeutic target in breast cancer: Brk promotes breast carcinoma cell proliferation. Oncogene. 2003;22:5006–5010. doi: 10.1038/sj.onc.1206577. [DOI] [PubMed] [Google Scholar]
- 7.Harvey AJ, Pennington C, Porter S, et al. Brk protects breast cancer cells from autophagic cell death induced by loss of anchorage. Am J Pathol. 2009;175(3):1226–1234. doi: 10.2353/ajpath.2009.080811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kamalati T, Jolin HE, Fry MJ, Crompton MR. Expression of the Brk tyrosine kinase in mammary epithelial cells enhances the coupling of EGF signaling to PI 3-kinase and Akt, via erbB3 phosphorylation. Oncogene. 2000;19(48):5471–5476. doi: 10.1038/sj.onc.1203931. [DOI] [PubMed] [Google Scholar]
- 9.Kamalati T, Jolin HE, Mitchell PJ, et al. Brk, a breast tumor-derived non-receptor protein-tyrosine kinase, sensitizes mammary epithelial cells to epidermal growth factor. J Biol Chem. 1996;271(48):30956–30963. doi: 10.1074/jbc.271.48.30956. [DOI] [PubMed] [Google Scholar]
- 10.Kang SA, Lee ES, Yoon HY, et al. PTK6 inhibits downregulation of EGF receptor through phosphorylation of ARAP1. J Biol Chem. 2010;285(34):26013–26021. doi: 10.1074/jbc.M109.088971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.King LE, Jr, Gates RE, Stoscheck CM, Nanney LB. The EGF/TGF alpha receptor in skin. J Invest Dermatol. 1990;94:164S–170S. doi: 10.1111/1523-1747.ep12876141. [DOI] [PubMed] [Google Scholar]
- 12.Llor X, Serfas MS, Bie W, Vasioukhin Y, Polonskaia M, Derry J, Abbott CM, Tyner AL. BRK/Sik expression in the gastrointestinal tract and in colon tumors. Clin Cancer Res. 1999;5(7):1767–1777. [PubMed] [Google Scholar]
- 13.Mitchell PJ, Barker KT, Martindale JE, Kamalati T, Lowe PN, Page MJ, Gusterson BA, Crompton MR. Cloning and characterisation of cDNAs encoding a novel non-receptor tyrosine kinase, brk, expressed in human breast tumours. Oncogene. 1994;9(8):2383–2390. [PubMed] [Google Scholar]
- 14.Nanney LB, Stoscheck CM, King LE, Jr, et al. Immunolocalization of epidermal growth factor receptors in normal developing human skin. J Invest Dermatol. 1990;94(6):742–748. doi: 10.1111/1523-1747.ep12874601. [DOI] [PubMed] [Google Scholar]
- 15.Petro BJ, Tan RC, Tyner AL, Lingen MW, Watanbabe K. Differntial expression of the non-receptor tyrosine kinase BRK in oral squamous cell carcinoma and normal oral epithelium. Oral Oncol. 2004;40(10):1040–1047. doi: 10.1016/j.oraloncology.2004.05.010. [DOI] [PubMed] [Google Scholar]
- 16.Peus D, Hamacher L, Pittelkow MR. EGF-receptor tyrosine kinase inhibition induces keratinocyte growth arrest and terminal differentiation. J Invest Dermatol. 1997;109:751–756. doi: 10.1111/1523-1747.ep12340759. [DOI] [PubMed] [Google Scholar]
- 17.Polakowska RR, Piacentini M, Bartlett R, et al. Apoptosis in human skin development: morphogenesis, periderm, and stem cells. Dev Dyn. 1994;199:176–188. doi: 10.1002/aja.1001990303. [DOI] [PubMed] [Google Scholar]
- 18.Sun TT, Green H. Differentiation of the epidermal keratinocyte in cell culture: formation of the cornified envelope. Cell. 1976;9:511–521. doi: 10.1016/0092-8674(76)90033-7. [DOI] [PubMed] [Google Scholar]
- 19.Vasioukhin V, Serfas MS, Siyanova EY, et al. A novel intracellular epithelial cell tyrosine kinase is expressed in the skin and gastrointestinal tract. Oncogene. 1995;10(2):349–357. [PubMed] [Google Scholar]
- 20.Vasioukhin V, Tyner AL. A role for the epithelial-cell-specific tyrosine kinase Sik during keratinocyte differentiation. Proc Natl Acad Sci. 1997;94(26):14477–14482. doi: 10.1073/pnas.94.26.14477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang TC, Jee SH, Tsai TF, et al. Role of breast tumor kinase in the in vitro differentiation of HaCaT cell. Br J Dermatol. 2005;153(2):282–289. doi: 10.1111/j.1365-2133.2005.06604.x. [DOI] [PubMed] [Google Scholar]
- 22.Wang X, Bolotin D, Chu DH, Polak L, Williams T, Fuchs E. AP-2α: a regulator of EGF receptor signalling and proliferation in skin epidermis. J Cell Biol. 2006;172(3):409–421. doi: 10.1083/jcb.200510002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Watt FM (1994) In: Leigh, Watt (eds) Keratinocyte Methods, Cambridge University Press
- 24.Weil M, Raff MC, Braga VM. Caspase activation in the terminal differentiation of human epidermal keratinocytes. Curr Biol. 1999;9(7):361–364. doi: 10.1016/S0960-9822(99)80162-6. [DOI] [PubMed] [Google Scholar]
- 25.Zhang P, Ostrander JH, Faivre EJ, et al. Regulated association of protein kinase B/Akt with breast tumour kinase. J Biol Chem. 2005;280(3):1982–1991. doi: 10.1074/jbc.M412038200. [DOI] [PubMed] [Google Scholar]